Abstract
Hepatocellular carcinoma (HCC) is characterized by hypervascularity. Hepatic stellate cells (HSCs) play very important roles in HCC malignant progression, as these cells facilitate HCC tumorigenesis and metastasis. We demonstrated that HSCs induce angiogenesis in HCC by upregulating the expression of Gli-1, which stimulates reactive oxygen species (ROS) production and potentiates increases in HCC cell invasiveness. Resveratrol abolished HSC-induced angiogenesis and suppressed ROS production and IL-6 and CXCR4 receptor expression in HepG2 cells by down-regulating Gli-1 expression. These findings indicate that Gli-1 may be a target for the prevention of angiogenesis in HCC and that resveratrol may have beneficial effects with respect to preventing HCC progression.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Hepatocellular carcinoma (HCC), one of the most lethal human cancers, is the third-leading cause of cancer-related death worldwide [1]. HCC is a highly vascularized tumor that requires angiogenesis to grow, invade, and metastasize [2]. As a result of advances in technology and clinical practice, the patient survival rates associated with tumor resection have improved within the last three decades. However, approximately 70% of patients with HCC present with tumor recurrence within 5 years after resection, and most of these patients present with recurrence within the first 2 years after resection [3]. Transarterial chemoembolization and sorafenib are systemic chemotherapy options for patients who are not suitable candidates for surgical resection. However, several clinical trials have shown that these therapies have marginal anti-cancer effects and are non-beneficial with respect to improving patient survival [3, 4]. Elucidating the mechanisms underlying HCC progression and identifying new therapeutic targets for the treatment of the disease may contribute to improvements in disease control.
The tumor microenvironment has been shown to play a critical role in liver carcinogenesis [5]. Hepatic stellate cells (HSCs) are important components of the normal liver, and their activation, which subsequently leads to phenotypic changes, is a key event in fibrosis. HSCs can also influence HCC initiation and evolution. Recent studies have shown that HSCs promote tumor cell proliferation and migration by secreting growth factors and cytokines [6]. HSC-mediated extracellular matrix production also modulates tumor growth. In addition, HSCs function in angiogenesis regulation and immune evasion.
Aberrant hedgehog (Hh) pathway activation has also been linked to HCC. Intrinsic Hh signaling in tumor cells is associated with hepatitis B virus X protein-induced carcinogenesis [7], and paracrine Hh signaling involving ligand-secreting tumor cells [8] and Hh-responsive HSCs are also well-characterized phenomena [9–11]. Gli-1 is a key Hh pathway transcription factor [12]. Resveratrol, or 3,4′,5 trihydroxy-trans-stilbene, is a natural polyphenol with multiple properties. Resveratrol is a phytoalexin produced by vines and is widely found in plants (such as grapes, blackberries, blackcurrants, and blueberries) and some traditional Chinese medicines (such as Polygonum cuspidatum) [13, 14]. Resveratrol has been shown to exert anti-cancer effects by sensitizing cancerous tumors to chemotherapy [15]. For example, in chronic myeloid leukemia (CML), resveratrol inhibited Hh signaling in both imatinib mesylate-sensitive cells and imatinib mesylate-resistant cells [16].
Here, we evaluated whether Gli-1 plays a vital role in HCC progression and investigated the potential protective effects of resveratrol on HSC-induced HCC angiogenesis and invasiveness. We showed that resveratrol suppressed HSC-induced HCC angiogenesis and invasion by inhibiting Gli-1 expression.
Materials and methods
Cell lines and cell culture
The indicated HCC cell lines (HepG2) were obtained from American Type Culture Collection (USA), and human umbilical vein endothelial cells (HUVECs) were purchased from the Shanghai Institution for Biological Science. Human HSC lines were obtained from ScienCell Research Laboratory (Carlsbad, CA, USA). All cell lines were maintained in Dulbecco’s modified Eagle medium (DMEM, Gibco, Grand Island, NY, USA) containing 10% fetal bovine serum (FBS, Gibco), 100 U/mL penicillin, and 100 μg/mL streptomycin supplemented with 2 mmol/L l-glutamine in a humidified atmosphere at 37 °C with 5% CO2.
Reagents
Anti-CXCR4 was purchased from Bioworld, St. Louis Park, MN, USA, and the other antibodies, namely, anti-Gli-1, anti-MMP-9, anti-uPA, and anti-β-actin, were obtained from Santa Cruz Biotechnology, Santa Cruz, CA, USA. DCF-DA was obtained from Molecular Probes (Eugene, OR, USA), and resveratrol was obtained from Sigma-Aldrich (St. Louis, MO, USA).
Real-time quantitative PCR (RT-qPCR)
Total RNA was isolated using TRIzol reagent, and first-strand cDNA was synthetized using a PrimeScript™ Synthesis Kit (Takara Bio, Inc., Dalian, China). The sequences for the primers used in the experiments are as follows. RT-qPCR was performed on an Applied Biosystems 7500 Real-Time PCR system (Applied Biosystems, White Plains, NY, USA). Fold changes in gene expression were determined using the △△CT method, and gene expression levels were normalized to those of GAPDH. The following PCR primers were used in the study: MMP-9, forward: 5′-GAACCAATCTCACCGACAGG-3′ and reverse: 5′-GCCACCCGAGTGTAACCATA-3′; uPA, forward: 5′-GGAATGGTCACTTTTACCGAG-3′ and reverse: 5′-GGGCATGGTAGTTTGCTG-3′; VEGF, forward: 5′-TGCAGATTATGCGGATCAAACC-3′ and reverse: 5′-TGCATTCACATTT GTTGTGCTGTAG-3′; Gli-1, forward: 5′-GGGATGATCCCACATCCTCAGTC-3′ and reverse: 5′-CTGGAGCAGCCCCCCCAGT-3′; IL-6, forward: 5′-AGTTCCTGCAGTCCAGCCTGAG-3′ and reverse: 5′-TCAAACTGCATAGCCACTTTC C-3′; and GAPDH, forward: 5′-ACCACAGTCCATGCCATCAC-3′ and reverse: 5′-TCCACCACCCTGTTGCTGAT-3′.
Western blot analysis
Total cellular protein was isolated and heated for 5 min at 100 °C. The same amount of protein (20 μg per well) was subjected to 10% SDS-PAGE before being transferred to a PVDF membrane, which was blotted with the following primary antibodies: anti-CXCR4, anti-Gli-1, anti-MMP-9, and anti-uPA. The membrane was subsequently stripped and re-probed with anti-β-actin, which served as a loading control.
Cell invasion assay
Eight micrometer pore Millicell (Millipore, Billerica, MA, USA) inserts were coated with 25 mL of Matrigel. HepG2 cells (5 × 104) from the indicated groups were added to the top chamber and incubated in serum-free media for 20 h, after which the non-invading cells were removed from the top of the Matrigel, and the invading cells on the bottom surface of the filter were fixed with methanol and stained with crystal violet. We subsequently evaluated cell invasion ability by counting the number of migrated cells in ten randomly selected fields under a light microscope at × 100 magnification.
HUVEC tubule formation assay
Each well of a 24-well plate was coated with 200 μL of Matrigel. HUVECs (2 × 104) from each group were resuspended in 200 μL of conditioned media (CM) in each well and cultured in an atmosphere containing 5% CO2 for 24 h at 37 °C. Images were captured under a light microscope at a magnification of 100×, and we counted the number of capillary tubes that had formed by measuring the total length of each image. Briefly, three different fields per well were randomly chosen and photographed using a light microscope. Images were captured using Adobe Photoshop, and the data were imported as a TIFF file into ImageJ software. After calibration with a stage micrometer, the total length of all tubing with each field was measured, and the data were analyzed using Prism 5 software (GraphPad Software, San Diego, CA, USA). Experiments were performed a minimum of three times.
Assay of intracellular ROS
Previous publications have described the assay in which intracellular H2O2 production is assessed [17]. Briefly, the cells from the indicated groups were incubated in 5 μg/mL DCF-DA for 5 min and then washed with PBS before being lysed with 1 mL of RIPA buffer. H2O2 concentrations were subsequently detected by fluorimetric analysis at 510 nm. Total protein content was used to normalize the data.
Enzyme-linked immunosorbent assay (ELISA)
HCC cells from the indicated groups were cultured with serum-free medium for 72 h. We detected the concentrations of IL-6 and VEGF in the CM using an Enzyme-Linked Immunosorbent Assay (ELISA) Kit (R&D), according to the manufacturer’s instructions.
RNA interference
ShRNAs against Gli-1 (sc-37911-SH) and a negative control shRNA (sc-108060) were purchased from Santa Cruz Biotechnology and were used to transfect the HCC cells. Lipofectamine (Invitrogen) was used during RNA interference, according to the manufacturer’s instructions. After transfection, the silenced cells were selected using puromycin. Then, to avoid clonal selection, we selected the stably transfected cells for further use.
Statistical analysis
The data are presented as the mean ± standard deviation. Statistical comparisons between two samples were performed using the Student’s t test. Statistical comparisons of more than two groups were performed using one-way analysis of variance with Bonferroni’s post hoc test. Significance was defined as p < 0.05. Each experiment was performed at least three times.
Results
Resveratrol inhibits HCC angiogenesis induced by HSCs by targeting Gli-1
To examine the role of resveratrol in HSC-induced angiogenesis, we used HUVECs to perform a tube formation assay. HepG2 + HSC cell-derived CM significantly enhanced tube formation compared with HepG2 cell-derived CM. However, resveratrol significantly abrogated HSC-mediated enhancements of angiogenesis (p < 0.05; Fig. 1). To evaluate whether Gli-1 plays a key role in HCC angiogenesis, we silenced Gli-1 in HepG2 cells using shRNA. We found that Gli-1 interference significantly inhibited tube formation (Fig. 1). There was no significant difference in the number of tubes formed among the three groups (sh-Gli-1-HepG2, sh-Gli-1-HepG2 + HSC, and sh-Gli-1-HepG2 + HSC + Res). Taken together, these findings indicate that HSC enhanced HCC angiogenesis through Gli-1 and that resveratrol suppresses HSC-induced angiogenesis in HCC by targeting Gli-1.
Resveratrol inhibited HSC-induced angiogenesis mediated by Gli-1 upregulation. HUVECs were incubated with CM from the HepG2, HepG2 + HSC, HepG2 + HSC + Res, sh-Gli-1-HepG2, sh-Gli-1-HepG2 + HSC, and sh-Gli-1-HepG2 + HSC + Res groups. Res stands for Resveratrol. 80 µM resveratrol were added into the medium for 24 h in HepG2 + HSC + Res group or sh-Gli-1-HepG2 + HSC + Res group. a Angiogenesis was evaluated based on tube formation (indicated by arrows). b Tube numbers were counted. *p < 0.05 versus HepG2 group (n = 6), # p < 0.05 versus HepG2 + HSC (n = 6). c Gli-1 in HepG2 cells was silenced by shRNA. Gli-1 and β-actin expression levels were determined by immunoblotting. *p < 0.05, sh-control-HepG2 versus sh-Gli-1-HepG2, n = 3. d HepG2 cells were treated as in C, and Gli-1 and β-actin expression levels were determined by qRT-PCR. *p < 0.05, sh-control-HepG2 versus sh-Gli-1-HepG2, n = 3. All data are representative of at least three independent experiments
Resveratrol abolishes VEGF and IL-6 expression and ROS production in HCC by targeting Gli-1
Oxidative stress and inflammation have been shown to be related to tumor progression and enhanced malignancy [18–20]. VEGF and IL-6 expression levels in HepG2 cells were significantly increased after the cells had been cultured in HSC-derived CM (Fig. 2a–d). However, shRNA-mediated silencing of Gli-1 in HepG2 cells, as well as co-culturing HepG2 cells with resveratrol, could abrogate the increases in VEGF and IL-6 expression induced by HSC-derived CM (Fig. 2a–d), indicating that Gli-1 and resveratrol have significant effects on the inflammatory and angiogenic responses of HepG2 cells exposed to HSC-derived CM. Moreover, we found that the reactive oxygen species (ROS) production in HepG2 cells was increased after these cells had been exposed to HSCs (Fig. 2e) and that resveratrol and Gli-1 knockdown abolished HSC-induced ROS production in HepG2 cells.
Resveratrol suppressed HSC-induced IL-6 and VEGF expression and ROS production by targeting Gli-1. ELISA was assayed to assess IL-6 (a) and VEGF (b) expression in the CM of the indicated groups. IL-6 (c) and VEGF (d) mRNA expression was detected by qRT-PCR, as described in “Materials and methods” section. e Hydrogen peroxide production was determined using DCF-DA, and total protein content was used to normalize the data. *p < 0.05 versus HepG2 group (n = 6), # p < 0.05 versus HepG2 + HSC (n = 6). All data are representative of at least three independent experiments
However, resveratrol could not suppress VEGF and IL-6 expression or ROS production when Gli-1 was silenced by shRNA in HepG2 cells, indicating that the effects of resveratrol are facilitated by Gli-1 downregulation (Fig. 2e).
Resveratrol suppresses HCC invasion induced by HSCs by targeting Gli-1
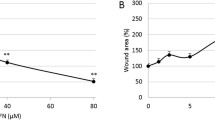
The tumor microenvironment has been shown to play a critical role in liver carcinogenesis [1]. Here, we investigated whether resveratrol could inhibit HSC-induced HCC invasion. We treated HepG2 cells with CM from HSCs in the presence of resveratrol and evaluated the expression of the indicated invasion-related proteins (e.g., CXCR4, MMP-9, and uPA) in HepG2 cells. Additionally, we assessed the invasive ability of the cells using a chamber invasion assay. We observed that resveratrol could abolish the increases in CXCR4, MMP-9, and uPA expression induced by HSC-derived CM in HepG2 cells (Fig. 3). However, we also observed that resveratrol could not suppress the upregulations in CXCR4, MMP-9, and uPA expression driven by HSC-derived CM in HepG2 cells when Gli-1 was silenced by shRNA in those cells (Fig. 3). We observed similar results in our studies of HepG2 cell invasion ability. The effects of HSC-derived CM on HepG2 cell invasiveness could be abrogated by resveratrol (Fig. 4). However, when Gli-1 was silenced by shRNA in HepG2 cells, HepG2 cell invasiveness was not suppressed by resveratrol (Fig. 4). These findings suggest that resveratrol suppresses HSC-induced HCC invasion by targeting Gli-1.
Resveratrol abolished HSC-induced increases in Gli-1, CXCR4, MMP-9, and uPA expression in HepG2 cells by targeting Gli-1. a Gli-1, CXCR4, MMP-9, uPA, and β-actin protein expression levels were evaluated by immunoblotting. *p < 0.05 versus HepG2 group (n = 6), # p < 0.05 versus HepG2 + HSC (n = 6). (B&C) Gli-1, CXCR4, MMP-9, uPA, and β-actin mRNA expression levels were determined by qRT-PCR *p < 0.05 versus HepG2 group (n = 3), # p < 0.05 versus HepG2 + HSC (n = 3). All data are representative of at least three independent experiments
Resveratrol abrogated HSC-induced invasion in PC3 cells by targeting Gli-1. The cells were placed in a Matrigel-coated invasion chamber for 20 h. a, b We evaluated invasion ability by counting the numbers of migrated cells in ten randomly selected fields under a light microscope at ×100 magnification. *p < 0.05 versus HepG2 group (n = 6), # p < 0.05 versus HepG2 + HSC (n = 6). All data are representative of at least three independent experiments
Discussion
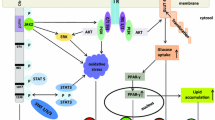
The fifth-most common cancer, HCC is also the third-most common cause of cancer-related mortality worldwide and is a particularly important cause of cancer-related mortality in China [11]. A main characteristic of HCC is hypervascularity, which is predictive of a poor prognosis [11]. However, the molecular mechanisms underlying angiogenesis remain poorly understood [21]. Tumor angiogenesis results from an imbalance between proangiogenic factors, including vascular endothelial growth factor (VEGF) family members, and anti-angiogenesis agents, such as endostatin, angiostatin, and other related molecules [22]. Sorafenib, an angiogenesis inhibitor, has been demonstrated to improve survival in HCC patients [23, 24]. Although the outcomes of studies investigating the efficacy of sorafenib treatment were not satisfactory, the results of those studies implied that angiogenesis was an important target for the treatment of HCC. In this study, we found that HSCs increased tube formation and VEGF expression through Gli-1 upregulation, indicating that Gli-1 is a promising target for HCC therapy. Moreover, our data showed that resveratrol suppressed tube formation and VEGF expression and that Gli-1 interference abolished these effects, indicating that resveratrol exerts significant therapeutic effects on HCC by targeting Gli-1.
HCC develops mostly in the setting of chronic liver disease and fibrosis. HSC activation, which is facilitated by the transition of these cells into myofibroblast-like cells, is a key event in liver fibrosis. Moreover, activated HSCs have been linked to HCC progression through various biological processes. Here, our data showed that HSCs enhanced HCC invasion through Gli-1 upregulation and that resveratrol can abolish the effects of HSCs on HCC by inhibiting Gli-1 expression. Several tumors are surrounded by many stromal fibroblasts, which can differentiate into activated fibroblasts and influence tumor progression [25–28]. Tumor cells can in turn secrete several cytokines, which are able to convert fibroblasts into so-called activated CAFs [29]. For example, prostate carcinoma cells secrete IL-6 to stimulate CAF activation [30]. It has previously been shown that CXCR4, which is associated with chemotaxis in cancer cells and endotheliocytes, and IL-6, which is associated with the inflammatory response, are downstream effectors of Gli-1 [31]. As shown in a study on pancreatic cancer, activated pancreatic cancer stellate cells can secrete stromal cell-derived factor 1 (SDF-1) and IL-6 to drive EMT in pancreatic cancer [31]. Our study showed that exposing HepG2 cells to HSCs boosts IL-6 and CXCR4 expression in HepG2 cells, indicating that HCC cells surrounded by HSCs may more easily metastasize to other sites than other cells. Therefore, the HCC stroma may exert crucial effects to accelerate the metastasis of HCC cells to other organs and trigger the satellite metastasis of HCC cells.
ROS play an important role in tumor aggression through several pathways. They can modulate the activity of transcription factors by causing DNA damage and genome instability and can also induce changes in gene expression. In prostate cancer, MAOA-induced upregulation of mitochondrial ROS production has been shown to be associated with EMT [32]. We illustrated that silencing Gli-1 reverses the increases in ROS production induced by exposing HepG2 cells to HSC-derived CM. Our findings suggest that Gli-1 plays an essential role in the regulation of HSC-induced ROS production.
Resveratrol, a natural polyphenol, exerts many beneficial effects. Resveratrol exerts its anti-inflammatory effects by suppressing the expression of pro-inflammatory factors (such as prostaglandins and interleukins) and by inhibiting immune cell chemo-attraction and the accumulation of immune cells at inflammation sites [33–36]. Moreover, resveratrol increases the accumulation of ROS and antioxidant enzymes, such as superoxide, catalase, heme oxygenase-1 (HO-1), and dismutase-1 (SOD-1) [37, 38]. Our data showed that resveratrol inhibits IL-6 and CXCR4 expression and ROS production and suppresses HCC invasion. Moreover, previous studies have shown that resveratrol can decrease VEGF expression and suppress vascular endothelial cell migration and proliferation [39, 40]. Our results indicate that resveratrol suppresses VEGF expression to attenuate HCC angiogenesis. Intriguingly, resveratrol could not inhibit VEGF or IL-6 expression or ROS production when Gli-1 was silenced by shRNA in HepG2 cells, which indicated that Gli-1 is a key factor in resveratrol-mediated suppression of HCC progression.
References
Forner A (2015) Hepatocellular carcinoma surveillance with miRNAs. Lancet Oncol 16(7):743–745
Abou-Alfa GK, Venook AP (2013) The antiangiogenic ceiling in hepatocellular carcinoma: does it exist and has it been reached. Lancet Oncol 14(7):e283–288
Forner A, Llovet JM, Bruix J (2012) Hepatocellular carcinoma. Lancet 379(9822):1245–1255
Jue C, Lin C, Zhisheng Z, Yayun Q, Feng J, Min Z, Haibo W, Youyang S, Hisamitsu T, Shintaro I, Shiyu G, Yanqing L (2016) Notch1 promotes vasculogenic mimicry in hepatocellular carcinoma by inducing EMT signaling. Oncotarget. doi:10.18632/oncotarget.12388
Han KQ, He XQ, Ma MY, Guo XD, Zhang XM, Chen J, Han H, Zhang WW, Zhu QG, Nian H, Ma LJ (2015) Inflammatory microenvironment and expression of chemokines in hepatocellular carcinoma. World J Gastroenterol 21(16):4864–4874
Li W, Miao S, Miao M, Li R, Cao X, Zhang K, Huang G, Fu B (2016) Hedgehog signaling activation in hepatic stellate cells promotes angiogenesis and vascular mimicry in hepatocellular carcinoma. Cancer Invest 34(9):424–430
Arzumanyan A, Sambandam V, Clayton MM, Choi SS, Xie G, Diehl AM, Yu DY, Feitelson MA (2012) Hedgehog signaling blockade delays hepatocarcinogenesis induced by hepatitis B virus X protein. Cancer Res 72(22):5912–5920
Li X, Wang Z, Ma Q, Xu Q, Liu H, Duan W, Lei J, Ma J, Wang X, Lv S, Han L, Li W, Guo J, Guo K, Zhang D, Wu E, Xie K (2014) Sonic hedgehog paracrine signaling activates stromal cells to promote perineural invasion in pancreatic cancer. Clin Cancer Res 20(16):4326–4338
Omenetti A, Choi S, Michelotti G, Diehl AM (2011) Hedgehog signaling in the liver. J Hepatol 54(2):366–373
Chan IS, Guy CD, Chen Y, Lu J, Swiderska-Syn M, Michelotti GA, Karaca G, Xie G, Krüger L, Syn WK, Anderson BR, Pereira TA, Choi SS, Baldwin AS, Diehl AM (2012) Paracrine hedgehog signaling drives metabolic changes in hepatocellular carcinoma. Cancer Res 72(24):6344–6350
Magistri P, Leonard SY, Tang CM, Chan JC, Lee TE, Sicklick JK (2014) The glypican 3 hepatocellular carcinoma marker regulates human hepatic stellate cells via Hedgehog signaling. J Surg Res 187(2):377–385
Lei J, Fan L, Wei G, Chen X, Duan W, Xu Q, Sheng W, Wang K, Li X (2015) Gli-1 is crucial for hypoxia-induced epithelial-mesenchymal transition and invasion of breast cancer. Tumour Biol 36(4):3119–3126
Lee SK, Zhang W, Sanderson BJ (2008) Selective growth inhibition of human leukemia and human lymphoblastoid cells by resveratrol via cell cycle arrest and apoptosis induction. J Agric Food Chem 56(16):7572–7577
Chen BY, Kuo CH, Liu YC, Ye LY, Chen JH, Shieh CJ (2012) Ultrasonic-assisted extraction of the botanical dietary supplement resveratrol and other constituents of Polygonum cuspidatum. J Nat Prod 75(10):1810–1813
Bonnet D, Dick JE (1997) Human acute myeloid leukemia is organized as a hierarchy that originates from a primitive hematopoietic cell. Nat Med 3(7):730–737
Liao HF, Su YC, Zheng ZY, Jhih CC, Hou MH, Chao KS, Chen YJ (2012) Sonic hedgehog signaling regulates Bcr-Abl expression in human chronic myeloid leukemia cells. Biomed Pharmacother 66(5):378–383
Chiarugi P, Pani G, Giannoni E, Taddei L, Colavitti R, Raugei G, Symons M, Borrello S, Galeotti T, Ramponi G (2003) Reactive oxygen species as essential mediators of cell adhesion: the oxidative inhibition of a FAK tyrosine phosphatase is required for cell adhesion. J Cell Biol 161(5):933–944
Comito G, Giannoni E, Di GP, Segura CP, Gerlini G, Chiarugi P (2012) Stromal fibroblasts synergize with hypoxic oxidative stress to enhance melanoma aggressiveness. Cancer Lett 324(1):31–41
Cat B, Stuhlmann D, Steinbrenner H, Alili L, Holtkötter O, Sies H, Brenneisen P (2006) Enhancement of tumor invasion depends on transdifferentiation of skin fibroblasts mediated by reactive oxygen species. J Cell Sci 119(Pt 13):2727–2738
Cronin PA, Wang JH, Redmond HP (2010) Hypoxia increases the metastatic ability of breast cancer cells via upregulation of CXCR4. BMC Cancer 10:225
Hanahan D, Folkman J (1996) Patterns and emerging mechanisms of the angiogenic switch during tumorigenesis. Cell 86(3):353–364
Zhu AX, Rosmorduc O, Evans TR, Ross PJ, Santoro A, Carrilho FJ, Bruix J, Qin S, Thuluvath PJ, Llovet JM, Leberre MA, Jensen M, Meinhardt G, Kang YK (2015) SEARCH: a phase III, randomized, double-blind, placebo-controlled trial of sorafenib plus erlotinib in patients with advanced hepatocellular carcinoma. J Clin Oncol 33(6):559–566
Llovet JM, Ricci S, Mazzaferro V, Hilgard P, Gane E, Blanc JF, de Oliveira AC, Santoro A, Raoul JL, Forner A, Schwartz M, Porta C, Zeuzem S, Bolondi L, Greten TF, Galle PR, Seitz JF, Borbath I, Häussinger D, Giannaris T, Shan M, Moscovici M, Voliotis D, Bruix J (2008) Sorafenib in advanced hepatocellular carcinoma. N Engl J Med 359(4):378–390
Cheng AL, Kang YK, Chen Z, Tsao CJ, Qin S, Kim JS, Luo R, Feng J, Ye S, Yang TS, Xu J, Sun Y, Liang H, Liu J, Wang J, Tak WY, Pan H, Burock K, Zou J, Voliotis D, Guan Z (2009) Efficacy and safety of sorafenib in patients in the Asia-Pacific region with advanced hepatocellular carcinoma: a phase III randomised, double-blind, placebo-controlled trial. Lancet Oncol 10(1):25–34
Kalluri R (2016) The biology and function of fibroblasts in cancer. Nat Rev Cancer 16(9):582–598
Visvader JE, Lindeman GJ (2008) Cancer stem cells in solid tumours: accumulating evidence and unresolved questions. Nat Rev Cancer 8(10):755–768
Desmoulière A, Guyot C, Gabbiani G (2004) The stroma reaction myofibroblast: a key player in the control of tumor cell behavior. Int J Dev Biol 48(5–6):509–517
Pietras K, Ostman A (2010) Hallmarks of cancer: interactions with the tumor stroma. Exp Cell Res 316(8):1324–1331
Chaiwun B, Sukhamwang N, Trakultivakorn H, Saha B, Young L, Tsao-Wei D, Naritoku WY, Groshen S, Taylor CR, Imam SA (2011) GSTPi-positive tumour microenvironment-associated fibroblasts are significantly associated with GSTPi-negative cancer cells in paired cases of primary invasive breast cancer and axillary lymph node metastases. Br J Cancer 105(8):1224–1229
Giannoni E, Bianchini F, Masieri L, Serni S, Torre E, Calorini L, Chiarugi P (2010) Reciprocal activation of prostate cancer cells and cancer-associated fibroblasts stimulates epithelial-mesenchymal transition and cancer stemness. Cancer Res 70(17):6945–6956
Lei J, Huo X, Duan W, Xu Q, Li R, Ma J, Li X, Han L, Li W, Sun H, Wu E, Ma Q (2014) α-Mangostin inhibits hypoxia-driven ROS-induced PSC activation and pancreatic cancer cell invasion. Cancer Lett 347(1):129–138
Wu JB, Shao C, Li X, Li Q, Hu P, Shi C, Li Y, Chen YT, Yin F, Liao CP, Stiles BL, Zhau HE, Shih JC, Chung LW (2014) Monoamine oxidase A mediates prostate tumorigenesis and cancer metastasis. J Clin Invest 124(7):2891–2908
Tili E, Michaille JJ, Adair B, Alder H, Limagne E, Taccioli C, Ferracin M, Delmas D, Latruffe N, Croce CM (2010) Resveratrol decreases the levels of miR-155 by upregulating miR-663, a microRNA targeting JunB and JunD. Carcinogenesis 31(9):1561–1566
Calamini B, Ratia K, Malkowski MG, Cuendet M, Pezzuto JM, Santarsiero BD, Mesecar AD (2010) Pleiotropic mechanisms facilitated by resveratrol and its metabolites. Biochem J 429(2):273–282
Hwang SH, Wecksler AT, Wagner K, Hammock BD (2013) Rationally designed multitarget agents against inflammation and pain. Curr Med Chem 20(13):1783–1799
Wei G, Chen X, Wang G, Fan L, Wang K, Li X (2016) Effect of resveratrol on the prevention of intra-abdominal adhesion formation in a rat model. Cell Physiol Biochem 39(1):33–46
Zheng Y, Liu Y, Ge J, Wang X, Liu L, Bu Z, Liu P (2010) Resveratrol protects human lens epithelial cells against H2O2-induced oxidative stress by increasing catalase, SOD-1, and HO-1 expression. Mol Vis 16:1467–1474
Luna C, Li G, Liton PB, Qiu J, Epstein DL, Challa P, Gonzalez P (2009) Resveratrol prevents the expression of glaucoma markers induced by chronic oxidative stress in trabecular meshwork cells. Food Chem Toxicol 47(1):198–204
Hua J, Guerin KI, Chen J et al (2011) Resveratrol inhibits pathologic retinal neovascularization in Vldlr(−/−) mice. Invest Ophthalmol Visual Sci 52:2809–2816
Yang SF, Lee WJ, Tan P et al (2015) Upregulation of miR-328 and inhibition of CREB-DNA-binding activity are critical for resveratrol-mediated suppression of matrix metalloproteinase-2 and subsequent metastatic ability in human osteosarcomas. Oncotarget 6:2736–2753
Acknowledgements
This study was funded by the National Natural Science Foundation of China (No. 81572734) and the Scientific and Technological Development Research Project Foundation of Shaanxi Province (2016SF-121).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflicts of interest
The authors declare that they have no competing interests.
Rights and permissions
About this article
Cite this article
Yan, Y., Zhou, C., Li, J. et al. Resveratrol inhibits hepatocellular carcinoma progression driven by hepatic stellate cells by targeting Gli-1. Mol Cell Biochem 434, 17–24 (2017). https://doi.org/10.1007/s11010-017-3031-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-017-3031-z