Abstract
Context
Evaluating connectivity and identifying corridors for protection is a central challenge in applied ecology and conservation. Rigorous validation and comparison of how approaches perform in capturing biological processes is needed to guide research and conservation action.
Objectives
We aim to compare the ability of connectivity surfaces optimised using home range and dispersal data to accurately capture lion movement during dispersal, using cost-distance and circuit theory approaches.
Methods
We delineate periods of dispersal in African lions (Panthera leo) to obtain movement trajectories of dispersing individuals across the Kavango Zambezi Transfrontier Conservation Area, southern Africa. We use these trajectories to assess comparative measures of connectivity values at dispersal points across surfaces and the ability of models to discriminate between observed and randomised paths.
Results
Encouragingly, results show that on average, all connectivity approaches and resistance surfaces used perform well in predicting movements of an independent set of dispersing lions. Cost-distance approaches were generally more sensitive to resistance input than circuit theory, but differences in performance measures between resistance inputs were small across both approaches.
Conclusions
Findings suggest that home range data can be used to generate resistance surfaces for connectivity maps in this system, with independent dispersal data providing a promising approach to thresholding what is considered as “connected” when delineating corridors. Most dispersers traversed through landscapes that had minimal human settlement and are likely highly connected by dispersal. Research into limiting factors and dispersal abilities will be critical to understanding how populations will respond to increasing habitat fragmentation and human expansion.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Human population growth has led to extensive changes in land-use and habitat fragmentation; these changes have in turn led to the increasing isolation of wild animal populations (Haddad et al. 2015). The long-term viability of these populations relies on the existence of corridors that facilitate key biological processes such as mating, dispersal and gene flow (Rudnick et al. 2012; Hilty et al. 2019). Corridors allow species to respond effectively to environmental changes, such as climate (Chen et al. 2011; Wasserman et al. 2012) and land-use (Cushman et al. 2016; Kaszta et al. 2019, 2020). Among the many species facing significant challenges due to habitat fragmentation is the African lion (Panthera leo), which has undergone a population decline of 43% between 1993 and 2014, with only 8% of their historic range remaining (Bauer et al. 2016). Smaller, more fragmented populations of lions experience increased vulnerability to local extinctions (Woodroffe and Ginsberg 1998), disease susceptibility and genetic diversity loss (Trinkel et al. 2011), and human-wildlife conflict (Broekhuis et al. 2017; Cushman et al. 2018). As landscapes become increasingly fragmented, population connectivity will increasingly rely on large-scale movement, and in particular dispersal, to ensure long-term population persistence (Björklund 2003; Cushman et al. 2016). It is therefore crucial to identify effective corridors outside of existing protected areas to maintain population connectivity for wide-ranging species such as the African lion (Cushman et al. 2016, 2018).
Modern approaches address the challenges of maintaining connectivity by assessing the functional connectivity of landscapes, given a species’ population size, their dispersal ability, and landscape resistance patterns (Cushman et al. 2013b). These approaches use a resistance to movement surface as their foundation (hereafter, resistance surface), which quantifies the cost of movement as a function of landscape features (Zeller et al. 2012; Spear et al. 2015; Cushman et al. 2016). While resistance surfaces can be generated using a wide variety of input data and analytical approaches (reviewed in Zeller et al. 2012), surfaces based on GPS collar data have been shown to perform well in capturing processes such as dispersal in pumas (Zeller et al. 2018) and road crossings in bears (Cushman et al. 2014). Statistical tools like path- and step-selection functions are commonly employed to estimate resistance for connectivity studies. However, animal movement and resource selection are context-dependent and can vary across seasons (Zeller et al. 2020) and life stages (e.g. Elliot et al. 2014).
Dispersal is the key biological process enabling movement of individuals between populations, and it follows that dispersal data should produce the most reliable resistance values to model connectivity. Previous studies comparing corridors from dispersal data with those based on alternative data inputs have demonstrated substantial differences in predicted connectivity for African lions, Iberian lynx and African wild dogs (Elliot et al. 2014b; Gastón et al. 2016; Jackson et al. 2016). Interestingly, growing evidence suggests that home range data can act as a suitable surrogate for dispersal data in capturing habitat use and movement during the dispersal process for diverse mammalian species including leopards, desert bighorn sheep, kinkajous, brown bears and pumas (Newby 2011; Fattebert et al. 2015; Mateo-Sánchez et al. 2015; Keeley et al. 2016, 2017; Zeller et al. 2018). While dispersal data is relatively rare, home range use data has already been collected for many large carnivore species and leveraging this would allow for rapid functional connectivity assessment (Fattebert et al. 2015). This is a timely question to address, particularly given the acute need for accurate and defensible connectivity models in service of conservation, and the considerable challenges associated with obtaining data during dispersal events (Fagan and Calabrese 2006).
Once a resistance surface has been generated, researchers must select an appropriate connectivity algorithm; these are commonly based on cost distance (CD) or circuit theory (e.g., CircuitScape; CS). Both approaches use resistance as a foundation but differ in their assumptions about an animal’s knowledge of unfamiliar landscapes. Source-destination models of CD (Cushman et al. 2009), such as least-cost paths (Adriaensen et al. 2003) and least-cost corridors, predict optimal, lowest cost routes between specified source and destination locations. CS models relax the assumption that animals will select optimal routes, but still require a priori destinations. Previous studies formally comparing CD and CS methods have found varying performance based on the species, the biological process, and the quality of input data used. (e.g. Cushman et al. 2014; McClure et al. 2016; Zeller et al. 2018). Consequently, it is important to understand how generalisable these connectivity approaches are, and consider their sensitivity to different data inputs. At present it is unclear to what extent the contrasting results are the result of system- or species-specific ecological differences, or differences in how connectivity is modelled. Repetition of studies across species and systems in a comparable manner is needed to disentangle biological differences from methodological artefacts and provide general recommendations.
Zeller et al. (2018) represented a substantial advancement in generating general recommendations for connectivity modelling by introducing a standardised approach for assessing the performance of multiple data types and connectivity algorithms in capturing biological processes. However, despite the importance of validating model outputs, few studies have used independent dispersal data for this purpose likely due to the inherent difficulties in acquiring dispersal data for wild animals (exemptions can be found in McClure et al. 2016; Zeller et al. 2018). Lions, in particular, exhibit highly variable and context-dependent dispersal patterns (Elliot et al. 2014c), making it challenging to accurately capture the onset and duration of dispersal with a GPS collar. Moreover, the identification of dispersal onset and endpoint from GPS data is further complicated by the fact that lions may exhibit prospecting searches outside their natal territory before dispersal, and may also sometimes exhibit secondary dispersal (Elliot et al. 2014a).
To address these challenges, we present a simple and repeatable method to identify dispersal periods from GPS data (adapted from Weston et al. 2013), demonstrating its applicability to lions. We then use this independent dataset on lion dispersal to investigate the predicative capability of home range vs. dispersal data in determining movement trajectories during dispersal, comparing the performance of CD and CS approaches and their sensitivity to different data inputs. Our findings offer a direct comparison to the results of Zeller et al. (2018), contributing to the understanding of how generalisable recommendations regarding connectivity approaches and data inputs are across species. Furthermore, we extend the analytical paradigms from Zeller et al. (2018) and McClure et al. (2016) by incorporating a spatial randomisation procedure first described by Cushman et al. (2010a), that addresses challenges related to corridor delineation thresholds and the specificity of connectivity models, which remain open challenges in the field.
In doing so, we address the following research questions: (1) do connectivity surfaces based on dispersal data perform quantitatively better than those using male and female home range data? And (2) do source-destination CD or CS connectivity approaches best capture the observed routes selected by African lions during dispersal? Prior research has demonstrated not only substantial demographic difference in connectivity for lions (Elliot et al. 2014b), but also that CS is less sensitive to data choice than CD, and that for path-based movement data the CD algorithm outperforms CS models (Zeller et al. 2018). In line with these findings, we expect that, across both algorithms, dispersal surfaces will out-perform those produced using home range data in capturing the observed dispersal process. We also expect that CS will be less sensitive to demographic differences in resistance surface values, and that CD models will outperform CS models.
Methods
Dispersal data
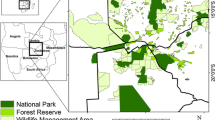
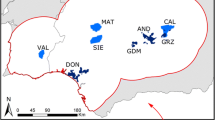
We identified periods of dispersal from Global Positioning System (GPS) collar data by adapting a distance-threshold method from Weston et al. (2013). We confirmed the applicability of this method to lions by applying it to five known subadult natal dispersers from Elliot et al. (2014a) and comparing our results to emigration dates determined by direct observation (Fig. 1 and Online Resource 1). We applied this method to 69 lions collared across Zimbabwe and Botswana as part of the Trans-Kalahari Predator Programme (Fig. 2). We estimated the point of emigration and settlement for dispersers by comparing movement distances and excursion durations to 20 known resident lions. The details for this procedure are given in supplemental information (Online Resource 1). This allowed us to identify a total of 28 dispersal events (Fig. 2), with 19 examples of natal dispersal (13 males, 6 females) and nine of adult dispersal (3 males, 6 females). We extracted GPS locations from these dispersal periods (n = 39,066) to use as our validation dataset. These data are independent from those used to parameterise the resistance surfaces described below.
Net displacement from the centroid of the first 3 months of data as a function of time showing labelled periods of residency and dispersal for one study animal. Vertical dashed lines represent point of dispersal and settlement, light red box indicates the window for dispersal estimated by field observation (Elliot et al. 2014). Inset graph shows a spatial representation of the same dataset. Horizontal lines represent the maximum and 98th percentile of ranging distances from the centre of their ranges for reference resident individuals and were used as distance thresholds to determine onset and departure from range restricted behaviour (see Online Resource 1)
Collar data was labelled as either resident or dispersing using a distance-threshold method adapted from Weston et al. (2013). Grey points represent the extent of the full GPS collar dataset, with points labelled as dispersing highlighted in colour. Green hatching represents National Parks and Game Reserves. All dispersal data presented here and used in the validation procedure were independent from the original dataset used by Elliot et al. (2014) to parameterise the resistance surfaces
As our approach is movement driven, the periods detected correspond to departures from (and return to) range-restricted behaviour. These include natal and secondary dispersal but may also include prospecting or exploratory trips outside of the normal range if these lasted for a sufficient period. Given our aim is to validate the movement component of connectivity, we focus on these periods of transience more generally to capture the routes lions selected when moving between established ranges. For simplicity, we hereafter refer to individuals exhibiting the range of transience behaviours as “dispersers”.
Connectivity models
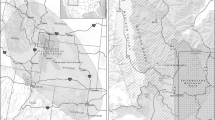
We used three demographic-specific resistance surfaces as the foundation for our connectivity models: adult females, adult males, and natal dispersers (Fig. 3, Step 1: a–c). These resistance surfaces were generated by Elliot et al. (2014b) using GPS data from 50 African lions (11 male natal dispersers, 20 adult males, and 19 adult females) in the Kavango Zambezi Transfrontier Conservation Area (KAZA). Elliot et al. (2014b) used a path level analysis (Cushman and Lewis 2010) to parameterise resistance, using three categories of environmental variables: land use, habitat, and anthropogenic factors (Online Resource 2). Variation in landscape permeability was largely driven by demographic differences in the strength of selection for protected areas, and avoidance of humans and agropastoral land (Elliot et al. 2014b). Details of this procedure are given in supplemental information (Online Resource 2). At the time of publication, authors estimated approximately 31% of their 1.4 million km2 study extent was managed for wildlife, including 26 National Parks, 297 Forest Reserves and 117 Wildlife Management Areas. Importantly, the study extent covered the Okavango-Hwange ecosystem, one of Africa’s 10 remaining lion ‘strongholds’ (Riggio et al. 2013) and the region where we obtained independent dispersal validation data. Surfaces based on GPS collar data, and particularly path selection functions, perform consistently well at capturing dispersal data (Zeller et al. 2018). These therefore provide a suitable basis from which to explore the impact of other factors, such as demography, on animal behaviour and movement as a function of landscape features.
Conceptual diagram representing (1) the inputs for connectivity modelling and (2) the validation process. Step 1 (a–c) depict resistance surfaces generated using dispersing individuals, adult females, and males, from left to right. Resistance is depicted from a gradient of low (white) to high (black) at a resolution of 500 m. Relative resistance values across the study extent range from 1–5312 for dispersers, 3–10,007 for adult males, and 2–23,497 for adult females. Overlaid are the start and end points of a single dispersal event represented as a lion and a cross, respectively. Step 2 (a) illustrates the spatial randomisation process, giving five examples (out of the total 100,000) of possible routes the same lion could have taken in grey, alongside the observed points in black. Step 2 (b–e and f–i) show performance measures 1–4 for CircuitScape (top row) and cost-distance (bottom row) for the disperser surface inputs. Mean percentile value (b) & (f) are shown with high connectivity values gaining increasingly blue colours, as with the 90th percentile corridors (e) & (i). In the latter case, everything outside the corridor has been cropped. (c) & (g) represent the null resistance comparison, and (d) & (h) the comparison with neighbourhood average landscape resistance. Both comparisons are shown with negative values gaining increasingly brown colours, and positive values in blue
We applied two connectivity approaches to our resistance surfaces: source-destination cumulative cost (cost distance; CD) and circuit theory (CircuitScape; CS). We applied each connectivity algorithm across the three resistance surfaces between the start and end points of each dispersal event to produce animal-specific surfaces (as in Zeller et al. 2018). We ran CS using Circuitscape 5.0 (Anantharaman et al. 2019) and CD using the gdistance R package (van Etten 2017). The CS approach produces a cumulative current flow surface between the start and end points, while the CD approach sums the two cost distance surfaces calculated on the start and end points. This produces a surface whose values are the joint cost of moving through a given pixel from the start to the end of a movement path. This is also known as the least cost corridor between the start and end points (Cushman et al. 2013b). We included a 100 km buffer around the extent of our validation dataset for our connectivity models to ensure any randomised paths created from dispersal trajectories (see Spatial Randomisation Procedure below) fell within the map extent. The final area covered approximately 250,868 km2 within the KAZA (Fig. 2).
Evaluating performance
We used five complementary validation methods to evaluate the performance of each connectivity surface (Fig. 3, Step 2: a–i), building on frameworks from previous studies (Cushman et al. 2014; McClure et al. 2016; Zeller et al. 2018),
Performance measure 1 (PM 1): connectivity value at observed dispersal points
We extracted the values at dispersal points on each connectivity surface to estimate mean connectivity as our first performance measure. To facilitate comparison across different connectivity algorithms, we percentile scaled connectivity values (Zeller et al. 2012; McClure et al. 2016). A higher performing model is assumed to result in higher connectivity values at observed dispersal points (Zeller et al. 2012; McClure et al. 2016), and would therefore evaluate how well the connectivity surface as a whole captures patterns of the dispersal process (represented here as GPS points from dispersal events).
Performance measure 2 (PM 2): comparison with null resistance
We then compared the connectivity values from our surfaces generated using empirical resistance to those using a null, isolation-by-distance model. We generated distance-only models by running connectivity algorithms on surfaces where resistance is set to a value of one for all pixels. We calculated the difference at each dispersal point between empirical and null resistance connectivity values. Any resulting surface with net positive values is identified as significantly outperforming the null model (Zeller et al. 2018). This isolates the performance of the resistance component of a connectivity surface by testing the null hypothesis that observed dispersal movement is unrelated to the resistance of the landscape i.e., that animals travel without regard to habitat quality (McClure et al. 2016).
Performance measure 3 (PM 3): comparison with resistance-only model
We generated local resistance-only models based on neighbourhood average landscape resistance (NALR). To do this, we calculated the focal mean of landscape resistance for each of our three resistance surfaces using a 5 × 5 matrix of neighbourhood cells (1 km buffer) centred on the focal pixel. This produced focal average resistance values, which we compared to empirical connectivity values sampled under dispersal points. As above, we calculated the difference at each dispersal point between empirical connectivity (based on CD and CS) and connectivity based on local resistance alone. This provides a counterpart to the null resistance comparison by testing the hypothesis that observed dispersal movement is driven largely by local resistance, rather than the wider pattern across the landscape (Cushman et al. 2014).
Performance measure 4 (PM 4): proportion of dispersal points in percentile corridors
Connectivity models are often used to delineate corridors for wildlife management by taking an upper percentage of the connectivity values (e.g., the 90th percentile, or the most traversable 10% of the landscape). In practice, a successful connectivity map for this purpose should therefore encompass a high proportion of dispersal points within the upper percentile of connectivity values (McClure et al. 2016; Zeller et al. 2018). We calculated the proportion of points (per lion) that fall in the top 10, and 1% of connectivity values to estimate mean proportion of points in corridors for each connectivity surface. Repeating the analysis for two percentile cut-offs allowed us to explore how the definition of what is considered “connected” impacts a simple corridor model’s performance.
We used the package lme4 (Bates et al. 2015) to fit mixed-effects models in R (R Core Team 2020) describing each of our above performance measures as a function of resistance type (male-, female-, disperser-, generated resistance) and connectivity approach (CD vs. CS). We also included a categorical variable describing the length of the dispersal event (short vs. long, i.e., > approximately 3 home range crossings). Animal ID was modelled as a random intercept to account for repeated samples from each individual. In the case of proportion of points, we used a binomial error distribution (with logit link). For PMs 1–4, we allowed up to three-way interactions in the maximal models, using likelihood ratio tests (of nested models) to provide an estimate of the statistical significance of each term and contrasts of estimated marginal means using the R package emmeans (Lenth 2020) to assess statistical differences across and within factor levels. This allowed us to test how PMs varied by resistance surface, if this was consistent between dispersal categories, and if these relationships depended on the connectivity approach used.
Performance measure 5 (PM 5): ranking in spatial randomisation procedure
Finally, we used a spatial randomisation procedure to compare connectivity values obtained at dispersal points to values obtained at random samples of points. We generated random samples of points by rotating each observed path using the trajR package in R (McLean and Volponi 2018) around its origin (by a value of radians drawn from a normal distribution) over 1 × 105 iterations (e.g. Cushman et al. 2010). By comparing values at observed points to the distribution of values from the random samples we can obtain an estimate of the probability of the observed outcome given the data (Fortin and Dale 2005). This allows us to test the hypothesis that routes taken during dispersal are unrelated to the predicted connectivity (Cushman et al. 2014). This measure is analogous to the second performance measure from McClure et al. (2016).
To quantify this comparison, we recorded the proportion of instances of 1 × 105 random samples, where a random draw of 39,066 points (total of observed dispersal points) produced a performance measure value higher than or equal to the value at the observed points. This allowed us to examine the specificity of the surfaces in capturing observed dispersal events and tells us which surfaces produce higher values than we would expect by chance given the distribution of connectivity across the landscape.
Comparing connectivity values at dispersal points to the distribution from the random samples provides a scale-independent means to compare surfaces without rescaling, as values obtained at points from the random samples implicitly fall on the same scale (per surface) as observed points. This is advantageous as it avoids the need to discretise the continuous connectivity values into bins. We therefore repeated the spatial randomisation procedure for PM 1, but directly using unscaled connectivity values to estimate the mean values. We included all connectivity surfaces (i.e., empirically-derived and null) in this ranking. This allowed us to examine how the process of discretising connectivity values (as is done in the generation of e.g., a percentile corridor) impacted the specificity of the surfaces. The response values used for the comparison of unscaled surface values were strictly positive, and right skewed. Therefore, we used the gamma and inverse gaussian families (with a “log link” function) in the mixed-effects models as a good, flexible option for bounded data. We selected the family which best fit each resistance/approach combination by comparing AIC values and examined the residuals of the models to confirm there were no major violations of model assumptions.
Results
Performance measures 1–4
For both CircuitScape (CS) and cost-distance (CD) surfaces, variation in mean percentile value at observed dispersal points (PM 1) was best described by an interaction term between resistance input and dispersal category (CS: 2(2) = 168.12, p = < 2.2e–16; CD: 2(2) = 751.2, p = < 2.2e–16), indicating that the effect of resistance input on mean percentile connectivity varied between long and short dispersal categories. Although statistically significant, differences between surfaces were small (< 1 percentile value) and all surfaces predicted mean values of > 97th percentile for all dispersal distance categories.
The data also supported an interaction term between resistance input and dispersal category in describing variation in the difference between empirical and null resistance surface (PM 2) percentile connectivity values for both connectivity approaches (CS: 2(2) = 443.63, p = < 2.2e–16; CD: 2(2) = 14.326, p = 0.00078). No combinations of connectivity approach and resistance input produced estimated differences where the lower boundary of 95% confidence intervals fell above 0 (i.e., no surfaces were identified that significantly outperformed a surface based on isolation-by-distance as measured by a higher percentile value on empirical surfaces under dispersal points).
Conversely, all empirical surfaces significantly outperformed null surfaces based on neighbourhood average local resistance (NALR) alone (PM 3). There was no support (at the 0.05 level) for the inclusion of either an interaction term, or the main effect of dispersal category in describing difference from NALR surfaces. Estimated marginal means (EMMs) for the CS and CD surface differences were 24, 20, and 30 for disperser, female and male surfaces, respectively. Contrasts between estimated marginal means (EMMs) for resistance inputs were all significant (p < 0.001, p-value adjustment: Bonferroni method for 3 tests).
All observed dispersal points in the short-distance dispersal category (bar two individuals on the CD-female surface combination) fell within the 90th percentile corridor (PM 4). This made modelling variation in the proportion of points redundant, and the predicted proportion of points for the long-distance category was > 0.999 for all surface-connectivity combinations. The 99th percentile corridors also captured a high proportion of dispersal points, with the lowest combination (long distance category on the CD-female resistance surface) predicting 72% of points in corridors. There was statistical support for the inclusion of a three-way interaction (2(3) = 135.27, p = < 2.2e–16) between connectivity algorithm, dispersal category and resistance input. The effect of resistance input on proportion of points in 99th percentile corridors varied between dispersal categories, and the magnitude of this effect varied between connectivity approaches. This implied there was a difference in the sensitivity of the approaches to resistance inputs, and that this relationship varied depending on the dispersal distance category. For short-distance dispersers, all combinations predicted 98% of points in corridors and above. For long-distance dispersers, surfaces optimised using disperser resistance consistently performed best (83 and 85% for CD and CS), followed by male resistance (80 and 85% for CD and CS), with female resistance capturing the lowest proportion of points (72 and 80% for CD and CS). The discrepancy between the top and bottom ranking combinations was larger for CD than CS (difference of 11 vs. 5%, respectively), and the difference between the top two CS surfaces was < 1%.
Rankings in spatial randomisation procedure
For both CD and CS, the mean percentile connectivity value of observed dispersal points was much higher than the distribution produced from the spatial randomization procedure (PM 5), indicating strong, non-random association of selected paths with routes through the landscape with higher predicted connectivity. The same pattern was observed using unscaled connectivity values to an even greater extent (Table 1). For percentile-scaled mean connectivity values, the relationship between the number of random draws (out of 100,000) that produced a mean of greater than or equal to the dispersal values and the resistance input differed between CS and CD approaches: CD surfaces performed better for shorter distance dispersers across all resistance surfaces, but while this also held true for male and disperser resistance under CS, for female resistance the opposite was true (Table 1).
For unscaled values, all CircuitScape (CS) and resistance combinations generated higher mean cumulative flow values at dispersal points than any of the randomised paths (Fig. 4, row 3). For short distance dispersers, all CD and resistance combinations had mean cost values lower than all randomised paths (Fig. 4, row 2). For animals travelling a longer distance (Fig. 4, row 1), a small number of the random draws for the female (67) and male (2) resistance inputs produced lower mean cost values than the observed dispersal points, with all observed values for disperser and null resistance inputs outperforming the random samples. Under the NALR scenario (Fig. 4, row 4), a proportion of 0.00003, 0.00054, and 0.01770 random samples produced mean average resistance values lower (i.e., performed equal to or better) than those observed at dispersal points for disperser, male and female resistance inputs, respectively. Note that, when considering unscaled connectivity, the generated values (cost for CD surfaces, cumulative flow for CS surfaces, and average focal resistance for NALR surfaces) differ in their interpretation, with lower values conferring higher connectivity for CD and NALR, and the converse for CS.
Observed mean connectivity values from unscaled surfaces within distribution of mean values from randomised paths. Mean values of cost (rows 1–2), current flow (row 3), and neighbourhood average resistance (row 4) from 100,000 random samples of points are presented at histograms, with the mean value obtained at dispersal points shown as a dashed vertical line. Where the mean value at dispersal points outperformed all random samples, the times smaller (or larger, for cumulative flow) than the lowest (or highest) value obtained in the random samples is shown next to the line. When connectivity surfaces were unscaled, cost-distance (CD) surfaces were the only connectivity approach that indicated statistical support for the inclusion of a term describing the maximum displacement of dispersers (Disperser: 2(1) = 7.43, p = 0.006; Female: 2(1) = 5.208, p = 0.007; Male: 2(1) = 7.406, p = 0.022, Null: 2(1) = 8.787, p = 0.003)
Both CS and CD again performed well for most combinations of approach, resistance input, and dispersal category in capturing proportion of points falling into both the 90th and 99th percentile corridors with a high level of specificity. CD and CS for all three resistance inputs (disperser, male and female) performed very well in the long-distance category, with a proportion less than 0.002 of randomized paths producing a higher number of points within the 90th percentile corridor and close to none in the 99th percentile corridor (Table 1). The short-distance dispersal category showed less discrimination in terms of proportion of points falling in percentile corridors, compared with the distribution of the randomized paths. This suggests that short distance dispersal is less strongly linked to predicted connectivity across these resistance surfaces than long distance dispersal. Using the CD method, the disperser resistance model performed best at discriminating short distance dispersal values, followed by the male resistance surface, with the female resistance surface performing least effectively (Table 1). In contrast, for CS methods in the low dispersal distance category, the disperser and male resistance surfaces both performed less well than the female resistance surface (Table 1).
Discussion
Our study draws together multiple approaches for evaluating connectivity models (e.g., Cushman et al. 2014; McClure et al. 2016; Zeller et al. 2018) and applies them concurrently in an integrated framework. By doing so we are able to draw conclusions about the relative performance of each connectivity-resistance surface, both as a whole and in its component parts. In addition, we can infer the likelihood that the observed dispersal process is related to the underlying connectivity.
Our first expectation—i.e., models based on dispersal-optimised resistance will outperform home range data in capturing dispersal movements—was supported in many, but not all, performance metrics and combinations of dispersal category and connectivity approach. For models based on local resistance alone (neighbourhood average landscape connectivity; NALR), the disperser surface received 18- and 590-times greater support than male and female resistance respectively, as captured by our spatial randomisation procedure (Table 1). This shows clear support for the idea that dispersal-optimised resistance provides the strongest match to observed dispersal routes. However, once a connectivity algorithm was applied, the performance for all three empirical resistance inputs was fundamentally very similar, and there was strong support that routes selected during dispersal are driven by landscape connectivity pattern (even for lower ranking combinations of resistance-connectivity; Table 1).
Our second expectation was that CS models would be less sensitive than CD to demographic differences in resistance surface values. This was clearly shown in our results from performance measure 4: proportion of points in percentile corridors. For both CD and CS approaches, models using disperser resistance captured the highest proportion of points for long-distance dispersal in a 99th percentile corridor, followed by male then female resistance. However, the discrepancy between the top and bottom ranking combinations was larger for CD than CS (with a difference of 11 vs. 5%, respectively), and the discrepancy between the top two CS surfaces was < 1%.
Our final expectation—that CD models will outperform CS models—was not met. Under the implementation we used to generate connectivity, both connectivity approaches and all resistance surfaces generated high connectivity values at dispersal points, and all were convincingly specific to points at dispersal. This is highly encouraging and supports the growing body of evidence that home range data can act as a surrogate to adequately capture the dispersal process (Newby 2011; Fattebert et al. 2015; Keeley et al. 2016, 2017; Zeller et al. 2018).
It is notable that no resistance-connectivity approach outperformed a null model based on distance alone, and that distance-only models showed perfect performance for both connectivity approaches in the spatial randomisation procedure (Table 1). In contrast, all resistance-connectivity combinations out-perform surfaces based on focal average resistance alone. The high relative support for disperser-NLAR over the male- and female-NALR surfaces suggests these demographic differences are biologically based and one part of the story. However, the strong performance of the null resistance model implies that resistance is not the major driver of performance in this system. Rather, our results suggest that it is the connectivity algorithms that drive good fit in our models, paired with known source-end points. The KAZA area is comprised of a large network of protected areas and forms one of the remaining strongholds for lions, and as such, the dispersal events we observed are likely moving through a highly-connected system. The fact that isolation by distance seems to play a large role in dispersal in our system is therefore consistent with previous findings that when landscapes are not highly limiting due to fragmentation, patterns of relative resistance and connectivity will not vary from isolation by distance (Cushman et al. 2011; Barros et al. 2019). Similar patterns have been shown for the effects of dispersal on genetic differentiation, both in simulation (Cushman et al. 2013a) and empirical studies (Short Bull et al. 2011; Vergara et al. 2017; Barros et al. 2019).
Conclusions regarding the relative importance of resistance in explaining emergent movement patterns is likely system- and process-specific. For example, Cushman et al. (2014) found substantial differences in performance between empirically derived resistance surfaces for both resistant kernel and factorial least cost path approaches in predicting the location of road crossing for American black bears. Were the landscape to contain greater variation in permeability, and more constraint in the routes available to lions, it is quite possible that the pattern of resistance across the landscape would be a much greater driver of the emergent pattern of observed dispersal paths. This highlights the need for large-scale replication of such studies across the species’ range to determine the location and effects of limiting factors and how they affect movement patterns. If resistance does not play a major role in determining the performance of connectivity models, then there is little need to further optimise resistance inputs (e.g., Cushman et al. 2013a). We would suggest future research might most productively focus on how to best define source points and identify suitable thresholds for defining corridors. Conversely, if different estimates of resistance are found to be important when landscapes are highly fragmented and limiting (e.g., Cushman et al. 2011; Short Bull et al. 2011; Vergara et al. 2017; Kaszta et al. 2021) this suggests that accurate estimation of resistance surfaces is important, especially when landscape variation limits movement. That would, in turn, focus future work on quantifying when landscape features become limiting to movement (Short Bull et al. 2011) and gene flow (Cushman et al. 2013a).
After observing the strong relative performance of the null resistance input, we estimated post-hoc the proportion of points that fell inside a 90th percentile corridor. While the proportion of dispersal points inside the corridors was comparable to the empirical surfaces, the null surface corridor did not outperform even a single draw of random points, as all points—random or observed—fell inside the corridor. A similar pattern was seen in short-distance dispersers for the empirical surfaces, where the proportion of points from the random samples was at least as high as the observed dispersal points in around 10–20% of the cases. As we increased the cut-off for the corridor to the 99th percentile, the proportion of dispersal points in the corridor remained high, but the models’ ability to discriminate between the dispersal points and the random samples improved (Table 1). This highlights two key points. First, that selecting a percentile threshold to consider a corridor may not be a trivial choice, and second, that binning values from a continuum of connectivity into categories risks losing some of the match between pattern and process (e.g., Cushman et al. 2010b). All models were convincingly specific at discriminating observed paths from those randomly generated in terms of unscaled mean connectivity, and this held when connectivity was percentile scaled (100 bins), although to a lesser extent. The percentile corridors were our most extreme form of categorisation and performed the worst at discriminating observed from random paths, until a sufficiently high threshold was selected. Here, we binned the continuous connectivity values into percentiles; however, a finer level of discretisation might be more appropriate as an approximation of a continuous scale.
There is a clear need for further research on thresholding and optimising the delineation of corridors, particularly in areas where human populations are expanding and demand for land is at a premium. Where available, using independent dispersal data is one way to select an appropriate threshold. However, given the challenges in obtaining such data, explicitly including a dispersal threshold into the estimation of connectivity offers a powerful alternative. The use of resistant kernels (Compton et al. 2007) may offer a particular improvement for cost-distance approaches, given these relax assumptions about known destination points and are able to explicitly incorporate a biological threshold for dispersal ability (e.g. Cushman et al. 2013; Wasserman et al. 2013; Cushman et al. 2015, 2018). We used single source-destination implementations of CD and CS in this study as the start and end points of our dispersal trajectories were known a priori. This allowed us to directly evaluate the connectivity approaches and resistance inputs—the focus of our study—while controlling for uncertainty in source points. Further research could usefully evaluate the performance of resistant kernel approaches relative to CS and source-destination formulations of CD, particularly for movement at the population-level.
Our results provide important advances in understanding the performance of different connectivity modelling methods and the sensitivity of this performance to the resistance surfaces used. These results have also led us to believe that more attention should be given to areas such as limiting factors, the relative importance of different resistance levels, and to evaluating a broader range of connectivity modelling methods (such as all-directional, dispersal limited modelling with methods like resistant kernels). Several studies have suggested that density, distribution, and dispersal ability of the study species can be as important, if not more than, differential resistance on functional connectivity (e.g., Cushman et al. 2013a). Future work should therefore build on this to also evaluate the effects of different numbers and spacing of dispersal source points and different dispersal abilities on connectivity predictions.
The overall outcome that all assessed connectivity-resistance combinations perform well is encouraging in its implications for modelling corridors for conservation. It is also encouraging that the performance of connectivity models using resistance based on parameters from home range data captures the observed dispersal movements well in this system. This means connectivity maps can be generated for multiple sites across the lions’ range where these data already exist. For each new context, validation will still be a critical element of defensible connectivity maps; however, given the range of species now supported, the outlook is positive. Understanding how connectivity and dispersal varies across the range of environments where these animals exist will be important in understanding the population viability of this rapidly declining carnivore.
Data availability
The datasets generated and analysed during the current study are available from the corresponding author on reasonable request.
References
Adriaensen F, Chardon JP, De Blust G et al (2003) The application of ‘least-cost’ modelling as a functional landscape model. Landsc Urban Plan 64:233–247
Anantharaman R, Hall K, Shah V, Edelman A (2019) Circuitscape in julia: high performance connectivity modelling to support conservation decisions. Preprint at https://arxiv.org/abs/1906.03542
Barros T, Carvalho J, Fonseca C, Cushman SA (2019) Assessing the complex relationship between landscape, gene flow, and range expansion of a Mediterranean carnivore. Eur J Wildl Res 65:44
Bates D, Mächler M, Bolker BM, Walker SC (2015) Fitting linear mixed-effects models using lme4. J Stat Softw. https://doi.org/10.18637/jss.v067.i01
Bauer H, Packer C, Funston PJ et al (2016) Panthera leo (errata version published in 2017). In: IUCN red list threat. Species 2016 e.T15951A115130419. https://doi.org/10.2305/IUCN.UK.2016-3.RLTS.T15951A107265605.en. Accessed 29 Oct 2020
Björklund M (2003) The risk of inbreeding due to habitat loss in the lion (Panthera leo). Conserv Genet 4:515–523
Broekhuis F, Cushman SA, Elliot NB (2017) Identification of human–carnivore conflict hotspots to prioritize mitigation efforts. Ecol Evol 7:10630–10639
Chen IC, Hill JK, Ohlemüller R et al (2011) Rapid range shifts of species associated with high levels of climate warming. Science 333:1024–1026
Compton BW, McGarigal K, Cushman SA, Gamble LR (2007) A resistant-kernel model of connectivity for amphibians that breed in vernal pools. Conserv Biol 21:788–799
Cushman SA, Huettmann F (eds) (2010) Spatial complexity, informatics, and wildlife conservation. Springer Japan, Tokyo
Cushman SA, Lewis JS (2010) Movement behavior explains genetic differentiation in American black bears. Landsc Ecol 25:1613–1625
Cushman SA, McKelvey KS, Schwartz MK (2009) Use of empirically derived source-destination models to map regional conservation corridors. Conserv Biol 23:368–376
Cushman SA, Chase M, Griffin C (2010) Mapping landscape resistance to identify corridors and barriers for elephant movement in Southern Africa. Spatial complexity, informatics, and wildlife conservation. Springer, Tokyo, pp 349–367
Cushman SA, Gutzweiler K, Evans JS, McGarigal K (2010) The gradient paradigm: a conceptual and analytical framework for landscape ecology. Spatial complexity, informatics, and wildlife conservation. Springer Japan, Tokyo, pp 83–108
Cushman SA, Raphael MG, Ruggiero LF et al (2011) Limiting factors and landscape connectivity: the American marten in the Rocky Mountains. Landsc Ecol 26:1137–1149
Cushman SA, Landguth EL, Flather CH (2013) Evaluating population connectivity for species of conservation concern in the American Great Plains. Biodivers Conserv 22:2583–2605
Cushman SA, Mcrae B, Adriaensen F et al (2013) Biological corridors and connectivity. Key topics in conservation biology, 2nd edn. Wiley, Oxford, pp 384–404
Cushman SA, Lewis JS, Landguth EL (2014) Why did the bear cross the road? Comparing the performance of multiple resistance surfaces and connectivity modeling methods. Diversity 6:844–854
Cushman SA, Elliot NB, Macdonald DW, Loveridge AJ (2015) A multi-scale assessment of population connectivity in African lions (Panthera leo) in response to landscape change. Landsc Ecol. https://doi.org/10.1007/s10980-015-0292-3
Cushman SA, Elliot NB, Macdonald DW, Loveridge AJ (2016) A multi-scale assessment of population connectivity in African lions (Panthera leo) in response to landscape change. Landsc Ecol 31:1337–1353
Cushman SA, Elliot NB, Bauer D et al (2018) Prioritizing core areas, corridors and conflict hotspots for lion conservation in southern Africa. PLoS ONE. https://doi.org/10.1371/journal.pone.0196213
Elliot NB, Cushman SA, Loveridge AJ et al (2014) Movements vary according to dispersal stage, group size and rainfall: the case of the African lion. Ecology 95:2860
Elliot NB, Cushman SA, Macdonald DW, Loveridge AJ (2014) The devil is in the dispersers: predictions of landscape connectivity change with demography. J Appl Ecol 51:1169–1178
Elliot NB, Valeix M, Macdonald DW, Loveridge AJ (2014) Social relationships affect dispersal timing revealing a delayed infanticide in African lions. Oikos 123:1049–1056
Fagan WF, Calabrese JM (2006) Quantifying connectivity: balancing metric performance with data requirements. In: Crooks KR, Sanjayan M (eds) Connectivity conservation. Cambridge University Press, Cambridge, pp 297–317
Fattebert J, Robinson HS, Balme G et al (2015) Structural habitat predicts functional dispersal habitat of a large carnivore: how leopards change spots. Ecol Appl 25:1911–1921
Fortin MJ, Dale MRT (2005) Spatial analysis: a guide for ecologists. Cambridge University Press, Cambridge
Gastón A, Blázquez-Cabrera S, Garrote G et al (2016) Response to agriculture by a woodland species depends on cover type and behavioural state: insights from resident and dispersing Iberian lynx. J Appl Ecol 53:814–824
Haddad NM, Brudvig LA, Clobert J et al (2015) Habitat fragmentation and its lasting impact on Earth’s ecosystems. Sci Adv. https://doi.org/10.1126/sciadv.1500052
Hilty JA, Keeley ATH, Lidicker WZ, Merenlender AM (2019) Corridor ecology: linking landscapes for biodiversity conservation and climate adaptation, 2nd edn. Island Press, Washington
Jackson CR, Marnewick K, Lindsey PA et al (2016) Evaluating habitat connectivity methodologies: a case study with endangered African wild dogs in South Africa. Landsc Ecol 31:1–15
Kaszta Ż, Cushman SA, Hearn AJ et al (2019) Integrating Sunda clouded leopard (Neofelis diardi) conservation into development and restoration planning in Sabah (Borneo). Biol Conserv 235:63–76
Kaszta Ż, Cushman SA, Htun S et al (2020) Simulating the impact of belt and road initiative and other major developments in Myanmar on an ambassador felid, the clouded leopard, Neofelis nebulosa. Landsc Ecol 35:727–746
Kaszta Ż, Cushman SA, Slotow R (2021) Temporal non-stationarity of path-selection movement models and connectivity: an example of African elephants in Kruger National Park. Front Ecol Evol 9:207
Keeley ATH, Beier P, Gagnon JW (2016) Estimating landscape resistance from habitat suitability: effects of data source and nonlinearities. Landsc Ecol 31:2151–2162
Keeley ATH, Beier P, Keeley BW, Fagan ME (2017) Habitat suitability is a poor proxy for landscape connectivity during dispersal and mating movements. Landsc Urban Plan 161:90–102
Lenth R (2020) Emmeans: estimated marginal means, aka least-squares means. R Core Team, Vienna
Mateo-Sánchez MC, Balkenhol N, Cushman SA et al (2015) Estimating effective landscape distances and movement corridors: comparison of habitat and genetic data. Ecosphere. https://doi.org/10.1890/ES14-00387.1
McClure ML, Hansen AJ, Inman RM (2016) Connecting models to movements: testing connectivity model predictions against empirical migration and dispersal data. Landsc Ecol 31:1419–1432
McLean DJ, Volponi MAS (2018) Trajr: an R package for characterisation of animal trajectories. Ethology. https://doi.org/10.1111/eth.12739
Newby J (2011) Puma dispersal ecology in the central Rocky Mountains. University of Montana, Missoula
R Core Team (2020) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Riggio J, Jacobson A, Dollar L, Bauer H, Becker M, Dickman A et al (2013) The size of savannah Africa: a lion’s (Panthera leo) view. Biodivers Conserv 22:17–35
Rudnick DA, Ryan SJ, Beier P et al (2012) The role of landscape connectivity in planning and implementing conservation and restoration priorities. Issues Ecol 16:1–21
Short Bull RA, Cushman SA, Mace R et al (2011) Why replication is important in landscape genetics: American black bear in the Rocky Mountains. Mol Ecol 20:1092–1107
Spear SF, Cushman SA, McRae BH (2015) Resistance surface modeling in landscape genetics. Landscape genetics. Wiley, Chichester, pp 129–148
Trinkel M, Cooper D, Packer C, Slotow R (2011) Inbreeding depression increases susceptibility to bovine tuberculosis in lions: an experimental test using an inbred-outbred contrast through translocation. J Wildl Dis 47:494–500
van Etten J (2017) R package gdistance: distances and routes on geographical grids. J Stat Softw. https://doi.org/10.18637/jss.v076.i13
Vergara M, Cushman SA, Ruiz-González A (2017) Ecological differences and limiting factors in different regional contexts: landscape genetics of the stone marten in the Iberian Peninsula. Landsc Ecol 32:1269–1283
Wasserman TN, Cushman SA, Shirk AS et al (2012) Simulating the effects of climate change on population connectivity of American marten (Martes americana) in the northern Rocky Mountains, USA. Landsc Ecol 27:211–225
Wasserman TN, Cushman SA, Littell JS et al (2013) Population connectivity and genetic diversity of American marten (Martes americana) in the United States northern Rocky Mountains in a climate change context. Conserv Genet 14:529–541
Weston ED, Whitfield DP, Travis JMJ, Lambin X (2013) When do young birds disperse? Tests from studies of golden eagles in Scotland. BMC Ecol 13(42):1–13. https://doi.org/10.1186/1472-6785-13-42
Woodroffe R, Ginsberg JR (1998) Edge effects and the extinction of populations inside protected areas. Science 280:2126–2128
Zeller KA, McGarigal K, Whiteley AR (2012) Estimating landscape resistance to movement: a review. Landsc Ecol 27:777–797
Zeller KA, Jennings MK, Vickers TW et al (2018) Are all data types and connectivity models created equal? Validating common connectivity approaches with dispersal data. Divers Distrib. https://doi.org/10.1111/ddi.12742
Zeller KA, Lewison R, Fletcher RJ et al (2020) Understanding the Importance of dynamic landscape connectivity. Land 9:303–303
Acknowledgements
GF was supported by the U.K. Natural Environment Research Council (NERC) through the DTP in Environmental Research (Grant NE/L002612/1). We thank Botswana’s Department of Wildlife and Zimbabwe Parks and Wildlife Management Authority for permission to conduct the study. We are grateful to Kathy Zeller for constructive criticism of an earlier draft of manuscript and to Bill Peterman for his input during the review process. We thank Jane Hunt and the field team in Zimbabwe for data collection. The authors would like to acknowledge the use of the University of Oxford Advanced Research Computing (ARC) facility in carrying out this work. https://doi.org/10.5281/zenodo.22558.
Funding
Author GF was supported by the U.K. Natural Environment Research Council (NERC) through the DTP in Environmental Research (Grant Number NE/L002612/1). Field research was funded by the Recanati-Kaplan and Robertson Foundations and Darwin Initiative for Biodiversity grants DAR3270, 162-09-015, EIDPO002.
Author information
Authors and Affiliations
Contributions
GF and SC conceived the ideas and designed methodology; AL and DWM managed field research; AL, DB, NE, and KK collected the data; GF analysed the data and led the writing of the manuscript. All authors provided comments on manuscript drafts and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Finerty, G.E., Cushman, S.A., Bauer, D.T. et al. Evaluating connectivity models for conservation: insights from African lion dispersal patterns. Landsc Ecol 38, 3205–3219 (2023). https://doi.org/10.1007/s10980-023-01782-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10980-023-01782-z