Abstract
To what extent particular plant defences against herbivorous insects are constitutive or inducible will depend on the costs and benefits in their neighbourhood. Some defensive chemicals in leaves are thought to be costly and hard to produce rapidly, while others, including volatile organic compounds that attract natural enemies, might be cheaper and can be released rapidly. When surrounding tree species are more closely related, trees can face an increased abundance of both specialist herbivores and their parasitoids, potentially increasing the benefits of constitutive and inducible defences. To test if oaks (Quercus robur) respond more to herbivore attacks with volatile emission than with changes in leaf phenolic chemistry and carbon to nitrogen ratio (C: N), and whether oaks respond to the neighbouring tree species, we performed an experiment in a forest in Poland. Oak saplings were placed in neighbourhoods dominated by oak, beech, or pine trees, and half of them were treated with the phytohormone methyl jasmonate (elicitor of anti-herbivore responses). Oaks responded to the treatment by emitting a different volatile blend within 24 h, while leaf phenolic chemistry and C: N remained largely unaffected after 16 days and multiple treatments. Leaf phenolics were subtly affected by the neighbouring trees with elevated flavan-3-ols concentrations in pine-dominated plots. Our results suggest that these oaks rely on phenols as a constitutive defence and when attacked emit volatiles to attract natural enemies. Further studies might determine if the small effect of the neighbourhood on leaf phenolics is a response to different levels of shading, or if oaks use volatile cues to assess the composition of their neighbourhood.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Plant defences against herbivorous insects are tailored to counter herbivore pressure and can also respond to herbivory by induction and upregulation of various traits. Plant defences can prevent insect herbivory altogether when they repel foraging insects (Bernays and Chapman 1994; Gripenberg et al. 2010). For example, ovipositing Lepidoptera may avoid leaves with dense trichomes (Kumar 1992; War et al. 2013), high concentrations of defensive chemicals (Renwick and Chew 1994), predatory ants (Freitas and Oliveira 1996), or signs of existing herbivore infestation (De Moraes et al. 2001). Subsequently, plant defences can hamper feeding or digestion, or otherwise harm insects, thus lowering the survival and growth of insect herbivores (Feeny 1968; Forkner et al. 2004; Frost and Hunter 2008; Nykänen and Koricheva 2004). Plants can also defend themselves against insect herbivores indirectly by increasing the natural enemy pressure on their herbivores by slowing insect growth (Kondoh and Williams 2001), hosting natural enemies with shelter or food (Wäckers et al. 2005; Walter 1996), or signalling to natural enemies that herbivores are present (Amo et al. 2013; Pearse et al. 2020; Price et al. 1980). These signals usually include volatile organic compounds (VOCs) that attract natural enemies of herbivores such as parasitoids, predatory insects, and birds (Amo et al. 2013; Dicke 2016; Janssen et al. 2002). How such defence traits are integrated depends on their costs and benefits in their ecological context (Agrawal and Fishbein 2006).
Constitutive plant defences have different benefits and costs than induced defences. Constitutive defences are present regardless of insect attacks, while induced defences are generated or upregulated when a plant is attacked (Tollrian and Harvell 1999). Constitutive defences are effective at repelling foraging herbivores as these insects tend to avoid better-defended or less nutritious plants (e.g. ovipositing insects; Gripenberg et al. 2007). Subsequently, constitutive defences reduce the growth and survival of herbivores (Nykänen and Koricheva 2004). However, maintaining high levels of constitutive defences can be energetically costly for the plant, resulting in trade-offs with other physiological processes (Monson et al. 2022; Moreira et al. 2014; Stamp 2003). Induced defences can be less costly, as defence investments are only higher during actual herbivore attacks (Karban 2011).
The speed of induction is a key factor for such defences, as they must increase rapidly to ensure efficiency. Defences that take relatively long to accumulate in sufficient concentrations are not likely candidates for induction by herbivore attacks. However, delayed induction of these types of defences can still be adaptive when current herbivory predicts future herbivory (Mertens et al. 2021; Rieske and Dillaway 2008). This can lead to current herbivory having knock-on effects on future herbivores (Ekholm et al. 2020; Karban and Baldwin 2007; Karban and Takabayashi 2018). In contrast, VOCs are usually of lower molecular weight and may peak immediately after herbivory (e.g. green leaf volatiles (GLV); Hoballah et al. 2004; Robert et al. 2013) or in the next photosynthetic cycle (e.g. terpenes; Robert et al. 2013). This makes them a particularly fast response against herbivores. Fast induction has also been reported for non-volatile chemical defences, such as terpenoids and lipophilic flavonoids (e.g. Keefover-Ring et al. 2016; Martemyanov et al. 2012).
The inducibility of many VOCs and their upregulation in response to external stimuli is linked to at least two ecological factors. First, signalling to natural enemies of herbivores will only work if the signal is a reliable indicator of current or recent herbivory. Second, VOCs can also have an ecological cost when volatiles attract herbivores rather than repel them (Ghirardo et al. 2012; Heil 2004). This could potentially explain the extensive variation in VOC production among individual plants within populations (Bertić et al. 2021).
The efficacy and relative importance of various plant defences depend on the composition of local communities of herbivores and their natural enemies, and by the predictability of herbivory. When herbivore pressure is low and unpredictable, inducible defences are favoured, since they are produced only when needed and do not require a constitutive investment into their maintenance (Åström and Lundberg 1994; Mertens et al. 2021). This may in part explain why the relative importance of constitutive and induced plant defences differs between habitats that differ in herbivore pressure (Agrawal and Hastings 2019; Pellissier et al. 2016).
Due to their size and longevity, trees encounter numerous insects during their lifetimes, which selects for strong constitutive defences (Feeny 1976). However, growing far from conspecifics or other phylogenetically closely related individuals can reduce the apparency of small trees (circumference < 1 m) to specialised herbivores (Vialatte et al. 2010). Therefore, the rate of insect attacks on small trees may be reduced or less predictable when they are in a neighbourhood consisting mainly of distantly related species (Mallick et al. 2023; Yguel et al. 2011; but see opposite trend on larger trees Molleman et al. 2022). Specifically, oak trees surrounded by distantly related neighbours experience less herbivory than oak trees that are surrounded by conspecifics and closely related tree species (Yguel et al. 2011). In addition, the efficacy of VOCs in attracting natural enemies of herbivores may depend on the neighbourhood because the neighbourhood affects the natural enemy pool and ambient VOCs (Jactel et al. 2011; Janssen et al. 2002). However, the effect of neighbourhood on the effectiveness of plant defences has rarely been studied (Dicke and Hilker 2003).
Since neighbouring trees can affect pressure by insect herbivores, plants might benefit from tailoring their defences to their neighbours. Populations of oaks in which individuals tend to be surrounded by conspecifics appear to have strong constitutive defences against insects (provenance experiment; Pihain et al. 2019). Similarly, we can expect evolution of defence induction in response to neighbourhood composition. Such a plastic response in defence strategies to neighbourhood composition requires interaction among plants, which can be mediated by ambient VOCs or through underground connections (Brosset and Blande 2021; Karban 2021; Simard 2018). Most studies on such interactions focus on within-species interactions where plants without herbivores are exposed to volatiles from plants with herbivores, and then often show priming to respond to herbivory or induction of defences (e.g. Soler et al. 2007; Tscharntke et al. 2001). However, plants can also respond to herbivore-induced VOCs from distantly related plant species (Karban et al. 2000; Kessler et al. 2006). Studies addressing how neighbourhood might affect plant defence without induction of neighbours are rare (Glinwood et al. 2011; Ninkovic et al. 2016). We might expect that trees will always host some herbivores so that on average neighbourhoods will be reflected in ambient herbivore-induced VOCs. As a result, lower abundance of conspecifics in a neighbourhood might translate into less priming or induction from conspecifics. Notably, the identity of neighbours can additionally affect the light environment and micro-climate, and this can also affect leaf chemistry (Barbier et al. 2008; Kovács et al. 2017). In summary, (1) neighbourhood can select for different levels of investment in constitutive defences, (2) plants can plastically upregulate defences, and (3) there are signals informing plants on their neighbourhood. We hypothesise that trees can plastically increase investment in leaf defences (higher concentration of defensive chemicals, reduced nutritional value) when they are placed among closely related neighbours where herbivore pressure is high.
We studied constitutive and induced chemical defences of oak saplings (Quercus robur) placed in different neighbourhoods in a forest in Poland. Oaks are important components of temperate forests in the northern hemisphere and harbour a high diversity of species of herbivorous insect (Southwood 1961). They have a rich secondary chemistry, including various tannins that affect insect herbivores through oxidative activity and protein precipitation (Barbehenn et al. 2006; Bertić et al. 2021; Feeny 1970; Forkner et al. 2004; Salminen and Karonen 2011; Volf et al. 2022). When induced by (simulated) caterpillar feeding, the oak species that we studied emit VOCs that are associated with the attraction of natural enemies of caterpillars (Mrazova et al. 2023; Volf et al. 2021). Oaks also show genetically-determined intraspecific variation in defences that have consequences for their susceptibility to insect herbivores (Ghirardo et al. 2012; Pihain et al. 2019; Roslin et al. 2006; Valdés-Correcher et al. 2021). However, the possible plastic response to neighbourhood composition has not been studied. To test oak responses to insect herbivory and neighbourhood composition, we placed potted oak saplings in three types of neighbourhood (dominated by either oak, beech, or pine), and treated half of the saplings with methyl jasmonate (MeJA). MeJA is a phytohormone that is central in mediating the response of plants to insect herbivory and is commonly used in ecological studies to mimic plant responses to leaf-chewing herbivores (e.g. Amo et al. 2022; Mrazova and Sam 2019; Semiz et al. 2012). After applying MeJA, we sampled VOCs, and intermittently monitored leaf traits using a leafclip sensor. After 16 days, we determined the carbon-nitrogen ratio (C: N), phenolic composition, and antioxidant activity of the leaves. We tested the following hypotheses: (1) leaf phenolics and C: N are mostly constitutive, while VOCs are rapidly induced by the MeJA treatment, and (2) oaks among closely related neighbours upregulate their phenolic defensive leaf chemistry.
Methods and Materials
Experimental Design
We carried the study out in Puszcza Zielonka forest near Kamińsko village in Western Poland, using potted saplings of Q. robur (purchased from Szkółki Kórnickie, Kórnik, Poland) in June 2022. On June 2nd, we placed 17 pairs of potted saplings (within-pair distance 10 m) in 10 sites that varied in neighbourhood. After excluding three saplings eaten by deer, we had ten oak saplings in four areas dominated by oaks (Q. petraea), seven in two areas dominated by beeches (Fagus sylvatica), and twelve in four areas dominated by pines (Pinus sylvestris). The undergrowth in these neighbourhoods was sparse due to browsing wildlife, and no other oak saplings were present. We planted the saplings levelled with the surface, but they remained in the pots and were watered as needed.
Two days after placing the saplings in the field (June 4th), we sprayed one individual within each pair with 2mM of the plant hormone MeJA, and the other with a control solution (distilled water with molecular-grade ethanol and triton X-100). After the solution had dried, we sampled VOCs from the oaks following Kallenbach et al. (2014). Specifically, we placed two 1.5 cm polydimethylsiloxane tubes (PDMS; Carl Roth GmbH, Karlsruhe, Germany) on a stainless-steel wire attached to the measured branch, and enclosed them in a polyamide bag (Stella Pack S. A. 35 × 43 cm). We attempted to enclose similar-sized twigs. The VOCs from the headspace were passively adsorbed onto the PDMS tubes that were collected after 24 h.
We then repeated the MeJA and control treatments every four days (June 8th, 12th, and 16th), and we switched the position of saplings within pairs twice during the experiment. To gauge how rapidly MeJA treatment and neighbourhood affect leaf chemistry over time, we took measurements using the Dualex+ (METOS Pessl Instruments GmbH, Weiz, Austria) five times during the study period (June 4th, 5th, 10th, 12th, and 16th ). For each sapling, we measured four leaves in two places. The Dualex + is a leafclip sensor that non-invasively measures the nitrogen balance index (NBI), and indices of chlorophyll, flavonol, and anthocyanin content in leaves (Goulas et al. 2004). Chlorophyll content and NBI can be expected to reflect light conditions (Šušić et al. 2023) and might also respond to herbivory (Visakorpi et al. 2020). Sixteen days after the start of the experiment (June 20th), we harvested all the leaves for analysis of phenolics and C: N. Leaves were dried at 35 °C for three days, chopped in a blender and then milled using an IKA mill MF 10.1 (IKA-Werke GmbH, Staufen, Germany). Sub-samples for C: N analysis were dried at 65 °C for 24 h.
Volatile Emission Measurements
The VOCs absorbed in the PDMS tubes were analysed using gas chromatography-mass spectrometry (GC/MS) using a Thermal Desorption (TD) TD100-xr (Markes International, Bridgend, UK) installed on a Trace 1300 gas chromatograph coupled to an ISQ quadrupole mass spectrometer (Thermo Fisher Scientific, Waltham, MA, USA). The PDMS tubes were inserted into metal tubes and placed in the TD instrument. The tube was dried with a helium flow (20 mL/min) for 5 min. Then it was heated to 200 °C for 5 min (the helium flow was 60 mL/min). The desorbed VOCs were captured in a cold trap (0 °C), which was then heated to 300 °C for 4 min. The split flow was 10 mL/min. GC separation was performed using a VF-5MS capillary column (30 m × 0.25 mm id, 0.25 μm film thickness, Agilent Technologies, Santa Clara, CA, USA) and helium as the carrier gas at a flow rate of 2 mL/min, following the temperature conditions: 39 °C for 1 min, increasing at a rate of 8 °C/min to 95 °C, then increasing to 300 °C at a rate of 20 °C/min. Finally, the temperature was held for 2 min. VOCs were detected using an electron ionisation (EI) ion source (70 eV, 230 °C) and a single quadrupole in a scan range of 33–500 m/z. The Thermo Xcalibur 2.0 software (Qual Browser, Quan Browser, Thermo Fisher Scientific) was used for data processing, method calibration, and validation. VOC emissions were quantified as the area under the chromatographic peak.
Leaf Phenolics And C: N analysis
We measured total phenolic content using the Folin–Ciocalteu method (Sokół-Łętowska et al. 2020; details in Supplementary Information 1) and antioxidant activity as Trolox equivalents (Gao 2000, Re 1999; details in Supplementary Information 1). The content of individual (classes of) phenolics and their total content were determined using an optimized high-performance liquid chromatography coupled photodiode array (HPLC-PDA) method (Sokół-Łętowska et al. 2020; details in Supplementary Information 1). Carbon and nitrogen contents (% d.m.) were determined using a 2400 CHN Elemental Analyzer (Perkin Elmer, Waltham, MA, USA).
Data analysis
For VOC emissions, we log-transformed the data to approach a normal distribution of residuals. We used concentration of phenolics as mg/g of the dry leaf tissue. We analysed the composition of VOCs and leaf phenolics using non-metric multidimensional scaling (NMDS) and PERMANOVA in the R package vegan (Oksanen et al. 2007; R_Core_Team 2023). As predictors in PERMANOVA models, we used treatment (sprayed with MeJA vs. control), and neighbourhood (dominated by oak, beech, or pine), and the interaction between treatment and neighbourhood.
We compared the log of total VOC emission, C: N, sums of classes of phenolics, and antioxidant activity across treatments using ANOVA analysis. When a predictor affected VOC or phenolic composition, we further explored which VOCs or phenols most responded to the predictor using ANOVAs. We used the Bonferroni method to correct for multiple testing within each set of ANOVAs.
To test if treatment, or neighbourhood and their interaction affected changes over time in NBI, chlorophyll content, flavanol index, and anthocyanin index (data from Dualex + leafclip sensor), we implemented mixed models with sapling ID as a random effect in R. As predictors we used day, treatment (sprayed with MeJA vs. control), and neighbourhood (dominated by oak, beech, or pine), and the interaction between treatment and neighbourhood. We then performed model selection by step-wise eliminating non-significant predictors.
Results
VOC Emissions
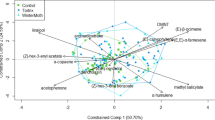
In total, 18 VOCs were identified in Q. robur saplings (Table 1). These belong to aldehydes (1), alkanes (4), GLVs (1), monoterpenes (6), and sesquiterpenes (6). The oak saplings treated with MeJA had on average much higher total VOC emissions than controls (5.5 times, N = 30, F = 6.89, p = 0.014). The VOC composition was also clearly affected by the MeJA treatment (Table 2; Fig. 1a). At least four individual Q. robur saplings appeared not to have responded to MeJA treatment (placed among control saplings in Fig. 1a).
NMDS plots of (a) VOC emissions, and (b) phenolic composition by MeJA treatment (C = control, T = MeJA treatment) and neighbourhood for Q. robur saplings. Indivdiuals that appeared not to have responded to MeJA treatment are encircled. Statistical results are in Table 2
Specifically, oak saplings treated with MeJA emitted significantly elevated amounts of six VOCs: the monoterpenes Eucalyptol, Linalool, o-Cymene, β-Ocimene, β-cis-Ocimene and the sesquiterpene α-Copaene (after correcting for multiple testing; Table 1).
Phenolics in Leaves
The treatment with MeJA did not affect the phenolic composition of leaves (Fig. 1b; Table 2). Neighbourhood also did not have an overall significant effect on phenolic composition (Fig. 1b; Table 2). The total concentrations of all major classes of phenolics were not significantly higher in saplings that received the MeJA treatment (Table 3a, Fig. 2a). Flavonols showed significantly lower concentrations in induced trees (Table 3a; Fig. 2a). Antioxidant activity and C: N also did not differ significantly between the MeJA and control treatments (Fig. 2a, Table 3a).
Effects of (a) MeJA (T = treated, C = control), and (b) neighbourhood (dominated by beech, oak, or pine) for all phenolics (All PP) in mg gallic acid equivalent/g, classes of phenolics as mg/g of the dry leaf tissue (summing concentrations within classes), and antioxidant activity (Antiox. ac.) expressed as Trolox equivalent in uM Tx/g for Q. robur saplings. The total flavonol concentration was significantly lower for MeJA-treated Q. robur saplings (Table 3a), and the total flavan-3-ols concentrations differed marginally significantly between neighbourhoods (Table 3b)
The total concentrations of phenolics in leaves of oak saplings were not significantly affected by neighbourhood (Table 3b). Only the sum of flavan-3-ols concentrations was significantly different between neighbourhoods (Table 3b), with higher concentrations in neighbourhoods dominated by pines (Fig. 2b). C: N and antioxidant activity did not differ significantly between neighbourhoods (Table 3b).
After correction for multiple testing, no phenolic compound differed significantly in concentration between saplings placed in different neighbourhoods (Table 3b). Without this correction, five of 39 phenolic compounds had significantly different concentrations among neighbourhoods (Table 3b).
Temporal Changes in Leaf Chemistry
To gauge the speed of leaf phenolics induction and changes in NBI and chlorophyll concentrations, we measured leaf parameters with the leafclip Dualex + at intervals during the study. All measured parameters changed over time (Table 4). For anthocyanin we found significantly lower values for oak saplings treated with MeJA, but the difference between treatment and control saplings decreased rather than increased over time despite multiple applications of MeJA (Table 4; Fig. 3).
Discussion
To test our hypotheses on effects of herbivory and neighbourhood on plant defences, we placed oak saplings in neighbourhoods dominated by oak, beech, or pine trees, and then treated half with the phytohormone MeJA. We showed that VOCs were induced with MeJA application but leaf phenolics, C: N, NBI, and chlorophyll concentration were not changed. Leaf phenolic composition did not differ significantly between saplings surrounded by oaks, beeches, and pines, but flavan-3-ols were elevated in neighbourhoods dominated by pines. These results show that VOCs can rapidly be induced, while phenolic composition and leaf nutritional value do not respond rapidly to a chemical mimic of herbivory. Our findings also suggest that the composition of neighbouring trees can induce rapid changes in leaf chemistry in oak saplings.
Response to MeJA
Many inducible VOCs serve as infochemicals, attracting natural enemies of herbivores (Dicke and Hilker 2003; Karban 2021). The significant increases in emission of three VOCs within 24 h after induction provide evidence that VOCs are readily inducible in oak saplings. The significantly increased emission of monoterpenes and a sesquiterpene corroborates other studies on oaks (Mrazova et al. 2023; Semiz et al. 2012). The emission of these VOCs indicates signalling to natural enemies (Rosenkranz et al. 2021). In particular, terpenoids often act as signals to natural enemies (Rosenkranz et al. 2021). Moreover, emission of these volatiles by oak saplings has been shown to be associated with the recruitment of natural enemies of caterpillars (Mrazova et al. 2023).
In contrast to VOC emissions, phenolics and nutritional quality of leaves (C: N and NBI) showed no response to MeJA induction over 16 days, although the leafclip measurements indicated slightly higher anthocyanin levels in MeJA-treated saplings. This weak or absent induction of phenolics corroborates delayed or weak responses to MeJA in leaf phenolic chemistry in mature oaks (Ekholm et al. 2020; Volf et al. 2021), and may be typical for woody plants (Haukioja and Hanhimäki 1985; Nykänen and Koricheva 2004). This difference in inducibility between VOC emissions and phenolics was expected based on purported benefits and costs of polyphenols such as tannins and volatiles. In short, recruiting natural enemies using VOC signalling probably needs to be rapid to be effective. VOC productions might be facilitated by the possibly low cost of production (Robert et al. 2013). In contrast, upregulating defensive leaf chemistry may have little effect on herbivores that have already established (Nykänen and Koricheva 2004), while producing such defences may be costly for some defensive chemicals (Stamp 2003).
Effect of Neighbourhood
We found some effects of neighbourhood on concentrations of flavon-3-ols and a few other polyphenols in oak leaves. This effect occurred over the experimental period of 18 days, which suggests that concentrations of some phenolics can change over relatively short periods, if triggered by stimuli other than MeJA induction. Phenolic compounds serve multiple functions in plants, including defence against herbivores and UV protection (Salminen and Karonen 2011). Since we used potted plants that were not affected by soil conditions at the given sites, the main factors in play could primarily involve either shading and the resulting reductions in UV and desiccation stress, or stimulation by VOCs emitted by the neighbouring trees. The shading gradient decreases from beech trees through oaks to pines (Barbier et al. 2008; Kovács et al. 2017). Indeed, in saplings surrounded by pines, we found the highest levels of flavan-3-ols, chemicals that can play important roles in protection against radiation (Hernández et al. 2011; Šamec et al. 2021; Zhang et al. 2013). However, if shading was the dominant factor affecting leaf chemistry of studied saplings, we would expect to also find differences between neighbourhoods in chlorophyll content or NBI (Šušić et al. 2023). No such effects were detected with the leafclip sensor. Perhaps chlorophyll and NBI take more time to respond to shading. The leaf phenolic composition may also be a plastic response to prime the plant for increased herbivore pressure in particular neighbourhoods (similar to inter-population difference; Pihain et al. 2019). In this case, the effect of pine VOCs on leaf chemistry may reflect lower abundance of parasitoids that are specialised on insects that feed on oaks in pine-dominated forests. In turn, this may select oak trees to rely more on defensive leaf chemistry than on emitting signals to parasitoids (no such reduction was apparent in our data but exposure to neighbourhood was only two days before VOC sampling). Elevated leaf phenolic concentrations would not be needed in oak-dominated neighbourhoods where VOCs reduce herbivore pressure by attracting parasitoids. While airborne communication between plants appears widespread (Karban 2021), further studies are needed to verify if ambient VOCs in the forest understory can induce changes in leaf chemistry of saplings, and if this correlates with herbivore pressure or effectiveness of VOC emission in recruiting natural enemies.
Individual Differences
We found individual differences in response to MeJA treatment among Q. robur saplings. Four MeJA-treated Q. robur saplings clustered with controls. The unresponsiveness to MeJA of these individuals indicates that there is variation among individuals in the response to induction, as shown before in the same species (Ghirardo et al. 2012). Specifically, Ghirardo et al. (2012) found that oaks that were classified as ‘susceptible’ to defoliation by the moth Tortrix viridana (often a major pest on oaks) emit a strong VOC signal that attracted T. viridana females, while ‘resistant’ oaks tended have low volatile emission. Therefore, not emitting VOCs may be adaptive in the face of insect herbivores that use the VOCs to locate profitable trees. This ecological cost of VOC emission may also vary with neighbourhood. Such alternative defence strategies emphasize the importance of considering not only differences between species (Mrazova et al. 2023) and populations (Pihain et al. 2019; Valdés-Correcher et al. 2021), but also variation within plant populations.
Conclusions
We found that most Q. robur saplings induced with the phytohormone MeJA emitted a significantly increased and altered VOC blend (six out of 18 identified VOCs were elevated), but some individuals maintained low volatile emissions. In contrast, responses to MeJA in terms of leaf phenolics and nutritional quality were absent or small. The neighbourhood in which saplings were placed affected flavan-3-ol concentration within 18 days of exposure. Our results suggest that oak saplings rely mostly on constitutive leaf chemistry to defend directly against insect herbivores, and most (but not all) oak saplings emit VOCs, some of which are known to attract natural enemies of insect herbivores. The effect of the neighbourhood on phenolics might be a response to variation in levels of abiotic stress, in particular the light environment, or a response to the way herbivore pressure and their natural enemies are affected by the neighbourhood of a tree.
References
Agrawal AA, Fishbein M (2006) Plant defense syndromes. Ecology 87(S132–S149). https://doi.org/10.1890/0012-9658(2006)87[132:PDS]2.0.CO;2
Agrawal AA, Hastings AP (2019) Trade-offs constrain the evolution of an inducible defense within but not between plant species. Ecology 100:e02857. https://doi.org/10.1002/ecy.2857
Amo L, Jansen JJ, van Dam NM, Dicke M, Visser ME (2013) Birds exploit herbivore-induced plant volatiles to locate herbivorous prey. Ecol Lett 16:1348–1355. https://doi.org/10.1111/ele.12177
Amo L, Mrazova A, Saavedra I, Sam K (2022) Exogenous application of methyl jasmonate increases emissions of volatile organic compounds in pyrenean oak trees, Quercus pyrenaica. Biology 11:84
Åström M, Lundberg P (1994) Plant defence and stochastic risk of herbivory. Evol Ecol 8:288–298
Barbehenn RV, Jones CP, Karonen M, Salminen J-P (2006) Tannin composition affects the oxidative activities of tree leaves. J Chem Ecol 32:2235–2251
Barbier S, Gosselin F, Balandier P (2008) Influence of tree species on understory vegetation diversity and mechanisms involved—a critical review for temperate and boreal forests. Forest Ecol Manag 254:1–15
Bernays E, Chapman R (1994) Behavior: the process of host-plant selection. In: Host-plant selection by phytophagous insects. pp 95–165
Bertić M et al (2021) European oak chemical diversity – from ecotypes to herbivore resistance. New Phytol 232:818–834. https://doi.org/10.1111/nph.17608
Brosset A, Blande JD (2021) Volatile-mediated plant–plant interactions: volatile organic compounds as modulators of receiver plant defence, growth, and reproduction. J Exp Bot 73:511–528. https://doi.org/10.1093/jxb/erab487
De Moraes CM, Mescher MC, Tumlinson JH (2001) Caterpillar-induced nocturnal plant volatiles repel conspecific females. Nature 410:577–580. https://doi.org/10.1038/35069058
Dicke M (2016) Plant phenotypic plasticity in the phytobiome: a volatile issue. Curr Opin Plant Biol 32:17–23
Dicke M, Hilker M (2003) Induced plant defences: from molecular biology to evolutionary ecology. Basic Appl Ecol 4:3–14
Ekholm A, Tack AJ, Pulkkinen P, Roslin T (2020) Host plant phenology, insect outbreaks and herbivore communities: the importance of timing. J Anim Ecol 89:829–841
Feeny PP (1968) Effect of oak leaf tannins on larval growth of the winter moth Operophtera brumata. J Insect Physiol 14:805–817
Feeny PP (1970) Seasonal changes in oak leaf tannins and nutrients as a cause of spring feeding by winter moth caterpillars. Ecol 51:565–581
Feeny P (1976) Plant apparency and chemical defense. Biochemical interaction between plants and insects. Springer, pp 1–40
Forkner RE, Marquis RJ, Lill JT (2004) Feeny revisited: condensed tannins as anti-herbivore defences in leaf‐chewing herbivore communities of Quercus. Ecol Entomol 29:174–187
Freitas AV, Oliveira PS (1996) Ants as selective agents on herbivore biology: effects on the behaviour of a non-myrmecophilous butterfly. J Anim Ecol :205–210
Frost CJ, Hunter MD (2008) Herbivore-induced shifts in carbon and nitrogen allocation in red oak seedlings. New Phytol 178:835–845. https://doi.org/10.1111/j.1469-8137.2008.02420.x
Ghirardo A, Heller W, Fladung M, Schnitzler JP, Schroeder H (2012) Function of defensive volatiles in pedunculate oak (Quercus robur) is tricked by the moth Tortrix Viridana. Plant Cell Environ 35:2192–2207. https://doi.org/10.1111/j.1365-3040.2012.02545.x
Glinwood R, Ninkovic V, Pettersson J (2011) Chemical interaction between undamaged plants – effects on herbivores and natural enemies. Phytochemistry 72:1683–1689. https://doi.org/10.1016/j.phytochem.2011.02.010
Goulas Y, Cerovic ZG, Cartelat A, Moya I (2004) Dualex: a new instrument for field measurements of epidermal ultraviolet absorbance by chlorophyll fluorescence. Appl Opt 43:4488–4496
Gripenberg S, Salminen J-P, Roslin T (2007) A tree in the eyes of a moth? Temporal variation in oak leaf quality and leaf-miner perform. Oikos 116:592–600. https://doi.org/10.1111/j.2007.0030-1299.15415.x
Gripenberg S, Mayhew PJ, Parnell M, Roslin T (2010) A meta-analysis of preference–performance relationships in phytophagous insects. Ecol Lett 13:383–393
Haukioja E, Hanhimäki S (1985) Rapid wound-induced resistance in white birch (Betula pubescens) foliage to the geometrid Epirrita autumnata: a comparison of trees and moths within and outside the outbreak range of the moth. Oecologia 65:223–228
Heil M (2004) Direct defense or ecological costs: responses of herbivorous beetles to volatiles released by wild lima bean (Phaseolus lunatus). J Chem Ecol 30:1289–1295
Hernández I, Alegre L, Munné-Bosch S (2011) Plant aging and excess light enhance flavan-3-ol content in cistus clusii. J Plant Physiol 168:96–102
Hoballah ME, Köllner TG, Degenhardt J, Turlings TCJ (2004) Costs of induced volatile production in maize. Oikos 105:168–180
Jactel H, Birgersson G, Andersson S, Schlyter F (2011) Non-host volatiles mediate associational resistance to the pine processionary moth. Oecologia 166:703–711. https://doi.org/10.1007/s00442-011-1918-z
Janssen A, Sabelis MW, Bruin J (2002) Evolution of herbivore-induced plant volatiles. Oikos 97:134–138
Kallenbach M, Oh Y, Eilers E, Veit D, Baldwin IT, Schuman MC (2014) A robust, simple, high-throughput technique for time‐resolved plant volatile analysis in field experiments. Plant J 78:1060–1072
Karban R (2011) The ecology and evolution of induced resistance against herbivores. Functional Ecology 25:339–347
Karban R (2021) Plant communication. Annu Rev Ecol Evol S 52:1–24
Karban R, Baldwin IT (2007) Induced responses to herbivory. University of Chicago Press
Karban R, Takabayashi J (2018) Chewing and other cues induce grass spines that protect meristems. Arthropod-Plant Interact 13:541–550. https://doi.org/10.1007/s11829-018-9666-1
Karban R, Baldwin IT, Baxter KJ, Laue G, Felton GW (2000) Communication between plants: induced resistance in wild tobacco plants following clipping of neighboring sagebrush. Oecologia 125:66–71. https://doi.org/10.1007/PL00008892
Keefover-Ring K, Trowbridge A, Mason CJ, Raffa KF (2016) Rapid induction of multiple terpenoid groups by Ponderosa pine in response to bark beetle-associated fungi. J Chem Ecol 42:1–12. https://doi.org/10.1007/s10886-015-0659-6
Kessler A, Halitschke R, Diezel C, Baldwin IT (2006) Priming of plant defense responses in nature by airborne signaling between Artemisia tridentata and Nicotiana attenuata. Oecologia 148:280–292 https://doi.org/10.1007/s00442-006-0365-8
Kondoh M, Williams IS (2001) Compensation behaviour by insect herbivores and natural enemies: its influence on community structure. Oikos 93:161–167
Kovács B, Tinya F, Ódor P (2017) Stand structural drivers of microclimate in mature temperate mixed forests. Agric for Meteorol 234:11–21
Kumar H (1992) Inhibition of ovipositional responses of Chilo partellus (Lepidoptera: Pyralidae) by the trichomes on the lower leaf surface of a maize cultivar. J Econ Entomol 85:1736–1739
Mallick S, Molleman F, Yguel B, Bailey R, Müller J, Jean F, Prinzing A (2023) Ectophagous folivores do not profit from rich resources on phylogenetically isolated trees. Oecologia 201:1–18
Martemyanov VV et al (2012) Rapid induced resistance of silver birch affects both innate immunity and performance of gypsy moths: the role of plant chemical defenses. Arthropod-Plant Interact 6:507–518. https://doi.org/10.1007/s11829-012-9202-7
Mertens D, Boege K, Kessler A, Koricheva J, Thaler JS, Whiteman NK, Poelman EH (2021) Predictability of biotic stress structures plant defence evolution. Trends Ecol Evol 36:444–456
Monson RK, Trowbridge AM, Lindroth RL, Lerdau MT (2022) Coordinated resource allocation to plant growth–defense trade-offs. New Phytol 233:1051–1066. https://doi.org/10.1111/nph.17773
Molleman F, Walczak U, Melosik I, Baraniak E, Piosik Ł, Prinzing A (2022) What drives caterpillar guilds on a tree: enemy pressure, leaf or tree growth, genetic traits, or phylogenetic neighbourhood? Insects 13(4):367. https://doi.org/10.3390/insects13040367
Moreira X, Mooney KA, Rasmann S, Petry WK, Carrillo-Gavilán A, Zas R, Sampedro L (2014) Trade‐offs between constitutive and induced defences drive geographical and climatic clines in pine chemical defences. Ecol Lett 17:537–546
Mrazova A, Sam K (2019) Exogenous application of methyl jasmonate to Ficus hahliana attracts predators of insects along an altitudinal gradient in Papua New Guinea. J Trop Ecol 35:157–164. https://doi.org/10.1017/s0266467419000117
Mrazova A, Houska Tahadlova M, Řehová V, Sam K (2023) The specificity of induced chemical defence of two oak species affects differently arthropod herbivores and arthropod and bird predation. Arthropod-Plant Interact 17:141–155
Ninkovic V, Markovic D, Dahlin I (2016) Decoding neighbour volatiles in preparation for future competition and implications for tritrophic interactions perspectives in plant ecology. Evol Syst 23:11–17. https://doi.org/10.1016/j.ppees.2016.09.005
Nykänen H, Koricheva J (2004) Damage-induced changes in woody plants and their effects on insect herbivore performance: a meta-analysis. Oikos 104:247–268. https://doi.org/10.1111/j.0030-1299.2004.12768.x
Oksanen J, Kindt R, Legendre P, O’Hara B, Stevens MHH, Oksanen MJ, Suggests M (2007) The vegan package. Community ecology package 10:719
Pearse IS et al (2020) Generalising indirect defence and resistance of plants. Ecol Lett 23:1137–1152. https://doi.org/10.1111/ele.13512
Pellissier L, Moreira X, Danner H, Serrano M, Salamin N, van Dam NM, Rasmann S (2016) The simultaneous inducibility of phytochemicals related to plant direct and indirect defences against herbivores is stronger at low elevation. J Ecol 104:1116–1125. https://doi.org/10.1111/1365-2745.12580
Pihain M, Gerhold P, Ducousso A, Prinzing A (2019) Evolutionary response to coexistence with close relatives: increased resistance against specialist herbivores without cost for climatic-stress resistance. Ecol Lett 22:1285–1296
Price PW, Bouton CE, Gross P, McPheron BA, Thompson JN, Weis AE (1980) Interactions among three trophic levels: influence of plants on interactions between insect herbivores and natural enemies. Annu Rev Ecol Syst 11:41–65
R_Core_Team (2023) R: a language and environment for statistical computing, 4.3 edn. R Foundation for Statistical Computing, Vienna, Austria
Renwick JAA, Chew FS (1994) Oviposition behavior in lepidoptera annual. Rev Entomol 39:377–400. https://doi.org/10.1146/annurev.en.39.010194.002113
Rieske L, Dillaway D (2008) Response of two oak species to extensive defoliation: tree growth and vigor, phytochemistry, and herbivore suitability. Forest Ecol Manag 256:121–128
Robert CAM et al (2013) Genetically engineered maize plants reveal distinct costs and benefits of constitutive volatile emissions in the field. Plant Biotechnol J 11:628–639
Rosenkranz M, Chen Y, Zhu P, Vlot AC (2021) Volatile terpenes – mediators of plant-to-plant communication. Plant J 108:617–631. https://doi.org/10.1111/tpj.15453
Roslin T, Gripenberg S, Salminen JP, Karonen M, O’Hara B, Pihlaja R, Pulkkinen K P (2006) Seeing the trees for the leaves–oaks as mosaics for a host-specific moth. Oikos 113:106–120
Salminen JP, Karonen M (2011) Chemical ecology of tannins and other phenolics: we need a change in approach. Funct Ecol 25:325–338. https://doi.org/10.1111/j.1365-2435.2010.01826.x
Šamec D, Karalija E, Šola I, Vujčić Bok V, Salopek-Sondi B (2021) The role of polyphenols in abiotic stress response: the influence of molecular structure. Plants 10:118
Semiz G, Blande JD, Heijari J, Işık K, Niinemets Ü, Holopainen JK (2012) Manipulation of VOC emissions with methyl jasmonate and carrageenan in the evergreen conifer Pinus sylvestris and evergreen broadleaf Quercus ilex. Plant Biol 14:57–65. https://doi.org/10.1111/j.1438-8677.2011.00485.x
Simard SW (2018) Mycorrhizal Networks Facilitate Tree Communication, Learning, and memory. In: Baluska F, Gagliano M, Witzany G (eds) Memory and learning in plants. Springer International Publishing, Cham, pp 191–213. https://doi.org/10.1007/978-3-319-75596-0_10
Sokół-Łętowska A, Kucharska AZ, Hodun G, Gołba M (2020) Chemical composition of 21 cultivars of sour cherry (Prunus cerasus) fruit cultivated in Poland. Molecules 25:4587
Soler R, Harvey JA, Bezemer TM (2007) Foraging efficiency of a parasitoid of a leaf herbivore is influenced by root herbivory on neighbouring plants. Funct Ecol 21:969–974
Southwood T (1961) The number of species of insect associated with various trees. J Anim Ecol 1–8
Stamp N (2003) Out of the quagmire of plant defense hypotheses. Q Rev Biol 78:23–55
Šušić N, Milić Komić S, Živanović B, Jelušić A, Marković S, Sedlarevic Zoric A, Veljovic Jovanovic S (2023) Acclimation of pedunculate oak seedlings to different light conditions in the first months after germination. In: 30th International Conference Ecological Truth And Environmental Research–EcoTER’23. University of Belgrade, Technical Faculty in Bor, pp 135–140
Tollrian R, Harvell CD (1999) The ecology and evolution of inducible defenses. Princeton University Press
Tscharntke T, Thiessen S, Dolch R, Boland W (2001) Herbivory, induced resistance, and interplant signal transfer in Alnus glutinosa. Biochem Syst Ecol 29:1025–1047. https://doi.org/10.1016/S0305-1978(01)00048-5
Valdés-Correcher E et al (2021) Search for top‐down and bottom‐up drivers of latitudinal trends in insect herbivory in oak trees in Europe. Global Ecol Biogeogr 30:651–665
Vialatte A et al (2010) Phylogenetic isolation of host trees affects assembly of local Heteroptera communities. Proc Royal Soc B: Biol Sci 277:2227–2236. https://doi.org/10.1098/rspb.2010.0365
Visakorpi K, Riutta T, Malhi Y, Salminen JP, Salinas N, Gripenberg S (2020) Changes in oak (Quercus robur) photosynthesis after winter moth (Operophtera brumata) herbivory are not explained by changes in chemical or structural leaf traits. PLoS ONE 15:e0228157. https://doi.org/10.1371/journal.pone.0228157
Volf M et al (2021) Branch-localized induction promotes efficacy of volatile defences and herbivore predation in trees. J Chem Ecol 47:99–111
Volf M et al (2022) A mosaic of induced and non-induced branches promotes variation in leaf traits, predation and insect herbivore assemblages in canopy trees. Ecol Lett 25: 729–739
Wäckers FL, van Rijn PCJ, Bruin J (2005) Plant-provided food for carnivorous insects: a protective mutualism and its applications. Cambridge University Press
Walter DE (1996) Living on leaves: mites, tomenta, and leaf domatia. Annu Rev Entomoll 41:101–114
War AR, Hussain B, Sharma HC (2013) Induced resistance in groundnut by jasmonic acid and salicylic acid through alteration of trichome density and oviposition by Helicoverpa armigera (Lepidoptera: Noctuidae). AoB Plants 5:plt053
Yguel B et al (2011) Phytophagy on phylogenetically isolated trees: why hosts should escape their relatives. Ecol Lett 14:1117–1124. https://doi.org/10.1111/j.1461-0248.2011.01680.x
Zhang Z-Z, Che X-N, Pan Q-H, Li X-X, Duan C-Q (2013) Transcriptional activation of flavan-3-ols biosynthesis in grape berries by UV irradiation depending on developmental stage. Plant Sci 208:64–74
Acknowledgements
Fieldwork was carried out with the kind permission of the Forestry Experimental Station in Murowana Goślina (Poznań University of Life Sciences). We are grateful to Fatmanur Selvi for her help in the field. C: N analysis was performed at the Institute of Dendrology of the Polish Academy of Sciences in Kórnik.
Funding
The research was funded by grant No. 2018/29/B/NZ8/00112 to Freerk Molleman from the National Science Centre (NCN, Poland), and Martin Volf acknowledges the Grant Agency of the Czech Republic (grant number 19-28126X).
Author information
Authors and Affiliations
Contributions
Freerk Molleman developed the initial concept of the study, led the fieldwork, analysed the data and wrote the first draft of the manuscript, Manidip Mandal participated in the planning and fieldwork, performed induction, collected the volatile samples, and participated in the VOC analysis, Anna Sokół-Łętowska performed phenolics analyses, Urszula Walczak participated in developing the concept, planning of the study, and in fieldwork, Martin Volf participated in developing the concepts and planning of the study, Soumen Mallick participated in fieldwork and collection of volatile samples, Martin Moos and Petr Vodrážka advised on volatile sample collection and performed volatile measurements, Andreas Prinzing participated in developing the concept, Priscila Mezzomo participated in developing the concept, provided training in VOC sample collection and interpretation of VOC measurements. All authors contributed to the manuscript writing.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Molleman, F., Mandal, M., Sokół-Łętowska, A. et al. Simulated Herbivory Affects the Volatile Emissions of Oak Saplings, while Neighbourhood Affects Flavan-3-ols Content of Their Leaves. J Chem Ecol 50, 250–261 (2024). https://doi.org/10.1007/s10886-024-01471-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-024-01471-4