Abstract
Headwater streams drain large proportions of landscapes and represent a large proportion of stream habitat. Understanding their ecology requires more comparative data from the smallest streams. We describe dynamics of benthic organic material (BOM) and invertebrate assemblages in an order-2 Australian tropical stream and compare samples from orders-1 and 2 sites. Litterfall and, especially, stream discharge determined BOM standing stock. Most material was in the > 1 mm fraction, pools stored more than riffles, and order-1 sites more than order-2. BOM increased in the dry season but was depleted by wet-season spates. Retention was greater in years without major spates, when riffle-pool stream sections were largely ecologically isolated. Invertebrate richness was greater in riffles than pools and in order-2 than order-1 sites, with overlap in composition. Current velocity was the primary variable determining distributions in order-2 sites, with particulate organic matter (63–250 µm) the main variable in order-1 sites. However, explanatory relationships were weak because of unusually benign conditions during the sampling period. We need more globally comprehensive spatially explicit assessment of the ecosystem dynamics of headwater streams to predict effects of environmental change. Assessments over extended periods are necessary to capture the variability of even apparently predictable systems.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Headwater streams represent the smallest tributaries in the landscape, and include first- and second-order streams (Meyer et al., 2003), although this definition is not adhered to universally (Clarke et al., 2008). Headwater streams typically contribute a large proportion of river length and provide important ecosystem services such as transfer of organic material, nutrients and energy downstream and between terrestrial and aquatic ecosystems (Vannote et al., 1980; Meyer & Wallace, 2001; Meyer et al., 2003; Wipfli et al., 2007; De Nadai-Monoury et al., 2014; Boyero et al., 2016). They also support substantial biological diversity (Gomi et al., 2002; Meyer et al., 2007). Diversity of invertebrates in streams varies substantially geographically but invertebrate richness tends to be greatest in mid-order streams (Clarke et al., 2008).
Although algae in headwater forest streams may be more important in food webs than their apparent abundance suggests (McNeely et al., 2007; Schmidt et al., 2017), these food webs are substantially detritus-based, and understanding of detrital dynamics and their relationship to invertebrate assemblages is a major goal in the understanding of these ecosystems (e.g. Wallace & Webster, 1996; Cheshire et al., 2005; Landeiro et al., 2010; Chara-Serna et al., 2012; Graça et al., 2015). Breakdown of terrestrially derived leaf litter is caused by mechanical abrasion and microbial and invertebrate activity, which are affected by flow, temperature, litter quality, phylogenetic diversity of the litter, and stream chemistry (Boyero et al., 2016). In temperate zones, litter inputs and breakdown are very seasonal, whereas in the humid tropics, equable temperatures and continuous litter inputs (albeit with seasonal peaks—e.g. Benson & Pearson, 1993) typically promote year-round litter processing (Cheshire et al., 2005).
Fine particulate organic matter is abundant in streams, but its quality and quantity in low-order streams has been investigated less frequently than the dynamics of coarse particulate organic matter (Callisto & Graça, 2013). Most work on detrital processing and dynamics has been done in temperate zones, and there is less, but increasing, information available from the tropics, including studies from South America (e.g. Ferreira et al., 2012), Central America (Colón-Gaud et al., 2008), Africa (Moss, 2005), Asia (Li & Dudgeon, 2009), Australia (Bastian et al., 2007) and globally (Boyero et al., 2016).
The distribution of benthic organic matter relates both to inputs (Bärlocher, 1983) and retention, which varies with stream morphology and flow (Hoover et al., 2006). Huryn & Wallace (1988) reported that, in North Carolina streams, habitats of greater roughness (riffles) or lower current (pools) were both sites of deposition of organic material. However, studies on the dynamics of litter may examine only riffles (Pearson et al., 1989) or pools (De Nadai-Monoury et al., 2014) or not mention specific habitats (Lisboa et al., 2015). Nevertheless, reports on litter dynamics typically include reference to substratum and flow, or correlates such as turbulence (Graça et al., 2015). While riffles are more retentive of experimentally released litter than pools (Speaker et al., 1984; Quinn et al., 2007), standing stock of organic material may or may not reflect this finding (Scullion et al., 1982; Angradi, 1996; Kobayashi & Kagaya, 2002; Nakajima et al., 2006), although comparisons of standing stock between pools and riffles are rather scarce.
Benthic detritus in tropical headwater streams is subject to similar variability of the flow regime as elsewhere, with retention mechanisms often more important than input in determining organic matter dynamics (Pearson et al., 1989; Lisboa et al., 2015). For example, floods redistributed litter substantially in an Australian tropical stream, but only temporarily, and responses were related more to individual floods than to overall discharge (Wootton et al., 2019), much as is the case in the temperate zone (Wallace et al., 1995). In coastal Ecuadorian streams, variation in benthic organic stocks were closely linked to seasonal rainfall (Molinero, 2019), whereas in order-1 Puerto Rican streams variation was largely explained not by physical conditions but by abundance of large crustaceans (Crowl et al., 2002).
Invertebrate assemblages in small forest streams globally are strongly influenced by physical variables (current velocity, substratum, etc.) and the availability of detrital resources (Cummins et al., 1973; Hawkins & Sedell, 1981; Chara et al., 2007; Walther & Whiles, 2011; Straka et al., 2012; Pearson, 2014; Pearson et al., 2017; Sarremejane et al., 2018). Many invertebrates use detritus as a source of food in the form of coarse material (leaves, etc.), or finer particular matter accumulating on the coarse material (Richardson, 1992; Dudgeon & Wu, 1999) and elsewhere.
There have been some comparisons between riffles and pool invertebrate assemblages because of their contrasting morphology and hydraulics, and their predominance in many stream systems (e.g. Logan & Brooker, 1983; Brown & Brussock, 1991), although most studies are undertaken in riffles, partly because of their ease of access and sampling. Contrasts in the fauna of riffles and pools vary among studies, probably as a result of differences in morphology, hydrology or hydraulics. Logan & Brooker (1983) reported greater invertebrate densities in riffles than pools, and similar numbers of taxa, in upland streams in North America and UK, with each habitat characterised by particular taxa. Similar results have been reported elsewhere—for example, Arkansas (Brown & Brussock, 1991), Costa Rica (Ramirez & Pringle, 1998), Hungary (Schmera & Erös, 2011), Wales (Scullion et al., 1982). Conversely, in lowland Texas streams, pools contained higher densities and number of taxa (McCulloch, 1986) whereas, in Uganda, pools and riffles had similar fauna (Tumwesigye et al., 2000).
Short-term changes in flow can have substantial effects on density and diversity of invertebrate assemblages (McElravy et al., 1989; Brown & Brussock, 1991; Ramirez and Pringle, 1998; Pearson, 2014). In the southern USA, invertebrate density was lower in pools than riffles but following high discharge there was no difference between them (Payne & Miller, 1991), while in Victoria, seasonal changes in assemblages occurred only in erosional habitats, as a result of changes in only a few species (Barmuta, 1989). Inter-annual differences may also be substantial: for example, in California, invertebrate richness was higher in riffles than pools in low-discharge years, but similar during a year of average discharge, with the annual peak discharge and the period of high flow being important factors. Research on Finnish streams suggests that around 6 years’ data are required to adequately understand inter-annual variability (Huttunen et al., 2018), with major flow events being the best predictors of invertebrate assemblages (Sarremejane et al., 2018).
Invertebrate assemblages typically change along streams from their source. In a series of sites from orders 1–7 in the USA, richness was greatest in mid-order reaches (Grubaugh et al., 1996). In Panama, assemblages were more heterogeneous in order-1 than orders-2 or 3 riffles, because the greater substratum heterogeneity in the lower order (Boyero & Bailey, 2001), and in Finland, riffles were more variable in second-order than in third-order sections (Heino et al., 2004). In the Australian Wet Tropics (hereafter, “Wet Tropics”), order-1 and 2 streams represent about 75% of total stream length and area of landscape drained, and about 26% of stream habitat area (Januchowski-Hartley et al., 2011). Therefore, they are major components of landscapes and the regional stream network, much as elsewhere (Meyer et al., 2003; Wipfli et al., 2007). Litterfall is predictable and occurs through the year, with a major peak in spring (Spain, 1984; Hopkins & Graham, 1989; Benson & Pearson, 1993), prior to the period of maximum discharge (summer wet season) (Pearson, 2014), unlike the situation in typical northern-hemisphere systems. Discharge and litterfall can be greatly boosted by unpredictable cyclones (hurricanes) any time during the summer (Benson & Pearson, 1993). Leaf-litter packs of varying density and leaf age cover much of the stream bed in the late dry season, but moderate floods remove or redistribute a substantial proportion of litter, some which may be exported without undergoing substantial breakdown (Wootton et al., 2019).
As elsewhere, allochthonous litter is a major component of the food web in Wet Tropics streams (Cheshire et al., 2005), and invertebrates show strong association with benthic organic matter (Hearnden & Pearson, 1991; Pearson et al., 2017). Invertebrate richness in Wet Tropics streams is high (Pearson et al., 1989; Vinson & Hawkins, 2004; Pearson & Boyero, 2009) and tends to increase in a downstream direction (Pearson et al., 2017). In Birthday Creek (the focus of the present study), invertebrate abundance and richness were higher in order-3 riffles than in pools (Cheshire et al., 2005). In Yuccabine Creek, about 100 km north of Birthday Creek, riffle assemblages varied with season, but showed strong resilience to flood and drought (Pearson, 2014), as they did in experiments in several Wet Tropics streams (Rosser & Pearson, 1995, 2018).
We aimed to understand the seasonal dynamics of invertebrates and organic material in Wet Tropics order-1 and 2 streams. We hypothesised that (1) order-1 streams, particularly pools, would store the largest proportion of organic matter; (2) standing stock would show a strong correlation with litterfall only during periods of low streamflow; (3) invertebrate assemblages would be strongly influenced by abundance of detritus; and (4) detritus and invertebrate assemblages would respond to stream size and the flow regime, with the wet season acting as a re-set mechanism on an annual basis.
Methods
Study site
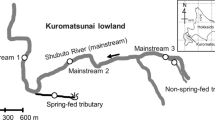
The study was conducted in the headwaters of Birthday Creek (stream orders 1 and 2) in largely undisturbed, forested granitic uplands in north-eastern Australia (~ 19.0°S, 146.2°E, 865 m a.s.l; Fig. 1). The study area has an average annual rainfall of 2585 mm, 70% of which falls in the summer wet season (December to March; Fig. 2). During this study, four cyclones approached the coast near Birthday Creek, bringing gale-force winds: Charlie, causing no change in rainfall; Aivu, causing a modest increase in rainfall; and Ivor and Joy, both producing very heavy rainfall (Fig. 2). During the study period, annual rainfall ranged from 1736 to 3367 mm (Australian Bureau of Meteorology data); air temperature from 5 to 26°C, and stream temperature from 11.5 to 19.5°C (measured with maximum–minimum thermometers); pH from 5.9 to 7.0, conductivity from 19 to 60 µScm−1, and dissolved oxygen from 7.3 to 9.8 mgl−1 (~ 85% saturation) with no discernible diurnal fluctuation (measured with YSI instruments). Soils of the catchment are acidic (mean pH 3.85–4.47) and of low fertility (Congdon & Herbohn, 1993). Nutrient concentrations in the stream are typically very low (Connolly & Pearson, 2013). Vegetation within the catchment is primarily simple notophyll vine forest (Tracey, 1982), with complex notophyll vine forest close to the stream (Webb, 1978).
Top panel: Birthday Creek climate showing mean monthly minimum and maximum temperatures (top lines), mean monthly rainfall (lower line) and monthly rainfall during study period (bars). Cyclones passing nearby are identified by asterisks: C, Charlie; A, Aivu; I, Ivor; J, Joy. Other panels from top: total riparian litterfall recorded at the order-2 site; and monthly AFDM ± SE of each fraction of benthic organic material in order-2 riffle and pool samples. Differences between years, ***P < 0.001; similar letters, a–d, indicate no difference in AFDM among years within each fraction (ANOVA followed by Tukey tests, α = 0.05; see Table S1). Absence of data indicates no sample collected
The main study reach was located upstream of a waterfall and had a total length (orders-1 and 2 sections) of 740 m with a gradient of 10.6% and catchment area of 35 ha (Fig. 1), comprising 44% pool, 33% riffle, 14% run and small proportions of race, cascade and waterfall. The substratum comprised about 30% sand and gravel, 25% cobbles and 40% boulders or bedrock, with occasional fallen branches and exposed roots. In the late dry season, about 30% of it was covered by leaf litter.
The order-2 study pool was 28 m long, had mean width of 4.1 m, mean depth of 0.24 m and indiscernible current except in spates. The order-2 study riffle was 37.5 m in length, with mean width of 4.1 m, mean depth of 0.10 m and mean non-spate current velocity of about 0.10 ms−1. Three order-1 perennial tributaries were sampled, each upstream of the order-2 site. We termed running-water habitat in these streams as “riffle”, even though in most circumstances they were not turbulent. Mean length of riffle and pool habitats was approximately 5.0 m, width 1.5 m, and depth < 0.15 m. Together, the selected riffles and pools (orders 1 and 2 combined) represented 16.9% and 18.1% of total habitat length in the study reach.
Sampling
We measured vertical litter inputs from June 1987 to May 1990 using 15 litter traps (0.25 m2 collection area, 0.5 mm mesh), placed randomly in a 28-m section of the order-2 reach, within 7 m of the stream centre; we also measured lateral litter input, but do not report it here as it was of minor importance (see Benson & Pearson, 1993). In the laboratory, litter samples were sorted into leaf, wood, reproductive material and trash, then oven dried to constant weight at 85°C and weighed.
We collected benthic samples (riffle, 5–10; pool, 5) of organic material and invertebrates from the order-2 section approximately monthly, April 1987 to September 1988, and then organic material only from May 89 (pool) and August 1989 (riffle) until August 1991. We collected three samples from each riffle and pool in each of the three order-1 streams before and after the wet season (October 1987, June 1988). Sampling was undertaken using a Surber sampler (Surber, 1937) of 0.25 × 0.25 m basal area, with a 63-µm-mesh collecting net and 250-µm mesh on the upstream and side faces of the sampler. The substratum was vigorously agitated in the sampler and washed into the collecting net until no more organic matter was visible within the sample area. The contents were preserved in 70% ethanol. Current, depth and substratum particle size were measured concurrently using a timed floating object, rule and templet, respectively.
In the laboratory, benthic samples were elutriated to separate organic and inorganic material and filtered through stacked sieves of 1 mm, 250 µm and 63 µm mesh size. Here we refer to benthic organic material as BOM, the fraction > 1 mm as coarse particulate organic matter (CPOM), the 250-µm to 1-mm fraction as fine particulate organic matter (FPOM) and the 63–< 250-µm fraction as very fine particulate organic matter (VFPOM) (Cummins, 1974). Invertebrates were removed from each fraction and the remaining organic matter was dried at 85°C for 24 h on pre-weighed filter paper (Whatman number 41), transferred to crucibles and ashed in a muffle furnace at 600°C for 4 h, and then reweighed to determine ash-free dry mass (AFDM). Invertebrates were identified to the lowest taxonomic level possible, and recognisably different taxa without positive identification were mostly assigned as operational taxonomic units. The Chironomidae was not separated to species or OTU level. Invertebrates were grouped by family or higher taxon for comparison among stream orders 1–3 (with order-3 data from published information).
Data analysis
We transformed benthic sample data to log (x + 1). For analysis of litterfall we combined data into four seasonal groups—Wet, Wet–Dry transition, Dry, and Dry–Wet transition, representing December–February, March–May, June–August and September–November, respectively; for comparison with litterfall we combined BOM data by the same groups and by years. We used correlation analysis to identify relationships between rainfall, litterfall and BOM, and ANOVA or ANCOVA followed by Tukey tests where appropriate to compare data among habitats and stream orders, using Statistix 10 (Analytical Software, Tallahassee, Florida, USA). We estimated invertebrate species richness taking into account sample sizes using the Chao2 rarefaction estimate in PC-ORD (MjM Software, Gleneden Beach, Oregon, USA). We used linear regression analysis to investigate relationships between BOM and rainfall, which was used as a surrogate for flow as the two have a very close relationship (Clayton & Pearson, 2016). We used PERMANOVA in Primer (Primer-E Software, Plymouth, UK) to compare invertebrate assemblages and functional groups among samples, with invertebrates assigned to functional groups following Cheshire et al. (2005), and multidimensional scaling (MDS) in Primer to visualise relationships among sites. We used the RELATE function in Primer to test whether cyclicity was evident in the monthly samples in the order-2 samples.
We used distance-based linear modelling (DistLM, in Primer) to determine the relationship between invertebrate assemblages and potential environmental drivers, using forward selection and the Akaike information criterion adjusted for small sample sizes (AICc). DistLM seeks the most significant relationships between a similarity matrix and environmental variables by modelling the matrix against the most influential variable, taking the residuals of that relationship, modelling the next most influential variable, and so on. Initial environmental variables used were substratum size; AFDM of CPOM, FPOM and VFPOM; 30-days antecedent rainfall; water depth; current; pH; conductivity; dissolved oxygen; and water temperature. Mean temperature correlated strongly with maximum and minimum temperature (r = 0.954 and 0.977, respectively) so only the mean was used, as recommended (Anderson et al., 2008). Depth correlated negatively with current velocity for all species so was removed, as current was more important in preliminary analysis. Dissolved oxygen was removed because it never fell below 80% saturation.
Results
Rainfall, litter inputs and benthic organic material
Monthly rainfall was below average for the first 2 years of the study (1736, 1920 mm, cf. 49-year mean = 2585 mm), about average for the third and fourth year (2697, 2601 mm) and above average for the final year (3367 mm) (Fig. 2). More importantly, peak monthly rainfall was very much above average in the final 2 years, although daily maxima were within the one-in-one-year return interval in all 5 years (Fig. S1). Litterfall was greatest in the late dry season, except for extra peaks following tropical cyclones (Fig. 2), with major contrasts between Dry season and other samples for total litterfall and leaf fall, between the Wet–Dry transition and other samples, and minor contrasts for reproductive material and trash (Fig. S2). Overall, litter comprised 73.1% leaf, 15.3% small wood, 2.5% reproductive and 9.1% trash. There was no correlation between recent rainfall and litterfall (DF = 36, r < 0.200, P > 0.05) except with small wood (r = 0.460, P < 0.01).
Benthic organic material in the order-2 sites indicated some seasonal pattern in the riffle samples over the first 18 months of sampling, with a peak during the wet season, but this was less clear subsequently, and was not evident in the pool samples; however, there were substantial differences in BOM mass among years for all fractions, with contrasts between earlier and later years and between seasons—mainly between the Dry season and the rest for CPOM in riffle and pool samples, and FPOM in riffles, but not for other fractions (Fig. 2; Table S1). The largest proportion of organic material was in the CPOM fraction, with the CPOM, VFPOM and FPOM fractions roughly in the ratios of 25:4:1 in the riffles (F2,90 = 24.69, P < 0.001) and 20:5:1 in the pools (F2,93 = 22.48, P < 0.001). The only correlations between litterfall and BOM were between reproductive material and FPOM in riffle samples (DF = 11, r = 0.615, P < 0.05) and between reproductive material and CPOM and FPOM in the pool samples (DF = 13, r = 0.610, P < 0.05 and r = 0.848, P < 0.001, respectively).
In the comparison between streams for October and June samples, ANCOVA (with months as the covariate) showed that order-1 pool samples contained more CPOM (approximately threefold) than did order-1 riffle or any order-2 samples, and order-1 samples mostly contained much more FPOM and VFPOM than order-2 samples, in both pools and riffles (Fig. 3, Table S2).
High wet-season flows strongly influenced the subsequent BOM mass, with regression analysis clearly pointing to the three highest flows as outliers, and little other relationship with flow, except when the three highest flows were removed. In the absence of these three data points, CPOM and VFPOM, but not FPOM, showed positive relationships with flow in riffles, although there was substantial variability among samples (Fig. 4, Table S3). For pools there was no such relationship, with or without outliers (Table S3). In the order-1 streams, only CPOM in riffles was affected by antecedent rainfall, but the results are not directly comparable with those from order-2 sites because the order-1 streams were only sampled in the dry season (Table S3).
Invertebrate assemblages
The dry-season invertebrate samples had greater abundances in riffles than in pools in order-1 but not order-2 streams, while similar habitats did not differ in invertebrate abundance between stream orders (Table 1). The order-2 invertebrate samples produced 130 taxa over 18 months of sampling—124 from riffles (89 samples) and 75 from pools (55 samples); the order-1 samples produced 69 taxa—64 from riffles (18 samples) and 44 taxa from pools (18 samples) (Table 1, Table S6). Chao2 estimates of richness via rarefaction were higher, but followed the same sequence of richness, as did the numbers of taxa per sample (Table 1). However, only 17 taxa contributed more than 1% of the fauna in any stream order/habitat combination; the only taxa contributing 5% or more were the Chironomidae in all combinations; Oligochaeta, Lectrides sp. (Leptoceridae) and Ceratopogonidae in order-1 pools; and Simuliidae in order-2 riffles (Table 1).
MDS and PERMANOVA of invertebrate samples from stream orders 1 and 2 separated order-1 from order-2 samples in riffles and pools in both October and June, mainly on axis 2; the order-1 samples showed a very broad spread on axis 1 and there was no difference among the order-1 samples in either habitat (Fig. 5, Table S4). In a similar analysis using functional-group data, there were only minor differences among samples, with riffle samples in October differing between one order-1 stream and the order-2 stream, and riffle samples for June differing between two of the order-1 streams and the order-2 stream (Table S4). For the order-2 dataset (11 samples, 18 months), riffle and pool samples were almost completely separated (Fig. 5b). Between-month trajectories were similar along axis 1 during the first three months, but deviated subsequently; the RELATE procedure identified moderate cyclicity among samples (P < 0.05, Table S4).
Results of multidimensional scaling of invertebrate assemblages: a centroids for order-1 and 2 sites (1a, 1b, 1c and 2, respectively) riffles (filled triangle) and pools (filled circle); October samples indicated by open symbols, June samples by closed symbols. Order-1 samples did not differ from each other, but differed from order-2 samples (PERMANOVA, P < 0.01, see Table S4). b Order-2 pool and riffle samples, showing moderate cyclicity over 18 months (RELATE, P < 0.05, Table S4)
In the order-2 stream samples taken over 18 months, abundances of invertebrates in riffle and pool samples differed in the wet season, whereas in the dry season they did not, and abundances in the riffle did not differ between wet and dry seasons, whereas in the pool abundance was greater in the dry season (Fig. 6, Table S5; Fig. S3 and S4). Linear modelling (DistLM) produced models relating invertebrate assemblages to environmental variables, but explained only small proportions of the variation in the data (Table 2). Most prominent variables were VFPOM, current, CPOM, temperature and substratum size. Correlations between occurrence of individual taxa and individual variables were not extensive: correlations with r2 > 0.15 in order-1 streams included two with current (both positive), three with temperature (two negative), one with FPOM (positive) and six with VFPOM (all positive); in order-2 samples, 11 with current (one negative) and four with VFPOM (two negative) (Table 3). In order-1 streams, the strongest relationships were between VFPOM and Leptoceridae, including Triplectides sp., and Nousia sp. 6; and between FPOM and Chironomidae. In order-2 samples, the strongest relationships were between current velocity and Nousia sp. A and Hydrochidae.
Discussion
Dynamics of organic material
Litterfall followed the expected pattern of continuing inputs through the year, peaking in the late dry season, with further peaks resulting from cyclones (Spain, 1984; Hopkins & Graham, 1989). During drier years the BOM standing stock in Birthday Creek was higher than in orders 1–3 streams worldwide, but similar during wet years (Stewart & Davies, 1990; Boulton & Lake, 1992), and similar to that in Yuccabine Creek, an order-3 site some 100 km north of Birthday Creek (Pearson et al., 1989). A general pattern of reduced density following the wet season and a gradual build-up through the dry season was apparent in the first year of sampling in riffles, but not in pools, as reported for an order-3 section of Birthday Creek (Wootton et al., 2019). Standing stock was highest in order-1 streams, which accords with other studies over a greater gradient (e.g. Naimann & Sedell, 1980; Benfield et al., 2000).
Overall, the only component of litterfall with which BOM standing stock correlated was reproductive material, which was a minor component of the litter. Conversely, stream flow, the predominant physical variable influencing stream ecology (Resh et al., 1988; Rosser & Pearson, 2018; Sarremejane et al., 2018), positively affected CPOM and VFPOM standing stock in riffles except when the largest flows were included, possibly because of variable redistribution of material under different flows. Retention of leaf litter, dependent on flow regime and microstructure of the stream bed (e.g. Minshall et al., 1983; Webster et al., 1994; Angradi, 1996; Hoover et al., 2006; Lisboa et al., 2015; Molinero, 2019), clearly determines the quantities of organic material in Birthday Creek as it did in Yuccabine Creek (Pearson et al., 1989). There was little indication of such patterns in the pool samples across the whole sampling period, and even the increased flood flows of the later years were insufficient to remove BOM completely from the pools. In contrast, much larger floods, such as a one-in-five-year event in 1994, cleaned virtually all organic material out of both riffles and pools (unpublished data). The wet season thus may regularly redistribute BOM in riffles, and occasionally act as a re-set mechanism, substantially removing the stores of organic matter from the headwaters.
Annual export of CPOM from headwater streams may be poorly correlated with annual discharge, with most export being fine and dissolved organic matter (Wallace et al., 1995). The majority of transport of organic material in Birthday Creek occurred over short periods in the wet season, when spates affected all size fractions of organic matter, while dry-season flows transported the finer fractions more readily than coarser fractions (Benson, 1999). Pools trapped organic material (particularly FPOM and VFPOM) and invertebrates exported from riffles during base flows and smaller floods, and the accumulation of fine material in the riffle appeared to be flow-limited. Therefore, it appears that, in the dry season, ecological processes are largely restricted to short stretches of the stream because of the inhibitory effect of pools on movement of fauna and organic matter, as suggested for other systems (Benson & Pearson, 1987; Bunn & Hughes, 1997; Brooks et al., 2017; De Nadai-Monoury et al., 2014).
Invertebrate assemblage composition and dynamics
The benthic fauna of Birthday Creek and its changes with stream order were similar to those of other sites in the Wet Tropics and elsewhere. In comparable riffle samples, order-3 streams had greater family richness (39 families in Birthday Creek, 38 in Yuccabine Creek) than our order-2 (20 families) or order-1 (19 families) samples from Birthday Creek (Cheshire et al., 2005; Pearson, 2014). Collector/filterers (e.g. Cheumatopsyche spp.) were more abundant in larger streams and some shredders (e.g. Lectrides spp.) were more abundant in lower-order streams, reflecting a gradual change in composition with stream size through the headwaters (Rosser & Pearson, 1995; Grubaugh et al., 1996; Waite et al., 2000), although changes in richness were not universal in the Wet Tropics (Pearson et al., 2017).
In our samples, distance-based linear modelling provided only limited explanations for invertebrate occurrence, with models for order-1 and order-2 riffles and pools each explaining less than 12% of the variation in the data—rather less than might be expected, as invertebrate assemblages are typically correlated with organic matter standing stock and with stream discharge (Ramirez & Pringle, 1998; Chara et al., 2007; Walther & Whiles, 2011; Callisto & Graça, 2013; Pearson, 2014; Sarremejane et al., 2018). For Wet Tropics streams generally, over 40% of variation was explained by habitat variables, including current velocity, substratum composition and amount of BOM (Pearson et al., 2017). It is probable that the unusually benign conditions during the sampling period tempered the influence of the environmental variables measured. The absence of major floods meant that any influence of increased current velocities following rainfall were very short-lived. For example, Wootton et al. (2019) found that heavy rainfall and associated spate caused by a one-in-one-year event led to redistribution and loss of some, but not all, leaf litter in Birthday Creek, and recovery of shredders from the disturbance was rapid. Additionally, the mostly consistent availability of BOM in the Wet Tropics, resulting from year-round inputs, may reduce seasonality in assemblages compared with more seasonal settings (Campbell & Fuchshuber, 1994), although life cycles of many species are influenced by seasonality in temperature or flow regime (Nolen & Pearson, 1992; Pearson, 2014). The continuous litterfall and leaf breakdown probably facilitates recovery of the stream fauna after wet-season spates (Benson & Pearson, 1987), with even fresh green litter being processed by shredders, providing a source of finer material to other species (Benson & Pearson, 1988; Pearson et al., 1989; Nolen & Pearson, 1993). Therefore, despite occasional loss of BOM, the resilience of shredders to moderate flood, their ability to process green leaves, and their continued presence across seasons contribute to seasonal consistency in detrital-based food webs in the study stream except, perhaps, after extreme floods (Cheshire et al., 2005; Wootton et al., 2019).
While most taxa were recorded in both riffles and pools, they showed distinct habitat preferences, with contrasts in current velocity the primary determining variable of invertebrate distributions in order-2 samples, and VFPOM being most important in order-1. In other locations, comparisons between riffles and pools are inconsistent (Logan & Brooker, 1983; McCulloch, 1986; Ramirez & Pringle, 1998; Herbst et al., 2018), although they may be more similar in order-1 and 2 sections than in higher-order streams (Logan & Brooker, 1983; Brown & Brussock, 1991).
Conclusion
Order-1 stream sections stored more organic material than order-2 sections and the bulk of stored BOM was in the coarse fraction, with pools accumulating more material than riffles, supporting our first hypothesis. However, the magnitude of wet-season spates determines whether organic matter is redistributed within, or exported from, a stream section, only partly supporting our second hypothesis. Therefore, despite high predictability of litterfall in the Wet Tropics, we show that long-term datasets are required to fully understand the variable dynamics of organic material resulting from unpredictability in the extent and duration of floods.
Proportional composition of the fauna in the headwaters of Birthday Creek changed with distance downstream, reflecting changes in biophysical characteristics, especially flow and transport/retention of organic matter. However, relationships between environmental variables and invertebrate assemblages were weak, contrary to our third and fourth hypotheses, because of benign conditions during the study period, in contrast with other studies in the region that included periods of drought and flood (Pearson, 2014) and a broad range of sites (Pearson et al., 2017).
Given that low-order headwater streams contribute the largest proportion of stream length, drain such a large proportion of the landscape and represent a large proportion of stream habitat (Januchowski-Hartley et al., 2011), they clearly contribute substantially to stream ecosystems, both through organic material inputs and processing, and invertebrate assemblages. We need more globally comprehensive assessment of ecosystem dynamics of these systems across multiple years if we are to predict effects of change to land use or climate. The longitudinal changes in headwater-stream invertebrate assemblages make it evident that such assessments need to be spatially explicit.
References
Anderson, M. J., R. N. Gorley & K. R. Clarke, 2008. PERMANOVA for PRIMER: Guide to Software and Statistical Methods. PRIMER-E Ltd., Plymouth.
Angradi, T. R., 1996. Inter-habitat variation in benthic community structure, function and organic matter storage in 3 Appalachian headwater streams. Journal of the North American Benthological Society 15: 42–63.
Bärlocher, F., 1983. Seasonal variation of standing crop and digestibility of CPOM in a Swiss Jura stream. Ecology 64: 1266–1272.
Barmuta, L. A., 1989. Habitat patchiness and macrobenthic community structure in an upland stream in temperate Victoria, Australia. Freshwater Biology 21: 223–236.
Bastian, M., L. Boyero, B. R. Jackes & R. G. Pearson, 2007. Leaf litter diversity and shredder preferences in an Australian tropical rain-forest stream. Journal of Tropical Ecology 23: 219–229.
Benfield, E. F., J. R. Webster, J. J. Hutchens, J. L. Tank & P. A. Turner, 2000. Organic matter dynamics along a stream-order and elevational gradient in a southern Appalachian stream. Internationale Vereinigung für theoretische und angewandte Limnologie: Verhandlungen 27: 1341–1345.
Benson, L. J., 1999. Allochthonous input and dynamics in an upland tropical stream. PhD thesis, James Cook University, Townsville, Australia.
Benson, L. J. & R. G. Pearson, 1987. The role of drift and effect of season in colonization of implanted substrata in a tropical Australian stream. Freshwater Biology 18: 109–116.
Benson, L. J. & R. G. Pearson, 1988. Diversity and seasonality of adult Trichoptera captured in a light-trap at Yuccabine Creek, a tropical Australian rainforest stream. Australian Journal of Ecology 13: 337–344.
Benson, L. J. & R. G. Pearson, 1993. Litter inputs to a tropical Australian upland rainforest stream. Australian Journal of Ecology 18: 377–383.
Boulton, A. J. & P. S. Lake, 1992. Benthic organic matter and detritivorous macroinvertebrates in two intermittent streams in south-eastern Australia. Hydrobiologia 241: 107–118.
Boyero, L. & R. C. Bailey, 2001. Organization of macroinvertebrate communities at a hierarchy of spatial scales in a tropical stream. Hydrobiologia 464: 219–225.
Boyero, L., R. G. Pearson, C. Hui, M. O. Gessner, J. Pérez, M. A. Alexandrou, M. A. S. Graça, B. J. Cardinale, R. J. Albariño, M. Arunachalam, L. A. Barmuta, A. J. Boulton, A. Bruder, M. Callisto, E. Chauvet, R. G. Death, D. Dudgeon, A. C. Encalada, V. Ferreira, R. Figueroa, A. S. Flecker, J. F. Gonçalves Jr., J. Helson, T. Iwata, T. Jinggut, J. Mathooko, C. Mathuriau, C. M’Erimba, M. Moretti, C. M. Pringle, A. Ramírez, L. Ratnarajah, J. Rincon & C. M. Yule, 2016. Biotic and abiotic variables influencing plant litter breakdown in streams: a global study. Proceedings of the Royal Society B 283: 20152664.
Brooks, A. J., B. Wolfenden, B. J. Downes & J. Lancaster, 2017. Do pools impede drift dispersal by stream insects? Freshwater Biology 62: 1578–1586.
Brown, A. V. & P. P. Brussock, 1991. Comparisons of benthic invertebrates between riffles and pools. Hydrobiologia 220: 99–108.
Bunn, S. E. & J. M. Hughes, 1997. Dispersal and recruitment in streams: evidence from genetic studies. Journal of the North American Benthological Society 16: 338–346.
Callisto, M. & M. A. S. Graça, 2013. The quality and availability of fine particulate organic matter for collector species in headwater streams. International Review of Hydrobiology 98: 132–140.
Campbell, I. C. & L. Fuchshuber, 1994. Amount, composition and seasonality of terrestrial litter accession to an Australian cool temperate rainforest stream. Archiv für Hydrobiologie 130: 499–512.
Chara, J. D., D. Baird, T. Telfer & L. Giraldo, 2007. A comparative study of leaf breakdown of three native tree species in a slowly-flowing headwater stream in the Colombian Andes. Hydrobiologia 92: 183–198.
Chara-Serna, A. M., J. D. Chara, M. C. Zuniga, R. G. Pearson & L. Boyero, 2012. Diets of leaf litter-associated invertebrates in three tropical streams. Annales de Limnologie – International Journal of Limnology 48: 139–144.
Cheshire, K., L. Boyero & R. G. Pearson, 2005. Food webs in tropical Australian streams: shredders are not scarce. Freshwater Biology 50: 748–769.
Clarke, A., R. MacNally, N. Bond & P. S. Lake, 2008. Macroinvertebrate diversity in headwater streams: a review. Freshwater Biology 53: 1707–1721.
Clayton, P. & R. G. Pearson, 2016. Harsh habitats? Waterfalls and their faunal dynamics in tropical Australia. Hydrobiologia 775: 123–137.
Colón-Gaud, C., S. Peterson, M. R. Whiles, S. S. Kilham, K. R. Lips & C. M. Pringle, 2008. Allochthonous litter inputs, organic matter standing stocks, and organic seston dynamics in upland Panamanian streams: potential effects of larval amphibians on organic matter dynamics. Hydrobiologia 603: 301–312.
Congdon, R. A. & J. L. Herbohn, 1993. Ecosystem dynamics of disturbed and undisturbed sites in north Queensland wet tropical rain forest. I. Floristic composition, climate and soil chemistry. Journal of Tropical Ecology 9: 349–363.
Connolly, N. M. & R. G. Pearson, 2013. Nutrient enrichment of a heterotrophic stream alters leaf-litter nutritional quality and shredder physiological condition via the microbial pathway. Hydrobiologia 718: 85–92.
Crowl, T. A., A. P. Covich, F. N. Scatena, R. Phillips, M. J. Townsend & D. K. Vinson, 2002. Particulate organic matter dynamics in tropical headwater streams: a comparison of biotic and abiotic factors. Internationale Vereinigung für theoretische und angewandte Limnologie: Verhandlungen 28: 923–927.
Cummins, K. W., 1974. Structure and function of stream ecosystems. BioScience 24: 631–641.
Cummins, K. W., R. C. Petersen, F. O. Howard, J. C. Waycheck & V. I. Holt, 1973. The utilisation of leaf litter by stream detritivores. Ecology 54: 336–345.
De Nadai-Monoury, E., F. Gilbert & A. Lecerf, 2014. Forest canopy cover determines invertebrate diversity and ecosystem process rates in depositional zones of headwater streams. Freshwater Biology 59: 1532–1545.
Dudgeon, D. & K. K. Y. Wu, 1999. Leaf litter in a tropical stream: food or substrate for macroinvertebrates? Archiv für Hydrobiologie 146: 65–82.
Ferreira, V., A. C. Encalada & M. A. S. Graça, 2012. Effects of litter diversity on decomposition and biological colonization of submerged litter in temperate and tropical streams. Freshwater Science 31: 945–962.
Gomi, T., R. C. Sidle & J. S. Richardson, 2002. Understanding processes and downstream linkages of headwater systems. BioScience 52: 905–916.
Graça, M. A. S., V. Ferreira, C. Canhoto, A. C. Encalada, F. Guerrero-Bolaño, K. M. Wantzen & L. Boyero, 2015. A conceptual model of litter breakdown in low order streams. International Review of Hydrobiology 100: 1–12.
Grubaugh, J. W., J. B. Wallace & E. S. Houston, 1996. Longitudinal changes of macroinvertebrate communities along an Appalachian stream continuum. Canadian Journal of Fisheries and Aquatic Sciences 53: 896–909.
Hawkins, C. P. & J. R. Sedell, 1981. Longitudinal and seasonal changes in functional organization of macroinvertebrate communities in four Oregon streams. Ecology 62: 387–397.
Hearnden, M. R. & R. G. Pearson, 1991. Habitat partitioning among mayflies (Insecta: Ephemeroptera) in an Australian tropical stream. Oecologia 87: 91–101.
Heino, J., P. Louhi & T. Muotka, 2004. Identifying the scales of variability in stream macroinvertebrate abundance, functional composition and assemblage structure. Freshwater Biology 49: 1230–1239.
Herbst, D. B., S. D. Cooper, R. B. Medhurst, S. W. Wiseman & C. T. Hunsaker, 2018. A comparison of the taxonomic and trait structure of macroinvertebrate communities between the riffles and pools of montane headwater streams. Hydrobiologia 820: 115–133.
Hoover, T. M., J. S. Richardson & N. Yonemitsu, 2006. Flow-substrate interactions create and mediate leaf litter resource patches in streams. Freshwater Biology 51: 435–447.
Hopkins, M. S. & A. W. Graham, 1989. Community phenological patterns of a lowland tropical rain forest in north-eastern Australia. Australian Journal of Ecology 14: 399–414.
Huryn, A. D. & J. B. Wallace, 1988. Community structure of Trichoptera in a mountain stream: spatial patterns of production and functional organization. Freshwater Biology 20: 141–155.
Huttunen, K.-L., H. Mykrä, R. Paavola & T. Muotka, 2018. Estimates of benthic invertebrate community variability and its environmental determinants differ between snapshot and trajectory designs. Freshwater Science 37: 769–779.
Januchowski-Hartley, S. R., R. G. Pearson, R. Puschendorf & T. Rayner, 2011. Fresh waters and fish diversity: distribution, protection and disturbance in tropical Australia. PLoS ONE 6: e25846.
Kobayashi, S. & Kagaya, 2002. Differences in litter characteristics and macroinvertebrate assemblages between litter patches in pools and riffles in a headwater stream. Limnology 3: 37–42.
Landeiro, V. L., N. Hamada, B. S. Godoy & A. S. Melo, 2010. Effects of litter patch area on macroinvertebrate assemblage structure and leaf breakdown in Central Amazonian streams. Hydrobiologia 649: 355–363.
Li, A. O. Y. & D. Dudgeon, 2009. Shredders: species richness, abundance, and role in litter breakdown in tropical Hong Kong stream. Journal of the North American Benthological Society 28: 167–180.
Lisboa, L. K., A. L. L. da Silva, A. E. Siegloch, J. F. Gonçalves Júnior & M. M. Petrucio, 2015. Temporal dynamics of allochthonous coarse particulate organic matter in a subtropical Atlantic rainforest Brazilian stream. Marine and Freshwater Research 66: 674–680.
Logan, P. & M. P. Brooker, 1983. The macroinvertebrate fauna of riffles and pools. Water Research 17: 263–270.
McCulloch, D. L., 1986. Benthic macroinvertebrate distributions in the riffle-pool communities of two east Texas streams. Hydrobiologia 135: 61–70.
McElravy, E. P., G. A. Lamberti & V. H. Resh, 1989. Year-to-year variation in the aquatic macroinvertebrate fauna of a northern California stream. Journal of the North American Benthological Society 8: 51–63.
McNeely, C., C. Finlay & M. E. Power, 2007. Grazer traits, competition, and carbon sources to a headwater-stream food web. Ecology 88: 391–401.
Meyer, J.L., L.A. Kaplan, J.D. Newbold, D.L. Strayer, C.J. Woltemade, J.B. Zedler, R. Beilfuss, Q. Carpenter, R. Semlitsch, M.C. Watzin & P.H. Zedler, 2003. Where rivers are born: the scientific imperative for defending small streams and wetlands. American Rivers and Sierra Club. https://stroudcenter.org/publications/where-rivers-are-born/. Accessed 8 Dec 2018.
Meyer, J. L. & J. B. Wallace, 2001. Lost linkages and lotic ecology: rediscovering small streams. In Press, M. C., N. J. Huntly & S. Levin (eds), Ecology: Achievement and Challenge. Blackwell Science, Oxford: 295–317.
Meyer, J. L., D. L. Strayer, J. B. Wallace, S. L. Eggert, G. S. Helfman & N. E. Leonard, 2007. The contribution of headwater streams to biodiversity in river networks. Journal of the American Water Resources Association 43: 86–103.
Minshall, G. W., R. C. Petersen, K. W. Cummins, T. L. Bott, J. R. Sedell, C. E. Cushing & R. L. Vannote, 1983. Interbiome comparison of stream ecosystem dynamics. Ecological Monographs 53: 1–25.
Molinero, J., 2019. Seasonality and composition of benthic coarse particulate organic matter in two coastal tropical streams with different land uses. Hydrobiologia 838: 29–43.
Moss, B., 2005. Rapid shredding of leaves by crabs in a tropical African stream. Internationale Vereinigung für theoretische und angewandte Limnologie: Verhandlungen 29: 147–150.
Naiman, R. J. & J. R. Sedell, 1980. Relationship between metabolic parameters and stream order in Oregon. Canadian Journal of Fisheries and Aquatic Sciences 37: 834–844.
Nakajima, T., T. Asaeda, T. Fujino & A. Nanda, 2006. Coarse particulate organic matter distribution in the pools and riffles of a second-order stream. Hydrobiologia 559: 275–283.
Nolen, J. A. & R. G. Pearson, 1992. Life history studies of Anisocentropus kirramus Neboiss (Trichoptera: Calamoceratidae) in a tropical Australian rainforest stream. Aquatic Insects 14: 213–221.
Nolen, J. A. & R. G. Pearson, 1993. Factors affecting litter processing by Anisocentropus kirramus (Trichoptera: Calamoceratidae) from an Australian tropical rainforest stream. Freshwater Biology 29: 469–479.
Payne, B. S. & A. C. Miller, 1991. The structure of dominant invertebrate assemblages in a small southeastern stream. Journal of Freshwater Ecology 6: 257–266.
Pearson, R. G., 2014. Dynamics of invertebrate diversity in a tropical stream. Diversity 6: 771–791.
Pearson, R. G. & L. Boyero, 2009. Gradients in regional diversity of freshwater taxa. Journal of the North American Benthological Society 28: 504–514.
Pearson, R. G., R. K. Tobin, R. E. W. Smith & L. J. Benson, 1989. Standing crop and processing of rainforest litter in a tropical Australian stream. Archiv für Hydrobiologie 115: 481–498.
Pearson, R. G., F. Christidis, N. M. Connolly, J. A. Nolen, R. M. St Clair, A. E. Cairns & L. Davis, 2017. Stream macroinvertebrate assemblage uniformity and drivers in a tropical bioregion. Freshwater Biology 62: 544–558.
Quinn, J. M., N. R. Phillips & S. M. Parkyn, 2007. Factors influencing retention of coarse particulate organic matter in streams. Earth Surface Processes and Landforms 32: 1186–1203.
Ramirez, A. & C. M. Pringle, 1998. Structure and production of a benthic insect assemblage in a neotropical stream. Journal of the North American Benthological Society 17: 443–463.
Resh, V. H., A. V. Brown, A. P. Covich, M. E. Gurtz, H. W. Li, G. W. Minshall, S. R. Reice, A. L. Sheldon, J. B. Wallace & R. C. Wissman, 1988. The role of disturbance in stream ecology. Journal of the North American Benthological Society 7: 433–455.
Richardson, J. S., 1992. Food, microhabitat, or both? Macroinvertebrate use of leaf accumulations in a montane stream. Freshwater Biology 27: 169–176.
Rosser, Z. C. & R. G. Pearson, 1995. Reponses of rock fauna to physical disturbance in two Australian tropical rainforest streams. Journal of the North American Benthological Society 14: 183–196.
Rosser, Z. C. & R. G. Pearson, 2018. Hydrology, hydraulics and scale influence macroinvertebrate responses to disturbance in tropical streams. Journal of Freshwater Ecology 33: 1–17.
Sarremejane, R., H. Mykrä, K.-L. Huttunen, K.-R. Mustonen, H. Marttila, R. Paavola, K. Sippel, N. Veijalainen & T. Muotka, 2018. Climate-driven hydrological variability determines inter-annual changes in stream invertebrate community assembly. Oikos 127: 1586–1595.
Schmera, D. & T. Erös, 2011. The role of sampling effort, taxonomical resolution and abundance weight in multivariate comparison of stream dwelling caddisfly assemblages collected from riffle and pool habitats. Ecological Indicators 11: 230–239.
Schmidt, K., M. Blanchette, R. G. Pearson, R. A. Alford & A. M. Davis, 2017. Trophic roles of tadpoles in tropical Australian streams. Freshwater Biology 62: 1929–1941.
Scullion, J., C. A. Parish, N. Morgan & R. W. Edwards, 1982. Comparison of benthic macroinvertebrate fauna and substratum composition in riffles and pools in the impounded River Elan and the unregulated River Wye, mid-Wales. Freshwater Biology 12: 579–595.
Spain, A. V., 1984. Litterfall and the standing crop of litter in three tropical Australian rainforests. Journal of Ecology 72: 947–961.
Speaker, R., K. Moore & S. Gregory, 1984. Analysis of the process of retention of organic matter in stream ecosystems. Internationale Vereinigung für theoretische und angewandte Limnologie: Verhandlungen 22: 1835–1841.
Stewart, B. A. & B. R. Davies, 1990. Allochthonous input and retention in a small mountain stream, South Africa. Hydrobiologia 202: 135–146.
Straka, M., V. Syrovátka & J. Helešic, 2012. Temporal and spatial macroinvertebrate variance compared: crucial role of CPOM in a headwater stream. Hydrobiologia 686: 119–134.
Surber, E. W., 1937. Rainbow trout and bottom fauna production in one mile of a stream. Transactions of the American Fisheries Society 66: 193–202.
Tracey, J. G., 1982. The vegetation of the humid tropical region of north Queensland. CSIRO, Melbourne.
Tumwesigye, C., S. K. Yusuf & B. Makanga, 2000. Structure and composition of benthic macroinvertebrates of a tropical forest stream, River Nyamweru, western Uganda. African Journal of Ecology 38: 72–77.
Vanotte, R. L., G. W. Minshall, K. W. Cummins, J. R. Sedell & C. E. Cushing, 1980. The river continuum concept. Canadian Journal of Fisheries and Aquatic Sciences 37: 130–137.
Vinson, M. R. & C. P. Hawkins, 2004. Broad-scale geographical patterns in local stream insect genera richness. Ecography 26: 751–767.
Waite, I. R., A. T. Herlihy, D. P. Larsen & D. J. Klemm, 2000. Comparing strengths of geographic and nongeographic classifications of stream benthic macroinvertebrates in the Mid-Atlantic Highlands, USA. Journal of the North American Benthological Society 19: 429–441.
Wallace, J. B. & J. R. Webster, 1996. The role of macroinvertebrates in stream ecosystem function. Annual Review of Entomology 41: 115–139.
Wallace, J. B., M. R. Whiles, S. Eggert, T. F. Cuffney, G. J. Lugthart & K. Chung, 1995. Long-term dynamics of coarse particulate organic matter in three Appalachian Mountain streams. Journal of the North American Benthological Society 14: 217–232.
Walther, D. A. & M. R. Whiles, 2011. Secondary production in a southern Illinois headwater stream: relationships between organic matter standing stocks and macroinvertebrate productivity. Freshwater Science 30: 357–373.
Webb, L. J., 1978. A general classification of Australian rain forests. Australian Plants 9: 349–363.
Webster, J. R., A. P. Covich, J. L. Tank & T. V. Crocket, 1994. Retention of coarse organic particles in streams in the southern Appalachian Mountains. Journal of the North American Benthological Society 9: 120–140.
Wipfli, M. S., J. S. Richardson & R. J. Naiman, 2007. Ecological linkages between headwaters and downstream ecosystems: transport of organic matter, invertebrates, and wood down headwater channels. Journal of the American Water Resources Association 43: 72–85.
Wootton, A., R. G. Pearson & L. Boyero, 2019. Patterns of flow, leaf litter and shredder abundance in a tropical stream. Hydrobiologia 826: 353–365.
Acknowledgements
We thank those people who assisted in the field, especially Jacqui Coughlan and Jacqui Nolen, and two anonymous reviewers for their constructive comments on the manuscript. Funding was provided by James Cook University.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling editor: Verónica Ferreira
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Benson, L.J., Pearson, R.G. Dynamics of organic material and invertebrates in a tropical headwater stream. Hydrobiologia 847, 121–136 (2020). https://doi.org/10.1007/s10750-019-04076-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-019-04076-1