Abstract
Paroxysmal atrial fibrillation (PAF) may be the cause of a substantial part of cryptogenic strokes (CS). Echocardiography could assist risk stratification for PAF to select patients in need of prolonged rhythm monitoring. We aimed to assess the value of left atrial (LA) strain and a revised diastolic dysfunction (DDF) model with LA strain for predicting PAF. This was a prospective study of 56 CS patients who had a cardiac monitor implanted for 3 year monitoring for PAF, and an echocardiogram performed prior to monitoring. Conventional echocardiography, global longitudinal strain (GLS) and LA strain were performed. LA speckle tracking provided the LA reservoir strain (LAs). Patients were stratified into high versus low LAs by ROC curves (28.2%), and this cut-off was used to refine DDF grading. During follow-up of median 20 months, 13 (23%) patients were diagnosed with PAF. No conventional echocardiographic parameters differed between patients who developed PAF and those without PAF. However, LAs was significantly impaired in PAF patients (LAs: 30 vs. 27% for non-PAF and PAF, p = 0.046). Low LAs significantly predicted PAF independent of LA volume and GLS [OR 5.88 (1.30; 26.55), p = 0.021]. Revised DDF grading significantly predicted PAF, even when adjusted for the CHADS2 risk-score (OR 1.88 [1.01;3.50], per increase in DDF grade, p for trend = 0.047), which was not the case for conventional DDF grading. In conclusion, LAs associates with PAF independent of GLS and LA size, and may be used to improve the performance of DDF grading for identifying PAF in CS patients.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Stroke is the second leading cause of death globally and often associated with great morbidity [1]. The majority of strokes are ischemic, and ischemic strokes can be further subdivided based on etiology according to the TOAST classification: lacunar infarcts, large-artery atherosclerosis, cardioembolic strokes, stroke of other determined etiology and strokes of undetermined etiology [2]. However, in about 25–30% of cases no underlying cause is found during basic work up and the event is designated a cryptogenic stroke (CS) [3]. It has long been hypothesized that subclinical paroxysms of atrial fibrillation (AF) may be the cause. This has prompted several studies to investigate the utility of prolonged cardiac rhythm monitoring [4, 5], and several studies now support the application of insertable cardiac monitors (ICM) for AF diagnostics after stroke [6,7,8]. The most recent guidelines from the European Society of Cardiology on the management of AF have set forth a Class IIa, level B, indication for the use of ICMs after ischemic strokes and states that long-term monitoring should be considered in the search for silent AF [9]. This warrants a more strict risk stratification for AF in patients after stroke in order to optimize cost–benefit and to avoid unnecessary use of long term monitoring in patients with low risk of AF. Risk stratification could be facilitated by the use of echocardiography, which provides direct visualization of the left atrium (LA) and allows detailed quantification of LA structural and functional properties.
In echocardiography the maximal LA volume (max LAV) and diastolic dysfunction (DDF) are well-established predictors of AF [10, 11]. Recently, LA reservoir strain (LAs) has been proposed as a novel measure of LA dysfunction and may be used to optimize characterization of DDF [12]. However, it was suggested that LAs does not provide clinically relevant information beyond left ventricular (LV) strain and max LAV [13, 14]. Furthermore, whether LAs or revised DDF could be useful in patients with CS for predicting device-detected AF is unclear. Hence, the purpose of this study of CS patients with continuous rhythm monitoring was: (1) to investigate the prognostic potential of LA strain, (2) to investigate if the prognostic value of LA strain is mediated by max LAV and LV strain and (3) to investigate if LA strain can refine DDF grading and how this relates to AF outcome.
Methods
This was an echocardiographic substudy of the SURPRISE study (Stroke Prior to Diagnosis of Atrial Fibrillation Using Long Term Observation with Implantable Cardiac Monitoring Apparatus Reveal®), which was undertaken at Bispebjerg Hospital, Copenhagen, Denmark from May 2010 to August 2012. The study population has previously been described in detail [4] and information on the echocardiographic substudy has also been published [15]. Overall, 87 patients with CS were included in the SURPRISE study, and 58 of these underwent an echocardiographic examination as part of the investigative work-up of their stroke to identify a potential underlying cardiac etiology. Of these, two patients were excluded because LV and/or LA speckle tracking was infeasible, leaving 56 for final inclusion. Inclusion criteria for the main study were patients with: (1) a verified ischemic stroke by cerebral computed tomography (CT) or cerebral magnetic resonance imaging (MRI), (2) age above 18 years, (3) no history of AF, (4) no AF detected during the admission based on 12-lead ECG and 12–24 h of telemetry, (5) modified ranking scale (mRS) ≤ 2 equalling independent in ADL-functions. Finally, patients were only designated cryptogenic if the work-up did not reveal occlusive carotid disease, cardiac embolic source, obvious signs of small vessel disease or thrombophilia.
Reveal apparatus and AF diagnosis
The Reveal XT® ICM provides continuous cardiac rhythm monitoring for up to 3 years (Fig. 1). The device is implanted subcutaneously and continuously monitors beat-to-beat variability in 2 min loops. When R–R intervals are irregular, data is stored and via the patient uploaded to a protected server with access for clinicians. All included patients, except two, had an ICM implanted as soon as possible after their stroke. The two patients who did not have an ICM implanted were diagnosed with PAF before the scheduled implantation date, which rendered implantation futile.
Patients actively transmitted data every fourth night. If a significant AF event was detected, patients were switched from antiplatelet treatment to anticoagulation treatment.
The AF episode had to last 120 s or longer to be classified as a significant AF event. Two cardiologists adjudicated all events.
Echocardiography
All patients were examined with conventional two-dimensional transthoracic echocardiography (using Philips iE 33, Netherlands) and analyzed off-line. All off-line analyses were done with commercially available software (Xcelera quantification software) with the investigator blinded to the results of all other investigations.
Conventional echocardiography
LV dimensions (interventricular septum wall thickness, LV internal dimension and LV posterior wall thickness) were obtained at end-diastole from the parasternal long-axis view at the level of the mitral valve leaflet tips. LV mass index was calculated as the anatomic mass by Deveraux’ formula and divided with body surface area (BSA). Pulsed wave Doppler in the apical four-chamber view was used to record mitral valve inflow patterns between the tips of the mitral leaflets. Peak velocity of early (E) and late (A) diastolic filling and deceleration time of the E-wave (DT) were measured and the E/A-ratio was calculated. Pulsed-wave tissue Doppler imaging (TDI) tracings were obtained with the range gate placed at the septal and/or lateral mitral annular segment in the apical 4-chamber view. The peak longitudinal early diastolic myocardial velocity (e′) was measured and used to obtain the E/e′. Diastolic function grading was assessed by max LAV, E/A-ratio, e′ and E/e′ according to a recently proposed DDF model [16] and in a revised form including LAs (Fig. 2). Fifty of the 56 patients had all DDF measures performed, and so 6 patients were not included in the DDF grading. The LV ejection fraction (LVEF) was measured using the modified Simpson’s biplane method. LA volumes were estimated by the biplane area-length method. The max LAV was measured at LV end-systole and minimal LA volume (min LAV) at LV end-diastole.
Revised diastolic dysfunction grading. Building on a recently suggested diastolic dysfunction grading model, we added the LAs split by into high versus low LAs based on receiver operating characteristics curves. Entry level for diastolic dysfunction was an impaired relaxation velocity (e′) and number of impaired E/e′, E/A ratio, max LAV and LAs redistributed patients into different degrees of diastolic dysfunction. e′ early myocardial relaxation velocity, E/e′ ratio of transmitral early inflow velocity to early myocardial relaxation velocity, max LAV Maximal left atrial volume, LAs left atrial strain
Speckle tracking echocardiography
Speckle tracking was performed as off-line analysis using commercially available software, Epsilon Imaging (EchoInsight® software revision 2.2.0.x), by an investigator blinded to outcome and baseline data. LV speckle tracking was performed in accordance with published guidelines [17]. Analysis was performed in all three apical views (4-chamber, 2-chamber and apical longitudinal long-axis view) by manual delineation of the LV endocardium. The region of interest was adjusted to include the endocardium, midmyocardium, and the epicardium. Segments could be excluded at the discretion of the investigator. If more than two segments were excluded in a projection, the investigation was deemed unsatisfactory. LA speckle tracking was performed as biplane analysis from the apical 4-chamber and 2-chamber view. Manual delineation of the endocardium was also performed in the LA and regional region of interest was adjusted as deemed appropriate by the investigator. The roof of the atrium was systematically excluded due to influence of pulmonary venous structures and non-longitudinal fiber orientation in this part of the LA [18, 19]. LV speckle tracking provided the global longitudinal strain (GLS), systolic strain rate (SRs), early diastolic strain rate (SRe) and late diastolic strain rate (SRa).
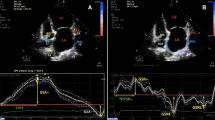
LA speckle tracking provided the LAs, the plateau phase strain at the p-wave, and also systolic strain rate (LA-SRs), early diastolic strain rate (LA-SRe) and late diastolic strain rate (LA-SRa). The plateau strain was subtracted from LAs to provide the conduit strain (Fig. 3). LAs was indexed to the max LAV (LAs/LAV) as proposed by other studies [20].
Left atrial speckle tracking. Left atrial speckle tracking performed in the apical 4-chamber view, which is shown on the left. Overall strain measures from the analysis are presented as the curves placed in the upper section on the right side. Measures of left atrial strain by different phases are highlighted in text. In the section below are curves which show the strain rates of the different phases in this analysis. LA-SRs left atrial systolic strain rate, LA-SRe left atrial early diastolic strain rate, LA-SRa left atrial late diastolic strain rate
Statistical analysis
STATA Statistics/Data analysis, SE 13.0 (StataCorp, Texas, USA) was used for statistical analysis. Patients were compared according to outcome of significant AF event (Tables 1, 2). Furthermore, patients were stratified into a high and low LAs group according to a cutoff value derived from receiver operating characteristics curves showing the highest area under the curve (cutoff = 28.2% with an area under the curve of 0.69) and these groups were compared in Tables 3 and 4.
In baseline Tables 1, 2, 3 and 4 proportions were compared using χ2-test, continuous Gaussian distributed variables with Student’s t test, and Wilcoxon-Mann–Whitney test if the variables were non-Gaussian distributed. All continuous variables are expressed as mean values ± standard deviation, continuous non-Gaussian distributed variables are presented by the median and IQR.
Univariable logistic regressions models were performed to correlate echocardiographic measures to the endpoint of significant AF event. Multivariable logistic regression models were performed to investigate whether low LAs was a predictor of AF independent of GLS and max LAV, and whether DDF grading predicted AF independent of clinical risk score (CHADS2). Spline curves were constructed from a univariable logistic regression spline model to assess the probability of AF according to LAs (Fig. 4a), and from a multivariable logistic regression spline model to assess the probability of AF according to LAs after adjusting for max LAV and GLS (Fig. 4b). Spline curves were created with two-tied knots, which provided the lowest Akaike information criterion (AIC) score.
a Spline curve illustrating the probability of AF with decreasing LAs. Black line shows unadjusted probability of device-detected AF with 95% confidence intervals (dotted lines) per decreasing percentages of LAs in patients with cryptogenic ischemic stroke. AF atrial fibrillation, LAs left atrial reservoir strain. b Black line shows probability of device-detected AF with 95% confidence intervals (dotted lines) per decreasing percentages of LAs in patients with cryptogenic ischemic stroke when adjusted for GLS and max LAV. AF atrial fibrillation, LAs left atrial reservoir strain, GLS global longitudinal strain; max LAV maximal left atrial volume
Ethics
The study was approved by the Ethics committee of the Capital Region of Denmark (H3-2010-083) and registered at clinicaltrials.gov (NCT01498146). All patients provided informed written consent.
Results
Of the 56 patients included in this study, 13 (23%) were diagnosed with PAF during a median follow-up period of 20 months (interquartile range (IQR) 11–29 months). There was a complete (100%) follow-up. Implantation was performed within a median 65 days (IQR 41–123 days) after the index stroke. PAF was diagnosed a median of 59 days (IQR 14–108 days) after ICM implantation. All significant AF events were asymptomatic and prompted conversion from antiplatelet to anticoagulant therapy.
The echocardiograms were performed at a median of 50 days (IQR 18–146 days) after the stroke event as part of the routine clinical work-up.
Clinical baseline
Clinical baseline characteristics for the entire population and based on outcome of AF are outlined in Table 1. Patients who developed AF were significantly older than patients who remained free of AF (65 vs. 50 years, p < 0.001). They also exhibited a higher risk of stroke by the CHADS2-score (2.9 vs. 2.4, p = 0.029). There was also a trend toward a higher CHA2DS2-VASc-score among AF patients (3.7 vs. 3.0, p = 0.07). Aside from this, the two groups did not differ clinically.
Left atrial measures
Echocardiographic baseline characteristics for the entire population and based on outcome are shown in Table 2. The patients did not differ by any conventional echocardiographic measures including LA volumes (min LAV and max LAV). However, the two groups did differ with respect to LAs, with the AF outcome group showing significant impairment in LAs: 27 versus 30%, p = 0.046. The LAs/LAV did not differ between outcome groups.
When patient characteristics were stratified based on the cutoff for LAs (28.2%), it became evident that only age and diabetes was higher and more prevalent, respectively, with worsening LAs, but beside this no other clinical feature differed between those with high versus low LAs (Table 3). For echocardiographic measures, the e′ was lower and E/e′ higher in patients with low LAs. For LA measures, no conventional LA measures differed between the high or low LAs groups, however, absolute GLS and all LA speckle tracking measures were significantly lower in the low LAs group, except for the late diastolic strain rate (Table 4).
No conventional echocardiographic parameters were univariable predictors of AF. Nor were the max LAV or min LAV predictors of AF, regardless of whether they were indexed to body surface area or not. A dilated LA (defined as max LAV ≥ 34 mL/m2) was present in 16 patients and was not associated with AF [OR 1.76 (0.47;6.54), p = 0.40] in univariable logistic regression. Patients with low LAs as determined by a value below 28.2% had an approximately five times greater risk of having occult AF [OR 5.01 (1.22;21.25), p = 0.025]. Even after adjusting for max LAV and GLS, those with low LAs had a significantly increased risk of AF compared to those with high LAs [OR 5.88 (1.30;26.55), p = 0.021]. Logistic spline curves illustrate the increased risk of AF with decreasing percentage of LAs in an unadjusted model (Fig. 4a) and when adjusted for GLS and max LAV (Fig. 4b), p value 0.039 and 0.029, respectively.
Diastolic dysfunction grading
Conventional DDF grading showed a trend towards being a predictor of AF: OR 1.83 [0.95;3.50], p for trend = 0.069, per worsening DDF grade, but was not a predictor when adjusted for the CHADS2-score [OR 1.87 (0.92;3.81), p for trend = 0.09, per worsening DDF grade]. In the revised DDF model, 7 patients were reclassified from having DDF grade 1 to 2, and 5 more were reclassified from grade 2 to 3 (Table 1). The revised DDF model was a stronger predictor than conventional DDF grading [OR 1.90 (1.07; 3.35), p for trend = 0.028, per worsening DDF grade] and remained a strong predictor even after adjusting for the CHADS2-score [OR 1.88 (1.01; 3.50), p for trend = 0.047, per worsening DDF grade].
Discussion
This is to our knowledge the first study to evaluate the applicability of speckle tracking echocardiography for the prediction of device-detected AF in patients with cryptogenic stroke. Continuous monitoring for up to 3 years by an ICM with a dedicated AF detection algorithm is considered the gold standard for AF detection since this method will also detect asymptomatic episodes. Our findings suggest LAs to be a valuable tool for prediction of AF, whereas LA volumes and speckle tracking of the LV was of no predictive value in this study. Interestingly, the predictive value of LAs did not appear to be mediated by GLS and max LAV, as previously suggested. Furthermore, diastolic dysfunction classification may be improved by including LAs and a revised version of diastolic dysfunction grading may indicate an increased risk of subclinical AF in cryptogenic stroke.
LA strain and AF
Findings from the Utah group have revealed an inverse linear relation between amount of fibrosis and LA reservoir strain [21], suggesting LA reservoir strain to be a sensitive marker of LA fibrosis, and thereby a marker of AF risk [22, 23]. This is in accordance with the results presented in the present report where LAs was found to be of value in identifying patients with subclinical AF following a CS. Recently, similar findings were published by Choi et al. [24], who found low LAs could predict subclinical AF in patients with cardiac implantable devices such as pacemakers or implantable cardioverter defibrillators. Pagola et al. [20] performed a prospective pilot study of 90 CS patients and found that both decreased LA reservoir strain and low ratio of LA reservoir strain to max LAV (LAs/LAV) were valuable parameters for predicting AF detected by 72 h Holter monitoring. Although our study showed predictive value of LA reservoir strain, the LAs/LAV-ratio did not prove of value in our population. The relationship between LA reservoir strain and incident AF was also recognized in a large-scale retrospective case–control study by Leong et al. [25], who compared 371 CS patients to 371 controls and revealed the LA reservoir function to be significantly impaired in the CS-group as a marker of LA dysfunction in these patients.
LA reservoir function and LA volumes
The max LAV is the current applied measure for determining LA dysfunction, however, failed to predict AF in this study. The explanation may be given by an invasive study investigating the relationship of echocardiographic measures to LV end-diastolic pressure (LVEDP), which showed that LAs was the parameter that most closely related to LVEDP [26]. Cameli et al. proposed that the reason for this was that LVEDP served as a measure of LA afterload, inducing stress on the LA which is detected by LAs prior to LA volumes. Indeed, LA strain has been hailed as a promising marker of LV filling pressure by experts in the field of diastolic function [27]. This is in line with several studies which have found that LAs becomes impaired before LA enlargement becomes evident, and even predict outcome in patients with non-enlarged LA. This was not possible to assess in this study due to a small sample size. Additionally, the max LAV is clinically meaningful when it indicates a dilated LA (≥ 34 mL/m2) and since the majority of patients did not exhibit a dilated LA in this study, this may explain why the max LAV failed to predict AF. However, even a dilated LA was not associated with device-detected AF in this study, and the fact that LAs can predict AF in a population with so few patients exhibiting a dilated LA only emphasized its potential value. Furthermore, LA dysfunction by atrial volumes poses another challenge as these are often indexed to BSA to compensate for body size, however, recent studies have found that such isometric standardization to BSA may not accurately reflect LA size. Instead it has been proposed that allometric standardization or standardization to other measures of body size may more accurately reflect pathologic enlargement of LA size [28]. This hypothesis is still under investigation, but may also explain why atrial volumes were not of value in our study, and thereby also explain why the LAs/LAV-ratio was not a predictor of AF in our study as this measure suffers from the same bias.
Global longitudinal strain as a marker of LA reservoir function
It is particularly interesting to note that LA reservoir function by LA speckle tracking seemed promising, whereas LA reservoir function by LV analysis (i.e., GLS) seemed unaffected in AF patients in our study. GLS has otherwise shown to predict AF [29], also in device-detected AF after transient ischemic attack [30], and since it indirectly reflects LA reservoir function it has been questioned whether LAs actually provide valuable information beyond GLS and max LAV. Ersbøll et al. showed that the predictive value of LAs was mediated by GLS and max LAV [13]. This notion is, however, challenged by our findings showing predictive value of LAs independent of GLS and max LAV.
Diastolic dysfunction grading
Guidelines for classifying diastolic dysfunction exist, however, a recurrent problem is that patients quite often cannot be placed into the established categories due to conflicting measures. Hence, diastolic assessment is quite complex. To resolve this issue Johansen et al. [16] tried to create a new method of classifying diastolic dysfunction from the Copenhagen City Heart study suggesting the e′ as an entry parameter for having diastolic dysfunction since impaired LV relaxation often is the first step in the process of developing diastolic dysfunction. By this method, a substantial proportion of patients could be classified into different degrees of diastolic dysfunction, and this classification also proved to relate to clinical outcomes better than the conventional DDF grading. Although LAs seems of predictive value in our study, measures of diastolic function should not stand alone in an echocardiographic investigation. By extension, recent studies have suggested that LAs could be of use in DDF grading [12], and showed that LAs progressively worsens with increasing DDF [31]. Although Singh et al. [12] suggested different cut-offs for different degrees of DDF, implementing a strategy of three different cutoffs in the already complex assessment of DDF classification seems clinically challenging. We rather propose a single cutoff for this purpose as is already the clinical way for the other diastolic measures. Although a cutoff of 28.2% seems to be efficient for prediction of AF, further studies are needed to investigate if this modified DDF grading can also be applied for prediction of other clinical outcomes.
Other measures of impaired LA function
Although impaired reservoir function seems to play a role in the pathology of AF, another important marker of AF risk is the LAEF. We have previously reported the prognostic value of LAEF in the same cohort and a similar cohort [15, 32]. This measure reflects both passive and active properties of the LA, but whether the dysfunction lies in the LA conduit function or LA contraction, cannot be determined by the LAEF. Even though our study indicates a decreased LA conduit function to be present in the AF patients, it could just as well be a reduced atrial contraction that mediates the predictive value of LAEF as has been suggested in related studies [33, 34].
Clinical translation and limitations of LA strain
Although LAs shows promise, the technique is still somewhat flawed. As we have previously outlined, several technical obstacles need to be clarified [18]. There is no consensus on how to perform LA speckle tracking. Furthermore, LA speckle tracking is performed with software designed for LV speckle tracking as there is no dedicated speckle tracking algorithm designed for LA analysis specifically. A previous study from Singh et al. reported a high intra-class correlation of 0.89 but with an inter-reader variability of 13% [35]. Keeping this in mind and the fact that the difference in LAs between outcome groups in this study was 3%, the high variability does not favor clinical application of the software and emphasizes the need for a dedicated LA speckle tracking software. Our findings should therefore be seen as an extension of similar hypothesis generating studies to also include patients with cryptogenic stroke and device-detected AF.
These issues may, however, be resolved in the near future since several initiatives from international imaging societies have been developed. The MASCOT-HIT study will investigate the technical aspects on how to optimize LA speckle tracking, an expert panel has been convened to standardize LA imaging [19], and the EACVI Echo Afib registry [36] has been developed to investigate how cardiac imaging can be used to guide prevention of ischemic stroke. All initiatives stress the importance of the subject and the dedication towards implementing LAs in clinical practice.
Limitations
Identifying echocardiographic predictors of PAF was not the primary focus of the SURPRISE study and only two-thirds of the study population underwent an echocardiographic examination as part of the diagnostic work-up. No (rare) structural cardio-embolic sources were diagnosed in these standardized echocardiographic examinations, which emphasizes the potential information about atrial pathophysiology and potential presence of PAF which can be acquired from the routine echocardiogram.
These results are from a somewhat small sample size, and results cannot be extrapolated to the entire cohort of CS patients.
Some medication may have potential protective effects for the development of AF such as beta blockers, unfortunately we did not have information on such medication use and we can therefore not exclude the possibility of residual confounding.
An important aspect to consider is that even though studies have revealed a high AF detection rate with increased rhythm monitoring, there is currently no evidence to support that initiation of anticoagulation treatment based on opportunistic screening for PAF also translates into a better outcome in these patients. This needs to be clarified before ICM implantation is considered routinely implemented in the clinic, and this also influences the need for echocardiographic selection of patients in need of prolonged rhythm monitoring.
Conclusion
Low LAs predicts device-detected subclinical AF in cryptogenic stroke patients, whereas left ventricular speckle tracking does not predict AF. LA reservoir strain provides prognostic value independent of LA size and GLS. Furthermore, LA reservoir strain may be used to revise diastolic dysfunction grading, which may translate into an increased risk of AF independent of clinical risk score.
References
Olsen FJ, Møgelvang R, Jensen GB et al (2018) Relationship between left atrial functional measures and incident atrial fibrillation in the general population: the copenhagen city heart study. JACC Cardiovasc Imaging 12:981–989. https://doi.org/10.1016/j.jcmg.2017.12.016
Adams HP, Bendixen BH, Kappelle LJ et al (1993) Classification of subtype of acute ischemic stroke. Definitions for use in a multicenter clinical trial. TOAST. Trial of Org 10172 in Acute Stroke Treatment. Stroke 24:35–41
Hart RG, Diener H-C, Coutts SB et al (2014) Embolic strokes of undetermined source: the case for a new clinical construct. Lancet Neurol 13:429–438. https://doi.org/10.1016/S1474-4422(13)70310-7
Christensen LM, Krieger DW, Højberg S et al (2014) Paroxysmal atrial fibrillation occurs often in cryptogenic ischaemic stroke. Final results from the SURPRISE study. Eur J Neurol 21:884–889. https://doi.org/10.1111/ene.12400
Sanna T, Diener H-C, Passman RS et al (2014) Cryptogenic stroke and underlying atrial fibrillation. N Engl J Med 370:2478–2486. https://doi.org/10.1056/NEJMoa1313600
Diamantopoulos A, Sawyer LM, Lip GYH et al (2016) Cost-effectiveness of an insertable cardiac monitor to detect atrial fibrillation in patients with cryptogenic stroke. Int J Stroke 11:302–312. https://doi.org/10.1177/1747493015620803
Burkowitz J, Merzenich C, Grassme K, Brüggenjürgen B (2016) Insertable cardiac monitors in the diagnosis of syncope and the detection of atrial fibrillation: a systematic review and meta-analysis. Eur J Prev Cardiol 23:1261–1272. https://doi.org/10.1177/2047487316632628
Olsen FJ, Biering-Sørensen T, Krieger DW (2015) An update on insertable cardiac monitors: examining the latest clinical evidence and technology for arrhythmia management. Future Cardiol 11:333–346. https://doi.org/10.2217/fca.15.15
Kirchhof P, Benussi S, Kotecha D et al (2016) 2016 ESC Guidelines for the management of atrial fibrillation developed in collaboration with EACTS. Eur Heart J 37:2893–2962. https://doi.org/10.1093/eurheartj/ehw210
Tsang TS, Barnes ME, Bailey KR et al (2001) Left atrial volume: important risk marker of incident atrial fibrillation in 1655 older men and women. Mayo Clin Proc 76:467–475. https://doi.org/10.4065/76.5.467
Jons C, Joergensen RM, Hassager C et al (2010) Diastolic dysfunction predicts new-onset atrial fibrillation and cardiovascular events in patients with acute myocardial infarction and depressed left ventricular systolic function: a CARISMA substudy. Eur J Echocardiogr 11:602–607. https://doi.org/10.1093/ejechocard/jeq024
Singh A, Addetia K, Maffessanti F et al (2017) LA strain for categorization of LV diastolic dysfunction. JACC Cardiovasc Imaging 10:735–743. https://doi.org/10.1016/j.jcmg.2016.08.014
Ersbøll M, Andersen MJ, Valeur N et al (2013) The prognostic value of left atrial peak reservoir strain in acute myocardial infarction is dependent on left ventricular longitudinal function and left atrial size. Circ Cardiovasc Imaging 6:26–33. https://doi.org/10.1161/CIRCIMAGING.112.978296
Solomon SD, Biering-Sørensen T (2017) LA strain when ejection fraction is preserved: a new measure of diastolic function? JACC Cardiovasc Imaging 10:744–746. https://doi.org/10.1016/j.jcmg.2016.09.018
Biering-Sørensen T, Christensen LM, Krieger DW et al (2014) LA emptying fraction improves diagnosis of paroxysmal AF after cryptogenic ischemic stroke: results from the SURPRISE study. JACC Cardiovasc Imaging 7:962–963. https://doi.org/10.1016/j.jcmg.2014.02.003
Johansen ND, Biering-Sørensen T, Jensen JS, Mogelvang R (2017) Diastolic dysfunction revisited: a new, feasible, and unambiguous echocardiographic classification predicts major cardiovascular events. Am Heart J 188:136–146. https://doi.org/10.1016/j.ahj.2017.03.013
Voigt J-U, Pedrizzetti G, Lysyansky P et al (2015) Definitions for a common standard for 2D speckle tracking echocardiography: consensus document of the EACVI/ASE/Industry Task Force to standardize deformation imaging. Eur Heart J Cardiovasc Imaging 16:1–11. https://doi.org/10.1093/ehjci/jeu184
Olsen FJ, Bertelsen L, de Knegt MC et al (2016) Multimodality cardiac imaging for the assessment of left atrial function and the association with atrial arrhythmias. Circ Cardiovasc Imaging 9:e004947. https://doi.org/10.1161/CIRCIMAGING.116.004947
Donal E, Lip GYH, Galderisi M et al (2016) EACVI/EHRA expert consensus document on the role of multi-modality imaging for the evaluation of patients with atrial fibrillation. Eur Heart J Cardiovasc Imaging 17:355–383. https://doi.org/10.1093/ehjci/jev354
Pagola J, González-Alujas T, Flores A et al (2014) Left atria strain is a surrogate marker for detection of atrial fibrillation in cryptogenic strokes. Stroke 45:e164–e166. https://doi.org/10.1161/STROKEAHA.114.005540
Kuppahally SS, Akoum N, Burgon NS et al (2010) Left atrial strain and strain rate in patients with paroxysmal and persistent atrial fibrillation: relationship to left atrial structural remodeling detected by delayed-enhancement MRI. Circ Cardiovasc Imaging 3:231–239. https://doi.org/10.1161/CIRCIMAGING.109.865683
Marrouche NF, Wilber D, Hindricks G et al (2014) Association of atrial tissue fibrosis identified by delayed enhancement MRI and atrial fibrillation catheter ablation: the DECAAF study. JAMA 311:498–506. https://doi.org/10.1001/jama.2014.3
Higuchi K, Akkaya M, Akoum N, Marrouche NF (2014) Cardiac MRI assessment of atrial fibrosis in atrial fibrillation: implications for diagnosis and therapy. Heart 100:590–596. https://doi.org/10.1136/heartjnl-2013-303884
Choi H-M, Yoon YE, Oh I-Y et al (2019) Global left atrial strain as a predictor of silent atrial fibrillation following dual chamber cardiac implantable electronic device implantation. JACC Cardiovasc Imaging 11:1537–1539. https://doi.org/10.1016/j.jcmg.2017.12.013
Leong DP, Joyce E, Debonnaire P et al (2017) Left atrial dysfunction in the pathogenesis of cryptogenic stroke: novel insights from speckle-tracking echocardiography. J Am Soc Echocardiogr 30:71–79.e1. https://doi.org/10.1016/j.echo.2016.09.013
Cameli M, Sparla S, Losito M et al (2016) Correlation of left atrial strain and doppler measurements with invasive measurement of left ventricular end-diastolic pressure in patients stratified for different values of ejection fraction. Echocardiography 33:398–405. https://doi.org/10.1111/echo.13094
Smiseth OA (2018) Evaluation of left ventricular diastolic function: state of the art after 35 years with Doppler assessment. J Echocardiogr 16:55–64. https://doi.org/10.1007/s12574-017-0364-2
Yao G-H, Vallurupalli N, Cui J et al (2011) Allometric model improves scaling of left atrial size in obese population: the use of body weight containing variables is challenged. Echocardiography 28:253–260. https://doi.org/10.1111/j.1540-8175.2010.01337.x
Russo C, Jin Z, Sera F et al (2015) Left ventricular systolic dysfunction by longitudinal strain is an independent predictor of incident atrial fibrillation: a community-based cohort study. Circ Cardiovasc Imaging 8:e003520. https://doi.org/10.1161/CIRCIMAGING.115.003520
Bruun Pedersen K, Madsen C, Sandgaard NCF et al (2019) Left atrial volume index and left ventricular global longitudinal strain predict new-onset atrial fibrillation in patients with transient ischemic attack. Int J Cardiovasc Imaging 35:1277–1286. https://doi.org/10.1007/s10554-019-01586-w
Khan UA, de Simone G, Hill J et al (2013) Depressed atrial function in diastolic dysfunction: a speckle tracking imaging study. Echocardiography 30:309–316. https://doi.org/10.1111/echo.12043
Skaarup KG, Christensen H, Høst N et al (2016) Diagnosing paroxysmal atrial fibrillation in patients with ischemic strokes and transient ischemic attacks using echocardiographic measurements of left atrium function. Am J Cardiol 117:91–99. https://doi.org/10.1016/j.amjcard.2015.10.022
Olsen FJ, Jørgensen PG, Møgelvang R et al (2016) Predicting paroxysmal atrial fibrillation in cerebrovascular ischemia using tissue doppler imaging and speckle tracking echocardiography. J Stroke Cerebrovasc Dis 25:350–359. https://doi.org/10.1016/j.jstrokecerebrovasdis.2015.10.004
Sanchis L, Montserrat S, Obach V et al (2016) Left atrial function is impaired in some patients with stroke of undetermined etiology: potential implications for evaluation and therapy. Rev Espanola Cardiol Engl Ed 69:650–656. https://doi.org/10.1016/j.rec.2015.11.033
Singh A, Medvedofsky D, Mediratta A et al (2019) Peak left atrial strain as a single measure for the non-invasive assessment of left ventricular filling pressures. Int J Cardiovasc Imaging 35:23–32. https://doi.org/10.1007/s10554-018-1425-y
Galderisi M, Donal E, Magne J et al (2018) Rationale and design of the EACVI AFib Echo Europe Registry for assessing relationships of echocardiographic parameters with clinical thrombo-embolic and bleeding risk profile in non-valvular atrial fibrillation. Eur Heart J Cardiovasc Imaging 19:245–252. https://doi.org/10.1093/ehjci/jex322
Acknowledgements
We would like to express our sincere gratitude to our late professor, Dr. Jan Skov Jensen, for his contribution to this paper.
Funding
FJO was financially supported by Herlev & Gentofte Hospital’s internal funds and the Danish Heart Foundation (Grant No.: 18-R125-A8534-22083). TBS was supported by the Fondsbørsvekselerer Henry Hansen og Hustrus Hovedlegat.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
JHS has received research grants and speaker fee from Medtronic. HC has received speaker fee from Medtronic, MSD and Bayer. HC also serves on the internal advisory board for Boehringer-Ingelheim. The SURPRISE study received financing form the capital region of Denmark, Medtronic Denmark, Sophus Jacobsens and the Arvid Nilssons foundation. The sponsors had no role in the study design, data collection, data analysis, data interpretation, or writing of the manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Olsen, F.J., Christensen, L.M., Krieger, D.W. et al. Relationship between left atrial strain, diastolic dysfunction and subclinical atrial fibrillation in patients with cryptogenic stroke: the SURPRISE echo substudy. Int J Cardiovasc Imaging 36, 79–89 (2020). https://doi.org/10.1007/s10554-019-01700-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10554-019-01700-y