Abstract
In the aftermath of 2019’ tropical cyclones Idai and Kenneth, we assessed ground beetle communities of the Gorongosa National Park (GNP) in Mozambique. The influence of habitat shelters, namely the buffering role of closed microhabitat conditions on Alpha diversity and community trait values during a long period of drought, was evaluated across the main habitat types of the park: miombo forests, mixed forests, transitional forests, and grasslands (open savannas). These habitat types comprised a distance gradient in relation to lake Urema, in the center of GNP. Miombo forests were farther from the lake while grasslands and transitional forests were in the floodplain area. Ground beetle communities were sampled using pitfall traps set up at 25 sites of each habitat type along an environmental gradient of tree canopy cover during the last twenty days of the dry season. Higher species richness of ground beetles was found in closed habitat shelters along the distance gradient to lake Urema. A higher functional diversity was also found along the canopy gradient, with larger sized and wingless species being more abundant in closed habitats than in open areas. This result highlights the crucial role of habitat closedness in the protection of sensitive ground beetles. In particular, the buffer effect of tree canopy and the protection of the understory plants and the litter layer was critical for the survival of hygrophilous species and will be key in conservation strategies to face climate aridification and habitat fragmentation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Climate change events related to El Niño/southern oscillation (ENSO) are a major driver of climatic variability in tropical biomes in Southern Africa such as the savannas in Mozambique (Kirichenko-Babko et al. 2020; Ariza et al. 2021). Particularly, the increase in long-lasting periods of drought is likely to severely affect African biodiversity due to increasing aridification (Engelbrecht et al. 2007; Massad and Castigo 2016; Mbokodo et al. 2020). Increasing drought intensity and frequency lead to tree dieback and soil desertification (Khaine and Woo 2015; Corlett 2016; Mbokodo et al. 2020), and extends beyond emblematic mammals, by impacting soil invertebrates which are pivotal for ecosystem functioning.
Among the various functional groups of soil communities, ground beetles (Coleoptera, Carabidae) serve as important ecological and biodiversity indicators of habitat quality and environmental changes (Rainio and Niemelä 2003; Koivula 2011). Specific carabid traits, namely body size and hind wing typology, reflect the species sensitivity to environmental disturbance, acting as proxies for species resilience (e.g., Ribera et al. 2001; Kotze and O’Hara 2003; Lambeets et al. 2009; Brooks et al. 2012; Nolte et al. 2017, 2019). Previous studies have found that species with smaller body size and fully developed wings can more easily escape habitat disturbance and quickly recolonize new areas (Ribera et al. 2001; Gobbi and Fontaneto 2008; Pizzolotto 2009; Gerisch 2011). In addition, while most ground beetle species are hygrophilous (Rainio 2013; Zajicek et al. 2021), some are better adapted to periodic floods (Kolesnikov et al. 2012; Kirichenko-Babko et al. 2020). Thus, the expected longer-lasting periods of drought, alternated by annual flooding, are expected to be key in determining ground beetle diversity and community changes. The outcomes will greatly depend on their dispersal ability and the availability of natural refuge habitats during desiccation and/or harsher flooding episodes.
Previous studies in Mediterranean and temperate systems have shown that ground beetle species diversity is strongly influenced by the presence of closed forest patches and shrub areas that act as shelter/refuge habitats in agroforestry landscapes (e.g., MacLeod et al. 2004; Martins da Silva et al. 2011, 2017; Zou et al. 2019). Specifically, forested shelter/refuge patches were key in supporting sensitive ground beetles, namely apterous or brachypterous species with larger body size, since they are more sensitive to changing climatic conditions and hence, require more protection (Blake et al. 1994; Brose 2003; Warnaffe and Lebrun 2004; Martins da Silva et al. 2008, 2017; Schirmel et al. 2015; Eyre et al. 2016; Wang et al. 2018). Conversely, small species with long wings are usually dominant in highly trampled open-habitat types (Blake et al. 1994; Grandchamp et al. 2000; Wang et al. 2018; Ariza et al. 2021), such as several European floodplains (e.g., Bates et al. 2006; Lambeets et al. 2009; Gerisch 2011). Yet, no study has addressed how local habitat shelters may influence ground beetle diversity and community trait patterns in dynamic tropical ecosystems, which are threatened by soil aridification due to climate change. In this context, the Gorongosa National Park (GNP) in Mozambique is an interesting case study, as comprises a dynamic ecosystem with a gradient of habitat types, from typical miombo forests to floodplain grasslands, where periodic (annual) flooding controls interannual variations in diversity, survival, and seasonal community patterns (Stalmans et al. 2019). Yet, GNP landscape configuration may change in the next decades due to deforestation and soil desertification (Herrero et al. 2020; Mbokodo et al. 2020), and for this reason, specific landscape features, such as tree canopy and soil ground cover will be key in influencing carabid survival and community patterns, particularly during the expected longer lasting periods of drought.
Therefore, in this study, we assessed ground beetle communities at the main GNP habitat types, covering a wide range of environmental conditions, from the typical miombo forests to transitional and grassland habitats in the floodplains. We focused on the role of microhabitat structure as shelters for ground beetles during intense drought periods. We hypothesized that, by providing more stable microhabitat conditions during the dry season, habitat shelters will promote higher ground beetle Alpha-diversity across a tree canopy cover gradient (H1). Accordingly, larger-sized apterous/brachypyterous species will be more sensitive to drought as they are more dependent on habitat shelters than the smaller species with higher dispersal abilities. In addition, we hypothesized that the number of sensitive carabid species will increase with the increasing distance of miombo habitats to the floodplain (H2), reflecting community patterns primarily shaped by the flooding gradient.
Materials and methods
Study site
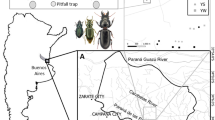
Gorongosa National Park (GNP) occupies approximately 4000 km2 in the Sofala Province, central Mozambique, at the southernmost end of Africa’s Great Rift Valley (18°58′04.84″ S, 34°21′41.64″ E) (Stalmans et al. 2019). The wet season in Mozambique normally lasts from November to April and mean annual rainfall within the Rift is 700–900 mm (Stalmans et al. 2019). Average annual temperatures range between 15 oC in the dry season and 30 oC in the wet season (Herrero et al. 2020).
GNP encompasses a vast ecosystem diversity, including Afromontane rainforest and riverine forest at Mount Gorongosa (> 700 m elevation), as well as wooded savannas and open floodplain at lower elevations (Massad and Castigo 2016). The plateaus encircling the park’s central area are covered with a closed-canopy savanna named “miombo” due to the dominant tree, a member of the genus Brachystegia. In the central part of the park (“lower Gorongosa”) savannas range from “open” savanna (floodplain grassland) to “mixed” savanna (grass-shrub-open tree: transitional forest), as well as “closed” savannas dominated by different tree species, i.e. mixed forests and miombo forests (Herrero et al. 2020). Hence, GNP low plateau comprises four main habitat types, from the woodland patches of miombo or mixed forests to the more open savannas, namely transitional forests, and floodplains near lake Urema (Stalmans et al. 2019).
Sampling design, carabid sample processing and record of species traits
Field work was carried out during October-November of 2019, following Cyclone Idai which occurred in March of the same year. In 2019 the dry season lasted till mid-November. The first mild rainfall started on November 14th (40 mm) and heavy rain only fell after November 20th (110–117 mm) (Martins da Silva et al. 2023a). Therefore, from October 25th to November 15th, pitfall traps were set to sample ground beetles, with beetle collections conducted each ten-day period.
We selected 100 sampling sites, 25 per each main habitat type (miombo forest, mixed forest, transitional forest, and open savanna) with a minimum distance between any two sites greater than 1 km (detailed sampling design in Serrano et al. 2023). This selection allowed us to analyze an environmental gradient, in terms of tree canopy cover, along with geographical distance to the lake Urema, located at the center of the park (Martins da Silva et al. 2023a, b; Serrano et al. 2023). Sampling sites within each of these habitat types also varied in terms of microhabitat conditions due to differences in tree canopy cover and local vegetation structure (Sect. 2.3 below).
At each sampling site, sampling was carried out using three pitfall traps (sub-samples) arranged in a triangle, with 5 m between pitfalls, summing a total of 600 pitfalls, although part of them (44) were destroyed by animals or fire during the dry season.
Pitfall traps consisted of plastic vials of 10 cm diameter filled with 5% ethylene glycol. The traps were covered with a plastic lid (10 cm diameter) fixed a few centimeters aboveground to minimize bycatches of small vertebrates.
Sampled beetles were preserved in absolute ethanol and transported to the entomological laboratory at the Centre for Ecology, Evolution and Environmental Changes, at the University of Lisbon (Portugal), for taxonomic identification of all specimens. A total of 62 species belonging to 32 genera and 17 tribes were taxonomically identified, resulting in a ground beetle dataset provided by Serrano et al. (2023). For pragmatic reasons we followed here the recent classification by Lorenz (2021), which includes tiger beetles and ground beetles in the Carabidae family. Information on species traits was collected from literature and by direct morphological measurement of the specimens.
We selected two species traits, average body size, i.e. species length (measured in mm) and wing typology, i.e., whether they are macropterous (fully developed hind wings) or brachypterous/apterous species, since the use of these traits proved to be informative for ecological studies (e.g., Ribera et al. 2001; Nolte et al. 2017). All data on species taxonomy and traits are provided in Martins da Silva et al. (2023a).
Environmental variables
Martins da Silva et al. (2023b) noted substantial variability in prey availability (e.g. Collembola) and abiotic conditions within each GNP habitat type, mainly associated with the understory structure (amount of herbaceous plants, shrubs and litter) and canopy cover. This intra-habitat variation can potentially influence ground beetle populations, namely due to differences in moisture conditions (Martins da Silva et al. 2023a), and niche availability or food resource (Magura and Lövei 2019; Boutaud et al. 2022) which are dependent on the buffer role of habitat shelters (e.g. Marañón et al. 2009; Rossetti et al. 2015; Widenfalk et al. 2015). As a proxy of habitat shelter, different local environmental variables related to the vegetation cover were selected at each sampling site, namely canopy area (CA - calculated as a percentage), as well as the levels of bare soil (BS), herbaceous cover (HC), herbaceous height (HH), shrub cover (SC), and litter (L), which were assessed on a scale from 0 to 3 (Appendix Table S1).
We then calculated the Shelter Index (SI) by attributing a weight to each of these variables:
where \(\frac{HC\times HH}{3}\) corresponds to the formula for Herbaceous Protection, calculated to minimize the impact of herbaceous height while emphasizing the influence of herbaceous cover. Based on the previous Collembola diversity information (Martins da Silva et al. 2023b) higher weight (4) was attributed to canopy area (CA) since it provides shadow and leaf litter material, and hence is a major driver of habitat protection against desiccation and of niche availability in woodlands and semi-open habitats (Rossetti et al. 2015; Magura and Lövei 2019; Boutaud et al. 2022; Martins da Silva et al. 2023b). Litter cover (L) was the second most relevant parameter, with twice the weight of herbaceous (HC) and shrub covers (SC), since its contribution is twofold, by directly protecting against soil desiccation but also supporting more food resources through a positive impact on microarthropods (Martins da Silva et al. 2023b). In contrast, the bare soil (BS) has a negative impact on the index as it reduces habitat capacity to retain moisture and refuge against predators (Brose 2003; Wang et al. 2018).
Another potentially important factor influencing ground beetle population survival and community patterns is the distance from lake Urema (distance gradient), which was determined using a GPS device. The two main environmental factors, i.e., habitat Shelter Index and geographical distance to lake Urema, were not correlated (Pearson: r = 0.103, p = 0.382; Fig. 1). Miombo forest sampling sites were significantly more distant from lake Urema than transitional forests and grasslands (F = 164.2, p < 0.001; Fig. 1, Table S1).
Data analyses
Pitfall sub-samples from each sampling point were pooled before data analysis. Alpha-diversity (abundance) was calculated using R BAT package, version 2.9.5 (Cardoso et al. 2015), implemented in the statistical software R version 4.1.3 (R Core Team 2022). Alpha-functional diversity (Alpha_FD) for average beetle body size (Alpha-FD_Body_size) and wing typology (Alpha-FD_Wings) was calculated using FD package, version 1.0-12.3 (Laliberté et al. 2014).
Before testing the influence of habitat shelter through the composite Shelter Index, the single role of each environmental predictor on ground beetle activity-density (N), Alpha-diversity (S) and Alpha-functional diversity (Alpha-FD) of carabid body size (Alpha-FD_Body_size) and wing typology (Alpha FD_Wings), as well as the responses of trait values of beetle body size (Trait-values_Body_size) and wing typology (Trait-values_Wings), was checked with generalized linear models (GLM). For each response variable (N, S, Alpha-FD_Body_size, Alpha-FD_Wings, Trait-values_Body_size, Trait-values_Wings), GLMs were fitted with different error distribution families (N – “GLM: Negative Binomial”, S – “GLM: Poisson”, FD Alpha_body_size – “GLM: Inverse-Gamma”, FD-Alpha_wing_typology – “GLM: Inverse-Gamma”, Trait-values_Body_size – “GLM: Inverse Gamma”, Trait-values_ Wings – “GLM: Binomial”).
Subsequently, we used generalized linear mixed-models (GLMM), fitted by maximum likelihood (Laplace approximation), with habitat type (i.e., grasslands, transitional forests, mixed forests, miombo forests) as a random factor, to test the effects of Shelter Index (SI) and the distance to lake Urema (floodplain center) on the variation of ground beetle N, S, Alpha-FD_Body_size, Alpha FD_Wings, Trait-values_Body_size and Trait-values_Wings. In line with the GLM models, each GLMM response variable was fitted with the correspondent error distribution family (N – “GLMM: Negative Binomial”, S – “GLMM: Poisson”, FD Alpha_body_size – “GLMM: Inverse-Gamma”, FD-Alpha_wing_typology – “GLMM: Inverse-Gamma”, Trait-values_Body_size – “GLMM: Inverse Gamma”, Trait-values_ Wings – “GLMM: Binomial”). GLMM analyses were performed using the “lme4” R package, version 1.1–33 (Bates et al. 2015).
Results
Individual GLM models showed a significantly positive canopy effect (through the variables canopy area and litter cover) on ground beetle activity-density (N), Alpha-diversity (S), Alpha-FD of carabid body size and wing typology (Table 1). The amount of shrub cover was also related to higher N values, in contrast to the herb cover and herb height, which were negatively related to N and S parameters (Table 1). Moreover, larger ground beetles and a higher number of brachypterous/apterous species were associated to forested sites (higher canopy area and litter cover), while macropterous beetles were positively associated to sites with higher amount of herb cover (Table 1).
With the GLMM models comparing the effects of habitat shelter vs. site distance to lake Urema, we observed that higher N values of ground beetles were significantly explained by the distance gradient, while the shelter gradient was not important across the GNP landscape (Table 2). As for the ground beetle S values, both factors were significant, although the Shelter Index effect was only borderline significant, with a positive effect on S while the distance factor showed a strong negative effect (Table 2).
Ground beetle functional diversity (Alpha-FD), in terms of species’ average body size only responded to the shelter gradient across the GNP habitat types, while Alpha-FD of wing typology was not significantly explained by the Shelter Index, and neither by the distance to lake Urema (Table 2).
Community trait values of carabid body size were influenced by both environmental gradients, as significantly bigger-sized species occurred in more closed habitat shelters (Table 2; Fig. 2a), as well as in habitats farther from the floodplains (Fig. 2b).
Higher habitats shelter also harbored a significantly higher number of apterous/brachypterous species compared to the open habitats, which were richer in beetles with higher dispersal power, i.e., macropterous species (Table 2; Fig. 3). This same pattern was observed for the distance gradient, but it was not significant (Table 2).
Discussion
Ground beetle richness along the tree canopy gradient
Climate change is expected to drive changes to future ground beetle diversity patterns in tropical ecosystems, mainly due to the longer periods of drought and severe flooding events related to the El Niño/southern oscillation (e.g., Ariza et al. 2021; Peterson et al. 2021). In this study, our main hypothesis was that habitat closedness was a major factor governing ground beetle diversity by protecting more sensitive species, namely bigger sized species with lower dispersal ability. Supporting this hypothesis, higher species richness of ground beetles was associated to habitat shelters along the distance gradient from miombo forest to the floodplains. This result highlights the important role of habitat closedness, namely the buffer effect of tree canopy and understory structure (e.g. litter layers). This is highly important for the protection of sensitive taxa such as those hygrophilous species (e.g. Abacetus percoides Fairmaire, 1868; Metagonum insolitum Péringuey. 1904) that need habitat shelters to withstand climatic extremes (Rainio 2013; Zajicek et al. 2021). In fact, most ground beetles benefit from moist environments promoted by canopy shading and thicker litter layers, enabling a higher number of species to co-exist (Niemelä 1993; Magura and Lövei 2019; Marrec et al. 2021; Zajicek et al. 2021). Contrarily, in more exposed habitat areas, ground beetle communities tend to be dominated by those few species (e.g. Microlestes zambezianus Mateu. 1960) that are more tolerant to drought (e.g., Brandmayr et al. 1983; Tsafack et al. 2020). This result is in line with earlier studies on Mediterranean and temperate ecosystems, which emphasized the importance of forested habitats as important refuges in grassland-forest mosaics to ensure biodiversity conservation of ground beetles (Romero-Alcaraz and Ávila 2000; Woodcock et al. 2005; Zamora et al. 2007; Martins da Silva et al. 2011).
Higher richness and activity-density were found in miombo habitats farther from lake Urema, i.e., more distant from the floodplain area. Previous authors have shown that ground-beetles’ activity and dynamics are driven by temporal flooding (Lambeets et al. 2008; Kirichenko-Babko et al. 2020). Carabid communities inhabiting floodplains are adapted to survive the periodic flooding episodes, although only a small number of species can survive in the inundated habitat (Tamm 1984, 1986; Zerm and Adis 2001; Kolesnikov et al. 2012). Except for a few tolerant species (e.g., Adis 1982; Amorim et al. 1997), most ground beetles avoid flooding by dispersing to adjacent, non-inundated habitats (Zerm and Adis 2001; Andersen 2005; Kolesnikov et al. 2012), i.e., the miombo forest in the case of the GNP. In contrast to the eurytopic species, and those with higher dispersal ability, the dynamics of stenotopic species may be confined to sheltered habitats located farther from the disturbance source, i.e., those associated with the miombo forested habitats that are significantly less affected by annual inundation (Kirichenko-Babko et al. 2020). Hence, the alternative hypothesis that ground beetle communities could be also shaped by the event of seasonal flooding is also supported by the results of this study.
Response of carabid functional diversity and community traits
According to our results, higher levels of species richness in habitat shelters were reflected in higher functional diversity in terms of species body size and wing morphology, with bigger sized and brachypterous/apterous species being mostly found in closed habitats in relation to open areas. Previous studies showed that wingless species with a larger body size are typically more sensitive to disturbance (Halme and Niemela 1993; Koivula 2002; Jelaska and Durbešic 2009; Wamser et al. 2011; Brandmayr and Pizzolotto 2016; Wang et al. 2018). Body size and dispersal ability respond to local habitat configuration, particularly vegetation structure and canopy cover, and are the main factors explaining community patterns in European agricultural and forested ecosystems (e.g., Brose 2003; Woodcock et al. 2005; Vandewalle et al. 2010; Spake et al. 2016; Martins da Silva et al. 2017). In this study, habitat shelters promoted by forest patches supported larger species with poorer dispersal abilities, as they require more stable habitat conditions (Warnaffe and Lebrun 2004; Martins da Silva da Silva et al. 2008, 2017; Schirmel et al. 2015; Eyre et al. 2016) and protection against natural predators (Blake et al. 1994; Brose 2003; Wang et al. 2018). Supporting these findings, and in line with our predictions, larger ground beetles and species with less dispersal potential have likely found refuge in closed habitats with denser vegetation cover that provided more suitable microclimate conditions, namely moisture, across the GNP landscape. Conversely, small species with long wings are usually dominant in disturbed and exposed open habitats (Blake et al. 1994; Grandchamp et al. 2000; Wang et al. 2018; Ariza et al. 2021). Previous studies have shown that smaller species with fully developed wings, i.e., with greater dispersal capabilities, are better adapted to disturbances and/or more able to escape and colonize new areas (Blake et al. 1994; Grandchamp et al. 2005; Wang et al. 2018). During periods of drought, these species not only benefit from closed habitats as shelter areas interspersed in the landscape due to their better dispersal abilities (Jelaska and Durbešic 2009; Eyre et al. 2016; Martins da Silva et al. 2017); they also benefit from finding refuges in ground slits, shrubs, and small tussocks in grasslands (MacLeod et al. 2004; Schirmel et al. 2015; Ariza et al. 2021).
In a previous study, Martins da Silva et al. (2023a) compared ground beetle communities in dry and wet seasons, and concluded that wet conditions, provided by rainfall after a long period of drought, were crucial to support species richness, particularly for larger species with poorer dispersal ability. In this study, we can conclude that during the drought period beetle survival and activity strongly depends on the provision of habitat shelters. This finding is relevant considering the anticipated increase in extreme climatic events in the next decades, namely hurricanes/flooding (e.g., Lambeets et al. 2008; Lafage and Pétillon 2016; Litavský et al. 2021) and longer-lasting periods of drought (e.g., Šustek et al. 2017; Šiška et al. 2020; Ariza et al. 2021) with consequent landscape changes in tropical ecosystems. These disruptive events are likely to affect ground beetle species, particularly those more moisture dependent, and favor more eurytopic species with smaller size and higher dispersal ability (Ariza et al. 2021; Zajicek et al. 2021). In contrast, larger-sized and wingless species will probably be more vulnerable to extinction in response to future climate aridification and landscape homogenization and habitat changes (Kirichenko-Babko et al. 2020; Ariza et al. 2021). Habitat shelters providing microhabitat conditions to buffer arid conditions during drought will be essential for ground beetle diversity conservation and should be integrated in future conservation activities.
Recommendations for land management
Our results highlighted the importance of habitat shelters for ground beetle diversity in the Gorongosa National Park, namely by supporting larger-sized sensitive species during a long-lasting drought season. Thus, conservation initiatives and management planning need to take into consideration the key role of closed habitat patches, provided by trees and shrubs, as essential buffers for ground beetle protection against environmental disturbance and climatic extremes. Land management towards the maintenance of woodland patches is critical considering the degradation of Gorongosa miombo expected in the long term, due to human activities (e.g. agriculture, fires) but also to the predicted longer lasting periods of drought (Massad and Castigo 2016; Herrero et al. 2020). Besides the heuristic importance of ground beetle diversity conservation, they play a key role in tropical food webs (Kotze et al. 2011), for instance as prey for several birds, reptiles, and mammals (Brose 2003). Hence, biodiversity protection of ground beetles, and especially the larger-sized species, will also contribute to higher habitat quality and life support of emblematic fauna of the GNP, also threatened by increasing environmental instability and climate extremes that are predicted to increase in Mozambique in the next decades.
Data availability
Dataset that supported the findings of this study is provided in a reference within the manuscript.
References
Adis J (1982) Eco-entomological observations from the Amazon: II. Carabids are adapted to inundation-forests. Coleopt Bull 36:439–440. https://www.jstor.org/stable/i384412
Amorim MA, Adis J, Paarmann W (1997) Ecology and adaptations of the tiger beetle Pentacomia egregia (Chaudoir) (Cicindelinae: Carabidae) to central amazonian floodplains. Ecotropica 3:71–82
Andersen JM (2005) Riparian beetles, a unique, but vulnerable element in the fauna of Fennoscandia. Biodiv Conserv 14:3497–3524. https://doi.org/10.1007/s10531-004-0825-6
Ariza GM, Jácome J, Kotze DJ (2021) Carabid beetles of tropical dry forests display traits that cope with a harsh environment. Int J Trop Insect Sci 41:3011–3021. https://doi.org/10.1007/s42690-021-00493-9
Bates AJ, Sadler JP, Fowles AP (2006) Condition-dependent dispersal of a patchily distributed riparian ground beetle in response to disturbance. Oecologia 150:50–60. https://doi.org/10.1007/s00442-006-0508-y
Bates D, Maechler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67:1–48. https://doi.org/10.18637/jss.v067.i01
Blake S, Foster GN, Eyre MD, Luff ML (1994) Effects of habitat type and grassland management practices on the body size distribution of carabid beetles. Pedobiologia 38:502–512. https://www.jstor.org/stable/23735413
Boutaud E, Nolte D, Harry I (2022) Conservation value of semi-open habitats for ground beetles (Coleoptera: Carabidae, Cicindelidae) in Central Europe. Biodivers Conserv 31:1469–1489. https://doi.org/10.1007/s10531-022-02402-z
Brandmayr P, Pizzolotto R (2016) Climate change impact on carabid beetles. Period biol 118:147–162. https://doi.org/10.18054/pb.2016.118.3.4062
Brandmayr P, Colombetta G, Polli S (1983) Waldcarabiden Des Triester Karstes als Indikatoren Des Makroklimatischen Uebergangs Vom Kontinentalen Europa Zur Mediterraneis. Zool Jb Syst 110:201–220
Brooks DR, Bater JE, Clark SJ, Monteith DT, Andrews C, Corbett SJ, Beaumont DA, Chapman JW (2012) Large carabid beetle declines in a United Kingdom monitoring network increases evidence for a widespread loss in insect biodiversity. J Appl Ecol 49:1009–1019. https://doi.org/10.1111/j.1365-2664.2012.02194.x
Brose U (2003) Bottom-up control of carabid beetle communities in early successional wetlands: mediated by vegetation structure or plant diversity? Oecologia 135:407–413. https://doi.org/10.1007/s00442-003-1222-7
Cardoso P, Rigal F, Carvalho JC (2015) BAT biodiversity assessment tools, an r package for the measurement and estimation of alpha and beta taxon, phylogenetic and functional diversity. Methods Ecol Evol 6:232–236. https://doi.org/10.1111/2041-210X.12310
Corlett RT (2016) The impacts of droughts in tropical forests. Trends Plant Sci. 21:584—593https://doi.org/10.1016/j.tplants.2016.02.003
Engelbrecht B, Comita L, Condit R, Kursar TA, Tyree MT, Turner BL, Hubbel SP (2007) Drought sensitivity shapes species distribution patterns in tropical forests. Nature 447:80–82. https://doi.org/10.1038/nature05747
Eyre MD, McMillan SD, Critchley CNR (2016) Ground beetles (Coleoptera Carabidae) as indicators of change and pattern in the agroecosystem: longer surveys improve understanding. Ecol Ind 68:82–88. https://doi.org/10.1016/j.ecolind.015.11.009
Gerisch M (2011) Habitat disturbance and hydrological parameters determine the body size and reproductive strategy of alluvial ground beetles. In: Kotze DJ, Assmann T, Noordijk J, Turin H, Vermeulen R (Eds) Carabid Beetles as Bioindicators: Biogeographical, Ecological and Environmental Studies. ZooKeys 100:353–370. https://doi.org/10.3897/zookeys.100.1427
Gobbi M, Fontaneto D (2008) Biodiversity of ground beetles (Coleoptera: Carabidae) in different habitats of the Italian Po lowland. Agric Ecosyst Environ 127:273–276. https://doi.org/10.1016/j.agee.2008.04.011
Grandchamp AC, Niemelä J, Kotze J (2000) The effects of trampling on assemblages of ground beetles (Coleoptera, Carabidae) in urban forests in Helsinki, Finland. Urban Ecosyst 4:321–332. https://doi.org/10.1023/A:1015707916116
Grandchamp AC, Bergamini A, Stofer S, Niemelä N, Duelli P, Scheidegger C (2005) The influence of grassland management on ground beetles (Carabidae, Coleoptera) in Swiss montane meadows. Agric Ecosyst Environ 110:307–317. https://doi.org/10.1016/j.agee.2005.04.018
Halme E, Niemelä J (1993) Carabid beetles in fragments of coniferous forest. Ann Zool Fenn 30:17–30. https://doi.org/10.2307/23735353
Herrero H, Waylen P, Southworth J, Khatami R, Yang D, Child B (2020) A healthy park needs healthy vegetation: the story of Gorongosa National Park in the 21st century. Remote Sens 12:476. https://doi.org/10.3390/rs12030476
Jelaska LŠ, Durbešić P (2009) Comparison of the body size and wing form of carabid species (Coleoptera: Carabidae) between isolated and continuous forest habitats. Ann Soc Entomol Fr 45:327–338. https://doi.org/10.1080/00379271.2009.10697618
Khaine I, Woo SY (2015) An overview of interrelationship between climate change and forests. For Sci Technol 11:11–18. https://doi.org/10.1080/21580103.2014.932718
Kirichenko-Babko M, Danko Y, Musz-Pomorksa A, Widomski MK, Babko R (2020) The impact of climate variations on the structure of ground beetle (Coleoptera: Carabidae) assemblage in forests and wetlands. Forests 11:1074. https://doi.org/10.3390/f11101074
Koivula M (2002) Alternative harvesting methods and boreal carabid beetles (Coleoptera, Carabidae). For Ecol Manag 167:103–121https://doi.org/10.1016/S0378-1127(01)00717-4
Koivula MJ (2011) Useful model organisms, indicators, or both? Ground beetles (Coleoptera, Carabidae) reflecting environmental conditions. In: Kotze DJ, Assmann T, Noordijk J, Turin H, Vermeulen R (eds) Carabid Beetles as Bioindicators: Biogeographical, Ecological and Environmental Studies. ZooKeys 100:287–317. https://doi.org/10.3897/zookeys.100.1533
Kolesnikov F, Karamyan AN, Hoback WW (2012) Survival of ground beetles (Coleoptera: Carabidae) submerged during floods: field and laboratory studies. Eur J Entomol 109:71–76. https://doi.org/10.14411/eje.2012.009
Kotze DJ, O’Hara RB (2003) Species decline—but why? Explanations of carabid beetle (Coleoptera, Carabidae) declines in Europe. Oecologia 135:138–148. https://doi.org/10.1007/s00442-002-1174-3
Kotze DJ, Brandmayr P, Casale A, Dauffy-Richard E, Dekoninck W, Koivula MJ, Lövei GL, Mossakowski D, Noordijk J, Paarmann W, Pizzolotto R, Saska P, Schwerk A, Serrano J, Szyszko J, Taboada A, Turin H, Venn S, Vermeulen R, Zetto T (2011) Forty years of carabid beetle research in Europe – from taxonomy, biology, ecology and population studies to bioindication, habitat assessment and conservation. ZooKeys 100:55–148. https://doi.org/10.3897/zookeys.100.1523
Lafage D, Pétillon J (2016) Relative importance of management and natural flooding on spider, carabid and plant assemblages in extensively used grasslands along the Loire. Basic Appl Ecol 17:535–545 https://doi.org/10.1016/j.baae.2016.04.002
Laliberté E, Legendre P, Shipley B (2014) FD: measuring functional diversity from multiple traits, and other tools for functional ecology. R package version 1.0-12.3. https://cran.r-project.org/web/packages/FD/FD.pdf
Lambeets K, Vandegehuchte ML, Maelfait J-P, Bonte D (2008) Understanding the impact of flooding on trait-displacements and shifts in assemblage structure of predatory arthropods on river banks. J Animal Ecol 77:1162–1174. https://www.jstor.org/stable/20143299
Lambeets K, Vandegehuchte ML, Maelfait JP, Bonte D (2009) Integrating environmental conditions and functional life-history traits for riparian arthropod conservation planning. Biol Conserv 142:625–637. https://doi.org/10.1016/j.biocon.2008.11.015
Litavský J, Majzlan O, Stašiov S, Svitok M, Fedor P (2021) The associations between ground beetle (Coleoptera: Carabidae) communities and environmental condition in floodplain forests in the Pannonian Basin. Eur J Entomol 118:14–23. https://doi.org/10.14411/eje.2021.002
Lorenz W (2021) Carabcat database. In: Bánki O, Roskov Y, Döring M, Ower G, Hernández Robles DR, Plata Corredor CA, Stjernegaard Jeppesen T, Örn A, Vandepitte L, Hobern D, Schalk P, DeWalt RE, Ma K, Miller J, Orrell T, Aalbu R, Abbott J, Adlard R, Aedo C, Catalogue of Life Checklist (v.03 (08/2021)). https://doi.org/10.48580/dfrdl-3dk
MacLeod A, Wratten SD, Sotherton NW, Thomas MB (2004) Beetle banks’ as refuges for beneficial arthropods in farmland: long-term changes in predator communities and habitat. Agr for Entomol 6:147–154. https://doi.org/10.1111/j.1461-9563.2004.00215.x
Magura T, Lövei GL (2019) Environmental filtering is the main assembly rule of ground beetles in the forest and its edge but not in the adjacent grassland. Insect Sci 26:154–163. https://doi.org/10.1111/1744-7917.12504
Marañón T, Pugnaire FI, Callaway RM (2009) Mediterranean-climate oak savannas: the interplay between abiotic environment and species interactions. Web Ecol 9:30–43. https://doi.org/10.5194/we-9-30-2009
Marrec R, Le Roux V, Martin L, Lenoir J, Brunet J, Cousins SAO, De Smedt P, Deconchat M, Diekmann M, Ehrmann S, Gallet-Moron E, Giffard B, Liira J, Lindgren J, Valdes A, Verheyen K, Wulf M, Decocq G (2021) Multiscale drivers of carabid beetle (Coleoptera: Carabidae) assemblages in small European woodlands. Global Ecol Biogeogr 30:165–182. https://doi.org/10.1111/geb.13208
Martins da Silva P, Aguiar CAS, Niemelä J, Sousa JP, Serrano ARM (2008) Diversity patterns of ground-beetles (Coleoptera: Carabidae) along a gradient of land-use disturbance. Agric Ecosyst Environ 124:270–274. https://doi.org/10.1016/j.agee.2007.10.007
Martins da Silva P, Aguiar CAS, de Faria e Silva I, Serrano ARM (2011) Orchard and riparian habitats enhance ground dwelling beetle diversity in Mediterranean agro-forestry systems. Biodivers Conserv 20:861–872. https://doi.org/10.1007/s10531-010-9987-6
Martins da Silva P, Oliveira J, Ferreira A, Fonseca F, Pereira JA, Aguiar CA, Serrano ARM, Sousa JP, Santos SA (2017) Habitat structure and neighbor linear features influence more carabid functional diversity in olive groves than the farming system. Ecol Ind 79:128–138. https://doi.org/10.1016/j.ecolind.2017.04.022
Martins da Silva P, Carvalho R, Boieiro M, Sousa JP, Serrano ARM (2023a) Long droughts decrease tiger- and ground-beetle’ beta diversity and community body size in savannas of the Gorongosa National Park (Mozambique). J Insect Conserv 27:927–940. https://doi.org/10.1007/s10841-023-00509-4
Martins da Silva P, Bartz M, Mendes S, Boieiro M, Timóteo S, Azevedo-Pereira HMVS, Alves da Silva A, Alves J, Serrano ARM, Sousa JP (2023b) Tree canopy enhances collembola functional richness and diversity across typical habitats of the Gorongosa National Park (Mozambique). Appl Soil Ecol 190:105010. https://doi.org/10.1016/j.apsoil.2023.105010
Massad TJ, Castigo T (2016) Investigating possible effects of climate change on tree recruitment: responses of abundant species to water stress in Gorongosa National Park. S Afr J Bot 106:96–100https://doi.org/10.1016/j.sajb.2016.06.002
Mbokodo I, Bopape M-J, Chikoore H, Engelbrecht F, Nethengwe N (2020) Heatwaves in the future warmer climate of South Africa. Atmosphere 11:712. https://doi.org/10.3390/atmos11070712
Niemelä J (1993) Interspecific competition in ground-beetle assemblages (Carabidae): what have we learned? Oikos 66:325–335. https://doi.org/10.2307/3544821
Nolte D, Schuldt A, Gossner MM, Ulrich W, Assmann T (2017) Functional traits drive ground beetle community structures in central European forests: implications for conservation. Biol Conserv 213:5–12. https://doi.org/10.1016/j.biocon.2017.06.038
Nolte D, Boutaud E, Kotze DJ, Schuldt A, Assmann T (2019) Habitat specialization, distribution range size and body size drive extinction risk in carabid beetles. Biodivers Conserv 28:1267–1283https://doi.org/10.1007/s10531-019-01724-9
Peterson KNR, Browne RA, Erwin TL (2021) Carabid beetle (Coleoptera, Carabidae) richness, diversity, and community structure in the understory of temporarily flooded and non-flooded amazonian forests of Ecuador. Zookeys 1044:831–876. https://doi.org/10.3897/zookeys.1044.62340
Pizzolotto R (2009) Characterization of different habitats on the basis of the species traits and eco-field approach. Oecologica 35:142–148. https://doi.org/10.1016/j.actao.2008.09.004
R Core Team (2022) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. https://www.R-project.org
Rainio J (2013) Seasonal variation of carabid beetle (Coleoptera: Carabidae) abundance and diversity in Ranomafana National Park, Madagascar. J Entomol Zool Stud 1:92–98
Rainio J, Niemelä J (2003) Ground beetles (Coleoptera: Carabidae) as bioindicators. Biodivers Conserv 12:487–506. https://doi.org/10.1023/A:1022412617568
Ribera I, Dolédec S, Downie IS, Foster GN (2001) Effect of land disturbance and stress on species traits of ground beetle assemblages. Ecology 82:1112–1129. https://doi.org/10.1890/0012-9658(2001)082[1112:EOLDAS]2.0.CO;2
Romero-Alcaraz E, Ávila JM (2000) Landscape heterogeneity in relation to variations in epigaeic beetle diversity of a Mediterranean ecosystem. Implications for conservation. Biodivers Conserv 9:985–1005. https://doi.org/10.1023/A:1008958720008
Rossetti I, Bagella S, Cappai C, Caria MC, Lai R, Roggero PP, Martins da Silva P, Sousa JP, Querner P, Seddaiu G (2015) Isolated cork oak trees affect soil properties and biodiversity in a Mediterranean wooded grassland. Agric Ecosyst Environ 202:203–216. https://doi.org/10.1016/j.agee.2015.01.008
Schirmel J, Mantilla-Contreras J, Gauger D, Blindow I (2015) Carabid beetles as indicators for shrub encroachment in dry grasslands. Ecol Ind 49:76–82. https://doi.org/10.1016/j.ecolind.2014.09.041
Serrano ARM, Baptista M, Carvalho R, Boieiro M, Mendes S, Bartz M, Timóteo S, Azevedo-Pereira HMVS, Aguiar CAS, Alves da Silva A, Alves J, Briones MJ, Sousa JP, da Martins P (2023) Inventory of tiger- and ground-beetles (Coleoptera Caraboidea: Cicindelidae, Carabidae) in two sampling seasons of the Gorongosa National Park. Biodivers Data J 11:e101280. https://doi.org/10.3897/BDJ.11.e101280
Šiška B, Eliašová M, Kollár J (2020) Carabus population response to drought in lowland oak hornbeam forest. Water 12:3284. https://doi.org/10.3390/w12113284
Spake R, Barsoum N, Newton AC, Doncaster CP (2016) Drivers of the composition and diversity of carabid functional traits in UK coniferous plantations. Ecol Manage 359:300–308. https://doi.org/10.1016/j.foreco.2015.10.008
Stalmans ME, Massad TJ, Peel MJS, Tarnita CE, Pringle RM (2019) War-induced collapse and asymmetric recovery of large-mammal populations in Gorongosa National Park, Mozambique. PLoS ONE 14:e0212864. https://doi.org/10.1371/journal.pone.0212864
Šustek Z, Vido J, Škvareninová J, Škvarenina J, Šurda P (2017) Drought impact on ground beetle assemblages (Coleoptera, Carabidae) in Norway spruce forests with different management after windstorm damage – a case study from Tatra Mts. (Slovakia). J Hydrol Hydromech 65:333–342. https://doi.org/10.1515/johh-2017-0048
Tamm JC (1984) Surviving long submergence in the egg stage – a successful strategy of terrestrial arthropods living on flood plains (Collembola, Acari, Diptera). Oecologia 61:417–419
Tamm JC (1986) Temperature-controlled under-water dormancy and postflood hatching in Isotoma viridis (Collembola) as forms of adaptation to annual long-term flooding. Oecologia 68:241–245
Tsafack N, Xie Y, Wang X, Fattorini S (2020) Influence of climate and local habitat characteristics on carabid beetle abundance and diversity in Northern Chinese steppes. Insects 11:19. https://doi.org/10.3390/insects11010019
Vandewalle M, de Bello F, Berg MP, Bolger T, Dolédec S, Dubs F, Feld CK, Harrington R, Harrison PA, Lavorel S, da Silva PM, Moretti M, Niemelä J, Santos P, Sattler T, Sousa JP, Sykes MT, Vanbergen AJ, Woodcock BA (2010) Functional traits as indicators of biodiversity response to land use change across ecosystems and organisms. Biodivers Conserv 19:2921–2947. https://doi.org/10.1007/s10531-010-9798-9
Wamser S, Dauber J, Birkhofer K, Wolters V (2011) Delayed colonisation of arable fields by spring breeding ground beetles (Coleoptera: Carabidae) in landscapes with a high availability of hibernation sites. Agric Ecosyst Environ 144:235–240. https://doi.org/10.1016/j.agee.2011.08.019
Wang X, Steiner M, Schütz M, Vandegehuchte ML, Risch AC (2018) Progressively excluding mammals of different body size affects community and trait structure of ground beetles. Oikos 127:1515–1525. https://doi.org/10.1111/oik.05198
Warnaffe GB, Lebrun P (2004) Effects of forest management on carabid beetles in Belgium: implications for biodiversity conservation. Biol Conserv 118:219–234. https://doi.org/10.1016/j.biocon.2003.08.015
Widenfalk LA, Bengtsson J, Berggren Å, Zwiggelaar K, Spijkman E Huyer-Brugman F, Berg MP (2015) Spatially structured environmental filtering of collembolan traits in late successional salt marsh vegetation. Oecologia 179:537–549. https://doi.org/10.1007/s00442-015-3345-z
Woodcock BA, Westbury DB, Potts SG, Harris SJ, Brown VK (2005) Establishing field margins to promote beetle conservation in arable farms. Agric Ecosyst Environ 107:255–266. https://doi.org/10.1016/j.agee.2004.10.029
Zajicek P, Welti EAR, Baker NJ et al (2021) Long-term data reveal unimodal responses of ground beetle abundance to precipitation and land use but no changes in taxonomic and functional diversity. Sci Rep 11:17468. https://doi.org/10.1038/s41598-021-96910-7
Zamora J, Verdú JR, Galante E (2007) Species richness in Mediterranean agroecosystems: spatial and temporal analysis for biodiversity conservation. Biol Conserv 134:113–121. https://doi.org/10.1016/j.biocon.2006.08.011
Zerm M, Adis J (2001) Further observations on the natural history and survival strategies of riverine tiger beetles (Coleoptera: Cicindelidae) from open habitats in central amazonian floodplains (Brazil). Ecotropica 7:115–137
Zou Y, Sang W, Bai F, Brennan E, Diekman M, Liu Y, Li L, Marples A, Shi H, Zhongzhou S, Sun X, Wang C, Wang X, Warren-Thomas E, Yang X, Yu Z, Axmacher JC (2019) Large-scale α‐diversity patterns in plants and ground beetles (Coleoptera: Carabidae) indicate a high biodiversity conservation value of China’s restored temperate forest landscapes. Divers Distrib 25:1613–1624. https://doi.org/10.1111/ddi.12964
Acknowledgements
This work was supported by FCT - Fundação para a Ciência e Tecnologia, I.P., in the framework of the Project UIDB/04004/2020 and DOI identifier https://doi.org/10.54499/UIDB/04004/2020 (https://doi.org/10.54499/UIDB/04004/2020), and Associate Laboratory TERRA (LA/P/0092/2020) and DOI identifier https://doi.org/10.54499/LA/P/0092/2020 (https://doi.org/10.54499/LA/P/0092/2020). Project ECOASSESS – A biodiversity and ECOlogical ASSESSment of soil fauna of Gorongosa National Park (Mozambique) (PTDC/BIA¬CBI/29672/2017) was funded through national funds by FCT / MCTES (PIDDAC) under the Programme All Scientific Domains. Pedro Martins da Silva and Mário Boieiro were supported by FCT under contracts DL57/2016/IT057-18-7285 and DL57/2016/CP1375/CT0001, respectively. Marie Bartz was contracted by the University of Coimbra (contract nr. IT057-19-7955) through financial support by the Project/R&D Institution ECOASSESS. Sara Mendes and Rui Carvalho were financially supported by FCiências – Associação para a investigação e Desenvolvimento de Ciências through research grants funded by the Project/R&D Institution ECOASSESS. ST was funded by FCT under the contract CEECIND/00135/2017/CP1460/CT0002 and DOI identifier https://doi.org/10.54499/CEECIND/00135/2017/CP1460/CT0002 (https://doi.org/10.54499/CEECIND/00135/2017/CP1460/CT0002). JA was funded by FCT under the contract 2022.05848.CEECIND/CP1714/CT0008 and DOI identifier https://doi.org/10.54499/2022.05848.CEECIND/CP1714/CT0008 (https://doi.org/10.54499/2022.05848.CEECIND/CP1714/CT0008). ECOASSESS field sampling was carried out with the logistic support of Gorongosa National Park under supervision of Jason Denlinger (Lab manager) and Mark Stalmans (Director of Scientific Service).
Funding
Open access funding provided by FCT|FCCN (b-on).
Author information
Authors and Affiliations
Contributions
PMS and MB(Baptista) wrote the first draft of the manuscript. MB(Baptista) and RC analized the data. MB(Boieiro), ST, JA, AAdS, MJIB, ARMS and JPS reviewed previous versions of the ms. MB(Bartz), ARMS, SM, ST, MB(Boieiro), HMVSAP, MJIB, JPS and PMS collected the data. PMS and JPS coordinated the data management and field sampling. ARMS performed taxonomic identification of all Caraboidea specimens. PMS concieved the conceptual framework of the study.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Communicated by Andreas Schuldt.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Baptista, M., Carvalho, R., Boieiro, M. et al. The role of sheltered habitats in biodiversity conservation of species sensitive to drought: a case study using ground beetles (Coleoptera, Carabidae) in the Gorongosa National Park. Biodivers Conserv 33, 2345–2361 (2024). https://doi.org/10.1007/s10531-024-02853-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-024-02853-6