Abstract
The tropical dry forest (TDF) ecosystem is characterised by strong seasonality exasperated periodically by the El Niño/southern oscillation (ENSO). The environment produced by this event could constrain the survival of small organisms, such as insects. Carabid beetles were collected in a TDF in Armero, Colombia, during wet and dry seasons in both El Niño and non-El Niño periods. A series of traits linked to desiccation resistance were measured to characterise their adaptation to the TDF environment and to investigate changes experienced by carabid beetles during both episodes in quantitative (assemblage) and qualitative (traits) parameters. We found no difference in the presence of traits between El Niño and non-El Niño episodes, but carabid assemblages changed significantly in composition and assemblage structure between these episodes. During both periods, small-sized and nocturnal species dominated the assemblages, but in terms of number of individuals, medium and large-sized, and visual hunter species dominated. Calosoma alternans and Megacephala affinis were the most abundant species with high dispersal capacity. Carabid beetles exhibited morphological traits well-adapted to drought experienced in TDF, including when it is exasperated by ENSO. However, long-term studies can help to elucidate the real effects of ENSO and to confirm the adaptation of carabid beetles to cope with this extreme environment.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The tropical dry forest (TDF) ecosystem is characterised by strong seasonal rainfall with four to six dry months (Murphy and Lugo 1986), making the availability of moisture crucial to the survival of organisms (Maass and Burgos 2011). These natural fluctuations between wet and dry periods throughout the year are exasperated by the El Niño/southern oscillation (ENSO). In South America, ENSO is characterised by high temperatures and low precipitation (Poveda et al. 2000), as has happened in 2015/2016, which was one of the strongest ENSO episodes on record of the 21th Century (Luo et al. 2018). ENSO can be critical for the maintenance of the TDF ecosystem, considering its effects on plant and animal communities (Holmgren et al. 2001) species can face local or global extinction if their populations do not have sufficient time to recover between ENSO episodes (Charrete et al. 2006). For tropical insects, ENSO has shown strong community effects. For example, Chrysomelidae beetles experienced a considerable loss of species during the event, with partial population recovery after the dry period (Kishimoto-Yamada and Itioka 2008; Kishimoto-Yamada et al. 2009). For butterflies, ENSO’s effects can vary due to temporal migratory responses to drought (Srygley et al. 2010, 2014). Evidence exists that Cantharidae decrease in species richness due to this climatic event in TDF (Hernández and Caballero 2016). In general, it appears that the responses of insects to ENSO are related to resources, which are indirectly affected by the weather (White 2008).
The configuration of insect bodies (high surface area/volume ratio) puts an additional constraint to the persistence and success of species in this environment (Schowalter 2006). As such, an insect’s survival in ENSO-affected TDF landscapes will not only depend on their behavioural adaptations, but also their morphology (Cloudsley-Thompson 1975; Crawford 1981), and the effectiveness of those adaptations will contribute to the persistence of species (Chown et al. 2011). Species traits have become an important tool to predict the presence and persistence of species in the environment (Keddy 1992; Cadotte et al. 2011; Kraft et al. 2015). Carabid beetles have a wide range of traits linked to environment conditions (Homburg et al. 2014; Fountain-Jones et al. 2015), yet knowledge regarding this group’s traits are lacking in the tropics. Changes in the environment, as a result of disturbance, can play an important role in filtering traits in ground beetles (Shibuya et al. 2011; Pakeman and Stockan 2014; Piano et al. 2017; Magura and Lövei 2019; but see Kraft et al. 2015). ENSO is a recurring event in the TDF landscape (Caviedes 2001; Grove and Adamson 2018), and is likely to have had a strong filtering effect on insect communities (see Kotze and Lawes 2007; Meir and Pennington 2011). If this is the case, species in this landscape are expected to display traits that cope with harsh conditions, but abundances may fluctuate substantially between wet and dry periods, particularly so during ENSO events.
The aims of this study were to characterise the responses of TDF carabid beetles, in terms of drought tolerance, by investigating changes in TDF carabid assemblages during a period of El Niño (2015) and non-El Niño (2016) in both (1) quantitative assemblage parameters (number of species and abundances) and (2) qualitative parameters (trait dominance). We hypothesise that carabid species that are larger in size and with functional wings are well-adapted to drought episodes in the TDF ecosystem. A larger beetle body has a lower surface area-to-volume ratio, conferring to desiccation resistance (Hood and Tschinkel 1990; Chown et al. 1995; Le Lagadec et al. 1998), while macroptery – a dominant trait in unstable habitats – facilitates an individual’s escape from unfavourable conditions (Darlington 1943; Venn 2016). On the other hand, smaller bodied beetles can benefit from this environment for other reasons, including requiring fewer resources to satisfy their energetic requirements and protection against predators (Blanckenhorn 2000; Chown and Gaston 2010). As such, we expected small-sized carabid beetles of elongate or narrow form, fossorial legs and/or nocturnal habits to be able to escape the risk of water loss (Forsythe 1987; Erwin 1979; Bauer and Kredler 1993; Bauer et al. 1998) in the TDF landscape. These smaller species are also expected to have a long metatrochanter to aid in mobility through confined habitats (Forsythe 1981). In terms of flight, even though macroptery is beneficial, flight is energetically expensive, especially during periods of limited resources (Nelemans 1987). Finally, a relationship between coloration and thermoregulation in carabids have been observed in the Palearctic zone, where a dark dorsal surface is beneficial to gain heat (Schweiger and Beierkuhnlein 2016); as such, we expect that most species in this hot landscape would have lightly coloured bodies. However, colour could have a minor role in thermoregulation in TDF carabids but a prominent role in predation avoidance, due to the prominence of predator avoidance behaviour in ground beetles, although it implies some thermal cost (Schultz 1986; Hadley et al. 1988, 1992).
Quantitatively, we expect a decrease in carabid beetle species richness and abundance during the El Niño episode in Colombia TDF, similar to what occurred in Ecuadorian Amazonian rain forests (Lucky et al. 2002). Drought produced by ENSO may stimulate a diapause and escape response in some species to avoid desiccation and thus diminish their temporal occurrence (Dingle 1972; Lövei and Sunderland 1996; Venn 2016). We presume temperature and moisture act as clues to start and end diapause during an ENSO episode (Cloudsley-Thompson 1975; Wolda and Denlinger 1984; Tauber et al. 1998; Hodek 2003, 2012). This means that species richness and abundance can decline drastically during drought events, but can also recover in relatively short time scales when precipitation returns. However, we do not suspect drastic changes in trait dominance between these two climatic states due to the strong adaptation to drought that organisms show in TDF (Dirzo et al. 2011; Pizano and García 2014; Pulla et al. 2015).
Material and methods
Study area
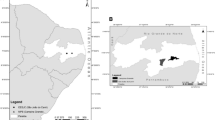
Ground beetles were surveyed in the dry forest biome in Armero (Tolima), Colombia (Fig. 1). Average temperatures during the surveys were 45 °C and 35 °C for the El Niño dry and wet seasons respectively, while the non-El Niño dry and wet seasons were around 30 °C. Air humidity were 36% (dry season) and 61% (wet season) during the El Niño episode and around 70% during the non-El Niño period (see Supplementary information 1). Given the current fragmented status of tropical dry forest and that the mostly dry forest of the Valley of Magdalena River in Colombia are immersed in a mosaic of pastures and areas at different successional stages (Pizano et al. 2014, 2016), we characterised the beetle assemblage and their traits in the TDF landscape by sampling three dominant habitat types: five forest patches (see F1–5 in Fig. 1b), four early successional patches (3–7 years of age, ES1–4) and three pastures (P1–3). The minimum distance between any of the 12 sites was 240 m.
Carabid beetle sampling
Carabid beetles were collected during an El Niño (2015) and non-El Niño (2016) event. During each period (El Niño and non- El Niño), beetles were collected in one month during the dry season (September) and one month during the wet season (October). Ten pitfall traps of 300 ml with water plus a few drops of detergent were used at each site to collect the ground beetles. The traps were installed 10 m apart along a transect of 100 m, and were operated continuously for three days per month. Each transect was at least 20 m from the edge of the site to minimize edge effects. Adult carabid beetles were identified to genus level using Martínez (2005), and to species level using Dejean (1829, 1831); Putzeys (1846, 1866); Reichardt (1967); Ball and Shpeley (2002, 2009); Vitolo (2004); Will (2005) and Bruschi (2010). However, due to the scarcity of taxonomic keys for the Neotropics, some of the identifications at species level should be confirmed. Voucher specimens are deposited in the Entomological Museum of the Universidad del Tolima, Colombia (MENT-UT).
Trait measurements
Based on a literature review, a series of traits related to the adaptation to desiccation were measured (Supplementary information 2). Information about the ecology and dispersal power (at genus level) were obtained from Larochelle and Larivière (2003), Vitolo (2004), Martínez (2005) and Will (2005). However, in an attempt to develop ecological information at species level, a set of traits were measured from the specimens collected to deduce habit and microhabitat use: desiccation resistance, daily activity time (nocturnal, diurnal), microhabitat use (burrowing habit and capacity to shelter in confined habitats, fast runner, slow runner), and dispersal capacity (high, low) (Table 1). The specimens collected were mounted on an entomological pin, and photographed with a Canon camera (PowerShot SX200 IS) through a stereomicroscope (Motic SMZ-168). Measurements were taken with ImageJ 1.52 k software (Schneider et al. 2012). Ten individuals per species were used for measurements (means were used), unless fewer than 10 individuals were collected, in which case all of the individuals were measured (see Supplementary information 3). The ratio between traits that involves size and body length was used to compare between species. For the capacity to shelter in confined spaces (microhabitat use), the ratio between prothorax width-depth and abdomen width-depth was used. The range of measures to classify and characterise certain attributes were from Forsythe (1981, 1987) and Bauer and Kredler (1993). Flight muscle development was determined by comparing the flight muscles of specimens to the flight muscle figures in Desender (2000).
Data analyses
We used the χ2 test in Past 3.x (Hammer et al. 2001) to compare the distribution of each trait among the El Niño and non-El Niño episodes.
Results
Carabid beetle trait characteristics in the tropical dry forest landscape
The traits of 15 species were measured (Supplementary information 3); Meotachys sp. was excluded due to its small body size (2.2 mm). 73.3% of the species collected were classified as small (4–12 mm), and 26.6% as either medium or large (Table 2). The literature (see Trait measurements section above) classified 80% of the collected species as nocturnal, 13.3% intermediate (both diurnal and nocturnal activity) and for one species, daily activity period is unknown. However, the most abundant species, Calosoma alternans and Megacephala affinis, were intermediate. All nocturnal species had short antennae (ANT/BS = 0.28–0.47) except Galerita sp., whose antennae were longer (ANT/BS = 0.62) (Supplementary information 4). Head width also did not show clear differences between nocturnal and intermediate species, only two species had wide heads; Barysomus hoepfneri (nocturnal, HW/BS = 0.29) and M. affinis (intermediate HW/BS = 0.27). On the contrary, eye surface area reflected behaviour presented in the literature, i.e., nocturnal species had small eyes (CES/BS = 0.01–0.05) and intermediate species had large eyes (CES/BS = 0.08–0.13).
Twenty percent of the species had fossorial forelegs (Aspidoglossa crenata, Clivina sp. and Camptodontus sp.), and had a prothorax width/abdomen width and prothorax depth/abdomen depth ratio of almost 1 (Supplementary information 4). Two runner species Athrostictus paganus and Enceladus gigas had the same body configurations. In terms of the fore- and hindleg total length, differences between fossorial and runner species were also clear; these were shorter for fossorial species (Fore-LTL/BS = 0.36–0.42, Hind-LTL/BS = 0.40–0.57): except for the runner species Apenes morio (both fore- and hindlegs shorter), Stolonis interceptus, which had shorter hindlegs and, A. paganus and E. gigas, which had shorter forelegs. Most species had a long metatrochanter (73.3%; MTL/BS = 0.09–0.13), but M. affinis was the only species with a long and slender metafemora, long metatibiae and small metatrochanter.
Most species were macropterous (80%), however only 41% of these had developed flight muscles. Apenes prasinus was brachypterous and E. gigas was apterous. None of the species collected showed hindwing polymorphism. Also, 80% of the species were dark in body colour and 53% had dark legs. Apenes coriacea was unique with a lightly coloured body.
Assemblage changes between El Niño and non-El Niño episodes
Distribution of species
Sixteen carabid beetle species (70 individuals) were collected; six species (17 individuals) during the El Niño period, and 14 species (53 individuals) during the non-El Niño period (Table 3). During the El Niño episode, the most abundantly collected species was C. alternans, but during the non-El Niño episode, only one individual of this species was collected. During the non-El Niño period, the most abundantly collected species was M. affinis, followed by E. gigas and Tetragonoderus sp.; these two last mentioned species were not collected during the El Niño event. Despite the low abundance of carabids, a marked change in assemblage composition and structure was observed. There is a clear substitution in dominance and the disappearance of many species during the El Niño period.
The wet season during both El Niño and non-El Niño periods had the highest number of individuals (88% and 71% respectively). Only two species were collected during the dry season of the El Niño period; Galerita sp. and M. affinis. During the non-El Niño period, similar numbers of species were collected during the dry (9 species) and wet (10 species) seasons. Aspidoglossa crenata, B. hoepfnery, E. gigas, M. affinis and Tetragonoderus sp. were present in both seasons.
Distribution of functional response traits
All measured traits and attributes were present in both episodes, except for light coloured bodies, which was not present during the El Niño event. The ratios of attributes within each trait during these two periods, and their significant differences are presented in Fig. 2. During both El Niño and non-El Niño periods, small-sized species dominated the assemblages, but in terms of individuals, medium and large-size dominated. In terms of daily activity period, most species collected were nocturnal (which was also reflected in the traits associated with daily activity period; head width, antennal length and compound eye surface area), while most individuals were intermediate (reflected only in compound eye surface area). This applied to both El Niño and non-El Niño periods. The runner/poor digger trait was dominant during both periods, with long fore- and hind legs. Short metatrochanter was abundant in the non-El Niño period, so too were metallic body colour and pale legs. High dispersal capacity, in terms of the proportion of individuals collected, was dominant during both periods.
Distribution of carabid beetle functional response traits among El Niño and non-El Niño periods in Armero, Colombia. Abbreviations are explained in Supplementary information 2. χ2 and p values are presented that test for differences in the distribution of attributes within each trait between the two climatic periods. spp = species, ind = individuals
Discussion
Despite the fact that ecological information on tropical carabid beetles is sparse, studies have shown that there are direct relationships between traits and habits/lifestyles (Forsythe 1983, 1987, 1991; Talarico et al. 2007). This was also confirmed in our study, which showed that the traits displayed by carabids are reflective of this group being well-adapted to environmental change experienced in TDF, including when it is exasperated by the El Niño/southern oscillation (ENSO). As predicted, changes in the assemblage between El Niño and non-El Niño were more quantitative than qualitative. All traits and attributes (except light body colour) were present during both climatic episodes and marked changes were perceived in the number of species and individuals, which recovered relatively fast after the climatic anomaly ended. Most of the carabid species collected were small and nocturnal, although in terms of numbers of individuals collected, medium and large sizes and intermediate activity trait attributes were most dominant, contrary to our expectation. It appears that resource availability is a limiting factor for large-sized species during droughts, while small-sized species persist in a low-resource environment and benefit from being nocturnal, thus avoiding desiccation. Similarly, runner species was a dominant trait, but with a long metatrochanter that reduces the ability to run, yet aids in the species’ ability to move through confined spaces or litter. A long metatrochanter was also present in medium-sized and large species. Almost all species were macropterous (80%), although only five species showed developed flight muscles, perhaps as a consequence of limited resources (Nelemans 1987; Nelemans et al. 1989). Calosoma alternans and M. affinis were the most abundant species with high dispersal capacity.
Quantitative carabid beetle changes between El Niño and non-El Niño periods
Quantitatively, TDF carabid beetles were affected by El Niño (ENSO), as has happened with other tropical beetle groups (Lucky et al. 2002; Kishimoto-Yamada and Itioka 2008; Kishimoto-Yamada et al. 2009; Hernández and Caballero 2016). The number of species and individuals decreased more than two fold during the El Niño period. However, carabids showed differential responses to drought, similarly to the Chrysomelidae in Borneo during the 1998 ENSO event (Kishimoto-Yamada et al. 2009). In Colombian TDF, 62% of the collected species were not present during the El Niño period, C. alternans was the only species showing a substantial decrease during the non-El Niño period, its numerical decrease could be related to its life span (see Burgess 1911): its larvae were seen in high numbers in pastures in October and November (Ariza 2016; pers. obs.), however long-term studies can help to elucidate the life cycle of this species. On the contrary, M. affinis benefited considerably from an improved environment during non-El Niño periods. This fast running and flight capable species may be particularly vulnerable to desiccation during dry ENSO periods (Pearson and Vogler 2001). In general, the carabid beetle assemblage recovered quickly (within three months after El Niño ended), which may be due to diapause as an adaptive mechanism to survive harsh conditions (see Burgess 1911; Jeffords and Case 1987; Jacobs et al. 2011).
Carabid beetle trait distribution in the tropical dry forest landscape
Even though the carabid beetle assemblage in TDF was dominated by small species, more individuals of medium and large sized species were collected; the two most abundant species C. alternans (large) and M. affinis (medium) possibly benefitting from their lower volume-to-surface area ratio, thus resisting desiccation during dry conditions (Hood and Tschinkel 1990; Chown et al. 1995; Le Lagadec et al. 1998). These species were observed walking during the day (Ariza 2016; pers. obs.), but are considered to be active both during the day and night (intermediate activity) (Larochelle and Larivière 2003; Vitolo 2004). Another medium-size species, Galerita sp. is consider nocturnal (Larochelle and Larivière 2003), and is the only nocturnal species of TDF that meets all the characteristic traits described as typical of this life-style: long antennae, small eyes and a narrow head (Bauer and Kredler 1993). The rest of the nocturnal species (which are also small) have short antennae, or at least shorter than Galerita sp. and M. affinis. However, antennal length and head width differences between nocturnal and intermediate species groups were small, making it difficult to characterize daily activity using these traits. Carabid beetles use three methods to detect prey: visual, tactile and olfactory, or a combination of these; species that do not hunt visually, use their antennae and palps (Wheater 1989). Antennae are an important sensory structure (Chapman 1998; Ploomi et al. 2003), but it is unclear how prominent its role is in prey detection. On the contrary, eye surface area has distinct differences between nocturnal and diurnal active species. Studies have shown that eyes are a better trait to reflect activity period (Bauer 1985; Talarico et al. 2007, 2011, 2018). For instance, C. alternans and M. affinis have large eyes, and although they can hunt both during the day and night, they are probably better visual hunters.
Small-sized species in dry ecosystems risk desiccation (Schoener and Janzen 1968), yet most species in TDF are small but at low abundance (27% of the total number of individuals). Although a large size has physiological advantages, it also has disadvantages in terms of food resources (high energetic requirements), and are more visible to predators (Blanckenhorn 2000). Small insects resolve the challenge to conserve moisture through, amongst others, behavioural adaptations, for instance by minimizing their exposure to harsh conditions (Chown and Klok 2003). In TDF, those adaptations include nocturnal activity and a digger habit (Hadley 1974; Remmert 1981); all small carabid species captured are nocturnal, and although only three species are burrowing specialists, all non-fossorial species have a long metatrochanter, which is related to the ability to push the body into confined habitats and leaf litter, both to hunt and for shelter (Forsythe 1981, 1987). Burrowing species are characterized by fossorial legs and short fore- and hindlegs, which help with entering the ground (Forsythe 1981). Additional to these morphological adaptations, burrowing species like A. crenata, Clivina sp. and Camptodontus sp., and runner species like A. paganus have similar proportions of the prothorax and hind body (width and depth) that permit them to move in fissures and avoid friction and obstruction (Forsythe 1987).
Based on the traits measured, we can infer that all small species have low desiccation resistance, are olfactory/tactile hunters and good diggers or with good abilities to move in restricted spaces, while medium and large sized species have higher desiccation resistance (Table 2). Galerita sp. is the only species from this last group with an olfactory/tactile hunter strategy. This species and C. alternans have long metatrochanter, probably as a mechanisms to hunt in the litter layer or shelter from predation (Forsythe 1991; Larochelle and Larivière 2003). Enceladus gigas was the biggest and only apterous species, and although its metatrochanter does not aid in its ability to push into narrow spaces, its pedunculate body facilitate movement through them (Forsythe 1987). Finally, M. affinis could be consider a fast visual hunter, with large and slender legs, and a short metatrochanter (Forsythe 1981). Both C. alternans and M. affinis are macropterous with flight muscles developed, allowing these open-habitat species to escape predation (Forsythe 1987). Additionally, the iridescent body colour of M. affinis and iridescent shades of C. alternans provides additional protection against predators, which may get disorientated when these carabids fly between sunny and shady areas (Seago et al. 2009).
Conclusions
We showed that the ratios of attributes in carabid beetle response traits between the El Niño and non-El Niño periods differed in the tropical dry forest ecosystem, yet trait occurrence was similar between the two periods. Species were generally small in size, with nocturnal activities, while in terms of abundance, medium and large sized beetles with intermediate daily activity dominated. It appears that in this dry ecosystem, resource limitation is a greater challenge to the presence of carabid beetles than desiccation risk. Carabid beetles possess a set of traits that show adaptation to harsh conditions experience during El Niño in the TDF. Diapause could have a prominent role in species present in the TDF. Yet, despite the importance of diapause to survive bad conditions, insects experience mortality and other costs during diapause (Nelemans et al. 1989; Matsuo 2006). Long term studies on the effects of ENSO linked with other anthropogenic pressures can clarify the real risks to carabid beetle communities during ENSO, especially given additional threats, such as climate change.
Data availability
The authors confirm that the data supporting the findings of this study are available within the article (and/or) its supplementary materials.
References
Ball GE, Shpeley D (2002) The Neotropical subgenera and species of the Pantropical genus Anaulacus MacLeay (sensu novo) (Coleoptera: Carabidae: Masoreini): A taxonomic revision, with notes about way of life, evolution, and geographical history. Trans Am Entomol Soc 128:265–343. https://www.jstor.org/stable/25078782
Ball GE, Shpeley D (2009) A taxonomic review of the genus Apenes Leconte (Coleoptera: Carabidae: Lebiini) in the West Indies, with descriptions of new species and notes about classification and biogeography. Ann Carnegie Mus 78:79–191. https://doi.org/10.2992/007.078.0201
Bauer T (1985) Different adaptation to visual hunting in three ground beetle species of the same genus. J Insect Physiol 31:593–601. https://doi.org/10.1016/0022-1910(85)90057-5
Bauer T, Desender K, Morwinsky T, Betz O (1998) Eye morphology reflects habitat demands in three closely related ground beetle species (Coleoptera: Carabidae). J Zool 245:467–472. https://doi.org/10.1111/j.1469-7998.1998.tb00121.x
Bauer T, Kredler M (1993) Morphology of the compound eyes as an indicator of life-style in carabid beetles. Can J Zool 71:799–810. https://doi.org/10.1139/z93-105
Blanckenhorn WU (2000) The evolution of body size: What keeps organisms small? Q Rev Biol 75:385–407. https://doi.org/10.1086/393620
Bruschi S (2010) Key to the species of subgenus Castrida Motschulsky. http://www.calosomas.com/Castrida/Castrida_keys.html. Accessed 17 Sept 2018
Burgess AF (1911) Calosoma sycophanta: Its life history, behaviour, and successful colonization in New England. US Dept. of Agriculture, Bureau of Entomology, Washington. https://doi.org/10.5962/bhl.title.65273
Cadotte MW, Carscadden K, Mirotchnick N (2011) Beyond species: Functional diversity and the maintenance of ecological processes and services. J Appl Ecol 48:1079–1087. https://doi.org/10.1111/j.1365-2664.2011.02048.x
Caviedes CN (2001) El Niño in history storming through the ages. University Press of Florida, Florida
Chapman RF (1998) The insects structure and function. Cambridge University Press, NewYork
Charrete NA, Cleary DFR, Mooers AØ (2006) Range-restricted, specialist bornean butterflies are less likely to recover from ENSO-induced disturbance. Ecology 87:2330–2337. https://www.jstor.org/stable/20069234
Chown SL, Gaston KJ (2010) Body size variation in insects: A macroecological perspective. Biol Rev 85:139–169. https://doi.org/10.1111/j.1469-185X.2009.00097.x
Chown SL, Klok CJ (2003) Water-balance characteristics respond to changes in body size in subantarctic Weevils. Physiol Biochem Zool 76:634–643. https://doi.org/10.1086/376919
Chown SL, Scholtz CH, Klok CJ, Joubert FJ, Coles KS (1995) Ecophysiology, range contraction and survival of a geographically restricted African dung beetle (Coleoptera: Scarabaeidae). Funct Ecol 9:30–39. https://doi.org/10.2307/2390087
Chown SL, Sørensen JG, Terblanche JS (2011) Water loss in insects: An environmental change perspective. J Insect Physiol 57:1070–1084. https://doi.org/10.1016/j.jinsphys.2011.05.004
Cloudsley-Thompson JL (1975) Adaptations of Arthropoda to arid environments. Annu Rev Entomol 20:261–283. https://doi.org/10.1146/annurev.en.20.010175.001401
Crawford CS (1981) Biology of desert invertebrates. Springer-Verlag, Berlin
Darlington PJ (1943) Carabidae of mountains and islands: Data on the evolution of isolated faunas, and on atrophy of wings. Ecol Monogr 13:37–61. https://doi.org/10.2307/1943589
Dejean PFMA (1829) Spécies génйral des Coléoptčres, de la collection de M. le Comte Dejean, Crevot, Paris
Dejean PFMA (1831) Spécies génйral des Coléoptčres, de la collection de M. le Comte Dejean, Crevot, Paris
Desender K (2000) Flight muscle development and dispersal in the life cycle of carabid beetles: Patterns and process. Bull Inst Roy Sci Nat Belgique 70:13–31
Dingle H (1972) Migration strategies of insects. Science 175:1327–1335. https://doi.org/10.1126/science.175.4028.1327
Dirzo R, Young HS, Mooney HA, Ceballos G (2011) Seasonally dry tropical forests: Ecology and conservation. Island Press, Washington
Erwin TL (1979) Thoughts on the evolutionary history of ground beetles: Hypotheses generated from comparative faunal analyses of lowland forest sites in temperate and tropical regions. In: Erwin TL, Ball GE, Whitehead DR, Halpern AL (eds) Carabid beetles their evolution, natural history, and classification. Springer, Netherlands, Dordrecht, pp 539–592
Forsythe TG (1981) Running and pushing in relationship to hind leg structure in some Carabidae (Coleoptera). Coleopts Bull 35:353–378. https://www.jstor.org/stable/4007954
Forsythe TG (1983) Locomotion in ground beetles (Coleoptera carabidae): An interpretation of leg structure in functional terms. J Zool 200:493–507. https://doi.org/10.1111/j.1469-7998.1983.tb02811.x
Forsythe TG (1987) The relationship between body form and habit in some Carabidae (Coleoptera). J Zool 211:643–666. https://doi.org/10.1111/j.1469-7998.1987.tb04477.x
Forsythe TG (1991) Feeding and locomotory functions in relation to body form in five species of ground beetle (Coleoptera: Carabidae). J Zool 223:233–263. https://doi.org/10.1111/j.1469-7998.1991.tb04763.x
Fountain-Jones NM, Baker SC, Jordan GJ (2015) Moving beyond the guild concept: Developing a practical functional trait framework for terrestrial beetles. Ecol Entomol 40:1–13. https://doi.org/10.1111/een.12158
Grove R, Adamson G (2018) El Niño in World history. Palgrave Macmillan UK, London
Hadley NF (1974) Adaptational biology of desert Scorpions. J Arachnol 2:11–23. https://www.jstor.org/stable/3704992
Hadley NF, Savill A, Schultz TD (1992) Coloration and its thermal consequences in the New Zealand tiger beetle Neocicindela perhispida. J Therm Biol 17:55–61. https://doi.org/10.1016/0306-4565(92)90020-G
Hadley NF, Schultz TD, Savill A (1988) Spectral reflectances of three tiger beetle subspecies (Neocicindela perhispida): Correlations with their habitat substrate. New Zeal J Zool 15:343–346. https://doi.org/10.1080/03014223.1988.10422624
Hammer Ø, Harper DAT, Ryan PD (2001) PAST: Paleontological statistics software package for education and data analysis. Palaeontol Electron 4:1–9
Hernández XCP, Caballero SZ (2016) Temporal variation in the diversity of Cantharidae (Coleoptera), in seven assemblages in Tropical dry forest in Mexico. Trop Conserv Sci 9:439–464. https://doi.org/10.1177/194008291600900124
Hodek I (2003) Role of water and moisture in diapause development (A review). Eur J Entomol 100:223–232. https://doi.org/10.14411/eje.2003.037
Hodek I (2012) Adult diapause in coleoptera. Psyche 249081:1–10. https://doi.org/10.1155/2012/249081
Holmgren M, Scheffer M, Ezcurra E, Gutiérrez JR, Mohren GMJ (2001) El Niño effects on the dynamics of terrestrial ecosystems. Trends Ecol Evol 16:89–94. https://doi.org/10.1016/S0169-5347(00)02052-8
Homburg K, Homburg N, Schäfer F, Schuldt A, Assmann T (2014) Carabids.org - a dynamic online database of ground beetle species traits (Coleoptera, Carabidae). Insect Conserv Divers 7:195–205. https://doi.org/10.1111/icad.12045
Hood WG, Tschinkel WR (1990) Desiccation resistance in arboreal and terrestrial ants. Physiol Entomol 15:23–35. https://doi.org/10.1111/j.1365-3032.1990.tb00489.x
Jacobs JM, Colin Bergeron JA, Work TT, Spence JR (2011) Low intensity surface fire instigates movement by adults of Calosoma frigidum (Coleoptera, Carabidae). ZooKeys 147:641–649. https://doi.org/10.3897/zookeys.147.2084
Jeffords M, Case L (1987) Effect of prey density on diurnal activity and ovarian development in Calosoma calidum (Coleoptera: Carabidae): Implications for biological control of the Gypsy moth, Lymantria dispar (Lepidoptera: Lymantriidae) in the Midwest. Great Lakes Entomol 20:9. https://scholar.valpo.edu/tgle/vol20/iss2/9
Keddy PA (1992) Assembly and response rules: Two goals for predictive community ecology. J Veg Sci 3:157–164. https://doi.org/10.2307/3235676
Kishimoto-Yamada K, Itioka T (2008) Consequences of a severe drought associated with an El Niño-Southern Oscillation on a light-attracted leaf-beetle (Coleoptera, Chrysomelidae) assemblage in Borneo. J Trop Ecol 24:229–233. https://doi.org/10.1017/S0266467408004811
Kishimoto-Yamada K, Itioka T, Sakai S, Momose K, Nagamitsu T, Kaliang H, Meleng P, Chong L, Karim AAH, Yamane S, Kato M, Reid CA, Nakashizuka T, Inoue T (2009) Population fluctuations of light-attracted chrysomelid beetles in relation to supra-annual environmental changes in a Bornean rainforest. Bull Entomol Res 99:217–227. https://doi.org/10.1017/S000748530800624X
Kotze DJ, Lawes MJ (2007) Viability of ecological processes in small Afromontane forest patches in South Africa. Austral Ecol 32:294–304. https://doi.org/10.1111/j.1442-9993.2007.01694.x
Kraft NJB, Adler PB, Godoy O, James EC, Fuller S, Levine JM (2015) Community assembly, coexistence and the environmental filtering metaphor. Funct Ecol 29:592–599. https://doi.org/10.1111/1365-2435.12345
Le Lagadec MD, Chown SL, Scholtz CH (1998) Desiccation resistance and water balance in southern African keratin beetles (Coleoptera, Trogidae): The influence of body size and habitat. J Comp Physiol B 168:112–122. https://doi.org/10.1007/s003600050127
Larochelle A, Larivière MC (2003) A natural history of the ground-beetles (Coleoptera: Carabidae) of America north of Mexico. Pensoft Publishers, Bulgaria
Lövei GL, Sunderland KD (1996) Ecology and behavior of ground beetles (Coleoptera: Carabidae). Annu Rev Entomol 41:231–256. https://doi.org/10.1146/annurev.en.41.010196.001311
Lucky A, Erwin TL, Witman JD (2002) Temporal and spatial diversity and distribution of arboreal Carabidae (Coleoptera) in a Western Amazonian rain forest. Biotropica 34:376–386. https://doi.org/10.1111/j.1744-7429.2002.tb00551.x
Luo X, Keenan TF, Fisher JB, Jiménez-Muñoz JC, Chen JM, Jiang C, Ju W, Perakalapudi NV, Ryu Y, Tadić JM (2018) The impact of the 2015/2016 El Niño on global photosynthesis using satellite remote sensing. Philos Trans R Soc B 373:20170409. https://doi.org/10.1098/rstb.2017.0409
Maass M, Burgos A (2011) Water dynamics at the ecosystem level in Seasonally dry tropical forests. In: Dirzo R, Young HS, Mooney HA, Ceballos G (eds) Seasonally dry tropical forests: Ecology and conservation. Island Press, Washington, pp 141–156
Magura T, Lövei GL (2019) Environmental filtering is the main assembly rule of ground beetles in the forest and its edge but not in the adjacent grassland. Insect Sci 26:154–163. https://doi.org/10.1111/1744-7917.12504
Martínez C (2005) Introducción a los escarabajos Carabidae. Instituto de Investigación de Recursos Biológicos Alexander von Humboldt, Bogotá
Matsuo Y (2006) Cost of prolonged diapause and its relationship to body size in a seed predator. Funct Ecol 20:300–306. https://doi.org/10.1111/j.1365-2435.2006.01097.x
Meir P, Pennington RT (2011) Climatic change and Seasonally dry tropical forests. In: Dirzo R, Young HS, Mooney HA (eds) Seasonally dry tropical forests: Ecology and conservation. Island Press, Washington, pp 279–299
Murphy PG, Lugo AE (1986) Ecology of Tropical dry forest. Annu Rev Ecol Evol Syst 17:67–88. https://doi.org/10.1146/annurev.es.17.110186.000435
Nelemans MNE (1987) Possibilities for flight in the carabid beetle Nebria brevicollis (F.). The importance of food during larval growth. Oecologia 72:502–509. https://doi.org/10.1007/BF00378974
Nelemans MNE, Den Boer PJ, Spee A (1989) Recruitment and summer diapause in the dynamics of a population of Nebria brevicollis (Coleoptera: Carabidae). Oikos 56:157–169. https://doi.org/10.2307/3565331
Pakeman RJ, Stockan JA (2014) Drivers of carabid functional diversity: Abiotic environment, plant functional traits, or plant functional diversity? Ecology 95:1213–1224. https://doi.org/10.1890/13-1059.1
Pearson DL, Vogler AP (2001) Tiger Beetles: The evolution, ecology, and diversity of the cicindelids. Cornell University Press, United States of America
Piano E, De Wolf K, Bona F, Bonte D, Bowler DE, Isaia M, Lens L, Merckx T, Mertens D, Van Kerckvoorde M, De Meester L, Hendrickx F (2017) Urbanization drives community shifts towards thermophilic and dispersive species at local and landscape scales. Glob Chang Biol 23:2554–2564. https://doi.org/10.1111/gcb.13606
Pizano C, García H (2014) El Bosque seco tropical en Colombia. Instituto de Investigación de Recursos Biológicos Alexander von Humboldt (IAvH), Bogotá
Pizano C, González-M R, González MF, Castro-Lima F, López R, Rodríguez N, Idárraga-Piedrahíta Á, Vargas W, Vergara-Varela H, Castaño-Naranjo A, Devia W, Rojas A, Cuadros H, Toro JL (2014) Las plantas de los Bosques secos de Colombia. In: Pizano C, García H (eds) El Bosque seco tropical en Colombia. Instituto de Investigación de Recursos Biológicos Alexander von Humboldt, Bogotá, pp 48–93
Pizano C, González-M R, López R, Jurado RD, Cuadros H, Castaño-Naranjo A, Rojas A, Pérez K, Vergara-Varela H, Idárraga Á, Isaacs P, García H (2016) El Bosque seco tropical en Colombia: Distribución y estado de conservación. In: Gómez MF, Moreno LA, Andrade GI, Rueda C (eds) Biodiversidad 2015: Estado y tendencias de la biodiversidad continental de Colombia. Instituto Alexander von Humboldt, Bogotá, pp ficha 202.
Ploomi A, Merivee E, Rahi M, Bresciani J, Ravn HP, Luik A, Sammelselg V (2003) Antennal sensilla in ground beetles (Coleoptera, Carabidae). Agron Res 1:221–228
Poveda G, Graham NE, Epstein PR, Rojas W, Quiñones ML, Vélez ID, Martens WJM (2000) Climate and ENSO variability associated with vector-borne diseases in Colombia. In: Diaz HF, Markgraf V (eds) El Niño and the Southern oscillation: Multiscale variability and global and regional impacts. Cambridge University Press, Cambridge, pp 183–204
Pulla S, Ramaswami G, Mondal N, Chitra-Tarak R, Suresh HS, Dattaraja HS, Vivek P, Parthasarathy N, Ramesh BR, Sukumar R (2015) Assessing the resilience of global Seasonally dry tropical forests. Int For Rev 17:91–113. https://doi.org/10.1505/146554815815834796
Putzeys J (1846) Monographie des Clivina et genres voisins, précedée d’un tableau synoptique des genres de la tribu des Scaritides. Mémoires de la Socíeté R. des Sciencies de Liege 2:521–663
Putzeys J (1866) Révision génйrale des Clivinides. Annales de la Sociétй Entomologique de Belgique 10:1–242
Reichardt H (1967) A monographic revision of the american Galeritini (Coleoptera:Carabidae). Arq Zool 15:1–176. https://doi.org/10.11606/issn.2176-7793.v15i1-2p1-176
Remmert H (1981) Body size of terrestrial Arthropods and biomass of their populations in relation to the abiotic parameters of their milieu. Oecologia 50:12–13. https://doi.org/10.1007/BF00378789
Schneider CA, Rasband WS, Eliceiri KW (2012) NIH Image to ImageJ: 25 years of Image Analysis. Nat Methods 9:671–675. https://doi.org/10.1038/nmeth.2089
Schoener TW, Janzen DH (1968) Notes on environmental determinants of tropical versus temperate insect size patterns. Am Nat 102:207–224. https://doi.org/10.1086/282538
Schowalter TD (2006) Insect ecology - An ecosystem epproaach. Academic Press, San Diego
Schultz TD (1986) Role of structural colors in predator avoidance by tiger beetles of the genus Cicindela (Coleoptera: Cicindelidae). Bull Entomol Soc Am 32:142–146. https://doi.org/10.1093/besa/32.3.142
Schweiger AH, Beierkuhnlein C (2016) Size dependency in colour patterns of Western Palearctic carabids. Ecography 39:846–857. https://doi.org/10.1111/ecog.01570
Seago AE, Brady P, Vigneron JP, Schultz TD (2009) Gold bugs and beyond: A review of iridescence and structural colour mechanisms in beetles (Coleoptera). J R Soc Interface 6:165–184. https://doi.org/10.1098/rsif.2008.0354.focus
Shibuya S, Kubota K, Ohsawa M, Kikvidze Z (2011) Assembly rules for ground beetle communities: What determines community structure, environmental factors or competition? Eur J Entomol 108:453–459. https://doi.org/10.14411/eje.2011.058
Srygley RB, Dudley R, Oliveira EG, Aizprúas R, Pelaez NZ, Riveros AJ (2010) El Niño and dry season rainfall influence hostplant phenology and an annual butterfly migration from Neotropical wet to dry forests. Glob Chang Biol 1:936–945. https://doi.org/10.1111/j.1365-2486.2009.01986.x
Srygley RB, Dudley R, Oliveira EG, Riveros AJ (2014) El Niño, host plant growth, and migratory butterfly abundance in a changing climate. Biotropica 46:90–97. https://doi.org/10.1111/btp.12081
Talarico F, Brandmayr P, Giglio A, Massolo A, Brandmayr TZ (2011) Morphometry of eyes, antennae and wings in three species of Siagona (Coleoptera, Carabidae). ZooKeys 100:203–214. https://doi.org/10.3897/zookeys.100.1528
Talarico F, Cavaliere F, Mazzei A, Brandmayr P (2018) Morphometry and eye morphology of three scaritine ground beetles relate to habitat demands and behavioural traits (Coleoptera, Carabidae, Scaritinae). Zool Anz 277:190–196. https://doi.org/10.1016/j.jcz.2018.10.002
Talarico F, Romeo M, Massolo A, Brandmayr P, Zetto T (2007) Morphometry and eye morphology in three species of Carabus (Coleoptera: Carabidae) in relation to habitat demands. J Zool Syst Evol Res 45:33–38. https://doi.org/10.1111/j.1439-0469.2006.00394.x
Tauber MJ, Tauber CA, Nyrop JP, Villani MG (1998) Moisture, a vital but neglected factor in the seasonal ecology of insects: Hypotheses and tests of mechanisms. Environ Entomol 27:523–530. https://doi.org/10.1093/ee/27.3.523
Venn S (2016) To fly or not to fly: Factors influencing the flight capacity of carabid beetles (Coleoptera: Carabidae). Eur J Entomol 113:587–600. https://doi.org/10.14411/eje.2016.079
Vitolo AL (2004) Guía para la identificación de los escarabajos tigre. Instituto de la Investigación de Recursos Biológicos Alexander von Humboldt, Bogotá
Wheater C (1989) Prey detection by some predatory Coleoptera (Carabidae and Staphylinidae). J Zool 218:171–185. https://doi.org/10.1111/j.1469-7998.1989.tb02531.x
White TCR (2008) The role of food, weather and climate in limiting the abundance of animals. Biol Rev 83:227–248. https://doi.org/10.1111/j.1469-185X.2008.00041.x
Will KW (2005) The Neotropical genera Oxycrepis Reiche and Stolonis Motschulsky: A taxonomic review, key to the described species and description of new Stolonis species from Ecuador (Coleoptera: Carabidae: Loxandrini). Zootaxa 17:1–17. https://doi.org/10.11646/zootaxa.1049.1.1
Wolda H, Denlinger DL (1984) Diapause in a large aggregation of a tropical beetle. Ecol Entomol 9:217–230. https://doi.org/10.1111/j.1365-2311.1984.tb00717.x
Acknowledgements
We thank Hector E. Esquivel for permission to use the Dendrology laboratory of the Universidad del Tolima to measure the specimens collected. Comments from the editor and two reviewers improved the quality of this contribution substantially.
Funding
This research was partially supported by funding from the Universidad del Tolima.
Author information
Authors and Affiliations
Contributions
GMA collected the data and performed laboratory activities. GMA and DJK performed the analyses. All authors participated in the writing of the manuscript.
Corresponding author
Ethics declarations
Consent for publication
The first author declares that the two co-authors of this contribution are aware of the fact and have agreed to being so named.
Conflict of interest
The authors declare that there are no conflicts of interest in conducting the research.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ariza, G.M., Jácome, J. & Kotze, D.J. Carabid beetles of tropical dry forests display traits that cope with a harsh environment. Int J Trop Insect Sci 41, 3011–3021 (2021). https://doi.org/10.1007/s42690-021-00493-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42690-021-00493-9