Abstract
The rapid expansion of planted forests harms native biodiversity. Few studies report the effects of replacing wetlands with planted forests on ground beetles. We analyze how the taxonomic and functional diversity of ground beetles are affected by intensive management of a planted forest landscape in the Lower Delta of the Parana River. We defined six habitat types (n = 3, N = 18): young and mature willow (YW, MW), young and mature poplar without cattle (YP, MP), young and mature silvopastoral poplar (YS, MS). Using pitfall traps (N = 1728), we recorded 35 species (1896 individuals). YW and MS reached the highest taxonomic diversity and richness. YW with more vertical heterogeneity showed higher species richness than MW. Hydrophilic species were more abundant in YW. Zoophagous species were more abundant in MS. YS, MS, and YW reached the highest functional evenness, which implies that a large part of the functional niches was used. Cattle dung and freshwater canals for livestock offer more resources for ground beetles. The planted tree species, stand age, and presence of cattle affects taxonomic and functional diversity of ground beetles. Willow and silvopastoral planted forests are the most suitable habitats for hosting wetland species. So, we recommend using willow species rather than poplar species when planted forests replace fluvial wetlands, increasing irrigation of poplar planted forests through ditches and canals, conserving or restoring different strata of understory to increase vertical heterogeneity, and maintaining the landscape heterogeneity. These management measures are essential to prevent the loss of wetland species and conserve ground beetle’s diversity.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Rapid anthropogenic expansion through planted forest meant to intensify productivity has led to the transformation or even the disappearance of natural ecosystems (Sica et al. 2016; Evans and Turnbull 2004). The total area of forest in the world is 31 percent. The planted forest area is increasing and already represents 7 percent of the total forest area (264 million hectares) (FAO 2010). Between 2000 and 2010, the area of the planted forest increased by about 5 million hectares per year. Given the current trend, an additional increase in the planted forest area to 300 million hectares can be anticipated by 2020 (MacGregor 2002). Argentinian plantation area reached 1.2 million ha in 2015 (SENASA 2015).
It is well known that the replacement of natural ecosystems by anthropogenic ecosystems harms native biodiversity (Sica et al. 2018; Suárez et al. 2021). Anthropogenic ecosystems are relatively homogeneous and composed of one or few tree species with individuals of the same size and age. Planted forests generally support a low species diversity because of the limited number of available habitats (Stephens and Wagner 2007; Bremer and Farley 2010). However, the species diversity can be favored, with a dominance of generalist species, when the vertical heterogeneity of the planted forest understory is high (Magnano et al. 2019). The diversity of organisms is highly dependent on the heterogeneity of the landscape and the complexity of the stand (Dias et al. 2013). In this context, one of the most affected groups of animals is usually soil arthropods (Paillet et al. 2010).
Beetles of the family Carabidae (Coleoptera) have been used as indicators of natural and anthropogenic disturbances and management practices (Lövei and Sunderland 1996) because they are taxonomically well known, common in most terrestrial habitats, and can easily be collected using standard methods (Magura 2017). Also, this family provides important ecosystem services (Cicchino 2006; Paleologos et al. 2007; Kwiatkowski 2011). Studies addressing the influence of planted forests and their management on ground beetle have been reported positive (Niemela et al. 2007), negative (Magura et al. 2003) or with no effect (Magura et al. 2000) on richness and diversity in the boreal region and the Hungarian Mountain Range. However, few studies report the effects of replacing wetlands with planted forests on ground beetles. For other taxa, such as mammals and birds, ecologically sustainable planted forest management could contribute to their conservation in wetlands of the Lower Delta of the Paraná River (Fracassi et al. 2017; Magnano et al. 2019; Krug et al. 2019). Establishing how management practices affect ground beetle communities is essential for their conservation and for carrying out appropriate practices for the development of each assembly.
Species abundance, richness and diversity of ground beetles communities in planted forests are influenced by biotic factors (tree species composition, understory, canopy cover) (Chungu 2014; Kaizuka and Iwasa 2015), abiotic factors (light availability, structural and chemical characteristics of leaf litter, soil characteristics) (Magura et al. 2003; Chungu 2014) and management characteristics (type of implemented management, presence of cattle, location of the plantation within the landscape, edge effect, size and age of the stands, vicinity to other habitat types and habitat type that was replaced) (Gormally and Fahy 1998; Koivula 2001; Magura 2002; Niemelä et al. 2007; Binkley and Fisher 2012; Magura et al. 2017; Nanni et al. 2019). Seasonality could also be a determinant factor in structuring ground beetle communities (Saska et al. 2013). Furthermore, these factors modify the diversity and abundance of functional groups of ground beetles (e.g., Allegro and Sciaky 2003; Paleologos et al. 2007; Liu et al. 2015; Magura 2017). Functional group diversity may affect the dynamics of the available resources (e.g., food and shelter), as well as the stability of the entire ecosystem in terms of resilience and resistance (Díaz and Cabido 2001). Changes in the stability of the ecosystem are of vital importance, especially when planted forests replace highly dynamic wetland environments, as in the present study. In this context, we aim to analyze how taxonomic and functional diversity of ground beetles are differentially affected by intensive management of a planted forest landscape in the Lower Delta of the Parana River, Argentina. For this, we compared ground beetles communities among land uses with different intensive management and across seasons. We hypothesize that abundance, richness, and diversity of the ground beetles change due to factors such as planted tree species with their associated environmental conditions, type of implemented management, stand age, presence of cattle, and season (hypothesis 1). We predict that abundance, richness, and diversity of ground beetles to be greatest in the habitat types with the most vertical heterogeneity. Also, we hypothesize that each planted forest has a specific set of environmental conditions (environmental filters e.g., understory, soil characteristics) that select species with specific traits associated with these (hypothesis 2). We predict more typical wetland species within the lowland planted forests than in the highland planted forests because the first preserves soil conditions similar to wetland environments.
Materials and methods
Study area
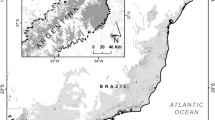
The Parana River Delta is a wetland macrosystem located along the final 320 km of the Parana Basin (Fig. 1). Its southernmost area, the Lower Delta (33° 45′S; 58° 51′W), encompasses approximately 4500 km2 of the mainland (southern Entre Rios province) and 3000 km2 of islands (northern Buenos Aires province, Sica et al. 2016). The hydrological regime is dominated by floods from the Parana River, combined with floods from Gualeguay and Uruguay rivers, tidal and storm surges from the De la Plata River estuary and local rainfall events. All these events produce a distinctive hydrological signature across the area (Baigún et al. 2008).
Map of the study area: A Forestal Nucleus in the Lower Delta of the Paraná River, B habitat types, C diagram of the arrangement of pitfall traps within each habitat type. MP Mature Poplar without cattle, YP Young Poplar without cattle, MW Mature Willow, YW Young Willow, MS Mature Silvopastoral poplar, YS Young Silvopastoral poplar. Pictures by Cicchino: Scarithes anthracinus (average length 3 cm), Polpochila flavipes (average length 1.5 cm), Selenophorus anceps (average length 1 cm)
The Lower Delta has a temperate-humid climate. The coldest month of the year corresponds to July, with an average temperature of 10.5 °C, while the warmest corresponds to January, with an average temperature of 24.4 °C. The average annual rainfall is 1066.1 mm, ranging from 230 in February to 31 mm in June (Malvárez 1999).
We focused on the insular area of the Lower Delta that has undergone a substantial transformation over more than 100 years, mainly due to hydrological changes caused by the construction of a dense network of dikes and canals for the establishment of planted forests with Salicaceae and, more recently, of silvopastoral systems (Quintana et al. 2014). As a result of these anthropogenic changes, the natural land cover composed of riparian forests and freshwater marshes had been degraded or destroyed (Quintana and Kalesnik 2008). This area represents the largest cover (c. 80,000 ha) of planted forests with Salicaceae in Argentina (Petray 2000), including willow (Salix babylonica, Salix nigra and hybrids), covering 75%, and poplar stands (Populus nigra, Populus deltoides and hybrids), covering 25% of the area (Borodowski 2006). Planted forests are not homogeneously distributed in the Lower Delta, but are concentrated in the so-called “Forest Nucleus” (Fig. 1), a 935 km2-area mainly occupied by tree-only systems, silvopastoral systems and ranches (Fracassi 2012; Quintana et al. 2014).
Planted forest management
Planted forests of willow and poplar are different systems. Willow trees are spaced at 3 × 2 m or 3 × 3 m and are primarily used for pulp production, while poplar trees, preferred for plywood and sawn timber, are spaced at 6 × 2 m, 6 × 4 m or 6 × 6 m. In general, planted forests of willow are not managed and are planted in lowlands that can retain waterlogging, unlike planted forests of poplar that are planted in highlands with landfill. Willow stand understory is dominated by Carex riparia subsp. chilensis and Rubus divaricatus. Young willow stands have a greater structural heterogeneity than mature willow stands, due to complementing species such as Eleocharis bonariensis and Hydrocotyle bonariensis (Bergonzi 2019). Management of young poplar plantations includes pruning to shape and land clearing using plows and rollers (Fracassi 2012). Pruning to shape consists of cutting the lower branches to favor the development of the tree trunk. The use of plows makes furrows in the soil to reduce compaction and eliminates the understory. The use of rollers crushes the herbaceous vegetation which reduces the competition with the young forest species. Management also involves the use of agrochemicals, which is recognized as a serious threat to biodiversity (Evans and Turnbull 2004). Mature poplar stands are not managed inside because they have a canopy closure that avoids the development of an abundant understory. Their understory is dominated by leaf litter and Carex bonariensis. Although young poplar stands are managed, they have abundant understory with the dominance of Lolium multiflorum and Eryngium pandanifolium (Krug 2018). The forest rotation of poplar planted forests shifts in the Lower Delta have an average of 17 years (Cobas et al. 2013). It is possible to obtain understory up to the 7th year of the rotation. From the eighth year, understory growth decreases due to the limited light penetrating the canopy (Cobas et al. 2013). Mátyás and Peszlen (1997) showed that the site conditions influence the maturation age of Populus deltoides implanted. In the region, juvenile to mature wood transition occurs between 7 and 9 years due to the rapid growth of Salicaceae (Borodowski 2006).
Silvopastoral systems integrate cattle husbandry with forest management (e.g., use of plows and rollers) in planted forests of poplar given their wider tree spacing. Both mature and young silvopastoral systems have canals or ditches as internal drinking fountains, unlike poplar planted forests without cattle where there is no such management. Cattle feed mainly on plants in the understory. The predominating grazing systems are continuous or involve rotations among stands. The annual average stocking rate varies between 0.3–0.5 and 0.7–1 LU/ha (LU: livestock unit) (Quintana et al. 2014). In the silvopastoral systems, the use of plows predominates over rollers. Young silvopastoral systems have a more diverse understory than mature ones, with species such as Eryngium pandanifolium, Carex spp., Carduus spp. and Solanum bonariense. The understory of mature silvopastoral systems is dominated by leaf litter. Species such as Muhlenbergia schreberi, Trifolium repens and Hydrocotyle bonariensis are less abundant (Krug 2018).
Sampling design
We characterized each habitat type in terms of three variables: tree species (willow and poplar), land-use (tree-only and silvopastoral system) and stand age (young: 5–6 years; mature: 11–14 years). We defined six habitat types: Young Willow (YW; ages = 5, 5, 5), Mature Willow (MW; ages = 14, 14, 14), Young Poplar without cattle (YP; ages = 5, 5, 5), Young Silvopastoral poplar (YS; ages = 6, 5, 5), Mature Poplar without cattle (MP; ages = 11, 11, 11) and Mature Silvopastoral poplar (MS; ages = 11, 11, 12) (Online Resource 1). We selected three stands for each habitat type (Fig. 1) of approximately 0.5 ha each one (n = 3, N = 18). The average distances between stands were 2.6 km (Min = 0.05 km; Max = 5.48 km).
Ground beetle sampling
We collected specimens using pitfall traps (Woodcock 2005) (diameter: 9 cm; height: 14 cm; volume: 600 ml). These contained 400 ml of a mixture of 2/3 of 96% alcohol and 1/3 of glycerin. We set six pitfall traps in each stand (Nper visit = 108) forming two triangles equal to 1.250 m2. Triangles were separated about 100 m from each other. Pitfall traps were separated about 50 m from each other (Fig. 1) and located centrally or at least 15 m away from field edges to avoid edge effect (Molnar et al. 2001). Traps were considered as independent observations because they were placed at a distance greater than 25 m (Woodcock 2005). We conducted this study from August 2012 to August 2014. We collected pitfall traps every 45 days, with two capture periods in all seasons (N = 1728) during two years. In the laboratory, we only counted the captured adult ground beetles and identified them up to the species level (Reichardt 1979). A reference collection was established with specimens deposited at the “Museo Argentino de Ciencias Naturales, Bernardino Rivadavia” and at the “Instituto de Investigación e Ingeniería Ambiental, UNSAM”.
Description of traits
Functional diversity has been proposed as an important feature of communities, enabling prediction of the rate and reliability of ecosystem processes (i.e., ecosystem function and ecosystem reliability, Mason et al. 2005). We selected the following functional traits for functional diversity analysis: dependence on soil moisture, adult feeding habits and seasonality.
Dependence on soil moisture (hydrophilic, mesophilic and xerophilic species) was selected because it has a direct association with the habitat requirements of each planted forest species and their management practices. Dependence on soil moisture (Table 1) was obtained from the bibliography (Sorensen 2006; Turienzo 2006) and field observation of the authors (Nanni et al. 2017). Adult feeding habits are highly varied (zoophagous, granivorous, and omnivorous species), with the majority of predatory species being polyphagous and consuming animal tissues, either from live prey or from carrion (Lövei and Sunderland 1996). There are also species with a partially or totally phytophagous diet as well as strict granivores in which the seeds are the main food (Kotze et al. 2011). This functional trait analysis of carabid diets can provide information about prey identity and help to determine predator–prey relationships, increasing knowledge of trophic links (Jelaska et al. 2014). Adult feeding habits (Table 1) was obtained from the bibliography (Cicchino and Farina 2005; Marasas et al. 2010; Nanni et al. 2017) and the direct observation of the mandibular structure from collected adults.
Finally, knowledge of seasonal fluctuations (Table 1) in ground beetle species abundance is important for adequate agroecosystem management, because it determines the reproductive period and consequently the survival of their populations (Castro et al. 2017). Surveying carabids and other invertebrates within a range of habitats over a vast region would not only help to understand the taxonomy, natural history, and biogeography of the species present, but would also help to develop guidelines for their conservation (Cividanes 2021).
Data analysis
We registered seasonal species richness (number of species), and we estimated seasonal species evenness and diversity. For the estimations, we used the Pielou and the Shannon–Wiener index, respectively (Pielou 1969; Shannon and Weaver 1949). We compared values of evenness and diversity with a generalized linear model (GLM) based on a Gaussian distribution with an identity link function and using a first-order autoregressive correlation matrix per replicate. We use these approaches because they are better fits than include replicate as a random factor. We considered season and habitat type as fixed factors (treatment). We modelled the variance with the varIdent function per habitat type. We analyzed the species richness with a generalized linear mixed model (GLMM) based on Poisson distribution with a log link function. We used this approach because species richness is a discrete variable. We considered season and habitat type as fixed factors (treatment) and the replicates as random factors. Including replicate as a random factor allows us to model the temporal dependence of the traps. Akaike’s information and parsimony criteria were used to select the final models (Crawley 2009).
To characterize each habitat type according to the composition of beetle communities, we computed a Principal Component Analysis (PCA) with a Hellinger pre-transformation (Borcard et al. 2011). We removed all rare species, i.e., those species that were recorded only once in a single sample during the study. Rare species do not characterize the species assemblage and its contribution to the principal components analysis is insignificant because it only contains one piece of data. Later, we evaluated species contribution to each component through Pearson’s correlation coefficients. To simplify the interpretation of the results, we plotted the centroids of the six habitat types for each season and the species with a significant correlation.
To evaluate and compare the different components of functional diversity in each habitat type, we assigned each species to a functional group using clusters with Gower function and Average linkage. We calculated the functional diversity indexes: functional richness (FRic), functional evenness (FEve), functional divergence (FDiv) and functional dispersion (FDis) (Mason et al. 2005). We compared them using GLMM based on a Gaussian distribution with an identity link function (Casanoves et al. 2010). We considered habitat type as a fixed factor and replicate as a random factor. We modelled the variance with the varIdent function per habitat type. Akaike’s information and parsimony criteria were used to select the final models (Crawley 2009). We did all analyses with the vegan package (Oksanen et al. 2019) of the R 3.6.0 (R Core Team 2019) and the Infostat (Di Rienzo 2011).
Results
We recorded 35 ground beetle species, distributed in 24 genera, and captured a total of 1896 individuals. Table 1 shows the species list collected during the study and their functional traits. Ground beetle species abundance per habitat type were given in Online Resource 2. Mature silvopastoral poplar, young poplar without cattle and young willow were the most abundant habitat types (Online Resource 2).
The general linear model of diversity showed a significant interaction among habitat types and seasons (F15,138 = 2.15; p = 0.0107). Young and mature willow and young and mature silvopastoral poplar reached the highest diversity in spring. The lowest diversity was found in young and mature poplar without cattle during summer and in mature willow during autumn (Fig. 2). The remaining combinations of habitat types and seasons yielded intermediate values.
Ground beetle diversity and evenness in a planted forest landscape in the Lower Delta of the Parana River (mean ± standard error). A Diversity (Shannon–Wiener index), B Evenness (Pielou index). MP Mature Poplar without cattle, YP Young Poplar without cattle, MW Mature Willow, YW Young Willow, MS Mature Silvopastoral poplar, YS Young Silvopastoral poplar. Winter (white), Autumn (light grey), Spring (grey) and Summer (black). Different letters indicate significant differences (p < 0.05)
On the other hand, there were significant differences in the generalized linear model of species richness among habitat types (F5138 = 5.09; p = 0.0003) and seasons (F3138 = 8.00; p < 0.0001), but not significant interaction (F15,138 = 1.38; p = 0.1637). Although no significant differences were found between mature silvopastoral poplar and young willow, the former showed higher species richness (Fig. 3). Young silvopastoral poplar, mature willow and young and mature poplars without cattle did not differ significantly in species richness. Young and mature poplars without cattle exhibited the lowest species richness. When comparing among seasons, species richness was significantly higher in spring than in other seasons (Fig. 3).
Ground beetle richness in a planted forest landscape in the Lower Delta of the Parana River (mean ± standard error). A Richness by habitat type, B Richness by season. MP Mature Poplar without cattle, YP Young Poplar without cattle, MW Mature Willow, YW Young Willow, MS Mature Silvopastoral poplar, YS Young Silvopastoral poplar. Different letters indicate significant differences (p < 0.05)
There was a significant interaction between habitat type and season for the general linear model of evenness (F15,138 = 5.27; p < 0.0001). The lowest evenness was recorded for mature and young poplar and young silvopastoral poplar in summer and young poplar and mature willow in autumn. The remaining combinations of habitat types and seasons yielded the highest evenness values (Fig. 2).
The first two PCA components (PC1 and PC2) accounted for 40.92% of the total variance (24.06% and 16.86% for PC1 and PC2, respectively). Paranortes cordicollis was significantly correlated with the PC1, while Scarites anthracinus and Argutoridius bonariensis were significantly correlated with the PC2. Loxandrus audouini was significantly correlated with both components (Table 2).
Regardless of the season, ground beetle communities in willow stands are located toward the positive end of PC1 and the positive end of PC2, differing from the rest of the habitat types. PC2 ordered the ground beetle communities according to the season, with spring and summer located toward the negative end of PC2 and autumn and winter toward its positive end (Fig. 4).
Principal component analysis (PC1 and PC2) of ground beetle species composition among habitat types in a planted forest landscape in the Lower Delta of the Parana River over the seasons. White—winter, light gray—spring, dark gray—autumn and black—summer. Gray ellipse shows willow planting sites. MP Mature Poplar without cattle, YP Young Poplar without cattle, MW Mature Willow, YW Young Willow, MS Mature Silvopastoral poplar, YS Young Silvopastoral poplar. We plotted only the species that presented a significant correlation. Paco Paranortes cordicollis, Arbo Argutoridius bonariensis, Loau Loxandrus audouini, Scaan Scarites anthracinus, Scame Scarites melanarius, Pastry Pachymorphus striatulus, Arab Argutoridius abacetoides, Asin Aspidoglossa intermedia, Oxfe Oxycheila femoralis, Pabre Paraclivina breviuscula and Peeg Pelmatellus egenus
Table 3 shows the abundance of individuals registered during our study by dependence on soil moisture and adult feeding habits. We observed that the abundance of zoophagous individuals was higher in the mature silvopastoral poplar. Mesophilic individuals were more abundant than hydrophilic and xerophilic individuals. Willow stand had more abundant hydrophilic individuals while poplar stand had more abundance of mesophilic individuals (Table 3).
Cluster analysis revealed eight functional groups during the study period (Table 4). Group 3 was characterized by zoophagous, mesophilic with annual activity species while group 4 was characterized by zoophagous, hydrophilic with annual activity species. Although functional groups 3 and 4 were the most abundant in all habitat types, group 3 was more abundant in poplar stands and group 4 in willow stands. Group 5 was characterized by zoophagous, hydrophilic summer activity species. Group 5 was mainly related to mature stands (independently of type) and young willow. Group 7 was exclusive to silvopastoral stands and group 8 was exclusive to young silvopastoral poplar. Group 6 was all land use. Groups 1 and 2 were the less abundant and showed no particular pattern (Table 4).
The general linear model showed significant differences for the indexes of functional divergence (FDiv) and functional evenness (FEve) (FDiv: F512 = 3.86, p = 0.0256; FEve: F512 = 3.27, p = 0.0430). Young and mature silvopastoral poplar and young willow reached the highest functional evenness (Table 5). Mature poplar was the only habitat type to differentiate in terms of functional divergence, presenting the lowest values (Table 5). We did not find significant differences in functional richness (FRic) and functional dispersion (FDis).
Discussion
In general, planted forests that replace natural ecosystems harm native biodiversity (Evans and Turnbull 2004). These changes in biodiversity depend on several factors, highlighting the type of habitat replaced (Corbelli et al. 2015). In particular, when the replaced ecosystem is a wetland, a drastic change in vegetation cover occurs and the natural hydrological regime is altered, losing the flood pulses. These changes limit the number of habitats available for wetland species (Magnano et al. 2019; Krug 2018; Nanni et al. 2019). In the Lower Delta of Parana River, there are few isolated remnants of natural grasslands and the original riparian forests have practically disappeared. The secondary forests are environments that most resemble the original riparian forests (Kalesnik and Sirolli 2011). These are abandoned planted forests of Salicaceae that were re-colonized by herbaceous, shrub, and arboreal native species and support a great number of wetland species (Magnano et al. 2019; Nanni et al. 2019). Our results indicate that planted tree species (willow or poplar) with their associated environmental conditions (light availability, soil conditions, waterlogging, understory vegetation) and the type of implemented management, contributed to the differences in ground beetles’ taxonomic diversity and richness. Particular associations between planted tree species and ground beetle communities have been observed before (Day et al. 1993; Chungu 2014; Kaizuka and Iwasa 2015). However, Stastna (2012) reported no difference in ground beetle communities between willow and poplar stands, probably, since these planted forests replaced grasslands, and they were set in areas with no difference in waterlogging. In our study, planted forests replaced wetlands. So, the differences in the ground beetle communities would be because the planted forest of willow are located in lowland areas within the dams and subject to temporary floods by rains that increase the richness of hydrophilic species, typical of wetland environments such as Loxandrus audouini and Aspidoglossa intermedia. Although planted forest of willow and poplar conserve many of the ground beetle species observed in other works for secondary forests in this region (Nanni et al. 2019), there is a loss of wetland species such as Apenes seriatus and Carbonellia platensis.
Cattle inside the planted forests of poplar were another factor that influenced ground beetles' richness. On one hand, cattle dung in silvopastoral systems offers new niches for soil arthropods. In particular, it is a suitable habitat for detritivorous species with different phenology (Cabrero-Sañudo and Lobo 2003), a fact reflected in the higher abundance of prey for zoophagous ground beetle species. We found that the abundance of zoophagous ground beetle species was much higher in silvopastoral systems than in the other habitat types. On the other hand, some management activities performed in silvopastoral systems such as shape pruning and/or weed clearing using rollers are known to affect soil microenvironmental conditions by increasing solar radiation and temperature, but also to favor the establishment of terrestrial open-habitat species (Kwiatkowski 2011; Batáry et al. 2007). Moreover, hydrophilic species found in these habitat types are likely favored by canals and ditches that act as a sink for freshwater for cattle. Alternatively, the adjacent patches of each stand could also affect the diversity due to the species flow, as seen in Magura and Lövei (2019). In future studies, adjacent patches could be incorporated at the landscape level to understand these potential interactions.
Also, ground beetles’ richness was modified by the stand age. Young willow showed higher species richness compared to mature willow in agreement with Rubio (2004) for the same region. Young willow stands have more vertical heterogeneity than mature stands due to an abundant understory dominated by shrubs (Casaubón et al. 2001). Ground beetles are typical soil beetles, usually found on caves, under rocks and litter, but some groups eventually inhabit trees and shrubs (Moraes et al. 2013). So, this vertical heterogeneity would support high species diversity because of an increased number of habitats available (Hardersen et al. 2020).
Seasonality is another modelling factor of the ground beetles’ richness and diversity. The spring was the most diverse and rich season for different habitat types. Although seasonality is species-specific, we detected patterns of richness for spring coinciding with active time reported for other ground beetle species in other parts of the world (Cartellieri and Lövei 2003). The environmental temperature during spring favors the activity of many ground beetles (Saska et al. 2013).
The habitat type mature poplar without cattle showed a low species diversity and richness, but the highest evenness. This suggests a stable climax community (Magurran 2005) resulting from the lack of forestry management practices. In addition, generalist species are most likely favored in highlands-where poplar stands are planted- and by the absence of cattle, because in the other habitat types they may compete with flood-tolerant, specialist species. Such predominance of generalist species has been previously reported for other planted forests of poplar with similar management practices (Ulrich et al. 2004; Stastna 2012). The low evenness of the community in young poplar (with a single dominating species, Paranortes cordicollis) is in line with that reported by Allegro and Sciaky (2003) for this habitat type. This result would be due to the presence of a dense cover of herbaceous vegetation that dominates the understory and affects the locomotion of medium- to large-sized species (Allegro and Sciaky 2003).
Thereby, the richness and diversity of the ground beetles change due to factors such as planted tree species with their associated environmental conditions, type of implemented management, stand age, presence of cattle, and season, as suggested in our hypothesis 1.
Functional evenness may be seen as the degree to which the biomass of a community is distributed in niche space to allow effective utilization of the entire range of resources available to it (Mason et al. 2005). The high functional evenness observed in young willow, and young and mature silvopastoral poplar implies that a large part of the functional niches was used. This situation would indicate an uniform exploitation of the niche space occupied by the different functional groups in these habitat types (Mason et al. 2005). The lowest functional divergence of mature poplar was likely because it is a stable climax community with a few dominant species, as mentioned above. Besides, the planted tree species determine the functional groups of the ground beetle. The planted tree species are an environmental filter, due to differences in waterlogging. These differences are reflected in the dependence on soil moisture of groups 3 (mesophilic species) and 4 (hydrophilic species). Also, both groups include annual zoophagous species. The high abundance of top predators suggests that the study habitat types harbor a large number of prey species and are environmentally complex (Liu et al. 2015). The xerophilic species Selenophorus anceps, the only member of the functional group 8, is unexpected in a wetland ecosystem. The finding of a terrestrial species in young silvopastoral poplar reflects the deep changes in the environmental conditions caused by the ongoing anthropogenic activities (Porrini et al. 2014) contributes to wetland desiccation. Alternatively, the appearance of this species could be due to the dispersal from other neighboring environments (Scharff et al. 2003), or the dispersal, mediated by anthropic actions such as the movement of machinery from the mainland to the islands. It is worthy of noting that the ground beetle functional groups represented in each habitat type are conditioned by environmental filters (Keddy 1992) associated with the management practices used according to the tree species (e.g., construction of dams and other hydraulic structures, use of plows and rollers to reduce the understory), as we hypothesized.
In conclusion, the planted tree species with their associated environmental conditions, stand age, and presence of cattle inside the plantations have relevant implications for planted forest management and taxonomic and functional diversity of ground beetles in the Lower Delta of the Parana River. Based on our results, planted forests of willow (set in lowland areas) host a greater number of hydrophilic species (typical of wetland) than planted forests of poplar. So, we recommend using willow species rather than poplar species when planted forests replace fluvial wetlands. Planted forests of willow are flood-resistant, thus if planted in lowlands can retain waterlogging. In fluvial wetlands, planted forests of willow are not managed, favoring occurrence of hydrophilic ground beetles. Silvopastoral planted forests were also habitat types suitable for the development of some hydrophilic species, probably due to freshwater sources for cattle. For this reason, increasing irrigation of poplar planted forests without cattle through the addition of ditches and canals would increase soil moisture, thus favoring hydrophilic species. Several of these ground beetles are potential predators of forest pests (Nanni et al. 2015). Therefore, if their populations are abundant, they could become natural enemies of pest species that are detrimental to production (Nanni et al. 2021). Also, other works showed that adding canals and ditches within poplar planted forests has a positive effect on the diversity of other taxa (e.g., mammals and birds) (Fracassi et al. 2017; Krug et al. 2019). Conserving or restoring different strata of understory to increase vertical heterogeneity would increase the number of habitats available for ground beetles, for example, reducing weed cleaning (e.g., use of plow and roller). Finally, the maintenance of heterogeneous landscape mosaic with different species trees, stand age, and silvopastoral systems could host almost the entire community of ground beetles found in secondary forests (Nanni et al. 2019). We recommend carrying out these management measures throughout planted forests that replace fluvial wetlands in areas with similar climate and conditions. These management measures favor ground beetle diversity, particularly hydrophilic species whose niche is limited by being in a planted forest landscape under intensive management. We believe in the need to implement management practices that are ecosystem friendly and that guarantee the conservation of biodiversity and the sustainable use of these modified fluvial wetlands.
Data availability
The generated and analysed datasets during the current study are available from the corresponding author on reasonable request.
References
Allegro G, Sciaky R (2003) Assessing the potential role of ground beetles (Coleoptera, Carabidae) as bioindicators in poplar stands, with a newly proposed ecological index (FAI). For Ecol Manag 175:275–284
Baigún CR, Puig A, Minotti PG, Kandus P, Quintana R, Vicari R, Bó R, Oldani N, Nestler JA (2008) Resource use in the Parana river delta (Argentina): moving away from an ecohydrological approach? Ecohydrol Hydrobiol 8:2–4
Batáry P, Báldi A, Szél G et al (2007) Responses of grassland specialist and generalist beetles to management and landscape complexity. Divers Distrib 13(2):196–202
Bergonzi A (2019) Análisis de la estructura de la vegetación en humedales del Bajo Delta del río Paraná sometidos a distintas modalidades productivas. PhD thesis. Facultad de Ciencias Exactas y Naturales, Universidad de Buenos Aires
Binkley D, Fisher R (2012) Ecology and management of forest soils. Wiley, Hoboken
Borcard D, Gillet F, Legendre P (2011) Numerical ecology with R. Springer Science & Business Media, New York
Borodowski ED (2006) Álamos y sauces en el Delta del Paraná: situación del sector y silvicultura. Actas Jornadas de Salicáceas, Buenos Aires, pp 61–70
Bremer LL, Farley KA (2010) Does plantation forestry restore biodiversity or create green deserts? A synthesis of the effects of land-use transitions on plant species richness. Biodivers Conserv 19(14):3893–3915
Cabrero-Sañudo FJ, Lobo JM (2003) Reconocimiento de los factores determinantes de la riqueza de especies: El caso de los Aphodiinae (Coleoptera, Scarabaeoidea, Aphodiidae) en la península ibérica. Graellsia 59(2–3):155–177
Cartellieri M, Lövei GL (2003) Seasonal dynamics and reproductive phenology of ground beetles (Coleoptera: Carabidae) in fragments of native forest in the Manawatu, North Island. New Zealand n z J Zool 30(1):31–42
Casanoves F, Pla L, Di Rienzo JA, Díaz S (2010) FDiversity: a software package for the integrated analysis of functional diversity. Methods Ecol Evol. https://doi.org/10.1111/j.2041-210X.2010.00082.x
Casaubon EA, Gurini LB, Cueto GR (2001) Diferente calidad de estación en una plantación de Populus deltoides cv Catfish 2 del Bajo Delta bonaerense del Río Paraná (Argentina). For Syst 10(2):217–231
Castro AV, Porrini DP, Cicchino AC (2017) Diversidad de Carabidae (Insecta: Coleoptera) en distintos ambientes de un agroecosistema del sudeste bonaerense, Argentina. Ecol Austral 27:252–265
Chungu D (2014) Impact of land use on assemblages of carabid beetles (Coleoptera: Carabidae) in Zambia. PhD thesis. Department of Biology of the University of Marburg
Cicchino AC (2006) Diversidad de Carábidos (Insecta, Coleoptera, Carabidae) de dos asociaciones de tala en la Laguna de los Padres, partido de General Pueyrredón, provincia de Buenos Aires. In: Mériday E, Athor J (eds) Talares Bonaerenses y su Conservación. Fundación de Historia Natural Félix de Azara-Universidad Maimónides, Buenos Aires, pp 128–136
Cicchino AC, Farina JL (2005) Carabidofauna de los suelos colindantes con la laguna litoral de la Reserva Natural del Puerto de Mar del Plata, provincia de Buenos Aires, Argentina. In: V REBIOS, San Salvador de Jujuy, 2005, Área temática I, Comunidades Terrestres III, pp 1–15
Cividanes FJ (2021) Carabid beetles (Coleoptera: Carabidae) and biological control of agricultural pests in Latin America. Ann Entomol Soc Am. https://doi.org/10.1093/aesa/saaa051
Cobas AC, Area MC, Monteoliva S (2013) Juvenile to mature wood transition in one clone of Populus deltoides implanted in Buenos Aires, Argentina. Maderas-Cienc Tecnol 15(2):223–234
Corbelli JM, Zurita GA, Filloy J, Galvis JP, Vespa NI, Bellocq I (2015) Integrating taxonomic, functional and phylogenetic beta diversities: interactive effects with the biome and land use across Taxa. PLoS ONE 10(5):e0126854. https://doi.org/10.1371/journal.pone.0126854
Crawley MJ (2009) The R book. Wiley, London
Day KR, Marshall S, Heaney C (1993) Associations between forest type and invertebrates: ground beetle community patterns in a natural Oakwood and juxtaposed conifer plantations. Forestry 66(1):37–50
Di Rienzo JA (2011) “InfoStat versión 2011” Grupo InfoStat, FCA, Universidad Nacional de Córdoba, Argentina. http://www.infostat.com.ar
Dias RA, Bastazini VA, Gonçalves MS, Bonow FC, Müller SC (2013) Shifts in composition of avian communities related to temperate-grassland afforestation in Southeastern South America. Iheringia Ser Zool 103:12–19
Dı́az S, Cabido M (2001) Vive la difference: plant functional diversity matters to ecosystem processes. Trends Ecol Evol 16(11):646–655
Evans J, Turnbull JW (2004) Plantation forestry in the tropics: the role, silviculture, and use of planted forests for industrial, social, environmental, and agroforestry purposes, 3rd edn. Oxford University Press, Oxford
FAO (2010) Evaluación de los recursos forestales mundiales 2010: Informe principal. Organización de las Naciones Unidas para la Agricultura y la Alimentación. Italy
Fracassi NG, Pereira JA, Mujica G, Hauri B, Quintana RD (2017) Estrategias de conservación de la biodiversidad en paisajes forestales del Bajo Delta del Paraná-uniendo a los actores clave de la región. Mastozool Neotrop 24(7):59–68
Fracassi N (2012) Diversidad de mamíferos y aves en pajonales y forestaciones de salicáceas del Bajo Delta del río Paraná. Magister de la Universidad de Buenos Aires, Área Recursos Naturales, Facultad de Agronomía
Gormally M, Fahy O (1998) A comparison of plant and carabid beetle communities in an Irish oak woodland with a nearby conifer plantation and clearfelled site. For Ecol Manag 110:263–273
Hardersen S, Macagno ALM, Chiari S, Audisio P, Gasparini P, Lo Giudice G, Nardi G, Masong F (2020) Forest management, canopy cover and geographical distance affect saproxylic beetle communities of small-diameter beech deadwood. For Ecol Manag 467:118–152. https://doi.org/10.1016/j.foreco.2020.118152
Jelaska LS, Franjevic D, Jelaska SD, Symondson WOC (2014) Prey detection in carabid beetles (Coleoptera: Carabidae) in woodland ecosystems by PCR analysis of gut contents. Eur J Entomol 111:631–638
Kaizuka J, Iwasa M (2015) Carabid beetles (Coleoptera: Carabidae) in coniferous plantations in Hokkaido, Japan: effects of tree species and environmental factors. Entomol Sci 18(2):245–253
Kalesnik FA, Sirolli H (2011) La vegetación del Bajo Delta Insular del Paraná. Diversidad, situación y uso. In: RD Quintana y otros (Eds) El Patrimonio natural y cultural del Bajo Delta Insular del Río Paraná. Bases para su conservación y uso sostenible. 317: 89–100.
Keddy PA (1992) Assembly and response rules: two goals for predictive community ecology. J Veg Sci 3(2):157–164
Koivula M (2001) Carabid beetles (Coleoptera, Carabidae) in boreal managed forests—meso-scale ecological patterns in relation to modern forestry. PhD thesis, University of Helsinki, p. 120
Kotze DJ, Brandmayr P, Casale A, Dauffy-Richard E, Dekoninck W, Koivula MJ, Lövei GL et al (2011) Forty years of carabid beetle research in Europe: from taxonomy, biology, ecology and population studies to bioindication, habitat assessment and conservation. ZooKeys 100:55–148
Krug P (2018) Integridad ecológica de los humedales del Bajo Delta del Río Paraná bajo diferentes modalidades productivas. PhD Thesis. Facultad de Ciencias Exactas y Naturales, Universidad de Buenos Aires
Krug P, García Cabrera MS, Quintana RD (2019) Salicaceae afforestations: advantage or disadvantage for Neotropical otter in its southernmost distribution? Hystrix It. J Mamm 30(2):166–171
Kwiatkowski A (2011) Assemblages of carabid beetles (Coleoptera, Carabidae) in humid forest habitats of different stages of succession in the Puszcza Knyszynska forest (Northeastern Poland). Zookeys 100:447–459
Liu Y, Duan M, Zhang X, Yu Z, Axmacher JC (2015) Effects of plant diversity, habitat and agricultural landscape structure on the functional diversity of carabid assemblages in the North China Plain. Insect Conserv Divers 8(2):163–176
Lövei GL, Sunderland KD (1996) The ecology and behavior of ground beetles (Coleoptera: Carabidae). Annu Rev Entomol 41:241–256
MacGregor J (2002) Global Trends in Trade of Forest Products and services presents at the FAO Expert Consultation on Trade and Sustainable Forest Management: Impact and Interactions. Italy
Magnano AL, Fracassi NG, Nanni AS, Quintana RD (2019) Changes in bird assemblages in an afforestation landscape in the lower delta of the Paraná River, Argentina. Emu-Austral Ornith 119(4):346–354
Magura T (2002) Carabids and forest edge: spatial pattern and edge effect. For Ecol Manag 257:23–37
Magura T (2017) Ignoring functional and phylogenetic features masks the edge influence on ground beetle diversity across forest-grassland gradient. For Ecol Manag 384:371–377
Magura T, Lövei G (2019) Environmental filtering is the main assembly rule of ground beetles in the forest and its edge but not in the adjacent grassland. Insect Sci 26:154–163
Magura T, Tothmeresz B, Bordan Z (2000) Effects of nature management practice on carabid assemblages (Coleoptera: Carabidae) in a non-native plantation. Biol Conserv 93:95–102
Magura T, Tothmeresz B, Elek Z (2003) Diversity and composition of carabids during a forestry cycle. Biodivers Conserv 12:73–85
Magura T, Lövei GL, Tóthmérész B (2017) Edge responses are different in edges under natural versus anthropogenic influence: a meta-analysis using ground beetles. Ecol Evol 7:1009–1017
Magurran AE (2005) Measuring biological diversity. Blackwell Publishing Company, Hoboken
Malvárez AI (1999) El Delta del Río Paraná como mosaico de humedales. Tópicos sobre humedales subtropicales y templados de Sudamérica. p. 35–54
Marasas ME, Sarandón SJ, Cicchino AC (2010) Semi-natural habitats and field margins in a typical agroecosystem of the Argentinean pampas as a reservoir of carabid beetles. J Sustain Agric 34:1–16
Mason NW, Mouillot D, Lee WG, Wilson JB (2005) Functional richness, functional evenness and functional divergence: the primary components of functional diversity. Oikos 111(1):112–118
Mátyás C, Peszlen I (1997) Effect of age on selected wood quality traits of poplar clones. Silvae Genet 46(2):64–72
Molnar T, Magura T, Tothmérész B, Elek Z (2001) Ground beetles (Carabidae) and edge effect in oak-hornbeam forest and grassland transects. Eur J Soil Biol 37(4):297–300
Moraes RM, de Souza MM, Ott R (2013) Carabid beetle assemblages in three environments in the Araucaria humid forest of Southern Brazil. Rev Bras Entomol 57(1):67–74
Nanni AS, Cicchino AC, Quintana RD (2015) Can Carabidae of the Paraná River Delta control forestry plagues? Enfoques interdisciplinarios para la sustentabilidad del ambiente. II Congreso Internacional de Ciencia y Tecnología Ambiental. II Congreso Nacional de la Sociedad Argentina de Ciencia y Tecnología Ambiental, Argentina, pp 151–156
Nanni AS, Quintana RD, Fracassi NG, Cicchino AC (2017) Ground beetles (Coleoptera: Carabidae) inhabiting anthropogenic habitats in the lower delta of the Paraná River, Argentina: geographic distribution and ecological characteristics. Austral Entomol 56:244–254
Nanni AS, Fracassi NG, Magnano AL, Cicchino AC, Quintana RD (2019) Ground beetles in a changing world: communities in a modified wetland landscape. Neotrop Entomol 48(5):729–738
Nanni AS, Quintana RD, Sfara V (2021) Could Scarithes anthracinus (Coleoptera: Carabidae) be a biological control agent of Nematus oligospilus (Himenoptera: Tenthredinidae)? III Jornadas Internacionales y V Nacionales de Ambiente. In press
Niemelä J, Koivula M, Kotze DJ (2007) The effects of forestry on carabid beetles (Coleoptera: Carabidae) in boreal forests. J Insect Conserv 11(1):5–18
Oksanen J, Blanchet FG, Friendly M, Kindt R, et al. (2019) vegan: Community Ecology Package. R package version 2.5–5. https://github.com0/begandevs/began
Paillet Y, Bergès L, Hjalten J et al (2010) Biodiversity differences between managed and unmanaged forests: meta-analysis of species richness in Europe. Conserv Biol 24:101–112
Paleologos M, Cicchino A, Marasa ME, Sarandon SJ (2007) Las estructuras de dominancia de los ensambles carabidológicos como indicadoras de disturbio en agroecosistemas. Un ejemplo en dos viñedos bajo diferentes manejos en la costa de Berisso, Buenos Aires. Rev Bras Agroecol 2(2):655–659
Petray E (2000) Las actividades relativas al cultivo y la utilización del álamo y del sauce. Período 1966–1999. Comisión Nacional del Álamo de Argentina.
Pielou EC (1969) An introduction to mathematical ecology. Wiley, Hoboken
Porrini DP, Castro AV, Cicchino AC (2014) Los carábidos (Coleoptera: Carabidae) asociados a los remanentes de bosque nativo en la Reserva Natural Municipal Laguna de los Padres. Buenos Aires Rev Soc Entomol Arg 73(1–2):23–37
Quintana RD, Kalesnik FA (2008) Wetlands modification and wildlife habitat suitability: can capybaras survive in a matrix of poplar and willow afforestations? Insugeo, Misceláncea 17:161–168
Quintana RD, Bó R, Astrada E, Reeves C (2014) Lineamientos para una ganadería ambientalmente sustentable en el Delta del Paraná, 1st edn. Fundación Humedales/Wetlands International, Buenos Aires
R Core Team (2019) R: a language and environment for statistical computing. R 392 Foundation for Statistical Computing, Vienna, Austria. http://www.r393project.org/.r Development Core Team, Vienna, Austria
Reichardt H (1979) The South American carabid fauna: endemic tribes and tribes with African relationships. In: Erwin TL, Ball GE, Whitehead DR (eds) Carabid beetles: their evolution natural history and classification. The Hague, Dr. W. Junk, Boston, pp 319–325
Rubio A (2004) Variaciones en comunidades de artrópodos durante el turno de cosecha del Sauce Americano (Salix babylonica) en el Delta del Paraná. Lic. Thesis. Facultad de Ciencias Exactas y Naturales. Departamento de Ecología, Genética y Evolución
Saska P, van der Werf W, Hemerik L, Luff ML, Hatten TD, Honěk A (2013) Temperature effects on pitfall catches of epigeal arthropods: a model and method for bias correction. J Appl Ecol 50:181–189
Scharff N, Coddington JA, Griswold CE, Hormiga G, Bjorn P (2003) When to quit? Estimating spider species richness in a northern European deciduous forest. J Arachnol 32:246–276
SENASA (Servicio Nacional de Sanidad y Calidad Agroalimentaria) (2015) Bosques Argentinos, Actividad Forestal y Economías Regionales. Infografía
Shannon CE, Weaver W (1949) The mathematical theory of communication. University Illinois Press, Urbana
Sica YV, Quintana RD, Radeloff VC, Gavier-Pizarro GI (2016) Wetland loss due to land use change in the lower Paraná river delta, Argentina. Sci Total Environ 568:967–978
Sica Y, Gavier-Pizarro G, Pidgeon A, Travaini A, Bustamante J, Radeloff V, Quintana RD (2018) Changes in bird assemblages in a wetland ecosystem after 14 years of intensified cattle activity. Austral Ecol 43:786–797
Sorensen M (2006) Composición y riqueza específica de carábidos (Coleoptera: Carabidae) en paisajes agrícolas de la E.E.A. INTA Balcarce, sudeste de Buenos Aires. Lic. Thesis. FCEyN, Universidad Nacional de Mar del Plata., Mar del Plata. Argentina
Stastna P (2012) Diversity of ground beetles (Carabidae) in the plantations of fast growing trees. Acta Universitatis Agriculturae Et Silviculturae Mendelianae Brunensis, Volumen LX 6:309–315
Stephens S, Wagner MR (2007) Forest plantations and biodiversity: a fresh perspective. J For Res 105(6):307–313
Suárez RP, Goijman AP, Cappelletti S, Solari LM, Cristos D, Rojas D, Krug P, Babbitt KJ, Gavier-Pizarro GI (2021) Combined effects of agrochemical contamination and forest loss on anuran diversity in agroecosystems of east-central Argentina. Sci Total Environ. https://doi.org/10.1016/j.scitotenv.2020.143435
Turienzo P (2006) Estudio de una comunidad estival de Carabidae (Insecta: Coleoptera) en seis ambientes diferentes del centro del partido de Balcarce (Buenos Aires). Lic. Thesis. Facultad de Ciencias Agrarias, Universidad Nacional de Mar del Plata. Mar del Plata. Argentina
Ulrich W, Buszko J, Czarnecki A (2004) The contribution of poplar plantations to regional diversity of ground beetles (Coleoptera: Carabidae) in agricultural landscapes. Ann Zool Fennici. 41:501–512
Woodcock BA (2005) Pitfall trapping in ecological studies. In: Leather SR (ed) Insect sampling in forest ecosystems. Wiley, Hoboken, pp 37–57
Acknowledgements
We thank the Instituto de Investigación e Ingeniería Ambiental (IIIA), CONICET-UNSAM. We are grateful to the Las Carabelas staff of Papel Prensa S.A. and to the Gómez family for allowing access during this field study.
Funding
This work was supported by the projects PICT Bicentenario 2227 and PICT 2982 of the National Agency for Science and Technology Promotion, Argentina, and PIP 0092 of the National Council for Scientific and Technological Research (CONICET).
Author information
Authors and Affiliations
Contributions
AN, AC, RQ: Conceptualization, Methodology: AN; Formal analysis and investigation: AN, PK; Writing—original draft preparation: AN, PK; Writing—review and editing: AN, PK, AC, RQ; Funding acquisition: RQ; Resources: RQ, AC; Supervision: RQ. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Communicated by Andreas Schuldt.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article belongs to the Topical Collection: Forest and plantation biodiversity.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Nanni, A.S., Krug, P., Cicchino, A.C. et al. Effects of intensive human management on the taxonomic and functional diversity of ground beetles in a planted forest landscape. Biodivers Conserv 30, 3717–3735 (2021). https://doi.org/10.1007/s10531-021-02273-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-021-02273-w