Abstract
Microbial fuel cells (MFCs) face major hurdles for real-world applications as power generators with the exception of powering small sensor devices. Despite tremendous improvements made in the last two decades, MFCs are still too expensive to build and operate and their power output is still too small. In view of this, in recently years, intensive researches have been carried out to expand the applications into other areas such as acid and alkali production, bioremediation of aquatic sediments, desalination and biosensors. Unlike power applications, MFC sensors have the immediate prospect to be practical. This review covers the latest developments in various proposed biosensor applications using MFCs including monitoring microbial activity, testing biochemical oxygen demand, detection of toxicants and detection of microbial biofilms that cause biocorrosion.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
As shown in Fig. 1, a classical microbial fuel cell (MFC) setup has an anodic chamber with an anode covered with an anaerobic biofilm to digest an organic carbon (electron donor) source for the extraction of electrons. The electrons are passed to the anode by the electrogenic anodic biofilm. Electrons flow from the anode via an external load (used to harvest the electric power) to a cathode where they are used for the reduction of an oxidant (electron acceptor) such as oxygen on its surface. The following anodic and cathodic reactions use acetate as an example for organic carbon with oxygen as the terminal electron acceptor for the MFC in Fig. 1. Energy is harvested by the external load.
MFCs have been investigated intensively in the past two decades for their potential applications in electricity generation from various types of organic matters, especially those in wastewater. Tremendous improvements have been made in various aspects of MFCs including better reactor designs, less expensive anode and cathode materials and much improved power output (Ren et al. 2015; Liu et al. 2015; Zhang et al. 2015). MFC power output has been improved by 104-fold in the last two decades (Debabov 2008). However, major hurdles still exist for MFCs in practical power-generation applications. The highest reported power density is about several W m−3 under optimized conditions, which is far from the target value of 1 kW m−3 for an energy self-sufficient implementation (Li et al. 2014), but is far greater than that needed for a small sensor device.
There is still a long way to go for MFCs to be feasible as power generators except powering small sensor devices (Zhou et al. 2013). In recent years, more attention has been paid to hybrid MFCs applications such as acid and alkali production, desalination and biosensors (Chen et al. 2012). One particularly bright spot in recent MFC research is their application as biological sensors.
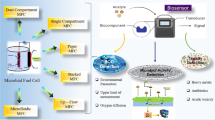
A biosensor is an analytical device containing one or more biological sensing elements and transducers to produce a signal output (Lei et al. 2006). Due to ever increasing concerns over the environment and stricter environmental regulations, there is growing demand for pollution monitoring, especially water quality monitoring (Sara et al. 2006). Compared with conventional sensors, electrochemical biosensors especially MFC-based biosensors have shown great promises in recent years due to their advantages, including high sensitivity, stability, and applicability for remote sites without an electricity supply (Jouanneau et al. 2014; Sara et al. 2006). Researchers have investigated MFC-based biosensor devices to test BOD, microbial activity, microorganism load, presence of corrosive biofilms and substances with cytotoxicity (Table 1).
MFC voltage output and power output are both influenced by various environmental factors such as organic compound type and concentration, temperature, pH, toxicants and inhibitors. This means apart from serving as an alternate power source for remote sensors, MFCs themselves may be used as sensors to detect many parameters (Jiang et al. 2015; Modin and Wilén 2012; Uríaa et al. 2012). Though some reviews (Dewan et al. 2014; Sun et al. 2015) briefly mention MFC sensors with limited applications, this present review summarizes the latest developments in various biosensor applications using MFCs including monitoring microbial activity, testing BOD, detection of toxicants, detection of microbial biofilms that cause biocorrosion and some other unusual application.
Different types of MFC-based biosensors
BOD sensor for wastewaters
Biochemical oxygen demand (BOD) is a key parameter for water quality. A conventional assay is to incubate a water sample at 20 °C for 5 days and compare its dissolved oxygen (DO) with that before incubation (Rice et al. 2012). This method is time and labor intensive, and the data sometimes are not consistent. Therefore, it is desirable to develop alternative methods. One promising alternative is to use an MFC for real-time and on-line BOD monitoring. Once the MFC reactor has acclimated, it can detect BOD in its feed water stream with fast response, good sensitivity and a wide measurement range with low maintenance (Jouanneau et al. 2014).
Some conventional BOD biosensors utilize physical transducers to monitor the change of the dissolved O2 (DO) or light emission caused by bacterial respiration, and then use the change of the electrical or optical signal to estimate the BOD value. However, a power supply is needed for these sensors (Liu and Mattiasson 2002). In comparison, an MFC-based BOD sensor is self-powered. It readily provides the voltage and current output without a transducer (Peixoto et al. 2011). The current output or the coulombic efficiency (CE) from the MFC has a linear relationship with the (organic carbon) strength of the wastewater (Chang et al. 2004; Peixoto et al. 2011). This makes MFC-based BOD sensors attractive for practical applications. Min and Angelidaki (2008) designed a submersible MFC (SMFC)-based sensor to detect BOD. Both the anode and cathode electrodes in the anaerobic reactor were submerged in water (Fig. 2a). Instead of partitioning the reactor vessel to provide a cathodic chamber, a proton exchange membrane provides an envelope that serves as the cathode chamber. Air is fed into the envelope by a tube to allow oxygen reduction on the cathode.
Similar to MFCs for power generations, there are many factors that affect the stability and sensitivity of the MFC-based BOD sensors, such as the DO concentration in the cathode compartment, external resistance and effluent flow rate. Optimization of the various operating conditions can improve the performance of the BOD sensors. The effluent flow rate in a continuous flow BOD sensor has a direct relationship with the response time and CE. Chang et al. (2004) found that the CE of their BOD sensor was over 90 % when the feeding rate was lower than 0.71 ml/min and the yield fell to 77 % at 1.20 ml/min. The decrease was due to an insufficient residence time for fuel consumption.
High reproducibility and good performances using different organic matters as fuel substances are key advantages for an MFC-based BOD sensor. However, it still has some drawbacks because bacteria varieties are often limited, the limited types of organic substances present and the metabolic rates are much lower than those in other typical biosensors. Therefore, to improve the performances of the sensors, research is underway to select better electrogenic microorganisms or microbial biofilm consortia that are capable of metabolizing different organic substances (Abrevaya et al. 2015a).
Toxicity sensors
Chemical analysis tools, such as GC, GC–MS, HPLC and LC–MS, are routinely used to detect toxic chemicals. However, they are not suitable or are too expensive for on-site real-time monitoring (Choi and Gu 2003). MFC-based sensors can fill the void, providing a simple, fast, sensitive and low maintenance method. Toxic substances inhibit the performance of biofilms. An inhibition rate (I) is used to show the effect of a toxic substance. I can be calculated by the following equation (Kim et al. 2007):
The coulombic yield (CY) is calculated by integrating MFC current output over time. CYnor and CYtox are the CY of the MFC reactor fed with normal wastewater and toxic wastewater, respectively. When a toxin is present in the effluent to the MFC reactor, the electrogenic biofilm covering the anode will be inhibited in its ability to oxidize the organic matter in the effluent. Fewer electrons are harvested, resulting in fewer electrons transferred to the anode by the biofilm. This is reflected by a decline in the observed current output (Fig. 2b). Thus, MFC-based toxicity biosensors can be standardized to yield quantitative results because the inhibition rate or the decline of current output is related to amounts of the toxic substances. A new on-line biomonitoring system using an MFC for the detection of toxic compounds in water systems has been developed (Kim et al. 2007).
Davila et al. (2011) microfabricated a 144 μl sensor (Fig. 2c) consisting of a proton exchange membrane placed between two silicon wafers (anode and cathode). Geobacteraceae sulfurreducens was used to form the anodic biofilm to oxidize organic carbon in the effluent. O2 was reduced on the cathodic surface. The device performed well as a toxicity biosensor when tested with formaldehyde.
Various types of toxicants can be monitored by MFC-based sensors. Kim et al. (2007) detected Cd(II) and Pb(II) at 0.1 to 1 mg/l using a dual chamber MFC. Sodium dodecyl sulfate and bentazon at, respectively, 10–50 mg/l and 1–3 mg/l were also been monitored using an MFC-based sensor (Stein et al. 2012b).
Compared with traditional methods, using MFC-based sensors to detect toxic chemicals has many advantages. However, there are still some shortcomings, such as non-specificity for the involved compounds and the requirement for microorganisms with sufficiently high biological activities. Furthermore, the main disadvantage limiting their application is due to the low sensitivity, which needs optimization in terms of control modes and flow configurations (Jiang et al. 2015).
Monitoring microbial activities
MFC-based sensors can also monitor microbial activities (MA) and the number of microorganism in situ using relationships between the current and the microorganisms. The principle of in situ monitoring of MA and quantification of microbial biomass can be divided into two categories. One of the common methods used to determine MA is through the measurement of microbial respiration. Tront et al. (2008) found that the current of their MFCs correlated with the activity of the biofilm covering the anodes because the respiration of the sessile cells was responsible for the electron transfer to the anode. One limitation of this method is that this type of MFC-based biosensor is good only for anaerobic biofilms. Another drawback is that the sensors assessed the MA of the anodic biofilms and used the MA to reflect sessile cells elsewhere at a monitoring site. Thus, the measurements were indirect and might be inaccurate since the anodic biofilm could change over time during the operation of the MFC-based sensor. Fresh anodes are needed from time to time to allow the adhesion of new biofilms that better reflect the microbes at the monitoring site (Jiang et al. 2010).The other method used to monitoring MA is through utilizing other suitable parameters to replace the biomass concentration as an expression of the active microorganisms concentration. Zhang and Angelidaki (2011) selected the concentration of ATP, that could indicate the total number of living microorganisms accurately as the indicator for the presence of microbial activities and then investigated the relationship between ATP concentration and current density. The results showed that the current density had a linear relationship with the concentrations of active microorganisms from 0 to 6.5 nM ATP. Compared with the first method, this method is faster and more accurate.
One specific example is the fast detection and quantification of Escherichia coli. Patchett et al. (1988) tested an MFC with thionine as an electron mediator that exhibited a proportional relationship between the steady-state current output and the number of E. coli cells. This sensor provided a new method for the rapid enumeration of bacteria. It was also very sensitive with a detection limit of 105 cells m1−1. Thus, the MFC method has a great appeal.
Compared with other methods used for monitoring microorganisms, the current generated in MFC-based sensors by bacteria grown under different conditions must be investigated. Furthermore, not all of the bacteria can be monitored effectively. For example, non-electrogenic bacteria do not utilize electron mediators. Thus, they are not directly involved in the current generation.
Monitoring of corrosive biofilms
Biocorrosion, also known as microbiologically-influenced corrosion (MIC), is caused by corrosive biofilms. It is a major problem in the oil and gas industry as well as some other industries such as water utilities. Flemming (1994) estimated that 20 % of all corrosion losses could be attributed to biocorrosion, which amounts to more than $50B each year in the US alone. There are two primarily types of biocorrosion mechanisms (Xu et al. 2013). Fermentative microbes secrete corrosive metabolites such as organic acids. This type of biocorrosion is relatively easy to detect because it typically involves a low pH. The other type is more difficult to detect. Oil pipelines provide an anaerobic environment for anaerobic microbes such as sulfate-reducing bacteria (SRB). SRB can switch to elemental iron (Fe0) as an electron donor to replace the carbon source during starvation. This respiration process causes corrosion that is explained by the following reactions (Xu and Gu 2014):
The extracellular electrons in Eq. (4) must be transported across cell wall to the SRB cytoplasm where sulfate reduction occurs under enzyme catalysis. Only electrogenic biofilms can transfer electrons across a cell wall.
The detection of corrosive biofilms is crucial in the decision-making of whether to use biocides and/or mechanical pigs for their mitigation. Existing biofilm sensors rely on applying an external electrical field to detect electrical resistance changes across a biofilm (George et al. 2006; Bruijs et al. 2001). However, this interferes with the biofilm metabolism (Janknecht and Melo 2003). Also as these sensors cannot distinguish a mineral film from a biofilm, a passive sensor without an external voltage is desired to avoid false results (Janknecht and Melo 2003).
Gu (2012) proposed the measurement of electrogenicity as an indicator of the presence of a corrosive biofilm and its ability to attack a metal. Figure 3 shows a passive MFC-based biofilm sensor for electrogenic biofilms. A solid-state anode provides electrons to the feed the cathodic biofilm. If a corrosive biofilm, such as an SRB biofilm, attaches to the cathode the electrogenic biofilm will transfer the biofilm to the cytoplasm of sessile cells to reduce sulfate (Xu and Gu 2014). The open circuit potential can be used to tell whether sulfate reduction, nitrate reduction, etc. is occurring at the cathode after calibration. The closed-circuit current flow measures the ability of the cathodic biofilm to transport extracellular electrons (i.e., electrogenicity), which is a bottleneck for an electrogenic biofilm to biocorrosion (Zhang et al. 2015).
Schematic of a membrane-less MFC-based corrosive biofilm sensor with a biocathode in a liquid solution containing oxidant(s) such as sulfate and nitrate (after Gu 2012)
An inert coupon (e.g., graphite coupon) may be placed in a pipeline or storage tank to allow a biofilm to grow on its surface. The coupon can be retrieved and inserted as the biocathode into a test cell on a base station for voltage and current measurements.
Because of its simplicity, this kind of disposable biofilm detection kit will likely be ready for field uses in the near future. For online detection, much more development work will be needed. Field systems have fluids that cannot be replaced in situ with a simple sulfate or nitrate solution to avoid interference. Moving parts in a sensor or probe are undesirable for pressurized pipelines. It may be possible to prevent fouling of the anode by wrapping it with a membrane. It is more likely that such an online biofilm sensor will be deployed in a seawater injection pipeline, before a more complicated oil transport pipeline.
Other sensor applications
There are several other potential applications based on published research. Liu et al. (2011) designed a low-cost MFC system to monitor an anaerobic digester. The system consisted of an up-flow anaerobic fixed-bed (UAFB), a gas–liquid separator and a wall-jet MFC. External recirculation coupled the liquid–gas separator and the two MFC sensors. One of the two MFCs had an anode covered with an electrogenic biofilm, and the other had no pre-immobilized biofilm. Therefore, by analyzing the potential data, the conditions in the digester could be monitored. These kinds of MFCs are low cost and can be coupled with different types of reactors.
Volatile fatty acid (VFA) concentrations are a key parameter for some bioprocesses such as anaerobic digestion. MFC voltage and current output can be correlated with VFA concentrations in the effluent (Kaur et al. 2013). It is even possible to distinguish among different species of VFA using cyclic voltammetry and columbic efficiency. MFCs may be used to distinguish non-fermentable substrates (e.g., acetate and butyrate) from fermentable substrates (e.g., glucose and starch) by analyzing their peak areas (Feng et al. 2013).
Conclusion and future prospects
MFC technology still faces major hurdles in real-world applications for power generation using organic matter in wastewaters except powering small sensor devices. Some researchers have developed applications in other areas. One particularly promising MFC application is the use of MFCs as biosensors. MFC-based biosensors show remarkable promise in the monitoring of BOD, the presence of toxic substances, DO, presence and corrosivity of corrosive biofilms, VFA and anaerobic digester performances. These sensors tend to be low cost and self-powered with real-time online monitoring capabilities. Some of them may see actual deployment in the next few years.
References
Abrevaya XC, Sacco NJ, Bonetto MC, Hilding-Ohlsson A, Corton E (2015a) Analytical applications of microbial fuel cells. Part II: toxicity, microbial activity and quantification, single analyte detection and other uses. Biosens Bioelectron 63:591–601
Abrevaya XC, Sacco NJ, Bonetto MC, Hilding-Ohlsson A, Corton E (2015b) Analytical applications of microbial fuel cells. Part I: biochemical oxygen demand. Biosens Bioelectron 63:580–590
Bruijs MCM, Venhuis LP, Jenner HA, Licina GJ, Daniels D (2001) Biocide optimization using an on-line biofilm monitor. Power Plant Chem 3:400–405
Chang IS, Jang JK, Gil GC, Kim M, Kim HJ, Cho BW, Kim BH (2004) Continuous determination of biochemical oxygen demand using microbial fuel cell type biosensor. Biosens Bioelectron 19:607–613
Chen S, Liu G, Zhang R, Qin B, Luo Y (2012) Development of the microbial electrolysis desalination and chemical-production cell for desalination as well as acid and alkali productions. Environ Sci Technol 46:2467–2472
Choi SH, Gu MB (2003) Toxicity biomonitoring of degradation byproducts using freeze-dried recombinant bioluminescent bacteria. Anal Chim Acta 481:229–238
Davila D, Esquivel JP, Sabate N, Mas J (2011) Silicon-based microfabricated microbial fuel cell toxicity sensor. Biosens Bioelectron 26:2426–2430
Debabov VG (2008) Electricity from microorganisms. Microbiology 77:123–131
Dewan A, Ay SU, Karim MN, Beyenal H (2014) Alternative power sources for remote sensors: a review. J Power Sources 245:129–143
Feng Y, Barr W, Harper WF Jr (2013) Neural network processing of microbial fuel cell signals for the identification of chemicals present in water. J Environ Manag 120:84–92
Flemming HC (1994) Microbial deterioration of materials - fundamentals economical and technical overview. Mater Corros 45:5–9
Flemming HC, Schaule G (1994) Microbial deterioration of materials - biofilm and biofouling: biofouling. Mater Corros 45:29–39
George RP, Muraleedharan P, Dayal RK, Khatak HS (2006) Techniques for biofilm monitoring. Corros Rev 24:123–150
Gil G-C, Chang I-S, Kim BH, Kim M, Jang J-K, Park HS, Kim HJ (2003) Operational parameters affecting the performance of a mediator-less microbial fuel cell. Biosens Bioelectron 18:327–334
Gu T (2012) Methods and devices for the detection of biofilms. World Intellectual Property Organization: Patent WO2012/149487
Jacobson GA (2007) Corrosion at Prudhoe Bay—a lesson on the line. Mater Perform 46:26–34
Janknecht P, Melo LF (2003) Online biofilm monitoring. Rev Environ Sci Biotechnol 2:269–283
Jiang D, Li B, Jia W, Lei Y (2010) Effect of inoculum types on bacterial adhesion and power production in microbial fuel cells. Appl Biochem Biotechnol 160:182–196
Jiang Y, Liang P, Zhang CY, Bian YH, Yang XF, Huang X, Girguis PR (2015) Enhancing the response of microbial fuel cell based toxicity sensors to Cu (II) with the applying of flow-through electrodes and controlled anode potentials. Biores Technol 190:367–372
Jouanneau S, Recoules L, Durand MJ, Boukabache A, Picot V, Primault Y, Lakel A, Sengelin M, Barillon B, Thouand G (2014) Methods for assessing biochemical oxygen demand (BOD): a review. Water Res 49:62–82
Kang KH, Jang JK, Pham TH, Moon HS, Chang IS, Kim BH (2003) A microbial fuel cell with improved cathode reaction as a low biochemical oxygen demand sensor. Biotechnol Lett 25:1357–1361
Kaur A, Kim JR, Michie I, Dinsdale RM, Guwy AJ, Premier GC (2013) Microbial fuel cell type biosensor for specific volatile fatty acids using acclimated bacterial communities. Biosens Bioelectron 47:50–55
Kim T, Han JI (2013) Fast detection and quantification of Escherichia coli using the base principle of the microbial fuel cell. J Environ Manag 130:267–275
Kim M, Park HS, Jin GJ, Cho WH, Lee DK, Hyun MS, Choi CH, Kim HJ (2006) A novel combined biomonitoring system for BOD measurement and toxicity detection using microbial fuel cells. IEEE Sens 1247–1248
Kim M, Hyun SM, Gadd GM, Kim HJ (2007) A novel biomonitoring system using microbial fuel cells. J Environ Monitor 9:1323–1328
Lei Y, Chen W, Mulchandani A (2006) Microbial biosensors. Anal Chim Acta 568:200–210
Li WW, Yu HQ, He Z (2014) Towards sustainable wastewater treatment by using microbial fuel cells-centered technologies. Energy Environ Sci 7:911–924
Liu J, Mattiasson B (2002) Microbial BOD sensors for wastewater analysis. Water Res 36:3786–3802
Liu Z, Liu J, Zhang S, Xing XH, Su Z (2011) Microbial fuel cell based biosensor for in situ monitoring of anaerobic digestion process. Bioresour Technol 102:10221–10229
Liu Q, Zhou Y, Chen SL, Wang ZJ, Hou HQ, Zhao F (2015) Cellulose-derived nitrogen and phosphorus dual-doped carbon as high performance oxygen reduction catalyst in microbial fuel cell. J Power Sources 273:1189–1193
Logan BE (2009) Exoelectrogenic bacteria that power microbial fuel cells. Nat Rev Microbiol 7:375–381
Lorenzoa MD, Curtis TP, Head IM, Scott K (2009) A single-chamber microbial fuel cell as a biosensor for wastewaters. Water Res 43:3145–3154
Min B, Angelidaki I (2008) Innovative microbial fuel cell for electricity production from anaerobic reactors. J Power Sources 180:641–647
Modin O, Wilén BM (2012) A novel bioelectrochemical BOD sensor operating with voltage input. Water Res 46:6113–6120
Moon HS, Chang IS, Kang KH, Jang JK, Kim BH (2004) Improving the dynamic response of a mediator-less microbial fuel cell as -biochemical oxygen demand (BOD) sensor. Biotechnol Lett 26:1717–1721
Moon HS, Chang IS, Jang JK, Kim KS, Lee J, Lovitt RW, Kim BH (2005) On-line monitoring of low biochemical oxygen demand through continuous operation of a mediator-less microbial fuel cell. J Microbiol Biotechn 15:192–196
Oh S, Min B, Logan BE (2004) Cathode performance as a factor in electricity generation in microbial fuel cells. Environ Sci Technol 38:4900–4904
Patchett RA, Kelly AF, Kroll RG (1988) Use of a microbial fuel cell for the rapid enumeration of bacteria. Appl Microbiol Biotechnol 28:26–31
Peixoto L, Min B, Martins G, Brito AG, Kroff P, Parpot P, Angelidaki I, Nogueira R (2011) In situ microbial fuel cell-based biosensor for organic carbon. Bioelectrochemistry 81:99–103
Quek SB, Cheng L, Cord-Ruwisch R (2015) In-line deoxygenation for organic carbon detections in seawater using a marine microbial fuel cell-biosensor. Bioresour Technol 182:34–40
Rabaey K, Lissens G, Siciliano SD, Verstraete W (2003) A microbial fuel cell capable of converting glucose to electricity at high rate and efficiency. Biotechnol Lett 25:1531–1535
Ren H, Pyo SJ, Lee JI, Park YJ, Gittleson FS, Leung FCC, Kim JB, Taylor AD, Lee HS, Chae JS (2015) A high power density miniaturized microbial fuel cell having carbon nanotube anodes. J Power Sources 273:823–830
Rice EW, Baird RB, Eaton AD, Clesceri LS (2012) Standard methods for the examination of water and wastewater, 22nd edn. American Public Health Association/American Water Works Association/Water Environment Federation, Washington DC
Sara RM, Maria JLDA, Damià B (2006) Biosensors as useful tools for environmental analysis and monitoring. Anal Bioanal Chem 386:1025–1041
Stein NE, Hamelers HV, Buisman CN (2010) Stabilizing the baseline current of a microbial fuel cell-based biosensor through overpotential control under non-toxic conditions. Bioelectrochemistry 78:87–91
Stein NE, Hamelers HMV, van Straten G, Keesman KJ (2012a) On-line detection of toxic components using a microbial fuel cell-based biosensor. J Process Contr 22:1755–1761
Stein NE, Hamelers HV, Straten GV, Keesman KJ (2012b) Effect of toxic components on microbial fuel cell-polarization curves and estimation of the type of toxic inhibition. Biosensors 2:255–268
Sun JZ, Kingori GP, Si RW, Zhai DD, Liao ZH, Sun DZ, Zheng T, Yong YC (2015) Microbial fuel cell-based biosensors for environmental monitoring: a review. Water Sci Technol 71(6):801–809
Tront JM, Fortner JD, Plotze M, Hughes JB, Puzrin AM (2008) Microbial fuel cell biosensor for in situ assessment of microbial activity. Biosens Bioelectron 24:586–590
Uríaa N, Sánchez D, Masb R, Sánchez O, Munozb FX, Mas J (2012) Effect of the cathode/anode ratio and the choice of cathode catalyst on the performance of microbial fuel cell transducers for the determination of microbial activity. Sensor Actuat B-Chem 170:88–94
Vishwanathan AS, Rao G, Siva Sankara Sai S (2013) A novel minimally invasive method for monitoring oxygen in microbial fuel cells. Biotechnol Lett 35:553–558
Wang X, Gao N, Zhou Q (2013) Concentration responses of toxicity sensor with Shewanella oneidensis MR-1 growing in bioelectrochemical systems. Biosens Bioelectron 43:264–267
Xu D, Gu T (2014) Carbon source starvation triggered more aggressive corrosion against carbon steel by the Desulfovibrio vulgaris biofilm. Int Biodeterior Biodegrad 91:74–81
Xu D, Li Y, Song F, Gu T (2013) Laboratory investigation of microbiologically influenced corrosion of C1018 carbon steel by nitrate reducing bacterium bacillus licheniformis. Corros Sci 77:385–390
Zhang Y, Angelidaki I (2011) Submersible microbial fuel cell sensor for monitoring microbial activity and BOD in groundwater: focusing on impact of anodic biofilm on sensor applicability. Biotechnol Bioeng 108:2339–2347
Zhang Y, Angelidaki I (2012) A simple and rapid method for monitoring dissolved oxygen in water with a submersible microbial fuel cell (SBMFC). Biosens Bioelectron 38:189–194
Zhang CY, Liang P, Jiang Y, Huang X (2015) Enhanced power generation of microbial fuel cell using manganese dioxide-coated anode in flow-through mode. J Power Sour 273:580–583
Zhou M, Wang H, Hassett DJ, Gu T (2013) Recent advances in microbial fuel cells (MFCs) and microbial electrolysis cells (MECs) for wastewater treatment, bioenergy and bioproducts. J Chem Technol Biotechnol 88:508–518
Acknowledgments
This work was financially supported by Natural Science Foundation of China (nos. 21328602, 51178225 and 21273120), GEFC09-12, NCET-08-0296 and the Fundamental Research Funds for the Central Universities.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Yang, H., Zhou, M., Liu, M. et al. Microbial fuel cells for biosensor applications. Biotechnol Lett 37, 2357–2364 (2015). https://doi.org/10.1007/s10529-015-1929-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-015-1929-7