Abstract
This study aimed to examine the dynamics of soil functional indicators under the influence of deadwood in old mixed beech forests in northern Iran. In January and February 2006, several strong wind events occurred in the forest of Khanikan, which led to severe windthrow. All the fallen trees remained in the protected parcel and were not removed. Immediately after tree felling in the studied areas, five common tree species [Fagus orientalis (Fagus), Acer velutinum (Acer), Carpinus betulus (Carpinus), Alnus subcordata (Alnus), and Tilia begonifolia (Tilia)] with the highest abundance of deadwood (with 14 individual trees) were marked as permanent plots for monitoring their effect on soil functions. Wood samples were collected from the trunks and soil samples (0–10 cm depth) from the lower part of deadwood in August 2011, 2016, and 2021. In addition, soil samples were taken from areas that did not have deadwood as control areas. For the study of soil biological parameters, soil samples were collected in August and November (2011, 2016, and 2021). However, for monitoring soil N transformation, soil samples were taken in August, September, November, and December (2011, 2016, and 2021). We found that Alnus and Carpinus deadwood had higher N, K, P, and Ca contents than deadwood of other species. Higher values of C and Mg were observed in Fagus deadwood in the final stages of sampling (2021). Anlus deadwood enhanced the stability of soil aggregates, soil nutrient contents, as well as soil enzyme and microbial activities, and soil fauna and microflora populations during the decay process. Based on our findings, the intensity of soil functions increased in the order control < Fagus < Tilia < Acer < Carpinus < Alnus, which corresponds to the increasing quality of the deadwood of the tree species. Our data emphasize that retaining deadwood is highly effective in boosting carbon and nutrient cycling in forest ecosystems, which may have implications for forest management strategies to improve soil ecological functions and forest sustainability. Although our observations were obtained in an old mixed beech forest, the importance of deadwood on soil functional characteristics can also be assumed in a broader range of forest environments, but this requires further investigation to determine the mechanisms that explain these phenomena.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The Caspian forest (Hyrcanian region) is a natural green belt that covers the northern slopes of the Alborz Mountains and extends to the southern slopes of the Caspian Sea. The Hyrcanian region is approximately 110 km wide, 800 km long and has a total area of 2.1 million ha, equivalent to 15% of Iran’s total forest area and 1.1% of Iran’s entire area. Hyrcanian region or Caspian forests is one of the last remnants of natural temperate deciduous forests in the world (Sagheb Talebi et al. 2014), which are very valuable in terms of ecological, social, economic, and cultural terms and recently were included in the UNESCO World Heritage List (Hosseini 2019). Forest areas located at high altitudes are often exposed to strong wind events. In this regard, some tree species are more affected by the wind and uprooted due to their special crown and root structures. Deadwoods that appear due to the uprooting of trees in the forest will have many ecological effects on the habitat and affect the soil functions over time. Deadwood plays a vital role in protecting biodiversity, forest ecosystem dynamics, soil fertility, and C stocks (Shannon et al. 2022; Uhl et al. 2022; Sacher et al. 2022). The type and amount of deadwood are the results of several factors occurring in forest ecosystems, including climatic factors such as humidity and temperature conditions (Herrmann and Bauhus 2013), tree species (Kahl et al. 2017; Minnich et al. 2021), decay age (Bond-Lamberty et al. 2002; Jomura et al. 2022), chemical properties such as nitrogen (N) and lignin content (Berg and McClaugherty 2008; Błońska et al. 2017), fungal community structure (Fukami et al. 2010; Odriozola et al. 2021), enzyme activities, and decomposer biomass (Bradford et al. 2014; Jiang et al. 2022). Since felled deadwood is in direct contact with the forest floor, the soil microbial community penetrates the wood and accelerates the wood degradation process (Tláskal et al. 2021). The main processes involved in deadwood decomposition include fragmentation, respiration, transformation, and leaching (Magnússon et al. 2016).
Over time, as dead wood decomposes, residues enter the soil and alter the fertility (Błońska et al. 2019; Jiang et al. 2022) and several characteristics of the soil (Błonska et al. 2017; Piaszczyk et al. 2019; Shannon et al. 2021; Hollands et al. 2022). Carbon includes almost 50% of the woody dry mass (Weiss et al. 2000). During the decomposition process, C is partially released into the atmosphere and at certain shares stored in the soil, thereupon increasing C resources in the forest soil (Błońska et al. 2019). Biologically, deadwood is an important component of resilience and belongs to the above-ground litter fraction, and its accumulation leads to significant interactions with below-ground soil components. Deadwoods are major contributors to soil organic matter (SOM), providing input to long-term nutrient cycling, ensuring nutrients for beneficial soil organisms, and forming ectomycorrhizal root tips. Despite the global significance of forest sustainability, the N and C cycle in forest ecosystems, and the effects of dead wood in nutrient cycling and C dynamics (Persoh 2015), very little information is available concerning nutrient dynamics, soil functional indicators and providing of microclimates in temperate forests (Woodall et al. 2020; Shannon et al. 2022). Deadwood is a central structural element influencing various essential ecological and biogeochemical functions by generating heterogeneous microsites in old mixed forests (Perreault et al. 2021). Natural old mixed forests are trees of different ages with a high amount of CWD in different sizes and species in all stages of decay in the forest ground (Lassauce et al. 2011). Deadwood is not only an essential reservoir of terrestrial biomass, soil carbon (C), and nutrients but also provides a habitat and food source for a variety of forest species, including microbes and fungi (Norden et al. 2004; Mayer et al. 2022), fauna communities (Zuo et al. 2021), vertebrate species (Spiering and Knight 2005), bryophytes and lichens (Paltto et al. 2008), and also vascular plants (Scheller and Mladenoff 2002).
Assessing the sustainability of forest management requires indicators, and in this regard, forest soils are of great importance due to their ability to supply the nutrients necessary to sustain forest productivity and sustainability (Dale et al. 2008; Jamil et al. 2016). Soils provide a wide range of vital ecosystem services that maintain global sustainability and ecological balance (Drobnik et al. 2018). Sustainable forest management includes soil quality as the criterion for sustainability. In the past decades, several attempts were made to introduce terms such as soil functions, soil quality, soil health, and soil resilience to support politicians and managers related to soil conservation and rehabilitation (Álvarez et al. 2003; Wienhold et al. 2004). However, in recent years the concept of soil functions has become synonymous with soil quality. (Kooch et al. 2022). Soil quality can be determined by appropriate indicators, which are the results of the physical, chemical and biological processes of the soil. Soil science has embraced the concept of soil functions, which brings various capabilities of soils to the fore and has emphasized the multi-functionality of soils and their very different properties (Greiner et al. 2017). Soil functions cannot be measured independently, but soil functions can be evaluated by measuring the physical, chemical, and biological soil characteristics (Doran and Parkin 1994; Larson and Pierce 1994). Therefore, new studies are needed to explore ecological processes that can explain changes in various soil functions when various indicators are taken into account (Teixeira et al. 2020). In forest ecosystems, various factors can affect soil functions and quality; deadwood, or coarse woody debris (CWD), which contributes to forest ecosystem functioning and carbon, nutrient and gas fluxes, improving microclimate, increasing nutrient availability, and habitat quality (Magnússon et al. 2016; Błonska et al. 2017).
Although deadwood is increasingly being included in ecological studies and environmental assessments, even in the Hyrcanian forest (Kooch et al. 2012; Moghimian et al. 2020), here we tried to monitor the long-term effects (15 years) of deadwood, in a mixed beech forest in northern Iran, on a wide range of functional characteristics of the soil (various parameters of soil physical, chemical, and biological) in order to achieve its effects on sustainable forest management. We hypothesized that the tree species with a lower C/N ratio and higher nutrient content would be degraded faster and, therefore, would contribute to soil fertility more instantly than other, more recalcitrant wood species. We also expected that during the monitoring time and further decomposition of deadwood, the biological activities of the soil are changed and differ depending on the deadwood of different tree species.
Materials and methods
Study area
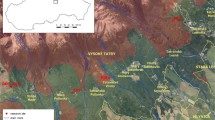
Khanikan forest, covering an area of 2807 hectares, is located near the city of Chalus in Mazandaran Province, northern Iran (Fig. 1a). The study area has an elevation range of 100–1600 m above sea level with a slope range of 0–30%. The average annual temperature is 16.05 °C, the average annual rainfall is 1313 mm, and the dry season runs from May to August (Fig. 1b). From late December to early April, the land is usually covered with snow in higher altitudes. According to the USDA Soil Classification, the parent material is dolomitic limestone from the late Jurassic to the early Cretaceous. The soil is in Alfisol composition with a loamy texture from silty clay. The area consists of a mixed beech forest dominated by oak (Quercus castaneifolia C. A. Mey.), besides oriental beech (Fagus orientalis Lipsky), zelkova (Zelkova carpinifolia Dippe), maple tree (Acer velutinum Boiss.), wingnut (Pterocarya fraxinifolia Lam.), lime tree (Tilia begonifolia Stev.), Caucasian hornbeam (Carpinus betulus L.), ash (Fraxinus excelsior L.), Caucasian alder (Alnus subcordata C.A. Mey.), and Cappadocian maple (Acer cappadocicum Gled). The average height and diameter of the trees were 20–25 m and 50–60 cm, respectively. Tree density was 250–300 Nha−1 in the Khanikan forest. Other less common species (< 10%) are the wild service tree (Sorbus torminalis Crantz), elm (Ulmus glabra Huds.), and the wild cherry (Prunus avium L.). Herbaceous plants such as Hypericum androsaemum L., Euphorbia amygdaloides L., Asperula odorata L., and Polystichum sp. covered more than 85% of the forest floor.
Sampling and laboratory analysis
In January and February 2006, several strong wind events occurred in the forest of Khanikan, which led to the windthrow of some trees (Kooch 2008). All of the fallen trees remained in the protected parcel (an area of about 60 ha−1 of the forest where there is no human intervention) and were not exited of the forest. This study was carried out in the protected parcel (control), where harvesting and any human activity that leads to the cutting, logging, and removal of trees from the protected parcel (control) is prohibited (in all the years). The windthrow event caused the falling of individual trees across the protected parcel. Immediately after tree felling in that field, five common tree species of the Hyrcanian forest [Fagus orientalis (Fagus; with 17.6% of the volume and 30% of the total number), Acer velutinum (Acer; with 5.8% of the volume and 2.7% of total number), Carpinus betulus (Carpinus; with 30.5% of the volume and 30% of the total number), Alnus subcordata (Alnus; with 9.1% of volume and 5% of the total number), and Tilia begonifolia (Tilia; with 2.7% of the volume and 1% of the total number)] (Sagheb Talebi et al. 2014) with the highest abundance of deadwood (with 14 individual trees) were marked as permanent plots to be monitored in order to study their effect on soil functions. All selected deadwoods (with a range of 40–50 cm diameter at breast height and height 20–25 m) are in close proximity to each other with a distance of less than 1000–1500 m between them and were considered if physiographic characteristics (i.e., elevation between 1500 and 1600 m above sea level, the slope between 10 and 15% and north aspect) of bedrock and climate had similar conditions (Fig. 1c). Wood samples were collected by using a core driller (12-mm drill bit) taking samples from the trunks at the lower part of deadwood (0–10 cm depth) in August of 2011, 2016, and 2021 (Fig. 1d). Three soil samples (0–10 cm depth) were taken from the right and the left side along the trunks using iron frames (30 × 30 cm area) and mixed. In addition, 14 soil (0–10 cm depth) samples were taken from areas that did not have deadwood (at a distance of about 50 m from the deadwood) as control areas using iron frames (30 × 30 cm area). For the study of soil biological parameters, samples were collected in August and November (2011, 2016, and 2021). However, soil samples for monitoring N transformation were taken in August, September, November, and December (2011, 2016, and 2021).
Wood and soil samples were immediately transferred to cooled and insulated containers, transported to the laboratory, and stored at 4 °C until further analysis. Several properties of deadwood and soil were measured at the laboratory based on standard protocols (see Table 1). In detail, deadwood properties, including carbon (DWC), phosphorous (DWP), potassium (DWK), calcium (DWCa), and magnesium (DWMg) were determined in quadruplicate using dry combustion with an elemental analyzer (Fisons EA1108, Milan, Italy) calibrated by the BBOT [2, 5-bis-(5-tert-butyl-benzoxazol-2-yl)-thiophen] standard (ThermoQuest Italia s.p.a.) (Kooch et al. 2017). The pycnometer method (Blake and Hartge 1986) and the cold method (Plaster 1985) were applied to measure the particle and bulk density, respectively. We used the formula [1—(bulk density/particle density)] for soil porosity calculated that proposed by Pires et al. (2014). Yoder’s method was used to determine soil stability (Kemper and Rosenau 1986). The Bouyoucos hydrometer method proposed by (Bouyoucos 1962) was used for measuring soil texture. We used a wet sieve with a sieve size of 0.25–0.50 mm to determine the particle size distribution of the aggregates. The size of micro- and macrograins was calculated between 0.053–0.25 mm and 0.25–0.50 mm, respectively (Cambardella and Elliott 1992). The Walkley–Black method was used for measuring soil organic carbon (Allison 1975), and then the formula (OM % = Organic C % × 1,724) was applied for calculated organic matter content (OM) (Ezeigbo et al. 2013). The stratification of soil organic matter was calculated based on the OM (0-10 cm)/OM (10–20 cm) depths (Bongiorno et al. 2019). The semi-micro-Kjeldahl method was used to measure total N (Bremner and Mulvaney 1982). Soil C and N stocks were calculated using the bulk density data and C and N concentrations at the studied depth (e.g., 0–10 cm) (Sariyildiz et al. 2015). The carbon pool index (CPI) was calculated from the change in total carbon, and the lability index (LI) was determined based on changes in the proportion of labile C in the soil. These two indices were applied to calculate the carbon management index (CMI), with CMI = C pool index (CPI) × lability index (LI) × 100 (Blair, et al. 1995). The soil/water solution was used to measure soil pH and EC of Orion Ionalyzer Model 901 pH and EC meter in a 1:2.5. Available phosphorous (P) was measured spectrophotometrically using Olsen’s method (Homer and Pratt 1961), and available potassium (K), calcium (Ca), and magnesium (Mg) were determined using atomic absorption spectrophotometry (Bower et al. 1952). Determination of particulate organic carbon (POC) and particulate organic nitrogen (PON) was done by physical fractionation. Determination of dissolved organic carbon (DOC) and dissolved organic nitrogen (DON) was performed using a total organic carbon (TOC) analyzer (Shimadzu TOC-550A) (Cambardella and Elliot, 1992). The buried-bag technique was used to estimate soil N mineralization, nitrification, and ammonification rates over 30 days. NH4 + and NO3- extraction was done with 2 M KCl solution (soil/solution, 1:5) and filtered using a 0.45 µm filter. The NH4 + and NO3- concentrations in the extraction solution were determined colorimetrically at 645 and 420 nm, respectively (Li et al. 2014).
Earthworms were collected by manual identification at the same time as soil samples, which were divided into ecological groups (i.e., epigeic, anecic, and endogeic) based on external characteristics and weighed (Jeffery et al. 2010). Soil Acarina and collembola were extracted using a modified Tullgren funnel (Hutson and Veitch 1987). Soil nematodes were extracted from a 100 g soil sample (fresh weight) using a modified filter method as described by Hutson and Veitch (1987). Soil protozoa population density was calculated microscopically according to the extraction method (Mayzlish and Steinberger 2004). Potato dextrose agar (PDA) and nutrient agar (NA) were used for bacterial and fungal culture, respectively (Wollum, 1982; Asadu et al. 2015). The evolved CO2 was determined during a 3 day incubation at 25 °C to measure soil basic respiration (BR) (Alef 1995). Glucose 1% was used as a substrate to measure substrate-induced respiration (SIR) and, after 72 h of incubation, the released CO2 was measured. CO2 evolved was absorbed in NaOH and then measured by titration with HCl (Anderson and Domsch 1990). The chloroform fumigation method was used for extraction of soil microbial biomass C (MBC) and microbial biomass N (MBN) (Brookes et al. 1985). Activities of urease (EC 3.5.1.5), acid phosphatase (EC 3.1.3.2), arylsulfatase (EC 3.1.6.1), and invertase (EC 3.2.1.26) were determined according to the method described by Schinner and von Mersi (1990).
Statistical analysis
The normality of the variables (p-value > 0.05) was checked by the Kolmogorov–Smirnov test, and the homogeneity of the variances was assessed using Levene’s test. Two-way repeated measured analysis of variance (ANOVA) was employed for deadwood and soil physical and chemical properties concerning different tree species and years. Three-way repeated measured analysis of variance (ANOVA) was used for N transformation and soil biological properties in relation to deadwood trees, years, and seasons. A post hoc test (using Duncan’s test) was used to make multiple comparisons of features with significant differences (p-value < 0.05). All analyzes were performed in SPSS ver. 22.0 statistical software. Principal component analysis (PCA) was applied to analyze the relationships between deadwood and soil properties using PC-ORD ver. 5.0. Correlations between soils and PCA components (i.e., PC1 and PC2) were analyzed using the Pearson correlation coefficient. The heat plots for soil function indicators were identified as part of the PCA output.
Results
Deadwood properties
The results of repeated measured analysis of variance (ANOVA) indicated that there were significant differences (p-value < 0.05) in the deadwood properties among different deadwood tree species (Table S1). Among the deadwood properties studied, only N and Ca had significant differences (p-value < 0.01) comparing different years of sampling (2011, 2016, and 2021). During our study period, the highest amount of OC was in Fagus deadwood, whereas the lowest amount was detected in Alnus (by 24–32%; Alnus < Carpinus ≈ Acer ≈ Tilia < Fagus) (Fig. S1a). However, Alnus had 1.5–2 times higher deadwood N at three sampling years than in the other deadwood trees (Alnus > Carpinus > Acer ≈ Tilia ≈ Fagus) (Fig. S1b). The C/N ratio of Fagus deadwood increased by 30 units in Fagus ≈ Tilia > Acer > Carpinus ≈ Alnus (Fig. S1c). The total P content in deadwood of Alnus was 45% higher than under Fagus and Tilia (Alnus > Carpinus ≈ Acer > Tilia ≈ Fagus) (Fig. S1d). Total amounts of deadwood K and deadwood Ca in Alnus were nearly half of those found in the other deadwood tree species (Alnus > Carpinus ≈ Acer ≈ Tilia > Fagus) (Fig. S1e and f). Deadwood of different tree species did not significantly differ (p-value < 0.01) concerning the amount of total Mg (Fig. S1g).
Soil properties
ANOVA results indicated that there was a significant difference (p-value < 0.05) in the soil’s physical and chemical properties in the vicinity of the deadwood of several tree species (Table S2). The bulk density was significantly higher in the control plot than for soil around deadwood of the other studied tree species with the lowest values for Alnus and Carpinus (18 and 25%) (Control > Fagus ≈ Tilia ≈ Acer < Carpinus < Alnus), respectively. The soil around deadwood from five tree species had no significant differences in soil particle density (Table S2). Different years (2011, 2016, and 2021) did not affect bulk density and particles among tree species. However, there were significant differences in the porosity of soil around deadwood, with the highest values under Alnus and Carpinus and lowest values for the control (Alnus ≈ Acer ≈ Carpinus ≈ Tilia > Fagus > control), respectively (Table S2). The soil under the studied tree species had the same texture, but sand decreased in the vicinity of deadwood under Alnus and Carpinus species (by 40–50%) compared to the control (control > Fagus > Tilia > Acer > Carpinus > Alnus), whereas the clay content increased (by 40–50%) under deadwood from Alnus and Carpinus compared to the control (Alnus > Carpinus > Acer > Tilia > Fagus > control). Soils under deadwood from the trees under study had no significant differences (p-value > 0.05) in silt content (Table S2). The amount of soil clay increased significantly in 2021 compared to other years, but this amount of silt decreased in 2021 in soils under all deadwood trees. Soil pH and EC under Alnus (pH = 7.04; EC = 0.32) were higher compared to other deadwood in ranked order of (Alnus > Acer ≈ Carpinus ≈ Tilia ≈ Fagus ≈ control) (Table S2).
Soil organic C and C stock did not vary significantly in deadwood trees, except in 2021, when Acer, Carpinus, and Fagus had significantly higher values than other species and lower values for the control (Acer ≈ Carpinus ≈ Fagus > Alnus > Tilia > control). The amount of soil C in 2021 was significantly higher than in other years (p-value < 0.05). The total soil N varied among deadwood tree species under Alnus ≈ Acer ≈ Carpinus, being higher than Fagus and the control (Table S2). The C/N ratio in soils under Alnus deadwood was almost half of those in the control and Fagus, whereas available forms of P, K, Ca, and Mg in Alnus and Carpinus were nearly triple those in the control and Fagus (Alnus > Carpinus > Acer > Tilia > Fagus > control). Annual changes from 2011 to 2021 showed higher effects on Alnus for available forms of P, K, Ca, and Mg, with higher values in 2021 and 15 years after the wind throw (Table S2). Soil DOC, POC, and contents of N in macro- and microaggregates, the N stock as well as N mineralization, the contents of NH4+and NO3−, nitrification, ammonification, PON, and DON indicated a significant decrease of almost 50–80% from soils under Alnus deadwood to the control (Alnus > Acer > Carpinus > Tilia > Fagus ≈ control), respectively (Table S2 and Fig. 2a–e). Based on the data from the three studied sampling periods, lower values for soil C in macro- and microaggregates were found under control compared to other deadwood tree species. The C in macro- and microaggregates in 2016 and 2021 were significantly higher than in 2011 (p-value < 0.05). The stratification ratio (OMstr) was higher in soils around Alnus and Carpinus deadwood (almost 25%) compared to Tilia, Acer, Fagus, and the control (Fig. S2a). The carbon management index (CMI) showed differences in the deadwood of different tree species, with the highest value detected under Tilia (Fig. S2b).
Mean (± SE) of NO3− − N (a) and NH4 + − N (b) concentration, nitrification rate (c), net ammonification rate (d) and N mineralization in the top mineral soil. Different lower case letters indicate significant difference (P < 0.05) among sampling times (i.e., 2011, 2016, and 2021), and different capital letters indicate significant differences (P < 0.05) among deadwood from different tree species and control plots
All studied soil enzymatic activities (i.e., urease, arylsulfatase, invertase, and acid phosphatase) around deadwood were significantly affected by deadwood tree species (Alnus > Carpinus > Acer > Tilia > Fagus > control), i.e., by a decrease of approximately 60% from Alnus to the control. The annual variation from 2011 to 2021 was most effective in soils around deadwood under Alnus (Table S3). Deadwood tree species significantly affected all studied soil biological characteristics (Tables S3 and S4; Table 2). Except for the total earthworms, the anecic and endogenic biomasses and the seasons significantly affected soil biological characteristics (Tables S3 and S4). Alnus and Carpinus deadwood increased soil density and biomass (Alnus > Carpinus > Acer ≈ Tilia ≈ Fagus ≈ control) of ecological earthworm groups (epigeic, anecic, and endogeic). The populations of collembola, Acarina, protozoa, nematodes, as well as soil fungi and bacteria around deadwood were significantly affected by tree species, seasons, and years (Tables S3 and S4); and declined along the order of Alnus > Carpinus > Acer > Tilia > Fagus > control, respectively (Table S4). Soil microbial indicators (i.e., BR, SIR, MBN, and MBC) had the highest values under Alnus and Carpinus deadwood and lowest values in the control and under Fagus (Table S4).
Relationship among deadwood and soil properties
The PCA output clustered properties of soil and deadwood (Fig. 3a–i; Table 3). Significant differences were found in the soil surrounding the deadwood of the five species. Furthermore, the studied tree species, deadwood quality, and soil biochemical characteristics in the years 2011–2021 could be separated by PCA output. In general, 59.76 and 11.34% (in 2011), 57.74 and 13.12% (in 2016), and 57.06 and 12.42% (in 2021) of the variance were explained by axis 1 and axis 2, respectively. The deadwood quality, soil functional, and soil biological properties had the highest values under Alnus and Carpinus. In contrast, the lowest values of soil biochemical characteristics were observed under control and Fagus. In this regard, Alnus and Carpinus had more deadwood nutrients (i.e., N, K, P, and Ca). The soil under these deadwood species had higher aggregate stability and soil porosity, higher EC, pH values and nutrient contents, as well as increased organic matter fractions, soil fauna and microbial populations. The lower values of deadwood C, deadwood C/N ratio, soil bulk density, sand, organic C, and C stock were assigned to Alnus and Carpinus deadwoods. Soils around deadwood having a higher fertility and increased biota abundance can be attributed to Alnus in the disintegration development stage (regarding the heat map plots; see Fig. 4), while the controls had a lower fertility and biota abundance.
Grouping of deadwood species and soil properties based on principal component analyses (PCA) at different sampling times. Abbreviations are explained in Table 1
Ranking of the tree species, deadwood species and soil properties based on axis 1 (a) and 2 (b) of principal component analyses (PCA). Abbreviations are explained in Table 1. The intensity in red color (from weak to strong) indicates the increase of values for deadwood species and soil properties during the years 2011–2021
Discussion
Deadwood properties
During our study period, C concentration in deadwood of Fagus and Tilia was significantly higher than that in Alnus, reflecting the higher C concentration in undecomposed wood of Fagus compared to Alnus (Thomas and Martin 2012; Kahl et al. 2017; Herrmann et al. 2018). With advancing decomposition from 2011 to 2021, a slight increase in C concentration was observed for all wood species, but this difference was not significant. Lignin (with C content of 63–66%) has a higher C concentration than cellulose (with C content about 44%), so this change is probably due to an increase in lignin and a decrease in cellulose (Herrmann et al. 2015). Accordingly, the results of Christensen and Vestural (2003) that followed C concentrations in beech did not observe a significant difference in the C concentration of deadwood with different decomposition stages. Beech (Fagus) stores more C than alder (Alnus) because of different wood densities among these species (Herrmann et al. 2018). Generally, we obtained the most significant increase in nutrient concentrations in dead Alnus trees. In our study, all species showed an increase in N concentration with a progressive decomposition stage. The N content increased after 15 years of decay, probably due to N fixation by bacteria, accumulation of fungal biomass and the effects of rainfall input (Garrett et al. 2008). The increase in N content resulted in a declining C/N ratio with the decay process in 2021; thus, the C/N ratio could be a potential indicator of the decay rate (Yang et al. 2010). With the ongoing decomposition from 2011 to 2021, a slight increase in P and K concentration was observed for all wood species, but this difference was not significant. Generally, we observed the most significant increase in nutrient concentrations in CWD of Alnus and Carpinus compared to Fagus. The variation in wood properties between the tree species (e.g., wood density) may cause these differences in deadwood nutrient concentration during decomposition (Harmon et al. 2013). This has been attributed to lower absolute lignin concentrations and higher nutrient contents (Stutz et al. 2017) in Alnus compared to Fagus. According to Laiho and Prescott (2004), the contents of P, K, Ca, and N in CWD depend mainly on the wood’s initial N, P, K, and Ca concentrations, which are determined by tree species and the nutrient status of the site, besides the characteristics of the wood (e.g., wood structure and type, C/N ratio). We tested the effect of each nutrient on the degradation of CWD and found that most nutrients were positively related to the duration of deadwood decay after the windthrow event. The content of most nutrients increases during the study period, suggesting that nutrient content changes with mass loss in CWD (Krankina et al. 1999; Yang et al. 2010).
Soil properties
Soil physical properties
In the present research, a wide variety of soil properties were assessed to inform how soil functions change and interact during the decay of deadwood from different tree species. Our study demonstrated the effects of deadwood decomposition on several physical properties of forest soils. Differences in the bulk density between soil around deadwood and control plots were found for five species, especially for Alnus and Carpinus deadwood. As organic matter increased due to the ongoing decomposition of deadwood, soil bulk density decreased. According to Yue et al. (2017), organic matter content effectively affects soil bulk density, thus, organic matter concentration predicts soil bulk density (Prévost et al. 2004; Perie and Ouimet 2008). In general, wood quality influences soil properties that provide sources for SOM formation (Wambsganss et al. 2017). We observed a positive relationship between soil structural stability, clay content, porosity, and macro- and microaggregation around dead trees across tree species. Soil properties such as soil type, texture, aggregate stability, and moisture influence the soil’s organic matter content (Matus 2021) and affect the decomposition process. Organic matter derived from dead trees enriches soil and determines soil particles’ structure and cohesion. Plant residues such as decaying wood stimulate microbial activity and contribute to stabilizing soil structure (Piaszczyk et al. 2017; Piaszczyk et al. 2019). Releasing organic matter from deadwood can stimulate the formation of soil aggregates, improve soil porosity, significantly increase the number of micropores, and increase soil water-holding capacity (Villada 2013; Matus 2021; Jiang et al. 2022).
Soil chemical properties
The soil pH was lower in control compared to the soil around deadwood species. Jiang et al. (2022) pointed to the leaching of nutrients from deadwood decomposition into the soil. In addition, Kooch et al. (2021) demonstrated that increasing urease activity contributes to increasing soil pH. In our study, urease enzyme activity increased in the soil around deadwood for all species compared to the control. We observed an increase in several soil biochemical characteristics located directly under decomposing deadwood compared to control soils. Thus, the wood decomposition process determines the activity of enzymes and nutrient contents in the soil under the deadwood. Available forms of P, K, Ca, and Mg in Alnus and Carpinus were nearly triple those in the control areas. Deadwoods are rich in macroelements such as N, P, K, and Ca, which ultimately end up in the soil and significantly improve soil quality (Piaszczyk et al. 2019). In our study, examining the characteristics of wood showed that N, K, P, and Ca concentrations in Alnus were higher compared to other species. Furthermore, our results claimed that wood with the highest stock of microelements (such as Alnus and Carpinus) released more ions to the soil surface than wood with lower concentrations of microelements (Lasota et al. 2018). In general, the concentration of nutrients in the topsoil layer released by dead trees depends on the type of wood and its decay stage (Jomura et al. 2022). Similar results were reported by Błońska et al. (2019) for the dead trees of seven tree species in the Czarnalozga reserve in central Poland.
Total N, contents of NH4+, NO3, nitrification, ammonification, N mineralization, DON, as well as N in macro- and microaggregates and PON concentrations are significantly higher in soils under degrading deadwood. Therefore, we hypothesize that the increased N concentrations are due to fungal N translocation (Hart 1999) and bacterial N fixation (Hicks et al. 2003) contributing to the higher concentration of N in wood and its transfer to the soil during the decomposition process, especially at the late decay process in Alnus. Ammonification rates are affected by C/N ratio, soil pH, and soil nutrient contents (Lee et al. 2009). Residues with a high C/N ratio cause strong competition for available soil N (Vallejo et al. 2011). Compared to other species, Alnus and Carpinus had higher values of NH4+-N as a result of stronger abiotic fixation or microbial competition (Giardina et al. 2001). Decreased nitrification rates in Fagus species were associated with higher C/N ratios in soil and deadwood (Burton et al. 2007). Also, soil pH is crucial in the nitrification reaction, as the nitrification rate decreases rapidly below pH 7.0 (Ahn 2006; Lee et al. 2009). It is generally assumed that acidic forest soils have negative nitrification levels because autotrophic nitrification is inhibited (Li et al. 2001). Soil N mineralization rate is impacted by forest conversion and site management (Yan et al. 2007; Scott et al. 2014). Correspondingly, Alnus had a lower C/N ratio than Fagus in the dataset of this study. Forest areas containing N-fixing species can increase soil N mineralization (Rothe et al. 2002). Differences in soil N mineralization may be due to differences in the availability of labile N substrates (Mc Kinley et al. 2008). High-quality deadwood reduces microbial immobilization of N, leading to N mineralization and improved plant N availability (Mc Kinley et al. 2008). The contents of soil organic C, DOC, C in macro- and microaggregates, and POC are significantly higher in the soil around deadwood than those in the control, corresponding to the conclusion by Błonska et al. (2019) and Jiang et al. (2022) in other forest ecosystems. These findings show that C input by deadwood contributes to decomposition processes that impact the composition of soil organic C, but this impact is a long-term process only to be observed in the long-term because most of the C characteristics were not yet significant in 2011 and 2016. The content of DOC in the soil below fallen wood at the Alnus site is higher than that of the control and for Fagus, which may be caused by a lower decomposition rate of the fallen Fagus wood compared to Alnus. Furthermore, the high concentration of DOC in leachates from deadwood in Alnus can be explained by wood structure and state of fragmentation. The Alnus wood features a soft and loose structure, resulting in greater water-holding capacity and hydrophilicity (Błońska et al. 2018).
Soil enzymes properties
The present study demonstrates the prominent role of the tree species and decay processes in promoting soil enzyme activity in the immediate vicinity of deadwood. Activities of all enzymes studied (i.e., urease, arylsulfatase, acid phosphatase, and invertase) showed a trend of increase for all tree species compared to control plots. This finding is most likely due to the increased N and P content of soils under deadwood. Moreover, the increased SOM accumulation contributes to enhanced extracellular enzymatic activity by providing a more comprehensive range of C and N substrates that can be accessed and utilized by different groups of soil microbes (Cenini et al. 2016). Urease is an enzyme widely considered to be a proxy of nitrogen (N) mineralization; therefore, its activity strongly depends on the amount of N (Keeler et al. 2009). Urease plays an influential role in the hydrolysis of urea to carbon dioxide and ammonia and may contribute to increasing soil pH. The results of Ling et al. (2014) claimed that increasing amounts of clay increased the activity of arylsulfatase and invertase (Zvyagintsev and Velikanov 1968; Mahmoud and Attia 1970). It can be concluded that increased clay accumulation in the soil around dead trees of various tree species plays an essential role in the increased activity of this enzyme (Kooch et al. 2021). P is an essential element for life, which is often limiting in terrestrial ecosystems, with organic P being the most important P fraction for soil microorganisms and acid phosphatase activity (Cleveland and Townsend 2006; Zhao et al. 2017). Other studies also determined an increased soil enzymatic activity directly impacted by decomposing deadwood (Błońska et al. 2017). The increased enzymatic activity may be related to the translocation of deadwood degradation products or the leaching of soluble compounds to the soil directly beneath. Accordingly, changes in soil microbial activity and changes in the structure of soils directly exposed to deadwood have been reported by Angst et al. (2018) and Bani et al. (2018).
Soil microbial properties
Throughout the present study, BR, SIR, MBC, and MBN showed a clear seasonality with a peak in summer. Statistically, significantly higher values for BR, SIR, MBC, and MBN were observed in soils under the direct impact of decomposing deadwood, with a maximum in the Alnus site and a minimum in the control. The high BR for Alnus may result from conditions favorable to microbial growth and enzymatic activities, due to increased input of labile organic substrates, especially in the upper soil layers (Kooch et al. 2020). In the current study, the lowest respiration activities were observed in the controls, which is probably due to the low organic matter contents and microbial activities. Deadwood had a strong effect on MBC for all tree species. Even deadwood in Fagus led to an increase in MBC with slow decomposition conditions in 2021. Microbial biomass is directly related to soil C and N contents. According to Peacock et al. (2001), the amount and quality of detritus input impact the amount of soil microbial biomass, which can be easily measured, indicating changes in soil organic C content at an early stage (Wiesmeier et al. 2019). Furthermore, microbial biomass C and N reflect the size and activity status of the soil microflora and provide a biologically bonded pool of nutrients in the soil (Nair et al. 2012). The results of this study support the view that soil microbial biomass can be efficiently used to readily detect long-term changes in soil functions, especially organic C transformation.
Soil fauna and flora properties
Earthworms are known as ecosystem engineers. It has been mentioned in many studies that earthworms have a specific food preference (Kleeman et al. 2017; Capowiez et al. 2020). Accordingly, populations around deadwood are very different from species in other environments. The results of the present study indicate that the abundance of earthworms from the Lumbricidae family and the epigeic ecological group was increased around deadwoods. According to several literature reviews (e.g., Nahidan and Ghasemzadeh 2022; Xiao et al. 2022; Zhao et al. 2022), Lumbricus terrestris is one of the most active species of earthworms in forest ecosystems of the temperate regions. Accordingly, in our study, among the different species of earthworms, Lumbricus terrestris was observed in all the treatments and years throughout the study area (see Table 2). Furthermore, the populations of collembola, Acarina, protozoa, nematodes, as well as fungi and bacteria in the soil around deadwood were significantly affected by tree species, seasons, and years; and were declining along the order of Alnus > Carpinus > Acer > Tilia > Fagus > control, respectively. Various studies have shown that in fresh wood, the density of bacteria, fungi, or microbial communities is generally minimal and increases with the progress of the decomposition process (Johnston et al. 2016; Wilhelm et al. 2019). Consistent with Gómez-Brandón et al. (2020), more ecological niches become available for microbial colonization as decomposition progresses, increasing the wood surface area and the availability of nutrients (e.g., N and P). Moreover, our results suggest increased metabolic diversity is required within bacterial and fungal communities to take advantage of the gradually available substrates as wood decay proceeds. Total N and soil pH were the main factors driving microbial communities, which is confirmed by the conclusions of Dong (2021). Particularly, increasing the N content provides more N sources for soil microbial growth and improves microbial community richness.
Relationships among deadwood species and soil properties
Generally, it can be pointed out that tree species and processes of deadwood decay have various effects on soil properties and functions (Gómez-Brandón et al. 2020; Pastorelli et al. 2021; Jomura et al. 2022). The output of PCA showed apparent differences in soil properties between plots with different tree species, showing that the decomposition period and tree species are essential factors determining soil functions. The current findings indicate that soil biological and chemical properties are decreased following tree species in the rank order of Alnus > Carpinus > Acer > Tilia > Fagus > control. Retaining deadwood trees or biological legacies can prepare a hotspot of soil functions, such as biological activities, abundance and diversity of microbial communities, and nutrient transformation in old mixed beech forests. While removing dead trees in the forest ecosystems, especially in case of Alnus and Carpinus, can threaten soil functional indicators and disrupt ecosystem services in the forests. The beneficial effects of deadwood on soil functions can suggest various strategies to improve forest sustainability and ecosystem conditions for forest management. A first step should be to prevent the removal of deadwood from forests, especially in areas of high disturbance. The next step is to determine the amount and type of deadwood that should be left in the forest to improve soil functional indicators. However, more research on deadwood is needed to plan for protection of soil function and health in sustainable forest ecosystems.
Conclusions
The deadwood of five tree species (i.e., Alnus, Tilia, Carpinus, Acer, and Fagus) strongly controls the dynamics of soil functional indicators during chemical modifications of the wood in an old mixed beech forest of northern Iran. Most of the deadwood properties differed significantly among the tree species of measured parameters for Alnus. Consequently, deadwood nutrients distinctly change with degradation and differ between tree species. In the case of Fagus deadwood, the C/N ratio was higher, and macroelement contents were lower values than for other tree species. This issue has also caused a reduction of soil nutrients around Fagus deadwood compared to other dead trees. Thus, in support of the first hypothesis of our study, we can claim that the tree species with lower C/N ratio and higher nutrient content would contribute to soil fertility more rapidly than species with more recalcitrant wood quality. Although not all features showed exactly the same pattern, our data strongly confirm the effects of deadwood on soil biological activities during the stages of wood decomposition, confirming our second hypothesis. In general, soil functions increased ranked in the order of control < Fagus < Tilia < Acer < Carpinus < Alnus, which can be assigned to the higher quality of deadwood in the different trees. It can be concluded that tree deadwood is essential in promoting soil functions and sustainability in old-growth beech forest ecosystems. Moreover, the present investigation provides a theoretical basis for the importance and applications of deadwood for sustainable forest management that can be useful in a broader range of forest ecosystems in determining the mechanisms that explain these phenomena.
References
Ahn AH (2006) Sustainable nitrogen elimination biotechnologies: a review. Process Biochem 41:1709–1721. https://doi.org/10.1016/j.procbio.2006.03.033
Alef K (1995) Soil respiration. Methods Appl Soil Microbiol Biochem 214:222
Alef, K. and Nannipieri, P. 1995. Estimation of microbial activities. Methods in Applied Soil Microbiology and Biochemistry, Academic Press, London, pp. 193–270
Allison LE (1975) Organic carbon in: black CA. Methods of soil analysis. American Society of Agronomy, Part, 2
Álvarez AG, Ibáñez JJ and Bello A (2003) Soil functions, soil quality or soil health. Scientific, metaphorical or utilitarian concepts in soil sciences.
Anderson TH, Domsch KH (1990) Application of eco-physiological quotients (qCO2 and qD) on microbial biomasses from soils of different cropping histories. Soil Biol Biochem 22:251–255. https://doi.org/10.1016/0038-0717(90)90094-G
Angst G, Messinger J, Greiner M, Häusler W, Hertel D, Kirfel K, Kögel-Knabner I, Leuschner C, Rethemeyer J, Mueller CW (2018) Soil organic carbon stocks in topsoil and subsoil controlled by parent material, carbon input in the rhizosphere, and microbial-derived compounds. Soil Biol Biochem 122:19–30. https://doi.org/10.1016/j.soilbio.2018.03.026
Asadu CLA, Nwafor IA, Chibuike GU (2015) Contributions of microorganisms to soil fertility in adjacent forest, fallow and cultivated land use types in Nsukka, Nigeria. International Journal of Agriculture and Forestry 5:199–204
Bani A, Pioli S, Ventura M, Panzacchi P, Borruso L, Tognetti R, Tonon G, Brusetti L (2018) The role of microbial community in the decomposition of leaf litter and deadwood. Appl Soil Ecol 126:75–84. https://doi.org/10.1016/j.apsoil.2018.02.017
Berg, B. and McClaugherty, C. 2008. Plant litter: decomposition, humus formation, carbon sequestration (No. 04; QH541. 5. S6, B4 2008.). Berlin: Springer.
Blair GJ, Lefroy RD, Lisle L (1995) Soil carbon fractions based on their degree of oxidation, and the development of a carbon management index for agricultural systems. Aust J Agric Res 46:1459–1466. https://doi.org/10.1071/AR9951459
Blake, G.R., Hartge, K.H., 1986. Particle density. In: Klute, A. (Ed.), Methods of soil analysis. Part 1. Physical and mineralogical methods, 2nd ed. SSSA Book Ser. 5. ASA and SSSA, Madison, WI, pp. 377–382.
Błońska E, Kacprzyk M, Spolnik A (2018) Effect of deadwood of different tree species in various stages of decomposition on biochemical soil properties and carbon storage. Ecol Res 32:193–203. https://doi.org/10.1007/s11284-016-1430-3
Błońska E, Lasota J, Tullus A, Lutter R, Ostonen I (2019) Impact of deadwood decomposition on soil organic carbon sequestration in Estonian and Polish forests. Ann for Sci 76:1–14. https://doi.org/10.1007/s13595-019-0889-9
Bond-Lamberty B, Wang C, Gower ST (2002) Annual carbon flux from woody debris for a boreal black spruce fire chronosequence. J Geophys Res Atmosph 107(D23):WFX-1. https://doi.org/10.1029/2001JD000839
Bongiorno G, Bünemann EK, Oguejiofor CU, Meier J, Gort G, Comans R, Mäder P, Brussaard L, de Goede R (2019) Sensitivity of labile carbon fractions to tillage and organic matter management and their potential as comprehensive soil quality indicators across pedoclimatic conditions in Europe. Ecol Ind 99:38–50
Bouyoucos GJ (1962) Hydrometer method improved for making particle size analyses of soils 1. J Agron 54:464–465. https://doi.org/10.2134/agronj1962.00021962005400050028x
Bower CA, Reitemeier RF, Fireman M (1952) Exchangeable cation analysis of saline and alkali soils. Soil Sciences 73:251–262
Bradford MA, Warren RJ II, Baldrian P, Crowther TW, Maynard DS, Oldfield EE, Wieder WR, Wood SA, King JR (2014) Climate fails to predict wood decomposition at regional scales. Nat Clim Chang 4:625–630. https://doi.org/10.1038/nclimate2251
Bremner, J.M. and Mulvaney, C.S. 1982. Nitrogen-total (In: Methods of Soil Analysis, Part 2, Eds: RH Miller, RR Keeney).
Brookes PC, Landman A, Pruden G, Jenkinson DS (1985) Chloroform fumigation and the release of soil nitrogen: a rapid direct extraction method to measure microbial biomass nitrogen in soil. Soil Biol Biochem 17:837–842. https://doi.org/10.1016/0038-0717(85)90144-0
Burton JM, Chen CR, Xu ZH, Ghadiri H (2007) Gross nitrogen transformations in adjacent native and plantation forests of subtropical Australia. Soil Biol Biochem 39:126–443. https://doi.org/10.1016/j.soilbio.2006.08.011
Cambardella CA, Elliott ET (1992) Particulate soil organic-matter changes across a grassland cultivation sequence. Soil Sci Soc Am J 56:777–783. https://doi.org/10.2136/sssaj1992.03615995005600030017x
Capowiez Y, Lévèque T, Pelosi C, Capowiez L, Mazzia C, Schreck E, Dumat C (2020) Using the ecosystem engineer concept to test the functional effects of a decrease in earthworm abundance due to an historic metal pollution gradient. Appl Soil Ecol 158:103816
Cenini VL, Fornara DA, McMullan G, Ternan N, Carolan R, Crawley MJ, Clément JC, Lavorel S (2016) Linkages between extracellular enzyme activities and the carbon and nitrogen content of grassland soils. Soil Biol Biochem 96:198–206. https://doi.org/10.1016/j.soilbio.2016.02.015
Christensen M, Vesterdal L (2003) Physical and chemical properties of decaying beech wood in two Danish forest reserves. Nat-Man Work Report 24:16
Cleveland CC, Townsend AR (2006) Nutrient additions to a tropical rain forest drive substantial soil carbon dioxide losses to the atmosphere. Proc Natl Acad Sci 103:10316–10321. https://doi.org/10.1073/pnas.0600989103
Dale VH, Peacock AD, Garten CT Jr, Sobek E, Wolfe AK (2008) Selecting indicators of soil, microbial, and plant conditions to understand ecological changes in Georgia pine forests. Ecol Ind 8:818–827. https://doi.org/10.1016/j.ecolind.2007.08.001
Dong X, Gao P, Zhou R, Li C, Dun X, Niu X (2021) Changing characteristics and influencing factors of the soil microbial community during litter decomposition in a mixed Quercus acutissima Carruth and Robinia pseudoacacia L. forest in Northern China. Catena 196:104811. https://doi.org/10.1016/j.catena.2020.104811
Doran JW, Parkin TB (1994) Defining and assessing soil quality. In: Doran JW, Coleman DC, Bezdicek DF, Stewart BA (eds) Defining soil quality for a sustainable environment. Soil science society of America and American society of agronomy, Madison, WI, USA, pp 1–21. https://doi.org/10.2136/sssaspecpub35.c1
Drobnik T, Greiner L, Keller A, Grêt-Regamey A (2018) Soil quality indicators–from soil functions to ecosystem services. Ecol Ind 94:151–169. https://doi.org/10.1016/j.ecolind.2018.06.052
Elliott ET, Cambardella CA (1991) Physical separation of soil organic matter. Agr Ecosyst Environ 34:407–419. https://doi.org/10.1016/0167-8809(91)90124-G
Ezeigbo OR, Ukpabi CF, Abel-Anyebe O, Okike-Osisiogu FU, Ike-Amadi CA, Agomoh NG (2013) Physicochemical properties of soil contaminated with refined petroleum oil in Eluama community, Abia State, Nigeria. Int J Sci Res Manag 1:405–413
Fukami T, Dickie IA, Paula Wilkie J, Paulus BC, Park D, Roberts A, Buchanan PK, Allen RB (2010) Assembly history dictates ecosystem functioning: evidence from wood decomposer communities. Ecol Lett 13:675–684. https://doi.org/10.1111/j.1461-0248.2010.01465.x
Garrett LG, Oliver GR, Pearce SH, Davis MR (2008) Decomposition of Pinus radiata coarse woody debris in New Zealand. For Ecol Manage 255:3839–3845. https://doi.org/10.1016/j.foreco.2008.03.031
Gómez-Brandón M, Probst M, Siles JA, Peintner U, Bardelli T, Egli M, Insam H, Ascher-Jenull J (2020) Fungal communities and their association with nitrogen-fixing bacteria affect early decomposition of Norway spruce deadwood. Soil Sci Soc Am J 65:1272–1279. https://doi.org/10.2136/sssaj2001.6541272x
Greiner L, Keller A, Grêt-Regamey A, Papritz A (2017) Soil function assessment: review of methods for quantifying the contributions of soils to ecosystem services. Land Use Policy 69:224–237. https://doi.org/10.1016/j.landusepol.2017.06.025
Harmon ME, Fasth B, Woodall CW, Sexton J (2013) Carbon concentration of standing and downed woody detritus: effects of tree taxa, decay class, position, and tissue type. For Ecol Manage 291:259–267. https://doi.org/10.1016/j.foreco.2012.11.046
Hart SC (1999) Nitrogen transformations in fallen tree boles and mineral soil of an old-growth forest. Ecology 80:1385–1394. https://doi.org/10.1890/0012-9658(1999)080[1385:NTIFTB]2.0.CO;2
Herrmann S, Bauhus J (2013) Effects of moisture, temperature and decomposition stage on respirational carbon loss from coarse woody debris (CWD) of important European tree species. Scand J for Res 2:346–357. https://doi.org/10.1080/02827581.2012.747622
Herrmann S, Bauhus J (2018) Nutrient retention and release in coarse woody debris of three important central European tree species and the use of NIRS to determine deadwood chemical properties. Forest Ecosyst 5:1–15. https://doi.org/10.1186/s40663-018-0140-4
Herrmann S, Kahl T, Bauhus J (2015) Decomposition dynamics of coarse woody debris of three important central European tree species. Forest Ecosyst 2:1–14. https://doi.org/10.1186/s40663-015-0052-5
Hicks WT, Harmon ME, Myrold DD (2003) Substrate controls on nitrogen fixation and respiration in woody debris from the Pacific Northwest, USA. For Ecol Manage 176:25–35. https://doi.org/10.1016/S0378-1127(02)00229-3
Hollands C, Shannon VL, Sawicka K, Vanguelova EI, Benham SE, Shaw LJ, Clark JM (2022) Management impacts on the dissolved organic carbon release from deadwood, ground vegetation and the forest floor in a temperate Oak woodland. Sci Total Environ 805:150399. https://doi.org/10.1016/j.scitotenv.2021.150399
Homer DC, Pratt PF (1961) Methods of analysis for soils, plants and waters. University of California Division of Agricultural Sciences, Berkeley, CA, p 309
Hosseini SM (2019) Outstanding universal values of Hyrcanian Forest, the newest Iranian property, inscribed in the UNESCO’s World Heritage List. Tourism Research, 10:1(3):1–7.
Hutson BR, Veitch LG (1987) Densities of collembola and Acarina in the soil and litter of three indigenous South Australian forests related to layer, site and seasonal differences. Austral Ecol 12:239–261. https://doi.org/10.1111/j.1442-9993.1987.tb00946.x
Jamil N, Sajjad N, Ashraf H, Masood Z, Bazai ZA, Khan R, Anjum N, Akmal F, Arbab N, Tareen P (2016) Physical and chemical properties of soil quality indicating forests productivity: a review. J Toxicol Sci 8:60–68. https://doi.org/10.5829/idosi.aejts.2016.8.2.10312
Jeffery S, Gardi C, Jones A, Montanarella L, Marmo L, Miko L, Ritz K, Peres G, Rombke J, van der Putten WH (eds) (2010) European atlas of soil biodiversity. European Commission, Publications Office of the European Union, Luxembourg
Jiang, X., Dang, P., Yan, W., Zheng, W., Xu, Y. and Chen, Y. 2022. Significant effects of dead trees on active organic carbon components and microbial communities in subtropical forests.
Johnston SR, Boddy L, Weightman AJ (2016) Bacteria in decomposing wood and their interactions with wood-decay fungi. FEMS Microbiol Ecol 92(11):fiw179. https://doi.org/10.1093/femsec/fiw179
Jomura M, Yoshida R, Michalčíková L, Tláskal V, Baldrian P (2022) Factors controlling dead wood decomposition in an old-growth temperate forest in Central Europe. J Fungi 8(7):673. https://doi.org/10.3390/jof8070673
Jones DL, Willett VB (2006) Experimental evaluation of methods to quantify dissolved organic nitrogen (DON) and dissolved organic carbon (DOC) in soil. Soil Biol Biochem 38:991–999. https://doi.org/10.1016/j.soilbio.2005.08.012
Kahl T, Arnstadt T, Baber K, Bässler C, Bauhus J, Borken W, Buscot F, Floren A, Heibl C, Hessenmöller D, Hofrichter M (2017) Wood decay rates of 13 temperate tree species in relation to wood properties, enzyme activities and organismic diversities. For Ecol Manage 391:86–95. https://doi.org/10.1016/j.foreco.2017.02.012
Keeler BL, Hobbie SE, Kellogg LE (2009) Effects of long-term nitrogen addition on microbial enzyme activity in eight forested and grassland sites: implications for litter and soil organic matter decomposition. Ecosystems 12:1–15. https://doi.org/10.1007/s10021-008-9199-z
Kemper WD, Rosenau RC (1986) Aggregate stability and size distribution. In: Klute A (ed) Methods of soil analysis, Part 1: physical and mineralogical methods, 2nd edn. American Society of Agronomy, Madison, Wisconsin, pp 383–411
Kleeman J, Baysal G, Bulley HNN, Furst C (2017) Assessing driving forces of land use and land cover change by a mixed-method approach in North-Eastern Ghana, West Africa. J Environ Manage 196:411–442
Kooch Y (2012) Response of earthworms’ ecological groups to decay degree of dead trees (Case study: sardabrood forest of Chalous, Iran). Europ J Experim Biol 2:532–538
Kooch Y, Noghre N (2020) Nutrient cycling and soil-related processes under different land covers of semi-arid rangeland ecosystems in northern Iran. Catena 193:104621. https://doi.org/10.1016/j.catena.2020.104621
Kooch Y, Zaccone C, Lamersdorf NP, Tonon G (2014) Pit and mound influence on soil features in an oriental beech (Fagus orientalis Lipsky) forest. Eur J Forest Res 133:347–354. https://doi.org/10.1007/s10342-013-0766-2
Kooch Y, Samadzadeh B, Hosseini SM (2017) The effects of broad-leaved tree species on forest floor quality and soil properties in a plain forest stand. CATENA 150:223–229
Kooch Y, Ghorbanzadeh N, Wirth S, Novara A, Piri AS (2021) Soil functional indicators in a mountain forest-rangeland mosaic of northern Iran. Ecolog Indicat 126:107672. https://doi.org/10.1016/j.ecolind.2021.107672
Kooch Y, Ghorbanzadeh N, Hajimirzaaghaee S, Egli M (2022) Soil functional indicators in mixed beech forests are clearly species-specific. J Forestry Res. https://doi.org/10.1007/s11676-022-01548-4
Kooch Y (2008) Determination and differentiation of plant ecological units and relation to some soil properties in Khanikan Forest of Chalus. M. Sc. thesis of Forestry, Mazandaran University, 149p.
Krankina ON, Harmon ME, Griazkin AV (1999) Nutrient stores and dynamics of woody detritus in a boreal forest: modeling potential implications at the stand level. Can J for Res 29:20–32. https://doi.org/10.1139/x98-162
Laiho R, Prescott CE (2004) Decay and nutrient dynamics of coarse woody debris in northern coniferous forests: a synthesis. Can J for Res 34:763–777. https://doi.org/10.1139/x03-241
Larson WE, Pierce FJ (1994) The dynamics of soil quality as a measure of sustainable management. Defin Soil Quality Sustain Environ 35:37–51. https://doi.org/10.2136/sssaspecpub35.c3
Lasota J, Błońska E, Piaszczyk W, Wiecheć M (2018) How the deadwood of different tree species in various stages of decomposition affected nutrient dynamics? J Soils Sediments 18:2759–2769. https://doi.org/10.1007/s11368-017-1858-2
Lassauce A, Paillet Y, Jactel H, Bouget C (2011) Deadwood as a surrogate for forest biodiversity: meta-analysis of correlations between deadwood volume and species richness of saproxylic organisms. Ecol Ind 11:1027–1039. https://doi.org/10.1016/j.ecolind.2011.02.004
Lee Ch, Tim D, Sun FG (2009) Nitrogen removal in constructed wetland systems. Eng Life Sci 9:11–22. https://doi.org/10.1002/elsc.200800049
Li ZA, Peng SL, Rae DJ, Zhou GY (2001) Litter decomposition and nitrogen mineralization of soils in subtropical plantation forests of southern China, with special attention to comparisons between legumes and non-legumes. Plant Soil 229:105–116. https://doi.org/10.1023/A:1004832013143
Li M, Zhou X, Zhang Q, Cheng X (2014) Consequences of afforestation for soil nitrogen dynamics in central China. Agr Ecosyst Environ 183:40–46. https://doi.org/10.1016/j.agee.2013.10.018
Liang W, Lou Y, Li Q, Zhong S, Zhang X, Wang J (2009) Nematode faunal response to long-term application of nitrogen fertilizer and organic manure in Northeast China. Soil Biol Biochem 41:883–890. https://doi.org/10.1016/j.soilbio.2008.06.018
Ling N, Sun Y, Ma J, Guo J, Zhu P, Peng C, Shen Q (2014) Response of the bacterial diversity and soil enzyme activity in particle-size fractions of mollisol after different fertilization in a long-term experiment. Biol Fertil Soils 50:901–911. https://doi.org/10.1007/s00374-014-0911-1
Magnússon RJ, Tietema A, Cornalissen JHC, Hefting MM, Kalbitz K (2016) Tamm review: sequestration of carbon from coarse woody debris in forest soils. For Ecol Manage 377:1–15. https://doi.org/10.1016/j.foreco.2016.06.033
Mahmoud SAZ and Attia RM (1970) Amylase adsorption in soils. J Soil Sci United Arab Rep 10, 143–153. https://www.cabdirect.org/cabdirect/abstract/19711902235.
Matus FJ (2021) Fine silt and clay content is the main factor defining maximal C and N accumulations in soils: a meta-analysis. Sci Reports 11:1–17. https://doi.org/10.1038/s41598-021-84821-6
Mayer M, Rosinger C, Gorfer M, Berger H, Deltedesco E, Bässler C, Müller J, Seifert L, Rewald B, Godbold DL (2022) Surviving trees and deadwood moderate changes in soil fungal communities and associated functioning after natural forest disturbance and salvage logging. Soil Biology Biochem 166:108558. https://doi.org/10.1016/j.soilbio.2022.108558
Mayzlish E, Steinberger Y (2004) Effects of chemical inhibitors on soil protozoan dynamics in a desert ecosystem. Biol Fertil Soils 39:415–421. https://doi.org/10.1007/s00374-004-0723-9
Mc Kinley DC, Rice CW, Blair JM (2008) Conversion of grassland to coniferous woodland has limited effects on soil nitrogen cycle processes. Soil Biol Biochem 40:2627–2633. https://doi.org/10.1016/j.soilbio.2008.07.005
Minnich C, Peršoh D, Poll C, Borken W (2021) Changes in chemical and microbial soil parameters following 8 years of deadwood decay: an experiment with logs of 13 tree species in 30 forests. Ecosystems 24:955–967. https://doi.org/10.1007/s10021-020-00562-z
Moghimian N, Jalali SG, Kooch Y, Ana REY (2020) Downed logs improve soil properties in old-growth temperate forests of northern Iran. Pedosphere 30(3):378–389. https://doi.org/10.1016/S1002-0160(17)60424-7
Nahidan S, Ghasemzadeh M (2022) Biochemical phosphorus transformations in a calcareous soil as affected by earthworm, cow manure and its biochar additions. Appl Soil Ecol 170:104310
Nair A, Ngouajio M (2012) Soil microbial biomass, functional microbial diversity, and nematode community structure as affected by cover crops and compost in an organic vegetable production system. Appl Soil Ecol 58:45–55. https://doi.org/10.1016/j.apsoil.2012.03.008
Norden B, Ryberg M, Götmark F, Olausson B (2004) Relative importance of coarse and fine woody debris for the diversity of wood-inhabiting fungi in temperate broadleaf forests. Biolog Conser 117:1–10. https://doi.org/10.1016/S0006-3207(03)00235-0
Odriozola I, Abrego N, Tláskal V, Zrůstová P, Morais D, Větrovský T, Ovaskainen O, Baldrian P (2021) Fungal communities are important determinants of bacterial community composition in deadwood. Msystems 6(1):01017–01020. https://doi.org/10.1128/mSystems.01017-20
Paltto H, Nordén B, Götmark F (2008) Partial cutting as a conservation alternative for oak (Quercus spp.) forest—response of bryophytes and lichens on dead wood. Forest Ecology Manag 256:536–547. https://doi.org/10.1016/j.foreco.2008.04.045
Parsapour MK, Kooch Y, Hosseini SM, Alavi SJ (2018) Litter and topsoil in Alnus subcordata plantation on former degraded natural forest land: a synthesis of age-sequence. Soil and Tillage Res 179:1–10. https://doi.org/10.1016/j.still.2018.01.008
Pastorelli R, Paletto A, Agnelli AE, Lagomarsino A, De Meo I (2021) Microbial diversity and ecosystem functioning in deadwood of black pine of a temperate forest. Forests 12(10):1418. https://doi.org/10.3390/f12101418
Peacock AG, Mullen MD, Ringelberg DB, Tyler DD, Hedrick DB, Gale PM, White DC (2001) Soil microbial community responses to dairy manure or ammonium nitrate applications. Soil Biol Biochem 33(7–8):1011–1019. https://doi.org/10.1016/S0038-0717(01)00004-9
Perie C, Ouimet R (2008) Organic carbon, organic matter and bulk density relationships in boreal forest soils. Canad J Soil Sci 88:315–325. https://doi.org/10.4141/CJSS06008
Perreault L, Forrester JA, Mladenoff DJ, Lewandowski TE (2021) Deadwood reduces the variation in soil microbial communities caused by experimental forest gaps. Ecosystems 24:1928–1943. https://doi.org/10.1007/s10021-021-00624-w
Persoh D (2015) Plant-associated fungal communities in the light of metalogics. Fungal Div 75:1–25. https://doi.org/10.1007/s13225-015-0334-9
Piaszczyk W, Błoska E, Lasota J (2019) Soil biochemical properties and stabilization of soil organic matter in relation to deadwood of different species. FEMS Microbiol Ecol 95:1–11. https://doi.org/10.1080/11263500903561114
Pires LF, Brinatti AM, Saab SC, Cássaro FA (2014) Porosity distribution by computed tomography and its importance to characterize soil clod samples. Appl Radiat Isot 92:37–45. https://doi.org/10.1016/j.apradiso.2014.06.010
Plaster, E.J. 1985. Soil science and management. Delmar Publishers Inc., Albany, p 124.
Prévost M (2004) Predicting soil properties from organic matter content following mechanical site preparation of forest soils. Soil Sci Soc Am J 68:943–949. https://doi.org/10.2136/sssaj2004.9430
Rothe A, Cromack JK, Resh SC, Makeneci E, Son Y (2002) Soil carbon and nitrogen changes under Douglas-fir with and without red alder. Soil Sci Soc Am J 66:1988–1995. https://doi.org/10.2136/sssaj2002.1988
Sacher P, Meyerhoff J, Mayer M (2022) Evidence of the association between deadwood and forest recreational site choices. Forest Policy Econ 135:102638
Sariyildiz T, Savaci G, Kravkaz IS (2015) Effects of tree species stand age and land use change on soil carbon and nitrogen stock rates in northwestern Turkey. Iforest 9:165–170
Scheller RM, Mladenoff DJ (2002) Understory species patterns and diversity in old-growth and managed northern hardwood forests. Ecol Appl 12:1329–1343. https://doi.org/10.1890/1051-0761(2002)012[1329:USPADI]2.0.CO;2
Schinner F, Von Mersi W (1990) Xylanase-, CM-cellulase-and invertase activity in soil: an improved method. Soil Biol Biochem 22(4):511–515
Scott CE, Rap A, Spracklen DV, Forster PM, Carslaw KS, Mann GW, Pringle KJ, Kivekäs N, Kulmala M, Lihavainen H, Tunved P (2014) The direct and indirect radiative effects of biogenic secondary organic aerosol. Atmos Chem Phys 14:447–470
Shannon VL, Vanguelova EI, Morison JIL, Shaw LJ, Clark JM (2022) The contribution of deadwood to soil carbon dynamics in contrasting temperate forest ecosystems. Europ J Forest Res 214:1–12. https://doi.org/10.1007/s10342-021-01435-3
Spiering DJ, Knight RL (2005) Snag density and use by cavity-nesting birds in managed stands of the black hills national forest. For Ecol Manage 214:40–52. https://doi.org/10.1016/j.foreco.2005.03.054
Robertson GP, Coleman DC, Sollins P and Bledsoe CS eds (1999) Standard soil methods for long-term ecological research (Vol. 2). Oxford University Press on Demand.
Stutz KP, Dann D, Wambsganss J, Scherer-Lorenzen M, Lang F (2017) Phenolic matter from deadwood can impact forest soil properties. Geoderma 288:204–212. https://doi.org/10.1016/j.geoderma.2016.11.014
Talebi KS, Sajedi T and Pourhashemi M (2014) Forests of Iran. A treasure from the past, a hope for the future 10.
Teixeira HM, Cardoso IM, Bianchi FJ, da Cruz Silva A, Jamme D, Peña-Claros M (2020) Linking vegetation and soil functions during secondary forest succession in the Atlantic forest. Forest Ecology and Management 457:117696. https://doi.org/10.1016/j.foreco.2019.117696
Thomas SC, Martin AR (2012) Carbon content of tree tissues: a synthesis. Forests 3:332–352. https://doi.org/10.3390/f3020332
Tláskal V, Brabcová V, Větrovský T, Jomura M, López-Mondéjar R, Oliveira Monteiro LM, Saraiva JP, Human ZR, Cajthaml T, Nunes da Rocha U, Baldrian P (2021) Complementary roles of wood-inhabiting fungi and bacteria facilitate deadwood decomposition. Msystems 6(1):01078–01120. https://doi.org/10.1128/mSystems.01078-20
Uhl B, Krah FS, Baldrian P, Brandl R, Hagge J, Müller J, Thorn S, Vojtech T, Bässler C (2022) Snags, logs, stumps, and microclimate as tools optimizing deadwood enrichment for forest biodiversity. Biolog Conser 270:109569. https://doi.org/10.1016/j.biocon.2022.109569
Vallejo VE, Gómez MM, Cubillos AM, Roldán F (2011) Effect of land use on the density of nitrifying and denitrifying bacteria in the colombian coffee region. Agronom Colomb J 29:455–464
Villada A (2013) Evaluation of tree species and soil type interactions for their potential for long-term C sequestration (Doctoral dissertation, University of Reading).
Wambsganss J, Stutz KP, Lang F (2017) European beech deadwood can increase soil organic carbon sequestration in forest topsoil’s. For Ecol Manage 405:200–209. https://doi.org/10.1016/j.foreco.2017.08.053
Weiss P, Schieler K, Schadauer K, Radunsky K, Englisch M (2000) Carbon budget of Austrian forests and considerations about Kyoto Protocol. Monograph M-106 94:221–228
Wienhold BJ, Andrews SS, Karlen DL (2004) Soil quality: a review of the science and experiences in the USA. Environ Geochem Health 26:89–95. https://doi.org/10.1023/B:EGAH.0000039571.59640.3c
Wiesmeier M, Urbanski L, Hobley E, Lang B, von Lützow M, Marin-Spiotta E, van Wesemael B, Rabot E, Ließ M, Garcia-Franco N, Wollschläger U (2019) Soil organic carbon storage as a key function of soils-a review of drivers and indicators at various scales. Geoderma 333:149–162. https://doi.org/10.1016/j.geoderma.2018.07.026
Wilhelm RC, Singh R, Eltis LD, Mohn WW (2019) Bacterial contributions to delignification and lignocellulose degradation in forest soils with metagenomics and quantitative stable isotope probing. ISME J 13:413–429. https://doi.org/10.1038/s41396-018-0279-6
Wollum AG (1982) Cultural methods for soil microorganisms. In: Page AL et al (eds) Methods of soil analysis, Part 2. American society of agronomy and soil science society of America, Madison, WI, pp 781–801
Woodall CW, Evans DM, Fraver S, Green MB, Lutz DA, Damato AW (2020) Real-time monitoring of deadwood moisture in forests Lessons learned from an intensive case study. Canad J Forest Res 50(11):1244. https://doi.org/10.1139/cjfr-2020-0110
Xiao R, Ali A, Xu Y, Abdelrahman H, Li R, Lin Y, Bolan N, Shahen SMJ, Zhang Z (2022) Earthworms as candidates for remediation of potentially toxic elements contaminated soils and mitigating the environmental and human health risks: a review. Environ Int 158:106924
Yan E, Wang XH, Chen XY (2007) Impacts of evergreen broad-leaved forest degradation on soil nutrients and carbon pools in Tiantong, Zhejiang province. Acta Ecol Sin 26:706–714
Yang FF, Li YL, Zhou GY, Wenigmann KO, Zhang DQ, Wenigmann M, Liu SZ, Zhang QM (2010) Dynamics of coarse woody debris and decomposition rates in an old-growth forest in lower tropical China. For Ecol Manage 259:1666–1672. https://doi.org/10.1016/j.foreco.2010.01.046
Yue CHEN, Huang Y, Wenjuan SUN (2017) Using organic matter and pH to estimate the bulk density of afforested/reforested soils in Northwest and northeast China. Pedosphere 27:890–900. https://doi.org/10.1016/S1002-0160(17)60372-2
Zhao M, Feng H, Han J, Ao H, Qian Z (2017) Multi-stimuli responsive copper nanoclusters with bright red luminescence for quantifying acid phosphatase activity via redox-controlled luminescence switch. Anal Chim Acta 984:202–210. https://doi.org/10.1016/j.aca.2017.06.046
Zhao S, Zhang Z, Chen L, Cui Q, Cui Y, Song D, Fang L (2022) Review on migration, transformation and ecological impacts of micro plastics in soil. Appl Soil Ecol 176:104486
Zuo J, Berg MP, Van Hal J, Van Logtestijn RS, Goudzwaard L, Hefting MM, Poorter L, Sterck FJ, Cornelissen JH (2021) Fauna community convergence during decomposition of deadwood across tree species and forests. Ecosystems 24:926–938. https://doi.org/10.1007/s10021-020-00558-9
Zvyagintsev DG, Velikanov LL (1968) Influence of adsorbents on the activity of bacteria growing on media with amino acids. Microbiology (USSR) 37:861–866
Acknowledgements
The authors express their thanks to Tarbiat Modares University for the financial support of the study reported in this paper.
Author information
Authors and Affiliations
Contributions
Y.K. Statistics, Writing-original draft, Supervision. Azam Nouraei: Writing-original draft. Stephan Wirth: Writing-revision and editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Communicated by Gediminas Brazaitis.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kooch, Y., Nouraei, A. & Wirth, S. Deadwoods are hotspots for soil functions in old-growth beech forests: monitoring during 15 years after a windthrow event. Eur J Forest Res 142, 1401–1421 (2023). https://doi.org/10.1007/s10342-023-01598-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10342-023-01598-1