Abstract
The diagnosis of amyotrophic lateral sclerosis (ALS) requires both upper and lower motor neuron signs. However, quite a few patients with ALS lack the upper motor neuron sign during the disease. This study sought to investigate whether metabolites, including glutamate (Glu), N-acetyl aspartate (NAA), and gamma aminobutyric acid (GABA), in the supplementary motor area (SMA) measured by magnetic resonance spectroscopy (MRS), could be a surrogate biomarker for ALS. Twenty-five patients with ALS and 12 controls underwent 3.0-T MR scanning, which measured Glu, NAA, and GABA. Finally, receiver operating characteristic (ROC) curves were created and the area under curve (AUC) was calculated to assess the diagnostic power. Logistic regression analysis revealed the usefulness of both Glu and NAA for the differentiation of ALS from controls (Glu, P = 0.009; NAA, P = 0.033). The ratio of Glu to NAA or GABA was significantly increased in patients with ALS (Glu/NAA, P = 0.027; Glu/GABA, P = 0.003). Both the AUCs were more than 0.7, with high specificity but low sensitivity. The present findings might indicate that both the Glu/NAA and the Glu/GABA ratios in the SMA could be potential biomarkers for the diagnosis of ALS.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The clinical diagnosis of amyotrophic lateral sclerosis (ALS) requires both upper and lower motor neuron signs. The upper motor neuron signs depend on excitability of the neurons in the primary motor cortex and anterior horn of the spinal cord, and the volume of the muscles. These factors change during disease progression and contribute to the clinical presentation of upper motor neuron signs, which might be associated with the ALS cases without upper motor neuron signs during the disease course. Indeed, one pathological study reported that 84.6% of the patients with primary muscular atrophy, one of the motor neuron diseases without upper motor neuron signs, showed degeneration of the upper motor neurons [1]. There are concerns that quite a few patients actually having ALS are not diagnosed with ALS because of the lack of upper motor neuron signs and thus fail to subsequently receive future effective therapies. To address this issue, surrogate biomarkers are required for upper motor neuron signs. Magnetic resonance spectroscopy (MRS) is an emerging method for the assessment of metabolites, including glutamate (Glu), N-acetyl aspartate (NAA), and gamma aminobutyric acid (GABA), as candidate biomarkers [2]. Glu-induced excitotoxicity is accepted as one of the causes of ALS [3]. Previous studies have focused on metabolites measured by MRS in the primary motor cortex (M1), as M1 is key to the pathology/pathophysiology of ALS [4,5,6,7,8,9,10,11,12,13,14,15,16,17,18,19,20,21]. The M1s in the two hemispheres are distant from each other; therefore, a double scan is needed to measure the metabolites, and leads to prolongation of scan time. In contrast, the supplementary motor area (SMA) allows for a single scan to measure the metabolites on both sides. Given the frontotemporal-lobar-degeneration-ALS spectrum [22], SMA is expected to be another key player in ALS. Additional measurements in the SMA, derived from both diffusion tensor imaging (DTI) and resting state functional MRI, could further improve accuracy for the diagnosis of ALS. We aimed at investigating whether the abnormalities in metabolites including Glu and GABA, and in the MRI-derived measurements, were detectable in the SMA of patients with ALS.
Material and methods
Subjects
A total of 37 subjects were consecutively recruited from the Department of Neurology of the Tokushima University Hospital between August 2014 and March 2018. All subjects underwent both MRI and MRS. We obtained data on age, sex, handedness, disease duration from symptom onset, disease severity, and mini-mental state examination (MMSE) scores. Furthermore, the revised El Escorial criteria were used for the clinical diagnosis of ALS. All patients satisfied the clinical diagnostic criteria of possible, probable-laboratory-supported, probable, or definite ALS. In addition, 6 of 25 patients were pathologically diagnosed with ALS. The progression rate was calculated according to the following formula: (48 - ALS functional rating scale revised (ALSFRS-R))/disease duration from symptom onset. In contrast, subjects with an aging brain MRI and without neurological deficits were defined as normal controls. The features of the included subjects are shown in Table 1.
Image acquisition
Images were acquired on a 3.0-T Discovery 750 scanner (GE, Milwaukee, WI) equipped with a standard head coil. Scan parameters have been separately described in the following section.
MRS
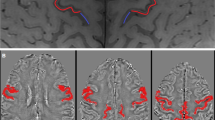
T2-weighted images were acquired before the 1H-MRS examination, and a single volume of interest (VOI: 30 × 30 × 30 mm) was included in the SMA (Fig. 1a). 1H-MR spectra were obtained using a point resolved echo spectroscopic sequence (PRESS) with the following parameters: repetition time (TR) = 2000 ms, echo time (TE) = 30 ms, and number of signals averaged = 128, to measure the concentration of both Glu and NAA (Fig. 1b). A Mesher-Garwood PRESS (MEGA-PRESS) was used to determine the GABA concentration with the following parameters: TR = 1500 ms, TE = 30 ms, and number of signals averaged = 256 (Fig. 1c). Metabolite concentrations were estimated by the LCModel (version 6.3) and quantified using Cramer-Rao lower bounds (CRLB), which yield the estimated error of the metabolite quantification [19]. The measured values with CRLB below 20% were included in the analyses for Glu and NAA, while those with a CRLB below 25% were included in the analyses for GABA. One patient was missing Glu concentration due to large CRLB.
VOIs for both MRS and MRI, and spectra. The MRS VOI in the SMA (a). The MRS spectra of PRESS (b) and MEGA-PRESS (c). The VOI in the SMA for DTI and RS-fMRI (d). GABA, gamma aminobutyric acid; Gln, glutamine; Glu, glutamate; DTI, diffusion tensor imaging; MEGA-PRESS, Mesher-Garwood point resolved echo spectroscopic sequence; MRS, magnetic resonance spectroscopy; NAA, N-acetyl aspartate; PRESS, point resolved echo spectroscopic sequence; RS-fMRI, resting state functional magnetic resonance imaging; SMA, supplementary motor cortex; VOI, volume of interest
DTI
Scan parameters included a diffusion gradient direction of 33, b value of 800 s/mm2, field of view (FOV) of 240 mm, matrix of 128 × 128, TR of 15,000 ms, TE of 84.9 ms, flip angle of 90, and slice thickness of 2.5 mm. Details on image preprocessing have been described elsewhere [23]. Briefly, FA maps were generated after both correction of Eddy current-induced distortions and head motion, and extraction of non-brain tissue. Non-linear registration was employed for automatic calculation of the FA value in the SMA followed by smoothing (8 × 8 × 8 mm3 kernel). The VOI of the SMA was derived from an automated anatomical labeling (AAL) atlas (Fig. 1d) [24]. The FA value was the mean in the SMA. Two patients were missing FA maps, either because of an extremely large distortion or discontinuation of the scan.
Resting state functional MRI
Details of the methods of image acquisition and preprocessing have been described elsewhere [25, 26]. Scan parameters were as follows: FOV, 240 mm; matrix, 64 × 64; TR, 2000 ms; TE, 27.2 ms; flip angle, 77; slice thickness, 3.0 mm. All volumes were subjected to motion correction, slice-timing correction, and non-linear registration to the Montreal Neurological Institute (MNI-152: 2 × 2 × 2 mm3) template [27]. The time series for each voxel was temporally band-pass filtered (0.01–0.08 Hz) to remove the effects of very-low-frequency drift and high-frequency respiratory and cardiac noise [28, 29]. Regional homogeneity (ReHo) is a method to measure regional synchronization of the blood oxygen level–dependent (BOLD) signal across the whole brain using Kendall’s coefficient of concordance, which assumes that synchronization of the BOLD signal within a functional cluster could be changed or modulated by different conditions [30]. Individual ReHo maps were divided by each mean value of the ReHo and were then smoothed with a Gaussian filter of 8 mm full width at half-maximum (FWHM). The VOI of the ReHo was the same as that of the DTI (Fig. 1d). Two patients were missing their ReHo maps, either because of artifacts or because of discontinuation of the scan.
Statistics
This study was performed according to STARD recommendation. The between-group difference was analyzed using a two-sided unpaired Student’s t test. Furthermore, forward stepwise logistic regression analysis was used as a multivariate analysis to determine which factors contributed to the diagnosis of ALS with the maximum likelihood estimation technique. The factors used for multivariate analysis were GABA, Glu, NAA, ReHo, FA, sex, and age. Pearson’s product moment correlation coefficient was used for correlation analysis between two factors, such as GABA, Glu, NAA, ReHo, FA, age, disease duration, ALSFRS-R, progression rate, and MMSE. Each receiver operating characteristic (ROC) curve was created and an area under curve (AUC) was calculated to assess the diagnostic power. We defined a P value of less than 0.05 as statistically significant. All statistical analyses were performed using the Statistical Package for the Social Sciences version 21 (IBM, Armonk, NY).
Results
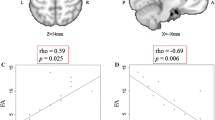
Neither metabolites, FA, nor ReHo reach significant difference between groups, although Glu, NAA, GABA, and ReHo tended to show significant differences (Glu, P = 0.103; NAA, P = 0.138; GABA, P = 0.202; ReHo, P = 0.064; FA, P = 0.600; Fig. 2a and supplementary Fig. 1A). Specifically, the ratio of Glu to NAA or GABA was significantly increased in patients with ALS when compared with control subjects (Glu/NAA, P = 0.027; Glu/GABA, P = 0.003; Fig. 2b). This result indicated an increased concentration of Glu and a decreased concentration of NAA and GABA in the ALS group when compared with the control group. Both the AUCs were more than 0.7, with high specificity, suggesting that both the Glu/NAA and the Glu/GABA ratios were suitable for definitive diagnosis (Glu/NAA, AUC = 0.74, P = 0.019, cut off = 0.95, sensitivity = 0.50, specificity = 0.917; Glu/GABA, AUC = 0.71, P = 0.048, cut off = 3.93, sensitivity = 0.54, specificity = 0.917; Fig. 2b). Furthermore, logistic regression analysis revealed the usefulness of Glu and NAA for the differentiation between ALS and controls (omnibus test of model coefficients, P = 0.004; Glu, P = 0.009, odds ratio = 2.10; NAA, P = 0.033, odds ratio = 0.33; Hosmer-Lemeshow goodness-of-fit, P = 0.115; GABA, P = 0.258; ReHo, P = 0.292; FA, P = 0.816; Sex, P = 0.261; Age, P = 0.850; Fig. 2a). Finally, there were significant correlations between NAA and age or Glu, and between FA and ReHo (NAA against Age, P = 0.049, r = −0.40; NAA against Glu, P = 0.001, r = 0.65; FA against ReHo, P < 0.001, r = 0.70; Fig. 3 and supplementary Fig. 1B).
Group differences in Glu, NAA, GABA, and the Glu/NAA and Glu/GABA ratios. Logistic regression analysis revealed significant differences in Glu and NAA between groups (a). Both the Glu/NAA and Glu/GABA ratios were found to be significantly increased in the ALS group when compared with the control group. Furthermore, the ROC curve demonstrated a moderate accuracy of the ratios for the diagnosis of ALS (b). ALS, amyotrophic lateral sclerosis; Ctr, control; GABA, gamma aminobutyric acid; Glu, glutamate; NAA, N-acetyl aspartate; ROC, receiver operating characteristic. # and * indicate significant difference with logistic regression analysis and Student’s t test, respectively
Discussion
The VOI in the SMA allowed for a single measurement of the metabolite concentration on both sides, in contrast to the double measurement required for the VOI in the M1. In this study, the VOI in the SMA helped measure the concentrations of Glu, NAA and GABA, and revealed an increased concentration of Glu and a reduced concentration of NAA in the patients with ALS. Specifically, both the Glu/NAA and Glu/GABA ratios were elevated in the ALS group when compared with the control group. Multivariate analysis demonstrated that the combination of Glu and NAA was best to differentiate patients with ALS from the controls, whereas the univariate analysis did not show any significant difference in each level of either the Glu or the NAA between the groups due to a lack of statistical power. Several lines of evidence suggest the presence of reduced levels of NAA in several regions, including the M1, in ALS [4,5,6,7,8,9,10,11,12,13,14,15, 17, 20, 21], while results of Glu or Glx (Glu + glutamine) were heterogeneous. Previous studies reported a decreased level of Glx in the medulla [5], an increased level of Glx and Glu in the M1 [15, 16], no significant difference in Glx or Glu in the M1, occipital cortex or brainstem [4, 31], and a decreased level of Glu in the M1 [6]. Furthermore, a 7-T MRI was applied to assess Glu concentration, and previous studies reported two different results: one reported no significant difference in the M1 or pons [21]; the other reported reduced level of Glu in the M1 [20]. The results on Glu concentration were heterogeneous in the M1, in spite of the 7-T MRI, although one longitudinal study reported the association of dynamic changes in motor cortex NAA with progression of upper limb function, and in pons Glx with progression of bulbar function [32]. Excitotoxicity is considered to play a key role in ALS pathophysiology from the point of view of Glu [3]. Riluzole, an inhibitor of Glu release, is one option for the treatment of patients with ALS on the basis of the concept of Glu-induced excitotoxicity [3]. Glu concentration in the cerebrospinal fluid was increased in patients with ALS [33], and knock-down of the metabotropic glutamate receptor 1 prolonged the survival of mouse model of ALS [34]. Given these results, Glu concentration was expected to be elevated in the brain of patients with ALS and to contribute to the pathophysiology underlying excitotoxicity. The increased ratio of Glu/NAA might imply that glutamatergic neurons were less vulnerable to excitotoxicity than other types of neurons in ALS if Glu and NAA indicated the number of glutamatergic neurons and all types of neurons, respectively [19]. Alternatively, the extracellular concentration of Glu might be increased due to dysfunction of glial glutamate transporter [35].
A reduced concentration of GABA in the M1 was repeatedly reported by one group [16,17,18]. GABA concentration in the SMA tended to be decreased when compared with controls, although it did not reach the statistical significance. However, the Glu/GABA ratio was significantly increased, which suggested a reduction in GABA concentration in the SMA. More number of subjects are needed to determine whether the GABA concentration is decreased or not in patients with ALS. The results of FA and ReHo showed the same tendency described for GABA concentration.
Of note, the VOI in the SMA reduced the frequency of measurement, given that one scan provides the concentration in both sides of the brain. Considering that the frontotemporal lobar degeneration spectrum includes ALS [22], it is pertinent to generate the SMA VOI for detection of abnormalities in ALS. This was supported by several studies using neuroimaging [23, 36,37,38,39]. Decreased FA [23, 36, 38], hyperactivity within a sensori-motor network [37], and cortical thinning was reported in the SMA [39]. Furthermore, microglial activation was reported in the SMA as well as in the M1 [40]. Collectively, the method used here is thought to be biologically and technically plausible for future clinical practice and research.
The limitations of this study will now be highlighted. A moderate number of subjects could lead to a lack of sufficient statistical power. As previously discussed [19], the source of Glu concentration was not distinguished by MRS [19]. We speculated that the Glu concentration measured by MRS was associated mainly with the intracellular fluid, given that the intracellular Glu concentration was at least three-orders-of-magnitude higher than that in the extracellular compartment [20, 41]. The time-dependent change was ignored because of the cross-sectional design of the study. Between-group difference in metabolites, including Glu and GABA, could depend on disease stage. A longitudinal study is required to reveal the dynamics of the levels of metabolites, including Glu and GABA.
In conclusion, the ratios of both Glu/NAA and Glu/GABA in the SMA exhibited low sensitivity but high specificity to distinguish ALS through a single scan. Finally, the increased level of Glu per NAA in patients with ALS might indicate that Glu neurons are resistant to Glu-related excitotoxicity relative to other types of neurons. Further work is needed to confirm the presented findings.
References
Riku Y, Atsuta N, Yoshida M, Tatsumi S, Iwasaki Y, Mimuro M, Watanabe H, Ito M, Senda J, Nakamura R, Koike H, Sobue G (2014) Differential motor neuron involvement in progressive muscular atrophy: a comparative study with amyotrophic lateral sclerosis. BMJ Open 4(5):e005213
Novotny EJ Jr, Fulbright RK, Pearl PL, Gibson KM, Rothman DL (2003) Magnetic resonance spectroscopy of neurotransmitters in human brain. Ann Neurol 54(Suppl 6):S25–S31
Kiernan MC, Vucic S, Cheah BC, Turner MR, Eisen A, Hardiman O, Burrell JR, Zoing MC (2011) Amyotrophic lateral sclerosis. Lancet 377(9769):942–955
Bradley WG, Bowen BC, Pattany PM, Rotta F (1999) 1H-magnetic resonance spectroscopy in amyotrophic lateral sclerosis. J Neurol Sci 169(1-2):84–86
Pioro EP, Majors AW, Mitsumoto H, Nelson DR, Ng TC (1999) 1H-MRS evidence of neurodegeneration and excess glutamate + glutamine in ALS medulla. Neurology 53(1):71–79
Bowen BC, Pattany PM, Bradley WG, Murdoch JB, Rotta F, Younis AA, Duncan RC, Quencer RM (2000) MR imaging and localized proton spectroscopy of the precentral gyrus in amyotrophic lateral sclerosis. AJNR Am J Neuroradiol 21(4):647–658
Pohl C, Block W, Karitzky J, Traber F, Schmidt S, Grothe C et al (2001) Proton magnetic resonance spectroscopy of the motor cortex in 70 patients with amyotrophic lateral sclerosis. Arch Neurol 58(5):729–735
Schuff N, Rooney WD, Miller R, Gelinas DF, Amend DL, Maudsley AA, Weiner MW (2001) Reanalysis of multislice (1)H MRSI in amyotrophic lateral sclerosis. Magn Reson Med 45(3):513–516
Rule RR, Suhy J, Schuff N, Gelinas DF, Miller RG, Weiner MW (2004) Reduced NAA in motor and non-motor brain regions in amyotrophic lateral sclerosis: a cross-sectional and longitudinal study. Amyotroph Lateral Scler Other Motor Neuron Disord 5(3):141–149
Wang S, Poptani H, Woo JH, Desiderio LM, Elman LB, McCluskey LF et al (2006) Amyotrophic lateral sclerosis: diffusion-tensor and chemical shift MR imaging at 3.0 T. Radiology 239(3):831–838
Mitsumoto H, Ulug AM, Pullman SL, Gooch CL, Chan S, Tang MX et al (2007) Quantitative objective markers for upper and lower motor neuron dysfunction in ALS. Neurology 68(17):1402–1410
Unrath A, Ludolph AC, Kassubek J (2007) Brain metabolites in definite amyotrophic lateral sclerosis. A longitudinal proton magnetic resonance spectroscopy study. J Neurol 254(8):1099–1106
Sivak S, Bittsansky M, Kurca E, Turcanova-Koprusakova M, Grofik M, Nosal V et al (2010) Proton magnetic resonance spectroscopy in patients with early stages of amyotrophic lateral sclerosis. Neuroradiology 52(12):1079–1085
Verma G, Woo JH, Chawla S, Wang S, Sheriff S, Elman LB, McCluskey LF, Grossman M, Melhem ER, Maudsley AA, Poptani H (2013) Whole-brain analysis of amyotrophic lateral sclerosis by using echo-planar spectroscopic imaging. Radiology 267(3):851–857
Han J, Ma L (2010) Study of the features of proton MR spectroscopy ((1)H-MRS) on amyotrophic lateral sclerosis. J Magn Reson Imaging 31(2):305–308
Foerster BR, Callaghan BC, Petrou M, Edden RA, Chenevert TL, Feldman EL (2012) Decreased motor cortex gamma-aminobutyric acid in amyotrophic lateral sclerosis. Neurology 78(20):1596–1600
Foerster BR, Pomper MG, Callaghan BC, Petrou M, Edden RA, Mohamed MA et al (2013) An imbalance between excitatory and inhibitory neurotransmitters in amyotrophic lateral sclerosis revealed by use of 3-T proton magnetic resonance spectroscopy. JAMA Neurol 70(8):1009–1016
Foerster BR, Carlos RC, Dwamena BA, Callaghan BC, Petrou M, Edden RA et al (2014) Multimodal MRI as a diagnostic biomarker for amyotrophic lateral sclerosis. Ann Clin Transl Neurol 1(2):107–114
Sako W, Abe T, Izumi Y, Harada M, Kaji R (2016) The ratio of N-acetyl aspartate to glutamate correlates with disease duration of amyotrophic lateral sclerosis. J Clin Neurosci 27:110–113
Atassi N, Xu M, Triantafyllou C, Keil B, Lawson R, Cernasov P, Ratti E, Long CJ, Paganoni S, Murphy A, Salibi N, Seethamraju R, Rosen B, Ratai EM (2017) Ultra high-field (7tesla) magnetic resonance spectroscopy in amyotrophic lateral sclerosis. PLoS One 12(5):e0177680
Cheong I, Marjanska M, Deelchand DK, Eberly LE, Walk D, Oz G (2017) Ultra-high field proton MR spectroscopy in early-stage amyotrophic lateral sclerosis. Neurochem Res 42(6):1833–1844
Seelaar H, Rohrer JD, Pijnenburg YA, Fox NC, van Swieten JC (2011) Clinical, genetic and pathological heterogeneity of frontotemporal dementia: a review. J Neurol Neurosurg Psychiatry 82(5):476–486
Sako W, Abe T, Izumi Y, Harada M, Kaji R (2016) Fractional anisotropy in the supplementary motor area correlates with disease duration and severity of amyotrophic lateral sclerosis. Neurol Sci 37(4):573–577
Tzourio-Mazoyer N, Landeau B, Papathanassiou D, Crivello F, Etard O, Delcroix N, Mazoyer B, Joliot M (2002) Automated anatomical labeling of activations in SPM using a macroscopic anatomical parcellation of the MNI MRI single-subject brain. Neuroimage 15(1):273–289
Sako W, Fujita K, Vo A, Rucker JC, Rizzo JR, Niethammer M, Carbon M, Bressman SB, Uluğ AM, Eidelberg D (2015) The visual perception of natural motion: abnormal task-related neural activity in DYT1 dystonia. Brain 138(Pt 12):3598–3609
Sako W, Abe T, Izumi Y, Yamazaki H, Matsui N, Harada M, Kaji R (2017) Spontaneous brain activity in the sensorimotor cortex in amyotrophic lateral sclerosis can be negatively regulated by corticospinal fiber integrity. Neurol Sci 38(5):755–760
Jenkinson M, Bannister P, Brady M, Smith S (2002) Improved optimization for the robust and accurate linear registration and motion correction of brain images. Neuroimage 17(2):825–841
Biswal B, Yetkin FZ, Haughton VM, Hyde JS (1995) Functional connectivity in the motor cortex of resting human brain using echo-planar MRI. Magn Reson Med 34(4):537–541
Lowe MJ, Mock BJ, Sorenson JA (1998) Functional connectivity in single and multislice echoplanar imaging using resting-state fluctuations. Neuroimage 7(2):119–132
Zang Y, Jiang T, Lu Y, He Y, Tian L (2004) Regional homogeneity approach to fMRI data analysis. Neuroimage 22(1):394–400
Block W, Karitzky J, Traber F, Pohl C, Keller E, Mundegar RR et al (1998) Proton magnetic resonance spectroscopy of the primary motor cortex in patients with motor neuron disease: subgroup analysis and follow-up measurements. Arch Neurol 55(7):931–936
Cheong I, Deelchand DK, Eberly LE, Marjańska M, Manousakis G, Guliani G, Walk D, Öz G (2019) Neurochemical correlates of functional decline in amyotrophic lateral sclerosis. J Neurol Neurosurg Psychiatry 90(3):294–301
Fiszman ML, Ricart KC, Latini A, Rodriguez G, Sica RE (2010) In vitro neurotoxic properties and excitatory aminoacids concentration in the cerebrospinal fluid of amyotrophic lateral sclerosis patients. Relationship with the degree of certainty of disease diagnoses. Acta Neurol Scand 121(2):120–126
Milanese M, Giribaldi F, Melone M, Bonifacino T, Musante I, Carminati E, Rossi PIA, Vergani L, Voci A, Conti F, Puliti A, Bonanno G (2014) Knocking down metabotropic glutamate receptor 1 improves survival and disease progression in the SOD1(G93A) mouse model of amyotrophic lateral sclerosis. Neurobiol Dis 64:48–59
Rothstein JD, Martin LJ, Kuncl RW (1992) Decreased glutamate transport by the brain and spinal cord in amyotrophic lateral sclerosis. N Engl J Med 326(22):1464–1468
Sage CA, Peeters RR, Gorner A, Robberecht W, Sunaert S (2007) Quantitative diffusion tensor imaging in amyotrophic lateral sclerosis. Neuroimage 34(2):486–499
Mohammadi B, Kollewe K, Samii A, Krampfl K, Dengler R, Munte TF (2009) Changes of resting state brain networks in amyotrophic lateral sclerosis. Exp Neurol 217(1):147–153
Keil C, Prell T, Peschel T, Hartung V, Dengler R, Grosskreutz J (2012) Longitudinal diffusion tensor imaging in amyotrophic lateral sclerosis. BMC Neurosci 13:141
Zhang J, Yin X, Zhao L, Evans AC, Song L, Xie B, Li H, Luo C, Wang J (2014) Regional alterations in cortical thickness and white matter integrity in amyotrophic lateral sclerosis. J Neurol 261(2):412–421
Corcia P, Tauber C, Vercoullie J, Arlicot N, Prunier C, Praline J, Nicolas G, Venel Y, Hommet C, Baulieu JL, Cottier JP, Roussel C, Kassiou M, Guilloteau D, Ribeiro MJ (2012) Molecular imaging of microglial activation in amyotrophic lateral sclerosis. PLoS One 7(12):e52941
Danbolt NC (2001) Glutamate uptake. Prog Neurobiol 65(1):1–105
Acknowledgements
We would like to thank all the subjects for joining this study.
Funding
This study was supported by the Uehara Memorial Foundation, Japan Society for the Promotion of Science (JSPS) KAKENHI Grant Number 20K12670, and Grants-in Aid from the Research Committee of CNS Degenerative Diseases, Research on Policy Planning and Evaluation for Rare and Intractable Diseases, Health, Labour and Welfare Sciences Research Grants, the Ministry of Health, Labour and Welfare, Japan.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval and informed consent statement
All subjects provided written informed consent, following detailed explanation of the procedures, and the study was approved by the local ethics committee of Tokushima University Hospital. This study was performed according to the Declaration of Helsinki.
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Supplementary Figure 1.
Differences in FA and ReHo, and results of correlation analyses. There were no significant differences in FA and ReHo between groups (A). ALS, amyotrophic lateral sclerosis; Ctr, control; FA value was positively correlated with ReHo. FA, fractional anisotropy; ReHo, regional homogeneity. (PNG 90 kb)
Rights and permissions
About this article
Cite this article
Sako, W., Izumi, Y., Abe, T. et al. MR spectroscopy and imaging-derived measurements in the supplementary motor area for biomarkers of amyotrophic lateral sclerosis. Neurol Sci 42, 4257–4263 (2021). https://doi.org/10.1007/s10072-021-05107-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-021-05107-3