Abstract
Amyotrophic lateral sclerosis (ALS) is progressive and fatal neurodegenerative disorder with upper and lower motor neuron signs. There are no biomarkers to track disease progression. To address this issue, we investigated regions in which fractional anisotropy (FA) values derived from diffusion weighted images correlated with both disease severity and duration in ALS patients. Fourteen patients with ALS were enrolled in this study. Voxel-based analysis revealed volume of interests (VOIs) showing significant correlation. Finally, Spearman rank correlation coefficient was assessed between FA value in each VOI and disease severity or duration. In the VOI of left supplementary motor area (SMA), FA value significantly correlated with disease severity and duration both (disease severity, rho = 0.59, p = 0.025; disease duration, rho = −0.69, p = 0.006). The present finding suggested the possibility that the abnormality in motor-related region including SMA could be a candidate for a biomarker to track disease progression.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Amyotrophic lateral sclerosis (ALS) is progressive and fatal neurodegenerative disorder with TAR-DNA binding protein-43 (TDP-43) inclusion bodies in neurons, which affects upper and lower motor neurons [1]. The revised El Escorial criteria has been accepted as a tool for a clinical diagnosis, and required upper and lower motor neuron sign [2]. However, quite a few patients pathologically diagnosed as ALS showed only lower motor neuron sign during the whole course of disease [3]. Furthermore, there are no biomarkers to track disease progression. To directly address these issues, state-of-the-art magnetic resonance imaging (MRI) techniques have been applied for diagnosis of ALS and assessment of disease progression, such as diffusion weighted images (DWI). The fractional anisotropy (FA) is derived from DWI, and is an index of the degree of directionality of water diffusivity. A voxel-based meta-analysis revealed the significant FA reduction in corticospinal tract in ALS patients [4]. FA values in the widespread brain region including corticospinal tract and frontal lobe significantly correlated with disease severity [5–7]. We hypothesized that the region showing FA correlation with both disease severity and duration could be a more useful biomarker to track disease progression because these two factors are derived from two different sources (i.e. physical examination or interview). To test this hypothesis, we investigated where FA values correlated with both disease severity and duration in ALS patients.
Materials and methods
Subjects
Fourteen patients with ALS [9 men and 5 women, age 64.5 ± 9.2 (mean ± SD)] were diagnosed according to the revised El Escorial criteria for possible, probable-laboratory-supported, probable or definite ALS, and enrolled in this study. Clinical characteristics of these subjects are summarized in Table 1. Informed consent was obtained from all participants under protocols approved by the local ethical committee.
MRI acquisition and analysis
The present study was performed using a 3.0 T MRI (GE, Milwaukee, WI, USA) with a standard head coil. DWI were acquired with 6 diffusion gradient directions using echo-planar single-shot spin-echo sequence. Scan parameters were as follows: b value, 1000 s/mm2; FOV, 240 mm; matrix, 128 × 128; TR, 9500 ms; TE, 82.3 ms; flip angle 90°; slice thickness, 4.0 mm. In-plane resolution of reconstructed images were 1.9 × 1.9 mm, respectively.
Images were corrected for Eddy current-induced distortions and head motion using the Eddy current correction routine in FSL. Non-brain tissue was removed from the images using the brain extraction tool in the FMRIB software library. After brain extraction, diffusion tensor components for each brain pixel were calculated, and FA maps were determined for each subject using the DTIFIT routine in FSL to fit the diffusion tensor model. B0 maps were registered to the Montreal Neurological Institute (MNI-152: 2 × 2 × 2 mm3) template using a 12-parameter affine transformation (FLIRT) and the resulting transformation was then applied to the FA maps to register them to MNI space. These images were smoothed using 8 × 8 × 8 mm3 kernel [8]. SPM5 (http://www.fil.ion.ucl.ac.uk/spm/software/spm5/) were used to identify the regions showing the significant correlation. The regions were considered significant for both a voxel-level threshold of p < 0.001 and cluster-level threshold of p < 0.05. The sphere-shaped volumes of interest (VOI) were generated with a radius of 5 mm and center at the highest t value voxel in each region detected by SPM analysis using WFU Pickatlas [9]. Finally, Spearman rank correlation coefficient was assessed between FA value in each VOI and ALSFRS-R or disease duration with IBM SPSS Statistics version 21(IBM Corp., Armonk, NY, USA).
Results
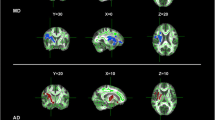
Significant negative correlation was found between FA value and disease duration in left and middle supplementary motor area (SMA; Table 2; Fig. 1a, b). There were no regions which showed significant positive correlation (Table 2). In contrast, significant positive correlation was found between FA value and ALSFRS-R in right cerebellum (IX, X), right posterior medulla, left anterior medulla including pyramid and left middle cerebellar peduncle (Table 3). No regions were found which showed significant negative correlation between FA value and ALSFRS-R (Table 3).
Fractional anisotropy (FA) value in volume of interest (VOI) in the supplementary motor area (SMA) correlated with disease severity and duration. a, b The VOI in the left SMA in the axial (a) and sagittal view (b). The sphere-shaped VOI was identified as the region where FA value correlated with a revised amyotrophic lateral sclerosis functional rating scale (ALSFRS-R) and disease duration both. c, d Rank correlations between FA value and ALSFRS-R (c), or disease duration (d) in the left SMA VOI. Significant correlations were observed, respectively (rho = −0.59, p = 0.025; rho = −0.69, p = 0.006). The vertical axis depicted FA value, and the horizontal axis depicted ALSFRS-R (c) or disease duration (d)
Furthermore, we examined whether FA value correlated with disease duration and ALSFRS-R both in each VOI. In the left SMA VOI, FA value significantly correlated with disease duration and ALSFRS-R both (disease duration, rho = −0.69, p = 0.006; ALSFRS-R, rho = 0.59, p = 0.025; Fig. 1c, d).
Discussion
We identified the region in which FA value correlated with disease duration and ALSFRS-R both in the left SMA. The present finding suggested the possibility that the abnormality in motor-related region could reflect disease progression. Several lines of evidence have supported the notion that the abnormality in the SMA is involved in ALS pathology. ALS patients showed decreased activity in the SMA in the sensorimotor network identified in resting state functional MRI compared to healthy volunteers [10]. Furthermore, cortical thinning was reported in the right SMA in ALS [11]. These results were not surprising because the key protein of ALS, TDP-43, was common to frontotemporal lobar degeneration (FTLD)-TDP and ALS was considered as a same spectrum disorders of FTLD-TDP [12]. It is possible that the potential abnormality of frontal lobe was detected in our study.
Keil and colleagues reported that FA value in the right SMA correlated not with ALSFRS-R but with disease duration [13]. The other study found significant correlation between FA value and ALSFRS in the prefrontal cortex including SMA [6]. Their results, based on only a voxel-level analysis, supported our finding, however, each study found separate correlation (i.e. significant correlation between FA value and disease duration or disease severity). In contrast, less-biased VOI centered at the highest t value voxel allowed for identification of the region which reflected disease duration and severity both, even though the VOI was derived from a voxel-based analysis. Detailed condition was different among studies, such as ALS patients (i.e. we included “possible” “laboratory-supported” “probable” and “definite” patients), disease duration, severity, MRI machine and analysis software. Therefore, other studies detected different regions which presented significant association between FA value and disease duration or severity [6, 7, 13, 14].
In terms of a biomarker, cerebrospinal fluid (CSF) is another candidate [15]. As suggested previously [16], the integration of multimodal methods including MRI and CSF was expected to allow for development of more reliable biomarkers, which could be adjusted for clinical information and genetic data.
Our results raised further question on how low FA value in the SMA was caused and contributed to pathophysiology of ALS. Given glutamate-induced excitotoxicity [1], we speculated that neurons in the SMA were damaged by excitatory projection, and reduced integration of fibers originated from SMA. Further studies are required to determine whether FA value is related to glutamate concentration in the SMA.
References
Kiernan MC, Vucic S, Cheah BC, Turner MR, Eisen A, Hardiman O, Burrell JR, Zoing MC (2011) Amyotrophic lateral sclerosis. Lancet (London, England) 377(9769):942–955. doi:10.1016/s0140-6736(10)61156-7
Brooks BR, Miller RG, Swash M, Munsat TL (2000) El Escorial revisited: revised criteria for the diagnosis of amyotrophic lateral sclerosis. Amyotroph Lateral Scler Other Motor Neuron Disord 1(5):293–299
Riku Y, Atsuta N, Yoshida M, Tatsumi S, Iwasaki Y, Mimuro M, Watanabe H, Ito M, Senda J, Nakamura R, Koike H, Sobue G (2014) Differential motor neuron involvement in progressive muscular atrophy: a comparative study with amyotrophic lateral sclerosis. BMJ Open 4(5):e005213. doi:10.1136/bmjopen-2014-005213
Li J, Pan P, Song W, Huang R, Chen K, Shang H (2012) A meta-analysis of diffusion tensor imaging studies in amyotrophic lateral sclerosis. Neurobiol Aging 33(8):1833–1838. doi:10.1016/j.neurobiolaging.2011.04.007
Agosta F, Pagani E, Petrolini M, Sormani MP, Caputo D, Perini M, Prelle A, Salvi F, Filippi M (2010) MRI predictors of long-term evolution in amyotrophic lateral sclerosis. Eur J Neurosci 32(9):1490–1496. doi:10.1111/j.1460-9568.2010.07445.x
Sage CA, Peeters RR, Gorner A, Robberecht W, Sunaert S (2007) Quantitative diffusion tensor imaging in amyotrophic lateral sclerosis. NeuroImage 34(2):486–499. doi:10.1016/j.neuroimage.2006.09.025
Thivard L, Pradat PF, Lehericy S, Lacomblez L, Dormont D, Chiras J, Benali H, Meininger V (2007) Diffusion tensor imaging and voxel based morphometry study in amyotrophic lateral sclerosis: relationships with motor disability. J Neurol Neurosurg Psychiatry 78(8):889–892. doi:10.1136/jnnp.2006.101758
Jenkinson M, Bannister P, Brady M, Smith S (2002) Improved optimization for the robust and accurate linear registration and motion correction of brain images. NeuroImage 17(2):825–841
Maldjian JA, Laurienti PJ, Kraft RA, Burdette JH (2003) An automated method for neuroanatomic and cytoarchitectonic atlas-based interrogation of fMRI data sets. NeuroImage 19(3):1233–1239
Mohammadi B, Kollewe K, Samii A, Krampfl K, Dengler R, Munte TF (2009) Changes of resting state brain networks in amyotrophic lateral sclerosis. Exp Neurol 217(1):147–153. doi:10.1016/j.expneurol.2009.01.025
Zhang J, Yin X, Zhao L, Evans AC, Song L, Xie B, Li H, Luo C, Wang J (2014) Regional alterations in cortical thickness and white matter integrity in amyotrophic lateral sclerosis. J Neurol 261(2):412–421. doi:10.1007/s00415-013-7215-5
Seelaar H, Rohrer JD, Pijnenburg YA, Fox NC, van Swieten JC (2011) Clinical, genetic and pathological heterogeneity of frontotemporal dementia: a review. J Neurol Neurosurg Psychiatry 82(5):476–486. doi:10.1136/jnnp.2010.212225
Keil C, Prell T, Peschel T, Hartung V, Dengler R, Grosskreutz J (2012) Longitudinal diffusion tensor imaging in amyotrophic lateral sclerosis. BMC Neurosci 13:141. doi:10.1186/1471-2202-13-141
Ellis CM, Simmons A, Jones DK, Bland J, Dawson JM, Horsfield MA, Williams SC, Leigh PN (1999) Diffusion tensor MRI assesses corticospinal tract damage in ALS. Neurology 53(5):1051–1058
Sako W, Ishimoto S (2014) Can cystatin C in cerebrospinal fluid be a biomarker for amyotrophic lateral sclerosis? A lesson from previous studies. Neurol Clin Neurosci 2(3):72–75. doi:10.1111/ncn3.82
Sako W, Murakami N, Izumi Y, Kaji R (2015) Neurofilament light chain level in cerebrospinal fluid can differentiate Parkinson’s disease from atypical parkinsonism: evidence from a meta-analysis. J Neurol Sci 352(1–2):84–87. doi:10.1016/j.jns.2015.03.041
Acknowledgments
This work was supported by Japan Society for the Promotion of Science (JSPS) KAKENHI Grant Numbers 15K19487.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
WS has received compensation for lecture from Novartis Pharma, Otsuka Pharmaceutical and Kyowa Hakko Kirin. The other authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Sako, W., Abe, T., Izumi, Y. et al. Fractional anisotropy in the supplementary motor area correlates with disease duration and severity of amyotrophic lateral sclerosis. Neurol Sci 37, 573–577 (2016). https://doi.org/10.1007/s10072-016-2487-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-016-2487-z