Abstract
Executive function (EF) allows for self-regulation of behavior including maintaining focus in the face of distraction, inhibiting behavior that is suboptimal or inappropriate in a given context, and updating the contents of working memory. While EF has been studied extensively in humans, it has only recently become a topic of research in the domestic dog. In this paper, I argue for increased study of dog EF by explaining how it might influence the owner–dog bond, human safety, and dog welfare, as well as reviewing the current literature dedicated to EF in dogs. In “EF and its Application to “Man’s Best Friend” section, I briefly describe EF and how it is relevant to dog behavior. In “Previous investigations into EF in dogs” section, I provide a review of the literature pertaining to EF in dogs, specifically tasks used to assess abilities like inhibitory control, cognitive flexibility, and working memory capacity. In “Insights and limitations of previous studies” section, I consider limitations of existing studies that must be addressed in future research. Finally, in “Future directions” section, I propose future directions for meaningful research on EF in dogs.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
In modern society, domestic dogs (Canis lupus familiaris) serve not only as companions, but also work alongside humans in sectors including law enforcement, search and rescue, scent detection, service to the disabled, and therapy. Given their pervasive role in human society, it is critical that dogs behave in a context-appropriate manner. For example, dogs are generally expected to refrain from jumping up on visitors and destroying furniture at home and must follow their owners’ commands to “stay,” “come,” or “leave it,” even in distracting environments such as dog parks or family gatherings. Meeting these and other expectations of their human caregivers relies in part on executive function.
Executive function (EF) is an umbrella term referring to a constellation of abilities that allow for context-appropriate responding and adapting to changes in our environment. It encompasses behaviors such as focusing on a goal in the face of distraction (i.e., selective attention), inhibiting behavior that is inappropriate for a given situation (i.e., inhibitory control), holding information in an active state (i.e., working memory capacity), and adapting behavior to accommodate changes in the environment (i.e., cognitive flexibility). In humans, EF deficits lead to a host of negative outcomes, including trouble interacting with peers, financial trouble, risky behavior, and substance abuse (Moffitt et al. 2011, 2015; Nigg 2001). Dire consequences exist for dogs as well; dogs can be fulfilling companions and diligent workers, but are also relatively large carnivores that can bite, maul, and cause destruction. Behaviors such as aggression, destructiveness, and disobedience often lead to relinquishment to an animal shelter and, in some cases, euthanasia (Salman et al. 2000).
The quality of dog welfare, human safety, and even dog-specific legislation relies on a body of rigorously conducted research on how dogs inhibit natural (but often suboptimal) urges and attend to humans amid distraction. Thus, the purpose of this paper is to illustrate EF’s relevance to dogs and encourage further, more rigorous inquiry into this topic. To achieve these ends, I have divided this paper into four sections. In “EF and its Application to “Man’s Best Friend”” section, I briefly define EF and describe abilities that fall under this term, including selective attention, inhibitory control, working memory capacity (WMC), and cognitive flexibility. I also explain how EF applies to dogs, with an emphasis on how increased knowledge of canine EF can benefit both dogs and humans. In “Previous investigations into EF in dogs” section, I provide a review of behavioral methods used by canine researchers to measure EF, and how they have conceptualized presumed causal factors like genetics and ontogeny. In “Insights and limitations of previous studies” section, I discuss limitations of the current body of research that must be addressed going forward. Although several studies have addressed EF in dogs, many contain methodological issues that limit the interpretation and applicability of their findings. Such issues, addressed in greater detail later in this paper, include pervasive use of small samples (Arden et al. 2016) and using task batteries without first assessing the reliability of each task (Bray et al. 2014; MacLean et al. 2014; Fagnani et al. 2016; Marshall-Pescini et al. 2015; Müller et al. 2016). These issues are not insurmountable, but must be remedied if we are to have an accurate understanding of how dogs regulate (or fail to regulate) their behavior. The paper concludes in “Future directions” section with suggested directions for future research from which we could learn how self-regulatory processes in dogs compare to ours and lay a theoretical foundation for better training and breeding practices, evidence-based legislation, and improved dog welfare and human safety.
EF and its application to “Man’s Best Friend”
To ensure their day-to-day survival, animals must attend to a variety of tasks including food acquisition, avoidance of danger, and the forging and maintenance of social ties. Some of these tasks become well practiced and can be carried out automatically; others require one’s full attention and, sometimes, behavioral adaptation to accommodate changes in the environment. Success in these latter tasks relies on EF, a term referring to processes that allow an animal to coordinate behavior in a manner appropriate for a given context, especially in the face of distraction. Given the complexity of EF, a full review is beyond the scope of this paper. For the sake of brevity, I constrain this section to briefly explaining how EF guides behavior, naming abilities encompassed by EF as proposed by researchers, and considering some of the consequences of one’s level of EF. The remainder of “EF and its Application to ‘Man’s Best Friend’” section is dedicated to how EF is applicable to dog behavior and how further study has the potential to benefit both dogs and humans.
Executive function: a brief primer on effortful self-regulation
A defining feature of EF is its reliance on the effortful allocation of attention, as opposed to mental processes that operate automatically. This distinction between such “controlled” executive and automatic behavior has existed since William James (1890) distinguished between “ideomotor acts,” which follow more or less immediately after conception in the mind, and “willed acts” which require an added element of volition. Many researchers have since expanded upon this automatic–executive distinction (for further reading, please see Baddeley and Hitch 1974; Norman and Shallice 1986; Schneider and Shiffrin 1977).
Depending on the demands of the environment, either automatic actions or those requiring EF can be adequate. In some cases, the ability to complete tasks automatically is beneficial; it enables an individual to behave in ways that have been effective in the past without requiring sustained attention that could be better used on other tasks (Norman and Shallice 1986). However, because many animals live in environments that are constantly changing, automatic responding can be erroneous and even dangerous. When troubleshooting, navigating a difficult or dangerous situation, and/or overcoming an automatic response are required, engagement of EF is preferred to automaticity. In these cases, one must be consciously aware of environmental conditions and be able to respond appropriately, even if this means abandoning an automatic response that had hitherto been effective (Miller and Cohen 2001). Classic models have illustrated how automatic responding is overridden in favor of a new, more appropriate behavior. For example, Norman and Shallice (1986) proposed that an executive system (known as the Supervisory Attentional System, or SAS) biases the activation of the less practiced behavior to increase the likelihood it will be expressed in place of the automatic response elicited by the environment. Further, this new behavioral sequence must be internally maintained, while it is being performed (Schneider and Shiffrin 1977), an act that requires attentional resources and generally prevents the individual from performing other behaviors at the same time.
Researchers have proposed that successfully overriding automatic responses and maintaining behavioral sequences relies on the prefrontal cortex (PFC), based on research showing that damage to this part of the brain causes deficits in behavioral flexibility and sustained attention while leaving well-learned, automatic actions intact (see Miller and Cohen 2001, for a review). Additionally, researchers have found that human and nonhuman subjects alike exhibit greater sustained activation in the PFC in tasks requiring the maintenance of information over a delay or in the face of distraction (De Pisapia and Braver 2006; Fuster and Alexander 1971; Kubota and Niki 1971; Miller et al. 1996). According to Miller and Cohen (2001), this activation is due to the PFC acting as a sort of “switchboard operator” to allow for effective behavior. Seating EF within a biological structure has made it more tractable to scientific study in human and nonhuman animals.
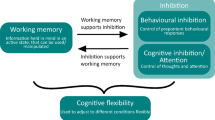
Rather than being a unitary construct, EF is a multifaceted phenomenon which draws from several distinct abilities to allow for successful responding. Across the literature, abilities proposed to be encompassed by EF include selective attention, cognitive flexibility, inhibitory control, and WMC, all of which rely heavily on the PFC (Jurado and Rosselli 2007; Miller and Cohen 2001). The reader should note that these abilities are not completely independent and, in many cases, interact.
Selective attention to stimuli
Selective attention refers to the ability to focus on one stimulus in the environment at the expense of others, achieved by an increased focus on the relevant stimulus and inhibition of signals from other, irrelevant stimuli (Miller and Cohen 2001). It is most strongly engaged when there are many salient stimuli in the environment, and reduces interference through attenuated sensitivity to distracting information (Conway et al. 2001; Hutchison 2011; Hutchison et al. 2016). Selective attention requires a certain degree of WMC; individuals with high WMC are better at focusing on relevant information and suppressing irrelevant information than individuals with low WMC (Conway and Engle 1994; Engle 2002).
Cognitive flexibility
The ability to flexibly attend and respond to the environment under changing conditions is critical in everyday life. Cognitive flexibility requires an individual to disengage attention from one task or set of rules to engage in another. Successful switching relies on preparation for the new task by mentally reconfiguring task rules, and inhibition of obsolete responses during the new task (Cepeda et al. 2001), both of which are made possible by the frontal cortex. Perseveration—the repeating of obsolete or incorrect responses—often accompanies frontal lobe damage (reviewed by Gazzaniga 2009) and age-related changes in the PFC and can be indicative of dementia risk (Berg 1948; Balota et al. 2010).
Working memory capacity
Although its name suggests the limits of memory, WMC is better described as how much information an individual can hold in an active, retrievable state in the face of distraction. Engle’s (2002) illustration of WMC consists of two distinct components: a short-term memory component that permits active maintenance of a small set of information, and an executive component that directs attention in a goal-directed manner. In fact, researchers have proposed that this executive component is the underlying factor responsible for the observed relationship between WMC and abilities such as reading comprehension, multitasking, SAT scores, and ability to multitask (Baddeley and Hitch 1974; Engle 2002; Miyake et al. 2000; Shipstead et al. 2014; Unsworth and Engle 2007).
Inhibitory control of behavior
Inhibitory control refers to one’s ability to withhold a prepotent response (one that is well practiced and has been rewarded consistently in the past). It requires top-down control to withhold or cease a behavior in favor of one that is novel, less practiced, and/or appropriate for the current situation. In humans, inhibitory control failure is related to engagement in dangerous or unhealthy behaviors (Duckworth et al. 2013), suboptimal cognitive aging (Balota et al. 2010), and dementia (Balota et al. 2010).
Implications of EF
The importance of EF can be best realized by observing consequences that follow from its presence or deficit. For example, individuals with attention-deficit hyperactivity disorder (ADHD) demonstrate marked deficits in inhibitory control, selective attention, and WMC. Compared to their typically functioning peers, they are at a greater risk of academic difficulties, having poor social relationships, aggression, experimentation with and abuse of drugs, and unemployment (Barkley 1997; Duckworth et al. 2013; Gardner and Gerdes 2015). In addition to difficulties experienced by the individual, these consequences carry a considerable cost for society in terms of public safety, productivity, and use of social programs like welfare.
In the general population, researchers have observed individual differences in EF. For example, people testing higher in various complex span tasks (referred to as “high spans” in the literature) are better able to respond appropriately to stimuli in a distracting environment than are low spans due to their ability to suppress the distracting information (Engle 2002). They are also better able to think creatively (Lee and Therriault 2013) and sustain attentional focus (Braver et al. 2007). Research with human participants has provided robust evidence that one’s capacity for EF affects problem-solving ability,Footnote 1 adapting to novel situations, and social relationships (Barkley 1997; Braver et al. 2007; Duckworth et al. 2013; Engle 2002; Gardner and Gerdes 2015; Lee and Therriault 2013; Mischel et al. 1989; Moffitt et al. 2011).
A case for increased study of dog EF
While the human EF literature is vast, we are only beginning to understand this construct in dogs. How dogs regulate (or fail to regulate) their own behavior is useful knowledge given the dog’s ubiquity in human society. Dogs often occupy households with children, other animals, and household items that can be soiled, torn, or eaten. To peacefully coexist with humans, dogs must be able to inhibit behaviors like eliminating in the house, playing too boisterously with children or other pets, and being destructive. Although training can help prevent these unwanted behaviors, a dog’s level of EF likely plays a large role in facilitating such training (Hasher and Zacks 1979) to allow it to overcome natural urges to obey its owner’s expectations for proper behavior. Conducting more, and better, research on canine EF, including the effects of genetics and ontogeny, has the potential to produce several tangible benefits to both humans and dogs, as I shall now discuss.
Modeling human EF
One benefit of a greater understanding of EF in dogs is, perhaps unexpectedly, a greater understanding of our own EF. In the field of comparative psychology, researchers have leveraged similarities between humans and other animals to study human psychology and its evolution.Footnote 2 Although humans and dogs are certainly different animals, researchers have noted many parallels between natural human and canid society: Both are communal creatures, living in cooperative family groups in which individuals hunt and move together, forge and maintain bonds, and rear young (Mech 2000; Schleidt and Shalter 2003). Selection pressures imposed by social living are believed to have shaped cognition in a variety of ways. For example, cooperative hunting requires that members of a group be able to coordinate maneuvers and inhibit chasing behavior until the right moment (Marshall-Pescini et al. 2015). They must also be able to inhibit impulsive, aggressive behavior for the sake of maintaining group cohesion. Additionally, information processing might become more complex as the social group increases in size. That is, individuals must be able to identify each member of the group and know where each member ranks relative to other members if there is a social hierarchy. Overall, one would expect that compared to solitary animals, animals living in social groups should be capable of more complex cognition and better self-control. Evidence for this notion includes the positive correlation between neocortex size and number of members of the social group in primates (Dunbar 1998), as well as the greater levels of inhibitory control exhibited by primates living in dynamic “fission–fusion” groups (Amici et al. 2008).
Dogs have been particularly useful models given their trainability, availability, and cooperative nature, especially in the area of cognitive aging. For example, researchers have found that, like humans, dogs experience declines in EF as they age (Cepeda et al. 2001; Mongillo et al. 2013; Tapp et al. 2003; Wallis et al. 2014, 2016). Researchers have also found canine analogs of attentional deficit hyperactivity disorder (ADHD; Vás et al. 2007) and Alzheimer’s disease (canine cognitive dysfunction; Azkona et al. 2009; Landsberg et al. 2003, 2012; Ruehl et al. 1995). Such research provides a starting point in developing methods of testing various cognitive abilities, as well as the efficacy of interventions, such as those aiming to reduce age-related cognitive decline (Cotman et al. 2002; Milgram 2003; Milgram et al. 2002, 2005). Increased knowledge of EF in dogs can show us which qualities (and associated brain regions) of this ability are shared between humans and dogs and which are distinct to each species, and inform the conclusions we can draw from research using dogs as models for human cognition and mental illness.
Improved owner–dog dynamics
As pointed out by Arden et al. (2016), most canine cognition studies are approached from an ethological angle. Ethology is the biological study of animal behavior, with a focus on how animals behave in their natural environment and how these behaviors contribute to their survival (Tinbergen 1963; Range and Virányi 2015). Many dog researchers are interested in understanding dog behavior in the context of the species’ shared environment with humans, both in the present-day and during the domestication process. Many contentions in the literature have arisen from different views regarding how genetics and ontogeny determine cognitive abilities of dogs (Dorey et al. 2009, 2010; Hare et al. 2002, 2009; Hare and Tomasello 2005; Merritt 2015; Virányi et al. 2008), and both of these factors likely influence EF in important ways.
Given the close relationship dogs share with humans, the dynamic between a dog and its owner is a crucial element of the dog’s development and how it responds to the environment. Clark and Chalmers (1998) argued that, although they are generally discussed as occurring internally, cognitive processes extend into our environment. For example, we often use physical aids in memory, self-regulation, and problem-solving including calculators, writing, and even the use of language. As such, we “extend” our mind into the environment. Regarding canine cognition, Merritt (2015) agrees with this notion and, further, discusses how dogs use us as physical aids, nearly always thinking “with” us. He further argues that researchers should approach the study of canine cognition with an appreciation for the “co-cognition” that takes place between humans and dogs. Researchers have found evidence for such co-cognition, showing how it has been influenced by domestication (Miklósi et al. 2003), breed (Udell et al. 2014), and training (Marshall-Pescini et al. 2009; Marshall-Pescini et al. 2016).
One of the primary ways humans influence their dogs’ behavior and cognition is through training. In terms of training techniques, positive punishment (e.g., striking a dog or rubbing its nose in its own excrement) can increase the likelihood of human-directed aggression (Casey et al. 2014), the end result of a failure to inhibit an inappropriate response. These findings mirror those found in human children subject to harsh parenting (Deater-Deckard et al. 2012). In the child psychology literature, researchers have shown that a mother’s level of EF shapes parenting practices that can ultimately help or hinder the child’s development of EF. In particular, mothers with low levels of EF were more likely to get frustrated and use harsh parenting practices than mothers with higher EF (Deater-Deckard et al. 2010, 2012).
Future research could investigate how owner EF influences training and discipline practices. Are owners with low EF more likely to endorse and use harsh training methods than owners with high EF? If so, does this predict lower EF or increased problem behaviors in their dogs? Interactions between owner and dog EF could be probed in more benign settings as well. For example, the sport of dog agility requires attentional control and flexible responding on the part of both canine and human participants. The owner must decide how to navigate a complex obstacle course while visually tracking their dog, and the dog must attend to the owner’s body language as well as the obstacles on the course. EF, particularly WMC and selective attention components, is likely involved in successful navigation of the course and subsequent scoring. Researchers could examine how much of a team’s performance is predicted by EF of the owner, EF of the dog, and the interaction between dog and owner EF. In short, the role human behavior plays in a dog’s ability to self-regulate and interact with its environment cannot be neglected in the midst of more intuitive factors like breed, training, and ontogeny.
Improved dog welfare
As a general rule, humans dictate most events in their dogs’ lives: Whether they receive training and what type of training they receive, how much physical and mental stimulation they receive, and whether they ultimately remain in the home. The amount and quality of attention owners pay their dogs can influence the development and occurrence of problem behaviors and thus increase likelihood of aggression, relinquishment, and euthanasia. In fact, one study (Salman et al. 2000) found that the top five reasons cited by owners for relinquishing their dogs to a shelter were biting, house soiling, aggression toward people, escaping, and being destructive indoors, all behaviors related to low levels of EF.
Learning more about how human behavior—including training philosophy, amount of owner–dog interaction, and previous experience with dogs—influences EF-related problem behaviors in dogs has the potential to reduce the number of dogs relinquished to shelters each year, as well as euthanasia rates of otherwise healthy dogs. Although many owners cite behavioral issues as the reason for relinquishing their dog to a shelter (Salman et al. 1998, 2010), these issues might be less reflective of a dog’s ability to behave and more reflective of their former owner’s inability to provide proper care, exercise, and training. Evidence of this notion comes from a study reporting that owners who relinquish their dogs are more likely to be young, first-time pet owners and have idealistic expectations for how much “work” the dog would require (Salman et al. 1998). Further, very few dogs who were relinquished had ever received obedience training. Unfortunately, dogs relinquished for behavioral reasons are less likely to be adopted than dogs relinquished due to cost or changing home dynamics (Lepper et al. 2002), and of the approximately 3.3 million dogs that are relinquished to shelters in the USA each year, about 670,000 of these dogs are euthanized (ASPCA, n.d.).
Increased human safety and evidence-based legislation
Legislation, local regulations, and even landlord-enacted breed restrictions would greatly benefit from improved knowledge of dog EF. With a better understanding of the factors underlying EF in dogs, we might be able to more accurately predict which dogs are at greater risk of responding to stress with aggression, allowing restrictions to be enacted on a case-by-case basis, rather than on the basis of size or breed, as is often the case. For example, researchers might find that low levels of EF are related to higher levels of aggression in dogs, as has been found to be the case in humans. Sprague et al. (2011) found that humans with low EF were more likely to respond to perceived stress with physical aggression. They concluded that EF, particularly inhibitory components, is important in managing inappropriate behavioral responses to stressors, as well as down-regulation of emotions that lead to such reactions. This supports the notion that behavioral inhibition might be an underlying factor uniting the various EF abilities (Barkley 1997). Further research might also show that problem behaviors owing to low EF are more influenced by owner characteristics and training than breed, reducing negative breed stereotypes and restrictions based on these stereotypes. If aggression or destructive tendencies are better predicted by EF than breed or size, restrictions should be modified accordingly. Further, if EF can be modified by training, this could provide a powerful intervention for dogs that might otherwise be relinquished or euthanized.
Summary
EF encompasses several distinct cognitive functions that promote flexible and context-appropriate responding. These functions include selective attention, cognitive flexibility, inhibitory control, and WMC, all of which rely, to some degree, on the frontal lobe. Researchers in the human domain have found that EF influences performance in academic contexts, finances, and social interactions, all of which are consequential to our survival. Canine cognition research suggests that EF is important for dogs as well, possibly implicated in behaviors like obedience, aggression, and suitability for working contexts. Given the potential impact of these behaviors on human safety and dog welfare, continued study of canine EF is needed.
Previous investigations into EF in dogs
EF has proven a challenging phenomenon to understand in our own species. It should not come as a surprise, therefore, that researchers have met with considerable difficulty in studying this construct in dogs, who do not share our linguistic abilities or perceptual world. Although the field of canine cognition has often focused on the dog’s understanding of human pointing and emotional expression, EF has received more attention in recent years. Hoping to better understand how genetics and ontogeny influence EF, researchers have employed a variety of cognitive tasks. Many of these tasks have been adapted from tasks used to study object permanence in human infants (e.g., A-not-B and cylinder task), whereas others were created specifically for use with dogs (e.g., social task and leash task). The purpose of this section is to review some of the behavioral tasks that have been used to study EF in dogs.
Cylinder task
The cylinder task is a modified form of detour-reaching task, in which subjects must suppress the tendency to reach directly for a visible food reward (hereafter referred to as a “treat”) in favor of approaching it via a learned “detour,” and traces its roots back to studies on object permanence in infants (Lockman 1984; Piaget 1954). It has since been modified to assess inhibitory control in a variety of nonhuman animals (see Amici et al. 2008; Boogert et al. 2011, Rørvang et al. 2015; Wallis et al. 2001), including dogs (Bray et al. 2014; Fagnani et al. 2016; MacLean et al. 2014; Marshall-Pescini et al. 2015). Using this task, researchers test a dog’s ability to successfully retrieve a treat from inside an open-ended cylindrical apparatus laid on its side. First, the dog is trained to retrieve the treat from the apparatus. During training, the dog is held in place approximately two meters from an opaque cylindrical apparatus and watches as a human experimenter places a treat into the cylinder. Once the cylinder has been baited, the dog is released and allowed to retrieve the treat. This protocol is repeated until the dog successfully retrieves the treat in four of five consecutive trials (Bray et al. 2014; Fagnani et al. 2016; MacLean et al. 2014; Marshall-Pescini et al. 2015), at which point the dog advances to the testing phase, which is identical to the training procedure with one exception: The cylinder is now transparent rather than opaque. To successfully retrieve the treat, the dog must inhibit the response of approaching the now visible reward directly (thus contacting the barrier) and instead approach it by using the detour technique learned during training. The dog’s EF is measured in terms of accuracy across ten trials.
To better understand how domestication might have affected EF, Marshall-Pescini et al. (2015) compared the performance of human-reared wolves on the cylinder task with that of trained and untrained pet dogs and human-reared “free-ranging” dogs. They found that dogs, regardless of training or housing situation, outperformed wolves and suggested that dogs might have used inadvertent human cues that led to their successful performance; however, there is conflicting evidence as to whether there exists a difference in the ability to follow human social cues between wolves and dogs (Hare and Tomasello 2005; Udell et al. 2008; Virányi et al. 2008). Further, researchers should use caution when comparing the performance of dogs to that of modern-day wolves when trying to understand the effects of domestication (see Kruska 1988).
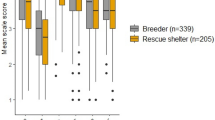
Researchers have also examined the influence of a dog’s environment. Because shelter dogs have previously shown decrements in socio-cognitive tasks in comparison with pet dogs (Udell et al. 2010a, b), Fagnani et al. (2016) reasoned that differences in lifetime experience with humans might influence a dog’s level of EF. Residing in a home requires more context-appropriate behavior to coexist peacefully with human owners. One would then expect that pet dogs would exhibit greater EF as indicated by better performance on the cylinder task in comparison to shelter dogs. However, Fagnani et al.’s data indicated no difference in performance between the two groups. Although it is possible that shelter dogs and pet dogs simply do not differ on this measure, two limitations of this study should be addressed before drawing such a conclusion. First, the researchers used a relatively small sample when collecting their data. As discussed by Arden et al. (2016), many canine cognition studies are too underpowered to detect existing effects. Replicating this study with a larger sample would provide more reliable data and, possibly, differential performance between shelter dogs and pet dogs. Second, accuracy is often remarkably high in the cylinder task (Fagnani et al. 2016; MacLean et al. 2014; Marshall-Pescini et al. 2015), even for dogs who did not partake in training (refer to Table 1), leading the researchers to suggest a possible ceiling effect. Indeed, Fagnani et al. suggested that, if the task was “too easy” for the dogs, existing differences between the two groups might have washed out.
Finally, Bray et al. (2014) found that age-related differences were reflected in cylinder task performance, but not the other two tasks in their battery. Age was negatively correlated with accuracy during test trials, such that older dogs were less accurate than younger dogs. This finding is consistent with previous observations that cognitive functioning, particularly EF, tends to decline with increasing age in humans (Cepeda et al. 2001; Craik and Bialystok 2006) and dogs (Wallis et al. 2014, 2016). For example, Wallis et al. (2014) found that, similar to humans, selective attention assumes a quadratic function over the lifespan in dogs, such that middle-aged dogs show the highest level of selective attention, while adolescent and older dogs show lower levels. The authors suggest that these age-related reductions in attention are due to deficits in EF. Older dogs have also been shown to exhibit impaired inhibitory control relative to young dogs (Mongillo et al. 2013; Lazarowski et al. 2014; Tapp et al. 2003) as well as patterns observed in humans, with inhibitory processes breaking down in the later years of life (Spieler et al. 1996).
Future studies using the cylinder task should include modification to make it more challenging. As has been shown in human research, individual differences are more apparent when the task is difficult than when it is easy. Researchers have imposed such difficulty by increasing the amount of information a participant must hold in memory (Conway and Engle 1994; Turner and Engle 1989), requiring the participant to engage in self-control before partaking in the task (Baumeister et al. 1998), and reducing external reminders of the task goal (Hutchison 2011; Kane and Engle 2003). Canine researchers could use similar strategies by requiring a dog to engage in self-control prior to completing the cylinder task (refer to the self-control task devised by Miller et al. 2010 discussed later in this section) or test the dog in a distracting environment, which could increase working memory load. As shown in Table 1, the ceiling effect observed in the previously described studies is absent in the study by Bray et al. (2014). Because dogs in this study performed another socio-cognitive task (the A-not-B) prior to the cylinder task, it is possible that this reduction in accuracy is due to fatigue.
It is also important to consider the relative novelty of the cylinder task in testing canine EF. Its first usage with dogs was by Bray et al. (2014) who tested thirty dogs on a battery of three tasks—the cylinder task, the social task, and the A-not-B task—presumed to reflect a dog’s level of EF. Thus far, there have been no demonstrations of its reliability. Before seeking correlations between tasks that presumably measure the same thing (as was done in all three studies discussed), a researcher must know that the tasks in the battery are reliable (Miyake et al. 2000). That the cylinder task has not been subject to this kind of testing limits its utility, particularly in task batteries.
Detour-fence task
The detour-fence task is another detour task in which a small section of fencing is used to separate a dog from a treat. The treat is placed on the side of the fence opposite to the dog, and the dog must go around the fence to obtain it. In other words, the dogs must first move away from the food to obtain it, requiring dogs to overcome the prepotent response of approaching the food head-on. EF has been measured in terms of latency to successfully retrieve the treat (Marshall-Pescini et al. 2015); however, this task might be more reflective of problem-solving ability than EF.
In their study, Marshall-Pescini et al. (2015) tested wolves and dogs on both the cylinder task and the detour-fence task. Although the tasks are thought to be conceptually similar, the researchers failed to find any correlation in performance between the two tasks. Additionally, they found that wolves outperformed dogs on the detour-fence task, while dogs outperformed wolves on the cylinder task. Did selection for cooperation with humans increase EF or decrease it? The former possibility would lead to better performance in both tasks by dogs, while the latter would lead to better performance by wolves. However, this line of thinking assumes that both tasks are measuring the same thing and that no other factors should significantly influence performance in either task. Given the lack of correlation in performance between the tasks and the evidence for species-level specialization, the data shed little light on the original research question. There is also a possibility that the detour task is more reliant on another ability, such as spatial problem-solving, than is the cylinder task. Indeed, numerous studies (Bray et al. 2015; Marshall-Pescini et al. 2016; Pongrácz et al. 2005; Smith and Litchfield 2010) have labeled the detour-fence task as a measure of problem-solving ability. Thus, researchers should consider omitting this task from studies examining EF.
Leash task
Used in only one study as part of a three-part task battery, the leash task is similar in nature to the cylinder and detour-fence tasks in that it requires dogs to first move away from a desired object in order to ultimately make contact with it. In this task, each dog was trained by being walked on a leash by its owner. During the walk, the dog encountered a situation in which the leash became caught on an object, such as a tree or post. The owner then called to the dog, at which point the dog was required to go around the object to return to its owner within 20 s (Müller et al. 2016). The distance the dog had to travel to untangle itself from the obstacle and reach its owner was gradually increased up to two meters. After the dog had been trained for on this task for approximately 1 month, the owner tested the dog, videotaping the footage to be scored later by the experimenter. Scores on the task ranged from 0 (poor performance) to 2 (good performance).
Ultimately, Müller and colleagues found significant, but weak, correlations between performance on the leash task and two other supposed EF tasks included in their battery. They suggested that this might imply context specificity of EF (similar to the argument presented by Bray et al. 2014 based on similar results). However, one could argue that, like the detour-fence task, the leash task requires a substantial degree of spatial problem-solving ability; dogs might simply not understand how to negotiate the obstacle presented by a tangled leash. This could have negatively impacted correlations between tasks, in addition to possible low reliability of this measure. As with the cylinder task, the leash task is a novel method of testing EF and has not been subject to tests of its reliability. Reliability must first be demonstrated before searching for evidence of validity (i.e., correlation with other supposedly EF tasks). Thus, that the leash task reflects EF should be treated cautiously until it has been subject to such scrutiny.
Delay discounting
Delay discounting refers to the devaluing of future rewards relative to immediate ones (Riemer et al. 2014). Generally, preference for a reward decreases as the delay to achieve such an award increases. Inability to delay gratification can lead to negative outcomes in humans including poor financial habits, obesity, and risky behavior (Duckworth et al. 2013; Moffitt et al. 2011) and is also related to various problem behaviors in dogs as reported by their owners (Wright et al. 2011).
The use of delay-discounting tasks to assess EF in dogs is more intensive in terms of time to train and test the dogs, as well as constructing the apparatus, than some of the other tasks discussed in this paper. In these tasks, dogs are allowed to choose between a small immediate reward and a larger, delayed reward. Using delay discounting as a measure of canine inhibitory control, Riemer et al. (2014) differentiated between cognitive impulsivity (the ability to delay gratification) and motor impulsivity (the ability to inhibit prepotent responses) and used a delay-discounting task to examine how stable these two abilities were over time. Dogs were first given ten forced-choice trials on two different apparatuses. One apparatus dispensed one treat after each press, and the other dispensed three treats after a 3-s delay. After learning the contingencies, dogs were given free access to both apparatuses for 15 min. During this time, the delay increased on the second apparatus by 1 s each time the dogs chose the delayed reward. The maximum delay reached by the dogs during the 15 min served as a measure of cognitive impulsivity and the number of extra presses between the first press and the delivery of the reward served as a measure of motor impulsivity. Dogs were tested again 6 years later in the same manner.
For the ten dogs that completed testing, the maximum delay achieved was stable over the 6-year period, whereas the number of extra presses increased. In other words, a dog’s ability to delay gratification seems more stable than the ability to inhibit prepotent responses, which declined over time. From this finding, Riemer et al. (2014) concluded that cognitive and motor aspects of EF, particularly inhibitory control, might be independent. The cognitive aspect might be more of a trait that remains stable over the lifetime, whereas the motor aspect might be more influenced by the current environment. Their results are consistent with the notion that inhibitory control, at least the inhibition of prepotent responses, is context specific (Bray et al. 2014; Fagnani et al. 2016). Additionally, the finding that inhibition of prepotent responses declined with age mirrors the results obtained in the cylinder task (Bray et al. 2014).
Self-control task
While not directly used to test EF, Miller et al. (2010) created a delay-type task to better understand the mechanisms responsible for ego depletion. Ego depletion refers to the observation that people tend to persist less in a difficult cognitive task after being required to exert self-control (Baumeister et al. 1998). The causes for this phenomenon have been hotly disputed (Baumeister 2014; Beurms and Miller 2016; Inzlicht and Schmeichel 2012). One theory is that both self-control and performance on difficult cognitive tasks are dependent upon a shared, limited resource. Miller and her team tested blood glucose level as a contender for this limited resource using a sample of dogs.
Dogs in the study were first assigned to one of two conditions: kennel or self-control. Those in the kennel condition were led into a kennel which was then closed by the owner. The owner left the room, and the dog remained in the kennel for 10 min before being released. For dogs in the self-control condition, owners instructed the dogs into a sit–stay position and then left the room such that they were able to view the dog by use of a mirror without being visible to the dog. If the dog broke the sit–stay, the owner walked back in, told the dog to sit and stay again, and then left the room. This continued until 10 min had passed. An experimenter recorded both the number of sit–stay “breaks” and at what intervals they occurred.
For their first experiment, Miller et al. (2010) found that, compared to dogs who had spent 10 min in a kennel, dogs who exerted self-control persisted less on an “impossible” task requiring them to retrieve a treat from a Tug-a-Jug toy (the opening to the toy was blocked such that the dog could see the treat inside but could not get it to fall out). However, in a follow-up 2 (condition: kennel/self-control) × 2 (drink: sugar-free/glucose) factorial design, the researchers found that consuming 2 oz. of a glucose drink erased the difference in persistence between dogs who had been in the kennel and dogs who had been in a sit–stay. For dogs who had consumed the sugar-free drink, exerting self-control still reduced persistence in the impossible task compared to dogs who had been in the kennel. The authors thus argued that both dogs and humans rely on glucose as a limited resource. Regardless of how ego depletion functions, engaging in sustained inhibition clearly reduced later persistence in a difficult task. The authors later extended these findings by showing that dogs who held the 10-min sit–stay were more likely to approach and maintain proximity to an aggressive caged dog, suggesting that engaging in self-control can also increase risky, impulsive behavior (Miller et al. 2012).
Although this self-control task was intended as a manipulation, it seems to reflect EF in its own right. Remaining in a seated position for 10 min is challenging for most dogs (Miller et al. 2010, 2012) and likely requires sufficient WMC to maintain the goal of obeying the “stay” command in the absence of the commanding human.Footnote 3
This task might be useful in assessing WMC, with number of “breaks” and time elapsed prior to the first “break” used as dependent measures (similar to the conceptualizations of motor impulsivity and cognitive impulsivity measured by Riemer et al. 2014). It could also be used to remedy ceiling effects reported in the cylinder task. Having dogs engage in self-control using this method prior to engaging in these tasks could increase subsequent task difficulty and increase the likelihood of observing individual differences in EF.
Wait-for-treat task
The wait-for-treat task was used by Müller et al. (2016) as a part of an inhibitory control battery to learn whether EF influences problem-solving ability in dogs. In this task, dogs were first trained by their owners to wait for permission before consuming a treat. During training, the owner placed a treat on the floor within 10 cm in front of the dog and commanded the dog to “wait.” After a few seconds had passed, the owner said “go” and permitted the dog to consume the treat. The delay between the “wait” and “go” command then became progressively longer with each successive trial. During testing, the dog was presented with the same task and was given an inhibitory control score ranging from 0 (dog never waited at least 5 s to consume treat) to 2 (dog waited for “go” command before consuming the treat following a duration of at least 5 s during first two trials, or during three of four consecutive trials). This task was intended to measure a dog’s ability to delay gratification in a shorter, simpler manner than the delay-discounting task mentioned earlier in this section. Upon creating a composite score from this and the other two tasks (the leash task and middle cup task), the researchers found that EF (specifically, inhibitory control ability) predicted subsequent problem-solving ability better than experience with the object used in the problem-solving task.
One might argue that this task merely reflects obedience, that is, a dog’s willingness to follow its owner’s commands. Although this might be true for the initial response to the command, sustaining the command to “wait” likely relies on more than obedience, especially at longer durations. To be successful at the task, a dog must (1) inhibit the response of consuming the treat in favor of following the command to “wait” on a moment-to-moment basis and (2) internally maintain the command to wait over a progressively longer period of time. Thus, this task might be reflective of both inhibitory control and WMC. However, because the authors acknowledge that performance might rely partly on a dog’s level of trainability, training history should be accounted for when using and interpreting findings from this task. Additionally, researchers should report the range of maximum delays achieved by the subjects to evaluate task difficulty, as well as the potential influences of breed and training.
Reversal task
Reversal tasks are used to measure EF, particularly cognitive flexibility, and are especially popular in the aging literature. These discrimination tasks typically consist of an acquisition phase and a reversal phase. During the acquisition phase, subjects are rewarded for making a specific response, such as selecting a particular item from an array (wooden blocks: Tapp et al. 2003), or exiting a maze using a specific route (Mongillo et al. 2013). The acquisition phase continues until the subject has met pre-specified criteria for having learned the task, at which point the stimulus-reward contingencies are reversed. During the reversal phase, subjects must overcome the urge to perform the previously rewarded response in favor of performing the opposite response (e.g., selecting a small block when the large block had previously been rewarded; Tapp et al. 2003). This task has been used with a variety of species to examine how aging affects EF, as well as testing the efficacy of treatments for age-related cognitive decline (Cotman et al. 2002; Milgram 2003; Milgram et al. 2002, 2005).
Using a reversal task, Tapp et al. (2003) tested young, middle-aged, old, and senior laboratory-raised beagles to examine aging effects on initial acquisition and reversal ability. They found that young and middle-aged dogs were able to learn the initial discrimination more quickly than old and senior dogs and were also more successful in responding to the change in contingencies. Whereas younger dogs were able to quickly adapt to the new contingency, older dogs took markedly longer to respond correctly to the reversal. Although both old and senior dogs showed difficulty in reversing, they showed different patterns of responding, leading authors to suggest different mechanistic failures underlying each group’s performance. Specifically, old dogs showed impairment in learning stimulus–response (S-R) contingencies, whereas senior dogs showed impairment in withholding responses to previously rewarded locations. Such patterns are not specific to laboratory-raised beagles: Similar results were obtained in a sample of Labrador retrievers trained as explosive detection dogs (Lazarowski et al. 2014), as well as a large sample of pet dogs by Mongillo et al. (2013), who used a T-maze to test learning and reversal abilities rather than the size-discrimination task used by Tapp et al.
The finding that EF diminished with increasing age in this study mirrors age-related patterns found by other dog researchers (Bray et al. 2014; Wallis et al. 2014, 2016), as well as those observed in monkeys (Gray et al. 2017; Lai et al. 1995), humans (Cepeda et al. 2001; Craik and Bialystok 2006), and those diagnosed with Alzheimer’s disease (Hutchison et al. 2010). Together, these results also suggest that older dogs are more susceptible to the effects of proactive interference, a process by which information from previous trials interferes with performance on the current trial. In humans, overcoming proactive interference requires attention during encoding of the new S-R contingency as well as retrieval during subsequent trials (Kane and Engle 2000). Resilience in the face of proactive interference thus relies on EF made possible by proper functioning of the PFC which has been shown to deteriorate during the aging process of both humans and dogs, especially for individuals with Alzheimer’s disease or canine cognitive dysfunction syndrome (Hutchison et al. 2010; Landsberg et al. 2003, 2012; Ruehl et al. 1995).
A-not-B task
Like the cylinder task, the A-not-B task originated with Piaget’s (1954) study of the development of object permanence in human infants. In these studies, an experimenter places a toy under one of two boxes while an infant watches and then allows the infant to attempt retrieving the toy. For several successive trials, the toy is placed under the same box (typically referred to as “box A”). On the final, critical trial, the experimenter places the toy under box A and then removes it and places it under the other box (“box B”), while the infant watches. Resolution of the fourth stage of object permanence (and onset of inhibitory control ability; Garavan et al. 1999) is inferred if the infant successfully reaches for box B. The A-not-B error (also known as the perseveration error) occurs when the infant reaches for the now-empty box A instead of box B.
Initially created to test object permanence, the A-not-B task has since adapted and used to assess aspects of EF in human infants and nonhuman primates (Amici et al. 2008; Diamond 2002; Diamond and Goldman-Rakic 1989; Topál et al. 2008, 2009), as well as dogs (Bray et al. 2014; Cook et al. 2016a, b; Fagnani et al. 2016; MacLean et al. 2014). Successful performance on the A-not-B requires holding an object’s new location in WM, overcoming the proactive interference from previous trials, and inhibiting the prepotent response of looking for the object at the previously rewarded location. In the canine cognition literature, the A-not-B task typically consists of two phases: training and testing. In the training phase, a dog observes the experimenter place a treat under one of three opaque cups. The dog is then released and permitted to search for the treat and eat it (if it is found on the first attempt). Across a variable number of trials, the dog observes as the same cup (cup A) is baited with the treat. If the dog successfully retrieves the treat on a pre-specified majority of the trials, it moves on to the testing phase. In the ten testing trials, the dog once again watches as a human experimenter baits cup A with a treat. Then, the dog watches as the experimenter removes the reward from the baited cup (container A) and places it under the other cup (container B). The subject is then released and must avoid approaching the now-empty cup A and instead approach cup B to successfully retrieve the treat. Performance metrics in the A-not-B task vary from study to study, with researchers reporting the percentage of dogs that respond accurately during a singular test trial (Bray et al. 2014; MacLean et al. 2014), dogs’ accuracy rates across multiple test trials (Fagnani et al. 2016), or the number of trials preceding the first accurate response (Cook et al. 2016a, b; Fagnani et al. 2016).
Overall, dogs tend to perform exceptionally well during the A-not-B task (see Table 1), with eighty to ninety percent of dogs responding correctly in studies using only one test trial (Bray et al. 2014; MacLean et al. 2014). As with the cylinder task, the A-not-B task might not be sufficiently challenging to allow individual differences to emerge. Some researchers have argued that this task is not a strong measure of aspects of EF, such as inhibitory control or WMC, but rather reflect the domestic dog’s ability to use inadvertent social cues from the human experimenter. Indeed, studies have shown that dogs show significantly lower accuracy when human cues are hidden than when they are present (Kis et al. 2012; Péter et al. 2015; Topál et al. 2009; Topál et al. 2014). However, authors of two separate studies (Bray et al. 2014; Fagnani et al. 2016) used the same precaution to minimize the effect of such cuing; after baiting cup A, the experimenters stepped away from the cups and faced the wall opposite the dog, making cuing impossible. Thus, susceptibility to the A-not-B error likely reflects a combination of factors, including EF failures owing to proactive interference and/or inhibitory control failure, sensitivity to human cuing (if precautionary measures are not taken during testing), and motivation to perform the task (Sümegi et al. 2014).
Departing from other studies showing generally high performance in this task, Fagnani et al. (2016) have provided data suggestive of lower EF in shelter dogs compared to pet dogs. In their design, both populations of dogs were given fifteen test trials, and performance was measured in terms of number of trials before the first accurate response, as well as the overall percentage of accurate responses. While the pet dogs showed a mean accuracy similar to that obtained in previous studies (M = 78%), shelter dogs showed an accuracy that was only half that (M = 39%). Future research should examine why shelter dogs exhibit such difficulty on this task. Is this finding truly due to lower EF in shelter dogs, the consequences of which might have led to their relinquishment? Might the stresses inherent in the shelter environment deplete a dog’s capacity for EF as previous research suggests (Alexander et al. 2007; Arnsten and Goldman-Rakic 1998; Baumeister et al. 1998)? Learning whether shelter and pet dogs differ in this respect, as well as the origins of reduced EF in shelter dogs, could help lay the foundation for interventions to increase EF while in the shelter, or reducing potential environmental stressors that may lead to suboptimal behavior while interacting with shelter staff and potential adoptive families.
Overall, the A-not-B task seems to be a shorter version of the reversal task mentioned earlier in this paper. Unlike the results of the aforementioned literature showing detrimental effects of aging on human and dog EF, it has not yet been shown to be affected by age. However, the ostensive ceiling effect suggests that the task is too easy to make age differences readily apparent. As with the cylinder task, future studies using the A-not-B task should (1) assess reliability of this measure, (2) use a larger sample sizes (such as that used by MacLean et al. 2014) to further examine differences relating to age and environment, and (3) explore the effects of proactive interference on EF in dogs.
Visual displacement task
The visual displacement task is another task derived from object permanence studies initially developed by Piaget (1954) and further modified to study other aspects of cognition (Uzgiris and Hunt 1975). In this task, the dog watches as an experimenter “hides” a desired object (such as a toy) behind one of four boxes. The dog’s view of the boxes is then obstructed by an opaque screen. After the passage of a pre-specified delay (ranging from 0 s to over 2 min), the screen is removed and the dog is permitted to search for the object. This task requires the dog to encode and maintain information about the object’s location over a delay and is similar to the Brown–Peterson task used to examine WMC in humans (Brown 1958; Peterson and Peterson 1959).
Using this task, Fiset et al. (2003) found that all dogs in their sample performed at above chance levels across all delays (ranging from 0 to 240 s); however, performance was significantly worse at longer delays. In their first experiment, accuracy was significantly higher at delays of 0 and 10 s than at delays of 30 and 60 s. In their second experiment, performance was significantly better at 0 and 30 s than at 60, 120, and 240 s. This pattern suggests that dogs experience greater difficulty holding spatial information in working memory for durations surpassing 30 s which could be due to surpassing the limits of a dog’s short-term memory capacity. However, it could instead reflect proactive interference from previous trials (see Keppel and Underwood 1962), which increases at longer current trial delays by impairing temporal discrimination between current and recent trials (Bjork and Whitten 1974). In tasks using nonhuman animals, proactive interference is inferred on any given trial when a subject revisits the location rewarded on the previous trial. Researchers found no evidence of proactive interference in either of the two experiments. Specifically, the dogs were no more likely to search the box rewarded on the previous trial at long delays than short delays. Further, only two out of eleven and two out of eight dogs (from Experiment 1 and Experiment 2, respectively) searched a previously rewarded box at above chance levels, suggesting proactive interference was not a significant issue in this task. According to the authors, using the visual displacement task including the introduction of an opaque screen can be a useful method for examining an animal’s working memory for disappearing objects. Although their results are compelling, this study should be replicated with a larger sample.
Summary
A variety of tasks have been used to understand EF in dogs, from the limits of their WMC to how they withhold counterproductive urges. Studies using these tasks have uncovered more parallels between human and dog cognition, such as the negative effect of aging on processes like cognitive flexibility and selective attention. However, some of the frequently used tasks (e.g., cylinder and A-not-B tasks) show robust ceiling effects, which might limit their utility in the study of EF, which requires effortful allocation of attention. Researchers might consider modifying these tasks to make them more difficult and thus more reflective of a dog’s EF.
Insights and limitations of previous studies
So far, I have described the importance of EF, presented an argument for its continued study in dogs, and reviewed some of the research methods used to increase scientific understanding of canine EF. I now summarize what researchers have learned thus far (i.e., what we know) and limitations of the current body of research (i.e., what we do not know).
First, researchers have provided preliminary evidence that the aging process affects dog and human EF similarly (Bray et al. 2014; Mongillo et al. 2013; Tapp et al. 2003; Wallis et al. 2014, 2016). Specifically, older dogs have a harder time inhibiting prepotent responses (Bray et al. 2014), selectively attending to information in their environment and sustaining attention (Wallis et al. 2014), learning new tasks, and flexibly adjusting their responding based on changing environmental conditions (Mongillo et al. 2013; Tapp et al. 2003). Because small and large dogs age at different rates, future research could examine whether dogs demonstrate different trajectories of cognitive decline based on their relative size.
Second, it is possible that shelter dogs possess lower inhibitory control than pet dogs (Fagnani et al. 2016). Although performance was not significantly different between these two groups on the cylinder tasks, pet dogs performed twice as well on the A-not-B task as shelter dogs. If this finding reflects a true deficit of EF in shelter dogs, it is of practical importance that this deficit be examined more closely. From where does this deficit emerge? Might the stressful environment of the shelter place a cognitive load on these dogs, making it difficult for them to engage EF? Does the reduced level of human contact, sometimes both quantitatively and qualitatively, lead to this deficit? Is this deficit preexisting, with low EF being the reason for relinquishment? These hypotheses are not mutually exclusive, and further exploration of the root of this deficit carries practical implications for owners, shelters, and the dogs themselves. If EF can be somehow increased through training, relinquishment can be attenuated, likelihood of adoption can be enhanced, and length of stay at the shelter can be shortened.
Although all of the studies mentioned in this paper ask (and attempt to test) important questions regarding canine EF, myriad methodological issues limit the interpretation of the findings contained therein. I have already discussed many of these issues, but briefly reiterate them here. Recurrent issues in the current body of research are consistently small sample sizes, lack of reliability assessment of measures, weak or absent attempts to address the task-impurity problem, and inconsistency in conceptualizing factors like breed and training.
First, the use of small sample sizes in dog research must be mitigated, especially when researchers are testing for potential differences between groups. The first concern is statistical: Using a small sample can prevent a researcher from detecting existing effects or lead to the finding of spurious effects that do not truly exist (Arden et al. 2016; Button et al. 2013). This can lead to low replicability or contradictory findings, even when using the exact same methods. This slows the acquisition of knowledge in the field and can create misperceptions of dogs’ abilities. The second concern is both ethical and economical. If the use of small samples provides unreliable and incorrect data, the researcher has wasted resources—in terms of time and money—that were used while conducting the study, and the accumulation of such wasted resources across several underpowered studies is likely substantial. Additionally, the subjects themselves can be viewed as wasted resources, a problem that violates reduction, one of the “three Rs” imposed by the Institutional Animal Care and Use Committee. Reduction demands that animal researchers use as few animals as possible to address their research question and is likely an important contributor to the use of small sample sizes in studies described in this paper. In other words, researchers may not be using small samples for the sake of expediency, but rather to satisfy requirements that allow them to conduct research in the first place. Striking a balance between ethical and methodological considerations thus poses a challenge to animal researchers when determining a sample size. Power analysis could be a useful tool in guiding researchers’ efforts to arrive at an appropriate sample size. Alternatively, researchers could turn to citizen science, in which the general public assist in data collection and analysis (Hecht and Rice 2014, 2015; Stewart et al. 2015).
Second, some of the tasks currently being used to measure EF have not been tested in terms of their reliability. The cylinder task, the leash task, and the wait-for-treat task are examples of measures new to the field of canine cognition which have been used, sometimes repeatedly, without knowledge of their reliability. This problem is not unique to dog research; Miyake et al. (2000) describe several tasks used in human cognition research without adequate inquiry regarding their reliability. In particular, this poses a problem interpreting the null or weak correlations observed in various test batteries. Using unreliable tasks will attenuate correlations between tasks, even if the tasks measure the same ability. This might explain why, for example, there were no correlations among tasks in batteries that included the cylinder task; assertions made by researchers that EF is context specific are thus premature. In short, reliability estimates must be obtained before a task is included in a battery.
Third, some of the existing tasks used to measure EF in dogs likely reflect other abilities including spatial problem-solving and trainability. For example, dogs who are better at inhibiting suboptimal responding might have a higher level of EF or might simply have a stronger training history. In particular, the wait-for-treat task seems to rely on a dog knowing both “sit” and “wait” commands. Additionally, the detour-fence task and leash task seem to heavily rely on spatial awareness and problem-solving ability. A dog that cannot untangle its leash from a tree might have high EF, but lack the spatial skills required to free itself. As has been learned by human cognitive researchers, no task is “process pure,” measuring only the ability of interest to the researcher. Using larger task batteries and latent variable analyses can help canine researchers extract underlying abilities that are common across tasks.
Finally, our knowledge of the effects of genetics and training are limited by the variety of ways by which these factors are defined across studies. For example, Marshall-Pescini et al. (2016) found that trained dogs were faster and more accurate at navigating the detour-fence tasks than untrained dogs. This finding should be considered in light of two things: (1) The detour-fence task was used in this study as a measure of problem-solving, not executive or inhibitory ability, and (2) the researchers defined “trained” dogs as those who had carried out agility or another dog sport at a competitive level or were certified as working dogs (police dogs, guard dogs, etc.), whereas “untrained” dogs had either no training at all or only basic obedience level training.Footnote 4 These definitions are not problematic in and of themselves, but should be considered by readers when interpreting the data. When examining the impact of genetics, researchers often use breed as a factor. However, breed can be defined in many different ways. Researchers can use AKC designations (e.g., Great Dane), breeds grouped based on genetic similarity (e.g., Mastiff type), or breeds grouped by intended function (e.g., herding breeds). Again, it is not necessarily problematic that researchers conceptualize “breed” differently, but is a fact that should be taken into account when comparing results of different studies and drawing conclusions on how breed might (or might not) determine abilities like inhibitory control or working memory capacity.
Summary
Although canine scientists have illuminated potential influences of aging and environment on a dog’s EF, future studies must address methodological issues to achieve a more complete understanding of this construct in dogs. These issues include using small samples, not accounting for variables reflected in EF task performance, and including tasks in batteries without knowing the reliability of these tasks. Increased knowledge of how genetics and training determine EF is also a fruitful area for future research.
Future directions
Because of dogs’ unique niche in human society, we can study them to better understand how to improve their behavior and welfare and to better understand ourselves. However, their continued success in our society will be enhanced if we dedicate more research to learning which factors influence their ability to behave in appropriate ways. The following are suggestions for future research that could allow us to better understand how EF works in “man’s best friend.”
Task battery development and latent variable analysis
In both dogs and humans, EF is better viewed as a multifaceted phenomenon rather than a unitary construct (Miyake et al. 2000; Müller et al. 2016; Pennington and Ozonoff 1996). Because of EF’s complexity, human researchers have used methods including task batteries and latent variable analysis to learn how it contributes to the use of semantic memory, aggression, and general fluid intelligence (Hutchison 2007; Sprague et al. 2011; Unsworth et al. 2014). When used properly, these methods indicate how much of the variance observed in performance among participants is due to specific factors, such as primary memory, secondary memory, and attentional control (Shipstead et al. 2014; Unsworth et al. 2014). Given the utility of latent variable analysis demonstrated in the human literature and the repeated calls for task batteries measuring EF in dogs (Marshall-Pescini et al. 2015; Bray et al. 2014; Fagnani et al. 2016), this is a fruitful direction for future research.
To conduct a latent variable analysis, researchers first have participants complete several cognitive tasks that have been shown to measure specific cognitive abilities, and then conduct an exploratory factor analysis to find commonality in task performance (Hutchison 2007; Miyake et al. 2000; Unsworth et al. 2014). The reason behind using such a large number of tasks is the task-impurity problem; using several tasks allows researchers to extract what is common among the tasks in terms of underlying ability. Because of variance introduced by task-specific abilities irrelevant to the underlying construct of interest, latent variable analysis has been described as a more “pure” method of assessing complex abilities like EF than using only one task (see Conway et al. 2003 for a discussion).
As previously discussed, task batteries created for use with dogs show either weak or null correlations. Although “context specificity” has become the favorite explanation for these findings, the pattern seems reminiscent of problems encountered by cognitive psychologists in studying EF in humans. As discussed by Miyake et al. (2000), some of the tasks being used to measure EF were not well validated and, further, had low reliability. The authors warn against testing correlations between such tasks as unreliable tasks will necessarily be unlikely to correlate with other tasks. Nonetheless, researchers at this time used a similar explanation as some canine researchers (Bray et al. 2014; Fagnani et al. 2016) that the measures did not correlate because independent processes were being used for each. Given that reliability estimates for tasks used in recent EF batteries in dogs are unknown, Miyake’s explanation seems more likely.
The careful construction of task batteries and use of latent variable analysis has the potential to shed light on the structure of EF in dogs and whether it is, indeed, context specific. The utility of such methods is contingent on researchers addressing problems well known to researchers studying human cognition, as well as those unique to the canine cognition field. First, any task included in a battery must first be shown to be reliable. Test–retest reliability seems most feasible for tasks like the cylinder task and the A-not-B, whereas split-half reliability could be used for the visual displacement task; however, measures of EF tend to be most valid when the task is novel (Miyake et al. 2000), a fact posing a potential challenge to assessing reliability of tasks used with dogs. Additionally, the reliability of a measure depends on researchers using the same methods when conducting their studies. Second, larger samples must be used. The consistent use of only a few dozen subjects in canine research can hinder a researcher’s power to uncover an existing effect. This problem is compounded when task reliability is questionable. Finally, researchers can test the possibility of context specificity. As described by Bray et al. (2014), their devised social task might have been more difficult for socially motivated dogs, whereas the cylinder and A-not-B tasks might have been more difficult for food-motivated dogs. A variety of questionnaires have been created to assess traits in dogs including trainability, extraversion, and excitability (Hsu and Serpell 2003; Ley et al. 2008; Mirkó et al. 2012). If Bray et al.’s proposal is correct, we should expect that dogs rated high on extraversion or affection-seeking should perform better on the social task than dogs rating lower on these factors. In conclusion, meticulous battery creation and discovery of latent EF variables could be an invaluable way to learn about relationships between EF and factors like breed, training, and environmental demands, as well as predicting a dog’s potential success in a working role, general trainability, or likelihood of adoption from a shelter.
Training EF
Because researchers have proposed that shelter dogs may have lower EF (inhibitory control, more specifically) than pet dogs (Fagnani et al. 2016), it is of practical and ethical interest whether dogs suffering from low EF can overcome this deficit through training. Studies in the human domain have shown that, despite having a strong genetic component, EF can be improved through training (Rueda et al. 2005); however, others argue that the benefits of such training are limited to short-term, task-specific abilities (Redick et al. 2015). Some canine researchers have found evidence that training can increase problem-solving ability (Marshall-Pescini et al. 2008), but the effects of training on EF are, as of yet, unexplored. Discovering whether training can positively influence EF has the potential to lead to the development of interventions that could enhance the bond between a dog and its owner, increase successful adoptions, and reduce euthanasia rates at shelters.
Eye tracking
Eye-tracking technology has been used in human research since the 1960s and uses an apparatus that reflects infrared light off the retina to observe changes in ocular activity such as pupil dilation and gaze fixation in response to presented stimuli. Researchers using this technology have found, for example, that pupil diameter increases when an individual is engaging in a challenging task, relative to an easy task (Beatty 1982; Peavler 1974), and in response to emotionally arousing stimuli relative to neutral stimuli (Partala and Surakka 2003). The use of eye trackers has been useful in understanding not only effortful cognitive processing, but also affective processing in humans (reviewed in Goldwater 1972), and could potentially shed light on EF in dogs.
Although using such methods with dogs may seem implausible, their high level of trainability makes them amenable to study with eye trackers and, as I discuss next, imaging techniques. In fact, several researchers have successfully employed eye trackers to study dog cognition in ways that traditional behavioral measures cannot. To learn how dogs process visual information presented in pictures, Somppi et al. (2012, 2014) used a contact-free eye tracker typically used in human cognition studies. Dogs were trained to lay still with their chin resting on a pad and then presented with pictures belonging to different categories (including upright and inverted human faces, upright and inverted dog faces, inanimate objects, and alphabetic characters) using a computer monitor. The researchers found that dogs tend to focus their gaze on information-rich areas of the images, such as the eye regions of dogs and humans, and preferred images of dog faces to other image categories (e.g., inanimate objects and alphabetical characters). The researchers concluded that dogs are amenable to study with the use of eye-tracking technology and that data from such studies could provide useful information regarding visual perception in dogs, including how they attend to humans and other dogs.
Subsequent investigations using eye tracking have focused on the roles of familiarity, species, and experience with humans on visual responses. Somppi et al. (2016) showed that dogs’ attend in different ways to threatening emotional expressions, depending on whether they are exhibited by a human or conspecific. Specifically, subjects in this study increased visual attention toward expressions of other dogs, but visually avoided those of humans. Other research has showed that processing of human faces is influenced by familiarity and lifetime experience with humans (Barber et al. 2016) and that administration of oxytocin can influence visual attention (Somppi et al. 2017).
Although designs using contact-free eye trackers are often used, head-mounted, portable eye-tracking systems have also been created for use with dogs. Using a head-mounted eye tracker, Rossi et al. (2014) examined gazing behavior during an object choice task to see where dogs look to obtain information about a hidden object’s location. In this task, dogs watched as a human demonstrator baited one of two plastic cups with a treat. The cups were then shuffled out of view of the dog to prevent the dog from knowing which cup had the treat. Then, the demonstrator pointed to one of the cups, at which point the dog was released and permitted to approach the cups (and eat the treat under the target cup if it was selected). Consistent with the findings of Somppi et al. (2014), the researchers found that dogs gazed mostly at the demonstrator’s face during the task. However, the dogs did not show significant differences in gazing behavior between familiar and unfamiliar demonstrators. Such an apparatus has the advantage of being portable, making it possible to study dogs in a variety of settings outside the laboratory. A disadvantage is the challenge of finding dogs that are tolerant of wearing such a device (see also Williams et al. 2011).
Eye-tracking technology can provide insight regarding dogs’ visual behavior in social situations with dogs and humans and during cognitive tasks that is unattainable through behavioral methods (Téglás et al. 2012; Somppi et al. 2012, 2014). For example, researchers could measure where dogs look as a human demonstrator places a treat under a cup or in a transparent cylinder or measure exerted effort during progressively longer durations in the visual displacement task. Important for researchers to consider is that, for both contact-free and portable devices, some dogs might be more tolerant of the apparatus (introducing the same potential for bias as mentioned by Berns and Cook 2016).
Functional magnetic resonance imaging (fMRI)
Another departure from strictly behavioral methods is the use of imaging technology with dogs. Magnetic resonance imaging (MRI) allows researchers to noninvasively view the structure of a dog’s brain, whereas functional magnetic resonance imaging (fMRI) shows the brain’s function in response to presented stimuli in real time. A challenge inherent to the use of imaging techniques is keeping subjects awake during scanning, a challenge that was met by Tóth et al. (2009), who published the first study in which brains of awake, unsedated dogs were imaged using MRI. The two subjects in this study were habituated to the confinement and sounds of the scanner and trained to lay still, while the MRI collected images. Berns et al. (2012) further extended this line of research by training dogs to lay still inside an fMRI scanner (for further reading, please see Thompkins et al. 2016).
The addition of fMRI technology to the dog researchers’ toolkit has proven to be a rich source of information. While traditional methods require researchers to infer underlying processes from a dog’s behavior, imaging techniques show the brain’s responses to presented stimuli in real time. This has advanced scientific understanding of how dogs process visual, acoustic, and olfactory information from both humans and other dogs (Andics et al. 2014; Berns et al. 2015; Cuaya et al. 2016; Dilks et al. 2015), individual preferences food or praise when being rewarded (Cook et al. 2016a, b), lexical processing of human speech (Andics et al. 2016), and, of particular importance to this paper, individual differences in EF (Cook et al. 2016a, b). In a recent study, Cook et al. 2016a, b had dogs complete two EF tasks, one while inside the scanner (the go-no-go task) and one while outside the scanner (the A-not-B task). Results showed greater activation in the frontal lobe when a dog successfully inhibited a response during the go/no-go task and that dogs who showed better performance on the go/no-go task committed fewer errors in the A-not-B task. From these findings, the researchers concluded that dogs exhibit consistent individual differences, just as humans do (Engle 2002; Lee and Therriault 2013; Braver et al. 2007).
Using fMRI provides previously inaccessible information of the dog’s perceptual world and could inform more educated creation of tasks to study the human–dog relationship, EF, and social behavior. For example, researchers could see how performance and brain activity during a cognitive task relates to behavior outside of the scanner, such as owner-reported aggression and impulsivity or level and type of training. As Berns and Cook (2016) suggest, it could also be used to determine which dogs might be best suited for working roles, potentially reducing the high costs associated with training dogs who are ultimately “unemployable.” Importantly, the authors caution the reader to be mindful of the possible bias inherent in data drawn from dogs willing to undergo fMRI scanning. Firstly, training a dog to willingly sit in a scanner, which is a noisy, enclosed space, requires 2–4 months of extensive shaping and training. Because not all dogs will be suited for such training, the dogs whose data end up being used and published might be more trainable, more intelligent, or less reactive than the dogs to whom the results are generalized. Further, as dogs are always “on-task” in the scanner, it can be difficult to ascertain differences between baseline and experimental trials, particularly when it comes to EF tasks. That is, the act of remaining still in a noisy scanner for prolonged periods of time likely requires the maintenance of the goal to “remain still,” and ignoring the sounds of the scanner to focus on the cognitive task at hand. The exertion of such control during the course of the task may attenuate observable differences in activation between ostensible “baseline” trials and experimental trials.
Summary
The continued exploration of dog EF is a timely and worthy challenge for canine researchers. Given recent developments, scientists are not limited to behavioral methods, but can integrate technologies like eye tracking and fMRI into their experiments. Such physiological measures, as well as use of latent variable analysis, have proven invaluable in the domain of human cognition. Further, learning whether training can enhance EF and downstream behaviors might inform behavioral interventions that could reduce damage to the dog–owner bond, relinquishment to shelters, and aggressive or destructive behavior.
Conclusion
EF is a multifaceted cognitive construct that allows human and nonhuman animals to engage in flexible, context-appropriate behavior. In dogs, EF is essential for thriving in their human-dominated environments and is implicated in trainability and suitability for working roles, as well as undesirable behaviors like biting and disobedience. Thus, an improved understanding of dog EF is a worthy pursuit of canine researchers, which can be facilitated by more rigorous research designs and incorporation of technologies like eye tracking and imaging. Increased understanding of this important construct might allow us as humans to influence dog EF for the better through training, socialization, and breeding practices, especially with possible at-risk populations (e.g., shelter dogs). Additionally, future research might inform dog-specific legislation and allow restrictions to be enacted based on a given dog’s training history and temperament, rather than stereotypes related to breed or size.
Notes
Although problem-solving ability is a higher-order cognitive function, it is viewed as an outcome of EF rather than a part of it. To illustrate, the problem-solving requires the ability to take into account the full scope of a task, attend to the task while ignoring irrelevant stimuli, learn the task, and apply previously acquired knowledge to novel problems (see Diamond 1990; Duckworth and Seligman 2005; Tangney et al. 2004).
Such similarities can arise from cognitive continuity that exists across species or influences of convergent evolution, under which different species show similar adaptations as a result of similar selection pressures in the environment (Darwin 1871).
Researchers examining WMC in humans have found that people scoring in the upper range of the distribution on complex span tasks (termed high spans) are more likely to spontaneously engage and sustain EF, whereas people in the lower range (termed low spans) need external reminders of task goals to maintain accuracy (briefly reviewed by Engle 2002. See also Braver et al. 2007). In the self-control task, the human command to “sit” and “stay” can be viewed as an external reminder that may be needed in absence of sustained EF.
Also, size and baseline speed of the dogs were not accounted for. The speed by which dogs were able to complete the task might have been an artifact of the physical attributes of the dog rather than a reflection of training or cognitive ability (Helton 2010).
References
Alexander JK, Hillier A, Smith RM, Tivarus ME, Beversdorf DQ (2007) Beta-adrenergic modulation of cognitive flexibility during stress. J Cogn Neurosci 19:468–478
American Society for the Prevention of Cruelty to Animals (n.d.). Pet statistics. Retrieved from https://www.aspca.org/animal-homelessness/shelter-intake-and-surrender/pet-statistics
Amici F, Aureli F, Call J (2008) Fission-fusion dynamics, behavioral flexibility, and inhibitory control in primates. Curr Biol 18(18):1415–1419
Andics A, Gácsi M, Faragó T, Kis A, Miklósi Á (2014) Voice-sensitive regions in the dog and human brain are revealed by comparative fMRI. Curr Biol 24:574–578
Andics A, Gábor A, Gácsi M, Faragó T, Szabó D, Miklósi Á (2016) Neural mechanisms for lexical processing in dogs. Science 353(6303):1030–1032
Arden R, Bensky MK, Adams MJ (2016) A review of cognitive abilities in dogs, 1911 through 2016: more individual differences, please! Curr Dir Psychol Sci 25(5):307–312
Arnsten AF, Goldman-Rakic PS (1998) Noise stress impairs prefrontal cortical cognitive function in monkeys: evidence for a hyperdopaminergic mechanism. Arch Gen Psychiatry 55(4):362–368
Azkona G, García-Belenguer S, Chacón G, Rosado B, León M, Palacio J (2009) Prevalence and risk factors of behavioural changes associated with age-related cognitive impairment in geriatric dogs. J Small Anim Pract 50:87–91
Baddeley AD, Hitch G (1974) Working memory. Psychol Learn Motiv 8:47–89
Balota DA, Tse C, Hutchison KA, Spieler DH, Duchek JM, Morris JC (2010) Predicting conversion to dementia of the Alzheimer’s type in a healthy control sample: the power of errors in Stroop color naming. Psychol Aging 25(1):208–218
Barber ALA, Randi D, Müller CA, Huber L (2016) The processing of human emotional faces by pet and lab dogs: evidence for lateralization and experience effects. PLoS ONE 11(4):e0152393. https://doi.org/10.1371/journal.pone.0152393
Barkley RA (1997) Behavioral inhibition, sustained attention, and executive functions: constructing a unifying theory of ADHD. Psychol Bull 121(1):65–94
Baumeister RF (2014) Self-regulation, ego depletion, and inhibition. Neuropsychologia 65:313–319
Baumeister RF, Bratslavsky E, Muraven M, Tice DM (1998) Ego depletion: is the active self a limited resource? Personal Process Individ Differ 74(5):1252–1265
Beatty J (1982) Task-evoked pupillary responses, processing load, and the structure of processing resources. Psychol Bull 91(2):276–292
Berg EA (1948) A simple objective technique for measuring flexibility in thinking. J Gen Psychol 39(1):15–22
Berns GS, Cook PF (2016) Why did the dog walk into the MRI? Curr Dir Psychol Sci 25(5):363–369
Berns GS, Brooks AM, Spivak M (2012) Functional MRI in awake unrestrained dogs. PLoS ONE 7(5):e38027. https://doi.org/10.1371/journal.pone.0038027
Berns GS, Brooks AM, Spivak M (2015) Scent of the familiar: an fMRI study of canine brain responses to familiar and unfamiliar human and dog odors. Behav Proc 110:37–46
Beurms S, Miller HC (2016) Sharing more than the sofa: what dogs can teach us about human self-control. Curr Dir Psychol Sci 25(5):351–356
Bjork RA, Whitten WB (1974) Recency-sensitive retrieval processes in long-term free recall. Cogn Psychol 6:173–189
Boogert NJ, Anderson RC, Peters S, Searcy WA, Nowicki S (2011) Song repertoire size in male song sparrows correlates with detour reaching, but not with other cognitive measures. Anim Behav 81(6):1209–1216
Braver TS, Gray JR, Burgess GC (2007) Explaining the many varieties of working memory variation: Dual mechanisms of cognitive control. In: Conway A, Jarrold C, Kane MJ, Miyake A, Towse JN (eds) Variation in working memory. Oxford University Press, New York, pp 76–106
Bray EE, MacLean EL, Hare BA (2014) Context specificity of inhibitory control in dogs. Anim Cogn 17(1):15–31
Bray EE, MacLean EL, Hare BA (2015) Increasing arousal enhances inhibitory control in calm but not excitable dogs. Anim Cogn 18(6):1317–1329
Brown J (1958) Some tests of the decay theory of immediate memory. Q J of Exp Psychol 10(1):12–21
Button KS, Ioannidis JPA, Mokrysz C, Nosek BA, Flint J, Robinson ESJ, Munafð MR (2013) Power failure: why small sample size undermines the reliability of neuroscience. Nat Rev Neurosci 14:365–376
Casey RA, Loftus B, Bolster C, Richards GJ, Blackwell EJ (2014) Human directed aggression in domestic dogs (Canis familiaris): occurrence in different contexts and risk factors. Appl Anim Behav Sci 152:52–63
Cepeda NJ, Kramer AF, Gonzalez de Sather JCM (2001) Changes in executive control across the lifespan: examination of task-switching performance. Dev Psychol 37(5):715–730
Clark A, Chalmers D (1998) The extended mind. Analysis 58(1):7–19
Conway ARA, Engle RW (1994) Working memory and retrieval: a resource-dependent inhibition model. J Exp Psychol Gen 123(4):354–373
Conway AR, Cowan N, Bunting MF (2001) The cocktail party phenomenon revisited: the importance of working memory capacity. Psychon Bull Rev 8(2):331–335
Conway ARA, Kane MJ, Engle RW (2003) Working memory capacity and its relation to general intelligence. Trends Cognit Sci 7(12):547–552
Cook PF, Prichard A, Spivak M, Berns GS (2016a) Awake canine fMRI predicts dogs’ preference for praise vs food. Soc Cognit Affect Neurosci 11(12):1853–1862
Cook PF, Spivak M, Berns G (2016b) Neurobehavioral evidence for individual differences in canine cognitive control: an awake fMRI study. Anim Cogn 19:867–878
Cotman CW, Head E, Muggenburg BA, Zicker S, Milgram NW (2002) Brain aging in the canine: a diet enriched in antioxidants reduces cognitive dysfunction. Neurobiol Aging 23:809–818
Cuaya LV, Hernández-Pérez R, Concha L (2016) Our faces in the dog’s brain: functional imaging reveals temporal cortex activation during perception of human faces. PLoS ONE 11(3):e0149431. https://doi.org/10.1371/journal.pone.0149431
Darwin C (1871) The descent of man, and selection in relation to sex. Murray, London
De Pisapia N, Braver TS (2006) A model of dual control mechanisms through anterior cingulate and prefrontal cortex interactions. Neurocomputing 69(10):1322–1326
Deater-Deckard K, Sewell MD, Petrill SA, Thompson LA (2010) Maternal working memory and reactive negativity in parenting. Psychol Sci 21(1):75–79
Deater-Deckard K, Wang Z, Chen N, Bell MA (2012) Maternal executive function, harsh parenting, and child conduct problems. J Child Psychol Psychiatry 53(10):1084–1091
Diamond A (1990) Developmental time course in human infants and infant monkeys, and the neural bases of, inhibitory control in reaching. Ann N Y Acad Sci 608(1):637–676
Diamond A (2002) Normal development of prefrontal cortex from birth to young adulthood. In: Stuss DT, Knight RT (eds) Principles of frontal lobe function. Oxford University Press, New York, pp 466–503
Diamond A, Goldman-Rakic P (1989) Comparison of human infants and rhesus monkeys on Piaget’s AB task: evidence for dependence on dorsolateral prefrontal cortex. Exp Brain Res 74:24–40
Dilks DD, Cook P, Weiller SK, Berns HP, Spivak M, Berns GS (2015) Awake fMRI reveals a specialized region in dog temporal cortex for face processing. PeerJ 3:1115. https://doi.org/10.7717/peerj.1115
Dorey NR, Udell MAR, Wynne CDL (2009) Breed differences in dogs’ sensitivity to human points: a meta-analysis. Behav Proc 81(3):409–415
Dorey NR, Udell MAR, Wynne CDL (2010) When do domestic dogs, Canis familiaris, start to understand human pointing? The role of ontogeny in the development of interspecies communication. Anim Behav 79(1):37–41
Duckworth AL, Seligman ME (2005) Self-discipline outdoes IQ in predicting academic performance of adolescents. Psychol Sci 16(12):939–944
Duckworth AL, Tsukayama E, Kirby TA (2013) Is it really self-control? Examining the predictive power of the delay of gratification task. Perspect Soc Psychol Bull 39(7):843–855
Dunbar RI (1998) The social brain hypothesis. Brain 9(10):178–190
Engle RW (2002) Working memory as executive attention. Curr Dir Psychol Sci 11(1):19–23
Fagnani J, Barrera G, Carballo F, Bentosela M (2016) Is previous experience important for inhibitory control? A comparison between shelter and pet dogs in A-not-B and cylinder tasks. Anim Cogn 19(6):1165–1172
Fiset S, Beaulieu C, Landry F (2003) Duration of dogs’ (Canis familiaris) working memory in search for disappearing objects. Anim Cogn 6:1–10
Fuster JM, Alexander GE (1971) Neuron activity related to short-term memory. Science 173(3997):652–654
Garavan H, Ross TJ, Stein EA (1999) Right hemispheric dominance of inhibitory control: an event-related functional MRI study. Proc Natl Acad Sci 96(14):8301–8306
Gardner DM, Gerdes AC (2015) A review of peer relationships and friendships in youth with ADHD. J Atten Disord 19(10):844–855
Gazzaniga MS (2009) Neuroanatomy and development. In: Gazzaniga MS, Ivry RB, Mangun GR (eds) Cognitive Neuroscience: The Biology of the Mind. W. W. Norton & Company, New York, pp 59–109
Goldwater BC (1972) Psychological significance of pupillary movements. Psychol Bull 77(5):340–355
Gray DT, Smith AC, Burke SN, Gazzaley A, Barnes CA (2017) Attentional updating and monitoring and affective shifting are impacted independently by aging in macaque monkeys. Behav Brain Res 322:329–338
Hare B, Tomasello M (2005) Human-like social skills in dogs? Trends Cognit Sci 9(9):439–444
Hare B, Brown M, Williamson C, Tomasello M (2002) The domestication of social cognition in dogs. Science 298(5598):1634–1636
Hare B, Rosati A, Kaminski J, Bräuer J, Call J, Tomasello M (2009) The domestication hypothesis for dogs’ skills with human communication: a response to Udell et al. (2008) and Wynne et al. (2008). Anim Behav 79:e1–e6
Hasher L, Zacks RT (1979) Automatic and effortful processes in memory. J Exp Psychol Gen 108(3):356–388
Hecht J, Rice ES (2014) Citizen science in canine science. J Vet Behav 9(6):e15
Hecht J, Rice ES (2015) Citizen science: a new direction in canine behavior research. Behav Proc 110:125–132
Helton WS (2010) Does perceived trainability of dog (Canis lupus familiaris) breeds reflect differences in learning or differences in physical ability? Behav Proc 83:315–323
Hutchison KA (2007) Attentional control and the relatedness proportion effect in semantic priming. J Exp Psychol Learn Mem Cogn 33(4):645–662
Hutchison KA (2011) Attentional control and the relatedness proportion effect in semantic priming. J Exp Psychol Learn Mem Cogn 33(4):645–662
Hutchison KA, Bugg JM, Lim YB, Olsen MR (2016) Congruency precues moderate item-specific proportion congruency effects. Atten Percept Psychophys 78(4):1087–1103
Inzlicht M, Schmeichel BJ (2012) What is ego depletion? Toward a mechanistic revision of the resource model of self-control. Perspect Psychol Sci 7(5):450–463
James W (1890) The principles of psychology. Holt, New York
Jurado MB, Rosselli M (2007) The elusive nature of executive functions: a review of our current understanding. Neuropsychol Rev 17(3):213–233
Kane MJ, Engle RW (2000) Working-memory capacity, proactive interference, and divided attention: limits on long-term memory retrieval. J Exp Psychol Learn Mem Cogn 26(2):336–358
Kane MJ, Engle RW (2003) Working-memory capacity and the control of attention: the contributions of goal neglect, response competition, and task set to Stroop performance. J Exp Psychol Gen 132(1):47–70
Keppel G, Underwood BJ (1962) Proactive inhibition in short-term retention of single items. J Verbal Learn Verbal Behav 1(3):153–161
Kis A, Topál J, Gácsi M, Range F, Huber L, Miklósi Á, Virányi Z (2012) Does the A-not-B error in adult pet dogs indicate sensitivity to human communication? Anim Cogn 15(4):737–743
Kruska D (1988) Mammalian domestication and its effect on brain structure and behavior. Intelligence and evolutionary biology. Springer, Berlin, pp 211–250
Kubota K, Niki H (1971) Prefrontal cortical unit activity and delayed cortical unit activity and delayed alternation performance in monkeys. J Neurophysiol 34:337–347
Lai ZC, Moss MB, Killiany RJ, Rosene DL, Herndon JG (1995) Executive system dysfunction in the aged monkey: spatial and object reversal learning. Neurobiol Aging 16(6):947–954
Landsberg GM, Hunthausen W, Ackerman L (2003) The effects of aging on the behavior of senior pets. Handbook of Behavior Problems of the Dog and Cat, 2nd edn. WB Saunders, Philadelphia
Landsberg GM, Nichol J, Araujo JA (2012) Cognitive dysfunction syndrome: a disease of canine and feline brain aging. Vet Clin N Am Small Anim Pract 42:749–768
Lazarowski L, Foster ML, Gruen ME, Sherman BL, Case BC, Fish RE, Milgram NW, Dorman DC (2014) Acquisition of a visual discrimination and reversal learning task by Labrador retrievers. Anim Cogn 17(3):787–792
Lee CS, Therriault DJ (2013) The cognitive underpinnings of creative thought: a latent variable analysis exploring the roles of intelligence and working memory in three creative thinking processes. Intelligence 41:306–320
Lepper M, Kass PH, Hart LA (2002) Prediction of adoption versus euthanasia among dogs and cats in a California animal shelter. J Appl Anim Welfare Sci 5(1):29–42
Ley J, Bennett P, Coleman G (2008) Personality dimensions that emerge in companion canines. Appl Anim Behav Sci 110:305–317
Lockman JJ (1984) The development of detour ability during infancy. Child Dev 55(2):482–491
MacLean EL, Hare B, Nunn CL, Addessi E, Amici F, Anderson RC, Zhao Y et al (2014) The evolution of self-control. Proc Natl Acad Sci 111(20):E2140–E2148. https://doi.org/10.1073/pnas.13235333111
Marshall-Pescini S, Valsecchi P, Petak I, Accorsi PA, Previde EP (2008) Does training make you smarter? The effects of training on dogs’ performance (Canis familiaris) in a problem-solving task. Behav Proc 78:449–454
Marshall-Pescini S, Passalacqua C, Barnard S, Valsecchi P, Prato-Previde E (2009) Agility and search and rescue training differently affects pet dogs’ behaviour in socio-cognitive tasks. Behav Proc 81(3):416–422
Marshall-Pescini S, Virányi Z, Range F (2015) The effect of domestication on inhibitory control: wolves and dogs compared. PLoS ONE 10(2):e0118469
Marshall-Pescini S, Frazzi C, Valsecchi P (2016) The effect of training and breed group on problem-solving behaviours in dogs. Anim Cogn 19:571–579
Mech DL (2000) Leadership in wolf, Canis lupus, packs. Can Field Nat 114(2):259–263
Merritt M (2015) Dismantling standard cognitive science: it’s time the dog has its day. Biol Philos 30:811–829
Miklósi Á, Kubinyi E, Topál J, Gácsi M, Virányi Z, Csányi V (2003) A simple reason for a big difference: wolves do not look back at humans, but dogs do. Curr Biol 13(9):763–766
Milgram NW (2003) Cognitive experience and its effect on age-dependent cognitive decline in beagle dogs. Neurochem Res 28(11):1677–1682
Milgram NW, Zicker SC, Head E, Muggenburg BA, Murphey H, Ikeda-Douglas CJ, Cotman CW (2002) Dietary enrichment counteracts age-associated cognitive dysfunction in canines. Neurobiol Aging 23:737–745
Milgram NW, Head E, Zicker SC, Ikeda-Douglas CJ, Murphey H, Muggenburg B, Siwak C, Tapp D, Cotman CW (2005) Learning ability in aged beagle dogs is preserved by behavioral enrichment and dietary fortification: a two-year longitudinal study. Neurobiol Aging 26:77–90
Miller EK, Cohen JD (2001) An integrative theory of prefrontal cortex function. Ann Rev Neurosci 24:167–202
Miller EK, Erickson CA, Desimone R (1996) Neural mechanisms of visual working memory in prefrontal cortex of the macaque. J Neurosci 16(16):5154–5167
Miller HC, Pattison KF, DeWall CN, Rayburn-Reeves R, Zentall TR (2010) Self-control without a “self”? Common self-control processes in humans and dogs. Psychol Sci 21(4):534–538
Miller HC, DeWall CN, Pattison K, Molet M, Zentall TR (2012) Too dog tired to avoid danger: self- control depletion in canines increases behavioral approach toward an aggressive threat. Psychon Bull Rev 19:535–540
Mirkó E, Kubinyi E, Gácsi M, Miklósi Á (2012) Preliminary analysis of an adjective-based dog personality questionnaire developed to measure some aspects of personality in the domestic dog (Canis familiaris). Appl Anim Behav Sci 138:88–98
Mischel W, Shoda Y, Rodriguez M (1989) Delay of gratification in children. Science 244:933–938
Miyake A, Friedman NP, Emerson MJ, Witzki AH, Howerter A, Wager TD (2000) The unity and diversity of executive functions and their contributions to complex “frontal lobe” tasks: a latent variable analysis. Cogn Psychol 41:49–100
Moffitt TE, Arsenault L, Belsky D, Dickson N, Hancox RJ, Harrington HL, Caspi A (2011) A gradient of childhood self-control predicts health, wealth, and public safety. Proc Natl Acad Sci 108(7):2693–2698
Moffitt TE, Houts R, Asherson P, Belsky DW, Corcoran DL, Hammerle M, Caspi A (2015) Is adult ADHD a childhood-onset neurodevelopmental disorder? Evidence from a four-decade longitudinal cohort study. Am J Psychiatry 172:967–977
Mongillo P, Araujo JA, Pitteri E, Carnier P, Adamelli S, Regolin L, Marinelli L (2013) Spatial reversal learning is impaired by age in pet dogs. Age 35:2273–2282
Müller CA, Riemer S, Virányi Z, Huber L, Range F (2016) Inhibitory control, but not prolonged object- related experience appears to affect physical problem-solving performance of pet dogs. PLoS ONE 11(2):e0147753. https://doi.org/10.1371/journal.pone.0147753
Nigg JT (2001) Is ADHD a disinhibitory disorder? Psychol Bull 127(5):571–598
Norman DA, Shallice T (1986) Attention to action: willed and automatic control of behavior. In: Davidson R, Schwartz RG, Shapiro D (eds) Consciousness and self-regulation: advances in research and theory. Plenum Press, New York, pp 1–18
Partala T, Surakka V (2003) Pupil size variation as an indication of affective processing. Int J Hum Comput Stud 59:185–198
Peavler WS (1974) Pupil size, information overload, and performance differences. Psychophysiology 11(5):559–566
Pennington BF, Ozonoff S (1996) Executive functions and developmental psychopathology. J Child Psychol 37(1):51–87
Péter A, Gergely A, Topál J, Miklósi Á, Pongrácz P (2015) A simple but powerful test of perseverative search in dogs and toddlers. Q J Exp Psychol 68(5):940–951
Peterson LR, Peterson MJ (1959) Short-term retention of individual verbal items. J Exp Psychol 58(3):192–198
Piaget J (1954) The Construction of Reality in the Child. Basic, Oxford
Pongrácz P, Miklósi Á, Vida V, Csányi V (2005) The pet dog’s ability for learning from a human demonstrator in a detour task is independent from the breed and age. Appl Anim Behav Sci 90:309–323
Range F, Virányi Z (2015) Tracking the evolutionary origins of dog-human cooperation: the “Canine Cooperation Hypothesis”. Front Psychol 5:1582
Redick TS, Shipstead Z, Wiemers EA, Melby-Lervag M, Hulme C (2015) What’s working in working memory training? An educational perspective. Edu Psychol Rev 27(4):617–633
Riemer S, Mills DS, Wright H (2014) Impulsive for life? The nature of long-term impulsivity in domestic dogs. Anim Cogn 17(3):815–819
Rørvang MV, Ahrendt LP, Christensen JW (2015) Horses fail to use social learning when solving spatial detour tasks. Anim Cogn 18(4):847–854
Rossi A, Smedema D, Parada FJ, Allen C (2014) Visual attention in dogs and the evolution of non-verbal communication. In: Horowitz A (ed) Domestic dog cognition and behavior. Springer Verlag, Berlin, pp 133–154
Rueda MR, Rothbart MK, McCandliss BD, Saccomanno L, Posner MI (2005) Training, maturation, and genetic influences on the development of executive function. Proc Natl Acad Sci USA 102(41):14931–14936
Ruehl WW, Bruyette DS, DePaoli A, Cotman CW, Head E, Milgram NW, Cummings BJ (1995) Canine cognitive dysfunction as a model for human age-related cognitive decline, dementia and Alzheimer’s disease: clinical presentation, cognitive testing, pathology and response to 1-deprenyl therapy. Prog Brain Res 106:217–225
Salman MD, New JG Jr, Scarlett JM, Kass PH, Ruch-Gallie R, Hetts S (1998) Human and animal factors related to relinquishment of dogs and cats in 12 selected animal shelters in the United States. J Appl Anim Welfare Sci 1(3):207–226
Salman MD, Hutchison J, Ruch-Gallie R, Kogan L, New JC, Hass PH, Scarlett JM (2000) Behavioral reasons for relinquishment of dogs and cats to 12 shelters. J Appl Anim Welfare Sci 3(2):93–106
Schleidt WM, Shalter MD (2003) Co-evolution of humans and canids: an alternative view of dog domestication: homo homini lupus? Evol Cognit 9(1):57–72
Schneider W, Shiffrin RM (1977) Controlled and automatic human information processing: detection, search, and attention. Psychol Rev 84(1):1–66
Shipstead Z, Lindsey DRB, Marshall RL, Engle RW (2014) The mechanisms of working memory capacity: primary memory, secondary memory, and attention control. J Mem Lang 72:116–141
Smith BP, Litchfield CA (2010) How well do dingoes, Canis dingo, perform on the detour task? Anim Behav 80:155–162
Somppi S, Tӧrnqvist H, Hänninen L, Krause C, Vainio O (2012) Dogs do look at images: eye tracking in canine cognition research. Anim Cogn 15:163–174
Somppi S, Tӧrnqvist H, Hänninen L, Krause CM, Vainio O (2014) How dogs scan familiar and inverted faces: an eye movement study. Anim Cogn 17:793–803
Somppi S, Tӧrnqvist H, Kujala MV, Hänninen L, Krause CM, Vainio O (2016) Dogs evaluate threatening facial expressions by their biological validity – evidence from gazing patterns. PLoS ONE 11(1):e0143047. https://doi.org/10.1371/journal.pone.0143047
Somppi S, Tӧrnqvist H, Topál J, Koskela A, Hänninen L, Krause CM, Vainio O (2017) Nasal oxytocin treatment biases dogs’ visual attention and emotional response toward positive human facial expressions. Front Psychol 8:1854. https://doi.org/10.3380/fpsyg.2017.01854
Spieler DH, Balota DA, Faust ME (1996) Stroop performance in healthy younger and older adults and in individuals with dementia of the Alzheimer’s type. J Exp Psychol Hum Percept Perform 22(2):461–479
Sprague J, Verona E, Kalkhoff W, Kilmer A (2011) Moderators and mediators of the stress-aggression relationship: executive function and state anger. Emotion 11(1):61–73
Stewart L, MacLean EL, Ivy D, Woods V, Cohen E, Hare B (2015) Citizen science as a new tool in dog cognition research. PLoS ONE 10(9):e0135176. https://doi.org/10.1371/journal.pone.0135176
Sümegi Z, Kis A, Miklósi Á, Topál J (2014) Why do adult dogs (Canis familiaris) commit the A-not-B search error? J Comp Psychol 128(1):21
Tangney JP, Baumeister RF, Boone AL (2004) High self-control predicts good adjustment, less pathology, better grades, and interpersonal success. J Pers 72(2):271–324
Tapp PD, Siwak CT, Estrada J, Head E, Muggenburg BA, Cotman CW, Milgram NW (2003) Size and reversal learning in the beagle dog as a measure of executive function and inhibitory control in aging. Learn Memory 10:64–73
Téglás E, Gergely A, Kupán K, Miklósi Á, Topál J (2012) Dogs’ gaze following is tuned to human communicative signals. Curr Biol 22(3):209–212
Thompkins AM, Deshpande G, Waggoner P, Katz JS (2016) Functional magnetic resonance imaging of the domestic dog: research, methodology, and conceptual issues. Comp Cognit Behav Rev 11:63–82
Tinbergen N (1963) On aims and methods of ethology. Zeitschrift für Tierpsychologie 20:410–433
Topál J, Gergely G, Miklósi Á, Erdӧhegyi Á, Csibra G (2008) Infants’ perseverative search errors are induced by pragmatic misinterpretation. Science 321(5897):1831–1834
Topál J, Gergely G, Erdӧhegyi Á, Csibra G, Miklósi Á (2009) Differential sensitivity to human communication in dogs, wolves, and human infants. Science 325(5945):1269–1272
Topál J, Kis A, Oláh K (2014) Dogs’ sensitivity to human ostensive cues: a unique adaptation. The social dog: behavior and cognition 319–346
Tóth L, Gácsi M, Miklósi Á, Bogner P, Repa I (2009) Awake dog brain magnetic resonance imaging. J Vet Behav 4(2):50
Turner ML, Engle RW (1989) Is working memory capacity task dependent? J Mem Lang 28:127–154
Udell MAR, Dorey NR, Wynne CDL (2008) Wolves outperform dogs in following human social cues. Anim Behav 76(6):1767–1773
Udell MAR, Dorey NR, Wynne CDL (2010a) What did domestication do to dogs? A new account of dogs’ sensitivity to human actions. Biol Rev 85:327–345
Udell MAR, Dorey NR, Wynne CDL (2010b) The performance of stray dogs (Canis familiaris) living in a shelter on human-guided object-choice tasks. Anim Behav 79(3):717–725
Udell MA, Ewald M, Dorey NR, Wynne CD (2014) Exploring breed differences in dogs (Canis familiaris): does exaggeration or inhibition of predatory response predict performance on human-guided tasks? Anim Behav 89:99–105
Unsworth N, Engle RW (2007) The nature of individual differences in working memory capacity: active maintenance in primary memory and controlled search from secondary memory. Psychol Rev 114(1):104–132
Unsworth N, Fukuda K, Awh E, Vogel EK (2014) Working memory and fluid intelligence: capacity, attention, control, and secondary memory retrieval. Cogn Psychol 71:1–26
Uzgiris IC, Hunt JM (1975) Assessment in infancy: Ordinal scales of psychological development. University of Illinois Press, Champaign
Vás J, Topál J, Péch É, Miklósi Á (2007) Measuring attention deficit and activity in dogs: a new application and validation of a human ADHD questionnaire. Appl Anim Behav Sci 103:105–117
Virányi Z, Gácsi M, Kubinyi E, Topál J, Belényi B, Ujfalussy D, Miklósi Á (2008) Comprehension of human pointing gestures in young human-reared wolves (Canis lupus) and dogs (Canis familiaris). Anim Cogn 11:373–387
Wallis JD, Dias R, Robbins TW, Roberts AC (2001) Dissociable contributions of the orbitofrontal and lateral prefrontal cortex of the marmoset to performance on a detour reaching task. Eur J Neurosci 13(9):1797–1808
Wallis LJ, Range F, Müller CA, Serisier S, Huber L, Virányi Z (2014) Lifespan development of attentiveness in domestic dogs: drawing parallels with humans. Frontiers in Psychology 5(71):1–13
Wallis LJ, Virányi Z, Müller CA, Serisier S, Huber L, Range F (2016) Aging effects on discrimination learning, logical reasoning and memory in pet dogs. Age 38(1):6
Williams FJ, Mills DS, Guo K (2011) Development of a head-mounted, eye-tracking system for dog. J Neurosci Methods 194(2):259–265
Wright HF, Mills DS, Pollux PMJ (2011) Development and validation of a psychometric tool for assessing impulsivity in the domestic dog Canis familiaris. Int J Comp Psychol 24:210–225
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The author declares that she has no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by the author.
Rights and permissions
About this article
Cite this article
Olsen, M.R. A case for methodological overhaul and increased study of executive function in the domestic dog (Canis lupus familiaris). Anim Cogn 21, 175–195 (2018). https://doi.org/10.1007/s10071-018-1162-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10071-018-1162-6