Abstract
Background
Minimally invasive esophagectomy (MIE) has been shown to be a feasible technique for the treatment of esophageal cancer; however, its postoperative morbidity remains high. This retrospective study aimed to evaluate the effect of postoperative complications on long-term outcomes in patients who have undergone MIE for esophageal squamous cell carcinoma (ESCC).
Methods
This retrospective study enrolled patients who had undergone MIE for ESCC between September 2009 and November 2014; all procedures were performed by a single surgical team. Relevant patient characteristics and postoperative variables were collected and evaluated. The disease-free survival (DFS) and disease-specific survival (DSS) were determined by the Kaplan–Meier method, and compared by log-rank tests. Possible predictors of survival were subjected to univariate analysis and multivariate Cox proportional hazard regression analysis.
Results
In all, data on 214 patients with ESCC were analyzed, including 170 men and 44 women. All study subjects had undergone thoracoscopic or thoracoscopic–laparoscopic esophagectomy and cervical esophagogastric anastomosis. One hundred and thirty patients (60.7%) had postoperative complications (Grades 1–4). The overall DFS and DSS rates were 80.0 and 88.9% at 1 year, 48.6 and 54.2% at 3 years, and 43.2 and 43.5% at 5 years, respectively. Univariate analysis and multivariate Cox proportional hazard regression analysis showed that T stage, N stage, and tumor grade were independent prognostic factors for long-term survival; however, postoperative complications had no significant effect on the DFS or DSS of this patient cohort (log-rank test, p = 0.354 and 0.160, respectively).
Conclusions
Postoperative complications have no significant effect on long-term survival in patients who have undergone MIE for ESCC.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Esophageal cancer (EC) is one of the deadliest and most aggressive cancers. It is the sixth most common cause of cancer deaths in men and the ninth most common in women globally [1]. In China, EC mainly occurs in the form of esophageal squamous cell carcinoma (ESCC), which accounts for ~90% of all cases of EC [2]. Surgical resection is the mainstay of treatment aimed at prolonging survival and achieving definitive cure [3].
The two most common minimally invasive esophagectomy (MIE) procedures, combined thoracoscopic and laparoscopic esophagectomy and combined thoracoscopic and laparotomy esophagectomy, were developed to reduce the trauma resulting from surgical treatment of esophageal cancer. The established benefits of MIE over open esophagectomy include less estimated blood loss, more lymph nodes resected, and shorter length of hospital stay [4,5,6]. This procedure is also associated with superior overall survival and perioperative mortality and fewer severe postoperative complications [7]. However, MIE remains a complex procedure with some risk of postoperative morbidity and mortality.
The effect of postoperative complications on long-term survival of EC patients who have undergone esophagectomy is currently controversial. Several studies have reported that major perioperative morbidity does not affect long-term survival of patients undergoing esophagectomy for cancer of the esophagus or gastroesophageal junction [8, 9], whereas Lerut et al. [10] reported a strong correlation between severity of complications and time to tumor recurrence. In a recent study, Luc et al. [11] found a significant association between occurrence of complications and shorter time interval until death owing to recurrence. However, most of these studies included both patients with ESCC and esophageal adenocarcinoma (EAC); additionally, the surgical procedures varied. Therefore, evaluation of the impact of postoperative complications on long-term survival has been difficult. The aim of this study was to explore the impact of postoperative complications on the long-term survival of patients who had undergone MIE for ESCC in a single institution.
Patients and methods
Patients
From September 2009 to November 2014, 249 thoracoscopic esophagectomies for ESCC of the thoracic esophagus were performed in the Department of Thoracic Surgery, Daping Hospital, Third Military Medical University (Chongqing, China). Exclusion criteria were as follows: loss to follow-up, non-curative (R1 or R2) resection (tumor-free margin <1 mm), postoperative death, and death from causes unrelated to esophageal cancer. All surgeries were performed by the same surgeon (W.G.). Data were collected retrospectively by chart review and relevant patient characteristics and intraoperative and postoperative variables analyzed. Preoperative staging was performed by endoscopic ultrasound and thoracoabdominal computed tomography scanning. Postoperative TNM stage was classified in accordance with the 7th edition of the American Joint Committee on Cancer staging protocol [12]. Postoperative death was defined as death within 30 days after surgery or before discharge from hospital. The study patients had given written consent to the operative procedures, and the study was approved by the Ethics Committee of Daping Hospital.
Operative procedures
All operations were performed by three-stage combined thoracoscopic and laparoscopic esophagectomy or combined thoracoscopic and laparotomy esophagectomy; 228 cases were placed in the left lateral position and 21 cases in the prone position, all operations were performed under general anesthesia and double-lumen intubation. These procedures have been described previously [13,14,15]. Briefly, the mediastinal pleura overlying the anterior aspect of the esophagus was cauterized and the esophagus mobilized away from the hilum and pericardium. Mobilization extended to the level of the azygos vein, which was skeletonized and ligated with 10-mm ligaclips. Using electrocautery, the parietal pleura posterior to the esophagus was opened from the level of the azygos vein to the crus. After dissecting the esophagus and mediastinal lymph nodes, the thoracic duct was routinely mass ligated immediately above the diaphragmatic hiatus [12]. For the second stage, the patient was placed in a supine position and laparotomy or laparoscopy performed. Following dissection of the pericardiac and celiac nodes, the stomach was prepared for reconstruction by dividing it at the cardia with a stapler. In the third stage, the cervical esophagus was dissected through a left oblique incision and the stomach pulled up through the posterior mediastinum. The procedure was completed with a cervical esophagogastric anastomosis.
Postoperative complications and classification of patient groups
Postoperative surgical and medical complications were graded according to a modification of the Clavien–Dindo classification [16] as follows: Grade 0, no complications; Grade 1, deviation from normal postoperative course without need for medical intervention; Grade 2, complications requiring pharmacological treatment; Grade 3, complications requiring invasive or radiological intervention; Grade 3a, does not require general anesthesia; Grade 3b, requires general anesthesia; Grade 4, life-threatening complications requiring intensive care unit management; and Grade 5, death. Patients who developed two or more than grades of postoperative complications were classified as having the more serious grade. The patients were divided into the following three groups: Group 1 (Grade 0 complications), Group 2 (Grades 1–3a), and Group 3 (Grades 3b–4b).
Follow-up
After surgery, all patients were followed up regularly in the outpatient clinic every 3 months during the first year, every 6 months until the fifth year, and then annually. All included subjects had complete follow-up information until death or November 2014. The duration of follow-up ranged between 2.7 and 63.9 months (mean 26.3 ± 15.0 months). At follow-up visits, specific examinations such as barium swallow, magnetic resonance imaging, biopsy, and positron emission tomography were performed as indicated. Recurrence was diagnosed based on pathologic or radiologic findings and classified as locoregional recurrence, distant metastasis, or death from ESCC. Disease-specific survival (DSS) was defined as survival from operation to death from ESCC.
Statistical analysis
Numerical data are expressed as the mean ± standard deviation. Continuous variables were compared by using χ2 test and quantitative variables by one-way analysis of variance. Life table analysis was used to calculate the 1-, 2-, 3-, 4-, and 5-year survival rates. Patient survival curves were constructed by the Kaplan–Meier method. The log-rank test was used to compare survival difference between groups for each variable, and multivariate Cox proportional hazard regression analysis were used to identify potential prognostic factors. All statistical calculations were performed using SPSS statistical software, version 19.0 (IBM SPSS, Chicago, IL, USA). A p value of <0.05 was considered statistically significant.
Results
During the study period, 249 video-assisted thoracoscopic esophagectomies for squamous cell carcinoma of the thoracic esophagus were performed by the same surgeon (W.G.). Twenty-four of these cases were lost to follow-up (24/249, 9.64%), two suicided postoperatively, three had undergone R1 or R2 resection, one died from oropharyngeal cancer, and one died from asphyxia for an unknown reason in the 51st day after operation. Furthermore, four patients who died within the first 30 postoperative days were also excluded because our aim was to analyze the effect of complications on long-term outcomes. Exclusion of the above-listed patients left 214 consecutive patients in this study. The overall 30-day mortality after surgery was 1.6% (4/249).
Among the 214 study patients, 170 were men, the sex ratio being 3.86 men to 1 woman (Table 1 ). The average age was 60.2 ± 8.1 years (range, 37–79 years). One hundred and fifty patients (70.1%) had a smoking history and 64 (29.9%) were never-smokers. The average operative time was 281.0 ± 64.6 min, and the mean postoperative hospital stay was 21.3 ± 12.2 days. There were significant differences in average operative time and mean postoperation hospital stay among the different groups (p = 0.049 and 0.000, respectively). Patient characteristics (TNM classification, lung function, intraoperative blood loss) did not differ significantly between the groups.
The complications encountered are shown in detail in Table 2. Eighty-four patients (39.3%) had an unremarkable postoperative course (Grade 0, Group 1). The majority of patients (130/214, 60.7%) did have postoperative complications (Grades 1–4). As described in the Materials and Methods section, these patients were subdivided into two groups: Group 2 (Grade 1–3a, n = 110) and Group 3 (Grade 3b–4b, n = 20). The most common complications were anastomotic leakage (9.3%, 20/214), pneumothorax (8.9%, 19/214), pleural infusion (8.9%, 19/214), pneumonia (7.9%, 17/214), adult respiratory distress syndrome (5.6%, 12/214), and chylothorax (3.7%, 8/214).
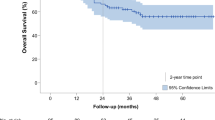
Seventy-two of the 214 study patients (33.6%) had died from ESCC, whereas 142 (66.4%) remained alive at the end of the study period. Ten of the 142 survivors had locoregional recurrence or distant metastasis at the end of the follow-up period. In this study, the disease-free survival (DFS) and disease-specific survival (DSS) were 80.0 and 88.9% at 1 year, 56.1 and 67.7% at 2 years, 48.6 and 54.2% at 3 years, 43.2 and 51.1% at 4 years, and 43.2 and 43.5% at 5 years after surgery, respectively (Fig. 1). The mean DFS and DSS times were 37.6 ± 2.0 and 41.7 ± 1.9 months, respectively.
Univariate analysis indicated that sex (p = 0.254), smoking history (p = 0.618), tumor location (p = 0.254), and grade of complication (p = 0.160) did not significantly influence survival, whereas tumor grade (p = 0.007), T stage (p = 0.002), and N stage (p = 0.025) were significantly associated with survival. According to multivariate Cox proportional hazard regression analysis, T stage (p = 0.002), N stage (p = 0.014), tumor grade (p = 0.021), and location (p = 0.033) were all significantly correlated with patient survival (Table 3). There were no significant differences in DFS and DSS among Groups 1–3 (p = 0.354 and 0.160, respectively) (Fig. 2).
Discussion
Previous studies have demonstrated that thoracoscopic esophagectomy is technically feasible, surgically safe, and oncologically adequate, with potentially faster postoperative recovery, reduced trauma, and shorter operative time than open surgery [5,6,7]. In a recent, multicenter randomized controlled trial, Biere et al. [17] provided evidence for the short-term benefits of MIE in patients with resectable esophageal cancer. Another study in which Ninomiya et al. [18] reviewed their 10-year experience also reported favorable oncologic outcomes of thoracoscopic esophagectomy with extended lymph node dissection in the left lateral position. However, the incidence of postoperative complications after MIE remains high, reportedly ranging from 10 to 57.8% [5,6,7, 9, 11, 17,18,19,20].
In our study, the overall 30-day mortality after surgery was 1.6% (4/249). The postoperative complication rate was 60.7% (130/214), the majority being minor complications (Group 2, 51.4%, 110/214) such as fever, hoarseness, and gastric motor disorders. The incidence of severe complications was 9.3% (20/214), which is clearly lower than that reported by others (15–30%) [9, 11]. Possible explanations for these apparent discrepancies include differences in surgical procedures and pathological types of cancer. Furthermore, we modified the Clavien classification [21] of postoperative complications according to the therapy needed to address the complication. For example, the main complication of anastomotic leakage was classified as Grade 1, whereas fistulas with local infection requiring antibiotics were classified as Grade 2. Empyema caused by anastomotic leakage requiring treatment by closed thoracic drainage was classified as Grade 3a. Thus, the same complication could be allotted different grades according to the therapy required.
In our study, the 5-year DFS and DSS rates were 43.2 and 43.5%, respectively. Until now, only a few case–control studies have reported long-term survival of patients who have undergone MIE. However, several studies reporting stage-specific survival following MIE have concluded that this procedure also achieves excellent long-term outcomes. For example, Smithers et al. [22] reported the following 5-year stage-specific survival rates after MIE: 85% for Stage I, 33% for Stage IIA, 37% for Stage IIB, and 16% for Stage III (TNM classification, 6th edition). In their non-randomized study, Burdall et al. [23] found significantly better survival following minimally invasive procedures than after open esophagectomy. Ninomiya et al. [18] also reported favorable oncologic outcomes of thoracoscopic esophagectomy with extended lymph node dissection. In their study, the overall 5-year survival rates for Stage I, II, and III disease after curative video-assisted thoracoscopic esophagectomy were 79.1, 77.9, and 56.7%, respectively. In the current study, the follow-up protocol included re-examination of all patients every 3 months; 249 patients attended for these regular follow-ups, whereas 24 were lost to follow-up. During the study period, 72 patients died. No port-site recurrences were observed. The DSS rates were 88.9% at 1 year, 54.2% at 3 years, and 43.5% at 5 years, whereas the DFS rates were 80.0% at 1 year, 48.6% at 3 years, and 43.2% at 5 years, indicating that MIE can achieve excellent long-term outcomes in patients with ESCC. Moreover, considering the proportion of T3 and T4a lesions in our series (131/214, 61.2%), we believe that MIE achieves satisfactory long-term results in patients with locally advanced esophageal cancer.
Univariate and multivariate Cox proportional hazard regression analyses identified T stage, N stage, and tumor grade as independent prognostic factors for long-term survival, which is in accordance with a previous study [13]. Notably, although patients in Group 1 (no complications) and Group 2 (minor complications) had shorter operative times and postoperative hospital stays than patients in Group 3 (severe complications), postoperative complications had no significant effect on the DFS and DSS of our patients (log-rank test, p = 0.354 and 0.160, respectively). Moreover, multivariate Cox proportional hazard regression analysis also showed that the grade of complications was not an independent prognostic factor in these patients (p = 0.211), confirming that postoperative complications did not affect the long-term survival.
There were several studies have investigated the possible effect of postoperative complications on survival of patients underwent esophagectomy [8,9,10,11]. The results from Lindner [8] and Xia [9] demonstrated that major perioperative morbidity had no obvious effect on long-term survival, whereas Lerut [10] and Luc [11] found that there was significant correlation between complications and time to tumor recurrence. Notably, these studies enrolled patients with ESCC and EAC, and the surgical procedures applied in these studies also varied. Therefore, evaluating the actual impact of postoperative complications on long-term survival was difficult, and the conclusions of these studies might lack conviction. In our series, we enrolled patients with ESCC and the surgical procedure applied was uniform, thus making the results and conclusion persuasive. Up to date, there was still few study mentioned the effect of postoperative complications on the long-term survival after MIE, so we believed that our study was helpful to understand the potential effect of complications on survival of patients underwent MIE.
As for the reason why major postoperative complications could not have clinical impact on the long-term outcomes, we speculated that major postoperative complications may have impact on short-time outcome by different paths like systemic inflammatory response and stress. According to previous study, inflammatory oncotaxis, the cytokines in response to stress, with the resulting inflammation, were associated with infection causing a hormonal milieu more conducive to the regrowth of cancer cells [24]. However, the effect of MIE on palliating the systemic inflammatory and catabolic response after surgical trauma has been demonstrated [25]. Therefore, we speculated that major postoperative complications may have impact on short-time outcome by different paths such as systemic inflammatory response and stress, while after the short-time effect was extincted, the long-term survival was not affected. In fact, the effect and potential paths of different postoperative complications on short-time and long-term outcome after MIE were the focus of our further research.
Although our sample was from a single institution and was relatively small compared with the worldwide collaborative esophageal cancer database, the database in our department and patients’ follow-up system were well established, and the surgical procedures, pathologic examinations, and patient follow-up were highly uniform throughout the entire study period. Additionally, all patients enrolled in this study had ESCC, and the operations were performed by a single surgeon using a uniform surgical approach. Therefore, we believe that our results are robust and valid. Of course, further studies with larger cohorts and longer follow-up are needed to validate our results.
In conclusion, MIE is a complex procedure with potential postoperative morbidities. Postoperative complications in patients who have undergone MIE for ESCC may have an impact on short-time survival and quality of life, but do not significantly affect long-term survival.
References
Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A (2015) Global cancer statistics, 2012. CA Cancer J Clin 65:87–108
Tran GD, Sun XD, Abnet CC, Fan JH, Dawsey SM, Dong ZW, Mark SD, Qiao YL, Taylor PR (2005) Prospective study of risk factors for esophageal and gastric cancers in the Linxian general population trial cohort in China. Int J Cancer 113:456–463
Ajani JA (2013) NCCN clinical practice guidelines in oncology, esophageal and esophagogastric junction cancers, v.2.2013. http://www.nccn.org. Accessed 23 July 2013
Cuschieri A, Shimi S, Banting S (1992) Endoscopic oesophagectomy through a right thoracoscopic approach. J R Coll Surg Edinb 37:7–11
Dolan JP, Kaur T, Diggs BS, Luna RA, Schipper PH, Tieu BH, Sheppard BC, Hunter JG (2013) Impact of comorbidity on outcomes and overall survival after open and minimally invasive esophagectomy for locally advanced esophageal cancer. Surg Endosc 27:4094–4103
Osugi H, Takemura M, Higashino M, Takada N, Lee S, Kinoshita H (2003) A comparison of video-assisted thoracoscopic oesophagectomy and radical lymph node dissection for squamous cell cancer of the oesophagus with open operation. Br J Surg 90:108–113
Palazzo F, Rosato EL, Chaudhary A, Evans NR, Sendecki JA, Keith S, Chojnacki KA, Yeo CJ, Berger AC (2015) Minimally invasive esophagectomy provides significant survival advantage compared with open or hybrid esophagectomy for patients with cancers of the esophagus and gastroesophageal junction. J Am Coll Surg 220:672–679
Lindner K, Fritz M, Haane C, Senninger N, Palmes D, Hummel R (2014) Postoperative complications do not affect long-term outcome in esophageal cancer patients. World J Surg 38:2652–2661
Xia BT, Rosato EL, Chojnacki KA, Crawford AG, Wekster B, Berger AC (2013) Major perioperative morbidity does not affect long-term survival in patients undergoing esophagectomy for cancer of the esophagus or gastroesophageal junction. World J Surg 37:408–415
Lerut T, Moons J, Coosemans W, Van Raemdonck D, De Leyn P, Decaluwé H, Decker G, Nafteux P (2009) Postoperative complications after transthoracic esophagectomy for cancer of the esophagus and gastroesophageal junction are correlated with early cancer recurrence: role of systematic grading of complications using the modified Clavien classification. Ann Surg 250:798–807
Luc G, Durand M, Chiche L, Collet D (2015) Major post-operative complications predict long-term survival after esophagectomy in patients with adenocarcinoma of the esophagus. World J Surg 39:216–222
Rice TW, Blackstone EH, Rusch VW (2010) 7th edition of the AJCC Cancer staging manual: esophagus and esophagogastric junction. Ann Surg Oncol 17:1721–1724
Guo W, Zhao YP, Jiang YG, Niu HJ, Liu XH, Ma Z, Wang RW (2012) Prevention of postoperative chylothoraxwith thoracic duct ligation during video-assisted thoracoscopic esophagectomy for cancer. Surg Endosc 26:1332–1336
Deng B, Tan QY, Jiang YG, Zhao YP, Zhou JH, Chen GC, Wang RW (2010) Prevention of early delayed gastric emptying after high-level esophagogastrostomy by pyloric digital fracture. World J Surg 34:2837–2843
Guo W, Zou YB, Ma Z, Niu HJ, Jiang YG, Zhao YP, Gong TQ, Wang RW (2013) One surgeon’s learning curve for video-assisted thoracoscopic esophagectomy for esophageal cancer with the patient in lateral positon: how many cases are needed to reach competence? Surg Endosc 27:1346–1352
Dindo D, Demartines N, Clavien PA (2004) Classification of surgical complications a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 240:205–213
Biere SS, van Berge Henegouwen MI, Maas KW, Bonavina L, Rosman C, Garcia JR, Gisbertz SS, Klinkenbijl JH, Hollmann MW, de Lange ES, Bonjer HJ, van der Peet DL, Cuesta MA (2012) Minimally invasive versus open oesophagectomy for patients with oesophageal cancer: a multicentre, open-label, randomised controlled trial. Lancet 379:1887–1892
Ninomiya I, Okamoto K, Fujimura T, Fushida S, Osugi H, Ohta T (2014) Oncologic outcomes of thoracoscopic esophagectomy with extended lymph node dissection: 10-year experience from a single center. World J Surg 38:120–130
Puntambekar SP, Agarwal GA, Joshi SN, Rayate NV, Sathe RM, Patil AM (2010) Thoracolaparoscopy in the lateral position for esophageal cancer: the experience of a single institution with 112 consecutive patients. Surg Endosc 24:2407–2414
Wang H, Feng M, Tan L, Wang Q (2010) Comparison of the short-term quality of life in patients with esophageal cancer after subtotal esophagectomy via video-assisted thoracoscopic or open surgery. Dis Esophagus 23:408–414
Dindo D, Clavien PA (2008) What is a surgical complication? World J Surg 32:939–941
Smithers BM, Gotley DC, Martin I, Thomas JM (2007) Comparison of the outcomes between open and minimally invasive esophagectomy. Ann Surg 245:232–240
Burdall OC, Boddy AP, Fullick J, Blazeby J, Krysztopik R, Streets C, Hollowood A, Barham CP, Titcomb D (2015) A comparative study of survival after minimally invasive and open oesophagectomy. Surg Endosc 29:431–437
DerHagopian RP, Sugarbaker EV, Ketcham A (1978) Inflammatory oncotaxis. JAMA 240:374–375
Scarpa M, Cavallin F, Saadeh LM, Pinto E, Alfieri R, Cagol M, Da Roit A, Pizzolato E, Noaro G, Pozza G, Castoro C (2015) Hybrid minimally invasive esophagectomy for cancer: impact on postoperative inflammatory and nutritional status. Dis Esophagus. doi:10.1111/dote.12418
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
Kun-Kun Li, Yin-Jian Wang, Xue-Hai Liu, Qun-You Tan, Yao-Guang Jiang, and Wei Guo have no conflict of interest or financial ties to disclose.
Rights and permissions
About this article
Cite this article
Li, KK., Wang, YJ., Liu, XH. et al. The effect of postoperative complications on survival of patients after minimally invasive esophagectomy for esophageal cancer. Surg Endosc 31, 3475–3482 (2017). https://doi.org/10.1007/s00464-016-5372-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00464-016-5372-1