Abstract
Purpose
Sarcomas are rare and heterogenic tumors with unclear etiology. They develop in bone and connective tissue, mainly in pediatric patients. To increase efficacy of current therapeutic options, natural products showing selective toxicity to tumor cells are extensively investigated. Here, we evaluated antitumor activity of bacterial pigment violacein in osteosarcoma (OS) and rhabdomyosarcoma (RMS) cell lines.
Methods
The toxicity of violacein was assessed in vitro and in vivo, using MTT assay and FET test. The effect of violacein on cell migration was monitored by wound healing assay, cell death by flow cytometry, uptake of violacein by fluorescence microscopy, generation of reactive oxygen species (ROS) by DCFH-DA assay and lipid peroxidation by TBARS assay.
Results
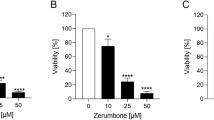
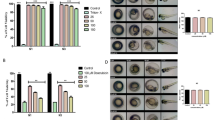
Violacein IC50 values for OS and RMS cells were in a range from 0.35 to 0.88 µM. Its selectivity toward malignant phenotype was confirmed on non-cancer V79-4 cells, and it was safe in vivo, for zebrafish embryos in doses up to 1 µM. Violacein induced apoptosis and affected the migratory potential of OS and RMS cells. It was found on the surfaces of tested cells. Regarding the mechanism of action, violacein acted on OS and RMS cells independently of oxidative signaling, as judged by no increase in intracellular ROS generation and no lipid peroxidation.
Conclusion
Our study provided further evidence that reinforces the potential of violacein as an anticancer agent and candidate to consider for improvement of the effectiveness of traditional OS and RMS therapies.






Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Angori S, Capanni C, Faulkner G, Bean C, Boriani G, Lattanzi G, Cenni V (2017) Emery-dreifuss muscular dystrophy-associated mutant forms of lamin a recruit the stress responsive protein ankrd2 into the nucleus, affecting the cellular response to oxidative stress. Cell Physiol Biochem 42:169–184. https://doi.org/10.1159/000477309
Aoyama H, Melo PS, Granjeiro PA, Haun M, Ferreira CV (2000) Cytotoxicity of okadaic acid and kinetic characterization of protein tyrosine phosphatase activity in V79 fibroblasts. Pharm Pharmacol Commun 6:331–334. https://doi.org/10.1211/146080800128736150
Ashkenazi A (2015) Targeting the extrinsic apoptotic pathway in cancer: lessons learned and future directions. J Clin Invest 125:487–489. https://doi.org/10.1172/JCI80420
Bertin G, Averbeck D (2006) Cadmium: cellular effects, modifications of biomolecules, modulation of DNA repair and genotoxic consequences (a review). Biochimie 88:1549–1559. https://doi.org/10.1016/j.biochi.2006.10.001
Borinstein SC, Steppan D, Hayashi M, Loeb DM, Isakoff MS, Binitie O, Brohl AS, Bridge JA, Stavas M, Shinohara ET, Meyer WH, Reed DR, Wagner LM (2018) Consensus and controversies regarding the treatment of rhabdomyosarcoma. Pediatr Blood Cancer. https://doi.org/10.1002/pbc.26809
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1006/abio.1976.9999
Bromberg N, Justo GZ, Haun M, Durán N, Ferreira CV (2005) Violacein cytotoxicity on human blood lymphocytes and effect on phosphatases. J Enzyme Inhib Med Chem 20:449–454. https://doi.org/10.1080/14756360500273052
Bromberg N, Dreyfuss JL, Regatieri CV, Palladino MV, Durán N, Nader HB, Haun M, Justo GZ (2010) Growth inhibition and pro-apoptotic activity of violacein in Ehrlich ascites tumor. Chem Biol Interact 186:43–52. https://doi.org/10.1016/j.cbi.2010.04.016
Cauz ACG, Carretero GPB, Saraiva GKV, Park P, Mortara L, Cuccovia IM, Brocchi M, Gueiros-Filho FJ (2019) Violacein targets the cytoplasmic membrane of bacteria. ACS Infect Dis 5:539–549. https://doi.org/10.1021/acsinfecdis.8b00245
Chaung W, Mi LJ, Boorstein RJ (1997) The p53 status of Chinese hamster V79 cells frequently used for studies on DNA damage and DNA repair. Nucleic Acids Res 25:992–994. https://doi.org/10.1093/nar/25.5.992
Choi SY, Yoon KH, Lee JI, Mitchell RJ (2015) Violacein: properties and production of a versatile bacterial pigment. Biomed Res Int. https://doi.org/10.1155/2015/465056
Choi SY, Im H, Mitchell RJ (2017) Violacein and bacterial predation: promising alternatives for priority multidrug resistant human pathogens. Future Microbiol 12:835–838. https://doi.org/10.2217/fmb-2017-0090
Choi SY, Lim S, Cho G, Kwon J, Mun W, Im H, Mitchell RJ (2020) Chromobacterium violaceum delivers violacein, a hydrophobic antibiotic, to other microbes in membrane vesicles. Environ Microbiol 22:705–713. https://doi.org/10.1111/1462-2920.14888
de Carvalho DD, Costa FT, Duran N, Haun M (2006) Cytotoxic activity of violacein in human colon cancer cells. Toxicol in Vitro 20:1514–1521. https://doi.org/10.1016/j.tiv.2006.06.007
Durán N, Justo GZ, Ferreira CV, Melo PS, Cordi L, Martins D (2007) Violacein: properties and biological activities. Biotechnol Appl Biochem 48:127–133. https://doi.org/10.1042/BA20070115
Durán N, Justo GZ, Durán M, Brocchi M, Cordi L, Tasic L, Castro GR, Nakazato G (2016) Advances in Chromobacterium violaceum and properties of violacein-Its main secondary metabolite: a review. Biotechnol Adv 34:1030–1045. https://doi.org/10.1016/j.biotechadv.2016.06.003
Durán N, Nakazato G, Durán M, Berti IR, Castro GR, Stanisic D, Brocchi M, Fávaro WJ, Ferreira-Halder CV, Justo GZ, Tasic L (2021) Multi-target drug with potential applications: violacein in the spotlight. World J Microbiol Biotechnol 37:151. https://doi.org/10.1007/s11274-021-03120-4
Eaton BR, Schwarz R, Vatner R, Yeh B, Claude L, Indelicato DJ, Laack N (2021) Osteosarcoma. Pediatr Blood Cancer. https://doi.org/10.1002/pbc.28352
Ferreira CV, Bos CL, Versteeg HH, Justo GZ, Durán N, Peppelenbosch MP (2004) Molecular mechanism of violacein-mediated human leukemia cell death. Blood 104:1459–1464. https://doi.org/10.1182/blood-2004-02-0594
Ganguly A, Yang H, Sharma R, Patel KD, Cabral F (2012) The role of microtubules and their dynamics in cell migration. J Biol Chem 287:43359–43369. https://doi.org/10.1074/jbc.M112.423905
Gartrell J, Pappo A (2020) Recent advances in understanding and managing pediatric rhabdomyosarcoma. F1000Res. https://doi.org/10.12688/f1000research.22451.1
Gupta R, Mitra S, Chowdhury S, Das G, Priyadarshini R, Mukhopadhyay MK, Ghosh SK (2021) Discerning perturbed assembly of lipids in a model membrane in presence of violacein. Biochim Biophys Acta Biomembr 1863:183647. https://doi.org/10.1016/j.bbamem.2021.183647
Harrison DJ, Geller DS, Gill JD, Lewis VO, Gorlick R (2018) Current and future therapeutic approaches for osteosarcoma. Exp Rev Anticancer Ther 18:39–50. https://doi.org/10.1080/14737140.2018.1413939
Hashimi SM, Xu T, Wei MQ (2015) Violacein anticancer activity is enhanced under hypoxia. Oncol Rep 33:1731–1736. https://doi.org/10.3892/or.2015.3781
Hassani HH, Hasan HM, Al-Saadi A, Ali AM, Muhammad MH (2012) A comparative study on cytotoxicity and apoptotic activity of pyocyanin produced by wild type and mutant strains of Pseudomonas aeruginosa. Europe J Exp Biol 2:1389–1394
Haun M, Pereira MF, Hoffmann ME, Joyas A, Campos V, Filardi LD, de Castro SL, Duran N (1992) Bacterial chemistry. VI. Biological activities and cytotoxicity of 1,3-dihydro-2H-indol-2-one derivatives. Biol Res 25:21–25
Jo VY, Fletcher CD (2014) WHO classification of soft tissue tumours: an update based on the 2013 (4th) edition. Pathology 46:95–104. https://doi.org/10.1097/PAT.0000000000000050
Kauanova S, Urazbayev A, Vorobjev I (2021) The frequent sampling of wound scratch assay revelas the “opportunity” window dor quantitative evaluation of cell motility-impending drugs. Front Cell Dev Biol 9:640972. https://doi.org/10.3389/fcell.2021.640972
Kim YJ, Yuk N, Shin HJ, Jung HJ (2021) The natural pigment violacein potentially suppresses the proliferation and stemness of hepatocellular carcinoma cells in vitro. Int J Mol Sci. https://doi.org/10.3390/ijms221910731
Kimmel CB, Ballard WW, Kimmel SR, Ullmann B, Schilling TF (1995) Stages of embryonic development of the zebrafish. Dev Dyn 203:253–310. https://doi.org/10.1002/aja.1002030302
Kodach LL, Bos CL, Durán N, Peppelenbosch MP, Ferreira CV, Hardwick JCH (2006) Violacein synergistically increases 5-fluorouracil cytotoxicity, induces apoptosis and inhibits Akt-mediated signal transduction in human colorectal cancer cells. Carcinogenesis 27:508–516. https://doi.org/10.1093/carcin/bgi307
Konzen M, De Marco D, Cordova CA, Vieira TO, Antônio RV, Creczynski-Pasa TB (2006) Antioxidant properties of violacein: possible relation on its biological function. Bioorg Med Chem 14:8307–8313. https://doi.org/10.1016/j.bmc.2006.09.013
Leal AM, de Queiroz JD, de Medeiros SR, Lima TK, Agnez-Lima LF (2015) Violacein induces cell death by triggering mitochondrial membrane hyperpolarization in vitro. BMC Microbiol 15:115. https://doi.org/10.1186/s12866-015-0452-2
Masuelli L, Pantanella F, La Regina G, Benvenuto M, Fantini M, Mattera R, Di Stefano E, Mattei M, Silvestri R, Schippa S, Manzari V, Modesti A, Bei R (2016) Violacein, an indole-derived purple-colored natural pigment produced by Janthinobacterium lividum, inhibits the growth of head and neck carcinoma cell lines both in vitro and in vivo. Tumour Biol 37:3705–3717. https://doi.org/10.1007/s13277-015-4207-3
Mehta T, Vercruysse K, Johnson T, Ejiofor AO, Myles E, Quick QA (2015) Violacein induces p44/42 mitogen-activated protein kinase-mediated solid tumor cell death and inhibits tumor cell migration. Mol Med Rep 12:1443–1448. https://doi.org/10.3892/mmr.2015.3525
Melo PS, Maria SS, Vidal BC, Haun M, Durán N (2000) Violacein cytotoxicity and induction of apoptosis in V79 cells. In Vitro Cell Dev Biol Anim 36:539–543. https://doi.org/10.1290/1071-2690(2000)0362.0.co;2
Melo PS, Justo GZ, de Azevedo MB, Durán N, Haun M (2003) Violacein and its beta-cyclodextrin complexes induce apoptosis and differentiation in HL60 cells. Toxicology 186:217–225. https://doi.org/10.1016/s0300-483x(02)00751-5
Pantanella F, Berlutti F, Passariello C, Sarli S, Morea C, Schippa S (2007) Violacein and biofilm production in Janthinobacterium lividum. J Appl Microbiol 102:992–999. https://doi.org/10.1111/j.1365-2672.2006.03155.x
Park H, Park S, Yang YH, Choi KY (2021) Microbial synthesis of violacein pigment and its potential applications. Crit Rev Biotechnol 41:879–901. https://doi.org/10.1080/07388551.2021.1892579
Petrovic I, Milivojevic M, Arsenijevic A, Lazic A, Kovacevic Grujicic N, Schwirtlich M, Popovic J, Stevanovic M (2021) Retinoic acid affects basic cellular processes and SOX2 and SOX18 expression in breast carcinoma cells. Biocell 45:1355–1367. https://doi.org/10.32604/biocell.2021.015817
Platt D, Amara S, Mehta T, Vercuyssee K, Myles EL, Johnson T, Tiriveedhi V (2014) Violacein inhibits matrix metalloproteinase mediated CXCR4 expression: potential anti-tumor effect in cancer invasion and metastasis. Biochem Biophys Res Commun 455:107–112. https://doi.org/10.1016/j.bbrc.2014.10.124
Potter TM, Neun BW, Stern ST (2011) Assay to detect lipid peroxidation upon exposure to nanoparticles. Methods Mol Biol 697:181–189. https://doi.org/10.1007/978-1-60327-198-1_19
Rivero Berti I, Rodenak-Kladniew B, Onaindia C, Adam CG, Islan GA, Durán N, Castro GR (2020) Assessment of in vitro cytotoxicity of imidazole ionic liquids and inclusion in targeted drug carriers containing violacein. RSC Adv 10:29336–29346. https://doi.org/10.1039/d0ra05101b
Rodrigues AL, Trachtmann N, Becker J, Lohanatha AF, Blotenberg J, Bolten CJ, Korneli C, de Souza Lima AO, Porto LM, Sprenger GA, Wittmann C (2013) Systems metabolic engineering of Escherichia coli for production of the antitumor drugs violacein and deoxyviolacein. Metab Eng 20:29–41. https://doi.org/10.1016/j.ymben.2013.08.004
Rudzinski ER, Anderson JR, Hawkins DS, Skapek SX, Parham DM, Teot LA (2015) The world health organization classification of skeletal muscle tumors in pediatric rhabdomyosarcoma: a report from the children’s oncology group. Arch Pathol Lab Med 139:1281–1287. https://doi.org/10.5858/arpa.2014-0475-OA
Saraiva VS, Marshall JC, Cools-Lartigue J, Burnier MN (2004) Cytotoxic effects of violacein in human uveal melanoma cell lines. Melanoma Res 14:421–424. https://doi.org/10.1097/00008390-200410000-00014
Schneider CA, Rasband WS, Eliceiri KW (2012) NIH Image to ImageJ: 25 years of image analysis. Nat Methods 9:671–675. https://doi.org/10.1038/nmeth.2089
Skapek SX, Ferrari A, Gupta AA, Lupo PJ, Butler E, Shipley J, Barr FG, Hawkins DS (2019) Rhabdomyosarcoma. Nat Rev Dis Primers 5:1. https://doi.org/10.1038/s41572-018-0051-2
de Souza KD, Perez KR, Durán N, Justo GZ, Caseli L (2017) Interaction of violacein in models for cellular membranes: Regulation of the interaction by the lipid composition at the air-water interface. Colloids Surf B Biointerfaces 160:247–253. https://doi.org/10.1016/j.colsurfb.2017.09.027
Suryawanshi RK, Patil CD, Borase HP, Narkhede CP, Stevenson A, Hallsworth JE, Patil SV (2015) Towards an understanding of bacterial metabolites prodigiosin and violacein and their potential for use in commercial sunscreens. Int J Cosmet Sci 37:98–107. https://doi.org/10.1111/ics.12175
Venegas FA, Köllisch G, Mark K, Diederich WE, Kaufmann A, Bauer S, Chavarría M, Araya JJ, García-Piñeres AJ (2019) The bacterial product Violacein exerts an Immunostimulatory effect Via TLR8. Sci Rep 9:13661. https://doi.org/10.1038/s41598-019-50038-x
Venil CK, Yusof NZ, Aruldass CA, Ahmad W-A (2016) Application of violet pigment from Chromobacterium violaceum UTM5 in textile dyeing. Biologia 71:121–127. https://doi.org/10.1515/biolog-2016-0031
Vishnu TS, Palaniswamy M (2018) Systematic approach on evaluating the in vitro antioxidant activity of violacein; novel isolate Chromobacterium Vaccinii CV5. Biomed Pharmacol J 11:703–709
Wu D, Wang Z, Lin M, Shang Y, Wang F, Zhou JY, Wang F, Zhang X, Luo X, Huang W (2019) In vitro and in vivo antitumor activity of cucurbatin C, a novel natural product from cucumber. Front Pharmacol 10:1287. https://doi.org/10.3389/fphar.2019.01287
Funding
This work was supported by the Ministry of Education, Science and Technological Development of the Republic of Serbia (project numbers 173008, 451–03-68/2020–14/200042, 451–03-9/2021–14/200042, 451–03-68/2022–14/200042).
Author information
Authors and Affiliations
Contributions
Conceptualization: SK and JJ; methodology and investigation: EM, NS, SB, NS, AB, VC and JJ; formal analysis: EM, NS, MN and JJ; resources: AB, VC and SK; visualization: EM and JJ; supervision: JJ and SK; writing—original draft: JJ and SK; writing—review and editing: EM, MN and VC; project administration and funding acquisition: SK. All authors have read and agreed to the submitted version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare they have no financial or nonfinancial interests to disclose.
Ethics approval
The study was conducted according to the guidelines of the EU Directive 2010/63/EU.
Consent to participate
Human subjects did not participate in this study and, therefore, consent to participate was not required.
Consent for publish
Human subjects did not participate in this study and, therefore, consent to publish was not required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary file2 (MP4 509 KB)
Supplementary file3 (MP4 829 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Milosevic, E., Stanisavljevic, N., Boskovic, S. et al. Antitumor activity of natural pigment violacein against osteosarcoma and rhabdomyosarcoma cell lines. J Cancer Res Clin Oncol 149, 10975–10987 (2023). https://doi.org/10.1007/s00432-023-04930-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-023-04930-9