Abstract
Background
We aim to assess the association between procedural time and outcomes in patients in unsuccessful mechanical thrombectomy (MT) for anterior circulation acute stroke.
Methods
We conducted a cohort study on prospectively collected data from patients with M1 and/or M2 segment of middle cerebral artery occlusion with a thrombolysis in cerebral infarction 0–1 at the end of procedure. Primary outcome was 90-day poor outcome. Secondary outcomes were early neurological deterioration (END), symptomatic intracranial hemorrhage (sICH) according to ECASS II and sICH according to SITS-MOST.
Results
Among 852 patients, after comparing characteristics of favourable and poor outcome groups, logistic regression analysis showed age (OR: 1.04; 95%CI: 1.02–1.05; p < 0.001), previous TIA/stroke (OR: 0.23; 95%CI: 0.12–0.74; p = 0.009), M1 occlusion (OR: 1.69; 95%CI: 1.13–2.50; p = 0.01), baseline NIHSS (OR: 1.01; 95%CI: 1.06–1.13; p < 0.001) and procedural time (OR:1.00; 95% CI: 1.00–1.01; p = 0.003) as independent predictors poor outcome at 90 days. Concerning secondary outcomes, logistic regression analysis showed NIHSS (OR:0.96; 95%CI: 0.93–0.99; p = 0.008), general anaesthesia (OR:2.59; 95%CI: 1.52–4.40; p < 0.001), procedural time (OR: 1.00; 95% CI: 1.00–1.01; p = 0.002) and intraprocedural complications (OR: 1.89; 95%CI: 1.02–3.52; p = 0.04) as independent predictors of END. Bridging therapy (OR:2.93; 95%CI: 1.21–7.09; p = 0.017) was associated with sICH per SITS-MOST criteria whereas M1 occlusion (OR: 0.35; 95%CI: 0.18–0.69; p = 0.002), bridging therapy (OR: 2.02; 95%CI: 1.07–3.82; p = 0.03) and intraprocedural complications (OR: 5.55; 95%CI: 2.72–11.31; p < 0.001) were independently associated with sICH per ECASS II criteria. No significant association was found between the number of MT attempts and analyzed outcomes.
Conclusions
Regardless of the number of MT attempts and intraprocedural complications, procedural time was associated with poor outcome and END. We suggest a deeper consideration of procedural time when treating anterior circulation occlusions refractory to MT.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Successful recanalization is one of the strongest predictors of favourable functional outcome after mechanical thrombectomy (MT) in patients with acute ischemic stroke (AIS) secondary to large vessel occlusion (LVO) [1]. However, poor or absent recanalization is reported in up to 30% of cases after MT2. Many causes have been suggested for unsuccessful recanalization, such as vessel tortuosity, thrombus characteristics or clot burden, site of occlusion, age, underlying vessel-wall pathology [2, 3]. In the setting of complex endovascular procedures, whether the optimal approach is to perform multiple thrombectomy attempts to achieve successful recanalization or stopping the procedure to avoid potential harm remains unclear. Indeed, despite the proven benefits of recanalization, longer procedures and more attempts might be detrimental in both successful and unsuccessful procedures [4, 5]. Furthermore, bridging therapy with intravenous thrombolysis (IVT) and MT might be associated with better outcomes compared with direct MT, even in case of partial recanalization [6].
Prognostic factors of outcome in patients who do not reach successful recanalization have been poorly investigated. Several factors such as procedural time (i.e. the time between groin puncture and the end of the procedure/last degree of recanalization) and number of retrieval attempts might influence the treatment decision-making process during the procedure.
The aim of this study was to investigate the associations between clinical, radiological and procedural factors and functional outcomes in patients with AIS due to anterior circulation LVO who had unsuccessful recanalization after MT.
Methods
This is a cohort study based on prospectively collected data of patients included in the Italian Registry of Endovascular Treatment in Acute Stroke (IRETAS). IRETAS is a multicenter, observational internet-based registry of patients with AIS secondary to LVO receiving endovascular treatment [7]. Participating centers were required to register consecutive stroke patients receiving endovascular procedures irrespective of whether treatment was according to guidelines.
Our analysis was conducted according to the STROBE criteria (Strengthening the Reporting of Observational Studies in Epidemiology) for observational studies.
Clinical and radiological data were collected by neurologists and neuroradiologists, respectively. The following clinical data were collected: baseline demographic characteristics such as age and sex; vascular risk factors such as arterial hypertension, diabetes mellitus, hypercholesterolemia, atrial fibrillation, current or previous smoking, previous stroke or transient ischemic attack, congestive heart failure, current tobacco use, baseline National Institutes of Health Stroke Scale (NIHSS), 24-h NIHSS, IVT. We also collected radiological and procedural data such as Alberta Stroke Program Early Computed Tomography Score [ASPECTS], site of occlusion, type of anaesthesia, the interval time between symptom onset and groin puncture time, procedural time (defined as the interval between groin puncture and last degree of recanalization), number of thrombectomy attempts and intraprocedural complications (vessel perforation or dissection).
Neuroradiologists assessed recanalization status at the end of the procedure according to the Thrombolysis in Cerebral Infarction (TICI) score.
Study population and outcome measures
We included patients older than 16 years old, with AIS secondary to isolated occlusion of M1 and/or M2 segment of middle cerebral artery (MCA). Patients were treated with MT within 24 h from symptom onset and had unsuccessful recanalization, defined as a TICI score from 0 to 1, between January 2011 and December 2021. Patients with a pre-stroke modified Rankin Scale (mRS) > 2, unknown time of onset and wake up stroke, tandem and T lesion or intracranial internal carotid artery (ICA) occlusion were excluded.
Primary outcome was 90-day poor outcome, defined as a mRS of 4 to 6. Secondary outcomes were death at 90 days, early neurological deterioration (END), defined as a worsening of at least 4 points on the NIHSS at 24 h or death within 24 h [8], symptomatic intracerebral haemorrhage (sICH) according to European Cooperative Acute Stroke Study (ECASS) II and sICH according to Safe Implementation of Thrombolysis in Stroke-Monitoring Study (SITS-MOST) classification [9, 10].
Statistical analysis
Continuous variables were presented as median (interquartile range, IQR) or mean (standard deviation, SD), as appropriate; dichotomous/categorical variables were presented as numbers and percentages. Student’s t test or Mann–Whitney U test were used for continuous data. Pearson’s χ2 test or Fisher’s exact test were used for dichotomous/categorical data as appropriate.
Univariate analysis was applied to look for associations between clinical and radiological variables and 90-day mRS 4–6, mortality and in-hospital death, END, sICH according to SITS-MOST and sICH according to ECASS II criteria [8,9,10]. The prognostic value of variables associated to outcome measures was tested using a logistic regression model including age, sex, year of treatment and variables found statistically significant (p < 0.10) at the univariate analysis. Probability values less than 0.05 were considered to be of statistical significance. Statistical analyses were performed using SPPS (V.28) ®(IBM) software.
Standard protocol approvals, registrations, and patient consents
Need for ethical approval or patient consent for participation in the IRETAS varied among participating hospitals. Ethical approval and informed consent were obtained when required.
Data availability statement
Anonymized data will be shared from any qualified investigator on reasonable request.
Results
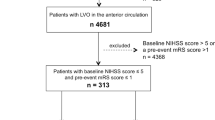
Among 19,069 patients, 852 (mean age: 74.92 ± 12.42; 60.2% female) fulfilled inclusion criteria and were collected for the analysis. Flow diagram of patient inclusion and exclusion is provided in Fig. 1. Among 852 patients included in the study, 57 were excluded from the analysis because of missing data concerning 90-day outcome. Table 1 summarizes baseline clinical and treatment characteristics of the entire cohort and of the two main outcome groups (90-day mRS 0–3 vs 4–6). Main vascular risk factors were hypertension (70%) followed by atrial fibrillation (38.4%) and diabetes (17.5); 47.1% of patients were treated with bridging therapy (IVT and MT) and the remaining with direct MT. The most frequent anaesthesia protocol was sedation (43.2%) followed by general anaesthesia (28.2%). Median baseline NIHSS was 16 (IQR: 11–20) and median baseline ASPECTS was 10 (IQR: 8–10). Median interval time between symptom onset and groin puncture and procedural time were 255 min (IQR: 196–337) and 87 min (IQR: 60–115), respectively.
The two 90-day outcome groups (mRS 0–3 and mRS 4–6) differed by age (76.87 ± 11.1 vs 70.71 ± 13.9; p < 0.001), atrial fibrillation (29.2% vs 42.8%; p < 0.001); hypertension (63.6% vs 74.4%; p = 0.003), baseline NIHSS [12 (IQR: 7–18) vs 18 (IQR: 14–21); p < 0.001], M1 occlusion (46.2% vs 63.1%; p < 0.001), M2 occlusion (53.8% vs 36.9%; p < 0.001), procedural time [79.5 min (IQR: 55–104) vs 89.0 min (IQR: 60–117); p = 0.002]. No difference in the number of thrombectomy attempts between the two outcome groups was found.
After logistic regression analysis, age (OR: 1.04; 95%CI: 1.02–1.05; p < 0.001), previous TIA/stroke (OR: 0.23; 95%CI: 0.12–0.74; p = 0.009), M1 occlusion (OR: 1.69; 95%CI: 1.13–2.50; p = 0.010), baseline NIHSS (OR: 1.01; 95%CI: 1.06–1.13; p < 0.001) and procedural time (OR: 1.00; 95% CI: 1.00–1.01; p = 0.003) were significantly associated with poor outcome at 90 days. Older age (OR: 1.06; 95%CI: 1.04–1.08; p < 0.001) and higher baseline NIHSS (OR: 1.04; 95%CI: 1.01–1.07; p = 0.003) were associated with a higher risk of mortality (Table 2).
Table 3 shows the comparison of clinical and treatment data of secondary outcome groups (END vs no END, sICH vs no sICH per ECASS II criteria, sICH vs no sICH per SITS MOST criteria). After logistic regression analysis, NIHSS (OR: 0.96; 95%CI: 0.93–0.99; p = 0.008), general anaesthesia (OR: 2.59; 95%CI: 1.52–4.40; p < 0.001), procedural time (OR: 1.00; 95% CI: 1.00–1.01; p = 0.002) and intraprocedural complications (OR: 1.89; 95%CI: 1.02–3.52; p = 0.044) were significantly associated with a higher risk of END. Bridging therapy (OR: 2.93; 95%CI: 1.21–7.09; p = 0.017) was significantly associated with sICH according to SITS-MOST criteria, whereas M1 occlusion (OR: 0.35; 95%CI: 0.18–0.69; p = 0.002), bridging therapy (OR: 2.02; 95%CI: 1.07–3.82; p = 0.03) and intraprocedural complications (OR: 5.55; 95%CI: 2.72–11.31; p < 0.001) were independently associated with sICH per ECASS II criteria. No significant association was found between the number of MT attempts and primary or secondary outcomes (Table 4).
Discussion
Several studies have investigated the association between the number of recanalization attempts during MT and functional outcomes [4, 5], mainly focusing on patients with successful recanalization and showing a clear reduction in the probability of achieving favourable outcome with increasing number of attempts [5]. Conversely, only few studies have examined the importance of the number of MT attempts in patients with unsuccessful recanalization and even less studies have investigated whether longer procedural times might be associated with worse outcomes in unsuccessful MT patients [4, 6]. Whether stopping the procedure after achieving a suboptimal degree of recanalization is more beneficial than pursuing successful recanalization with multiple attempts and longer procedural times is a debated question among stroke physicians.
In our study, age, baseline NIHSS, baseline ASPECTS, a previous TIA or stroke, proximal occlusion (M1 segment) of the MCA and procedural time were independently associated with 90-day outcomes, whereas no association was found between the number of MT attempts and outcomes (Table 2). In our study. only patients with M1 and M2 occlusions and witnessed symptom onset were included. This might have mitigated potential bias due to neurointerventionalists experience and skills in treating more technically complex occlusions (i.e. tandem and T lesions), allowing a more reliable analysis of variables and their association with main outcomes.
Independent prognostic factors of END were baseline NIHSS, general anaesthesia, procedural time and intraprocedural complications, (Table 4) whereas no association between the number of MT attempts and END was found despite an association in the univariable analysis (Table 3).
Therefore, our results suggest that procedural times may be associated with outcomes in patients with unsuccessful recanalization, whereas the number of MT attempts is not. Importantly, our results remained significant after adjusting our analysis for the presence of intraprocedural complications. An explanation for these findings might be found in the neurotoxicity of contrast agent during the endovascular procedure. Likely, the longer the interventional procedure lasts the greater is the amount of contrast agent used. Neurotoxicity of contrast agents has been well investigated in both non-pathological settings and pathological conditions where there is blood–brain barrier disruption such as brain tumors or stroke [11, 12]. However, while contrast-induced neurotoxicity in normal brain conditions is quite-well defined [13, 14], contrast-induced neurotoxicity during MT for ischemic stroke is poorly investigated. Interestingly, one study found a low incidence (1.7%) of contrast induced neurotoxicity during MT for ischemic stroke and found renal dysfunction and history of stroke to be associated with neurotoxicity[12]. In another study low ASPECTS and the amount of injected contrast agent were the only independent predictors of sICH which occurred in 9.3% of the patients [15]. No systematic evaluation of the amount of contrast agent used during endovascular procedure has been made in previous endovascular stroke trials. Thus, contrast agent induced neurotoxicity after MT for AIS might be an under recognized condition.
All the above-mentioned studies examined patients with all degrees of recanalization. Our study included only unsuccessful recanalized patients who, compared to patients who did achieve recanalization, might have suffered of a reduced contrast agent wash-out and of a more pronounced neurotoxicity coupled with enlarging ischemic core due to the failed recanalization.
Our study has some limitations. First, no comparison with a control group could be made due to lack of data on untreated patients in the IRETAS registry, hence no definitive conclusions can be drawn from our results. Second, despite the importance of collateral circulation as a prognostic factor of outcome, this could not be included in our analyses because of missing data in more than half of our patients [16]. Third, no data on the amount of contrast agent used were available in the IRETAS and thus our hypothesis of neurotoxicity cannot be tested.
Despite all the above-mentioned limitations, this is one of the largest cohort of non-recanalized ischemic stroke patients with proximal MCA occlusion. Our definition of no recanalization was rigorous (TICI 0–1, with exclusion of 2a) to avoid a possible confounding effect of IVT which could contribute in achieving minimal reperfusion [6]. Selection bias deriving from multivessel occlusion such as tandem or T lesion was reduced by inclusion of only single vessel pathology.
In conclusion, our study showed that procedural times are associated with END and 90-day poor outcome in ischemic stroke patients with failed MT, regardless of the number of MT attempts and intraprocedural complications. Careful consideration of procedural times should be performed when treating LVOs refractory to recanalization. Further larger studies systematically evaluating the correlation between functional outcomes and the amount of contrast agent used during MT are warranted.
References
Goyal M, Menon BK, van Zwam WH et al (2016) Endovascular thrombectomy after large-vessel ischaemic stroke: a meta-analysis of individual patient data from five randomised trials. Lancet 387:1723–1731
Flottmann F, Broocks G, Faizy TD, McDonough R, Watermann L, Deb-Chatterji M, Thomalla G, Herzberg M, Nolte CH, Fiehler J, Leischner H, Brekenfeld C, GSR investigators (2021) Factors associated with failure of reperfusion in endovascular therapy for acute ischemic stroke : a multicenter analysis. Clin Neuroradiol 31:197–205
Abdalla RN, Cantrell DR, Shaibani A, Hurley MC, Jahromi BS, Potts MB, Ansari SA (2021) Refractory stroke thrombectomy: prevalence, etiology, and adjunctive treatment in a North American cohort. Am J Neuroradiol 42:1258–1263. https://doi.org/10.3174/ajnr
Flottmann F, van Horn N, Maros ME, Leischner H, Bechstein M, Meyer L, Sauer M, Deb-Chatterji M, Alegiani A, Thomalla G, Fiehler J, Brekenfeld C, GSR investigators (2022) More retrieval attempts are associated with poorer functional outcome after unsuccessful thrombectomy. Clin Neuroradiol 32:361–368. https://doi.org/10.1007/s00062-021-01054-w
García-Tornel A, Requena M, Rubiera M, Muchada M, Pagola J, Rodriguez-Luna D, Deck M, Juega J, Rodríguez-Villatoro N, Boned S, Olivé-Gadea M, Tomasello A, Hernández D, Molina CA, Ribo M (2019) When to stop. Stroke 50:1781–1788. https://doi.org/10.1161/STROKEAHA.119.025088
Faizy TD, Broocks G, Heit JJ, Kniep H, Flottmann F, Meyer L, Sporns P, Hanning U, Kaesmacher J, Deb-Chatterji M, Vollmuth P, Lansberg MG, Albers GW, Fischer U, Wintermark M, Thomalla G, Fiehler J, Winkelmeier L, German Stroke Registry–Endovascular Treatment Investigators (2023) Association between intravenous thrombolysis and clinical outcomes among patients with ischemic stroke and unsuccessful mechanical reperfusion. JAMA Netw Open 6:e2310213. https://doi.org/10.1001/jamanetworkopen.2023.10213
Mangiafico S, Pracucci G, Saia V, Nencini P, Inzitari D, Nappini S, Vallone S, Zini A, Fuschi M, Cerone D, Bergui M, Cerrato P, Gandini R, Sallustio F, Saletti A, De Vito A, Romano DG, Tassi R, Causin F, Baracchini C, Piano M, Motto C, Ciccone A, Gasparotti R, Magoni M, Giorgianni A, De Lodovici M, Cavasin N, Critelli A, Gallucci M, Carolei A, Meloni T, Corso G, Vaudano G, Duc E, Zappoli F, Cavallini A, Padolecchia R, Tassinari T, Longoni M, Salmaggi A, Zampieri P, Bovi P, Puglioli M, Chiti A, Guidetti G, Simonetti L, Procaccianti G, Menozzi R, Scoditti U, Ricciardi F, Pezzella FR, Guarnieri G, Andreone V, Toni D (2015) The Italian Registry of Endovascular Treatment in Acute Stroke: rationale, design and baseline features of patients. Neurol Sci 36:985–993. https://doi.org/10.1007/s10072-014-2053-5
Seners P, Turc G, Oppenheim C, Baron JC (2015) Incidence, causes and predictors of neurological deterioration occurring within 24 h following acute ischaemic stroke: a systematic review with pathophysiological implications. J Neurol Neurosurg Psychiatry 86:87–94. https://doi.org/10.1136/jnnp-2014-308327
Hacke W, Kaste M, Fieschi C, von Kummer R, Davalos A, Meier D, Larrue V, Bluhmki E, Davis S, Donnan G, Schneider D, Diez-Tejedor E, Trouillas P (1998) Randomised double-blind placebo-controlled trial of thrombolytic therapy with intravenous alteplase in acute ischaemic stroke (ECASS II). Second European-Australasian Acute Stroke Study Investigators. Lancet 352:1245–1251. https://doi.org/10.1016/s0140-6736(98)08020-9
Mazya M, Egido JA, Ford GA, Lees KR, Mikulik R, Toni D, Wahlgren N, Ahmed N, SITS Investigators (2012) Predicting the risk of symptomatic intracerebral hemorrhage in ischemic stroke treated with intravenous alteplase: safe Implementation of Treatments in Stroke (SITS) symptomatic intracerebral hemorrhage risk score. Stroke 43:1524–1531. https://doi.org/10.1161/STROKEAHA.111.644815
Junck L, Marshall WH (1983) Neurotoxicity of radiological contrast agents. Ann Neurol 13:469–484. https://doi.org/10.1002/ana.410130502
Chu YT, Lee KP, Chen CH, Sung PS, Lin YH, Lee CW, Tsai LK, Tang SC, Jeng JS (2020) Contrast-induced encephalopathy after endovascular thrombectomy for acute ischemic stroke. Stroke 51:3756–3759. https://doi.org/10.1161/STROKEAHA.120.031518
Spina R, Simon N, Markus R, Muller DW, Kathir K (2017) Contrast-induced encephalopathy following cardiac catheterization. Catheter Cardiovasc Interv 90:257–268. https://doi.org/10.1002/ccd.26871
Vigano’ M, Mantero V, Basilico P, Cordano C, Sangalli D, Reganati P, Lunghi A, Rigamonti A, Salmaggi A (2021) Contrast-induced encephalopathy mimicking total anterior circulation stroke: a case report and review of the literature. Neurol Sci 42:1145–1150. https://doi.org/10.1007/s10072-020-04844-1
Lopez-Navarro ER, Delfs C, Jarre A, Sanio V, Greif G, Gutierrez J, Ringelstein EB, Meuth SG, Haensch CA, Ringelstein A, Ringelstein M (2022) Contrast neurotoxicity and its association with symptomatic intracranial hemorrhage after mechanical thrombectomy. Clin Neuroradiol 32:961–969. https://doi.org/10.1007/s00062-022-01152-3
Sallustio F, Motta C, Pizzuto S, Diomedi M, Giordano A, D’Agostino VC, Samà D, Mangiafico S, Saia V, Legramante JM, Konda D, Pampana E, Floris R, Stanzione P, Gandini R, Koch G (2017) CT angiography-based collateral flow and time to reperfusion are strong predictors of outcome in endovascular treatment of patients with stroke. J Neurointerv Surg 9:940–943. https://doi.org/10.1136/neurintsurg-2016-012628
Acknowledgements
We thank all patients and persons who participated in IRETAS.
Funding
The project ‘‘Registro Nazionale Trattamento Ictus Acuto’’ (RFPS-2006-1-336562) was funded by grants from the Italian Ministry of Health within the framework of 2006 Finalized Research Programmes (D.Lgs.n.502/1992).
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Conflict of interest
AZ declares consulting fees from Boehringer-Ingelheim, Alexion and CLS Behring; MC declares consultancy or advisory board fees or speaker’s honoraria from Pfizer/Bristol Meyer Squibb and Daiichi Sankyo.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sallustio, F., Nicolini, E., Saia, V. et al. Association between procedural time and outcome in unsuccessful mechanical thrombectomy for acute ischemic stroke: analysis from the Italian Registry of Endovascular Treatment in Acute Stroke. J Neurol 271, 5203–5212 (2024). https://doi.org/10.1007/s00415-024-12458-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-024-12458-2