Abstract
The incidence of Second Primary Tumors in the upper aerodigestive tract varies from 5 to 30 %. Most of them are located either in the same anatomical region or in the lungs, and are related to a poor overall survival. Our objective is to assess the incidence of Second Primary Tumors, factors related to its outcome, frequent associations, and impact on overall survival. 27 patients with Second Primary Tumors were reviewed out of 307 with head and neck cancer, between 2002 and 2011. Patients had a minimum follow-up period of 3 years, or until their death after the last treatment. Sex: 85.2 % were male; male:female ratio of 23:4. The mean age of appearance for the primary tumor was 66.8 years. Only 1 patient had a synchronous Second Primary Tumor, while 26 had metachronous Second Primary Tumors. Second neoplasms were grouped into synchronous and metachronous for statistical analysis. The following parameters were considered: age at diagnosis of the first tumor, gender, smoking and persistence of the habit, primary tumor’s location in the larynx, association between two squamous cell carcinomas (index and second primary tumor), and radiotherapy for the primary tumor. The incidence of Second Primary Tumors was 8.79 %. Overall survival of metachronous tumors was 95 months (7.9 years). The primary tumor’s location that prevailed when developing a Second Primary Tumor was the larynx. Regarding the histology, the most common association was head and neck squamous cell carcinoma with head and neck squamous cell carcinoma (9 patients). The Second Primary Tumor was the cause of death in 51.9 % of the patients and its most frequent location was the lungs. 19 patients underwent radiotherapy. Second Primary Tumors are common in the head and neck. The authors would like to enforce the importance of an extended follow-up, since second neoplasms worsen considerably the patient’s prognosis. Patients who keep on smoking after treatment decrease even more their overall survival.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Head and neck squamous cell carcinoma (HNSCC) is associated with high rates of subsequent primary malignancies [1]. Researchers report an incidence of 2–6 % per year and a cumulative second primary malignancy of 36 % at 20 years among patients with HNSCC [2].
A Primary Tumor (PT) is determined as the tumor that is diagnosed first, while a Second Primary Tumor (SPT) is determined as any other malignant neoplasm that is discovered simultaneously or later [3].
Moertel et al. [4] classified the second neoplasms according to their time sequence to appear as synchronous, those that are diagnosed simultaneously or during the first 6 months since the primary tumor was diagnosed, and metachronous, those that are diagnosed after that period of time. Regarding those tumors that appear simultaneously, a PT is determined as the tumor that caused the initial symptoms.
In the 19th century, Theodor Billroth (1829–1894) was the first one to state that any patient who had overcame a neoplasm could develop another one, only if both were malignant, had different locations and their metastasis could be individualized from each other [5].
In 1932, Warren and Gates [6] stated the criteria for second neoplasms:
-
Each tumor must be histologically malignant.
-
The possibility that the tumor is a metastasis from the other must be excluded.
-
There must be no submucous connection between both tumors and others. That means that if the PT and the second neoplasm appear in close areas and both have the same histological type, and then 2 cm of healthy tissue or dysplastic mucosa must exist between them.
Differential diagnosis between metastases versus second primary malignancies remains to be one of the most challenging tasks. The identification of a metastatic lesion is approached considering clinical and genetic parameters. “Differentiating advanced tumor spread from localized tumor growth has great influence on patient prognosis and could rationally guide evolving therapeutic strategies” [7].
Distinguishing metastases from second primary tumors depends on clinical criteria (localization of the lesion, tumor stage, and disease free interval) and genetic analysis [8]. Metastatic lesions are believed to occur at advanced stage HNSCCs with the antecedent spread to cervical lymph nodes [7]. There exists as well a close relationship between the index tumor and its metastases since both loose the same alleles on chromosomal arms.
On the other hand, in 1953, Slaughter et al. proposed the field cancerisation theory, which considered that the mucosa’s repeated exposure to carcinogenic agents, such as tobacco and alcohol, would determine the outcome of new tumors in different locations and in different time [9].
Our aim is to establish the incidence of second primary tumors, the frequent associations, and the impact on the patient’s overall survival when developing a second or third neoplasm.
Methods
A retrospective review of 307 patients with head and neck malignant neoplasms was made at the Churruca Visca Medical Center from January 2002 to December 2011. Out of them, 27 patients developed a second primary tumor. The second cancer’s diagnosis was based on Warren and Gates criteria.
This research was approved by the Churruca Visca Medical Center Ethics Committee. All of the patients from this center belong to the Federal Police Force, a closed population with no more than 170.000 members. This issue became a crucial aspect when analyzing conclusions, since the number of cases reviewed with second primary tumors is not significant compared to others in the literature.
We considered every patient under treatment for a head and neck cancer during the period previously mentioned, despite their gender or age; or that had developed a second primary tumor simultaneously, synchronically or metachronically in any other location; or that had a malignant neoplasm in the same location or far from it, with a minimum follow-up of 3 years or until their death after the last neoplasm was treated.
In order to analyze the facts, the second primary tumors were grouped into synchronous and metachronous.
The following variables with possible impact on the second primary tumor’s overall survival were analyzed: age at the time the index tumor was diagnosed, gender, primary tumor’s location in the larynx, association between two squamous cell carcinoma (index and second primary tumor), radiotherapy for the primary tumor, smoking, and persistence of this habit. Smoking persistence was defined as the continuous use of addictive substances that could lead to higher morbi- mortality [10]. We considered smoking persistence as the continuation of smoking of at least 1 cigarette/day after the diagnosis of the index tumor.
It would have been interesting to consider radiotherapy as a potential causative agent for second primary tumors; unfortunately as this research was done retrospectively, incomplete data made this impossible.
Statistical analysis
For the statistical analysis, Kaplan–Meier curves were used to assess overall survival. In order to compare mortality between two groups (SPT yes or no; smoking yes or no), the Log-Rank Test was used, considering statistically significant p < 0.05. In order to analyze the variables that determine mortality (age, gender, smoking), the Cox proportional hazards regression model was used.
Results
Out of the 307 patients assessed with malignant neoplasms, 27 developed a second primary tumor representing the 8.79 % of the population. The men–women ratio was 23:4 and the mean age of appearance of the PT is 66.8 years old. As regards the time in which the second primary tumors appear, the metachronous prevailed in 96.3 % of the cases (26 patients) with a mean time of diagnosis of 67.04 months; the synchronous represented only the 3.7 % (1 patient). Only one-third primary tumor was reported.
The Primary Tumor’s location that prevailed was head and neck, and the most frequent sites that developed SPT were larynx and hypopharynx (10 patients, 37.04 %) followed by oropharynx (4 patients, 14.81 %). The locations for SPT decreasingly were lungs (29.62 %), oropharynx (22.22 %), and larynx (18.52 %). The third primary tumor was located in the bladder.
Further analysis on the primary and second primary tumor’s location showed no differences on the following associations: head and neck cancer with head and neck cancer (10 patients), and head and neck cancer with infraclavicular cancers (10 patients).
In our series, we noticed that the location of the primary tumor influenced considerably the second primary tumor’s location. When the primary tumor was situated in the larynx–hypopharynx, oropharynx, and oral cavity, the second cancer frequently took place in the lungs (Table 1).
In our research, squamous cell carcinomas predominated in primary and second primary tumors. Regarding index tumors, squamous cell carcinomas represented 70.37 % of the group (52.6 % well differentiated, 31.6 % moderately differentiated, and 15.8 % poorly differentiated) and had a ratio with other histologies of 19:8. Squamous cell carcinomas in second cancers signified 81.48 % of this group, with a ratio of 22:5 with other histologies.
When comparing histological associations between PT and SPT, the association within two HNSCC prevailed (9 patients), followed by HNSCC—squamous cell carcinoma in another region (7 patients), and non-squamous cell carcinoma in another region—HNSCC (5 patients).
While estimating the different treatment modalities, radiotherapy alone or combined, was dominant in PT as well as in SPT. In primary cancers, 19 patients (70.3 %) underwent radiotherapy: 11 patients radiotherapy + surgery, 4 patients radiotherapy, 3 patients radiotherapy + chemotherapy, 1 patient radiotherapy + chemotherapy + surgery. In second malignancies, 11 patients underwent radiotherapy, most of them in association with chemotherapy (Table 2).
The overall survival was 33.3 % (9 patients), with a minimum follow-up of 3 years or until their death after the last neoplasm was treated. Out of the surviving patients, 29.6 % (8 patients) were healthy and with no disease.
The cause of death in 17 patients out of 27 (62.9 %) was cancer, and in 1 patient (3.7 %) was another disease.
Out of the patients who died of cancer, in 51.9 % (14 patients) was due to the second primary tumor: either because of recurrence (25.9 %), persistence (7.4 %), or metastasis (18.5 %). Only 11.1 % (3/27 patients) died because of the primary tumor.
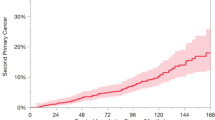
When comparing the overall survival among the patients who developed a second neoplasm to those who did not have a second primary tumor, there was not a statistically significant difference (Fig. 1).
Radiotherapy on the index tumor did not modify the overall survival of patients with SPT (p = 0.8632) (Fig. 2). On the contrary, smoking and its persistence did modify the overall survival, with p = 0.0295 and p = 0.0286, respectively (Figs. 3, 4). In Fig. 3, patients who did smoke seem to have better overall survival during the first months compared to those who did not. As time goes by, the overall survival of non-smokers remains constant, while that of smokers decreases abruptly.
Discussion
Due to advances in early diagnosis and treatment of cancer, loco-regional control was improved and recurrences were reduced; nevertheless, overall survival did not show substantial improvements. The outcome of second primary tumors and distant metastasis has increased [11, 12].
Some research has shown that patients with head and neck cancer have 3 to 7 more possibilities to develop a new primary tumor than the average population; this number could raise up to 30 times more [13].
We agree with other authors that the greatest incidence of SPT is due to an extended life rate of the population, an increased overall survival of oncological patients, an extended follow-up of treated patients with a primary tumor and an enlarged exposure to carcinogens [3].
In our series, the SPT rate was 8.79 %; this is consistent with that reported by other authors which varies from 1 to 47 %, being more frequent between 7 and 22 %.
As in the series reviewed, the metachronous tumors were more than the synchronous tumors, representing 96.3 % of our population [14].
In the only patient who had two simultaneous tumors, the one that caused the symptoms that lead to consultation was treated in the first place (advanced colon cancer) and afterwards the tumor in the oral cavity (early-stage hard palate cancer). Although these patients belong to a complex group, if their clinical general conditions allow it, an aggressive loco-regional treatment would be indicated [15].
Most authors consider that the outcome of the SPT is more frequent during the first 3 years; however, in our research, with a 12-year follow-up, the outcome risk of a new tumor was consistent with the 4–7 % annual risk of developing a SPT [14, 16].
It is considered that between 3 and 4 cases of second neoplasm appear per year; the most frequent locations are head and neck subsectors, lungs, and esophagus; less frequent locations are colon, genitourinary system, pancreas, and breast [9].
According to Licciardello et al, the primary tumors in the oral cavity seem to be related to SPT in the head and neck, while primary tumors in the larynx have to do with the outcome of SPT in the lungs. Epidemiological studies have shown that the risk of suffering lung cancer after a head and neck cancer, especially in the larynx, is from 3 to 6 times more frequent than in the general population [17].
In our series, the localization of the SPT was significantly influenced by the localization of the PT. As Alvarez Marcos et al. reported, when the PT was in the larynx, oropharynx, and oral cavity, the second neoplasm was localized more frequently in the lungs.
Consistently with the different authors considered, the dominant histology for the PT and for the SPT was the squamous cell carcinoma.
In a research based on data from the National Cancer Institute, Robinson et al. found that the second tumor in the head and neck showed a more limited extension than the index tumor in the head and neck, although the differences were not statistically significant [18].
The most frequent treatment with curative purposes on the PT and SPT was radiotherapy, alone or combined. In our series, 70.4 % of the SPT had received radiotherapy applied to the index tumor, but we could not demonstrate that this association had significantly reduced overall survival. Nevertheless, some authors state that overall survival of patients with second tumors in the head and neck was greater in those who did not receive radiotherapy as treatment modality for the primary tumor. Dolan et al. [19] reported a relationship between survival of SPT and the use of radiotherapy. In his research, the percentage of dead patients because of local recurrence of the second malignancy, when it was situated in a previously irradiated area, were 61 % compared to 30 % of those who did not receive radiotherapy.
Other authors such as Farhadieh et al. analyzed the effects of radiotherapy in laryngeal squamous cell carcinomas. In their study of 987 cases, they failed to prove any difference in overall survival between patients who received radiotherapy and those that were treated only with surgery of the primary tumor. Jones et al. also found that overall survival was not considerably different in those patients who had received radiotherapy [5, 20].
Leon et al. studied 427 squamous cell carcinomas SPT and concluded that overall survival in a 5-years follow-up of patients who had received radiotherapy in the PT and those who did not was similar (56 % radiotherapy and 60.5 % without radiotherapy).
Slaughter´s Field cancerisation theory is considered until these days the basis for the development of second tumors. Patients with squamous cell carcinomas of the head and neck develop most frequently other malignancies, synchronous, or metachronous, mainly of the upper aerodigestive tract; this might be related to the level of alcohol or tobacco intoxication [21, 22].
The repetitive exposure of the mucosa to these agents might determine the outcome of new tumors separated from the index tumor by anatomic site and time. They might produce, as well, genetic alterations because of the accumulative and persistent harm [23].
In line with what was previously mentioned and in coincidence with Kiyoto et al. and Leon et al., in our analysis we could see that smoking and its persistence have had a negative impact in the overall survival of patients with SPT [24, 25].
However, these factors alone do not explain the risk of developing a second malignancy. Many patients, including smokers and drinkers, will never develop a SPT, suggesting that genetic susceptibility may also contribute in the etiology. The identification of genetically susceptible groups would allow better perception of patients at high risk [26]. The recognition of risk markers for SPT would enable to improve initial treatment and subsequent better control. The expression of p53 protein identifies patients at higher risk of recurrences or SPT [27].
The overall survival of our series was 33.3 %; 29.6 % of them were free of disease. In similarity with López Molla et al., they reported a 33 % overall survival after 5 years of the diagnosis of the SPT, and Alvarez Marcos 30 %. We assume that the development of second malignancies necessarily worsens the prognosis compared to patients who develop a single cancer.
In a recent series of 1257 patients (595 squamous cell carcinomas of the oral cavity and 662 squamous cell carcinomas of the larynx), Lin et al. [28] reported a greater overall survival in patients with SPT of the upper aerodigestive tract. Recent investigations reinforce the fact that patients suffering from a SPT HNSCC would have a better overall survival than those with a SPT from another location (lungs or esophagus) [24, 29].
Liao et al. [30] in a research carried out in patients with carcinoma of the oral cavity perceived that SPT was diagnosed at a less advanced stage compared to index tumors. This is because of a rigorous follow-up of patients previously treated of a primary tumor in that location.
Several authors believe that early diagnosis and treatment of a second neoplasm would be critical to improve the overall survival. Despite being diagnosed at an early stage, overall survival of patients who develop a SPT is lower than that of patients with a single tumor.
Conclusions
Second primary tumors are frequent in the head and neck area, and their outcome worsens considerably the patient’s prognosis. Smoking persistence after treatment would have a negative impact on overall survival, but not the use of radiotherapy to treat the index tumor. The most common associations were larynx–lung, regarding the organ; squamous cell carcinomas, regarding the histology; and the head and neck, regarding the location.
We enforce the importance of an extended follow-up of patients successfully treated of cancer of the head and neck due to their increased risk of developing a second primary tumor.
Abbreviations
- H&N:
-
Head and neck
- CHT:
-
Chemotherapy
- RT:
-
Radiotherapy
- SPT:
-
Second primary tumor
- SM:
-
Smoking
- PT:
-
Primary tumor
References
Gran S, Dahlstrom K, Peck B, Caywood W, Li G, Wei Q, Zafereo M, Sturgis E (2013) Incidence and pattern of second primary malignancies in patients with index oropharyngeal cancers versus index non- oropharyngeal head and neck cancers. Cancer. 119(14):2593–2601
Morris L, Sikora A, Hayes R, Patel S, Ganly I (2011) Anatomic sites at elevated risk of second primary cancer after an index head and neck cancer. Cancer Causes Control 22(5):671–679
López Mollá C, Estellés Ferriola E, Carrasco Llatas M, Seijas Rosales T, Morales Suárez- Varela M, Dalmau Galofre J (2008) Neoplasias malignas múltiples en el cáncer escamoso de cabeza y cuello. Acta Otorrinolaringol Esp 59:427–432
Moertel C, Dockerty M, Baggenstoss A (1961) Multiple primary malignant neoplasms III: tumors of multicentric origin. Cancer 14:231–237
Jones A, Morar P, Phillips D, Field J, Husband D, Helliwell T (1995) Second primary tumors in patients with head and neck squamous cell carcinoma. Cancer 75:1343–1353
Licciardello J, Spitz M, Ki Hong W (1989) Multiple primary cancer in patients with cancer of the head and neck, esophagous and lung. Int J Radiation Oncology Biol Phys 17:467–476
Leong P, Rezai B, Koch W, Reed A, Eisele D, Lee D, Sidransky D, Jen J, Westra W (1998) Distinguishing second primary tumors from lung metastases in patients with head and neck squamous cell carcinoma. J Natl Cancer Inst 90(13):972–977
Geurts T, Nederlof P, Van den Brekel M, Van´t Veer L, Jong D, Hart A, Van Zandwijk N, Klomp H, Balm A, Van Velthuysen M (2005) Pulmonary squamous cell carcinoma following head and neck squamous cell carcinoma: metastasis or second primary? Clin Cancer Res 11(18):6608–6614
Yamamoto E, Shibuya H, Oshimura R, Miura M (2002) Site specific dependency of second primary cancer in early stage head and neck squamous cell carcinoma. Cancer 94:2007–2014
Sánchez Zamorano L, Hernández Avila M, Lazcano Ponce E (2006) El consumo inmoderado de alcohol como factor predictor de la persistencia del consumo de tabaco en jóvenes. Salud pública de México 48(1):S41–S47
Larson T, Adams G, Fatah H (1990) Survival statistics for multiple primaries in head and neck cancer. Otolaryngol Head Neck Surg 103:14–24
Vikram B (1984) Changing patterns of failure in advanced head and neck cancer. Arch Otolaryngol. 110:564–565
Pardo H, González Aguilar O, Pereyra A, Marolda S, Rubino A, Simkin D (2008) Cáncer de cabeza y cuello. Impacto pronóstico de los 2° y 3° primarios. Rev Argent Cirug 95:205–219
Álvarez Marcos C, Llorente Pendás J, Franco Gutiérrez V, Hermsen M, Cuesta Albalad M, Fernández Espina H et al (2006) Segundos tumores primarios en el cáncer escamoso de cabeza y cuello. Acta Otorrinolaringol Esp 57:462–466
Graff P, Schipman B, Desandes E, Mecellem H, Toussaint B, Cortese S et al (2011) Management of patients with head and neck tumors presenting at diagnosis with a synchronous second cancer at another anatomic site. Clin Oncol (R Coll Radioll). 23:174–181
Farhadieh R, Otahal P, Taghavi K, Salardini A, Russell P, Smee R (2011) Second primary tumors of the head and neck are not associated with adverse overall survival in oral squamous cell carcinomas. J Cancer Sci Ther 3:30–34
Argiris A, Brockstein B (2004) Competing causes of death and second primary tumors in patients with locoregionally advanced head and neck cancer treated with chemoradiotherapy. Cl Cancer Res 10:1956–1962
Robinson E, Neugut A, Murray T, Rennert G (1991) A comparison of the clinical characteristics of first and second primary head and neck cancers. Cancer 68:189–192
Dolan R, Vaughan C, Fuleihan N (1998) Metachronous cancer: prognostic factors including prior irradiation. Otolaryngol Head Neck Surg 119:619–623
León X, Martínez V, López M, García J, Venegas M, Esteller E et al (2012) Second, third, and fourth head and neck tumors. A progressive decrease in survival. Head Neck 34:1716–1719
Franco Gutierrez V, Álvarez Marcos C, Llorente J, Guervós M, Iglesias M, Tamargo L et al (2012) Genetic profile of second primary tumors and recurrences in head and neck squamous cell carcinomas. Head Neck 34:830–839
Gallegos Hernández F (2006) El cáncer de cabeza y cuello. Factores de riesgo y prevención. Cir Ciruj 74:287–293
Slaughter D, Southwick H, Smejkal W (1953) Field cancerization in oral stratified squamous epithelium. Clinical implications of multicentric origin. Cancer 6:963–968
León X, Quer M, Diez S, Orús C, López-Pousa A, Burgués J (1999) Second neoplasm in patients with head and neck cancer. Head Neck 21:204–210
Kiyoto S, Masaru T, Katsunori K, Ayako N, Takenori O, Yukinori A et al (2011) Distinct features of second primary malignancies in head and neck cancer patients in Japan. Tohoku J Exp Med 225:5–12
Wang Z, Sturgis E, Zhang F, Lei D, Liu Z, Xu L et al (2012) Genetic variants of p27 and p21 as predictors for risk of second primary malignancy in patients with index squamous cell carcinoma of head and neck. Mol Cancer 11:17
Zhang Y, Sturgis E, Huang Z, Zafereo M, Wei Q, Li G (2012) Genetic variants of the p53 and p73 genes jointly increase risk of second primary malignancies in patients after index squamous cell carcinoma of the head and neck. Cancer 118:485–492
Lin K, Patel S, Chu P, Matsuo J, Singh B et al (2005) Second primary malignancy of the aerodigestive tract in patients treated for cancer of the oral cavity and larynx. Head Neck 27:1042–1048
Vaamonde P, Martín C, del Río M, LaBella T (2003) Second primary malignancies in patients with cancer of the head and neck. Otolaryngol Head Neck Surg 129:65–70
Liao C, Kang C, Chang J (2007) Survival of second and multiple primary tumors in patients with oral cavity squamous cell carcinoma in the betel quid chewing area. Oral Oncol 43:811–819
Conflict of interest
Marina V. Aramendi and Marta S. Patrucco did not have conflicts of interests when performing this research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Patrucco, M.S., Aramendi, M.V. Prognostic impact of second primary tumors in head and neck cancer. Eur Arch Otorhinolaryngol 273, 1871–1877 (2016). https://doi.org/10.1007/s00405-015-3699-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00405-015-3699-1