Abstract
Background
Our prospective study of patients with early T-stage head and neck cancer indicated a high incidence of newly diagnosed secondary malignancies during the follow-up period. We aimed to determine the incidence rate and risk factors of secondary malignancies in early-stage head and neck cancer patients.
Methods
We sub-analyzed the patient data of a previous study focusing on secondary cancer incidence. The endpoints were statistical analyses of risk factors and survival and incidence rates.
Results
The incidence rate of secondary cancer was 37%, the crude incidence of second primary cancers was 10.6 per 100 person-years, and the 5 year secondary cancer-free survival rate was 63%. The hypopharynx as the primary site was an independent significant predictive factor (odds ratio 3.96, 95% confidence interval 1.07–14.6, p = 0.039).
Conclusions
Early stages of laryngeal, oropharyngeal, and hypopharyngeal cancer had a risk of secondary cancer, especially hypopharyngeal cancer. Attention to the secondary cancer has to be paid during the follow-up period after controlling the early-stage disease. These findings highlight the need for awareness of the incidence of secondary cancer in cases of early-stage primary head and neck cancer.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The incidence of head and neck cancer is about 5% among all malignancies in Japan [1]. About 60% of head and neck cancer patients are at an advanced stage at the first visit because of poor symptoms, except for glottis and oral cancers, and lack of awareness for head and neck cancers. These advanced-stage patients have a high risk of local recurrence or regional lymph node metastasis. Between 60 and 70% of these cases have unsuccessful treatments within the first year since the diagnosis, and 90–100% within 2 years. Moreover, the percentage of the presence of second primary cancers exceeded that of primary head and neck cancer treatment failures, which began at 4 years after the diagnosis of the primary cancer [2,3,4].
Recently, the number of early-stage patients has been increasing following the emergence of advanced diagnostic techniques, such as positron emission tomography-computed tomography (PET-CT) [5] and narrow-band imaging (NBI) endoscopy [6] as well as increased awareness of head and neck cancer among physicians in other fields of practice [7]. We proposed a prospective study to evaluate the efficacy of transoral surgery for early T-stage laryngeal, oropharyngeal and hypopharyngeal cancer patients and uncovered the risk factors of secondary malignancies [8, 9]. In that study, we found that 32% of patients had newly diagnosed secondary cancers during the follow-up period. A few reports describe the incidence rates in early-stage head and neck cancer patients after treatment. The incidence rates are seven to 26% for the observation period ranging from 1.0 to 25.5 years [10,11,12].
It is difficult to precisely describe the incidence rates of newly diagnosed secondary cancers in a retrospective setting because of the variable follow-up procedures among clinicians.
Here, we performed a sub-analysis of the prospective study data concerning the incidence of a secondary cancer in patients with early-stage head and neck cancer and tried to reveal the risk factors for developing a secondary malignancy.
Patients and methods
Study setting
Subset analysis of the previous study, entitled “a prospective clinical trial of the second-look procedure for transoral surgery in patients with T1 and T2 laryngeal, oropharyngeal and hypopharyngeal cancer (UMIN000012485)” [8, 9], was performed.
Endpoints
Statistical analysis between the incidence of secondary cancer and clinical characteristics was one of the primary endpoints. The other primary endpoint was incidence of newly diagnosed secondary cancer (ISC) free survival that was the presence of new malignant disease.
The secondary endpoints were survivals. The event for overall survival (OS) was death. Primary head and neck cancer-specific survival (PHNCSS) had the event of death due to primary cancer. Synchronous double cancer-specific survival (SDCSS) had the event of death due to synchronous double cancer diagnosed with primary head and neck cancer. The new secondary cancer-specific survival (NSCSS) had the event of death due to the new malignant disease during the follow-up period.
Eligibility criteria
Head and neck cancer was classified according to the 7th edition of the Tumor, Node, and Metastasis staging classification [13]. The enrolled patients had clinical T1- and T2-stage laryngeal, oropharyngeal, and hypopharyngeal carcinoma.
Inclusion criteria
Eligibility criteria for enrollment in the trial included a pathologically proven carcinoma and a primary tumor located in the larynx, oropharynx, or hypopharynx. Staging cT1 and cT2 tumor on visual and endoscopic examinations and imaging, such as computerized tomography (CT) or magnetic resonance imaging (MRI). The cN stage was evaluated by ultrasonic echo (US echo) or PET-CT. The primary site was assessed as resectable by transoral surgery and regional lymph node by neck dissection on CT, MRI, or US echo. There was no distant metastasis on PET-CT, no prior treatment for any head and neck cancer and patients over 20 years of age (legally adult in Japan). The performance status of 0–2 in compliance with the Eastern Cooperative Oncology Group criteria. The patients had no contraindications for surgery under general anesthesia and provided written informed consent.
Exclusion criteria
Exclusion criteria included clinically cervical lymph node-positive diagnosis, incurable synchronous malignancies [all patients were screened by PET-CT and magnifying upper gastrointestinal endoscope (UGIE) with NBI], and a history of prior systemic illness. Patients with previous malignancies were included in case these diseases were successfully cured or well-controlled (maintaining a complete response).
Treatment methods
Primary resection was performed using the transoral surgery technique. The mucosal lesion was confirmed by NBI endoscopy and stained with Lugol’s solution. The horizontal safety margin was set at a distance of 1–3 mm from the border of the lesion. Vertical resection was performed in the submucosal layer. After resection, a rapid pathological examination of the margin was performed on the horizontal and vertical sections. In cases with positive margins in the rapid pathological examination, additional resection was performed until a negative margin was confirmed. The resected specimen was stretched on a cork board to clarify the directions and was subsequently fixed with formalin for permanent pathological diagnosis. The wound in oropharyngeal and hypopharyngeal cancer patients was covered with a polyglycolic acid sheet.
Follow-up
Visual and endoscopic observations of the primary site were performed every month for the first and second years, and every 2–3 months from the third to the fifth year. Enhanced cervical CT or US of the primary site and regional lymph nodes was performed every 3–6 months for the first and second years and every 6–12 months from the third to fifth years. Finally, a PET-CT was performed every year for the first and second years, and enhanced whole-body CT was performed every year from the third to fifth years for the evaluation of distant metastasis. In case of the presence of new second primary cancer, the patient was referred to the appropriate department.
Statistical analysis
Patient characteristics were analyzed using the Mann–Whitney U test for the range of age, while Fisher’s exact test was performed for other factors between the new second cancer present and not present. Fisher’s exact test and the Cox proportional hazards model were used for the univariate and multivariate comparisons, respectively. The outcome variable was the incidence of newly diagnosed secondary cancer. The predictive variables were clinical characteristics, such as age, sex, smoking history, alcohol consumption, primary site, human papilloma virus (HPV) status in oropharyngeal cancer, T stage, tumor shape, enhanced CT observation. The pathological characteristics included histological type, differentiation, margin study, lymphatic invasion, vascular invasion, and nerve invasion. The Kaplan–Meier method was used to evaluate survival endpoints. The log-rank test analyzed the statistical differences.
Results
Patient characteristics, univariate and multivariate analyses
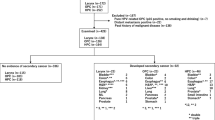
The patients’ clinical courses are shown diagrammatically in Fig. 1. One patient was excluded because of a short-term observation period after surgery due to drop out of the follow-up. Seven patients were excluded because of lymph node-positive status. A total of 43 patients were analyzed as stage I and II laryngeal, oropharyngeal, and hypopharyngeal cancer. All patients had squamous cell carcinoma on pathological examination. HPV was positive in three patients, negative in four patients and unknown in five patients of oropharyngeal cancer. Secondary cancer-positive and -negative groups included 16 and 27 patients, respectively. An enhanced CT examination was performed before surgery in 11 and 23 patients in secondary cancer-positive and -negative groups, respectively. For CT examination analysis, the cut-off values were defined by the receiver operating characteristic (ROC) curve between the presence of secondary cancer and tumor size (length and depth). The cut-off value of the length was 5 mm, and 3 mm in the depth. The clinical characteristics of patients and the results of the univariate analysis are summarized in table. There were no significant differences between these groups by the Mann–Whitney U test for the range of age and Fisher’s exact test for other factors. There was no statistically significant predictive factor for the presence of new secondary cancer by the univariate analysis.
Multivariate logistic regression analysis was performed to examine relationships between the clinical response and the results of univariate analysis for those factors with p value less than 0.15, i.e., range of age, primary site, tumor shape, pathological differentiation. Result revealed that primary head and neck cancer of hypopharynx was an independent significant predictive factor for the newly detected presence of secondary cancer (odds ratio 3.96, 95% CI 1.07–14.6, p = 0.039) (Table 1).
Primary endpoints
The 5 year ISC-free survival rate was 63% (Fig. 2). The incidence rate of secondary cancer was 37% (16/43), and the crude incidence of second primary cancers in this population was 10.6 per 100 person-years during the observation period, and there were three patients with head and neck cancer, two hypopharyngeal malignancies occurred at 13 (primary oropharyngeal cancer, secondary hypopharyngeal cancer) and 62 (both primary and secondary cancers were occurred in left pyriform sinus of the hypopharynx, but the different part) months after the first surgery, and one laryngeal cancer at 63 months (both primary and secondary cancers were occurred in left vocal cord of the larynx, but the different part). Three patients had lung cancers, two squamous cell carcinomas at eight and 25 months, and one adenocarcinoma at 25 months. Two patients developed esophageal cancers at 15 and 36 months, while two patients developed bladder cancers at 17 and 45 months. One patients developed duodenal cancer at 15 months; one developed colon cancer at 18 months, and one developed prostate cancer at 53 months. Similarly, one patients developed sarcoma at 13 months, one had a malignant mantle cell lymphoma at 36 months, and one had myelodysplastic syndrome (human T-lymphotropic virus-related) at 38 months.
Secondary endpoints
The 5 year OS rate was 82% (Fig. 3). The 5 year PHNCSS, SDCSS, and NSCSS were 98%, 98%, and 90%, respectively (Fig. 4). There were no statistically significant differences between these survivals (PHNCSS vs. SDCSS, p = 1; PHNCSS vs. NSCSS, p = 0.7114; and SDCSS vs. NSCSS, p = 0.7123) by log-rank test.
Discussion
At the time of diagnosis of head and neck cancer, about 60% of the patients are at an advanced stage. They have a high risk both of recurrence of the primary cancer and appearance of newly diagnosed secondary cancers after the definitive treatment. Cooper et al. [14] reported that the site of the primary head and neck cancer (except for nasopharynx) had no significant impact on the development of secondary cancers in all stages of head and neck cancer patients treated with radiotherapy during the 8-year observation period. McDonald et al. [15] reported that about 60% of the secondary cancers occurred in the aerodigestive tract, including 30% in the lungs, 20% of head and neck, and 8% in the esophagus, in all stages laryngeal cancer patients with a median follow-up of 10 years.
The acceptable mechanisms of secondary cancer incidence in advanced head and neck cancer patients are common risk factors, such as smoking and alcohol consumption, in head and neck, esophageal, and lung cancers, or field cancerization theory. However, Cooper et al. and McDonald et al. emphasized that although the per-year risk of developing secondary cancers might not be affected by the primary head and neck cancer stage, the cumulative incidence and survival impact was most significant in early-stage patients. Cooper et al. stated that this phenomenon was due to shorter survival and higher incidence of death from advanced primary head and neck cancers. Licciardello et al. [16] illustrated a lower incidence of secondary cancers in hypopharyngeal primary cancer, probably because it has a more aggressive clinical course and shorter survival than other primary head and neck cancers.
Although the diagnosis and treatment improvement [5,6,7], the fact that the high incidence of secondary cancer in advanced head and neck cancer patients has been accepted for head and neck oncologists until now. Lee et al. [17] retrospectively reported that the cumulative incidence of secondary malignancies was 17.9% at 5 years, and 23.1% at 10 years after the diagnosis of first head and neck cancer, which were all stage laryngeal, oral cavity, oropharyngeal, and hypopharyngeal cancers. They also indicated the risk factors were old age, hypopharyngeal cancer, and heavy drinker in multivariate analysis, and hypopharyngeal cancer was the independent variable for secondary cancer-specific survival. Xiang et al. [18] reported the crude incidence of second cancer per 100 patients-years was 1.55 in all stage nine tumor types, including head and neck cancer, treated with three-dimensional conformal, intensity-modulated, or proton beam radiotherapy.
There are a limited number of reports about the second primary cancers in the early-stage head and neck cancer [10,11,12, 18]. These reports are mainly described in oral and glottic malignancies, in which cancers are comparatively easy to diagnose at an early stage, because the symptoms, such as pain and hoarseness, tend to appear in the early phase and the accessibility of visual examination tends to be easy. The diagnosis of head and neck cancer in the early stages has increased due to the recent development of diagnostic tools, such as PET-CT [5] and NBI endoscopy [6], as well as increased awareness of head and neck cancers among physicians in other fields [7] with the fact that magnifying UGIE with NBI was indicated to be useful in detecting secondary primary head and neck cancers after endoscopic resection of esophageal squamous cell carcinoma [19]. These primary site cancers have been considered to have tended to appear at an advanced stage, and we have limited knowledge about the follow-up course of these primary early-stage cancers.
Bhatia et al. [10] reported that 45 out of 176 early-stage oral cavity, oropharyngeal, hypopharyngeal, or laryngeal cancer patients developed secondary cancers within a 1.0–25.5 year follow-up period. The secondary cancer primary sites included 11 head and neck, ten lung, six skin cancer (not melanoma), three breast, colorectal, and prostate, two bladder, and one esophageal, anal, brain, B-cell lymphoma, melanoma, endometrium, and gynecologic cancers. Ord et al. [12] reported that seven out of 112 stage I tongue cancer patients indicated secondary cancers at 32–91 month follow-up periods (three gingival cancers, two oropharyngeal cancers, one buccal cancer, and one adenocarcinoma of lung). Compared to advanced-stage patients, most of which are secondary cancers occurring in the head and neck, lung, or esophagus, the sites of secondary cancer in patients with early-stage head and neck cancer vary that are consistent to our result. In all stage analyses, the risk factors for the presence of secondary cancer were not the primary site of head and neck cancers, but the duration, type, and degree of continued tobacco use (e.g., smokeless tobacco correlating with secondary oral cancers). Another risk factor was the co-abuse of alcohol and tobacco correlating with esophageal or secondary head and neck cancers [14, 15]. Also, aldehyde dehydrogenase-2 genotype, of which 44% Japanese indicated mutation, and alcohol flushing were reported to be risk factors for hypopharyngeal and esophageal squamous cell carcinoma [20, 21]. However, many secondary cancer sites are difficult to consider that tobacco and alcohol consumption are the primary risk factors. The incidence of secondary cancer development depends on the extent of the evaluation, length of follow-up, and curability of the primary lesion, in addition to the patterns of tobacco and alcohol use [22]. In this study, the presence of hypopharyngeal cancer is an independent predictive factor for the development of secondary cancer, along with tobacco and alcohol use. These are well-known risk factors for hypopharyngeal cancer, and considered to affect this result as common risk factor theory or field cancerization theory.
The prognosis of early-stage head and neck cancers is relatively good. In Japan, the 5 year relative survival rates were 94.7% and 80.4% in stage I and stage II tongue cancer, respectively [1], 90% and 80% for oropharyngeal cancer [23], and 69.9% and 62.1% for hypopharyngeal cancer at each stage [1]. The 5 year PHNCSS of this study was 98%, which seemed superior, but the sample size was limited, and it was difficult to compare the survival rates. Eight patients had synchronous double cancers at diagnosis, and the 5 year SDCSS was 98%. The well-controlled survival rate was considered to be attributable to the exclusion criteria, which excluded incurable synchronous malignancies. The incidence of secondary cancer was 37% in the overall observation period, and the 5 year NSCSS was 90%. The incidence rate was higher than on previous reports (seven to 26% during six months to 25.5 year follow-up period [10,11,12]). One of the presumable reason for this discrepancy was fact that the patients in this study were older (median 69 years) than those in previous studies (median 61 years [10] and 63 years [11], and an average of 57.7 years [12]). It is reasonable that the incidence rate of malignant disease increases with age. The adequate management of early-stage primary head and neck cancer contributes to more prolonged survival, which may affect the secondary cancer onset in elderly cancer survivors explaining one possibility of high crude incidence of second cancer per 100 patients-years. Although second cancer has a high incidence, early detection of secondary cancer contributes to high curability. Systemic examinations, such as PET-CT and esophagoscopy, to detect, not only locoregional recurrence and distant metastasis but also secondary cancer, are necessary during the follow-up period. We also found that a preventive intervention to decrease the incidence of secondary cancer is an unmet medical need. According to our results, even at an early stage, hypopharyngeal cancer patients have a high risk of secondary cancer, possibly due to smoking habits, alcohol consumption, and advanced age.
A limitation of the present study was at a single institution with limited sample size.
Conclusions
We sub-analyzed early-stage laryngeal, oropharyngeal, and hypopharyngeal cancer patients and revealed the high incidence rates of secondary cancers with prospective data. The independent predictive factor was hypopharyngeal cancer. One possible reason may be smoking and alcohol consumption as common risk factors for aerodigestive tract cancers. Additionally, for all types of malignancies, the advanced age at the diagnosis of first head and neck cancers is another risk factor, which would be well controlled and longer survival after the first treatment.
Availability of data and materials
The datasets used during the present study are available from the corresponding author upon request. All data generated or analyzed during this study are included in this published article.
References
Cancer Registry and Statistics. Cancer Information Service, National Cancer Center, Japan. https://ganjoho.jp/reg_stat/
Vikram B (1984) Changing patterns of failure in advanced head and neck cancer. Arch Otolaryngol 110:564–565
Vokes EE, Kies M, Haraf DJ et al (1995) Induction chemotherapy followed by concomitant chemoradiotherapy for advanced head and neck cancer: impact on the natural history of the disease. J Clin Oncol 13:876–883
Khuri FR, Lippman SM, Spitz MR et al (1997) Molecular epidemiology and retinoid chemoprevention of head and neck cancer. J Natl Cancer Inst 89:199–211
Dammann F, Horger M, Mueller-Berg M et al (2005) Rational diagnosis of squamous cell carcinoma of the head and neck region: comparative evaluation of CT, MRI, and 18FDG PET. AJR Am J Roentgenol 184:1326–1331
Watanabe A, Taniguchi M, Tsujie H et al (2008) The value of narrow band imaging endoscope for early head and neck cancers. Otolaryngol Head Neck Surg 138:446–451
Morita M, Saeki H, Ito S et al (2014) Surgical strategies for esophageal cancer associated with head and neck cancer. Surg Today 44:1603–1610
Nishimura G, Sano D, Arai Y et al (2019) A prospective clinical trial of the second-look procedure for transoral surgery in patients with T1 and T2 laryngeal, oropharyngeal, and hypopharyngeal cancer. Cancer Med 8:7197–7206
Nishimura G, Sano D, Yabuki K et al (2017) The second-look procedure for transoral videolaryngoscopic surgery for T1 and T2 laryngeal, oropharyngeal, and hypopharyngeal cancer patients: protocol for nonrandomized clinical trial. JMIR Res Protoco 6:e235
Bhatia AK, Lee JW, Pinto HA et al (2017) Double-blind, randomized phase 3 trial of low-dose 13-cis retinoic acid in the prevention of second primaries in head and neck cancer: long-term follow-up of a trial of the eastern cooperative oncology Group-ACRIN Cancer Research Group (C0590). Cancer 123:4653–4662
Rogers SN, Swain A, Carroll C et al (2019) Incidence, timing, presentation, treatment, and outcomes of second primary head and neck squamous cell carcinoma after oral cancer. Br J Oral Maxillofac Surg 57:1074–1080
Ord RA, Isaiah A, Dyalram D et al (2018) Is long-term follow-up mandatory for stage i oral tongue cancer? J Oral Maxillofac Surg 76:2676–2683
Sobin L, Wittekind C (2009) TNM Classification of malignant tumours, 7th edn. Wiley-Liss, New York
Cooper JS, Pajak TF, Rubin P et al (1989) Second malignancies in patients who have head and neck cancer: incidence, effect on survival and implications based on the RTOG experience. Int J Radiat Oncol Biol Phys 17:449–456
McDonald S, Haie C, Rubin P et al (1989) Second malignant tumors in patients with laryngeal carcinoma: diagnosis, treatment, and prevention. Int J Radiat Oncol Biol Phys 17:457–465
Licciardello JT, Spitz MR, Hong WK (1989) Multiple primary cancer in patients with cancer of the head and neck: second cancer of the head and neck, esophagus, and lung. Int J Radiat Oncol Biol Phys 17:467–476
Lee DW, Roh JL, Baek S et al (2013) Second cancer incidence, risk factor, and specific mortality in head and neck squamous cell carcinoma. Otolaryngol Head Neck Surg 149:579–586
Xiang M, Chang DT, Pollom EL (2020) Second cancer risk after primary cancer treatment with three-dimentional conformal, intensity-modulated, or proton beam radiation therapy. Cancer 126(15):3560–3568
Shinozaki T, Katada C, Shiga K et al (2020) Effectiveness of planned surveillance for detecting second primary head and neck cancers after endoscopic resection of esophageal squamous cell carcinoma. Jpn J Clin Oncol. https://doi.org/10.1093/jjco/hyaa087[Epub ahead of print]
Yokoyama A, Kumagai Y, YokoyamaT, et al (2009) Health risk appraisal models for mass screening for esophageal and pharyngeal cancer: an endoscopic follow-up study of cancer-free Japanese men. Cancer Epidemiol Biomarkers Prev 18:651–655
Yamashita Y, Ikegami T, Suzuki M et al (2019) Hypopharyngeal cancer risk in Japanese: genetic polymorphisms related to the metabolism of alcohol- and tobacco-associated carcinogens. J Cancer Res Ther 15:556–563
Lippman SM, Hong WK (1989) Second malignant tumors in head and neck squamous cell carcinoma: the overshadowing threat for patients with early-stage disease. Int J Radiat Oncol Biol Phys 17:691–694
The Cancer Institute Hospital of JFCR, Japan. https://www.jfcr.or.jp
Funding
No funding was received.
Author information
Authors and Affiliations
Contributions
GN and NO conceived and statistical analyzed this study, and edited this manuscript. GN, NO, DS and YA designed this study. GN, TH, HT, YK, KT and TW acquired, analyzed and interpreted the date. GN, DS and YA evaluated the quality control of data and algorithms. GN prepared this manuscript and all authors reviewed and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflicts of interest.
Ethics approval and consent to participate
Ethical approval for the study was obtained from the Yokohama City University Institutional Review Board (#B131107003). Written informed consent was obtained from the study participants to publish of their data.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Nishimura, G., Sano, D., Arai, Y. et al. The incidence of newly diagnosed secondary cancer; sub-analysis the prospective study of the second-look procedure for transoral surgery in patients with T1 and T2 head and neck cancer. Int J Clin Oncol 26, 59–65 (2021). https://doi.org/10.1007/s10147-020-01779-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10147-020-01779-7