Abstract
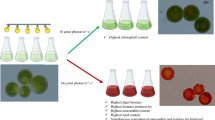
Pavlova sp. is not only one of the most popular microalgae in aquaculture, but also a source of high-quality biodiesel feedstock. With white light as a control (W), Pavlova sp. was cultured in this study under varying light quality, including monochromatic red light (R), blue light (B), and combinations thereof with different proportions (illuminators of mRnB, comprised of m red light units and n blue ones, m+n=7), to examine the effects of illuminating light quality on biocomponent production and biodiesel quality. The results show that combined monochromatic light, especially 2R5B, 3R4B, 4R3B, and 5R2B, could improve the growth of Pavlova sp. The dry weight of harvested algae powder in the 5R2B group reached 418.03 mg/L, and was 22.65% higher than that in the control group (W). Lipid production under combined monochromatic light of 4R3B reached 107.86 mg/L, and was 25.61% higher than in the control (W). In addition, illumination using 4R3B increased the proportion of C16:0 (palmitic acid) and C16: 1 (palmitoleic acid) fatty acids in Pavlova sp. by 15.55% and 21.94%, respectively, which translates into improved biodiesel quality. All cetane numbers (CN) for 4R3B–6R1B were over 51, while iodine values (IV) and degrees of unsaturation (DU) were reduced, leading to more stable biodiesel suitable for long-term storage. In addition, protein production under 6R1B was as high as 31.56 mg/L, 1.8 times greater than under W. Light quality is proposed as an effective tool to regulate biocomponent production by microalgae.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Data Availability Statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Altunoz M, Pirrotta O, Forti L et al. 2017. Combined effects of LED lights and chicken manure on Neochloris oleoabundans growth. Bioresource Technology, 244: 1261–1268, https://doi.org/10.1016/j.biortech.2017.04.094.
Ben Garali S M, Sahraoui I, de la Iglesia P et al. 2016. Effects of nitrogen supply on Pseudo-nitzschia calliantha and Pseudo-nitzschia cf. seriata: field and laboratory experiments. Ecotoxicology, 25(6): 1211–1225, https://doi.org/10.1007/s10646-016-1675-1.
Cerff M, Posten C. 2012. Enhancing the growth of Physcomitrella patens by combination of monochromatic red and blue light-a kinetic study. Biotechnology Journal, 7(4): 527–536, https://doi.org/10.1002/biot.201100044.
Cha T S, Chen J W, Goh E G et al. 2011. Differential regulation of fatty acid biosynthesis in two Chlorella species in response to nitrate treatments and the potential of binary blending microalgae oils for biodiesel application. Bioresource Technology, 102(22): 10633–10640, https://doi.org/10.1016/j.biortech.2011.09.042.
Chisti Y. 2007. Biodiesel from microalgae. Biotechnology Advances, 25(3): 294–306, https://doi.org/10.1016/j.biotechadv.2007.02.001.
Christenson L, Sims R. 2011. Production and harvesting of microalgae for wastewater treatment, biofuels, and bioproducts. Biotechnology Advances, 29(6): 686–702, https://doi.org/10.1016/j.biotechadv.2011.05.015.
Costa B S, Jungandreas A, Jakob T et al. 2013. Blue light is essential for high light acclimation and photoprotection in the diatom Phaeodactylum tricornutum. Journal of Experimental Botany, 64(2): 483–493, https://doi.org/10.1093/jxb/ers340.
Francisco É C, Neves D B, Jacob-Lopes E et al. 2010. Microalgae as feedstock for biodiesel production: carbon dioxide sequestration, lipid production and biofuel quality. Journal of Chemical Technology and Biotechnology, 85(3): 395–403, https://doi.org/10.1002/jctb.2338.
Fukuda H, Kondo A, Noda H. 2001. Biodiesel fuel production by transesterification of oils. Journal of Bioscience and Bioengineering, 92(5): 405–416, https://doi.org/10.1016/s1389-1723(01)80288-7.
Gao B Y, Chen A L, Zhang W Y et al. 2017. Co-production of lipids, eicosapentaenoic acid, fucoxanthin, and chrysolaminarin by Phaeodactylum tricornutum cultured in a flat-plate photobioreactor under varying nitrogen conditions. Journal of Ocean University of China, 16(5): 916–924, https://doi.org/10.1007/s11802-017-3174-2.
Gharajeh N H, Valizadeh M, Dorani E et al. 2020. Biochemical profiling of three indigenous Dunaliella isolates with main focus on fatty acid composition towards potential biotechnological application. Biotechnology Reports, 26: e00479, https://doi.org/10.1016/j.btre.2020.e00479.
Jin C L, Yu B Q, Qian S Y et al. 2021. Impact of combined monochromatic light on the biocomponent productivity of Dunaliella salina. Journal of Renewable and Sustainable Energy, 13(2): 023101, https://doi.org/10.1063/5.0041330.
Jungandreas A, Costa B S, Jakob T et al. 2014. The acclimation of Phaeodactylum tricornutum to blue and red light does not influence the photosynthetic light reaction but strongly disturbs the carbon allocation pattern. PLoS One, 9(8): e99727, https://doi.org/10.1371/journal.pone.0099727.
Kanamoto A, Kato Y, Yoshida E et al. 2021. Development of a method for fucoxanthin production using the haptophyte marine microalga Pavlova sp. OPMS 30543. Marine Biotechnology, 23(2): 331–341, https://doi.org/10.1007/s10126-021-10028-5.
Kim T H, Lee Y, Han S H et al. 2013. The effects of wavelength and wavelength mixing ratios on microalgae growth and nitrogen, phosphorus removal using Scenedesmus sp for wastewater treatment. Bioresource Technology, 130: 75–80, https://doi.org/10.1016/j.biortech.2012.11.134.
Kong Q X, Zhu L Z, Shen X Y. 2010. The toxicity of naphthalene to marine Chlorella vulgaris under different nutrient conditions. Journal of Hazardous Materials, 178(1–3): 282–286, https://doi.org/10.1016/jjhazmat2010.01.074.
Li D J, Yuan Y Z, Cheng D J et al. 2019. Effect of light quality on growth rate, carbohydrate accumulation, fatty acid profile and lutein biosynthesis of Chlorella sp. AE10. Bioresource Technology, 291: 121783, https://doi.org/10.1016/j.biortech.2019.121783.
Li Y F, Li L, Liu J G et al. 2020. Light absorption and growth response of Dunaliella under different light qualities. Journal of Applied Phycology, 32(2): 1041–1052, https://doi.org/10.1007/s10811-020-02057-9.
Lowrey J, Brooks M S, McGinn P J. 2015. Heterotrophic and mixotrophic cultivation of microalgae for biodiesel production in agricultural wastewaters and associated challenges—a critical review. Journal of Applied Phycology, 27(4): 1485–1498, https://doi.org/10.1007/s10811-014-0459-3.
Mata T M, Martins A A, Caetano N S. 2010. Microalgae for biodiesel production and other applications: a review. Renewable & Sustainable Energy Reviews, 14(1): 217–232, https://doi.org/10.1016/j.rser.2009.07.020.
Meher L C, Sagar D V, Naik S N. 2006. Technical aspects of biodiesel production by transesterification-a review. Renewable & Sustainable Energy Reviews, 10(3): 248–268, https://doi.org/10.1016/j.rser.2004.09.002.
Nascimento I A, Marques S S I, Cabanelas I T D et al. 2013. Screening microalgae strains for biodiesel production: lipid productivity and estimation of fuel quality based on fatty acids profiles as selective criteria. Bioenergy Research, 6(1): 1–13, https://doi.org/10.1007/s12155-012-9222-2.
Ponis E, Parisi G, Le Coz J R et al. 2006. Effect of the culture system and culture technique on biochemical characteristics of Pavlova lutheri and its nutritional value for Crassostrea gigas larvae. Aquaculture Nutrition, 12(4): 322–329, https://doi.org/10.1111/j.1365-2095.2006.00411.x.
Qari H A, Oves M. 2020. Fatty acid synthesis by Chlamydomonas reinhardtii in phosphorus limitation. Journal of Bioenergetics and Biomembranes, 52(1): 27–38, https://doi.org/10.1007/s10863-019-09813-8.
Ramos M J, Fernández C M, Casas A et al. 2009. Influence of fatty acid composition of raw materials on biodiesel properties. Bioresource Technology, 100(1): 261–268, https://doi.org/10.1016/j.biortech.2008.06.039.
Robert R, Gérard A. 1999. Bivalve hatchery technology: the current situation for the Pacific oyster Crassostrea gigas and the scallop Pecten maximus in France. Aquatic Living Resources, 12(2): 121–130, https://doi.org/10.1016/s0990-7440(99)80021-7.
Sharma N, Fleurent G, Awwad F et al. 2020. Red light variation an effective alternative to regulate biomass and lipid profiles in Phaeodactylum tricornutum. Applied Sciences, 10(7): 2531, https://doi.org/10.3390/app10072531.
Sørensen M, Berge G M, Reitan K I et al. 2016. Microalga Phaeodactylum tricornutum in feed for Atlantic salmon (Salmo salar)-effect on nutrient digestibility, growth and utilization of feed. Aquaculture, 460: 116–123, https://doi.org/10.1016/j.aquaculture.2016.04.010.
Sulochana S B, Arumugam M. 2016. Influence of abscisic acid on growth, biomass and lipid yield of Scenedesmus quadricauda under nitrogen starved condition. Bioresource Technology, 213: 198–203, https://doi.org/10.1016/j.biortech.2016.02.078.
Thiyagarajan S, Arumugam M, Kathiresan S. 2020. Identification and functional characterization of two novel fatty acid genes from marine microalgae for eicosapentaenoic acid production. Applied Biochemistry and Biotechnology, 190(4): 1371–1384, https://doi.org/10.1007/s12010-019-03176-x.
Vadiveloo A, Moheimani N R, Cosgrove J J et al. 2015. Effect of different light spectra on the growth and productivity of acclimated Nannochloropsis sp. (Eustigmatophyceae). Algal Research, 8: 121–127, https://doi.org/10.1016/j.algal.2015.02.001.
Wu H Q, Miao X L. 2014. Biodiesel quality and biochemical changes of microalgae Chlorella pyrenoidosa and Scenedesmus obliquus in response to nitrate levels. Bioresource Technology, 170: 421–427, https://doi.org/10.1016/j.biortech.2014.08.017.
Yu B Q, Qian S Y, Liu Q et al. 2022. The response of bio-component production of Nannochloris oculata to the combinations of monochromatic light. Journal of Ocean University of China, 21(1): 243–251, https://doi.org/10.1007/s11802-022-4896-3.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the National Natural Science Foundation of China (Nos. 42177459, 41776156, 41271521)
Rights and permissions
About this article
Cite this article
Jin, C., Dong, M., Zhang, Y. et al. Improvement in lipid production and biodiesel quality of Pavlova sp. by monochromatic illumination. J. Ocean. Limnol. 41, 1864–1875 (2023). https://doi.org/10.1007/s00343-022-2196-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00343-022-2196-9