Abstract
Objectives
To evaluate the performance of computed tomography angiography (CTA) ≥64 slices for detecting coronary in-stent restenosis (ISR) and determine the influence of separate characteristics on diagnostic accuracy.
Methods
We searched the PubMed, EMBASE and Cochrane databases for studies of CTA ≥64 slices in diagnosing ISR. We pooled data on bivariate modelling, and subgroup analysis was also performed.
Results
A total of 35 studies involving 4131 stents were included. The pooled positive likelihood ratio (LR+) and the negative likelihood ratio (LR–) were 14.0 and 0.10, for CTA in diagnosis-significant ISR ≥50%. LR+ and LR– were similar between CTA >64 slices versus 64 slices (both P > 0.99). LR– (0.10) was good for ruling out suspected ISR for <3-mm diameter. Time between CTA and stent implantation >6 months did not affect the ability of CTA for the high LR+ (12.3) and the LR– (0.10). Thick-strut stents ≥100 μm or bifurcation stenting demonstrated inferior accuracy, which was unfavourable for stent imaging.
Conclusions
With the high LR+ and LR– of CTA, patients with ISR may be appropriate for non-invasive angiographic follow-up. However, CTA imaging seems unsuitable for patients with characteristics unfavourable for stent imaging, such as thick-strut stents or bifurcation stenting.
Key points
• CTA may provide accurate information on characteristics of in-stent restenosis lesions.
• Using CTA, ISR patients may be appropriate for non-invasive angiographic follow-up.
• Stent diameter and the number of slices do not influence CTA.
• CTA seems unsuitable for patients with thick-strut stents or bifurcation stenting.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Coronary heart disease (CHD) is the most common cause of death and disability worldwide. Each year, more than one million CHD patients are treated with stent implantation [1]. Unfortunately, it is not a permanently curative treatment; even with drug-eluting stents (DESs), in-stent restenosis (ISR) will occur in a certain proportion of patients [2]. ISR can occur in 20–35% of patients for bare-metal stents, and in 5–10% for DES, as demonstrated by intravascular ultrasound [3]. The underlying process of atherosclerosis in the coronary lumen may progress to coronary restenosis, and in patients presenting with recurrent chest pain following DES implantation, invasive coronary angiography (ICA) is still frequently indicated to evaluate the presence of ISR [4]. However, ICA has limitations due to its invasiveness and association with potential risks of morbidity and mortality; therefore, a non-invasive alternative approach for the assessment of stent patency is highly desirable.
Computed tomography angiography (CTA) may have some effect for a more precise visualisation of coronary arteries and the patency of the lumen after stent implantation [5, 6]. This modality has been developed significantly in recent years, effectively allowing non-invasive coronary arteriography. However, the imaging of stents by CT scanner is more difficult than the native coronary artery. This is due to the presence of the stent metal, which can cause artefacts to interfere with the interpretation of lumen patency. With an increasing number of detector rows, promising results of CTA in coronary artery disease have been reported with improved spatial and temporal resolution. Primary studies have been published that compared anatomic imaging by CTA with functional imaging by various methods [7] and with standard care in the diagnosis of patients with suspected CHD [8]. However, currently, no major clinical trial has been conducted that relates to the use of CTA in the evaluation of stent patency. Current research uses different scanning protocols and scanner types, and the reported figures for ISR exhibit considerable variability. Furthermore, a multitude of stent sizes, stent types, strut thickness and other factors exist and have been evaluated with CTA, with varying results for diagnostic accuracy. Thus, the purpose of this study was to assess the pooled diagnostic accuracy of CTA with 64 slices or higher for the detection of ISR, and to determine the influence of separate characteristics of the CTA scanner and stent on the diagnostic performance.
Methods
We did a meta-analysis in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines [9] and the methods described in the Cochrane Handbook for Systematic Reviews of Diagnostic Test Accuracy.
We searched for the relevant studies in the EMBASE, PubMed and Cochrane databases for all published studies from the date of their inception until July 2016. We used the search terms and corresponding medical subject headings for ‘computed tomography’ and ‘in-stent restenosis’. No language restriction was placed. The references in all the retrieved articles were also searched for any additional relevant studies. Two reviewers were asked to look through all this literature and assess their eligibility for analysis.
Eligible studies had to meet the following criteria: (i) they used ≥64-slice CTA as a diagnostic test for coronary luminal restenosis, and reported patients’ baseline characteristics, and their sensitivity and specificity of modality; (ii) true positives (TPs), false positives (FPs), true negatives (TNs) and false negatives (FNs) were able to be calculated on the basis of sensitivity and specificity in respective publications; and (iii) substantial coronary artery restenosis was defined as ≥50% reduction in diameter using ICA as reference. Systematic reviews and meta-analyses were identified and their reference lists were screened. The reference lists of the retrieved articles were also screened. Finally, 35 studies were finalised, and any disagreement between them was resolved by discussion with a third party.
Two reviewers independently extracted relevant data from the selected studies in a standard form. Any identified discrepancies were discussed and corrected. Two-by-two contingency tables were constructed based on the data published, summarising TP, FP, TN and FN on the basis of sensitivity and specificity in respective papers. For each report, we extracted the following items: author, publication years, country, number of restenosis patients and stents, description of the study population (age, sex), scanner type, number of slices, stent size, mean heart rate during scanning, average months between the scan and the stent placement, and days between the CTA scan and the ICA (Table 1).
The quality of the selected studies and the potential bias were assessed using the Quality Assessment of Diagnostic Accuracy Studies (QUADAS-2) guidelines, including additional items as recommended by the Cochrane Collaboration [10]. Discrepancies were resolved by consensus.
Analysis was performed at the per-stent level, and a random effects model was performed for the primary meta-analysis. The primary objective was to estimate the sensitivity and specificity, and the positive and negative likelihood ratios (LR+ and LR–, respectively) with 95% confidence intervals (CIs). We assumed bivariate normal distributions for sensitivity and specificity and presented a forest plot. LR+ and LR– are metrics derived from the summarised sensitivity and specificity for assessing the discriminating ability of the imaging modality. If the LR+ is >10.0 and the LR– is <0.1, then the test can both rule in and rule out the disease [11]. Each data point of the summary receiver operator characteristic (SROC) graph comes from an individual study; then, the SROC curve is formed based on these points to form a smooth curve to reveal pooled accuracy. The sources of heterogeneity were explored at the stent level by using the bivariate generalised linear mixed model as previously described [12, 13]. We assessed the following covariates in meta-regression: sample size (divided by a median of 50 patients), average age of patients (divided by a median of 62 years), percentage of male subjects (divided by a median of 80%), number of slices (defined as either 64 slices or >64 slices), and the average time between the CT scan and the stent placement. The Deeks’ funnel plot was generated to assess the evidence of bias towards studies [14]. Significance testing was at the two-tailed 0.05 level. All analyses were performed using the software Stata SE version 14 (Stata Corp) and Meta Disc (Version 1.4).
Results
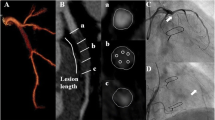
As illustrated in Fig. 1, 35 studies that constituted populations ranging between 15 and 231 patients were included in the final analysis [15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48]. All studies defined significant luminal restenosis as a cut-off ≥50%. One of the studies consisted both of a 64-slice and a 320-slice CT in the diagnosis of ISR simultaneously, and raw data of the two methods were involved in the analysis, respectively [16]. Baseline characteristics of included studies are listed in Table 1. The total included number of patients and atherosclerotic lesions was 2656 and 4131, respectively. The mean age of included patients was 62.2 years, and 80% of them were male. The mean peri-scan heart rate at the time of CTA was 62 beats/min (54–78 beats/min).
Of the 35 publications, 24 studies evaluating 1788 patients with a 64-slice CTA were used [16, 18, 21,22,23,24, 26, 29,30,31,32,33,34, 37,38,39,40, 42,43,44,45,46,47,48]; whereas 11 studies analysed 128- or 320-slice or dual-detector CTA in a total of 961 patients [15,16,17, 19, 20, 25, 27, 28, 35, 36, 41]. Six studies (482 patients) evaluated the effect of strut thickness on diagnostic performance [19, 22, 23, 26, 29, 34], whereas 11 studies (902 patients) analysed the effect of stent diameter [19, 22, 23, 25, 26, 29, 33, 34, 41, 43, 44]. Furthermore, six studies (475 patients) analysed stenting methods [19, 25, 34, 41, 44, 48].
Table S1 shows the results from the assessment of the methodological quality by QUADAS-2 tools. All the included studies exerted high quality in terms of applicability and satisfactory quality in terms of risk of bias. Risk of bias regarding flow and timing was unclear in 12 studies because the timing between the CTA scanning and the ICA was unknown.
The results were pooled from 35 studies with a total of 2656 patients and 4131 stents. Raw data (TP, FP, FN and TN) are depicted in Table S2. The coupled sensitivity and specificity values with a 95% CI are illustrated by the forest plot as shown in Fig. 2. In the per-stent analysis, the pooled sensitivity and specificity for CTA on the diagnosis of ISR were 0.90 (95% CI, 0.85–0.94) and 0.94 (95% CI, 0.91–0.96), respectively. LR+ and LR– were 14.0 (95% CI, 9.6–20.3) and 0.10 (95% CI, 0.07–0.17). For a test to be helpful in diagnosis, it is generally accepted that LR+ should be higher than 10 and LR− below 0.1 [11]. The SROC curve was symmetric, and the area under the curve (AUC) value was 0.97 (95% CI, 0.95–0.98; Fig. 3).
Summary receiver operator characteristic (SROC) curve of computed tomography angiography (CTA) in the diagnosis of in-stent restenosis. Pooled diagnostic accuracy of CTA for the detection of in-stent restenosis (ISR). Hollow circles are individual studies; the full curved line is the SROC curve; and the solid diamond is the pooled accuracy measures surrounded by the 95% confidence interval ellipse
The ROC space did not illustrate a curvilinear trend of points, and there was no presence of threshold effect (P = 0.603). Statistical heterogeneity was found for sensitivity (I 2 = 82.5%; P < 0.001) and specificity (I 2 = 93.2%; P < 0.001). According to meta-regression, all covariates [slice number, CT/percutaneous coronary intervention (PCI) interval (average number of months between scan and stent placement), sample size, male subjects and age] showed a significant effect on sensitivity and specificity. According to I 2 statistics, heterogeneity was present in the covariates of male subjects (I 2 = 55.0%; P = 0.11) and age (I 2 = 83.0%; P < 0.001). A publication bias existed in this study on the basis of Deeks’ test (P = 0.01).
All covariates showed a significant effect on sensitivity and specificity; a subgroup analysis was performed to estimate the level of the effect by classifying studies in certain covariates (Table 2). An analysis stratified according to the number of slices demonstrated a similar sensitivity (0.90) and specificity (0.94) for >64 slices compared with sensitivity (0.90) and specificity (0.94) for 64 slices (P > 0.99). Similarly, stratified according to the strut thickness, indicated a significantly favourable sensitivity (0.96) with thinner struts <100 μm compared with sensitivity (0.84) with thicker struts ≥100 μm (P < 0.001), with equal specificity (P = 0.231). Sensitivity and specificity of the CT/PCI period ≤6 months were similar to those >6 months (P = 0.399 and P = 0.085, respectively).
Diagnostic performances in stents with a diameter of ≥3 mm were more sensitive than those with a diameter of <3 mm (P = 0.024) and specificity (P = 0.073), whereas adjusted LR– (0.10) was good for ruling out ISR for a diameter of <3 mm. Sensitivity in the peri-scan heart rate <65 was significantly higher than the heart rate ≥65 (P = 0.028), but with equally high values of specificity in both groups (P = 0.421). Although LR+ (15.0) was enough to rule in ISR for the heart rate ≥65, the LR– (0.17) was modest. Sensitivity and specificity of CTA were significantly higher in simple stenting than in bifurcation stenting (P = 0.012 and P = 0.001, respectively).
Discussion
This study was performed to test the accuracy of CTA ≥64 slices in the diagnosis of suspected ISR. Our main finding was that CTA, using currently available technology, can effectively rule in and rule out significant restenosis; therefore, it may serve as a gatekeeper before an invasive method is necessary. Using ICA as the reference standard, it exhibited an excellent pooled LR+ (14.0) and LR− (0.10) for the inclusion and exclusion of ISR. ICA may not be suitable to assess or follow-up ISR lesions on a regular basis [49].With the high accuracy of CTA, patients assessed as no visible ISR may partly avoid ICA with its non-negligible risks for several complications.
The assessment of restenosis is essential for risk stratification and determination if concomitant coronary revascularisation is necessitated. CTA has been noted as being useful for the prognosis of high-risk patients and the prediction of coronary events to discriminate unstable plaque [50]. However, only a few studies with limited sample sizes have tested the evaluation of ISR using CTA, and definitive evidence advocating the use of this scanner in ISR requires further validation.
This study differs in two key aspects from the previous studies. First, we enriched the influence of more characteristics of CTA and stents to have adjudicated diagnostic accuracy compared with previous studies [6, 51,52,53,54,55]. Second, our study had more global representation and a larger number of patients, whereas the largest sample size of a previous study was 895 [52]. Six earlier meta-analyses explored the role of CTA with ≤64 slices in suspected restenosis [6, 51,52,53,54,55]. They concluded that the sensitivity was insufficient, and only in selected patients may CTA serve as alternative method to rule out ISR. However, they did not report CTA with >64 slices and adjudicated accuracy for some certain characteristics of the scanner and the stent. Accordingly, access to these data would have warranted further studies.
The present study indicated that the introduction of 128- and 320-slice and dual-detector armamentarium had similarly high diagnostic accuracy together with the 64-slice CTA. This result was clinically relevant because it indicated that CTA ≥64 slices could be effectively used to rule in and rule out ISR. A CT/PCI period >6 months did not affect the ability of CTA to reliably rule in and rule out significant ISR for the high LR+ (12.3) and LR– (0.10). A larger stent naturally provides fewer blooming artefacts and is more visible, which leads to more accurate ISR quantification, whereas stents with <3-mm in diameter were more likely to have ISR than those ≥3 mm in diameter [56]. Although analysis stratified by stent diameter demonstrated more favourable sensitivity and specificity for ≥3 mm in diameter, adjusted LR– (0.10) was also good for ruling out suspected ISR. CTA was probably enough to use as a rule-out triage modality in stents <3 mm in diameter. Strut thickness is also related to ISR detection, and partial volume artefacts from the stent led to blooming and artefactual lumen narrowing. Given that the stents are thick, they may be less likely to be visualised on CTA as they are within the shadow of the artefacts created by stent struts. Based on the present work, thick-strut stents (≥100 μm) had lower diagnostic accuracy than thin-strut stents, which suggested that thick-strut stents may reduce visualisation of the lumen.
Besides thick struts, diagnostic performances tend to be lower with other characteristics that are unfavourable for stent imaging, such as bifurcation stenting and an elevated heart rate. However, Yue et al. [16] revealed that the motion artefacts caused by slice misalignment may be optimised in patients with elevated heart rates for the improved information of 320-slice CTA. Additional research may be warranted to better define the efficiency of 320-slice CTA.
In this study, we clarified that a post-processed iterative reconstruction (IR) algorithm was not adopted in any included paper. The IR algorithms technique was developed in an attempt to decrease the radiation dose or improve image quality, it can significantly reduce image noise without loss in the diagnostic performance and thus holds the potential for radiation dose reduction [57,58,59]. In first-generation IR algorithms, a substantial noise reduction might be associated with the image, leading to a blotchy appearance of the IR-reconstructed studies [60, 61]. Second-generation IR algorithms allow for a more effective reduction of image noise without substantial effects on attenuation; this may have a role in reducing beam hardening and partial-volume artefacts associated with coronary artery stents and heavily calcified vessels [62, 63]. Reductions in stent volumes prompts less blooming artefacts and image noise along with improved in-stent visualisation and diagnostic accuracy [64, 65].
However, it is important to note that all IR techniques are vendor-specific with limited applicability to other CT systems [AIDR (Toshiba), ASIR (GE), IRIS (Siemens), iDose (Philips), SAFIRE (Siemens)], the choice of the IR algorithm and its strength level influences the image impression and noise characteristics. Accordingly, the selection of the preferred IR technique is a specific clinical task germane to the individual preferences for image quality [66]. In view of the fact that a large data base comparable to the one solely including filtered back-projection images is still lacking today, but that it would be very interesting to conduct a meta-analysis on whether performance of CTA for ruling in and ruling out of in-stent-restenosis could be further increased by using IR algorithms.
Some limitations exist in our study. First, there was heterogeneity between the results in our study, which is reflected in the wide predictive intervals around each estimate. We did not find a threshold effect in this study, while meta-regression was conducted to seek potential heterogeneity, and covariates of male subjects and age may have affected the pooled results. Second, not all the reports provided details regarding technical issues in the sub-analysis, which cast caution on the interpretation of the estimates of the adjusted effects. Third, in statistics, meta-analysis comprises statistical methods for contrasting and combining results from different studies in the hope of identifying patterns among study results. Unlike many clinical researches that rely on self-reported data, the meta-analysis is based on indirect observation in the context of multiple papers, and some specific issues may not be included caused by time and skill. Fourth, the vendor-specific IR algorithms were not elucidated in all studies, so we cannot assess the efficiency and efficacy of this advanced techniques. Further overview of CTA-adapted IR algorithms is warranted to investigate the potential usage of IR algorithms.
Conclusion
CTA could provide accurate information on ISR lesions or other characteristics of stents, and evaluation of these assessable lesions could be useful for detecting changes in the long term. Using this modality, patients with ISR may be appropriate for non-invasive angiographic follow-up. However, CTA imaging seems unsuitable for patients with characteristics unfavourable for stent imaging, such as thick-strut stents or bifurcation stenting.
Abbreviations
- CHD:
-
Coronary heart disease
- CTA:
-
Computed tomography angiography
- ICA:
-
Invasive coronary angiography
- ISR:
-
In-stent restenosis
- PCI:
-
Percutaneous coronary intervention
References
GBD 2013 Mortality and Causes of Death Collaborators (2015) Global, regional, and national age-sex specific all-cause and cause-specific mortality for 240 causes of death, 1990–2013: a systematic analysis for the Global Burden of Disease Study 2013. Lancet 385:117–171
Siontis GC, Stefanini GG, Mavridis D, Siontis KC, Alfonso F et al (2015) Percutaneous coronary interventional strategies for treatment of in-stent restenosis: a network meta-analysis. Lancet 386:655–664
James SK, Stenestrand U, Lindback J, Carlsson J, Schersten F et al (2009) Long-term safety and efficacy of drug-eluting versus bare-metal stents in Sweden. N Engl J Med 360:1933–1945
Sandfort V, Lima JA, Bluemke DA (2015) Noninvasive Imaging of Atherosclerotic Plaque Progression: Status of Coronary Computed Tomography Angiography. Circ Cardiovasc Imaging 8:e3316
Sato A, Aonuma K (2015) Role of cardiac multidetector computed tomography beyond coronary angiography. Circ J 79:712–720
Andreini D, Pontone G, Mushtaq S, Pepi M, Bartorelli AL (2010) Multidetector computed tomography coronary angiography for the assessment of coronary in-stent restenosis. Am J Cardiol 105:645–655
Douglas PS, Hoffmann U, Patel MR, Mark DB, Al-Khalidi HR et al (2015) Outcomes of anatomical versus functional testing for coronary artery disease. N Engl J Med 372:1291–1300
SCOT-HEART Investigation Group (2015) CT coronary angiography in patients with suspected angina due to coronary heart disease (SCOT-HEART): an open-label, parallel-group, multicentre trial. Lancet 385:2383–2391
Moher D, Liberati A, Tetzlaff J, Altman DG (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. BMJ 339:b2535
Whiting PF, Rutjes AW, Westwood ME, Mallett S, Deeks JJ et al (2011) QUADAS-2: a revised tool for the quality assessment of diagnostic accuracy studies. Ann Intern Med 155:529–536
Deeks JJ, Altman DG (2004) Diagnostic tests 4: likelihood ratios. BMJ 329:168–169
Hamza TH, Arends LR, van Houwelingen HC, Stijnen T (2009) Multivariate random effects meta-analysis of diagnostic tests with multiple thresholds. BMC Med Res Methodol 9:73
Liu LP, Cui LB, Zhang XX, Cao J, Chang N et al (2015) Diagnostic Performance of Diffusion-weighted Magnetic Resonance Imaging in Bone Malignancy: Evidence From a Meta-Analysis. Medicine (Baltimore) 94:e1998
Deeks JJ, Macaskill P, Irwig L (2005) The performance of tests of publication bias and other sample size effects in systematic reviews of diagnostic test accuracy was assessed. J Clin Epidemiol 58:882–893
Wan YL, Tsay PK, Chen CC, Juan YH, Huang YC et al (2016) Coronary in-stent restenosis: predisposing clinical and stent-related factors, diagnostic performance and analyses of inaccuracies in 320-row computed tomography angiography. Int J Card Imaging 32:105–115
Yue J, Chen J, Dou W, Hu Y, Li Q et al (2015) Comparative analysis between 64- and 320-slice spiral computed tomography in the display of coronary artery stents and diagnosis of in-stent restenosis. Exp Ther Med 10:1871–1876
Li M, Zhang J, Zhang Q, Pan J, Lu Z et al (2015) Coronary stent occlusion: reverse attenuation gradient sign observed at computed tomography angiography improves diagnostic performance. Eur Radiol 25:568–574
Yoshimura M, Nao T, Miura T, Okada M, Nakashima Y et al (2015) New quantitative method to diagnose coronary in-stent restenosis by 64-multislice computed tomography. J Cardiol 65:57–62
Wang J, Chen XM, Wang SH, Ye HH, Cui HB et al (2012) Diagnostic value of 320-slice computed tomography coronary angiography to assess in-stent restenosis. Zhonghua Xin Xue Guan Bing Za Zhi 40:487–491
Zhang XH, Yang L, Wu J, Ju HY, Zhang F et al (2012) Diagnostic accuracy and its affecting factors of dual-source CT for assessment of coronary stents patency and in-stent restenosis. Chin Med J 125:1936–1940
Pan J, Lu Z, Zhang J, Li M, Wei M (2013) Angiographic patterns of in-stent restenosis classified by computed tomography in patients with drug-eluting stents: correlation with invasive coronary angiography. Eur Radiol 23:101–107
Kwon W, Choi J, Kim JY, Kim SY, Yoon J et al (2012) In-stent area stenosis on 64-slice multi-detector computed tomography coronary angiography: optimal cutoff value for minimum lumen cross-sectional area of coronary stents compared with intravascular ultrasound. Int J Card Imaging 28:21–31
Hang CL, Lee YW, Guo GB, Youssef AA, Yip HK et al (2013) Evaluation of coronary artery stent patency by using 64-slice multi-detector computed tomography and conventional coronary angiography: a comparison with intravascular ultrasonography. Int J Cardiol 166:90–95
Zhao J, Zheng LL, Yang Y (2011) Evaluation of coronary artery in-stent patency using 64-slice computed tomography. Coron Artery Dis 22:540–552
Yang ZY, Wang Q, Guo SX, Zhang Y, Fang XM et al (2011) Value of detecting in-stent restenosis by dual source coronary computed tomography coronary angiography. Zhonghua Xin Xue Guan Bing Za Zhi 39:49–52
Andreini D, Pontone G, Bartorelli AL, Mushtaq S, Trabattoni D et al (2011) High diagnostic accuracy of prospective ECG-gating 64-slice computed tomography coronary angiography for the detection of in-stent restenosis: in-stent restenosis assessment by low-dose MDCT. Eur Radiol 21:1430–1438
Kong LY, Jin ZY, Zhang SY, Zhang ZH, Wang YN et al (2009) Assessment of coronary stents by 64-slice computed tomography: in-stent lumen visibility and patency. Chin Med Sci J 24:156–160
de Graaf FR, Schuijf JD, van Velzen JE, Boogers MJ, Kroft LJ et al (2010) Diagnostic accuracy of 320-row multidetector computed tomography coronary angiography to noninvasively assess in-stent restenosis. Investig Radiol 45:331–340
Chung SH, Kim YJ, Hur J, Lee HJ, Choe KO et al (2010) Evaluation of coronary artery in-stent restenosis by 64-section computed tomography: factors affecting assessment and accurate diagnosis. J Thorac Imaging 25:57–63
Abdelkarim MJ, Ahmadi N, Gopal A, Hamirani Y, Karlsberg RP et al (2010) Noninvasive quantitative evaluation of coronary artery stent patency using 64-row multidetector computed tomography. J Cardiovasc Comput Tomogr 4:29–37
Wykrzykowska JJ, Arbab-Zadeh A, Godoy G, Miller JM, Lin S et al (2010) Assessment of in-stent restenosis using 64-MDCT: analysis of the CORE-64 Multicenter International Trial. AJR Am J Roentgenol 194:85–92
Martuscelli E, Romagnoli A, D'Eliseo A, Sperandio M, Di Luozzo M et al (2010) Evaluation of coronary in-stent restenosis by 64-slice computed tomography in patients with optimal heart rate control by systematic administration of beta-blocker drugs. J Cardiovasc Med (Hagerstown) 11:431–439
Haraldsdottir S, Gudnason T, Sigurdsson AF, Gudjonsdottir J, Lehman SJ et al (2010) Diagnostic accuracy of 64-slice multidetector CT for detection of in-stent restenosis in an unselected, consecutive patient population. Eur J Radiol 76:188–194
Andreini D, Pontone G, Bartorelli AL, Trabattoni D, Mushtaq S et al (2009) Comparison of feasibility and diagnostic accuracy of 64-slice multidetector computed tomographic coronary angiography versus invasive coronary angiography versus intravascular ultrasound for evaluation of in-stent restenosis. Am J Cardiol 103:1349–1358
Pflederer T, Marwan M, Renz A, Bachmann S, Ropers D et al (2009) Noninvasive assessment of coronary in-stent restenosis by dual-source computed tomography. Am J Cardiol 103:812–817
Chen BX, Ma FY, Wen ZY, Luo W, Zhao XZ et al (2008) Diagnostic value of 128-slice CT coronary angiography in comparison with invasive coronary angiography. Zhonghua Xin Xue Guan Bing Za Zhi 36:223–228
Manghat N, Van Lingen R, Hewson P, Syed F, Kakani N et al (2008) Usefulness of 64-detector row computed tomography for evaluation of intracoronary stents in symptomatic patients with suspected in-stent restenosis. Am J Cardiol 101:1567–1573
Carrabba N, Bamoshmoosh M, Carusi LM, Parodi G, Valenti R et al (2007) Usefulness of 64-slice multidetector computed tomography for detecting drug eluting in-stent restenosis. Am J Cardiol 100:1754–1758
Das KM, El-Menyar AA, Salam AM, Singh R, Dabdoob WA et al (2007) Contrast-enhanced 64-section coronary multidetector CT angiography versus conventional coronary angiography for stent assessment. Radiology 245:424–432
Schuijf JD, Pundziute G, Jukema JW, Lamb HJ, Tuinenburg JC et al (2007) Evaluation of patients with previous coronary stent implantation with 64-section CT. Radiology 245:416–423
Pugliese F, Weustink AC, Van Mieghem C, Alberghina F, Otsuka M et al (2008) Dual source coronary computed tomography angiography for detecting in-stent restenosis. Heart 94:848–854
Cademartiri F, Palumbo A, Maffei E, La Grutta L, Runza G et al (2007) Diagnostic accuracy of 64-slice CT in the assessment of coronary stents. Radiol Med 112:526–537
Cademartiri F, Schuijf JD, Pugliese F, Mollet NR, Jukema JW et al (2007) Usefulness of 64-slice multislice computed tomography coronary angiography to assess in-stent restenosis. J Am Coll Cardiol 49:2204–2210
Ehara M, Kawai M, Surmely JF, Matsubara T, Terashima M et al (2007) Diagnostic accuracy of coronary in-stent restenosis using 64-slice computed tomography: comparison with invasive coronary angiography. J Am Coll Cardiol 49:951–959
Oncel D, Oncel G, Karaca M (2007) Coronary stent patency and in-stent restenosis: determination with 64-section multidetector CT coronary angiography--initial experience. Radiology 242:403–409
Rist C, von Ziegler F, Nikolaou K, Kirchin MA, Wintersperger BJ et al (2006) Assessment of coronary artery stent patency and restenosis using 64-slice computed tomography. Acad Radiol 13:1465–1473
Rixe J, Achenbach S, Ropers D, Baum U, Kuettner A et al (2006) Assessment of coronary artery stent restenosis by 64-slice multi-detector computed tomography. Eur Heart J 27:2567–2572
Van Mieghem CA, Cademartiri F, Mollet NR, Malagutti P, Valgimigli M et al (2006) Multislice spiral computed tomography for the evaluation of stent patency after left main coronary artery stenting: a comparison with conventional coronary angiography and intravascular ultrasound. Circulation 114:645–653
Nishimura RA, Otto CM, Bonow RO, Carabello BA, Erwin JR et al (2014) 2014 AHA/ACC guideline for the management of patients with valvular heart disease: executive summary: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol 63:2438–2488
Kashiwagi M, Tanaka A, Shimada K, Kitabata H, Komukai K et al (2013) Distribution, frequency and clinical implications of napkin-ring sign assessed by multidetector computed tomography. J Cardiol 61:399–403
Carrabba N, Schuijf JD, de Graaf FR, Parodi G, Maffei E et al (2010) Diagnostic accuracy of 64-slice computed tomography coronary angiography for the detection of in-stent restenosis: a meta-analysis. J Nucl Cardiol 17:470–478
Kumbhani DJ, Ingelmo CP, Schoenhagen P, Curtin RJ, Flamm SD et al (2009) Meta-analysis of diagnostic efficacy of 64-slice computed tomography in the evaluation of coronary in-stent restenosis. Am J Cardiol 103:1675–1681
Vanhoenacker PK, Decramer I, Bladt O, Sarno G, Van Hul E et al (2008) Multidetector computed tomography angiography for assessment of in-stent restenosis: meta-analysis of diagnostic performance. BMC Med Imaging 8:14
Abdulla J, Abildstrom SZ, Gotzsche O, Christensen E, Kober L et al (2007) 64-multislice detector computed tomography coronary angiography as potential alternative to conventional coronary angiography: a systematic review and meta-analysis. Eur Heart J 28:3042–3050
Hamon M, Champ-Rigot L, Morello R, Riddell JW, Hamon M (2008) Diagnostic accuracy of in-stent coronary restenosis detection with multislice spiral computed tomography: a meta-analysis. Eur Radiol 18:217–225
Garg P, Underwood SR, Senior R, Greenwood JP, Plein S (2016) Noninvasive cardiac imaging in suspected acute coronary syndrome. Nat Rev Cardiol 13:266–275
Willemink MJ, de Jong PA, Leiner T (2013) Iterative reconstruction techniques for computed tomography Part 1: technical principles. Eur Radiol 6:1623–1631
Stehli J, Fuchs TA, Bull S, Clerc OF, Possner M et al (2014) Accuracy of coronary CT angiography using a submillisievert fraction of radiation exposure: comparison with invasive coronary angiography. J Am Coll Cardiol 64:772–780
Beister M, Kolditz D, Kalender WA (2012) Iterative reconstruction methods in X-ray CT. Phys Med 28:94–108
Leipsic J, Labounty TM, Heilbron B, Min JK, Mancini GB et al (2010) Adaptive statistical iterative reconstruction: assessment of image noise and image quality in coronary CT angiography. AJR Am J Roentgenol 195(3):649–654
Mueck FG, Körner M, Scherr MK, Geyer LL, Deak Z et al (2012) Upgrade to iterative image reconstruction (IR) in abdominal MDCT imaging: a clinical study for detailed parameter optimization beyond vendor recommendations using the adaptive statistical iterative reconstruction environment (ASIR). Röfo 184:229–238
Renker M, Ramachandra A, Schoepf UJ, Raupach R, Apfaltrer P et al (2011) Iterative image reconstruction techniques: Applications for cardiac CT. J Cardiovasc Comput Tomogr 5:225–230
Ebersberger U, Tricarico F, Schoepf UJ, Blanke P, Spears JR et al (2013) CT evaluation of coronary artery stents with iterative image reconstruction: improvements in image quality and potential for radiation dose reduction. Eur Radiol 23:125–132
Eisentopf J, Achenbach S, Ulzheimer S, Layritz C, Wuest W et al (2013) Low-dose dual-source CT angiography with iterative reconstruction for coronary artery stent evaluation. JACC Cardiovasc Imaging 6:458–465
Gebhard C, Fiechter M, Fuchs TA, Stehli J, Müller E et al (2013) Coronary artery stents: influence of adaptive statistical iterative reconstruction on image quality using 64-HDCT. Eur Heart J Cardiovasc Imaging 14:969–977
Geyer LL, Schoepf UJ, Meinel FG, Nance JW, Bastarrika G et al (2015) State of the Art: Iterative CT Reconstruction Techniques. Radiology 276:339–357
Funding
This work was supported by grants from Science Foundation of Health and Family Planning Commission of Zhejiang Province (2015KYB273)
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Tao Dai.
Conflict of interest
The authors of this manuscript declare no relationships with any companies, whose products or services may be related to the subject matter of the article.
Statistics and biometry
The author Tao Dai has significant statistical expertise in diagnostic meta-analysis.
Informed consent
All analyses were based on previous published studies; thus, no ethical approval and patient consent are required.
Ethical approval
Institutional review board approval was not required because all analyses were based on previous published studies; thus, no ethical approval and patient consent are required.
Methodology
This is a diagnostic meta-analysis performed at one medical institution.
Rights and permissions
About this article
Cite this article
Dai, T., Wang, Jr. & Hu, Pf. Diagnostic performance of computed tomography angiography in the detection of coronary artery in-stent restenosis: evidence from an updated meta-analysis. Eur Radiol 28, 1373–1382 (2018). https://doi.org/10.1007/s00330-017-5097-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-017-5097-0