Abstract
Background
Breast augmentation or reconstruction is a major challenge in esthetic and reconstructive surgery. While autologous fat grafting (AFG) provides a natural filler and seems easy to harvest, AFG in breast surgery is still problematic especially due to the high resorption rate associated with megavolume transfer. Despite this pending issue, there is growing interest in this method, which is becoming more and more widespread, as can be seen by the recent increase in the number of clinical studies. This review aims to highlight recent knowledge in the technique of AFG to the breast and recent refined procedures to improve fat viability and long-term success of the graft.
Methods
Clinical publications and trials of AFG to the breast from the past 5 years were examined. Attention was focused on the different AFG steps and the clinical outcomes, in order to highlight the strengths and weaknesses of the available protocols.
Results
Recent studies have concentrated on new techniques to improve fat viability and graft intake. However, all of these studies use different protocols at each step of the procedure. Furthermore, results may vary depending on the technique used for fat harvesting and processing.
Conclusion
This review points out the recent advances in breast AFG techniques and their associated outcomes and complications. The bibliography has been carefully examined to reach a consensus so that recommendations could be made for each step of the technique with the aim of improving graft viability and long-term volume maintenance.
Level of Evidence V
This journal requires that authors assign a level of evidence to each article. For a full description of these Evidence-Based Medicine ratings, please refer to the Table of Contents or the online Instructions to Authors www.springer.com/00266.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
According to the International Society of Aesthetic Plastic Surgery (ISAPS) [1] breast augmentation and reconstruction represented around 20 % of plastic surgery procedures carried out in 2011. Surgical implants are predominantly used. However, prosthetic breast reconstruction is not without risk, since many cases of leakage or rupture, displacement, infection, and capsular contracture have been reviewed [2, 3]. Moreover, even if implants may last for many years, some of them need to be revised or replaced and they might present sometime complications such as capsular contracture.
An alternative is to use natural fillers for breast reconstruction. By utilizing natural fillers, surgeons use tissue from the patient, such as a flap taken from somewhere in the body and transferred to the breast. The flap can be musculocutaneous (dorsal, abdominal, or transversal) or adipo-cutaneous (DIEP). Finally, subcutaneous adipose tissue alone can be harvested via lipoaspiration and used as a natural filler in autologous fat grafting (AFG), alone or in combination with a flap.
AFG is a safe and easily adjustable technique that results in natural filling of the breast. Even if AFG use is rapidly expanding, the question about its legitimacy continues to be raised because of its main limits: the low rate of long-term graft maintenance [4] and the formation of oil cysts.
It is a fact that the results obtained with the AFG procedure are highly dependent upon the protocol and the experience of the surgeon [5], with these two parameters influencing cell death and resorption. Fortunately, an increasing number of studies and clinical trials are aiming to develop new techniques that limit graft resorption, leading to improvements in AFG efficiency.
The aim of this review is to discuss the pros and cons of AFG to the breast and to outline the different studies and procedures available, in order that recommendations can be made so that the procedures can be optimized to obtain long-lasting results.
Autologous Fat Grafting to the Breast: « The Pros and Cons »
Subcutaneous white adipose tissue is composed of two cell fractions: the adipocyte fraction providing fat volume [6] and the stromal vascular fraction (SVF) that contains numerous cell types (endothelial cells, pericytes, stromal cells, and hematopoietic cells) [7]. It is now widely accepted that these two fractions are critical for graft survival, with the efficacy of AFG being dependent upon their viability.
One of the major strengths of AFG to the breast compared to implant-based reconstruction is the natural aspect and palpation that it gives to the breast. AFG combined with liposuction can provide an esthetically pleasing result for the patient and is easy to perform by surgeons with minimal invasiveness. Another important point is that AFG to the breast gives an unlimited time effect, whereas a prosthesis usually needs to be replaced and does not age with the patient. Finally, the SVF of adipose tissue is a great source of mesenchymal stem cells (MSC), the reason for the increased interest in this tissue over the last decade. With advances in our knowledge of the SVF and cell-assisted lipotransfer, the trophic effect of stromal/stem cells on scars and irradiated zones, as well as the beneficial effects on angiogenesis and skin rejuvenation have been demonstrated. These effects can probably be linked to growth factor secretion by adipose stromal cells (ASC) [8] as well as their ability to differentiate into multiple lineages [9].
Nevertheless, AFG to the breast is also associated with limits and complications. Complications will depend upon the experience of the surgeon and the AFG protocol that is used. In 2013 Leopardi et al. [4] and Largo et al. [10] reviewed previous studies on breast AFG, focusing on the complications that were encountered. With regard to fat injection, the possible complications are: fat necrosis, oil cyst formation, and calcification. These are frequently reported and are not dependent upon the protocol used. Liponecrosis is believed to be caused when an excess of fat is injected, inhibiting the complete vascularization of the graft and causing cell death due to ischemia and nutrient deprivation. Cyst formation is often minor and can be easily detected by palpation or imaging. Of the minor complications, inflammation, striae, bruising, or superficial infections can occur.
One limitation of AFG to the breast comes with the patient morphology. For some patients, large volumes of fat for grafting cannot be harvested (at least not in a single operation without risking deformities of the donor site) especially for Cell-Assisted Lipotransfer (see the dedicated paragraph).
In recent years, a major concern as far as AFG to the breast is concerned, was the possible mammographic interference with cancer diagnosis due to tissue calcification [11]. However, in the 2013 study by Rubin et al, mammographic changes after AFG were less important than those following breast reduction, a commonly accepted procedure [12]. Therefore, care should be taken when analyzing mammographic images following AFG, with a high level of experience required to discern benign calcification from oncological dangers.
The two main remaining issues concerning AFG to the breast are the questions of the possible pro-cancerous role of injected adipose tissue (discussed in the safety paragraph) and the high resorption rate in the months following injection.
The principal limit of this technique addressed in this review is the low maintenance volume due to the use of large volumes of fat [13–15]. The efficacy is estimated to be approximately 50 % with nearly all techniques (Table 1). Most of the resorption occurs in the 3–6 months following adipose tissue injection. Nevertheless, it should be noted that many protocols have been developed since Coleman’s LipoStructure®, enhancing our knowledge concerning fat processing prior to grafting.
To summarize, AFG to the breast is an extremely promising technique with few complications, but protocol improvements are still required in order to offer the best possible outcome with enhanced fat maintenance.
Safety Concerns About AFG to the Breast
Even though breast autologous fat grafts are used worldwide, there is concern about the interaction between stem cells contained in the grafted adipose tissue and any possible residual cancerous cells from a mastectomy procedure. This debate on the role of stem cells in the fat transfer is of further concern when considering cell-assisted lipotransfer and the supra-physiological concentration of stem cells associated with this procedure.
There are numerous in vitro and in vivo models available to investigate the possible pro-cancerous role of fat injection; however, these models do not accurately simulate clinical results while often giving contradictory results [16–19]. There is currently a lack of large controlled studies with long-term follow-up to study this issue leaving the topic understudied. Several individual cases have been reported in which adipose tissue injection led to increased tumor growth and even cancer relapse [20, 21]. The mechanisms involved in these phenomena remain to be identified, but may be cancer dependent such as patients with intraepithelial neoplasia or other undetermined factors.
Despite these concerns, in 2012, the ASPS Fat Graft Task Force did not find “any reports suggesting an increased risk of malignancy associated with fat grafting” and so far, clinical reviews have not revealed any increased cancerous relapse when using AFG to the breast [18, 22, 23].
Recent Studies
AFG to the breast is of great interest in breast surgery with 12 clinical studies currently registered on the clinicaltrial.gov website (research that contains the key words “fat graft”, “fat grafting”, “adipose tissue injection”) [24].
In addition to these clinical trials, Table 1 resumes clinical studies published on breast AFG in the last 5 years.
This table highlights the main issue with breast AFG, which is the high resorption rate of the graft in the months following injection: 15–40 and 20–55 % resorption at 3 and 6 months, respectively (Table 1).
The table also demonstrates that currently, several different protocols are being used by surgeons. This demonstrates the complexity that surrounds adipose tissue handling in AFG and makes it difficult to compare the outcomes.
Moreover, one of the most striking observations is that despite the deleterious effect of high-speed centrifugation being demonstrated, over the past years, 17 studies have used centrifugation steps equal or above 700×g. This underlines the importance of determining a clear protocol for the purification of fat prior to reinjection.
A further piece of information that is highlighted by this table is that it now seems to be commonly accepted that microinjection of small lipid aliquots is employed in order to enhance fat revascularization upon reinjection.
Finally, the most recent breast AFG studies highlight the growing interest in enhancing harvested fat with a graft supplement (with SVF or platelet-rich plasma: PRP).
Processing of Adipose Tissue
Lipotransfer can be divided into four steps: infiltration, lipoaspiration, fat processing, and injection. Although numerous protocols are currently used in clinics for all of these steps, several studies compared the different methods with the common aim of reducing fat resorption [52–57].
Infiltration
Lipoaspiration can be carried out while simultaneously or previously injecting a physiological solution (wet lipoaspiration). Dry lipoaspiration, without infiltration, is no longer used for AFG because of the amount of associated blood loss [50]. The standard ratio for infiltration is commonly 1-cc infiltrated for 1 cc of fat tissue removed, but a higher ratio can be used with tumescent lipoaspiration techniques. The infiltration solution can contain local anesthetic (to avoid peri- and post-operative pain) and vasoconstrictor drugs (adrenaline) to prevent bleeding. Although adrenaline has been proven harmless at low doses, the use of local anesthetic (e.g., lidocaine) is still an issue. Indeed, these drugs have a cytotoxic effect on ASC or can affect their metabolism. Therefore, these drugs must be used at an appropriate concentration to prevent any damage to the tissue [51, 52]. Their use increases the importance of tissue washing prior to reinjection in order to avoid any prolonged contact.
Lipoaspiration
Harvesting Site
As far as fat viability and graft outcome are concerned, several studies have been unable to demonstrate a significant difference between harvesting sites [48, 53, 54]. Furthermore, the most common source for the harvesting of fat grafts is the abdomen [11].
Cannulae
In 2009 Erdim et al. evaluated the influence of cannula size and showed that a cannula that is too small could damage the tissue [55]. The inner diameter of the cannula varies between 2 and 4 mm, with the choice of cannula dependent upon the surgeon carrying out the technique (Table 1). Aside from the cannula size, the number and the size of the holes in the cannula is important. In fact a higher number of holes will enable faster fat harvesting with a concomitant reduction in depression pressure [56, 57]. The diameter of the holes will also affect the size of the harvested lobules, with smaller lobules supposedly being more viable, thus making revascularization easier [56].
Aspiration
The lipoaspiration cannula can either be connected to a syringe, so that aspiration can be carried out manually, or be power assisted. In all cases, the negative pressure caused by aspiration is a critical factor in graft survival [58]. Indeed, in 2001, Shiffman demonstrated that a depression of over 700 mmHg (by manual or by power-assisted liposuction) should not be used for lipotransfer [59]. Cheriyan obtained the same conclusion comparing −250 and −760 mmHg pressures [60]. A questionnaire was sent to the members of the American Society of Plastic Surgeons in 2013, with 55 % of the responders using manual suction instead of other techniques [11].
Pulling of the plunger should be carried out with caution because it will determine the negative pressure inflicted on the tissue [61]. With this in mind, special syringes with a controlled plunger can be used (e.g., Macrofill by Adip’sculpt). In any case, manual or power-assisted systems that enable the control of negative pressure (e.g., K-VAC Syringe by Lipocosm) are preferable.
A further factor that has given contradictory results is the effects of direct exposure to air [62]. Further data should be obtained to determine if non-closed systems should be avoided or not.
In addition to assisted aspiration, several other technological advances have been developed in an attempt to improve the quantity and the quality of harvested fat. Water-assisted lipoaspiration (WAL) uses continuous infiltration that enables “pre-dissection” of the tissue prior to its aspiration (e.g., BodyJet by human med) [30, 44] but this also results in a greater residual interstitial liquid phase.
Some devices also improve the speed of aspiration by making the cannula vibrate during the aspiration process (e.g., PAL by microaire). This technique allows easier and faster aspiration with less effort. However, even if it is possible to regulate the intensity of the cannula vibration, this technique might be more suited to performing high volume liposuction rather than lipotransfer due to the damage done to the harvested tissue with the vibrating cannula.
Purification
The purification step aims to remove the interstitial liquid whilst at the same time ensuring the viability of the graft cells. The two main purification protocols that are mostly used nowadays are decantation and centrifugation. In 2013, 45 % of surgeons used a decantation process and 34 % used centrifugation to purify the graft before AFG to the breast [11].
Decantation
The decantation process is easy to perform and does not necessarily require any additional devices. It can be performed directly in syringes or in a special device designed to trap the fat tissue and to isolate the liquid fraction (e.g., Lipocollector by human med; Tissu-Trans Filtron by Shippert Medical). Even if it is probably the cheapest and easiest to use for surgeons, the main limit of this process is that a significant fraction of liquid will still be trapped in the adipose fraction and will be the first to be reabsorbed after reinjection [30]. The remaining infiltration liquid can also be responsible for a higher concentration of pro-inflammatory cytokines secreted by the adipose tissue during aspiration. This may trigger inflammation at the recipient site, with recruitment of immune cells that can eventually lead to increased graft resorption [63].
Centrifugation
Due to this limitation in the decantation process, since the 1990s the lipoaspirate can also be centrifuged to remove the liquid fraction and improve graft uptake [11, 62, 64, 65]. Moreover, in 2010, Condé-Green found a greater concentration of ASC in the centrifuged fat than with the decantation method alone [61]. However, this step requires a centrifuge and probably more time to carry out than simple decantation. Since Coleman described the purification step with 3 min of centrifugation at 3000 rpm (rarely mentioned but corresponding to 900–1200 g, depending upon the rotor used), many studies have investigated the role of centrifugation (speed and time) on the viability and the maintenance of the reinjected tissue [61, 63, 65–67]. Caution must be taken with this centrifugation step to preserve the integrity of the adipose tissue. Indeed, the centrifugation speed is correlated with fat tissue compaction but also with the release of oil due to the death of adipocytes. A balance must then be found between interstitial liquid removal and adipose tissue damage. Tissue damage can lead to an increase in the secretion of pro-inflammatory cytokines, such as Monocyte Chimoattractant Protein 1 (MCP1) or Interleukin 6 (IL6) [63]. Hoareau and Kurita have both shown a decrease in survival with centrifugation forces higher than 400×g for 1 min [63, 67]. However, there are some protocols at the moment that still use a centrifugation step of 3 min or more at 3000 rpm. On the other hand, some protocols use one or several low speed/time centrifugation(s) [66, 68]. There are some new protocols that use a similar approach to reduce or limit damage to the tissue through the use of manual centrifugation (15×g). There is however little data in the literature to make conclusions regarding such a method.
Alternative Processes
The aim of the filtration method is the same as that of the decantation or centrifugation methods: removal of the liquid fraction to purify the fat before reinjection. It is accomplished by passing the lipoaspirate through a filter (e.g., PureGraft by Cytori or Lipivage by Genesis Biosystem) to remove particles below the chosen size. In 2013, Zhu found less oil and greater viability with this system compared to high-speed centrifugation [69].
Some protocols also integrate additional washing steps (e.g., Macrofill by Adip’sculpt; PureGraft by Cytori) that aim to remove the remaining infiltration solution [61, 69, 70], to get rid of most of the local anesthetic, vasoconstrictors, inflammatory molecules, and death factors (released during liposuction) that may damage the tissue and limit graft success [51, 63]. In 2013, only 28 % of surgeons were using a washing step with breast AFG [11].
Reinjection
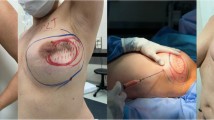
The final step of AFG is the reinjection of the adipose tissue. This step usually requires the use of different sized syringes (from 1 cc to 50 cc) depending on the quantity of adipose tissue to graft. The injection is carried out with a retrograde movement that deposes a small amount of fat tissue in multiple directions via microtunneling at the recipient site. If the graft deposit is too large it will take longer for vascularization to reach the center of the graft, resulting in fat necrosis (Fig. 1).
Therefore, the injection of small aliquots of fat is now commonly accepted, with a cannula of 2 mm being preferred for fat reinjection (Table 1). Surgeons usually graft in all tissue planes (except glandular areas) [56]. No consensus is available however on whether a particular grafting zone is preferable even though muscular zones are more vascularized and could thus prove to be good recipients.
To protect the injected tissue from injection-related shear stress, automated devices have been developed. These devices aim to inject the fat with rigorous continuous pressure. Control of the injection (with an adipose tissue injector) was shown to improve in vivo fat retention in a nude mouse model compared to a modified Coleman’s method following simple decantation [71].
Finally, care must be taken not to overgraft in the breast. Faced with resorption, many surgeons try to overcome this limit by “overcorrection” of the recipient site. However, exceeding breast volume might lead to an inverse effect with increased graft pressure resulting in tissue damage [27, 56].
More Complex Protocols
In the last five years, a new approach has been developed to improve fat grafting through the addition of stem cells or growth factors [25, 31, 34, 35, 37, 39]. In all of these protocols, whether the addition of PRP, SVF, or ASC is implied, the supra-physiological concentrations of stem cells and growth factors is a sensitive issue in reconstructive breast surgery with a risk of cancer relapse [72].
PRP Enriched Grafts
In surgical fields, great interest has been demonstrated in platelet concentrates, notably in PRP, which is easy to use and only requires a blood sample. This blood derived formula contains a concentrated number of platelets that releases several growth factors (contained in the alpha-granules) such as platelet-derived growth factor (PDGF), transforming growth factor beta (TGFß), vascular endothelial growth factor (VEGF), and epidermal growth factor (EGF). These growth factors are responsible for high angiogenesis and proliferation potential in vitro [73]. Because of its properties, the use of PRP in breast AFG has been considered. There are still limited and conflicting results in the literature. In 2011, Salgarello et al. showed no benefit of using PRP with Coleman AFG, whilst in 2012 Gentile et al. reported a 1-year graft survival of 69 % compared to 39 % for the Coleman technique alone [35, 37]. PRP clearly has a concentration-dependent effect, with standardization of PRP preparation still unavailable. These factors and the numerous available protocols [74] can explain the contradictory results that have been obtained. More studies with a higher number of patients will be required to reach a consensus on the use of PRP at a defined concentration.
Cell-Assisted Lipotransfer (CAL)
Over the past decade, studies have shown a great deal of interest in the SVF mainly because of its ASC content and its ability to secrete pro-angiogenic factors [75, 76]. Thus, new procedures have emerged, consisting of the injection of a stem cell enriched fat graft. The common name for this protocol is Cell-Assisted Lipotransfer (CAL) [25].
The goal of this approach is to inject more SVF cells to promote vascularization and survival of the graft thanks to the presence of more endothelial precursors and pro-angiogenic factors like the vascular endothelial growth factor (VEGF) secreted by the SVF [8]. Moreover, the enriched grafts contain more ASC that may be able to differentiate in the adipocyte lineage in situ to overcome the death of adipocytes that occurs during AFG. Finally, the immunoregulatory potential of ASC [77] may decrease the inflammation that is responsible for a sub-optimal outcome, causing adipocyte death. In the CAL protocol, the SVF is isolated of half of the harvested fat and then combined with the other half to create a stromal cell enriched fat graft. Several devices are now used in clinical practice to isolate the adipose SVF. These devices often use enzymatic digestion to digest the tissue and allow the isolation of the SVF cells as a pellet following centrifugation of the digested tissue [78].
So far the results obtained with CAL do not allow us to reach any conclusion as far as the superiority of this technique is concerned when compared with conventional fat transfer [45]. There is still a lack of real comparative studies with an objective assessment of graft survival. Moreover, the higher cost and quantity of tissue required for CAL remain limiting factors. It is likely that the CAL technique may not be suited to every AFG application. However, SVF seems to improve scar healing and might be suitable for low trophicity recipient sites [79].
ASC Enriched Graft
Last year, Trojahn Kølle et al. published a study carried out with volunteers in which fat grafts were compared with fat grafts enriched with cultured ASC. Even if this study was conducted on bolus injections of fat, the improvement in fat survival at 121 days (80.9 vs. 16.3 %) has now raised interest in cultured ASC supplemented fat for AFG [80]. We can expect in future breast AFG studies with cultured ASC to assess safety and cost effectiveness of this concept. However, this protocol is not a one-step procedure, with additional costs, time and the possibility of contamination related to the expansion of stromal cells. Furthermore, the intensive expansion of stromal cells increases the mutation risk in these cells (abnormal caryotype).
Breast Expansion System
Focusing on the recipient site, Khouri has developed the BRAVA system. The principle of this device is to extend the breast skin to make it a more favorable recipient for fat injection. The device applies a negative pressure (from 15 to 33 mmHg) to the breast area to (1) untighten the skin, increasing recipient volume, and (2) increasing blood supply via mechanotransduction effects [14]. Several studies and a clinical trial have supported the use of this technique and have demonstrated good results [26, 33, 38, 40, 49]. However, the BRAVA is quite expensive and its use is unwieldy for the patient. Considering its cost and its cumbersome nature it might be better suited for selected patients, for example with a “hostile” recipient breast (post-radiotherapy [49] ) or those who want a large augmentation with limited procedures [81]. Nowadays, 8 % of American plastic surgeons are using this device prior to AFG [11].
Guidelines
In Table 2, we resume some tips that, according to the literature, can help to improve outcome for breast AFG. There is still no ideal protocol but we believe that the recent knowledge can be used to avoid deleterious procedures.
Conclusion
AFG to the breast is now a standard practice for breast reconstruction and augmentation procedures. However, there remain serious limitations as far as graft intake is concerned. To improve outcome, many studies have focused on finding new approaches to treat the fat before reinjection. Here, we summarized current knowledge for each step of the AFG to the breast technique.
We believe it is possible to improve long-term outcomes by respecting both adipocyte and stromal fractions during the entire AFG to the breast process. Graft survival can be improved by controlled aspiration with low negative pressure (<700 mmHg) and with multi-small-holed cannulae. Before reinjection the fat needs to be concentrated while care is taken to preserve cell survival (positive balance between liquid removal and fat damages).
In the past 10 years, many research groups have shown interest in “stem cell enriched” grafts, especially the CAL protocol with SVF. It is still not clear if this protocol is better than conventional fat grafting. It might be promising for hostile hypotrophic recipient sites (an irradiated breast for example) justifying the higher cost, the necessity to harvest more tissue (half of it is digested) and the supplementary operation time that is required (approximately 2 h more). For all other applications high graft maintenance can be achieved by carefully processing the fat with simple and low-cost techniques.
The future of AFG to the breast is yet to be written; but it is clear that since the advent of the optimized Coleman’s lipostructure, a great deal of fat grafting knowledge has been gathered and we now have more clues about how to handle fat in order to offer the best esthetic outcomes for patients.
References
International Society of Aesthetic Plastic Surgery (ISAPS) Global statistics for year 2011: http://www.isaps.org/news/isaps-global-statistics. Accessed 31 July 2014)
Oulharj S, Pauchot J, Tropet Y (2013) PIP breast implant removal: a study of 828 cases. J Plast Reconstr Aesthet Surg 67:302–307
Cunningham B, McCue J (2009) Safety and effectiveness of Mentor’s MemoryGel implants at 6 years. Aesthet Plast Surg 33:440–444
Leopardi D, Thavaneswaran P, Mutimer KL, Olbourne NA, Maddern GJ (2013) Autologous fat transfer for breast augmentation: a systematic review. ANZ J Surg 84:225–230
Gutowski KA, Force AFGT (2009) Current applications and safety of autologous fat grafts: a report of the ASPS fat graft task force. Plast Reconstr Surg 124:272–280
Wronska A, Kmiec Z (2012) Structural and biochemical characteristics of various white adipose tissue depots. Acta Physiol (Oxf) 205:194–208
Bourin P, Bunnell BA, Casteilla L, Dominici M, Katz AJ, March KL, Redl H, Rubin JP, Yoshimura K, Gimble JM (2013) Stromal cells from the adipose tissue-derived stromal vascular fraction and culture expanded adipose tissue-derived stromal/stem cells: a joint statement of the International Federation for Adipose Therapeutics and Science (IFATS) and the International Society for Cellular Therapy (ISCT). Cytotherapy 15:641–648
Baer PC, Geiger H (2012) Adipose-derived mesenchymal stromal/stem cells: tissue localization, characterization, and heterogeneity. Stem Cells Int
Zuk PA, Zhu M, Ashjian P, De Ugarte DA, Huang JI, Mizuno H, Alfonso ZC, Fraser JK, Benhaim P, Hedrick MH (2002) Human adipose tissue is a source of multipotent stem cells. Mol Biol Cell 13:4279–4295
Largo RD, Tchang LA, Mele V, Scherberich A, Harder Y, Wettstein R, Schaefer DJ (2013) Efficacy, safety and complications of autologous fat grafting to healthy breast tissue: a systematic review. J Plast Reconstr Aesthet Surg 67:437–448
Kling RE, Mehrara BJ, Pusic AL, Young VL, Hume KM, Crotty CA, Rubin JP (2013) Trends in autologous fat grafting to the breast: a national survey of the american society of plastic surgeons. Plast Reconstr Surg 132:35–46
Rubin JP, Coon D, Zuley M, Toy J, Asano Y, Kurita M, Aoi N, Harii K, Yoshimura K (2012) Mammographic changes after fat transfer to the breast compared with changes after breast reduction: a blinded study. Plast Reconstr Surg 129:1029–1038
Del Vecchio DA, Del Vecchio SJ (2014) The graft-to-capacity ratio: volumetric planning in large-volume fat transplantation. Plast Reconstr Surg 133:561–569
Khouri RK, Rigotti G, Cardoso E, Khouri RK, Biggs TM (2014) Megavolume autologous fat transfer: part I. Theory and principles. Plast Reconstr Surg 133:550–557
Spear SL, Pittman T (2014) A prospective study on lipoaugmentation of the breast. Aesthet Surg J 34:400–408
Petit JY, Botteri E, Lohsiriwat V, Rietjens M, De Lorenzi F, Garusi C, Rossetto F, Martella S, Manconi A, Bertolini F, Curigliano G, Veronesi P, Santillo B, Rotmensz N (2012) Locoregional recurrence risk after lipofilling in breast cancer patients. Ann Oncol 23:582–588
Petit JY, Rietjens M, Botteri E, Rotmensz N, Bertolini F, Curigliano G, Rey P, Garusi C, De Lorenzi F, Martella S, Manconi A, Barbieri B, Veronesi P, Intra M, Brambullo T, Gottardi A, Sommario M, Lomeo G, Iera M, Giovinazzo V, Lohsiriwat V (2013) Evaluation of fat grafting safety in patients with intraepithelial neoplasia: a matched-cohort study. Ann Oncol 24:1479–1484
Rigotti G, Marchi A, Stringhini P, Baroni G, Galiè M, Molino AM, Mercanti A, Micciolo R, Sbarbati A (2010) Determining the oncological risk of autologous lipoaspirate grafting for post-mastectomy breast reconstruction. Aesthet Plast Surg 34:475–480
Zhang Y, Daquinag A, Traktuev DO, Amaya-Manzanares F, Simmons PJ, March KL, Pasqualini R, Arap W, Kolonin MG (2009) White adipose tissue cells are recruited by experimental tumors and promote cancer progression in mouse models. Cancer Res 69:5259–5266
Chaput B, Foucras L, Le Guellec S, Grolleau JL, Garrido I (2013) Recurrence of an invasive ductal breast carcinoma 4 months after autologous fat grafting. Plast Reconstr Surg 131:123–124
Smit JM, Tielemans HJ, de Vries B, Tuinder SM (2014) Recurrence of invasive ductal breast carcinoma 10 months after autologous fat grafting. J Plast Reconstr Aesthet Surg 67:e127–e128
Agha RF, Fowler AJ, Herlin C, Goodacre TE, Orgill DP (2014) Use of autologous fat grafting for breast reconstruction: a systematic review with meta-analysis of oncological outcomes. J Plast Reconstr Aesthet Surg 68:143–161
Gale KL, Takha EA, Ball G, Tan VKM, McCulley SJ, Macmilan RD (2015) A case controlled study of the oncological safety of fat grafting. Plast Reconstr Surg. Publish Ahead of Print
Registered clinical trials. http://www.clinicaltrial.gov. Accessed 24 May 2014
Yoshimura K, Sato K, Aoi N, Kurita M, Hirohi T, Harii K (2008) Cell-assisted lipotransfer for cosmetic breast augmentation: supportive use of adipose-derived stem/stromal cells. Aesthet Plast Surg 32:48–55
Zocchi ML, Zuliani F (2008) Bicompartmental breast lipostructuring. Aesthet Plast Surg 32:313–328
Delay E, Garson S, Tousson G, Sinna R (2009) Fat injection to the breast: technique, results, and indications based on 880 procedures over 10 years. Aesthet Surg J 29:360–376
Illouz YG, Sterodimas A (2009) Autologous fat transplantation to the breast: a personal technique with 25 years of experience. Aesthet Plast Surg 33:706–715
Panettiere P, Marchetti L, Accorsi D (2009) The serial free fat transfer in irradiated prosthetic breast reconstructions. Aesthet Plast Surg 33:695–700
Ueberreiter K, von Finckenstein JG, Cromme F, Herold C, Tanzella U, Vogt PM (2010) BEAULI™–a new and easy method for large-volume fat grafts. Handchir Mikrochir Plast Chir 42:379–385
Yoshimura K, Asano Y, Aoi N, Kurita M, Oshima Y, Sato K, Inoue K, Suga H, Eto H, Kato H, Harii K (2010) Progenitor-enriched adipose tissue transplantation as rescue for breast implant complications. Breast J 16:169–175
Caviggioli F, Maione L, Forcellini D, Klinger F, Klinger M (2011) Autologous fat graft in postmastectomy pain syndrome. Plast Reconstr Surg 128:349–352
Del Vecchio DA, Bucky LP (2011) Breast augmentation using preexpansion and autologous fat transplantation: a clinical radiographic study. Plast Reconstr Surg 127:2441–2450
Kamakura T, Ito K (2011) Autologous cell-enriched fat grafting for breast augmentation. Aesthet Plast Surg 35:1022–1030
Salgarello M, Visconti G, Rusciani A (2011) Breast fat grafting with platelet-rich plasma: a comparative clinical study and current state of the art. Plast Reconstr Surg 127:2176–2185
Cigna E, Ribuffo D, Sorvillo V, Atzeni M, Piperno A, Calò PG, Scuderi N (2012) Secondary lipofilling after breast reconstruction with implants. Eur Rev Med Pharmacol Sci 16:1729–1734
Gentile P, Orlandi A, Scioli MG, Di Pasquali C, Bocchini I, Curcio CB, Floris M, Fiaschetti V, Floris R, Cervell V (2012) A comparative translational study: the combined use of enhanced stromal vascular fraction and platelet-rich plasma improves fat grafting maintenance in breast reconstruction. Stem Cells Transl Med 1:341–351
Khouri RK, Eisenmann-Klein M, Cardoso E, Cooley BC, Kacher D, Gombos E, Baker TJ (2012) Brava and autologous fat transfer is a safe and effective breast augmentation alternative: results of a 6-year, 81-patient, prospective multicenter study. Plast Reconstr Surg 129:1173–1187
Pérez-Cano R, Vranckx JJ, Lasso JM, Calabrese C, Merck B, Milstein AM, Sassoon E, Delay E, Weiler-Mithoff EM (2012) Prospective trial of adipose-derived regenerative cell (ADRC)-enriched fat grafting for partial mastectomy defects: the RESTORE-2 trial. Eur J Surg Oncol 38:382–389
Auclair E, Blondeel P, Del Vecchio DA (2013) Composite breast augmentation: soft-tissue planning using implants and fat. Plast Reconstr Surg 132:558–568
Bonomi R, Betal D, Rapisarda IF, Kalra L, Sajid MS, Johri A (2013) Role of lipomodelling in improving aesthetic outcomes in patients undergoing immediate and delayed reconstructive breast surgery. Eur J Surg Oncol 39:1039–1045
Costantini M, Cipriani A, Belli P, Bufi E, Fubelli R, Visconti G, Salgarello M, Bonomo L (2013) Radiological findings in mammary autologous fat injections: a multi-technique evaluation. Clin Radiol 68:27–33
Fiaschetti V, Pistolese CA, Fornari M, Liberto V, Cama V, Gentile P, Floris M, Floris R, Cervelli V, Simonetti G (2013) Magnetic resonance imaging and ultrasound evaluation after breast autologous fat grafting combined with platelet-rich plasma. Plast Reconstr Surg 132:498e–509e
Hoppe DL, Ueberreiter K, Surlemont Y, Peltoniemi H, Stabile M, Kauhanen S (2013) Breast reconstruction de novo by water-jet assisted autologous fat grafting–a retrospective study. Ger Med Sci 11:Doc17
Peltoniemi HH, Salmi A, Miettinen S, Mannerström B, Saariniemi K, Mikkonen R, Kuokkanen H, Herold C (2013) Stem cell enrichment does not warrant a higher graft survival in lipofilling of the breast: a prospective comparative study. J Plast Reconstr Aesthet Surg 66:1494–1503
Khouri RK, Khouri RK, Rigotti G, Marchi A, Cardoso E, Rotemberg SC, Biggs TM (2014) Aesthetic applications of brava-assisted megavolume fat grafting to the breasts: a 9-year, 476-patient, multicenter experience. Plast Reconstr Surg 133:796–807
Maione L, Vinci V, Caviggioli F, Klinger F, Banzatti B, Catania B, Lisa A, Klinger M (2014) Autologous fat graft in postmastectomy pain syndrome following breast conservative surgery and radiotherapy. Aesthet Plast Surg 38:528–532
Small K, Choi M, Petruolo O, Lee C, Karp N (2014) Is there an ideal donor site of fat for secondary breast reconstruction? Aesthet Surg J 34:545–550
Uda H, Sugawara Y, Sarukawa S, Sunaga A (2014) Brava and autologous fat grafting for breast reconstruction after cancer surgery. Plast Reconstr Surg 133:203–213
Heymans O, Castus P, Grandjean FX, Van Zele D (2006) Liposuction: review of the techniques, innovations and applications. Acta Chir Belg 106:647–653
Girard A-C, Atlan M, Bencharif K, Gunasekaran MK, Delarue P, Hulard O, Lefebvre-d’Hellencourt C, Roche R, Hoareau L, Festy F (2013) New insights into lidocaine and adrenaline effects on human adipose stem cells. Aesthet Plast Surg 37:144–152
Moore JH, Kolaczynski JW, Morales LM, Considine RV, Pietrzkowski Z, Noto PF, Caro JF (1995) Viability of fat obtained by syringe suction lipectomy: effects of local anesthesia with lidocaine. Aesthet Plast Surg 19:335–339
Li K, Gao J, Zhang Z, Li J, Cha P, Liao Y, Wang G, Lu F (2013) Selection of donor site for fat grafting and cell isolation. Aesthet Plast Surg 37:153–158
Rohrich RJ, Sorokin ES, Brown SA (2004) In search of improved fat transfer viability: a quantitative analysis of the role of centrifugation and harvest site. Plast Reconstr Surg 113:391–397
Erdim M, Tezel E, Numanoglu A, Sav A (2009) The effects of the size of liposuction cannula on adipocyte survival and the optimum temperature for fat graft storage: an experimental study. J Plast Reconstr Aesthet Surg 62:1210–1214
Khouri RK, Rigotti G, Cardoso E, Khouri RKJ, Biggs TM (2014) Megavolume autologous fat transfer II. Plast Reconstr Surg 133:1369–1377
Del Vecchio D, Fichadia H (2012) Autologous fat transplantation—a paradigm shift in breast reconstruction, breast reconstruction—current techniques. In: Marzia Salgarello (ed) www.intechopen.com, p 217–236
Tambasco D, Arena V, Grussu F, Cervelli D (2013) Adipocyte damage in relation to different pressures generated during manual lipoaspiration with a syringe. Plast Reconstr Surg 131:645–646
Shiffman MA, Mirrafati S (2001) Fat transfer techniques: the effect of harvest and transfer methods on adipocyte viability and review of the literature. Dermatol Surg 27:819–826
Cheriyan T, Kai Kao H, Qiao X, Guo L (2014) Low harvest pressure enhances autologous fat graft viability. Plast Reconstr Surg 133:1365–1368
Condé-Green A, de Amorim NF, Pitanguy I (2010) Influence of decantation, washing and centrifugation on adipocyte and mesenchymal stem cell content of aspirated adipose tissue: a comparative study. J Plast Reconstr Aesthet Surg 63:1375–1381
Kaufman MR, Bradley JP, Dickinson B, Heller JB, Wasson K, O’Hara C, Huang C, Gabbay J, Ghadjar K, Miller TA (2007) Autologous fat transfer national consensus survey: trends in techniques for harvest, preparation, and application, and perception of short- and long-term results. Plast Reconstr Surg 119:323–331
Hoareau L, Bencharif K, Girard A-C, Gence L, Delarue P, Hulard O, Festy F, Roche R (2013) Effect of centrifugation and washing on adipose graft viability: a new method to improve graft efficiency. J Plast Reconstr Aesthet Surg 66:712–719
Coleman SR, Saboeiro AP (2007) Fat grafting to the breast revisited: safety and efficacy. Plast Reconstr Surg 119:775–787
Allen RJ, Canizares O, Scharf C, Nguyen PD, Thanik V, Saadeh PB, Coleman SR, Hazen A (2013) Grading lipoaspirate: is there an optimal density for fat grafting? Plast Reconstr Surg 131:38–45
Ferraro GA, De Francesco F, Tirino V, Cataldo C, Rossano F, Nicoletti G, D’Andrea F (2011) Effects of a new centrifugation method on adipose cell viability for autologous fat grafting. Aesthet Plast Surg 35:341–348
Kurita M, Matsumoto D, Shigeura T, Sato K, Gonda K, Harii K, Yoshimura K (2008) Influences of centrifugation on cells and tissues in liposuction aspirates: optimized centrifugation for lipotransfer and cell isolation. Plast Reconstr Surg 121:1033–1042
Delay E, Streit L, Toussoun G, La Marca S, Ho Quoc C (2013) Lipomodelling: an important advance in breast surgery. Acta Chir Plast 55:34–43
Zhu M, Cohen SR, Hicok KC, Shanahan RK, Strem BM, Yu JC, Arm DM, Fraser JK (2013) Comparison of three different fat graft preparation methods: gravity separation, centrifugation, and simultaneous washing with filtration in a closed system. Plast Reconstr Surg 131:873–880
Dos-Anjos Vilaboa S, Llull R, Mendel TA (2013) Returning fat grafts to physiologic conditions using washing. Plast Reconstr Surg 132:323–326
Chung MT, Paik KJ, Atashroo DA, Hyun JS, McArdle A, Senarath-Yapa K, Zielins ER, Tevlin R, Duldulao C, Hu MS, Walmsley GG, Parisi-Amon A, Momeni A, Rimsa JR, Commons GW, Gurtner GC, Wan DC, Longaker MT (2014) Studies in fat grafting: part I. Effects of injection technique on in vitro fat viability and in vivo volume retention. Plast Reconstr Surg 134:29–38
Spartalis ED, Tomos P, Konofaos P, Dimitroulis D, Kouraklis G (2014) Letter to Editor Breast reconstruction with autologous fat graft; does platelet-rich plasma affect patient’s survival? Int J Clin Exp Med 7:329–330
Lucarelli E, Beccheroni A, Donati D, Sangiorgi L, Cenacchi A, Vento AMD, Meotti C, Bertoja AZ, Giardino R, Fornasari PM, Mercuri M, Picci P (2003) Platelet-derived growth factors enhance proliferation of human stromal stem cells. Biomaterials 24:3095–3100
Kushida S, Kakudo N, Morimoto N, Hara T, Ogawa T, Mitsui T, Kusumoto K (2014) Platelet and growth factor concentrations in activated platelet-rich plasma: a comparison of seven commercial separation systems. J Artif Organs 17:186–192
Casteilla L, Planat-Benard V, Laharrague P, Cousin B (2011) Adipose-derived stromal cells: their identity and uses in clinical trials, an update. World J Stem Cells 3:25–33
Blaber SP, Webster RA, Hill CJ, Breen EJ, Kuah D, Vesey G, Herbert BR (2012) Analysis of in vitro secretion profiles from adipose-derived cell populations. J Transl Med 10:172–188
Puissant B, Barreau C, Bourin P, Clavel C, Corre J, Bousquet C, Taureau C, Cousin B, Abbal M, Laharrague P, Penicaud L, Casteilla L, Blancher A (2005) Immunomodulatory effect of human adipose tissue-derived adult stem cells: comparison with bone marrow mesenchymal stem cells. Br J Haematol 129:118–129
Zuk PA, Zhu M, Mizuno H, Huang J, Futrell JW, Katz AJ, Benhaim P, Lorenz HP, Hedrick MH (2001) Multilineage cells from human adipose tissue: implications for cell-based therapies. Tissue Eng 7:211–228
Tabit CJ, Slack GC, Fan K, Wan DC, Bradley JP (2012) Fat grafting versus adipose-derived stem cell therapy: distinguishing indications, techniques, and outcomes. Aesthet Plast Surg 36:704–713
Stig-Frederik Trojahn K, Anne F-N, Anders Bruun M, Jens Jørgen E, Roberto SO, Peter VG, Jens K, Maria K, Bo Sonnich R, Maj-Lis Møller T, Carsten T, Ebbe D, Krzysztof Tadeusz D (2013) Enrichment of autologous fat grafts with ex vivo expanded adipose tissue-derived stem cells for graft survival: a randomised placebo-controlled trial. Lancet 382:1113–1120
Del Vecchio D, Rohrich RJ (2012) A classification of clinical fat grafting: different problems, different solutions. Plast Reconstr Surg 130:511–522
Mestak O, Sukop A, Hsueh YS, Molitor M, Mestak J, Matejovska J, Zarubova L (2014) Centrifugation versus PureGraft for fatgrafting to the breast after breast-conserving therapy. World J Surg Oncol 12:178–186
Acknowledgments
Vincent Hivernaud, Franck Festy, Anne-Claire Girard and Regis Roche are employed by STEMCIS.
Author information
Authors and Affiliations
Corresponding author
Additional information
Anne-Claire Girard and Regis Roche have contributed equally.
Rights and permissions
About this article
Cite this article
Hivernaud, V., Lefourn, B., Guicheux, J. et al. Autologous Fat Grafting in the Breast: Critical Points and Technique Improvements. Aesth Plast Surg 39, 547–561 (2015). https://doi.org/10.1007/s00266-015-0503-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00266-015-0503-y