Abstract
Purpose
This study aimed to explore the value of dynamic magnetic resonance imaging (MRI) before and after operation for pelvic organ prolapse (POP).
Methods
Twenty-nine patients with POP (POP group) before and one month after surgery and 12 healthy women (control group) underwent static and dynamic MRI at rest and during straining (Valsalva maneuver). The preoperative MRI images of the POP and control groups were analyzed, and various measurements were recorded. Differences in measurements were compared between the POP and control groups; similarly, changes in measurements before and after operation were compared.
Results
In the POP group, MRI detected 29 anterior vaginal prolapses, 27 uterine prolapses, 1 rectoceles, and 14 pouch of Douglas hernias. In addition, 27 levator ani muscle defect and 15 pubocervical fascial defect cases were observed. The bladder-pubococcygeal line (B-PCL), uterus-pubococcygeal line (U-PCL), Douglas pouch-pubococcygeal line, the length of the hiatus, the descent of the levator plate, levator hiatus size, levator plate angle, iliococcygeus angle, and urethral inclination angle (UA) were larger in the POP group than in the control group. The pelvic organs’ positions returned to normal postoperatively in 9 of 21 cases. The B-PCL, U-PCL, and UA were smaller post-surgery than pre-surgery.
Conclusions
Static and dynamic MRI can comprehensively evaluate morphological and functional changes of the pelvic floor postoperatively.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Pelvic organ prolapse (POP) is an abnormal position and function of pelvic organs caused by weakness of the pelvic floor muscles and fascia [1]. It is a common condition in middle-aged and older women [2]. POP often involves multiple organs and chambers, and a comprehensive evaluation before surgery is particularly important [3]. Presently, the clinical diagnosis of POP mainly depends on pelvic organ prolapse quantification (POP-Q) [4]. However, for patients with POP with complex or multiple site defects, routine clinical examination is insufficient to comprehensively evaluate pelvic floor function, thus resulting in incomplete treatment and a high postoperative recurrence rate [5, 6].
Magnetic resonance imaging (MRI) can provide complete functional, anatomical information of the pelvic floor because of its imaging advantages, such as arbitrary orientation, excellent soft-tissue resolution, and no use of radiation [7]. This study aimed to determine the value of static and dynamic MRI in preoperative diagnosis and postoperative evaluation of patients with POP. The present findings provide an objective basis for individualized treatment of different patients from the perspective of medical imaging.
Materials and methods
Clinical data
From December 2011 to January 2014, 29 patients with POP underwent surgery in our hospital (POP group), with a mean age of 61.8 ± 10.4 years (range, 42–84 years), and a parity range of 1 to 7. The inclusion criteria included hospitalized patients with pelvic organ prolapse and no history of pelvic surgery. The main clinical manifestation was an orificium vaginae mass prolapse; the size of the prolapsed mass varied. The mass is painful when walking, and it can be restored without treatment in some cases. The diagnostic criteria for clinical admission in the POP group were based on the POP-Q system formulated by the International Continence Society [4] (Table 1). Twelve patients (mean age: 61.0 ± 9.9 years; range: 52–89 years) in the asymptomatic control group included hospitalized patients with no pelvic floor dysfunction and no history of pelvic surgery. Informed consent was obtained from all patients.
MRI examinations
All participants who underwent MRI examinations had no history of coronary heart disease or angina pectoris, cardiac pacemaker or intravascular stent, post-traumatic metal internal fixation, mental or nervous system diseases, or intrauterine device. Dynamic MRI examinations and static high-resolution volume scans were performed in the POP group before and one month after surgery and in the control group. Before the MRI examinations, the same physician trained all participants in the Valsalva maneuver (maximum abdominal pressure) until they understood the main points of the procedure. In addition, they were instructed to urinate 30 min before the examination. MR (magnetic resonance) images were captured with a Philips Achieva 1.5-Tesla scanner with a five-channel phased-array coil (Best, the Netherlands). The patient’s head was advanced in the supine position, and the knees were slightly flexed and had a valgus deformity. Static MRI was performed during natural breathing and included T2-weighted three-dimensional imaging in axial planes, with the following parameters: section thickness, 1 mm; repetition time, 2000 ms; echo time, 100 ms; matrix, 352 × 346; field of view, 35 cm × 35 cm; collection times, twice; and scanning time, 9–11 min. Dynamic MRI was performed with breath-holding during the Valsalva maneuver. T2-weighted single-shot fast spin-echo sequences in the sagittal, coronal, and axial planes were obtained, with the following parameters: section thickness, 4 mm; intersection gap, 0 mm; repetition time, 1000 ms; echo time, 80 ms; matrix, 280 × 250; and field of view, 35 cm × 35 cm.
MRI analysis
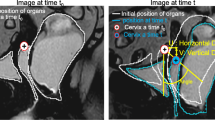
The locations of the POP and supporting structure of the pelvic floor (levator ani muscle and fascia) were evaluated. Three reference lines were determined on the median sagittal images at rest and during the Valsalva maneuver [8] as follows: 1) The pubococcygeal line (PCL), which is the line between the inferior margin of the pubic symphysis and the sacrococcygeal joint; 2) the length of the hiatus (H line), which is the distance from the posterior inferior symphysis to the posterior annular rectal fibers; and 3) the descent of the levator plate (M line), which is the vertical line from the attachment point of the posterior annular rectal fibers to the PCL. B-PCL, U-PCL, and D-PCL are the vertical distances from the lowest margin of the bladder, uterine neck, and Douglas’ cul-de-sac to PCL, respectively [9]. The diagnostic criteria for a relaxed pelvic floor were H line ≥ 6 cm (6 − 8 cm mild, 8 − 10 cm moderate, > 10 cm severe) and M line ≥ 2 cm (2 − 4 cm mild, 4 − 6 cm moderate, > 6 cm severe) [10]. The diagnostic criteria for POP were as follows: the farthest end of the pelvic organ was below the PCL (represented by a negative value); the greater the negative value, the lower the value [11]. The levator hiatus size (LHS) was calculated using the formula LHS = π × R1 × R2/4, where R1 and R2 are the maximum anteroposterior diameter and maximum transverse diameter of the levator ani muscle hiatus on the transverse section of the inferior margin of the pubic symphysis, respectively [12]. The levator plate angle (LPA) is the angle between the levator ani plate and the PCL in the median sagittal position [13]. The iliococcygeus angle (ICA) is the angle between the coronal iliococcygeal muscle and the horizontal plane of the body, and the left and right sides are measured separately [14]. The urethral inclination angle (UA) is the angle between the urethral and longitudinal axes of the body [15]. The reference values for the LPA, ICA, and UA during the Valsalva maneuver are 11.7 ± 4.8, 33.4 ± 8.2, and 30 degrees, respectively [8]. The MRI parameters were measured at rest and during the Valsalva maneuver (Fig. 1).
Schematic diagram of the B-PCL, U-PCL, D-PCL, H line, M line, LHS, LPA, R-ICA, and UA. a, b, c, d, and f: Sagittal T2-weighted magnetic resonance images; c and e: Axial T2-weighted magnetic resonance image. B-PCL: bladder-pubococcygeal line; U-PCL: uterus-pubococcygeal line; D-PCL: Douglas pouch-pubococcygeal line; H line: the length of the hiatus; M line: the descent of the levator plate; LHS: levator hiatus size; LPA: levator plate angle; R-ICA: Right-iliococcygeus angle; UA: urethral inclination angle
Statistical analyses
Statistical analysis was performed using SPSS 16.0. The normality of general clinical data and imaging measurement parameters were tested. Independent sample t-test was used to analyze normal or approximately normal distribution data, the rank-sum test was used to analyze skewed distribution data, and Pearson correlation analysis was used to analyze normally and non-normally distributed data. Spearman rank correlation analysis was used to measure the rank correlation strength between variables, with | r |> 0.8 indicating high correlation, | r |= 0.6–0.8 indicating moderate correlation, and | r |< 0.6 indicating low correlation. Statistical significance was set at P < 0.05.
Results
Baseline characteristics of the POP and control groups
In the POP group, 29 patients underwent dynamic MR examination of the pelvic floor before the operation, and 21 patients underwent MRI follow-up 1 month after the surgery. Eight patients were lost to follow-up. Of the 21 patients followed up after surgery, 8 patients underwent Prolift pelvic floor reconstruction, and 13 patients underwent traditional surgery (total hysterectomy and anterior and posterior colporrhaphy). There were no significant differences in age, parity, or bodyweight between the POP and control groups (P > 0.05) (Table 2).
Preoperative MRI analysis of the POP and control groups
MRI clearly showed levator ani muscle and fascia defects in patients with POP. In this group, there were 29 cases of anterior vaginal prolapse, 27 cases of uterine prolapse, 27 cases of posterior vaginal prolapse, one case of rectal prolapse, and 14 cases of hernia of the cul-de-sac of Douglas. Twenty-seven cases of weakness and defect of the pelvic floor muscle were observed (93.1%); among them, weakness and defect of the puborectalis were found in 3 cases (10.3%) weakness and defect of the pubococcygeus in 24 cases (82.8%), and weakness and defect of the iliococcygeus in 16 cases (55.2%). The weak and injured part of the bilateral levator ani muscle was located at the beginning of the tendon arch of levator ani muscle, and the defect of the puborectalis was mostly located at the beginning or the coordinate direction of the internal obturator muscle. Fifteen cases of weak and defective bladder and cervical fascia defects were observed (51.7%): right defect in 13 cases (44.8%) (Fig. 2a), left defect in 1 case (3.4%), and bilateral defect in 1 case (3.4%).
a: A 60-year-old patient had anterior vaginal, posterior vaginal, and uterine prolapse (POP-Q IV); the right posterior wall of the bladder bulged backward, suggesting a defect in the right bladder and cervical fascia. b, c: A 51-year-old patient had uterine and anterior vaginal prolapse (POP-Q III) and posterior vaginal prolapse (POP-Q II). b: At the resting state, the shape of bilateral iliococcygeus was symmetrical. c: During the Valsalva maneuver, the iliococcygeus was asymmetrical, thinned, and elongated, and bilateral discontinuous imaging revealed the “crack phenomenon” (arrow). d, e: A 54-year-old patient had uterine prolapse (POP-Q II) and anterior vaginal prolapse (POP-Q III). MR images show the weakness of puborectalis. d: The fissure of the inferior levator anus muscle was in the shape of “V” at rest. e: During the Valsalva maneuver, the bilateral puborectalis became thinner and dilated laterally, the fissure of the levator ani muscle was in an “o” shape, and the local fat was protruding into the defect. f, g: A 66-year-old patient had anterior vaginal, posterior vaginal, and uterine prolapse (POP-Q IV). MR sagittal images show that the iliococcygeus moved straight at rest, and the pelvic cavity expanded downwards during the Valsalva maneuver, resembling a hook shape. MR: magnetic resonance
Continuous observation on coronal MR images showed that in resting state, bilateral levator ani muscles of all patients were symmetrical, curved, and convex upward, showing imbricate shape(Fig. 2b). During the Valsalva maneuver, the bilateral levator ani muscles of most patients with POP were asymmetrical, thinned, elongated, curved downward, and funnel-shaped, and discontinuous imaging of the levator ani muscle could be seen in some cases, with a “crack phenomenon” (Fig. 2c). The axial view of the inferior margin of the pubic symphysis showed that the LHS during the Valsalva maneuver in the POP group was larger than that in the resting state (Fig. 2d, e). Continuous observation in the sagittal images indicated that the iliococcygeus had an arc-shaped convex shape at rest and a downward bulge during the Valsalva maneuver, resembling a fishhook shape (Fig. 2f, g). In the control group, the puborectal muscle was "U" shaped in the lower part of the pubis, the pubococcygeal muscle was "V" shaped in the middle part of the pubis, and the iliococcygeus muscle was an inverted "V" in the superior part of the pubis, and there were no significant changes in the bilateral levator ani muscle shape and size at rest and during the Valsalva maneuver.
Correlation between the LPA and POP
In patients with prolapse, there was a correlation between LPA and M line, D-PCL, or U-PCL. The correlation between LPA and M line was significant (P < 0.001, r = 0.844). LPA had a moderate correlation with D-PCL (P < 0.001, r = 0.606) and a low correlation with U-PCL (P = 0.004, r = − 0.516). However, there was no correlation between LPA and B-PCL (P > 0.05) (Table 3).
Comparisons of imaging measurements at rest and during the Valsalva maneuver between the POP and control groups before operation
As shown in Table 4, there were significant differences in B-PCL, U-PCL, H line, M line, LHS, LPA, and UA between the POP and control groups at rest and during the Valsalva maneuver. The differences in the L-ICA and R-ICA between the POP and control groups were not significant at rest but were statistically significant during the Valsalva maneuver.
Post-surgery changes in imaging measurements at rest and during Valsalva maneuver in the POP group
As shown in Table 5, there were statistically significant differences between the preoperative and postoperative measurements of B-PCL and U-PCL at rest and during the Valsalva maneuver (P < 0.01). LHS showed a statistically significant difference between preoperative and postoperative measurements at rest (P < 0.01); however, there was no significant difference during the Valsalva maneuver. The UA measurements showed no significant difference pre- and postoperatively at rest (P > 0.05); however, there was a significant difference during the Valsalva maneuver. There was no significant difference in the values of H line, M line, LPA, R-ICA, and L-ICA between preoperation and postoperation at rest and during the Valsalva maneuver.
Comparative analysis of MR images in the POP group before and after operation
Postoperative pelvic floor dynamic MRI showed that the bladder neck was below the PCL during the Valsalva maneuver in 12 patients (57.1%) (Fig. 3). The cervix was below the PCL in four patients (1.9 cm, 0.5 cm, 1.1 cm, and 1.1 cm, respectively), and the bladder and cervix (vaginal stump) were above the PCL in the remaining patients (Fig. 4). During the outpatient follow-up, eight patients reported persistent symptoms of urinary incontinence, such as persistent urination, or urination while laughing or coughing. Gynecological examination revealed four cases of mild anterior vaginal prolapse and no cases of uterine prolapse.
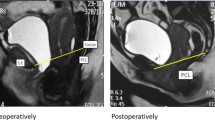
Preoperative dynamic MR on the midsagittal plane. a: The bladder neck decreased and developed into second-degree cystocele (red arrow) and third-degree uterine prolapse (green arrow) during Valsalva maneuver b: After the operation (hysterectomy + anterior and posterior colporrhaphy + vaginal constriction + high suspension of sacral ligament of vaginal stump), there was still mild stress incontinence. The bladder neck was located at 2.9 cm below the PCL, and UA increased. MR: magnetic resonance; PCL: pubococcygeal line; UA: urethral inclination angle
a: Preoperative dynamic MR on midsagittal images shows that the bladder neck decreased and developed into second-degree cystocele (red arrow) and third-degree uterine prolapse (green arrow) during Valsalva maneuver b: postoperative (Prolift for pelvic floor reconstruction (preservation of uterus) + inside-out transobturator vaginal tape) patients experienced no uncomfortable symptoms; dynamic MR shows that the bladder neck and cervix were above the PCL. MR: magnetic resonance; PCL: pubococcygeal line
Discussion
In this study, in the patients in the POP group, pelvic floor muscle weakness and defect occurred in 27 cases, including puborectal muscle weakness and defect in 3 cases, pubococcygeal muscle weakness and defect in 24 cases, and iliococcygeus muscle weakness and defect in 16 cases. Among them, the pubococcygeal muscle accounts for the highest proportion of all weaknesses and defects, which confirms the muscle stretching hypothesis of Krofta [16] that the pubococcygeal muscle is the most vulnerable muscle.
Between the pubic bone and paries anterior vaginae of the vagina is the pubovesicocervical fascia, which supports the bladder. The fascia of the levator anus muscle and the fascia of the obturator muscle constitute the tendinous arch of pelvic fascia and arcus tendineus musculi levatoris ani, respectively. The tendinous arch of pelvic fascia provides the lateral fulcrum for the paries anterior vaginae, and the arcus tendineus musculi levatoris ani provides support for the levator anus muscle. As the initial part of the levator anus muscle, the arcus tendineus musculi levatoris ani can be distinguished on MRI [17, 18]. In this study, it was observed that 5 patients had defects of the levator anus muscle at the beginning of the arcus tendineus musculi levatoris ani. In the POP group, there were 15 cases with weak fascia defect of the pubovesicocervical fascia: 13 cases with right defect, 1 case with left defect, and 1 case with bilateral defect. In addition, it was observed that the wall of the urinary bladder of 2 patients with prolapse was obviously protruding downward, and the symptoms of stress urinary incontinence were obvious, but no obvious weakness and defect of the levator anus muscle were found. It was speculated that the MRI manifestations and clinical symptoms were mainly related to the defect of the pubovesicocervical fascia supporting the bladder.
Dynamic MRI can quantify the diagnosis of POP and evaluate the function of the levator ani muscle. At present, the most widely used reference marker line in pelvic floor dynamic MRI research is the PCL line [19]. The LHS and H line can indirectly reflect the degree of weakness of the pelvic floor, and the M line can reflect the relaxation of the posterior pelvic floor muscles [20]. When the levator plate is damaged, the LPA increases; when the iliococcygeus and its anchored fascia and ligaments are torn and damaged, the ICA increases [13, 21]. The UA of healthy women is often less than 30 degrees, and the UA of patients with stress incontinence is often more than 30 degrees [22].
We investigated the correlation between the LPA and the decline of pelvic tissues and organs in this study and observed a significant correlation between the LPA and descent of the levator plate, uterus, and pouch of Douglas (P < 0.05; r = 0.844, − 0.516, and − 0.606, respectively). The larger the LPA, the more severe the prolapse of pelvic organs; this finding is consistent with the results of Li [23]. Therefore, the LPA can reflect the functional state of the levator plate and maintaining the normal position of the levator plate plays an important role in preventing POP.
In healthy women, the pelvic organs (bladder, uterus, small intestine, and sigmoid colon) are located above the PCL line at rest or during the Valsalva maneuver. In patients with prolapse, due to the weakness and defect of the levator ani muscle, the injury and loss of the supporting structure around the organs, or the weakness of the whole pelvic floor, the pelvic organs break away from their normal anatomical positions and fall below the PCL line [8]. In this study, MRI images were compared and analyzed for each measurement index in the prolapse and control groups at rest and during the Valsalva maneuver. In the control group, all pelvic organs of 11 patients were above the PCL line at rest or during the Valsalva maneuver. The bladder neck and cervix of patients in the POP group were all below the PCL line during the Valsalva maneuver. There were significant differences in the H line, M line, LPA, LHS, and UA between the two groups (P < 0.05). In addition, Handa et al. [24] showed that the degree of injury of the levator ani muscle was related to the degree of POP, probably due to the larger levator hiatus and weaker pelvic muscles after levator avulsion. Consistently, in this study, the preoperative values of all measurements in the POP group were higher than those in the control group, indicating that the impairment of levator ani muscle function and pelvic floor weakness in POP patients was more severe than that in the age-matched asymptomatic women. Notably, some participants in the control group similarly had defects in the levator ani muscle, fascia, and pelvic floor relaxation, indicating that some middle-aged and older women have potential pelvic floor weakness and may develop POP (Fig. 5).
A 57-year-old patient in the asymptomatic control group. a, b:MR images show the weakness and defect of the right pubococcygeal muscle and the left iliococcygeus muscle. c: MR sagittal images show that the pelvic organs are located above the PCL line during the Valsalva maneuver.MR: magnetic resonance
In this study, POP measurements in the POP group were compared and analyzed before and after surgery. It was observed that the measured values of U-PCL and B-PCL after the operation were smaller than those before the operation (P < 0.05). There was no significant difference between the preoperative and postoperative measurements of the H and M lines at rest or during the Valsalva maneuver. The postoperative measurements of LPA, and L/R-ICA were lower than those before the operation; however, the difference was not statistically significant (P > 0.05). The LHS was smaller after the operation than before the operation at rest (P < 0.05). The LHS during the Valsalva maneuver was not significantly different between pre-and postoperation (P > 0.05). UA was significantly reduced after surgery compared with before operation during the Valsalva maneuver (P < 0.05). The results are consistent with those in a comparative study of preoperative and postoperative MR images in patients with prolapse by Kasturi et al. [25]. These findings indicate that the position of the vesical uterus (or vaginal stump) was significantly improved after surgical treatment compared with before surgery. However, there were no significant changes in the H and M lines and minimal changes in LPA, and L/R-ICA after surgery compared with those before surgery; the finding indirectly suggests that the injury of the pelvic floor muscle and the weakness of the posterior pelvic floor structure could not be recovered by pelvic floor reconstruction alone. Indeed, the pathogenesis of pelvic floor dysfunction is extremely complicated and involves other tissues’ metabolism, neurophysiology, biomechanics, endocrine hormones, and other aspects.
We observed that the UA of postoperative POP patients was significantly lower than that before the operation; however, it was still greater than 30 degrees, indicating that the urethra still had high mobility. Clinical symptoms were assessed in the outpatient follow-up, and eight patients reported persistent symptoms of mild urinary incontinence, such as persistent urination and urination while laughing or coughing. Notably, Mahfouz et al. [26] observed that dynamic MRI of the pelvic floor was very helpful for postoperative evaluation of surgical repair for POP and demonstrated that there was a close correlation between UA and postoperative urinary incontinence symptoms.
This study has several limitations. First, most patients in the POP group had moderate to severe symptoms, and no further stratified analyses were conducted. Second, due to the small number of patients who underwent uterine-sparing surgery in this study, further studies with a large sample size are needed to determine the success rate of uterine-sparing surgery in POP patients.
In conclusion, static and dynamic MRI of the pelvis can accurately display changes in the position of prolapsed organs and the morphology of the pelvic floor muscles and fascia before and after surgery. In addition, they can be used for evaluating the function of the levator ani muscle, and especially the evaluation of multiple defects in the pelvis. MRI is also more useful than physical examination for detecting abnormalities in patients who remain symptomatic after surgery. Thus, dynamic MRI can guide the development of preoperative treatment plans and assess postoperative outcomes in patients with pelvic organ prolapse.
Data availability
The datasets used or analyzed during the current study are available from the corresponding author on reasonable request.
References
Bradley CS (2018) Progress toward understanding pelvic organ prolapse. Am J Obstet Gynecol 218:267-268. doi: https://doi.org/10.1016/j.ajog.2018.01.042
Huang L, Zhao Z, Wen J, Ling W, Miao Y, Wu J (2020) Cellular senescence: A pathogenic mechanism of pelvic organ prolapse (Review). Mol Med Rep 22:2155-2162. doi: https://doi.org/10.3892/mmr.2020.11339
Iglesia CB, Smithling KR (2017) Pelvic Organ Prolapse. Am Fam Physician 96:179-185
Haylen BT, Maher CF, Barber MD, Camargo S, Dandolu V, Digesu A, Goldman HB, Huser M, Milani AL, Moran PA, Schaer GN, Withagen MI (2016) An International Urogynecological Association (IUGA) / International Continence Society (ICS) Joint Report on the Terminology for Female Pelvic Organ Prolapse (POP). Neurourol Urodyn 35:137-168. doi: https://doi.org/10.1002/nau.22922
Easley DC, Abramowitch SD, Moalli PA (2017) Female pelvic floor biomechanics: bridging the gap. Curr Opin Urol 27:262-267. doi: https://doi.org/10.1097/MOU.0000000000000380
Pannu HK, Javitt MC, Glanc P, Bhosale PR, Harisinghani MG, Khati NJ, Mitchell DG, Nyberg DA, Pandharipande PV, Shipp TD, Siegel CL, Simpson L, Wall DJ, Wong-You-Cheong JJ (2015) ACR Appropriateness Criteria pelvic floor dysfunction. J Am Coll Radiol 12:134-142. doi: https://doi.org/10.1016/j.jacr.2014.10.021
Salvador JC, Coutinho MP, Venâncio JM, Viamonte B (2019) Dynamic magnetic resonance imaging of the female pelvic floor-a pictorial review. Insights Imaging 10:4. doi: https://doi.org/10.1186/s13244-019-0687-9
El Sayed RF, Alt CD, Maccioni F, Meissnitzer M, Masselli G, Manganaro L, Vinci V, Weishaupt D; ESUR and ESGAR Pelvic Floor Working Group (2017) Magnetic resonance imaging of pelvic floor dysfunction - joint recommendations of the ESUR and ESGAR Pelvic Floor Working Group. Eur Radiol 27:2067-2085. doi: https://doi.org/10.1007/s00330-016-4471-7
Maccioni F, Al Ansari N, Buonocore V, Mazzamurro F, Indinnimeo M, Mongardini M, Catalano C (2016) Prospective Comparison between two different magnetic resonance defecography techniques for evaluating pelvic floor disorders: air-balloon versus gel for rectal filling. Eur Radiol 26:1783-1791. doi: https://doi.org/10.1007/s00330-015-4016-5
Kobi M, Flusberg M, Paroder V, Chernyak V (2018) Practical guide to dynamic pelvic floor MRI. J Magn Reson Imaging 47:1155-1170. doi: https://doi.org/10.1002/jmri.25998
Reiner CS, Weishaupt D (2013) Dynamic pelvic floor imaging: MRI techniques and imaging parameters. Abdom Imaging 38:903-911. doi: https://doi.org/10.1007/s00261-012-9857-7
El-Haieg DO, Madkour NM, Basha MAA, Ahmad RA, Sadek SM, Al-Molla RM, Tantwy EF, Almassry HN, Altaher KM, Mahmoud NEM, Aly SA (2019) Magnetic resonance imaging and 3-dimensional transperineal ultrasound evaluation of pelvic floor dysfunction in symptomatic women: a prospective comparative study. Ultrasonography 38:355-364. doi: https://doi.org/10.14366/usg.19007
Hsu Y, Summers A, Hussain HK, Guire KE, Delancey JO (2006) Levator plate angle in women with pelvic organ prolapse compared to women with normal support using dynamic MR imaging. Am J Obstet Gynecol 194:1427-1433. doi: https://doi.org/10.1016/j.ajog.2006.01.055
Ansquer Y, Fernandez P, Chapron C, Frey C, Bennis M, Roy C, Salomon L, Mandelbrot L, Carbonne B (2006) Static and dynamic MRI features of the levator ani and correlation with severity of genital prolapse. Acta Obstet Gynecol Scand 85:1468-1475. doi: https://doi.org/10.1080/00016340600984837
Li M, Wang B, Liu X, Qiao P, Jiao W, Jiang T (2020) MR defecography in the assessment of anatomic and functional abnormalities in stress urinary incontinence before and after pelvic reconstruction. Eur J Radiol 126:108935. doi: https://doi.org/10.1016/j.ejrad.2020.108935
Krofta L, Havelková L, Urbánková I, Krčmář M, Hynčík L, Feyereisl J (2017) Finite element model focused on stress distribution in the levator ani muscle during vaginal delivery. Int Urogynecol J 28:275-284. doi: https://doi.org/10.1007/s00192-016-3126-1
Lamblin G, Delorme E, Cosson M, Rubod C (2016) Cystocele and functional anatomy of the pelvic floor: review and update of the various theories. Int Urogynecol J 27:1297-1305. doi: https://doi.org/10.1007/s00192-015-2832-4
Li JR, Lei L, Luo N, Chen N, Xu HT, Xin Hu, Song Y, Wu Y (2021) Architecture of female urethral supporting structures based on undeformed high-resolution sectional anatomical images. Anat Sci Int 96:30-41. doi: https://doi.org/10.1007/s12565-020-00554-y
García del Salto L, de Miguel Criado J, Aguilera del Hoyo LF, Gutiérrez Velasco L, Fraga Rivas P, Manzano Paradela M, Díez Pérez de las Vacas MI, Marco Sanz AG, Fraile Moreno E (2014) MR imaging-based assessment of the female pelvic floor. Radiographics 34:1417-1439. doi: https://doi.org/10.1148/rg.345140137
Schmid FA, Gomolka RS, Hötker AM, Boss A, Kessler TM, Rossi C, Eberli D (2020) Evaluation of Urinary Sphincter Function by Rapid Magnetic Resonance Diffusion Tensor Imaging. Int Neurourol J 24:349-357. doi: https://doi.org/10.5213/inj.2040208.104
Picchia S, Rengo M, Bellini D, Caruso D, Pironti E, Floris R, Laghi A (2019) Dynamic MR of the pelvic floor: Influence of alternative methods to draw the pubococcygeal line (PCL) on the grading of pelvic floor descent. Eur J Radiol Open 6:187-191. doi: https://doi.org/10.1016/j.ejro.2019.05.002
Su D, Wen Y, Lin Q (2021) Particle Swarm Algorithm-Based Analysis of Pelvic Dynamic MRI Images in Female Stress Urinary Incontinence. Contrast Media Mol Imaging 2021:8233511. doi: https://doi.org/10.1155/2021/8233511
Li N, Cui C, Cheng Y, Wu Y, Yin J, Shen W (2018) Association between Magnetic Resonance Imaging Findings of the Pelvic Floor and de novo Stress Urinary Incontinence after Vaginal Delivery. Korean J Radiol 19:715-723. doi: https://doi.org/10.3348/kjr.2018.19.4.715
Handa VL, Roem J, Blomquist JL, Dietz HP, Muñoz A (2019) Pelvic organ prolapse as a function of levator ani avulsion, hiatus size, and strength. Am J Obstet Gynecol 221:41.e1-41.e7. doi: https://doi.org/10.1016/j.ajog.2019.03.004
Kasturi S, Lowman JK, Kelvin FM, Akisik FM, Terry CL, Hale DS (2010) Pelvic magnetic resonance imaging for assessment of the efficacy of the Prolift system for pelvic organ prolapse. Am J Obstet Gynecol 203:504.e1-504.e5. doi: https://doi.org/10.1016/j.ajog.2010.06.034
Mahfouz W, Elmissiry M, Gomaa SA, Koraitim A, Hassan H (2020) Evaluation of the effect of abdominal sacrocolpopexy (ASC) on urethral anatomy and continence mechanism using dynamic MRI. Int Urol Nephrol 52:1429-1435. doi: https://doi.org/10.1007/s11255-020-02444-8
Acknowledgements
None.
Funding
This research did not receive any funding.
Author information
Authors and Affiliations
Contributions
HZ participated in data collection, analysis, manuscript writing, and approval; ZW participated in project development, data collection, analysis, and approval; BZ participated in project development, data collection, analysis, manuscript writing and approval; XX participated in project development, data analysis; JW participated in analysis.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest regarding this research.
Ethical approval
This manuscript has not been published or presented elsewhere in part or in entirety and is not under consideration by another journal. All study participants provided informed consent, and the study design was approved by the appropriate ethics review board.
Consent to participate and consent to publish
Informed consent was obtained from all individual participants included in the study. The authors affirm that the participants provided informed consent for publication of the images in Figs. 1, 2, 3, 4, and 5.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhang, H., Wang, Z., Xiao, X. et al. Dynamic magnetic resonance imaging evaluation before and after operation for pelvic organ prolapse. Abdom Radiol 47, 848–857 (2022). https://doi.org/10.1007/s00261-021-03361-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00261-021-03361-9