Abstract
Introduction
Treatment of pelvic organ prolapse (POP) associated with stress urinary incontinence (SUI) is a surgical challenge. Surgeons may perform combined prolapse and incontinence surgery or may correct prolapse first and evaluate incontinence afterwards. We present a prospective study to evaluate the effect of abdominal sacrocolpopexy (ASC) on urethral anatomy and continence using dynamic magnetic resonance imaging (MRI).
Methods
Twenty females with concomitant apical prolapse and SUI due to urethral hypermobility were included. Patients with intrinsic sphincteric deficiency (ISD) were excluded. All patients underwent ASC operation as a sole treatment without anti-incontinence procedure. Patients were informed they may need anti-incontinence procedure afterwards. Symptom-specific questionnaires assessing prolapse, incontinence, sexual function and quality of life (QoL), dynamic MRI and pressure flow urodynamic study were administered before and after surgery.
Results
Mean age was 53 years. All patients had apical prolapse; four with cystocele, and five with rectocele. Urethral hypermobility was positive in all patients. After performing ASC, all patients reported significant improvement of all prolapse and incontinence questionnaires as well as QoL and sexual function. Significant improvement of incontinence parameters on dynamic MRI (bladder neck descent, posterior urethrovesical angle and urethral inclination angle) was observed after ASC. Similarly, significant change in the position of the leading edge of prolapse and anorectal junction was observed.
Conclusions
In patients with prolapse and urethral hypermobility, ASC may return bladder neck and urethral anatomy towards normal as proved by dynamic MRI. However, further studies on larger number of patients with longer follow-up period are required.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Pelvic organ prolapse (POP) is a condition in which the urinary bladder, uterus, bowel or rectum protrudes into the vagina [1, 2]. Although various procedures have been tried to correct apical prolapse, until now, the most successful option has been abdominal sacrocolpopexy (open or laparoscopic), with success rates approaching 93–99% [3]. It is considered an excellent choice, because it maximizes functional vaginal length and attains a near-normal vaginal axis.
POP usually co-exists with other urinary tract disorders, with stress urinary incontinence (SUI) being the most socially unacceptable condition [1, 2]. SUI can be manifest or occult (i.e., evident only upon reduction of the POP) and in some cases, can develop after surgical POP repair (denovo SUI). Surgical challenges exist for urogynecologists when they treat POP with or without concomitant SUI. One of the challenging scenarios is treating females with POP without SUI, whether to perform prophylactic anti-incontinence procedure or not in these continent women.
The other challenging scenario occurs when both POP and SUI are co-existing. Two strategies are present: a one-step strategy (combined prolapse and incontinence surgery) and a two-step strategy (correct the prolapse first and evaluate incontinence afterwards). Evidence supporting the value of combination surgery is still limited in these women [4, 5]. Nevertheless, many physicians combine incontinence surgery during POP repair and there are probably large regional differences in treatment [6, 7].
The aim of this study is to evaluate the effect of abdominal sacrocolpopexy (ASC) on the urethral anatomy and continence mechanism using dynamic magnetic resonance imaging (MRI) and correlate this change with the clinical and urodynamic outcomes.
Materials and methods
Twenty female patients with symptomatic vaginal apical prolapse (vault or uterine) stage II or more on pelvic organ prolapse quantification (POP-Q) system and concomitant SUI due to urethral hypermobility were included in the trial. Hypermobility was diagnosed by observing the mobile urethra during examination with Valsalva maneuver, and preoperative urodynamics showing VLPP > 60 cmH2O. Patients presented to the Female Urology and Voiding Dysfunction Clinic at our Institute from April 2017 to May 2018. Our local institutional medical ethics review board approved the study, and informed consent was taken from all patients before enrollment in the study.
Exclusion criteria included preoperative fixed urethra or intrinsic sphincteric deficiency (ISD), preoperative valsalva leak point pressure (VLPP) < 60 cmH2O, patients with benign or malignant uterine or cervical lesions, active pelvic inflammatory diseases (PID) and previous anti-POP or anti-SUI surgeries.
All patients underwent open ASC operation as a sole treatment without concomitant anti-SUI procedure. Patients were informed that those who will present with persistent postoperative SUI will be offered anti-incontinence procedure at least 6 months postoperatively. All patients were subjected to clinical history taking, physical and gynecological examination, routine laboratory investigations. Symptom-specific questionnaires were administered in the form of Urogenital Distress Inventory (UDI-6), International Consultation on Incontinence Questionnaire short form (ICIQ-UI-SF) to evaluate incontinence, and Pelvic Organ Prolapse and Incontinence Sexual Function questionnaire (PISQ-12) short form to evaluate sexual function. All patients underwent urodynamic study (UDS) in the form of free uroflowmetry with PVR estimation, and pressure flow study with estimation of VLPP before and after surgery. The supervisor who was responsible for the follow-up assessment was blinded to the patients' baseline data.
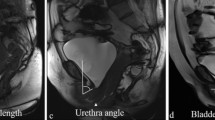
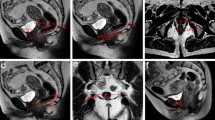
Dynamic MRI in the mid-sagittal plane, with T2-weighted images was performed for every patient. Patients were instructed not to urinate for at least 2 h prior to the examination, as we used the T2 hyperintense signal of urine as contrast to delineate the bladder from the surrounding structures. Valsalva maneuver was practiced with the patient before the procedure to ensure adequate straining during the examination. No urethral catheter was inserted. MRI was performed on a 1.5 T closed-configuration system using a body-array-surface coil. The resolution of the Tesla MRI provided excellent delineation of the urethra using the T2 hyperintensity of urine and hence better identification of the urethral axis. Rectal opacification by gel, which appeared T2 hyperintense, was done to delineate the rectal lumen for any concomitant pathologies such as rectal prolapse or rectocele.
The pubococcygeal line (PCL) was used as a reference line for the pelvic floor. PCL extends from the lower border of the symphysis pubis to the last coccygeal joint. The perpendicular distance from organs’ landmarks to the PCL was measured in millimeters (mm). Negative values were given to any measurement above the PCL, and positive values were given to any measurement below the PCL.
The following parameters were assessed in our study:
-
1.
Degree of bladder descent which is the vertical distance between PCL and the bladder base.
-
2.
Posterior urethrovesical angle (PUV): It is the angle between the urethral axis and the posterior border of the bladder base or the trigone [8], normally it is less than 115° [9].
-
3.
Angle of urethral inclination (UI): It is the angle of the urethral axis in relation to the vertical plane, normally it is less than 30° [10].
-
4.
Leading edge of the vaginal cuff (in cases of previous hysterectomy) or the location of the cervix (in cases of preserved uterus) in relation to the PCL.
-
5.
The anorectal junction in relation to the PCL.
ASC was performed in all patients. Use of vaginal retractor facilitated the dissection of both the anterior and posterior vaginal walls and also facilitated sutures placement and securing of the mesh to the vagina. Placing the retractor all the way into the vagina but not pushing the vagina upward establishes the proper length of the graft. Dissection of anterior vagina from the bladder continued caudally till reaching level of bladder neck, while dissection continued posteriorly along the rectovaginal space till reaching level of the perineal body. One to two permanent 1-prolene sutures were placed in the sacral promontory and anterior longitudinal ligament. Two polypropylene meshes were used, one was placed on the anterior vaginal wall and the other was placed on the posterior vaginal wall. If the uterus was preserved, the anterior vaginal mesh was Y shaped to be passed bilaterally through the broad ligaments. If the patient was posthysterectomy, a rectangular anterior vaginal mesh was used instead. The meshes were sutured to the exposed vaginal wall, respectively, using 2/0 non-absorbable monofilament sutures. A space of two fingerbreadths was left between the mesh and the rectum to prevent compression of the mesh over the rectum.
The patients were followed 6 months postoperatively to see the effect of sacrocolpopexy on the urethral anatomy and continence mechanism using Dynamic MRI. This was correlated with the clinical presentation and the urodynamic findings. Data were fed to the computer and analyzed using IBM SPSS software package version 20.0 (Armonk, NY: IBM Corp). The used tests were paired t test, Wilcoxon signed-rank test, Mann–Whitney test and McNemar test. Significance of the obtained results was judged at the 5% level.
Results
Twenty female patients presenting with POP and SUI were admitted to our department from April 2017 to May 2018. Mean age was 53.5 ± 8.9 years, with 17 postmenopausal patients (85%) and 3 premenopausal patients (15%). Gravidity ranged from 2 to 12, while the parity ranged from 2 to 9. Mean body mass index (BMI) was 29.37 ± 3.3. None of these patients underwent cesarean section for their deliveries, while six patients underwent abdominal hysterectomy. All the patients had apical prolapse, four had associated cystocele, while five had associated rectocele. All the patients had preoperative SUI with urethral hypermobility proved by visual inspection during Valsalva and preoperative VLPP > 60 cmH2O. Seven patients had mixed urinary incontinence (MUI) with predominant SUI.
After performing ASC, all the patients reported significant improvement of all prolapse and incontinence symptoms postoperatively. This was reflected on the change in the scores of the questionnaires postoperatively. UDI-6 score decreased from 48.6 ± 8.5 preoperatively to 7.4 ± 10.3 postoperatively (p < 0.001), ICIQ-UI score decreased from 16.6 ± 1.8 preoperatively to 1.7 ± 2.7 postoperatively (p < 0.001), while PISQ-12 score decreased from 17.3 ± 2 preoperatively to 13 ± 1.8 postoperatively (p < 0.001). PGI-I questionnaires showed significant improvement for both POP and SUI (90% and 85% improvement, respectively).
Mean VLPP preoperatively was 86 ± 2 cmH2O. Postoperative urodynamic evaluation showed improvement after ASC. Concerning VLPP, 11 patients had no VLPP postoperatively, while 9 patients had VLPP above 90 cmH2O with a mean of 101 ± 8.49 cmH2O (improved). Patients were satisfied with their urinary condition after surgery and only four of them underwent transobturator tape (TOT) insertion 6 months after ASC, as they were bothered by their SUI. The remaining five patients were satisfied with their continence status.
The results of the pre- and postoperative dynamic MRI are shown in Table 1. For incontinence parameters, bladder neck descent changed from 2.34 ± 0.71 cm preoperatively to − 2.23 ± 0.28 cm postoperatively (p < 0.001), PUV angle decreased from 173.15 ± 6.31° preoperatively to 127.7 ± 11.3° postoperatively (p < 0.001), while UI angle decreased from 69.8 ± 7.65° preoperatively to 29.15 ± 9.3° postoperatively (p < 0.001) (Figs. 1, 2 and 3). As for the prolapse, the distance between the anorectal junction and PCL changed from 2.7 ± 0.4 cm preoperatively to − 1 ± 0.9 cm postoperatively (p < 0.001), while the leading edge of the cervix or vaginal cuff in relation to the PCL changed significantly from 5.2 ± 0.72 cm to − 4.1 ± 0.68 cm postoperatively (p < 0.001) (Fig. 4).
Discussion
About 40–50% of women with POP have concomitant SUI (2). The relationship between POP and urinary incontinence (UI) is complex, as both UI and POP probably share some of the same etiological mechanisms [11, 12]. In 2008, Costantini et al. [13] evaluated the impact of Burch colposuspension as an anti-incontinence procedure in 47 patients with UI undergoing ASC for POP repair. After 1 year follow-up, they concluded that Burch colposuspension does not provide any additional benefit in POP repair in patients with UI. After 5 years [14], they re-evaluated the follow-up of these patients. The update of long-term follow-up confirmed the same conclusion.
On the contrary, in the CARE trial [15], 322 women with SUI and POP were enrolled and randomized, 157 underwent Burch procedure with ASC and 165 patients underwent ASC alone. They concluded that prophylactic Burch colposuspension significantly reduced postoperative symptoms of stress incontinence without increasing other lower urinary tract symptoms.
A large multicenter trial (OPUS trial) [16] included 327 women without symptoms of SUI but presenting with anterior vaginal prolapse, who were planning to undergo vaginal POP surgery. Women were randomly assigned to receive either a midurethral sling or sham incisions during the vaginal repair of the cystocele. They concluded that a prophylactic midurethral sling inserted during vaginal prolapse surgery resulted in a lower rate of urinary incontinence at 12 months, but higher rates of adverse events. However, this applies to vaginal repair of POP and not abdominal repair of prolapse.
In the CUPIDO- II trial, Van der Ploeg et al. [17] compared transvaginal prolapse repair combined with midurethral sling (group I) versus prolapse repair only (group II) in patients with concomitant prolapse and occult SUI. The authors found that, only 13% of women in group II needed additional MUS. Similarly, Borstad et al. [18] found that 25% of incontinent women were cured of SUI after prolapse repair only without anti-incontinence surgery. In another study done by Lensen et al. [19], they focused on the changes in UI rates after POP surgery without concomitant anti-incontinence surgery. At 1 year, 42% were cured from UUI and 39% were cured from SUI by POP surgery alone. Therefore, they considered it justified not to perform concomitant anti-incontinence surgery and await effects of POP surgery alone. Our study supports this consideration.
In our study, the majority of our patients were satisfied with their surgeries and symptom control. This satisfaction was manifest in their improvement in lower urinary tract symptoms, prolapse symptoms, and effect on symptom-specific and overall quality of life after the prolapse surgery. Eleven patients were dry after ASC, 5 patients had mild SUI but not bothering them, while the remaining 4 patients had bothering SUI after surgery and asked to be scheduled for TOT after 6 months. We also noticed significant improvement of sexual function after ASC, where 65% of the women became sexually active in comparison to 35% preoperatively. This was clearly demonstrated in results of PISQ-12. Similar results regarding improvement of sexual function after ASC were observed by Handa et al. [20].
The use of MRI to evaluate the efficacy of incontinence and prolapse surgery is not common in the clinical practice. However, few studies in the literature mentioned its use in this field [21, 22]. For incontinence, they measured the change in the bladder base level, PUV angle, and the UI angle after anti-incontinence surgery. However, for prolapse, they focused only on the change of the C point and bladder base level. In our study, we measured all the incontinence parameters and added two important parameters for prolapse (leading edge of vaginal cuff or cervix and the anorectal junction in relation to PCL) before and after ASC.
To the best of our knowledge, our study is the first prospective study to evaluate urethral anatomy and continence mechanism objectively by dynamic MRI in cases treated by ASC alone without concomitant anti-SUI procedure. The bladder base was significantly elevated above PCL after surgery in all patients, while the PUV angle and the angle of UI were both significantly reduced after surgical correction. The distance between the leading edge of the prolapse and PCL was also significantly changed after surgery (p < 0.01). Conventional thinking is that a cystocele protects against stress incontinence by causing angulation or kinking of the urethra and that prolapse repair often aggravates the degree of SUI. However, in our study, we showed with dynamic MRI images that, at least in certain cases, this thinking does not hold true. Perhaps, the cystocele has to be significant enough to cause angulation and in younger patients with no cystocele or minimal cystocele, patients may have improvement of their SUI symptoms.
The strengths of our study include the prospective nature with no patients lost to follow-up, use of the same questionnaires pre- and postoperatively, giving more exact information about changes in UI. In addition, the supervisor who was responsible for the follow-up assessment was blinded to the patients’ baseline data to eliminate any possible bias in the study. It is considered the first study in the literature to evaluate the effect of ASC on the urethral anatomy and continence mechanism using dynamic MRI. The major limitations of this study are the small sample size and short follow-up period, and the quantity of urine loss before and after surgery was not available.
Conclusions
In patients with prolapse and urethral hypermobility, ASC may return the bladder neck and urethral anatomy towards normal as proved by dynamic MRI. However, further studies on a larger number of patients with a longer follow-up period are required.
References
Brown JS, Waetjen LE, Subak LL, Thom DH, Van Den Eeden S, Vittinghoff E (2002) Pelvic organ prolapse surgery in the United States. Am J Obstet Gynecol 186:712–716
Olsen AL, Smith VJ, Bergstrom JO, Colling JC, Clark AL (1997) Epidemiology of surgically managed POP and urinary incontinence. Obstet Gynecol 89:501–506
Elliott DS, Frank I, DiMarco DS, Chow GK (2004) Gynecologic use of robotically assisted laparoscopic sacrocolpopexy for the treatment of high-grade vaginal vault prolapse. Am J Surg 188(4A Suppl):52S–56S
Maher C, Feiner B, Baessler K, Schmid C (2013) Surgical management of pelvic organ prolapse in women. Cochrane Database Syst Rev 30(4):CD004014. https://doi.org/10.1002/14651858.CD004014
Van Der Ploeg JM, Van Der Steen A, Rengerink KO, Van Der Vaart CH, Roovers JP (2014) Prolapse surgery with or without stress incontinence surgery for pelvic organ prolapse: a systematic review and meta-analysis of randomised trials. BJOG 121:537–547
Jha S, Cutner A, Moran P (2018) The UK National Prolapse Survey: 10 years on. Int Urogynecol J 22:517–528
Raman SV, Raker CA, Sung VW (2014) Concomitant apical prolapse repair and incontinence procedures: trends from 2001–2009 in the United States. Am J Obstet Gynecol 211:e1–e5
Jeffcoate TNA, Roberts H (1952) Stress incontinence of urine: opening papers. BJOG 59(5):685–697
Drutz HP, Shapiro BJ, Mandel F (1978) Do static cystourethrograms have a role in the investigation of female incontinence? Am J Obstet Gynecol 130(5):516–520
Green TH (1962) Development of a plan for the diagnosis and treatment of urinary stress incontinence. Am J Obstet Gynecol 83:632–648
Dietz HP, Hansell NK, Grace ME, Eldridge AM, Clarke B, Martin NG (2005) Bladder neck mobility is a heritable trait. BJOG 112:334–339
Chen B, Wen Y, Polan ML (2004) Elastolytic activity in women with stress urinary incontinence and pelvic organ prolapse. Neurourol Urodyn 23:119–126
Costantini E, Lazzeri M, Bini V, Del Zingaro M, Zucchi A, Porena M (2008) Burch colposuspension does not provide any additional benefit to pelvic organ prolapse repair in patients with urinary incontinence: a randomized surgical trial. J Urol 180:1007
Costantini E, Lazzeri M, Bini V, Del Zingaro M, Zucchi A, Porena M (2001) POP repair with and without prophylactic concomitant Burch colposuspension in continent women: a randomized, controlled trial with 8-year followup. J Urol 185(6):2236–2240
Brubaker L, Cundiff GW, Fine P, Nygaard I, Richter HE, Visco AG et al (2006) Abdominal sacrocolpopexy with Burch colposuspension to reduce urinary stress incontinence. N Engl J Med 354:1557–1566
Wei JT, Nygaard I, Richter HE, Nager CW (2012) A midurethral sling to reduce incontinence after vaginal prolapse repair. Obstet Gynecol Survey 366:2358–2367
van der Ploeg JM, Rengerink KO, van der Steen A, van Leeuwen JHS, van der Vaart CH, Roovers JPWR (2016) Vaginal prolapse repair with or without a midurethral sling in women with genital prolapse and occult stress urinary incontinence: a randomized trial. Int Urogynecol J 27(7):1029–1038
Borstad E, Abdelnoor M, Staff AC, Kulseng-Hanssen S (2010) Surgical strategies for women with pelvic organ prolapse and urinary stress incontinence. Int Urogynecol J 21(2):179–186
Lensen EJM, Withagen MIJ, Kluivers KB, Milani AL, Vierhout ME (2013) Urinary incontinence after surgery for pelvic organ prolapse. Neurourol Urodyn 32(5):455–459
Handa VL, Zyczynski HM, Brubaker L, Nygaard I, Janz NK, Richter HE et al (2007) Sexual function before and after sacrocolpopexy for pelvic organ prolapse. Am J Obstet Gynecol 197(6):e1–6
Perk H, Oral B, Yeşildağ A, Serel T, Özsoy M, Turgut T (2002) Magnetic resonance imaging for stress incontinence: evaluation of patients before and after surgical correction. Eur J Radiol 44(1):44–47
Ginath S, Garely AD, Luchs JS, Shahryarinejad A, Olivera CK, Zhou S et al (2012) Magnetic resonance imaging of abdominal versus vaginal prolapse surgery with mesh. Int Urogynecol J 23:1569–1576
Author information
Authors and Affiliations
Contributions
WM: project development, performing procedure, manuscript writing. ME: performing procedure, manuscript writing. S-AG: data collection, patient evaluation. AK: manuscript writing, project development. HH: performing procedure, project development.
Corresponding author
Ethics declarations
Conflict of interest
Wally Mahfouz, MD declares that he has no conflict of interest. Mostafa Elmissiry, MD declares that he has no conflict of interest. Saif-Alislam Gomaa, MD declares that he has no conflict of interest. Ashraf Koraitim, MD declares that he has no conflict of interest. Hebatallah H.M. Hassan, MD declares that she has no conflict of interest.
Ethical approval
All procedures performed in the study were in accordance with the ethical standards of the institutional research committee of Alexandria University, Egypt, and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mahfouz, W., Elmissiry, M., Gomaa, SA. et al. Evaluation of the effect of abdominal sacrocolpopexy (ASC) on urethral anatomy and continence mechanism using dynamic MRI. Int Urol Nephrol 52, 1429–1435 (2020). https://doi.org/10.1007/s11255-020-02444-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11255-020-02444-8