Abstract
Purpose
To investigate if multiphasic multidetector computed tomography (MDCT) enhancement profiles can distinguish clear cell renal cell carcinomas (ccRCCs) with high carbonic anhydrase-IX (CA-IX) expression from ccRCCs with low CA-IX expression.
Methods
With IRB approval for this retrospective study, we derived a cohort of 105 histologically proven ccRCCs with preoperative 4-phase renal mass MDCT from 2001 to 2013. Following manual segmentation, the computer-assisted detection algorithm selected a 0.5-cm-diameter region of maximal attenuation within each lesion in each phase. CA-IX expression level was determined by immunohistochemical staining of tumor specimens. In the high and low CA-IX expression subgroups, the magnitude of enhancement and washout were compared using t tests; the performance of contrast washout in differentiating between subgroups was assessed with logistic regression analysis.
Results
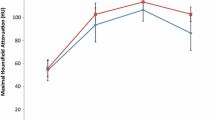
ccRCCs with high and low CA-IX expression both exhibited peak enhancement in the corticomedullary phase. ccRCCs with high CA-IX expression demonstrated significantly greater relative nephrographic washout than those with low CA-IX expression (18.4% vs. 7.8%, p = 0.03). ccRCCs with high CA-IX expression had greater relative excretory washout than ccRCCs with low CA-IX expression with a trend toward significance (33.4% vs. 25.2%, p = 0.05). After controlling for tumor size and stage, for distinguishing ccRCCs with high and low CA-IX expression, relative excretory washout had a sensitivity, negative predictive value, accuracy, and positive predictive value of 99% (65/66), 88% (7/8), 69% (72/105), and 67% (65/97), respectively.
Conclusion
Relative nephrographic and excretory washout may have the potential to help distinguish ccRCCs with high and low CA-IX expression, but this requires further validation.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Renal cell carcinoma (RCC) accounts for nearly 2% of all adult cancers worldwide with an estimated 210,000 new cases diagnosed each year [1]. Within the Vancouver classification, the most common histologic subtypes of renal cell carcinoma are clear cell, papillary, and chromophobe, accounting for 70–80%, 14–17%, and 4–8% of RCCs, respectively [2, 3]. Of these subtypes, clear cell RCC has the worst prognosis with a 5-year survival rate of 44–69%, accounting for 94% of metastatic RCC [4,5,6,7,8]. In patients with RCC, postoperatively determined pathological stage and grade have been used to predict patient prognosis, but these methods are invasive and can be unreliable due to heterogeneity within patient groups. Alternative biomarker analysis of a variety of molecular characteristics within lesions is being widely studied to address the shortcomings in the traditional approach. Identifying imaging biomarkers to predict specific gene and protein expression can be of great clinical value. As molecular analyses become an increasingly important part of patient care, imaging will need to provide more information beyond the size and location of a particular tumor. In this new paradigm, utilizing imaging features to predict the expression of molecular markers can help guide clinicians regarding which therapies to consider.
Carbonic anhydrase-IX (CA-IX), a molecular biomarker that has been investigated for its prognostic significance in clear cell RCC, is a transmembrane protein that regulates intra- and extra-cellular pH [9]. Prior studies have shown that low CA-IX expression, defined as a staining percentage less than or equal to 85%, is an independent prognostic marker of poorer survival outcomes in comparison to clear cell RCCs with high CA-IX expression [9,10,11]. In addition, patients with high CA-IX expression, defined as a staining percentage greater than 85%, are more likely to respond to interleukin-2 (IL-2) immunotherapy [9, 11].
Several recent studies have shown that the magnitude of enhancement on multiphasic multidetector computed tomography (MDCT) can help distinguish between clear cell RCC from other common RCC subtypes (papillary and chromophobe) and common benign mimics (oncocytomas and lipid-poor angiomyolipomas) [12,13,14,15,16,17,18,19]. Clear cell RCCs typically exhibit peak enhancement in the corticomedullary phase, which is significantly greater than that of other solid renal cortical tumors [12,13,14,15,16,17,18,19]. However, there are currently no published imaging biomarkers to predict the expression of CA-IX in clear cell RCCs. The purpose of this study was to investigate whether multiphasic MDCT enhancement can help differentiate between clear cell RCCs with high and low CA-IX expression.
Materials and methods
Patients
With Institutional Review Board approval for this Health Insurance Portability and Accountability Act (HIPAA)-compliant retrospective study and a waiver of informed consent, we queried our institution’s pathology database to derive all histologically proven cases of clear cell RCC from December 2001 to August 2013. Approximately 1900 histologically proven cases of clear cell RCC were obtained in this query. Approximately 1770 lesions were excluded due to lack of a preoperative dedicated four-phase renal mass protocol MDCT. An additional 29 cases were excluded due to the lack of availability of tissue specimens for immunohistochemistry, resulting in a study cohort of 105 clear cell RCCs, representing 100 unique patients. Five patients had two lesions each. The mean time from MDCT imaging to nephrectomy or biopsy in the study cohort was 65 days. The mean age at the time of nephrectomy or biopsy was 62 years (Table 1). Histopathology was obtained from partial nephrectomy [46 specimens (44%)], radical nephrectomy [32 specimens (31%)], and percutaneous 20 gauge core biopsy [27 specimens (26%)]. For lesions diagnosed at partial or radical nephrectomy, the T stage of each lesion was obtained by reviewing pathology reports for the resected specimens. For lesions diagnosed at percutaneous core biopsy, the T stage of each lesion was approximated based upon preoperative CT or MRI.
MDCT examination
All CT examinations were performed at our institution using multidetector row CT [mainly 64 (2005–present) or 16 detector (2003–2008) row helical scanners (Sensation 64 or Sensation 16, Siemens Medical Solutions, Erlangen, Germany); or 4-detector (2000–2003) scanners (Lightspeed Qx/i, GE Medical Systems, Milwaukee, WI)]. Images were obtained with the patient breath holding with the following parameters: 120 kVP, variable mA, section thickness interval of 3 mm. For each patient, a 4-phase renal mass protocol MDCT was performed. Patients first underwent an unenhanced scan of the abdomen from the top of the diaphragm to the iliac crest. Then, following the injection of 100–150 mL of nonionic intravenous iohexol based upon patient weight (Omnipaque 350, GE Healthcare, Milwaukee, WI) with a power injector at 3–4 mL per second, a bolus tracking algorithm (CareBolus, Siemens Medical Solutions; SmartPrep, GE Medical Systems) was employed. For bolus tracking, a region of interest was placed in the thoracoabdominal aorta junction with a trigger set to begin at 150 HU. Corticomedullary, nephrographic, and excretory phase imaging was performed 40 s, 90 s, and 8 min after the threshold of 150 HU was reached. Images were sent to our picture archiving and communications system (Centricity, GE Medical Systems, Milwaukee, WI) for interpretation.
CT image analyses
Each lesion was segmented in its entirety on axial images in each phase using proprietary non-commercial Food and Drug Administration (FDA) 510(k) cleared software to create a 3D lesion contour. Lesion contours were drawn slightly internal to the lesion’s outer margin to avoid partial volume averaging artifact [14, 20,21,22,23]. Each lesion contour was reviewed by two abdominal fellowship-trained genitourinary radiologists with 10 (MD) and 19 years (SSR) of experience, who were blinded to clinical, histopathologic, and immunohistochemical findings. If there was any discordance between the two readers regarding the appropriate placement of a contour, the case was discussed by both readers until a consensus was reached. Utilizing each whole lesion 3D contour, the computer-assisted detection (CAD) algorithm then selected a 0.5-cm-diameter circular region of maximal attenuation within each lesion in each of the four phases, excluding pixels with attenuation greater than 300 Hounsfield units (HU) to exclude calcifications. Each CAD-selected region of maximal attenuation was reviewed by each reader for appropriate placement. A similar small region of interest approach to quantifying lesion attenuation has been used in prior studies examining multiphasic MDCT enhancement as a means of differentiating clear cell RCC from other common subtypes and benign mimics [14], identifying prognostically significant cytogenetic abnormalities in clear cell RCC (the gain of chromosome 12, the gain of chromosome 20, and the loss of chromosome 8p) [22, 24, 25], differentiating type 1 from type 2 papillary RCC [21], and examining multiphasic MR enhancement as a means of differentiating clear cell RCC from papillary RCC, chromophobe RCC, and oncocytoma [23]. Relative nephrographic washout was calculated as 100* (corticomedullary attenuation − nephrographic attenuation)/(corticomedullary attenuation). Absolute nephrographic washout was calculated as 100*(corticomedullary attenuation − nephrographic attenuation)/(corticomedullary attenuation − unenhanced attenuation). Relative excretory washout was calculated as 100* (corticomedullary attenuation − excretory attenuation)/(corticomedullary attenuation). Absolute excretory washout was calculated as 100*(corticomedullary attenuation − excretory attenuation)/(corticomedullary attenuation − unenhanced attenuation). Lesion size was assessed by identifying the maximal lesion diameter on axial images.
Immunohistochemistry for carbonic anhydrase-IX (CA-IX)
Surgical pathology specimens obtained from nephrectomy or percutaneous core biopsy were stained with a rabbit polyclonal anti-CA-IX antibody NB100-417 (Novus Biologicals, Littleton, CO). Staining of tumor cells was assessed by a fellowship-trained genitourinary pathologist, and the percentage of positively staining tumor cells was calculated. High CA-IX expression was defined as a staining percentage greater than 85%; low CA-IX expression was defined as a staining percentage less than or equal to 85% [10, 11].
Statistical analyses
To compare the relative nephrographic, absolute nephrographic, relative excretory, and absolute excretory washout values between clear cell RCCs with high and low CA-IX expression, t tests were performed. Differences with p values less than 0.05 were considered to be statistically significant. Logistic regression classification models were used to evaluate the performance of contrast washout values in predicting the expression of CA-IX in clear cell RCCs, after controlling for lesion size and stage. To account for potential clustering effects, the data were also analyzed while including only one lesion per patient in each group. For patients with multiple lesions, the lesion included was selected at random. Analyses were performed using SPSS 23 for Windows (IBM Corp, Armonk, NY).
Results
Lesions
Of the 105 clear cell RCCs in our cohort, immunohistochemical staining revealed high and low CA-IX expression in 66 (63%) and 39 (37%), respectively. Baseline characteristics for clear cell RCCs in each group are presented in Table 1. There was no significant difference in mean lesion size between clear cell RCCs with high CA-IX expression and those with low CA-IX expression (4.4 cm vs. 3.6 cm, p = 0.12). The majority of the clear cell RCCs in both subcohorts were either stage T1a or T1b at the time of diagnosis.
Contrast washout parameters
Clear cell RCCs with both high and low CA-IX expression exhibited peak mean attenuation in the corticomedullary phase (Fig. 1). However, clear cell RCCs with high CA-IX expression had significantly greater relative nephrographic washout (18.4% vs. 7.8%, p = 0.03), absolute nephrographic washout (24.6% vs. 6.5%, p = 0.04), and absolute excretory washout (52.0% vs. 33.6%, p = 0.04) than clear cell RCCs with low CA-IX expression (Fig. 2, Table 2). The relative excretory washout of clear cell RCCs with high CA-IX expression was greater than that of clear cell RCCs with low CA-IX expression with a trend toward significance (33.4% vs. 25.2%, p = 0.05). To account for potential clustering effects, the data were also analyzed while including only one lesion per patient in each group. The results were similar (Table 3). Typical lesions in the high CA-IX and low CA-IX subcohorts are presented in Figs. 3 and 4.
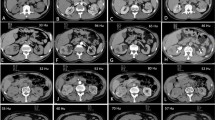
Corticomedullary, nephrographic, and excretory phase axial MDCT images of a clear cell RCC with high CA-IX expression in a 74-year-old man. Representative CAD-selected regions of maximal attenuation are shown in blue. A Maximal corticomedullary phase attenuation was 245 HU. B Maximal nephrographic phase attenuation was 153 HU. C Maximal excretory phase attenuation was 115 HU. Relative nephrographic washout was 38%. Relative excretory washout was 53%
Corticomedullary, nephrographic, and excretory phase axial MDCT images of a clear cell RCC with low CA-IX expression in a 70-year-old woman. Representative CAD-selected regions of maximal attenuation are shown in blue. A Maximal corticomedullary phase attenuation was 248 HU. B Maximal nephrographic phase attenuation was 245 HU. C Maximal excretory phase attenuation was 173 HU. Relative nephrographic washout was 1%. Relative excretory washout was 30%
Logistic regression classification models
Logistic regression classification models were developed to evaluate the performance of relative nephrographic and excretory washout in distinguishing clear cell RCCs with high CA-IX expression from clear cell RCCs with low CA-IX expression, after controlling for lesion size and lesion stage. As shown in Table 4, after controlling for these variables, relative nephrographic washout had an odds ratio of 1.02 (p = 0.077), and the model had an accuracy of 68% (71/105), sensitivity of 99% (65/66), specificity of 15% (6/39), negative predictive value of 86% (6/7), and positive predictive value of 66% (65/98) in differentiating clear cell RCCs with high CA-IX expression from clear cell RCCs with low CA-IX expression. After controlling for lesion size and stage, relative excretory washout had an odds ratio of 1.02 (p = 0.088), and the model had an accuracy of 69% (72/105), sensitivity of 99% (65/66), specificity of 18% (7/39), negative predictive value of 88% (7/8), and positive predictive value of 67% (65/97) in differentiating clear cell RCCs with high and low CA-IX expression.
Discussion
Consistent with prior published studies regarding the MDCT enhancement of benign and malignant renal tumors, clear cell RCCs in our cohort typically exhibited peak enhancement in the corticomedullary phase. In this study, we demonstrated that the degree of relative washout from the corticomedullary phase to the nephrographic and excretory phases may be helpful for differentiating between clear cell RCCs with high and low CA-IX expression. Clear cell RCCs with high CA-IX expression demonstrated a significantly greater degree of relative nephrographic washout than clear cell RCCs with low CA-IX expression. The relative excretory washout of clear cell RCCs with high CA-IX expression was greater than that of clear cell RCCs with low CA-IX expression with a trend toward significance. In a logistic regression classification model that controlled for lesion size and lesion stage, relative excretory washout had an accuracy of 69% (72/105), sensitivity of 99% (65/66), negative predictive value of 88% (7/8), and positive predictive value of 67% (65/97) in differentiating clear cell RCCs with high CA-IX expression from clear cell RCCs with low CA-IX expression.
CA-IX is a prognostically significant molecular marker in clear cell RCC. Low CA-IX expression is an independent prognostic marker of poorer survival outcomes in clear cell RCC [9,10,11]. Furthermore, patients with high CA-IX expression are more likely to respond to IL-2 immunotherapy [9, 11]. Because of the prognostic implications of CA-IX expression, a simple non-invasive means of identifying the level of CA-IX expression may be helpful clinically to assess patient prognosis and potentially guide further management, if validated in a large prospective trial.
Our study has several potential limitations. First, although all lesions in our cohort were evaluated with a dedicated four-phase renal mass protocol MDCT, due to the retrospective nature of this study, we could not account for variability of absolute attenuation within the same phase related to scanner differences. However, our institution’s four-phase renal mass MDCT protocol was designed to standardize these parameters as much as possible. Second, this was a preliminary discovery-phase study evaluating the relationship between CA-IX expression and contrast washout on multiphasic MDCT. Our findings should be validated in an independent testing cohort. We do not have a definite mechanistic explanation for the relationship between CA-IX expression and contrast washout, although we suspect that it is likely related to differences in angiogenic pathways. CA-IX is a transmembrane protein that regulates intracellular and extracellular pH [9]. By influencing the acidic microenvironment, CA-IX regulates the activation of angiogenic factors [26]. In addition, CA-IX is a downstream gene target of hypoxia-inducible factor-1α (HIF-1α) in the hypoxia-inducible factor pathway, which plays a significant role in modulating angiogenesis. Third, in a logistic regression classification model that controlled for lesion size and lesion stage, relative excretory washout had an accuracy of 69% (72/105) in differentiating clear cell RCCs with high CA-IX expression from clear cell RCCs with low CA-IX expression, which is not yet adequate to definitively guide further treatment. However, our findings suggest that there may be a consistent relationship between CA-IX expression in clear cell RCCs and contrast washout on multiphasic MDCT, if validated in a large prospective trial.
To our knowledge, our study is the first proof of concept study to demonstrate an MDCT-based difference between clear cell RCCs with high and low CA-IX expression. Because of the prognostic significance of CA-IX expression, a non-invasive means of identifying the level of CA-IX expression preoperatively may help guide clinical decision making and patient counseling. Additionally, with a baseline multiphasic MDCT scan prior to surgery or ablation, new lesions may be non-invasively evaluated to determine if their enhancement profiles are similar to previously treated lesions. Future work is needed to determine whether our findings can be replicated in a prospective cohort and whether similar findings can be replicated with gadolinium-enhanced magnetic resonance imaging and contrast-enhanced ultrasound imaging.
References
Gore ME, Larkin JM (2011) Challenges and opportunities for converting renal cell carcinoma into a chronic disease with targeted therapies. Br J Cancer 104(3):399–406. https://doi.org/10.1038/sj.bjc.6606084
Kovacs G, Akhtar M, Beckwith BJ, et al. (1997) The Heidelberg classification of renal cell tumours. J Pathol 183(2):131–133. 10.1002/(sici)1096-9896(199710)183:2<131::aid-path931>3.0.co;2-g
Truong LD, Shen SS (2011) Immunohistochemical diagnosis of renal neoplasms. Arch Pathol Lab Med 135(1):92–109. https://doi.org/10.1043/2010-0478-rar.1
Cheville JC, Lohse CM, Zincke H, Weaver AL, Blute ML (2003) Comparisons of outcome and prognostic features among histologic subtypes of renal cell carcinoma. Am J Surg Pathol 27(5):612–624
Hoffmann NE, Gillett MD, Cheville JC, et al. (2008) Differences in organ system of distant metastasis by renal cell carcinoma subtype. J Urol 179(2):474–477. https://doi.org/10.1016/j.juro.2007.09.036
Amin MB, Corless CL, Renshaw AA, et al. (1997) Papillary (chromophil) renal cell carcinoma: histomorphologic characteristics and evaluation of conventional pathologic prognostic parameters in 62 cases. Am J Surg Pathol 21(6):621–635
Beck SD, Patel MI, Snyder ME, et al. (2004) Effect of papillary and chromophobe cell type on disease-free survival after nephrectomy for renal cell carcinoma. Ann Surg Oncol 11(1):71–77
Moch H, Gasser T, Amin MB, et al. (2000) Prognostic utility of the recently recommended histologic classification and revised TNM staging system of renal cell carcinoma: a Swiss experience with 588 tumors. Cancer 89(3):604–614
Stillebroer AB, Mulders PF, Boerman OC, Oyen WJ, Oosterwijk E (2010) Carbonic anhydrase IX in renal cell carcinoma: implications for prognosis, diagnosis, and therapy. Eur Urol 58(1):75–83. https://doi.org/10.1016/j.eururo.2010.03.015
Bui MH, Seligson D, Han KR, et al. (2003) Carbonic anhydrase IX is an independent predictor of survival in advanced renal clear cell carcinoma: implications for prognosis and therapy. Clin Cancer Res 9(2):802–811
Lam JS, Pantuck AJ, Belldegrun AS, Figlin RA (2007) Protein expression profiles in renal cell carcinoma: staging, prognosis, and patient selection for clinical trials. Clin Cancer Res 13(2 Pt 2):703s–708s. https://doi.org/10.1158/1078-0432.ccr-06-1864
Young JR, Margolis D, Sauk S, et al. (2013) Clear cell renal cell carcinoma: discrimination from other renal cell carcinoma subtypes and oncocytoma at multiphasic multidetector CT. Radiology 267(2):444–453. https://doi.org/10.1148/radiol.13112617
Lee-Felker SA, Felker ER, Tan N, et al. (2014) Qualitative and quantitative MDCT features for differentiating clear cell renal cell carcinoma from other solid renal cortical masses. AJR Am J Roentgenol 203(5):W516–524. https://doi.org/10.2214/ajr.14.12460
Coy H, Young JR, Douek ML, et al. (2017) Quantitative computer-aided diagnostic algorithm for automated detection of peak lesion attenuation in differentiating clear cell from papillary and chromophobe renal cell carcinoma, oncocytoma, and fat-poor angiomyolipoma on multiphasic multidetector computed tomography. Abdom Radiol (NY) 42(7):1919–1928. https://doi.org/10.1007/s00261-017-1095-6
Zhang J, Lefkowitz RA, Ishill NM, et al. (2007) Solid renal cortical tumors: differentiation with CT. Radiology 244(2):494–504. https://doi.org/10.1148/radiol.2442060927
Kim JK, Kim TK, Ahn HJ, et al. (2002) Differentiation of subtypes of renal cell carcinoma on helical CT scans. AJR Am J Roentgenol 178(6):1499–1506. https://doi.org/10.2214/ajr.178.6.1781499
Sheir KZ, El-Azab M, Mosbah A, El-Baz M, Shaaban AA (2005) Differentiation of renal cell carcinoma subtypes by multislice computerized tomography. J Urol 174 (2):451–455; discussion 455. https://doi.org/10.1097/01.ju.0000165341.08396.a9
Jinzaki M, Tanimoto A, Mukai M, et al. (2000) Double-phase helical CT of small renal parenchymal neoplasms: correlation with pathologic findings and tumor angiogenesis. J Comput Assist Tomogr 24(6):835–842
Ruppert-Kohlmayr AJ, Uggowitzer M, Meissnitzer T, Ruppert G (2004) Differentiation of renal clear cell carcinoma and renal papillary carcinoma using quantitative CT enhancement parameters. AJR Am J Roentgenol 183(5):1387–1391. https://doi.org/10.2214/ajr.183.5.1831387
Rosenkrantz AB, Matza BW, Portnoy E, et al. (2014) Impact of size of region-of-interest on differentiation of renal cell carcinoma and renal cysts on multi-phase CT: preliminary findings. Eur J Radiol 83(2):239–244. https://doi.org/10.1016/j.ejrad.2013.10.020
Young JR, Coy H, Douek M, et al. (2017) Type 1 papillary renal cell carcinoma: differentiation from Type 2 papillary RCC on multiphasic MDCT. Abdom Radiol (NY) 42(7):1911–1918. https://doi.org/10.1007/s00261-017-1091-x
Young JR, Coy H, Douek M, et al. (2017) Clear cell renal cell carcinoma: identifying the gain of chromosome 12 on multiphasic MDCT. Abdom Radiol (NY) 42(1):236–241. https://doi.org/10.1007/s00261-016-0868-7
Young JR, Coy H, Kim HJ, et al. (2017) Performance of relative enhancement on multiphasic MRI for the differentiation of clear cell renal cell carcinoma (RCC) from papillary and chromophobe RCC subtypes and oncocytoma. AJR Am J Roentgenol 208(4):812–819. https://doi.org/10.2214/ajr.16.17152
Young JR, Margolis D, Sauk S, et al. (2014) Clear cell renal cell carcinoma: multiphasic MDCT enhancement can predict the loss of chromosome 8p. Abdom Imaging 39(3):543–549. https://doi.org/10.1007/s00261-014-0092-2
Young JR, Young JA, Margolis DJ, et al. (2016) Clear cell renal cell carcinoma: identifying the gain of chromosome 20 on multiphasic MDCT. Abdom Radiol (NY) 41(11):2175–2181. https://doi.org/10.1007/s00261-016-0813-9
Sedlakova O, Svastova E, Takacova M, et al. (2014) Carbonic anhydrase IX, a hypoxia-induced catalytic component of the pH regulating machinery in tumors. Front Physiol 4:400. https://doi.org/10.3389/fphys.2013.00400
Acknowledgments
This study was funded by the Society of Abdominal Radiology Howard S. Stern Research Grant (Grant No. 20163335). We thank the UCLA Computer Vision and Imaging Biomarker (CVIB) group, including Dr. Jonathan Goldin, Dr. Matthew Brown, Moe Moe Ko, and War War Ko, for their assistance with this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
The Institutional Review Board waived the requirement of informed consent for this study.
Rights and permissions
About this article
Cite this article
Young, J.R., Coy, H., Kim, H.J. et al. Utility of multiphasic multidetector computed tomography in discriminating between clear cell renal cell carcinomas with high and low carbonic anhydrase-IX expression. Abdom Radiol 43, 2734–2742 (2018). https://doi.org/10.1007/s00261-018-1546-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00261-018-1546-8