Abstract
Normalizing cardiovascular measurements for body size allows for comparison among children of different ages and for distinguishing pathologic changes from normal physiologic growth. Because of growing interest to use height for normalization, the aim of this study was to develop height-based normalization models and compare them to body surface area (BSA)-based normalization for aortic and left ventricular (LV) measurements. The study population consisted of healthy, non-obese children between 2 and 18 years of age enrolled in the Pediatric Heart Network Echo Z-Score Project. The echocardiographic study parameters included proximal aortic diameters at 3 locations, LV end-diastolic volume, and LV mass. Using the statistical methodology described in the original project, Z-scores based on height and BSA were determined for the study parameters and tested for any clinically significant relationships with age, sex, race, ethnicity, and body mass index (BMI). Normalization models based on height versus BSA were compared among underweight, normal weight, and overweight (but not obese) children in the study population. Z-scores based on height and BSA were calculated for the 5 study parameters and revealed no clinically significant relationships with age, sex, race, and ethnicity. Normalization based on height resulted in lower Z-scores in the underweight group compared to the overweight group, whereas normalization based on BSA resulted in higher Z-scores in the underweight group compared to the overweight group. In other words, increasing BMI had an opposite effect on height-based Z-scores compared to BSA-based Z-scores. Allometric normalization based on height and BSA for aortic and LV sizes is feasible. However, height-based normalization results in higher cardiovascular Z-scores in heavier children, and BSA-based normalization results in higher cardiovascular Z-scores in lighter children. Further studies are needed to assess the performance of these approaches in obese children with or without cardiac disease.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Normalization of the sizes of cardiovascular structures allows for comparison of measurements among children with different body sizes. In addition, it accounts for the physiologic effect of somatic growth and highlights the pathologic effect of disease processes on cardiovascular sizes [1]. The Pediatric Heart Network (PHN) recently calculated Z-scores based on body surface area (BSA) for common echocardiographic measurements to account for the effects of somatic growth on the sizes of cardiovascular structures in a large group of healthy and racially diverse children [2]. The PHN Echo Z-Score Project also determined that age, sex, race, and ethnicity did not have a significant effect on the relationship between cardiovascular measurements and BSA.
Many investigators have suggested the use of height instead of BSA to normalize cardiovascular measurements, because BSA does not characterize the individual effects of lean body mass and fat mass on cardiovascular growth, particularly in the overweight and obese populations [3].
Cardiovascular structures are sized to meet stroke volume requirements, but the effect of fat mass on stroke volume requirements is less than that of lean body mass [4]. In addition, visceral and subcutaneous fat have different effects on the cardiovascular system, with possibly pathologic impact by visceral fat on cardiovascular sizes [5]. This issue has been particularly highlighted in studies looking at normalized left ventricular (LV) mass to determine LV hypertrophy [6, 7].
Normalized aortic and LV measurements are often used to make clinical decisions related to medical and surgical interventions in children. The aim of this study was to develop and compare models for height-based and BSA-based normalization of aortic and LV sizes in a subset of the PHN study population of normal children. In addition, the impact of other demographic and anthropometric variables on both models was assessed. We hypothesized that height-based normalization models would result in a similar correlation coefficient as BSA-based normalization models, and the residual relationships of the models with BMI Z-score would be the same.
Methods
This study was a secondary analysis of children with normal echocardiograms collected from 19 North American centers as part of the PHN Echo Z-score Project. The retrospective study design assured adequate enrollment across the entire pediatric age range with equal numbers of boys and girls across a broad range of race and ethnicity categories, using specific inclusion and exclusion criteria as outlined in the methodology [2]. As previously reported because all submissions were de-identified, most children were retrospectively enrolled under a waiver of consent after Institutional Review Board (IRB) or Research Ethics Board approval. Race/ethnicity information was not routinely obtained at one center and was collected prospectively for eligible subjects after local regulatory approval. Some centers were able to perform research echocardiograms without charge and prospectively enrolled healthy children after IRB approval. For this analysis, the study population included healthy, non-obese children [body mass index (BMI) < 95th percentile for age and sex] over 2 years of age from the PHN Echo Z-Score Project. Subjects were categorized into 3 weight groups based on BMI: underweight (BMI < 5th percentile), normal weight (BMI in 5th to < 85th percentile), and overweight (BMI in 85th to < 95th percentile). Children younger than 2 years old from the original PHN cohort were excluded because the definition of underweight and overweight based on BMI is limited to children over 2 years old [8]. The study parameters for the analysis included the aortic annulus, aortic root, ascending aorta, LV end-diastolic volume, and LV mass, all measured using standard pediatric echocardiographic methods [9].
Statistical Analysis
The study parameters were normalized based on height using the same methodology based on BSA in the PHN Echo Z-Score Project [2]. In order to determine the best exponential transformation of height (heightα) for each parameter (X), several values of α were explored by nonparametric locally weighted scatterplot smoothing (LOESS) curve fitting [10], histograms, and linear regression plots of indexed parameter and height. The best height transformation was chosen when (1) there was a linear relationship between X and heightα, (2) the indexed parameter (X/heightα) was normally distributed, and (3) there was no clinically significant residual dependence of the indexed parameter on height (with a zero slope when the indexed parameter is plotted against height). Published reproducibility thresholds have reported that measurement variability is responsible for at least 5% of measurement differences for primary measurements such as aortic diameters and up to 10% of measurement differences for calculated parameters such as LV volume and mass [11]. Therefore, clinical significance for aortic measurements was defined as a difference of at least 5% between actual and predicted measurement values using models with and without the statistically significant effects. For LV volume and mass, a threshold of 10% was used to determine clinical significance.
The PHN Echo Z-Score Project determined the best exponential transformations of BSA (BSAα) for the study parameters and calculated BSA-based z-scores based on the mean and standard deviation for the indexed values from the full cohort [2]. Due to exclusion of subjects < 2 years of age from the current analysis, the full PHN cohort was not included in these analyses, and the mean and standard deviation for the BSA-based indexed values were recalculated for this subset of the study population. Height-based and BSA-based Z-scores for this study population were calculated using the following equation:
After the height transformations were chosen for the height-based models, multivariable regressions were performed on the indexed parameters to assess for statistically and clinically significant linear and nonlinear effects of age, sex, race, and ethnicity as well as their interactions. A p-value of ≤ 0.05 was used for significant main effects and ≤ 0.01 for significant interaction effects. Higher order interactions were considered first and removed from the model if not significant. Lower order interactions and main effects were kept in the model even if not significant if the effect was part of a significant higher order interaction. Age was assessed both linearly and piece-wise linearly with plotting and nonparametric LOESS curve fitting [10]. To determine whether statistically significant main effects and interactions were clinically significant, predicted values from the two models with and without effects were tested to see if the predicted raw echo parameters from the two models differed by more than 5% for the aortic measurements and 10% for the LV calculations. This was assessed with a t-test of the absolute proportion difference between the models.
To assess for a residual relationship with BMI, the relationships between BMI Z-score and the indexed parameters based on height and on BSA were evaluated via linear regression and tested for statistical and clinical significance. Demographics and height-based and BSA-based Z-scores were compared among the 3 weight groups with analysis of variance (ANOVA), while the effect of BMI Z-score was assessed via linear regression modelling.
Results
Among the 3215 subjects in the PHN cohort, 2299 were greater than 2 years old with demographic and clinical characteristics as listed in Table 1. The chosen height transformation exponents (α) as well as the published BSA transformation exponents (α) for each parameter (X) are listed in Table 2. All indexed parameters (X/heightα and X/BSAα) were normally distributed based on visual inspection. There was a nearly linear relationship between all parameters and transformed height and BSA with correlations that ranged from 0.87 to 0.93 (p < 0.001 for all). The relationships of the indexed parameters with height and with BSA were statistically significant with non-zero slopes, but none of these differences were considered clinically significant as defined for this analysis (Table 2). Z-scores based on height were then calculated from (1) the mean indexed parameter values, (2) the values for α, and (3) the standard deviations for each parameter as listed in Table 2.
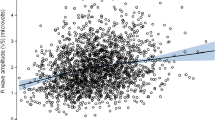
Multivariable regressions revealed statistically significant residual relationships of the indexed parameters with age, sex, race, and ethnicity, but none of the residual relationships were clinically significant (Supplementary Table A). There were also statistically, but not clinically, significant relationships between the indexed parameters and BMI Z-scores. Interestingly, when models that accounted for the effect of BMI Z-score were compared with models that ignored BMI Z-score, the percent differences between models increased from the 1st to the 3rd BMI quartile for the height-based models and decreased for the BSA-based models (Supplementary Table B). The median study parameter Z-scores based on height and BSA and their interquartile ranges for the underweight, normal weight, and overweight groups are depicted in Fig. 1. In the height-based normalization models, the median Z-scores were lower in the underweight group compared to the overweight group. In other words, the underweight group appeared to have smaller height-adjusted aortic and LV sizes than the overweight group with this model. On the other hand, in the BSA-based normalization models, the median Z-scores were increased in the underweight group compared to the overweight group. The relationship between body size parameter and BMI is further depicted in Fig. 2, showing that study parameter Z-scores based on height tended to increase with increasing BMI Z-score and those based on BSA tended to decrease with increasing BMI Z-score. Interestingly, for LV volume and mass, the absolute values for the slopes of the linear relationships were higher for Z-scores based on height compared to those based on BSA, indicating that normalization for height was less successful than normalization for BSA at eliminating residual dependence on body size in this non-obese population.
Discussion
This study calculated Z-scores based on height for aortic and LV measurements and compared them to Z-scores based on BSA in a large group of healthy, non-obese children. The models for normalization based on height and BSA are similar in terms of heteroscedasticity and absence of clinically significant residual relationships with age, sex, race, and ethnicity. Importantly, BMI Z-scores have a positive residual relationship with height-based Z-scores, which are higher for the overweight population, and a negative residual relationship with BSA-based Z-scores, which are lower for the overweight population.
This study used height to normalize aortic diameters and LV volumes in a large group of children. Previous studies used height to normalize LV mass as calculated from M-mode measurements in children and adults [12,13,14,15]. In contrast, the PHN study used 2-dimensional echocardiographic measurements and the area-length method to establish LV mass Z-scores based on BSA in children [2]. Studies comparing height-based and BSA-based normalization models in the adult population have resulted in variable conclusions, likely related to the variable prevalence of obesity in the study populations [3, 7, 16,17,18]. Krysztofiak and colleagues studied a group of adolescent athletes and, like our study, also found that height-based LV mass Z-scores were higher and BSA-based LV mass Z-scores lower in the overweight athletes [19].
Models based on BSA normalize measurements by assuming that the effects of lean body mass and fat mass are equivalent, whereas models based on height place more emphasis on the effects of lean body mass. Both models tend to adequately account for the effect of body size for individuals with normal weight and a more predictable ratio of lean body mass to fat mass, but both fail to account for potential differential effects of lean and fat tissue on cardiac output. If the goal is to normalize the potential effects of fat mass and assess the effects of other pathologic factors on the heart, then some have suggested that normalization based on BSA may be more useful [20, 21]. On the contrary, if the goal is to preferentially evaluate the effect of lean body mass over fat mass on cardiovascular growth, specifically when comparing normal weight to overweight subjects, then normalization based on height may be more useful. It is important to remember, however, that height, like BSA, does not provide information on the actual ratio of lean body mass to fat mass, limiting its ability to fully distinguish between the effects of lean versus fat mass on cardiovascular sizes.
With the increasing prevalence of obesity and obesity-related risk factors such as hypertension that affect cardiovascular size, assessment of the effect of fat mass in addition to other pathologic factors may be more relevant when caring for obese patients. Interestingly, Foster and colleagues found that, in obese children, normalization based on lean body mass was more concordant with normalization based on BSA than with normalization based on height [22]. In addition, Mahgerefteh and colleagues found that normalization by lean body mass removed the effect of blood pressure on LV mass [6].
The PHN Echo Z-Score Project excluded obese children from the study population, precluding a comparison of the normalization approaches in this group. When overweight children are compared with normal weight and underweight children, there are significant differences in aortic and LV Z-scores among the 3 groups. Cardiovascular structures seem bigger in the overweight group with height-based normalization and smaller with BSA-based normalization in the same population. An illustrative example of these differences can be provided by 2 children, one underweight and the other overweight, with a LV mass of 75 g. If both children had a height of 1.4 m, the height-based LV mass Z-score is 0.6 for both subjects but the BSA-based LV mass Z-score would be + 2.2 for the underweight child and − 0.4 for the overweight child. If we perform the same analysis for 2 children with a BSA of 1.15 m2, the BSA-based LV mass Z-score is + 1 for both subjects, but the height-based LV mass Z-score would be − 0.6 for the underweight child and + 2.8 for the overweight child (Supplementary Table C).
In fact, when looking specifically at the relationship between BMI Z-scores (as a measure of adiposity) and LV mass Z-scores based on both approaches (Fig. 2), the downward slope of the BSA-based relationship is less steep than the upward slope of the height-based relationship. In other words, height-based Z-scores were more likely to demonstrate the difference between underweight and overweight children than BSA-based Z-scores. The persistent relationship between height-based LV mass Z-scores and BMI Z-scores may provide some insight into the actual effect of adiposity on LV mass in underweight children.
Limitations
This was a retrospective study with the known biases associated with this type of analysis. Furthermore, this study was limited to non-obese children, so one cannot fully extrapolate the findings to obese children who usually have other comorbidities that may affect cardiovascular growth. Moreover, patients with systemic hypertension and other systemic disorders affecting the heart were also excluded from this evaluation. Simultaneous blood pressure and measurement data were not collected to assess the impact of blood pressure on aortic and LV sizes. The threshold of 5% and 10% for clinical significance of residual relationships for aortic and LV calculations, respectively, were based on previously published inter-observer variability. In order to decrease the complexity of the analysis, we limited the analysis to aortic and LV measurements, and the results cannot be generalized to other echocardiographic measures. Finally, BMI as an index of adiposity is limited, and evaluating the individual effects of lean body mass and adiposity on cardiovascular growth is beyond the scope of this study.
Conclusion
Normalization of aortic and LV sizes based on height is feasible and comparable to normalization based on BSA in healthy, non-obese children. The use of height results in higher cardiovascular Z-scores in heavier children, whereas the use of BSA results in higher cardiovascular Z-scores in lighter children. The performance of normalization based on height and BSA in the obese population warrants further studies.
References
Sluysmans T, Colan SD (2005) Theoretical and empirical derivation of cardiovascular allometric relationships in children. J Appl Physiol 99(2):445–457. https://doi.org/10.1152/japplphysiol.01144.2004
Lopez L, Colan S, Stylianou M, Granger S, Trachtenberg F, Frommelt P, Pearson G, Camarda J, Cnota J, Cohen M, Dragulescu A, Frommelt M, Garuba O, Johnson T, Lai W, Mahgerefteh J, Pignatelli R, Prakash A, Sachdeva R, Soriano B, Soslow J, Spurney C, Srivastava S, Taylor C, Thankavel P, van der Velde M, Minich L, Pediatric Heart Network I (2017) Relationship of echocardiographic Z scores adjusted for body surface area to age, sex, race, and ethnicity: the pediatric heart network normal echocardiogram database. Circ Cardiovasc Imaging. https://doi.org/10.1161/CIRCIMAGING.117.006979
de Simone G, Kizer JR, Chinali M, Roman MJ, Bella JN, Best LG, Lee ET, Devereux RB, Strong Heart Study I (2005) Normalization for body size and population-attributable risk of left ventricular hypertrophy: the Strong Heart Study. Am J Hypertens 18(2 Pt 1):191–196. https://doi.org/10.1016/j.amjhyper.2004.08.032
Frayn KN, Karpe F, Fielding BA, Macdonald IA, Coppack SW (2003) Integrative physiology of human adipose tissue. Int J Obes Relat Metab Disord 27(8):875–888. https://doi.org/10.1038/sj.ijo.0802326
Neeland IJ, Gupta S, Ayers CR, Turer AT, Rame JE, Das SR, Berry JD, Khera A, McGuire DK, Vega GL, Grundy SM, de Lemos JA, Drazner MH (2013) Relation of regional fat distribution to left ventricular structure and function. Circ Cardiovasc Imaging 6(5):800–807. https://doi.org/10.1161/CIRCIMAGING.113.000532
Mahgerefteh J, Linder J, Silver EJ, Hazin P, Ceresnak S, Hsu D, Lopez L (2016) The prevalence of left ventricular hypertrophy in obese children varies depending on the method utilized to determine left ventricular mass. Pediatr Cardiol 37(6):993–1002. https://doi.org/10.1007/s00246-016-1380-0
de Simone G, Devereux RB, Maggioni AP, Gorini M, de Divitiis O, Verdecchia P, Group MS (2005) Different normalizations for body size and population attributable risk of left ventricular hypertrophy: the MAVI study. Am J Hypertens 18(10):1288–1293. https://doi.org/10.1016/j.amjhyper.2005.05.027
Barlow SE, Expert C (2007) Expert committee recommendations regarding the prevention, assessment, and treatment of child and adolescent overweight and obesity: summary report. Pediatrics 120(Suppl 4):S164-192. https://doi.org/10.1542/peds.2007-2329C
Lopez L, Colan SD, Frommelt PC, Ensing GJ, Kendall K, Younoszai AK, Lai WW, Geva T (2010) Recommendations for quantification methods during the performance of a pediatric echocardiogram: a report from the Pediatric Measurements Writing Group of the American Society of Echocardiography Pediatric and Congenital Heart Disease Council. J Am Soc Echocardiogr 23(5):465–495. https://doi.org/10.1016/j.echo.2010.03.019 (quiz 576-467)
Cleveland WS, Devlin SJ (1988) Locally weighted regression: an approach to regression analysis by local fitting. J Am Stat Assoc 83(403):596–610. https://doi.org/10.1080/01621459.1988.10478639
Colan SD, Shirali G, Margossian R, Gallagher D, Altmann K, Canter C, Chen S, Golding F, Radojewski E, Camitta M, Carboni M, Rychik J, Stylianou M, Tani LY, Selamet Tierney ES, Wang Y, Sleeper LA, Pediatric Heart Network I (2012) The ventricular volume variability study of the Pediatric Heart Network: study design and impact of beat averaging and variable type on the reproducibility of echocardiographic measurements in children with chronic dilated cardiomyopathy. J Am Soc Echocardiogr 25(8):842–854. https://doi.org/10.1016/j.echo.2012.05.004
Chinali M, Emma F, Esposito C, Rinelli G, Franceschini A, Doyon A, Raimondi F, Pongiglione G, Schaefer F, Matteucci MC (2016) Left ventricular mass indexing in infants, children, and adolescents: a simplified approach for the identification of left ventricular hypertrophy in clinical practice. J Pediatr 170:193–198. https://doi.org/10.1016/j.jpeds.2015.10.085
de Simone G, Devereux RB, Daniels SR, Koren MJ, Meyer RA, Laragh JH (1995) Effect of growth on variability of left ventricular mass: assessment of allometric signals in adults and children and their capacity to predict cardiovascular risk. J Am Coll Cardiol 25(5):1056–1062
Khoury PR, Mitsnefes M, Daniels SR, Kimball TR (2009) Age-specific reference intervals for indexed left ventricular mass in children. J Am Soc Echocardiogr 22(6):709–714. https://doi.org/10.1016/j.echo.2009.03.003
Foster BJ, Mackie AS, Mitsnefes M, Ali H, Mamber S, Colan SD (2008) A novel method of expressing left ventricular mass relative to body size in children. Circulation 117(21):2769–2775
Chirinos JA, Segers P, De Buyzere ML, Kronmal RA, Raja MW, De Bacquer D, Claessens T, Gillebert TC, St John-Sutton M, Rietzschel ER (2010) Left ventricular mass: allometric scaling, normative values, effect of obesity, and prognostic performance. Hypertension 56(1):91–98. https://doi.org/10.1161/HYPERTENSIONAHA.110.150250
Kuznetsova T, Haddad F, Tikhonoff V, Kloch-Badelek M, Ryabikov A, Knez J, Malyutina S, Stolarz-Skrzypek K, Thijs L, Schnittger I, Wu JC, Casiglia E, Narkiewicz K, Kawecka-Jaszcz K, Staessen JA, European Project on Genes in Hypertension I (2016) Impact and pitfalls of scaling of left ventricular and atrial structure in population-based studies. J Hypertens 34(6):1186–1194. https://doi.org/10.1097/HJH.0000000000000922
Armstrong AC, Gidding S, Gjesdal O, Wu C, Bluemke DA, Lima JA (2012) LV mass assessed by echocardiography and CMR, cardiovascular outcomes, and medical practice. JACC Cardiovasc Imaging 5(8):837–848. https://doi.org/10.1016/j.jcmg.2012.06.003
Krysztofiak H, Mlynczak M, Malek LA, Folga A, Braksator W (2019) Left ventricular mass is underestimated in overweight children because of incorrect body size variable chosen for normalization. PLoS ONE 14(5):e0217637. https://doi.org/10.1371/journal.pone.0217637
de Simone G, Galderisi M (2014) Allometric normalization of cardiac measures: producing better, but imperfect, accuracy. J Am Soc Echocardiogr 27(12):1275–1278. https://doi.org/10.1016/j.echo.2014.10.006
Cuspidi C, Meani S, Negri F, Giudici V, Valerio C, Sala C, Zanchetti A, Mancia G (2009) Indexation of left ventricular mass to body surface area and height to allometric power of 2.7: is the difference limited to obese hypertensives? J Hum Hypertens 23(11):728–734. https://doi.org/10.1038/jhh.2009.16
Foster BJ, Gao T, Mackie AS, Zemel BS, Ali H, Platt RW, Colan SD (2013) Limitations of expressing left ventricular mass relative to height and to body surface area in children. J Am Soc Echocardiogr 26(4):410–418. https://doi.org/10.1016/j.echo.2012.11.018
Acknowledgements
A list of all Pediatric Heart Network Investigators is provided in the Supplementary Materials.
Funding
This work was supported by the National Institutes of Health Grants (HL068270, HL068290, HL109673, HL109737, HL109741, HL109743, HL109777, HL109778, HL109781, HL109816, and HL109818).
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Conflict of interest
No relevant or potentially relevant conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mahgerefteh, J., Lai, W., Colan, S. et al. Height Versus Body Surface Area to Normalize Cardiovascular Measurements in Children Using the Pediatric Heart Network Echocardiographic Z-Score Database. Pediatr Cardiol 42, 1284–1292 (2021). https://doi.org/10.1007/s00246-021-02609-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00246-021-02609-x