Abstract
Previous studies indicated a positive effect of vitamin K2 (VK2) supplementation on bone turnover biomarkers and bone mineral density (BMD), but the doses varied, and few studies have focused on the difference between VK2 supplementation alone and in combination with calcium and vitamin D3. The aim of this study was to explore a low and effective dose of VK2 for improving BMD, and to examine whether the co-supplementation of VK2, calcium and vitamin D3 would bring greater effects. In this trial, a total of 311 community-dwelling men and postmenopausal women aged 50 and 75 years were randomly assigned to four groups, receiving placebo, 50 µg/day, 90 µg/day or co-supplementation with calcium (500 mg/day) and vitamin D3 (10 µg/day) for 1 year. At the endpoint, the bone loss of femoral neck was significantly lower in postmenopausal women in the two 90 µg groups (treatment × time, p = 0.006) compared with placebo, but no effects in men. Serum biomarkers cOC/ucOC ratio increased in the intervention groups (treatment × time, p < 0.001). VK2 supplementation in dose of 90 µg/day performed a significant effect on reducing bone loss in postmenopausal women, but in combination with calcium and vitamin D3 brought no additional effects.
Trial registration This trial was registered at http://www.chictr.org.cn as chiCTR1800019240.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Osteoporosis is a major public health problem, which is characterized by low BMD and deterioration of bone microarchitecture, leading to increased risk to fragility fracture and having a negative impact on quality of life in high-risk populations [1]. Calcium and vitamin D supplementation is a common nutritional intervention to protect or treat osteoporosis over the past years [2,3,4], but some studies suggested it has no significant effect on increasing BMD or reducing fracture incidence [5, 6]. Therefore, more effective nutrition-related approaches are being explored.
From previous studies, vitamin K plays an important role in improving bone health by acting as a cofactor of γ-glutamyl carboxylase to activate vitamin K-dependent proteins [7]. Osteocalcin, one kind of vitamin K-dependent proteins, is an important bone turnover biomarker, and compared with undercarboxylated osteocalcin (ucOC), the carboxylated osteocalcin (cOC) can bind calcium ions to bone matrix to promote bone mineralization [8, 9]. To maintain sufficient levels, the carboxylation process relies on a vitamin K cycle in the body [10]. Nonetheless, adequate nutritional status of VK is essential.
Vitamin K (VK) is one kind of fat-soluble vitamin, with two dietary forms (VK1 and VK2) [11, 12]. VK1 is mainly found in plants, while VK2 is contained in some fermented foods, such as natto and cheese, and also endogenously synthesized by intestinal bacteria [13]. Compared with VK2, VK1 contributes majority parts of dietary sources of VK for its relatively high content in foods [14], but as for improving bone health, VK2 serves as a major contributor [15, 16], and has a greater effect on stimulating bone calcium deposition [17]. Two subtypes of vitamin K2 are commonly used in clinical trials, MK-4 and MK-7, MK-7 was chosen in this study for its longer half-time and lower effective dosage [18, 19].
Most observational studies indicated a direct relationship between VK intake, either VK1 or VK2, and BMD and risk of fracture [20]. However, in elderly, low food intake and poor nutrition condition of VK, especially VK2, has been a concern, and the increased risk of osteoporosis and fracture also in turn highlights a higher age-related requirement [21, 22]. Some randomized controlled trials, although designed for different intervention doses and periods, have shown that VK2 supplementation did promote bone health, which reflected in improving bone metabolism biomarkers, increasing BMD or reducing bone loss and preserving bone structure [23,24,25,26]. Thus, considering the liposolubility of VK2 and on the basis of the available evidence, we tried to explore the minimum effective dose of VK2 supplementation to improve BMD. To observe the treatment effect of VK2 on bone and reduce the interference of some possible factors caused by excessively long time, the intervention time was one year. Nowadays, the supplements of VK2 were mainly applied in two ways, alone or in combination with calcium and (or) vitamin D3, but few studies focused on their difference. So, another objective of this study was to examine if additional calcium and vitamin D3 supplementation would show a synergistic effect on improving BMD.
The aim of this 12-month randomized controlled trial was to assess the effects of VK2 supplementation on bone health in two doses in a monotherapy way, and simultaneously compared with in combination with calcium and vitamin D3 in middle-aged and elderly population in China. The primary outcome was BMD at the lumbar spine (L1–L4), femoral neck and total hip.
Methods
Participants
We recruited healthy community-dwelling men and postmenopausal women aged 50 and 75 years in November 2018 from the Harbin Cohort Study on Diet, Nutrition and Chronic Non-communicable Diseases (HDNNCDS) [27]. Inclusion criteria were T-scores of the lumbar spine (L1–L4) and (or) hip lower than − 1.0, body mass index (BMI) between 18 and 30 kg/m2. They were excluded if they had taken vitamin D, calcium, vitamin K, calcitonin, diphosphonate for osteoporosis in the past 6 months; vitamin K antagonists within the 1 year, such as warfarin; estrogen or other hormone therapy in past 1 years; history of chronic diseases of kidney, liver, lung, or pancreas; hyperthyroidism, hyperparathyroidism, osteomalacia; history of malignant tumors.
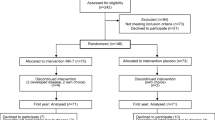
At first screening in April 2017, a total of 860 residents accepted bone mineral density measurement and accomplished the questionnaires and physical examination, there were 478 subjects meeting our inclusion criteria, and 311 subjects participated in the second screening in November 2018 and entered into the study (Fig. 1). All participants gave written informed consent before entering the study. Ethical requirements stated by the Declaration of Helsinki and other international regulations are met by this study. The study was approved by the Ethical Committee of Harbin Medical University (No. HMUIRB2018RCT002). This trial was registered at https://www.chictr.org.cn (code: chiCTR1800019240).
Study Design and Intervention
This study was designed as a 12-month, randomized controlled and single-blind trial to examine the effects of vitamin K2 (MK-7) supplementation on bone health. Two subtypes of vitamin K2 are used in clinical trials, MK-4 and MK-7, we chose MK-7 for its longer half-time and lower effective dosage [18]. The study was performed at the Harbin Medical University, Harbin, China, between September 2018 and December 2019.
According to baseline T-scores, 311 subjects were randomly assigned into four study groups (Placebo, VK2-1, VK2-2 and VK2DCa group) by block randomization with software-generated random numbers. The VK2-1 group and VK2-2 group received vitamin K2 (MK-7) 50 µg/day and 90 µg/day, respectively. The VK2DCa group received vitamin K2 (MK-7) 90 µg/day in combination with calcium 500 µg/day and vitamin D3 10 µg/day. The placebo group was set as a control group receiving placebo. All the three kinds of tablets contain MK-7 and placebo was provided by SUNGEN BIOSCIENCE CO., LTD. The tablets were similar in appearance and taste. All subjects were asked to take one tablet every evening after dinner and record their tablets intake. The records and empty bottles were returned per two months and then the next batch tablets were provided. Compliance was estimated with tablet counts.
Measurements
The primary outcome was the BMD of the lumbar spine (L1–L4) and left hip. BMDs of the lumbar spine (L1–L4) and left hip (total hip and neck) were measured by dual energy X-ray absorptiometry (DEXA, Hologic Acclaim QDR4500W, Hologic, USA) and analyzed according to standard protocol at Logistic Support Forces of the Chinese People's Liberation Army 962 Hospital.
Blood samples were collected in the morning after 8 h fasting. Serum vitamin K2, cOC, ucOC, 25-hydroxyvitamin D (25(OH)D) and calcium concentrations were measured at baseline and endpoint of intervention. Serum vitamin K2 was measured by double-antibody sandwich ELISA (MLBIO Biotechnology, Shanghai, China). Serum cOC and ucOC were measured by Gla-OC and Glu-OC EIA Kits (Takara, Japan), and the cOC/ucOC ratio was calculated from the cOC and ucOC concentrations. Serum 25(OH)D was extracted with Oasis HBL 96-well µElution Plates (Waters, Milford, MA, USA) and quantified with the ultra-performance liquid chromatography-tandem mass spectrometry (UPLC–MS/MS, Waters). Serum calcium was measured by a Roche Modular P800 Automatic Biochemical Analyzer (Roche Diagnostics, Mannheim, Germany).
Body fat was measured by using the electric impedance method with a body fat mass analyzer (OMRON HBF-306, Omron Corporation, Dalian, China). A food-frequency questionnaire was applied to assess dietary intake of subjects and estimated by using the Food Nutrition Calculater (V1.6; Chinese CDC).
Statistical Analysis
The sample size calculation was based on BMD change data from a previous study examining the effect of vitamin K2 supplementation on BMD in postmenopausal women, which observed BMD changes of 0.006 g/cm2 at lumber spine in the VK2 intervention group, and − 0.033 g/cm2 in the control group [23]. According to the data and considering a 15% dropout rate, 36 participants were needed in each group (2-sided, 80% power, and 0.05 α).
The results were expressed as means ± SDs, and changes between baseline and endpoint are presented as means (95% CI). The distribution of data was analyzed by descriptive statistics and Kolmogorov–Smirnov test. Univariate ANOVA was used to analyze the differences of continuous variables among four groups. Chi-square test was used to compare the differences among groups of classified variables. The effect of VK2 supplementation on the change of BMD, serum VK2, bone biomarkers and 25(OH)D was assessed by using linear mixed models with treatment, time and treatment × time interaction as fixed effects and participant included as a random effect. And if the linear mixed models analysis showed a significant of the interaction effect, pairwise comparisons the interaction effect were assessed by the model. For variables, values < 3 or > 3 SDs from the mean were considered outliers and removed. The p < 0.05 (2 tailed) was set as the threshold for statistical significant. Statistical analyses were performed with SPSS 21.0 (Beijing stats Data Mining Co. Ltd, Beijing, China).
Results
A total of 295 participants completed the study (Fig. 1). Sixteen subjects did not complete the study for the following reasons: 8 subjects could no longer be contacted, and 8 subjects withdrew from the study for personal reasons. The rates of tablets intake were 97.7%, 97.8%, 97.4% and 97.5% in the Placebo, VK2-1, VK2-2 and VK2DCa groups, respectively (p = 0.893).
The baseline characteristics were not significantly different among the four study groups (Table 1). The average age was 59.78 ± 6.60 years, and BMI of subjects was 24.08 ± 3.27 kg/m2. T-scores of the lumbar spine and total hip were − 1.62 ± 1.3 and − 1.06 ± 0.96, respectively. In this study, women constituted 64.73% of all subjects, mean menopausal age was 50.32 ± 3.84 years, and mean years since menopausal of female subjects was 10.19 ± 6.26 years.
After the 12-month intervention, no significant differences were observed in BMD changes of lumbar spine (L1–L4), femoral neck or total hip from baseline and endpoint among four groups in general population, but we observed a slight but non-significant BMD increase in the VK2DCa group at lumbar spine (0.002 g/cm2; 95% CI − 0.005, 0.009) but decreased in the placebo group (− 0.006 g/cm2; 95% CI − 0.017, 0.004), and a lower bone loss in the VK2DCa group (− 0.003 g/cm2; 95% CI − 0.012, 0.005) at femoral neck compared with the placebo group (− 0.015 g/cm2; 95% CI: − 0.031, 0.001). What’s more, difference of BMD changes between the VK2-2 group and the VK2DCa group was not statistically significant.
After 12-month vitamin K2 supplementation, serum vitamin K2 increased in all the three treatment groups (VK2-1 group: + 0.43 nmol/L; VK2-1 group: + 0.22 nmol/L; VK2DCa group: + 0.40 nmol/L) (treatment × time, p = 0.015). Similarly, in the three treatment groups, cOC/ucOC ratio increased (treatment × time, p < 0.001) but unchanged in the placebo group. Serum 25(OH)D of VK2DCa group increased significantly after additional vitamin D3 supplementation (treatment × time, p = 0.013) (Table 2).
A subgroup analysis was applied to examine the effects of vitamin K2 supplements on bone health in postmenopausal women. When analyzed with stratified for gender, changes in BMD at the femoral neck showed an obvious decline tendency from the placebo group to VK2DCa group in women (treatment × time, p = 0.006). Compared with the placebo group (− 0.025 g/cm2; 95% CI − 0.049, − 0.001), the VK2DCa and VK2-2 group both showed a decreased bone loss, − 0.000 g/cm2 (95% CI − 0.011, 0.011) (treatment × time, p = 0.036) for VK2DCa group and − 0.000 g/cm2 (95% CI − 0.02, 0.02) (treatment × time, p = 0.039) for VK2-2 group (Fig. 2). No significant difference was observed at the lumbar spine and total hip (Fig. 2). The cOC/ucOC ratio also increased in the three treatment groups (p = 0.013) (Fig. 3), and serum 25(OH)D also increased significantly in VK2DCa group (p = 0.004). We did not observe similar results in male subjects (Figs. 2, 3).
Effect of VK2 supplementation on changes from baseline in the lumbar spine, total hip and femoral neck BMD of female and male subjects during the intervention. Values were expressed as means (SEM). For female, n = 38, 52, 46 and 53, and for male n = 20, 22, 23 and 27 for placebo, VK2-1 (MK-7: 50 µg/day), VK2-2 (MK-7: 50 µg/day), VK2DCa (MK-7: 90 µg/day, Ca: 500 mg/day, VD3: 10 µg/day) groups, respectively. p were treatment × time, p values based on linear mixed model. *p < 0.05, significant change from the baseline value
Treatment effects on serum VK2, 25(OH)D and cOC/ucOC ratio in female and male subjects. Values were expressed as means (SEM). For female, n = 38, 51, 48 and 53, and for male n = 23, 26, 25 and 23 for placebo, VK2-1 (MK-7: 50 µg/day), VK2-2 (MK-7: 50 µg/day), VK2DCa (MK-7: 90 µg/day, Ca: 500 mg/day, VD3: 10 µg/day) groups, respectively. p were treatment × time, p values based on linear mixed model
Discussion
This 12-month randomized controlled trial was designed to examine the effects of VK2 supplementation alone and in combination with calcium and vitamin D3 on bone health in middle-aged and elderly women and men. We observed that VK2 supplementation in dose of 90 µg/day decreased bone loss at the femoral neck in postmenopausal women, but the combination with calcium and vitamin D3 did not bring additional benefits.
VK2 is also known as menaquinones, abbreviated as MK-n (n ranges from 1 to 13) depending on its number of isoprenyl units and length of side chain. Among the subtypes, MK-4 and MK-7 are more commonly used to improve bone health, but MK-7 has a longer half-life and higher bioavailability [18], and the number of clinical trials with MK-7 has increased in number recently. To improve bone health, a large range of doses has been used in previous studies. Although few adverse events have been reported in VK2 clinical trials [19], actually, for the micronutrients supplementation, more is not better. So we tried to find a relatively low and effective dose. A low dose of MK-7 around 50 µg/day was reported to maintain circulating status and improve bone biomarkers effectively [28]. We wanted to focus on not only biomarkers, also the BMD which is more closely related to bone health condition. Therefore, in our study, trying to provide some evidence for finding a threshold dose of MK-7 to improve BMD, we chose two relatively low doses of MK-7 (50 µg/day and 90 µg/day) supported by previous studies [29, 30]. Considering the key role of VK2 in the transport of calcium, we also designed a group in combination with calcium and vitamin D3 to explore whether there is a synergistic effect among them.
At the 1 year endpoint, we only observed a positive effect on inhibiting bone loss with 90 µg MK-7 supplementation in postmenopausal women. Some higher doses have been used in previous studies, but results showed that the effects on BMD did not seem to be proportional to the doses used. A study in 334 Norwegian women, aged between 50 and 60 years, with MK-7 supplementation alone (360 µg MK-7/day vs. placebo) showed no effect on bone loss rates at the total hip or any other measurement site after 1 year [31], and the effect also did not observed in a higher dose with 375 µg MK-7/day [26]. However, a 3-year study with a half dose MK-7 180 µg/day, carried out in 244 healthy Dutch postmenopausal women aged between 55 and 65 years demonstrated that MK-7 could maintain bone mass at lumbar spine [25]. Femoral neck, though not the same measurement site, reflected a similar effect in our study, and the results indicated that 90 µg MK-7/day might be a threshold dose to decrease bone loss effectively in postmenopausal women. Base on the above, it suggests a possibility that the relationship between the dose and effect of MK-7 supplementation might similar to be a bell-shaped curve. As recently reported, compared with the low-dose vitamin D3, when the dose increased to a certain extent, not only did it not improve BMD, but had a negative effect and resulted in significantly lower BMD [32].
VK2 plays an important role in the process of transporting calcium to bone [10]. In vitro experiments suggested that co-supplementation of VK2 and vitamin D3 might result in an optimal effect, similar to synergistic effect [33]. In our study, we investigated the effect of VK2 (90 µg MK-7/day) in combination with calcium and vitamin D3, 500 mg/day and 10 µg/day, respectively, which maintained the bone mineral density at femoral neck effectively as well. Consistent with our results, a 1-year study with MK-7 100 µg/day, calcium 800 mg/day and vitamin D3 10 µg/day showed a increase in BMD at lumbar spine and the whole body [24]. To our knowledge, few studies focused on the difference between MK-7 supplementation alone and in a combination way. In our study, compared with the monotherapy, a very close result, but not better, was observed in the co-supplementation. This result supported that the dose of 90 µg MK-7/day supplementation did have an effect on improving BMD. We guessed, in our target population, their body’s calcium and vitamin D reserves might be enough to meet the needs, therefore, no additional effects were observed.
BMDs of lumbar spine, total left hip and femoral neck were measured, after 1 year intervention, we only observed an expected effect at the femoral neck in postmenopausal women. This result also indicated a clinical value of MK-7 supplementation, because of such special anatomical location of the femoral neck, and fracture occurs at this site makes up the largest proportion in hip fracture [34]. As aging, the superolateral cortex of the femoral neck is getting thinning, which leads to fragility and risk of fracture increase [35]. Once fractures happen at this site, patients are prone to suffer a greater decrease in body function and quality of life than happen at elsewhere [36]. So, to reduce bone loss of this site is important. Normal bone remodeling is crucial to maintain bone mass, but osteopenia and osteoporosis patients always have a negative bone balance, i.e., the resorption of bone is more than formation [37]. Evidence from vitro studies prompted that VK2 also play a role in transcription regulation of bone remodeling, the corresponding mechanism is that VK2 can promote bone formation by suppressing expression of RANKL, a receptor activator of NF-κB, then inhibiting osteoclastogenesis, meanwhile, protecting osteoblasts from apoptosis and reduce cortical porosity [38,39,40].
So far, majority of relevant trials have been conducted in postmenopausal women, few carried out in men, thus, a part of male subjects was included in our study. The treatment effect of VK2 on BMD was not observed in male subjects, as female subjects. The serum biomarker related to VK2 supplementation just showed a lightly and non-significant change at the endpoint. A cross-sectional study including 1662 elderly Japanese men suggested that habitual natto intake was significantly associated with a beneficial effect on bone health, and the dose of VK2 intake was about 380 μg/day or more [41]. Another observational study in 1112 men and 1479 women aged 59 ± 9 years indicated that low dietary vitamin K intake was associated with low BMD in women instead of men [42]. So probably men need a higher dose of VK2 to observe an effect on biomarkers and bone loss. The rate of bone loss in men was very low, and a significantly improvement was hardly expected to observe if there was almost no bone loss during the one year of the study. Moreover, it could not exclude that fewer men than women in the analysis and there is a gender difference in the effect of VK2 supplementation on bone, further studies with larger sample size are needed to confirm it.
The strength of our study is that we designed two doses of MK-7, 50 µg/day and 90 µg/day, and tried to explore a dose–response relationship for improving BMD instead of just bone turnover biomarkers, and we found a relatively low but effective dose of 90 µg/day on reducing bone loss. In addition, we compared the differences between MK-7 supplementation alone and in combination with calcium and vitamin D3. There are also some limitations in our study. First, the sample size was relatively small when subjects were assigned into four study groups, as well as the subsequent subgroup analysis. What’s more, a similar effect was not observed in male subjects as female subjects, and we did not find out the exact reason in this study.
Conclusion
In conclusion, our findings showed that a 1-year vitamin K2 (MK-7) supplementation performed a positive effect on decreasing bone loss in postmenopausal women, but in combination with calcium and vitamin D3 brought no more effects. To improve bone health in postmenopausal women, we suggest a dose of 90 µg/day or more of MK-7 for a continuous supplementation.
References
Rachner TD, Khosla S, Hofbauer LC (2011) Osteoporosis: now and the future. Lancet 377(9773):1276–1287. https://doi.org/10.1016/S0140-6736(10)62349-5
Prince RL, Devine A, Dhaliwal SS, Dick IM (2006) Effects of calcium supplementation on clinical fracture and bone structure: results of a 5-year, double-blind, placebo-controlled trial in elderly women. JAMA Internal Med 166(8):869–875. https://doi.org/10.1001/archinte.166.8.869
Larsen ER, Mosekilde L, Foldspang A (2004) Vitamin D and calcium supplementation prevents osteoporotic fractures in elderly community dwelling residents: a pragmatic population-based 3-year intervention study. J Bone Miner Res 19(3):370–378. https://doi.org/10.1359/JBMR.0301240
Dawson-Hughes B, Harris SS, Krall EA, Dallal GE (1997) Effect of calcium and vitamin D supplementation on bone density in men and women 65 years of age or older. N Engl J Med 337(10):670–676. https://doi.org/10.1056/NEJM199709043371003
Kahwati LC, Weber RP, Pan H, Gourlay M, LeBlanc E, Coker-Schwimmer M, Viswanathan M (2018) Vitamin D, calcium, or combined supplementation for the primary prevention of fractures in community-dwelling adults: evidence report and systematic review for the US Preventive Services Task Force. JAMA 319(15):1600–1612. https://doi.org/10.1001/jama.2017.21640
Xiao Q, Murphy RA, Houston DK, Harris TB, Chow WH, Park Y (2013) Dietary and supplemental calcium intake and cardiovascular disease mortality: the National Institutes of Health-AARP diet and health study. JAMA Internal Med 173(8):639–646. https://doi.org/10.1001/jamainternmed.2013.3283
Shearer MJ, Fu X, Booth SL (2012) Vitamin K nutrition, metabolism, and requirements: current concepts and future research. Adv Nutr 3(2):182–195. https://doi.org/10.3945/an.111.001800
Atkins GJ, Welldon KJ, Wijenayaka AR, Bonewald LF, Findlay DM (2009) Vitamin K promotes mineralization, osteoblast-to-osteocyte transition, and an anticatabolic phenotype by {gamma}-carboxylation-dependent and -independent mechanisms. Am J Physiol Cell Physiol 297(6):C1358–1367. https://doi.org/10.1152/ajpcell.00216.2009
Booth SL (2009) Roles for vitamin K beyond coagulation. Annu Rev Nutr 29:89–110. https://doi.org/10.1146/annurev-nutr-080508-141217
Willems BA, Vermeer C, Reutelingsperger CP, Schurgers LJ (2014) The realm of vitamin K dependent proteins: shifting from coagulation toward calcification. Mol Nutr Food Res 58(8):1620–1635. https://doi.org/10.1002/mnfr.201300743
Gijsbers BL, Jie KS, Vermeer C (1996) Effect of food composition on vitamin K absorption in human volunteers. Br J Nutr 76(2):223–229. https://doi.org/10.1079/bjn19960027
Schurgers LJ, Teunissen KJ, Hamulyak K, Knapen MH, Vik H, Vermeer C (2007) Vitamin K-containing dietary supplements: comparison of synthetic vitamin K1 and natto-derived menaquinone-7. Blood 109(8):3279–3283. https://doi.org/10.1182/blood-2006-08-040709
Shearer MJ, Bach A, Kohlmeier M (1186S) Chemistry, nutritional sources, tissue distribution and metabolism of vitamin K with special reference to bone health. J Nutr 126(4 Suppl):1181S–1186S. https://doi.org/10.1093/jn/126.suppl_4.1181S
Booth SL, Pennington JA, Sadowski JA (1996) Food sources and dietary intakes of vitamin K-1 (phylloquinone) in the American diet: data from the FDA Total Diet Study. J Am Diet Assoc 96(2):149–154. https://doi.org/10.1016/s0002-8223(96)00044-2
Schurgers LJ, Vermeer C (2000) Determination of phylloquinone and menaquinones in food. Effect of food matrix on circulating vitamin K concentrations. Haemostasis 30(6):298–307. https://doi.org/10.1159/000054147
Schurgers LJ, Vermeer C (2002) Differential lipoprotein transport pathways of K-vitamins in healthy subjects. Biochim Biophys Acta 1570(1):27–32. https://doi.org/10.1016/s0304-4165(02)00147-2
Wu WJ, Gao H, Jin JS, Ahn BY (2019) A comparatively study of menaquinone-7 isolated from Cheonggukjang with vitamin K1 and menaquinone-4 on osteoblastic cells differentiation and mineralization. Food Chem Toxicol 131:110540. https://doi.org/10.1016/j.fct.2019.05.048
Sato T, Schurgers LJ, Uenishi K (2012) Comparison of menaquinone-4 and menaquinone-7 bioavailability in healthy women. Nutr J 11:93. https://doi.org/10.1186/1475-2891-11-93
Marles RJ, Roe AL, Oketch-Rabah HA (2017) US pharmacopeial convention safety evaluation of menaquinone-7, a form of vitamin K. Nutr Rev 75(7):553–578. https://doi.org/10.1093/nutrit/nux022
Palermo A, Tuccinardi D, D'Onofrio L, Watanabe M, Maggi D, Maurizi AR, Greto V, Buzzetti R, Napoli N, Pozzilli P, Manfrini S (2017) Vitamin K and osteoporosis: myth or reality? Metab Clin Exp 70:57–71. https://doi.org/10.1016/j.metabol.2017.01.032
Tsugawa N, Shiraki M, Suhara Y, Kamao M, Tanaka K, Okano T (2006) Vitamin K status of healthy Japanese women: age-related vitamin K requirement for gamma-carboxylation of osteocalcin. Am J Clin Nutr 83(2):380–386. https://doi.org/10.1093/ajcn/83.2.380
Theuwissen E, Magdeleyns EJ, Braam LA, Teunissen KJ, Knapen MH, Binnekamp IA, van Summeren MJ, Vermeer C (2014) Vitamin K status in healthy volunteers. Food Funct 5(2):229–234. https://doi.org/10.1039/c3fo60464k
Kanellakis S, Moschonis G, Tenta R, Schaafsma A, van den Heuvel EG, Papaioannou N, Lyritis G, Manios Y (2012) Changes in parameters of bone metabolism in postmenopausal women following a 12-month intervention period using dairy products enriched with calcium, vitamin D, and phylloquinone (vitamin K(1)) or menaquinone-7 (vitamin K (2)): the postmenopausal health study II. Calcif Tissue Int 90(4):251–262. https://doi.org/10.1007/s00223-012-9571-z
Moschonis G, Kanellakis S, Papaioannou N, Schaafsma A, Manios Y (2011) Possible site-specific effect of an intervention combining nutrition and lifestyle counselling with consumption of fortified dairy products on bone mass: the Postmenopausal Health Study II. J Bone Miner Metab 29(4):501–506. https://doi.org/10.1007/s00774-010-0256-2
Knapen MH, Drummen NE, Smit E, Vermeer C, Theuwissen E (2013) Three-year low-dose menaquinone-7 supplementation helps decrease bone loss in healthy postmenopausal women. Osteoporosis Int 24(9):2499–2507. https://doi.org/10.1007/s00198-013-2325-6
Ronn SH, Harslof T, Pedersen SB, Langdahl BL (2016) Vitamin K2 (menaquinone-7) prevents age-related deterioration of trabecular bone microarchitecture at the tibia in postmenopausal women. Eur J Endocrinol 175(6):541–549. https://doi.org/10.1530/EJE-16-0498
Na L, Wu X, Feng R, Li J, Han T, Lin L, Lan L, Yang C, Li Y, Sun C (2015) The harbin cohort study on diet, nutrition and chronic non-communicable diseases: study design and baseline characteristics. PLoS ONE 10(4):e0122598. https://doi.org/10.1371/journal.pone.0122598
Knapen MH, Braam LA, Teunissen KJ, Van't Hoofd CM, Zwijsen RM, van den Heuvel EG, Vermeer C (2016) Steady-state vitamin K2 (menaquinone-7) plasma concentrations after intake of dairy products and soft gel capsules. Eur J Clin Nutr 70(7):831–836. https://doi.org/10.1038/ejcn.2016.3
Theuwissen E, Cranenburg EC, Knapen MH, Magdeleyns EJ, Teunissen KJ, Schurgers LJ, Smit E, Vermeer C (2012) Low-dose menaquinone-7 supplementation improved extra-hepatic vitamin K status, but had no effect on thrombin generation in healthy subjects. Br J Nutr 108(9):1652–1657. https://doi.org/10.1017/S0007114511007185
Bruge F, Bacchetti T, Principi F, Littarru GP, Tiano L (2011) Olive oil supplemented with menaquinone-7 significantly affects osteocalcin carboxylation. Br J Nutr 106(7):1058–1062. https://doi.org/10.1017/S0007114511001425
Emaus N, Gjesdal CG, Almas B, Christensen M, Grimsgaard AS, Berntsen GK, Salomonsen L, Fonnebo V (2010) Vitamin K2 supplementation does not influence bone loss in early menopausal women: a randomised double-blind placebo-controlled trial. Osteoporos Int 21(10):1731–1740. https://doi.org/10.1007/s00198-009-1126-4
Burt LA, Billington EO, Rose MS, Raymond DA, Hanley DA, Boyd SK (2019) Effect of high-dose vitamin D supplementation on volumetric bone density and bone strength: a randomized clinical trial. JAMA 322(8):736–745. https://doi.org/10.1001/jama.2019.11889
Gigante A, Bruge F, Cecconi S, Manzotti S, Littarru GP, Tiano L (2015) Vitamin MK-7 enhances vitamin D3-induced osteogenesis in hMSCs: modulation of key effectors in mineralization and vascularization. J Tissue Eng Regen Med 9(6):691–701. https://doi.org/10.1002/term.1627
Li Y, Lin J, Cai S, Yan L, Pan Y, Yao X, Zhuang H, Wang P, Zeng Y (2016) Influence of bone mineral density and hip geometry on the different types of hip fracture. Bosn J Basic Med Sci 16(1):35–38. https://doi.org/10.17305/bjbms.2016.638
Mayhew PM, Thomas CD, Clement JG, Loveridge N, Beck TJ, Bonfield W, Burgoyne CJ, Reeve J (2005) Relation between age, femoral neck cortical stability, and hip fracture risk. Lancet 366(9480):129–135. https://doi.org/10.1016/S0140-6736(05)66870-5
Oden A, McCloskey EV, Johansson H, Kanis JA (2013) Assessing the impact of osteoporosis on the burden of hip fractures. Calcifi Tissue Int 92(1):42–49. https://doi.org/10.1007/s00223-012-9666-6
Langdahl B, Ferrari S, Dempster DW (2016) Bone modeling and remodeling: potential as therapeutic targets for the treatment of osteoporosis. Ther Adv Musculoskelet Dis 8(6):225–235. https://doi.org/10.1177/1759720X16670154
Urayama S, Kawakami A, Nakashima T, Tsuboi M, Yamasaki S, Hida A, Ichinose Y, Nakamura H, Ejima E, Aoyagi T, Nakamura T, Migita K, Kawabe Y, Eguchi K (2000) Effect of vitamin K2 on osteoblast apoptosis: vitamin K2 inhibits apoptotic cell death of human osteoblasts induced by Fas, proteasome inhibitor, etoposide, and staurosporine. J Lab Clin Med 136(3):181–193. https://doi.org/10.1067/mlc.2000.108754
Koshihara Y, Hoshi K, Okawara R, Ishibashi H, Yamamoto S (2003) Vitamin K stimulates osteoblastogenesis and inhibits osteoclastogenesis in human bone marrow cell culture. J Endocrinol 176(3):339–348. https://doi.org/10.1677/joe.0.1760339
Yamaguchi M, Weitzmann MN (2011) Vitamin K2 stimulates osteoblastogenesis and suppresses osteoclastogenesis by suppressing NF-kappaB activation. Int J Mol Med 27(1):3–14. https://doi.org/10.3892/ijmm.2010.562
Fujita Y, Iki M, Tamaki J, Kouda K, Yura A, Kadowaki E, Sato Y, Moon JS, Tomioka K, Okamoto N, Kurumatani N (2012) Association between vitamin K intake from fermented soybeans, natto, and bone mineral density in elderly Japanese men: the Fujiwara-kyo Osteoporosis Risk in Men (FORMEN) study. Osteoporos Int 23(2):705–714. https://doi.org/10.1007/s00198-011-1594-1
Booth SL, Broe KE, Gagnon DR, Tucker KL, Hannan MT, McLean RR, Dawson-Hughes B, Wilson PW, Cupples LA, Kiel DP (2003) Vitamin K intake and bone mineral density in women and men. Am J Clin Nutr 77(2):512–516. https://doi.org/10.1093/ajcn/77.2.512
Acknowledgements
The authors thank all the volunteers from the HDNNCDS for their participation in this study, and also thank all the colleagues who have taken part in the works for setting up, maintaining and following up this HDNNCDS cohort.
Funding
This study was funded by the Applied Technology Research and Development Plan of Heilongjiang Province (GA18C005).
Author information
Authors and Affiliations
Contributions
Author YL designed the study. She is guarantor. Author YZ prepared the first draft of the paper. Authors YZ, ZL, LD, YY, SY, YZ and HL contributed to the study investigation and experimental work. Authors YW and PW were responsible for statistical analysis of the data. All authors revised the paper critically for intellectual content and approved the final version. All authors agree to be accountable for the work and to ensure that any questions relating to the accuracy and integrity of the paper are investigated and properly resolved.
Corresponding authors
Ethics declarations
Conflict of interest
Yingfeng Zhang, Zhipeng Liu, Lili Duan, Yeyu Ji, Sen Yang, Yuan Zhang, Hongyin Li, Yu Wang, Peng Wang, Jiepeng Chen, and Ying Li declare that they have no conflicts of interest.
Human and Animal Rights
The study has been approved by the Ethical Committee of Harbin Medical University and was performed in accordance with the ethical standards as laid down in the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards.
Informed Consent
All participants gave written informed consent before entering the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhang, Y., Liu, Z., Duan, L. et al. Effect of Low-Dose Vitamin K2 Supplementation on Bone Mineral Density in Middle-Aged and Elderly Chinese: A Randomized Controlled Study. Calcif Tissue Int 106, 476–485 (2020). https://doi.org/10.1007/s00223-020-00669-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-020-00669-4