Abstract
Atrazine is an s-triazine herbicide widely used for the control of weeds, primarily in corn, sorghum and sugarcane crops. It is relatively persistent in the environment, moderately soluble in water and toxic to different organisms and humans. Its mobility through soil by leaching and runoff events frequently lead to contamination of sediments and water resources. Thus, atrazine has become a compound of public concern because it is frequently detected in surface, groundwater and rainfall samples in quantities exceeding the limit values set by regulatory agencies (the EU and the USA) for drinking water. In addition, several studies have shown its impact on the ecosystem and human health. For this reason, bioremediation strategies have been described to allow the removal of atrazine and avoid its dispersion in the environment. This chapter provides information on the behaviour and impact of atrazine in soil, aquatic ecosystems and non-target organisms and summarised current knowledge about bioremediation strategies for the clean-up of sites polluted with this herbicide. Recently, material-microbial-integrated technologies have been investigated in order to degrade atrazine, which will be also described. Finally, the bioremediation strategies are evaluated under laboratory and field conditions. Future advances related to atrazine degradation need to focus on an efficient removal and low environmental impact.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Atrazine is the most widely used s-triazine herbicides to control broadleaf weeds for many crops in the world. However, the benefit of weed elimination in crops is offset by the negative impact generated by the application of herbicides on the environment and on living beings. In fact, the atrazine contamination has become a growing public concern because it is one of the most commonly detected pesticides in soil, surface water and groundwater, representing a serious risk for the environment and public health (Nguyen et al. 2014). To minimise the damage caused by this herbicide, it is necessary to find and apply processes that allow its removal from contaminated sites. A great number of technologies are developed, such as adsorption, biodegradation and photochemical catalysis. Among the different methodologies proposed, bioremediation appears as a promising alternative that takes advantage of the metabolic potential of microorganisms to degrade contaminants and it can be carried out in different media such as sediments, soils, surface and groundwater, and biological sludges. In addition, both microbial-assisted plant remediation and the use of immobilised microorganisms have been also proposed for atrazine degradation. In the present chapter information on the behaviour and impact of atrazine on soil and aquatic ecosystems will be discussed. Besides, the progresses of researches based on the abovementioned biological treatment technologies for atrazine removal will be reviewed and summarised as well as the future prospects of these approaches.
2 Atrazine: Main Characteristics, Behavior and Environmental Impacts
2.1 Atrazine: Properties and Uses
Atrazine (2-chloro-4-ethylamino-6-isopropylamino-1,3,5-triazine) is an important s-triazine herbicide. Its molecular formula is C8H14ClN5; it is a solid crystalline powder and unstable at high temperature. Atrazine has a melting point of 175.8 °C, a water solubility of 33 mg L–1 (20 °C) and is readily soluble in organic solvents (Lewis et al. 2016). Atrazine is highly persistent with a half-life of 32–128 days in soil and of 100 days in water (Krutz et al. 2008). However, their half-life is increased in subsurface environments due to the low natural microbial degradation potential (Singh and Jauhari 2017).
Atrazine has been commercially available for more than 50 years. It was registered in Switzerland in 1958 and is widely used since the early 1960s in the USA, Europe, Africa, Asia and South America to control broadleaf weeds and some grasses weeds that affect mainly on corn, sorghum and sugarcane crops (Viegas et al. 2012). Nowadays, atrazine is the second most extensively used herbicide globally with an annual consumption of about 70,000–90,000 tons (Cao et al. 2021). The United States (USA) applied about 33,560 tons every year (Gaffar et al. 2021), while China has consumed 23,000 tons (Liu et al. 2021). South America is one of the areas of the world where atrazine is massively applied in agriculture. In Argentina, about 10,000 tons of atrazine are consumed annually (Alonso et al. 2018). Atrazine can increase yields by 6–50% depending on the crop (Rajendran et al. 2021). Commercial atrazine formulations are different and depend on each country: flowable, wettable powder, water-dispersible granules and soluble concentrate (Viegas et al. 2012). The main advantages of using atrazine are the versatility of its application and facility to mix it with other herbicides, such as S-metolachlor, alachlor, paraquat and linuron for broad-spectrum weed control (Rajendran et al. 2021).
Atrazine is used as a non-selective herbicide on both fallow and non-farmland land (He et al. 2019). It acts on the target weeds by blocking electron transport in photosystem II. This blocking occurs because atrazine inhibits the plastoquinone binding site (QB) of the D1 protein in chloroplasts and consequently suppressing the electron flow between photosystems (de Albuquerque et al. 2020). Therefore, electrons are not stored as chemical energy and chlorophyll molecules are heavily loaded with energy leading to lipid peroxidation in the membranes, inhibition of carbohydrate synthesis, decrease of the carbon (C) stock and accumulation of carbon dioxide (CO2) within plant cells and damage of leaf chlorophyll (Marchi et al. 2008). In the pre-emergence application, atrazine is first absorbed by the roots and then transported to the leaves, where its action produces chlorosis, necrosis and death. In post-emergence application, this herbicide is absorbed by the leaves (Souza et al. 2012; de Albuquerque et al. 2020). The action time of atrazine varies between 2 and 6 months, due to its stability in neutral and slightly alkaline or acid soil conditions (CASAFE, Cámara de Sanidad Agropecuaria y Fertilizantes 2013). The accumulation of atrazine in soil is prone to phytotoxicity to sensitive crops, such as soybeans, rice, oat and wheat (Chen et al. 2019).
The application of atrazine has been discussed due to its persistence and mobility in the environment, and consequently it is detected in the soil, surface water, groundwater, pastures, streams, lakes, sediments, foods and even glaciers in remote areas (Barchanska et al. 2012; Hansen et al. 2013; Sun et al. 2017; Pan et al. 2019; Wang et al. 2020). Consequently, the European Union (EU) banned its application in the year 2004. The USA and Canada have adopted restriction policies to minimise its potential environmental impact. However, it is still used extensively in agricultural practices in numerous countries, highlighting Argentina, China, Brazil and India (Sun et al. 2017; Montoya et al. 2019; de Albuquerque et al. 2020).
Massive application, mainly coinciding with rainy seasons, the high persistence and mobility are the main reasons for the atrazine detection in soil, surface water and groundwater at concentrations exceeding the limit established by normative values (Bachetti et al. 2021; Rajendran et al. 2021). The US Environmental Protection Agency (US EPA) has established the maximum atrazine concentration limit values in drinking water at 3 μg L–1 (US EPA 2007). However, the EU (EU 2004) and the World Health Organization (WHO 2011) have established the permissible limit to 0.1 and 2 μg L–1, respectively. The persistence of atrazine on environmental compartments poses a serious threat to human health. Furthermore, the US EPA has classified atrazine in toxicity class III and as an endocrine disruptor herbicide (Morales-Pérez et al. 2016; Singh and Jauhari 2017). The International Agency for Research on Cancer (IARC) has categorised atrazine in the list of carcinogenic herbicide (Mahler et al. 2017).
2.2 Behaviour and Impact of Atrazine on the Environment
After being applied, only 0.1% of the herbicide reaches the target organisms (weeds). The remainder can interact with the different environmental compartments through complexes biological, physical and chemical reactions. The environmental behaviour of atrazine depends upon several factors, including retention, transformation and transport processes, as well as by the interaction between them (Fig. 21.1) (Sun et al. 2019). These interactions are complex, being controlled simultaneously by biological, physical and chemical reactions. After being applied and before it reaches the soil, the herbicide may undergo photolysis, volatilisation and/or may be adsorbed or absorbed by the plant or by the stubble on the surface. Once in the soil, the xenobiotic is partitioned into solid, gas and liquid phases; in the latter, chemical and microbial degradation occurs, these processes being the most important for the dissipation of most herbicides. It is well known that the natural attenuation and fate of atrazine in soil environments are strongly related to adsorption, desorption and mineralisation processes (Liu et al. 2021).
The transformation of atrazine can be due to photochemical or biochemical processes, producing simpler molecules with different properties from that of the original compound (Mudhoo and Garg 2011). In fact, atrazine can be degraded in more than 15 metabolites, and each transformation product varies in its persistence (months to decades) and toxicity (Xue et al. 2021). The major atrazine degradation products are hydroxyatrazine (HA), deethylatrazine (DEA) and deisopropylatrazine (DIA). Due to their mobility, they have been frequently detected in many surface and groundwater of the world (Jablonowski et al. 2011; Nödler et al. 2013). Formation of HA occurs through different (biotic or abiotic) degradation mechanisms leading to the hydrolysis of atrazine molecule, and the microbial mediated process of atrazine N-dealkylation produces the mobiles metabolites DEA and DIA (Mudhoo and Garg 2011). The HA is the most important metabolite, with a higher retention in soils compared to other products (Chokejaroenrat et al. 2020).
A comprehensive understanding of the herbicide behaviour in the environmental compartments is extremely important to implement appropriate environmental management strategies to reduce its impact on human and animal health in the vulnerable areas.
2.3 Soil Contamination and Effects in Terrestrial Ecosystems
Soil is an important life-supporting system and plays a critical role for primary production, the regulation of biogenic gases, biogeochemical and hydrological cycles as well as the biodiversity preservation (Sun et al. 2017). However, soil pollution has become a worldwide concern because it acts as an important reservoir for numerous organic pollutants such as herbicides (Ali et al. 2019). In the soil, atrazine is distributed between aqueous and solid phases. Two different but coexisting processes are proposed for the atrazine movement in soils. One of them, is the rapid movement of the herbicide corresponding to a preferential flow through the soil macropores and the other one is a slow transport due to the sorption and degradation processes in the soil matrix (Mudhoo and Garg 2011).
The main processes that determine the persistence of atrazine in the soil environment are the physicochemical and microbiological properties (organic matter content, pH, texture, cation exchange capacity, microbial abundance and metabolic activity) of the soil, climatic characteristics (temperature, humidity, precipitation) and other parameters (mode and rate of application, prior history use, plant cover, topography) (Hernández et al. 2008; Prado et al. 2014). Atrazine breakdown in soil occurs mainly by chemical and microbial aerobic degradation (Viegas et al. 2012). Chemical degradation can occur by hydrolysis or by photodegradation. Atrazine is stable at room temperature, in the dark, at neutral pH and in the absence of microorganisms and organic matter (Prosen and Zupančič-Kralj 2005). This herbicide atrazine can be extremely persistent in soil environment due to its ability to bind to soil colloids (organic matter and clays) and become non-extractable residues (Martins et al. 2018). Recent studies have been dedicated to evaluate the behaviour and fate of atrazine in soils (Salazar-Ledesma et al. 2018; Sun et al. 2019; Liu et al. 2021), especially the adsorption and desorption processes between this herbicide and soil which influence its mobility and availability for weed control (Martins et al. 2018; Piratoba et al. 2021). Adsorption of atrazine seems to be positively correlated with organic matter and clay content and negatively with pH (Aparicio et al. 2015; Yue et al. 2017). It is reported that soil organic matter contains a variety of functional groups such as hydrophobic, hydrophilic and free radicals that can strongly entrap atrazine (Barriuso and Houot 1996). The pH is a factor affecting the adsorption of atrazine in soils, because when the pH increases, the soil surface tends to be negatively charged and organic molecules also tend to be ionised and negatively charged (Huang et al. 2013). This generates a repulsion of same-sign charges, which will be detrimental to the adsorption of organic molecules on the soil surface (Wang et al. 1999).
The accumulation of atrazine in soil (either dissolved or bound to colloids) is considered a long-term source of the compound leading to its possible occurrence to surface or groundwater. Numerous studies also reveal an accelerated degradation of atrazine due to the prolonged exposition of emergent native microbial populations capable to utilise the herbicide as a C (carbon) or N (nitrogen) source (Jablonowski et al. 2011; Sun et al. 2017). The exposure to some pesticides may change the resources that soil microorganisms use to obtain energy and nutrients, especially in soils with low levels of organic matter (Fernandes et al. 2020). Specific microorganisms are able to detoxify atrazine by N-dealkylation or dehalogenation reactions, and this may imply the development of microbial communities that can utilise the N in the triazine ring (Cycoń et al. 2017; Esquirol et al. 2020). Atrazine microbial degradation will be explained in Sect. 21.3 of this chapter. Soil macrofauna can also directly or indirectly affect the degradation and ultimately the fate of atrazine. In point of fact, the presence of earthworms in the soil may also affect the transport of atrazine. This is due to earthworms, which are keystone organisms that can ingest and transport the atrazine residues to deeper soil layers, enhancing the formation of non-extractable residues, thus reducing the leaching potential of this herbicide (Mudhoo and Garg 2011; Viegas et al. 2012).
Several studies have reported atrazine residues in soils and sediments from all over the world. Atrazine residues (19.5 g ha–1) and their degradate products were still found in agricultural soils a long time after the last herbicide application (more than 20 years), as well as in soils with no history of atrazine application (Jablonowski et al. 2010). Sun et al. (2017) showed that atrazine concentrations ranged from 1.0 to 113 ng g–1 dry weight, with a frequency of detection of 57.7% in soil samples of China (n = 241) and reported a close association between contamination and land use type. In Pakistan, Ali et al. (2019) evaluated the concentrations of 30 endocrine disrupting pesticides in soil and vegetable samples. Atrazine concentrations ranged from 1.7 to 120 μg kg–1 in soil samples, while no residues were detected in the vegetables studied. Alonso et al. (2018) showed atrazine residues (4–66 μg kg–1) in soil samples (n = 58) from the provinces of Córdoba and Buenos Aires (Argentina), an important corn producing area of Argentina. In addition, Mac Loughlin et al. (2017) detected atrazine in sediments of Carnaval creek (n = 10) (Buenos Aires, Argentina) at concentrations ranging from 5.1 to 32.7 μg kg–1, causing lethal and sublethal effects on benthic fauna.
The most susceptible group to the deposition of atrazine residues in the soil environment are non-target crops that may receive atrazine by spray drift, accidental spills or carryover. As a consequence of atrazine exposition, target and non-target plants often undergo oxidative stress because of an enhanced reactive oxygen species (ROS) production. Beker Akbulut and Yigit (2010) determined that ROS caused negative effects on peroxidase, ascorbate peroxidase and lipid peroxidation in Z. mays plants with postemergence atrazine application. Gao et al. (2011) reported that exposure to 10 μg L–1 atrazine significantly reduces plant fresh weight and total chlorophyll concentration. The authors also revealed a high plant mortality (up to 86.7%) at 100 μg L–1 concentration. Çanakci-Gülengül and Karabulut (2020) investigated the biochemical effects of atrazine concentrations (0, 200, 500 and 1000 μM) on wheat (Triticum aestivum L.) seedlings. The results indicated a decrease in reduced glutathione/oxidised glutathione (GSH:GSSG) ratio and catalase activity (CAT) in leaf and root and an increase in superoxide dismutase (SOD) activity. Gao et al. (2019) showed that seagrass Zostera marina L. exposed to high concentrations of atrazine (1, 3 and 10 μg L–1) significantly inhibited photosynthetic efficiency and reduced shoot sugar levels.
Atrazine also affected soil invertebrates, especially earthworm, mites, nematodes and collembolans species (Singh and Jauhari 2017). Lammertyn et al. (2021) exposed earthworms (Eisenia fetida) to different concentrations of atrazine to evaluate possible sublethal harmful effects. The results showed that atrazine (2 mg kg–1) affected the rate of cocoon production and increased lactate dehydrogenase and, especially, acetylcholinesterase activity. On the other hand, Dani et al. (2018) showed that earthworms exposed to sublethal concentrations of atrazine (362.4, 181.2, 90.6 and 45.3 ng cm–2) caused a general suppression in their metabolism, reduced ATP synthesis and had a negative impact on general health.
2.4 Water Contamination and Effects in Aquatic Ecosystems
Atrazine properties, such as low vapour pressures, moderate water solubility and low soil adsorption coefficient (KOC = 100 cm3 g–1), indicate its high leaching potential, particularly in well-structured soil profiles (de Albuquerque et al. 2020). Most atrazine movement occurs in the soil aqueous phase. Therefore, precipitation contributes to its dispersion in aquatic systems near the application zone (Jablonowski et al. 2011; Bachetti et al. 2021). Another, but less common, transport mechanism is adsorption of atrazine to eroded soil particles (Alonso et al. 2018). Atrazine is a frequently detected pesticides in groundwater and surface runoff from around the world (Table 21.1). Peng et al. (2018) identified the diversity and complexity of organic pollutants at 28 sampling sites in the Yangtze River Delta, finding an atrazine concentration of 1726 ng L–1. The authors concluded that this concentration exceeded the annual average environmental quality standards of Europe. In fact, the value was 9.4 times higher than the concentration reported by Battaglin et al. (2016) in seven US states for atrazine in surface water (183 ng L–1). These authors analysed a total of 86 water samples and atrazine was one of the most frequently detected herbicide (18% of the samples). In Europe, researchers collected 314 groundwater samples from the Júcar River European Union Pilot Basin (Spain), and the study showed that atrazine was frequently detected after terbuthylazine and bromacil (Menchen et al. 2017). Fingler et al. (2017) obtained samples from different surface and groundwater resources in Croatia, finding atrazine residues in all of them in concentrations of around 68 ng L–1. Almasi et al. (2020) founded atrazine concentrations from 0 to 2,175,800 ng L–1 in aquifers from Iran. In South America, atrazine is a common herbicide detected in various surface water and groundwater (De Gerónimo et al. 2014; Montagner et al. 2019; de Albuquerque et al. 2020). Particularly in Argentina, Montoya et al. (2019) reported its occurrence in 26% of groundwater samples (n = 95) with concentrations between 0.3 and 16.1 ng L–1. The values detected in this study were lower than those informed by Mas et al. (2020) in aquifers from Santiago del Estero (Argentina), where concentrations ranged from 1 to 7921 ng L–1. Recent studies performed in the Ctalamochita river basin (Córdoba, Argentina) showed the high ubiquity and persistence of the herbicide in surface water courses and at concentrations reaching 5000 ng L–1. The results revealed that atrazine residues in surface waters increased during the warm and rainy season as a consequence of atrazine application but also due to differences in textural and compositional characteristics of soil (Bachetti et al. 2021). Besides, atrazine has also been detected in rainwater from the Pampean plain of Argentina with detection frequency >80%, at concentrations from 500 to 67,280 ng L–1 (Alonso et al. 2018). The detection of atrazine in water resources, in concentrations above the maximum acceptable levels for drinking water, is of concern as it represents a direct risk to human health through drinking water consumption. To minimise the damage caused by this herbicide, it is necessary to apply processes that allow its removal from s-triazine-contaminated sites.
Aquatic ecosystems are complex environments as they contain a great diversity of organisms (algae, bacteria, fungi and protozoa) that play important roles in primary productivity, decomposition of organic compounds and nutrient cycling (Mauffret et al. 2017). Aquatic environments receive direct and indirect inputs of different compounds such as herbicides, causing qualitative and quantitative changes on microbial communities. These effects can impact on higher trophic levels and on processes that contribute to overall water quality (Verrhiest et al. 2002; Ensz et al. 2003). In this context, environmental impact on aquatic organisms associated with the application of atrazine has been widely reported (Bai et al. 2015; Baxter et al. 2016; Singh and Jauhari 2017). In addition, atrazine can be absorbed by algae and aquatic plants through cell walls, exerting toxic action mainly through inhibition of photosynthesis (DeLorenzo et al. 2001). For instance, Esperanza et al. (2017) evaluated the impact and action mode of the atrazine on the cellular senescence process of Chlamydomonas reinhardtii. The results indicated an increase in intracellular calcium levels, alterations in nuclear and cell morphology, as well as in the activity of biochemical and molecular markers, suggesting that short-term exposure to atrazine can promote death of microalgae, which are the basis of aquatic food webs. Zhao et al. (2018) showed that atrazine inhibited the growth of the microalga Selenastrum capricornutum. Sun et al. (2020a) observed acute toxicity of atrazine in the microalga Chlorella sp. because atrazine damaged the reaction centre of photosystem II. Religia et al. (2019) demonstrated that phytoplankton (Raphidocelis subcapitata) exposed to sublethal doses of atrazine affect the population dynamics of its predator, Daphnia magna, due to the production of non-viable broods. Simultaneously, atrazine toxicity can change water quality, due to increased concentrations of C, N and/or inorganic phosphorus (P), pH modifications and increased electrical conductivity (Viegas et al. 2012; de Albuquerque et al. 2020) and decreased dissolved oxygen (O2) concentration due to reduced primary productivity (C fixation) by photosynthetic organisms (DeLorenzo et al. 2001).
2.5 Effects on Higher Organisms
Toxicity effects of atrazine on other non-photosynthetic organisms (honeybees, birds and mammals) is lower in comparison to plants and algae. However, several works have demonstrated the effect of atrazine exposure on different higher organisms (Hirano et al. 2019; Soltanian 2016). Atrazine mainly affects the endocrine system (Mukherjee et al. 2019; Graceli et al. 2020), and it also causes oxidative stress due to the formation of reactive oxygen species, leading to reduced semen quality and infertility in fish, crustaceans and mammals (Gely-Pernot et al. 2015; Owolabi and Omotosho 2017; Stara et al. 2018). In aquatic species, the exposure to atrazine (<5 μg L–1) resulted in transgenerational reproductive dysregulation in Oryzias latipes (Cleary et al. 2019), disrupted immunity in Cyprinus carpio (Wang et al. 2019) and induced oxidative stress, reproductive dysfunction and neuroendocrine impairments in Danio rerio (Adeyemi et al. 2015). In addition, Hedayatirad et al. (2020) exposed Danio rerio to 0, 5 and 50 μg L–1 atrazine and observed that it increased cortisol level and decreased total immunoglobulin and lysozyme, affecting reproduction, thyroid function, stress reactivity and immunity of mature female zebrafish and subsequently their offspring. Abdulelah et al. (2020) demonstrated that exposure to atrazine (>10 ppb) causes significant DNA damage in crayfish lateral antennal cells, including olfactory sensory neurons, leading to impaired chemosensory abilities. Because crayfish rely on chemoreception for survival, changes in their ability to perceive odours following exposure to atrazine may have detrimental effects on their population size. Blahova et al. (2020) showed that common carp (Cyprinus carpio L.) chronically exposed to a range of atrazine concentrations (0.3; 300; 1000 and 3000 μg L–1) for 12 weeks negatively influenced many health status indicators, such as oxidative stress indices, immune system response, haematological and biochemical profile and organ histopathology.
In mammals, Komsky-Elbaz and Roth (2017) indicated that bovine spermatozoa exposed to atrazine (0.1–3 μg L–1) negatively affected sperm membranes, sperm viability, acrosome reaction and mitochondrial function. In females it produces imbalances in sex hormones and interferes with androgen or oestrogen receptors, altering instinctive abortion, ovarian cycles and defect in birth development (Bohn et al. 2011). It was suggested that the negative effects of atrazine on the neuroendocrine system are caused by altered hormone levels, mainly follicle-stimulating hormone (FSH) and luteinising hormone (LH) (Song et al. 2014). Altered LH levels contribute to prolonged prolactin secretion and subsequent stimulation of mammary gland changes and increased incidence of mammary fibroadenomas and adenocarcinomas (Jowa and Howd 2011; Simpkins et al. 2011). Foradori et al. (2009) exposed ovariectomised female rats to several atrazine doses (0 and 200 mg kg–1 day–1) for 4 days and reported that this herbicide reduced the number of activated gonadotropin-releasing hormone (GnRH) neurons. However, after 4 days of atrazine withdrawal, LH levels and GnRH activation markers returned to normal levels in treated animals. Although the negative effects could be reversed, it is unknown what may occur after longer exposure with the herbicide. Finally, Foradori et al. (2018) concluded that atrazine activates the hypothalamic-pituitary adrenal axis centrally and requires corticotropin-releasing hormone receptor activation. Atrazine also causes liver damage, the main metabolising organ of atrazine in mammals (Xing et al. 2015; Sagarkar et al. 2016). Cardiovascular system functioning is also affected by atrazine exposure (Cosselman et al. 2015). Besides, a possible association between atrazine contamination and a greater effect of several types of cancer in human cells, leukaemia and lymphoma has also been proposed (Thueson et al. 2015; Kirsten et al. 2017; Li et al. 2017; Brasil et al. 2018). There is limited epidemiological evidence on the adverse effects of prenatal atrazine exposure in humans. Consumption of drinking water with atrazine residues has been associated with an increased risk of preterm birth in Kentucky (Rinsky et al. 2012) and in four Midwestern states from the USA, where <10% of the population uses private well water (Stayner et al. 2017). In France, Chevrier et al. (2011) demonstrated that atrazine residues in maternal urine were associated with alterations in the babies, such as lower birth weight, head circumference and height. Xie et al. (2021) exposed human SH-SY5Y neuronal cells to 0.3, 3 and 30 μg L–1 atrazine, showing alterations in neurite outgrowth and SNCA pathology, which leads to epigenome changes and an increased risk of Parkinson’s disease. Previous epidemiological studies on the effect of atrazine on newborns are based on ecological estimates obtained from environmental monitoring data. Thus, a more reliable and accurate presence of atrazine is essential to ensure the safety of biota, human health and the environment.
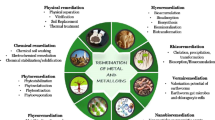
3 Bioremediation of Atrazine-Contaminated Environments
Bioremediation involves the utilisation of microorganisms, plants or their enzymes for the partial or complete transformation of organic pollutants present in environments, in order to protect the natural ecosystem and prevent further pollution (Viegas et al. 2019). This biotechnological tool has several advantages compared to the physicochemical treatments, such as lower operational costs, in situ application, efficient elimination and minimum disturbance of the treated site (Hernández et al. 2008). Due to this intensive method which must be adapted to site-specific conditions, small-scale pilot experiments are necessary before they can be carried out in the contaminated field. However, bioremediation sometimes has limitations, which will affect the efficiency of microbial degradation. Therefore, it is necessary to search microorganisms with better performance and environmental tolerance. Besides, the use of genetic technologies to improve the degradation properties of microorganisms is also receiving increasing attention (He et al. 2019).
Biostimulation and bioaugmentation are natural attenuation methods that allow in situ microbiological remediation of atrazine. Natural attenuation involves physical, chemical or biological processes in the environment to dissipate the contaminant, being a very slow process. Biostimulation involves treating the contaminated soils to increase the pollutant bioavailability or adding a co-substrate or nutritional compound to increase the population of indigenous (or introduced) bacteria that degrade contaminants (Tyagi et al. 2011). In this case, the elimination of atrazine depends on its initial concentration, the pH of the medium, the inoculation time and the type of stimulant (Rajendran et al. 2021). On the other hand, bioaugmentation involves the inoculation with microbial strains or consortia (indigenous or not) to improve the system’s biodegradation capacity of a specific organic pollutant in contaminated soils or water (Philp and Atlas 2005; Hernández et al. 2008). Furthermore, bioaugmentation may be required when indigenous degraders cannot degrade the pollutant rapidly or the degrading microorganisms are not present (Gentry et al. 2004). Both biostimulation and bioaugmentation are the most efficient methods of converting a pesticide into a less-harmful end product.
3.1 Bacterial Remediation
Bacteria are able to remove, degrade or breakdown xenobiotic compounds in less toxic or non-toxic ones by a process usually called bioremediation. This is a promising technology that includes different processes such as (a) pollutant transformation, (b) degradation to simple molecules, (c) mineralisation into inorganic compounds (such as СО2, Н2О, Н2, NH3, etc.), (d) cell surface sorption and (e) intracellular accumulation, among others (Krastanov et al. 2013). It is a profitable and increasingly popular technology to restore the environment quality (Lyon and Vogel 2013). In this process, microorganisms employ the contaminants as a source of nutrients or energy for their growth (Benimeli et al. 2008). However, at the time of its application, it is important to consider the susceptibility of the contaminant to microbiological transformation, the biological activity (bioactivity) to promote microbial growth and activity and the affordability of the contaminants to microorganisms (bioavailability) (Niti et al. 2013).
Numerous bacterial strains (Gram-positive and Gram-negative) have been described with the capability to use atrazine as a substrate (C and/or N source) for their growth (Udiković-Kolić et al. 2012). Among them, Pseudomonas sp. ADP has been the bacteria used for the study of the metabolic pathway of s-triazine degradation and its regulation (Cao et al. 2021). The two catabolic pathways of atrazine degradation, one upper and one lower, involve the atzABCDEF genes located in the pADP-1 plasmid (Martinez et al. 2001). The enzymes encoded by these genes catalyse six successive hydrolysis: one dechlorination, two dealkylations, biuret deamination, ring cleavage and an allophanate hydrolysis (Fig. 21.2). Thus, the upper catabolic pathway transforms atrazine to cyanuric acid, and the enzymes responsible for these transformations are coded by the atzA, atzB and atzC genes (Mandelbaum et al. 1995; De Souza et al. 1996, 1998; Sadowsky et al. 1998). An initial hydrolase different from atzA, but with identical function, called trzN, has also been described, mainly in Gram-positive bacteria, such as Arthrobacter aurescens TC1, Nocardioides sp. C190 and Nocardioides sp. SP12 (Topp et al. 2000a; Piutti et al. 2003; Sajjaphan et al. 2004; Smith et al. 2005). However, its presence has also been demonstrated in Gram-negative genera such as Sinorhizobium and Polaromonas (Devers et al. 2007). Thus, atrazine dechlorination results from the activity of atzA (atrazine chlorohydrolase) or trzN (triazine hydrolase), both aminohydrolases, which produce HA. It is known that atzA and trzN possess different substrate ranges: atzA hydrolyses s-triazine compounds whereas trzN hydrolyses radical groups (–OCH3, –SCH3, –CN, –F, –Cl) of both s-triazines and pyrimidines (De Souza et al. 1996; Seffernick et al. 2000, 2002; Strong et al. 2002; Shapir et al. 2006). Subsequently, HA is transformed into N-ethylammelide or N-isopropylammelide by hydrolysis of the N-ethyl or N-isopropyl side groups. Then, the transformation to N-isopropylammelide is encoded by atzB (hydroxyatrazine hydrolase) (Boundy-Mills et al. 1997) which is capable of catalysing the hydrolytic deamination of N-ethylammelide to cyanuric acid (Smith et al. 2005), an intermediate in the catabolism of s-triazines (Cook 1987). However, N-isopropylammelide is transformed to N-isopropylamine and cyanuric acid by the enzyme atzC (N-isopropylammelide hydrolase) (Sadowsky et al. 1998). These intermediates freed from the s-triazine ring by atzB and atzC can be used as sources of C, N and/or energy for the growth of the microorganism itself or others present in the environment (Strong et al. 2002; Kolić et al. 2007).
The lower pathway is carried out by three enzymes coded by the atzD, atzE and atzF genes (Cao et al. 2021) and leads to the final mineralisation of cyanuric acid to CO2 and NH3. It begins with ring cleavage in cyanuric acid catalysed by the enzyme cyanuric acid hydrolase (atzD) resulting in the production of the intermediate biuret carboxylate, which rapidly decomposes to biuret and CO2 (Seffernick et al. 2012). Alternatively, the trzD gene, with homologous function to the atzD gene, which encodes an enzyme involved in the cleavage of the s-triazine ring of cyanuric acid, has been identified in several s-triazine-degrading bacterial genera such as Pseudomonas, Paenarthrobacter, Arthrobacter, Aminobacter, Nocardioides, Klebsiella, Alcaligenes and Ralstonia (Cheng et al. 2005; Arbeli and Fuentes 2010; Yang et al. 2010; Fernández et al. 2013; Li et al. 2020). The hydrolysis of biuret to allophanate is mediated by biuret hydrolase (atzE) (Martinez et al. 2001; Cheng et al. 2005), and this deamination releases ammonium, which can be used by bacteria as a N source for growth. Finally, allophanate hydrolase (atzF) produces CO2 and NH4 from allophanate (Martinez et al. 2001; Cheng et al. 2005; Shapir et al. 2005, 2006). Comparisons among known degradative strains reveal substantial heterogeneity in the organisation and location of these catabolic genes in the genome. Thus, they may be located in (a) a single plasmid (Piutti et al. 2003; Aislabie et al. 2005; Devers et al. 2007), (b) several plasmids of varying size in the same host (Topp et al. 2000b; Rousseaux et al. 2002; Devers et al. 2007) and (c) occasionally in the microbial chromosome (Cai et al. 2003; Devers et al. 2007; Vaishampayan et al. 2007).
Bacterial strains that use partially degraded s-triazine as a N source were obtained from wastewater and soil from Switzerland and identified as Klebsiella pneumoniae strain 90 and 99 and Pseudomonas sp. (strains A, D and F) (Cook and Hütter 1981). Pseudomonas sp. YAYA6 was the first pure strain capable of mineralising atrazine and using it as a C source (Yanze-Kontchou and Gschwind 1994). A year later, Mandelbaum et al. (1995) identified from the US soils with a previous s-triazine herbicide application the bacterium Pseudomonas sp. ADP capable of growing with atrazine as C and N source, which was used for the characterisation of the enzymatic mechanism of atrazine mineralisation, as was previously described (De Souza et al. 1995, 1996; Mandelbaum et al. 1995; Boundy-Mills et al. 1997; Sadowsky et al. 1998; Martinez et al. 2001). Since then, numerous bacterial isolates capable of degrading s-triazines, either totally or partially, belonging to phylogenetically diverse groups have been isolated worldwide (Table 21.2). There are several parameters that influence the efficiency of the degradation process. The effect of N sources on the regulation of the atrazine catabolic pathway has been the subject of numerous studies (García-González et al. 2005). The external addition of N has shown a negative effect on atrazine biodegradation in most of the bacteria studied (García-González et al. 2003). High water salinity affected atrazine degradation in an industrial wastewater bacterial community (Udiković et al. 2003). Atrazine degradation efficiency both in the presence of O2 and in anaerobiosis showed no differences in both conditions, so that O2 would not influence the metabolism of this herbicide (Mandelbaum et al. 1995). Atrazine residues have a very ubiquitous distribution and can be found in different compartments of the environment, so these contaminated sites are the most appropriate for the isolation of tolerant microorganisms with the capacity to degrade them (Ortiz-Hernández et al. 2001). Although there is a great diversity of atrazine-degrading isolates obtained worldwide, most of them have been isolated from agricultural soils with a previous history of atrazine application (Fernández et al. 2013; Li et al. 2020; Cao et al. 2021) and only a few from effluents of agrochemical manufacturing industries (Li et al. 2008; Yang et al. 2010).
New investigations are being carried out to improve biodegradation of atrazine, including the use of bacterial consortia, immobilised cells on different natural or synthetic materials, among others, as will be described later.
3.2 Phytoremediation
The use of plant species for in situ treatment of contaminated sites is called phytoremediation. Pesticides can be transported across plant cell membranes and removed from the environment. Phytoremediation of pesticides involves several processes: pesticides in the soil can be absorbed by plant roots (rhizofiltration) or adsorbed by plant tissues (phytoextraction); pesticides in plant tissues can be transformed by plant enzymes (phytotransformation) or volatilised into the atmosphere (phytovolatilisation); and pesticides in the soil can be degraded by microorganisms in the rhizosphere (rhizoremediation) (Morillo and Villaverde 2017). Afterwards, incineration removes the compounds sequestered in the plant tissues (Gerhardt et al. 2009). Several studies have shown that root exudates of some plants significantly increase the desorption of organic pesticides in contaminated soils, increasing their bioavailability (Kidd et al. 2008; Muratova et al. 2009). Physical-chemical properties of the compounds and environmental and plant species characteristics are some of the causes that determine the rate of pesticide uptake (Singh and Jauhari 2017). Phytoremediation technology has many advantages: reduced costs compared to other remediation technologies; reduced erosion rate; improved physical, chemical and biological properties of the soil; aesthetic improvement; and environmentally friendly. However, it also presents some inconveniences such as longer restoration time of the contaminated site, extent and depth of the contaminated area, high dependence on climatic conditions, concentration and bioavailability of the contaminant and plant tolerance to contaminants (Morillo and Villaverde 2017; He et al. 2019). In addition, phytoremediation species can act as environmental filters at strategic water recharge points. For example, tree species capable of enhancing pesticide degradation in agricultural fields can be planted in alternating rows or as riparian forests to reduce or prevent their transport to rivers or groundwater (de Araújo et al. 2019). Inga striata and Caesalpinia ferrea are species that have shown their high tolerance to the atrazine herbicide (de Araújo et al. 2019; Aguiar et al. 2020).
Phytoremediation application is suitable in sites with low contamination and spread over large areas (Bini 2009). Several dicot and monocot species have been characterised as having high herbicide tolerance and some genotypes degrade herbicides in soil (Singh and Jauhari 2017). Herbicide removal is mainly attributed to the enzymes secreted by plant roots, such as peroxidases, polyphenol oxidases and invertases, followed by the uptake and transformation of the plants themselves (He et al. 2019). Merini et al. (2009) found that Lolium multiflorum had a higher atrazine removal capacity than natural attenuation in soil and water. Sui and Yang (2013) studied different rye grass genotypes, finding three genotypes capable of accumulating and degrading atrazine. Sánchez et al. (2017) investigated the phytoremediation of atrazine with Lolium perenne, Festuca arundinacea, Hordeum vulgare and Zea mays. The results showed that all plants had the ability to degrade atrazine, but Z. mays was the most efficient. Zhang et al. (2017) employed a genetically modified rice containing a metabolic enzyme glycosyltransferase 1 (ARGT1) capable of transforming atrazine. Cao et al. (2018) showed that the interaction between Pennisetum americanum and atrazine-contaminated soil influenced microbial communities and enhanced rhizosphere bacterial diversity by reshaping some soil physicochemical properties (urease activity, catalase activity, water-soluble organic carbon content and pH). In addition, some specific bacteria that could facilitate the degradation of organic pollutants or soil nutrient cycling were only identified in the rhizosphere of P. americanum. Aguiar et al. (2020) evaluated the remediation potential of Inga striata and Eremanthus crotonoides in atrazine-contaminated soils. They found that atrazine modified the physiological variables of these plants (photosynthetic rate, CO2 consumption and transpiration) but without compromising their development. Eremanthus crotonoides and I. striata were able to reduce atrazine residues even in soils with high concentrations, allowing their use in polluted sites. More recently, the electrokinetic-assisted phytoremediation (EKPR) is also used in the atrazine removal for improving the effect of phytoremediation, in soil mesocosms using ryegrass (Lolium perenne L.) (Sánchez et al. 2020).
3.3 Plant Microbial Remediation
Plant microbial remediation is a technology that uses plants and microorganisms to remove pollutants. Synergistic treatment between soil microorganisms and plant roots can promote the degradation of persistent organic pollutants in contaminated sites (Zhang et al. 2014; Asemoloye et al. 2017). Rhizodeposition and root exudation provide a source of nutrients for microorganisms present in the soil. Evidence suggests that organic acids, sugars, amino acids, tannins, phenolic compounds and vitamins found in root exudates have an important role in root-microbe communication (Tanimoto 2005). In addition, P solubilisation and N fixation occur in the rhizosphere, so bacterial populations benefit from increased availability of P and N (Shimp et al. 1993). Therefore, plants benefit from rhizosphere microorganisms through their metabolic detoxification of contaminants that can affect growth and these microorganisms, in turn, benefit from root exudates (Asemoloye et al. 2019). The detoxification mechanism may include three aspects: plants absorb organic pollutants to metabolise or accumulate them in their tissues, enzymes produced by plants favour the elimination of pollutants and microorganisms favour mineralisation.
A wide variety of plant species have demonstrated efficient degradation of organic pollutants present in the rhizosphere (Abhilash et al. 2012). Dong et al. (2016) combined Canna indica with Funnelliformis mosseae (arbuscular mycorrhizal fungi) to remove atrazine. They found that F. mosseae could reduce the inhibition of atrazine on photosynthesis and growth of Canna indica, while the combination of C. indica with F. mosseae increased the degradation rate to 95.7% compared with phytoremediation alone (68.1%). Bazhanov et al. (2017) used Arthrobacter ureafaciens DnL1-1 in combination with alfalfa and wheat to degrade atrazine and the results showed that DnL1-1 strain could help crops from the negative effect of the herbicide. The degradation rates of atrazine by the DnL1-1-alfalfa and DnL1-1-wheat combinations were 75.6% and 99.8%, respectively. James et al. (2018) isolated Pseudomonas strains from the roots of Typha latifolia, Acorus calamus and Phragmites karka and employed them in combination to remove atrazine. The results showed that A. calamus and Pseudomonas strains presented the highest degradation rate of atrazine, and the combination of plants-microbes could enhance the herbicide removal as compared with the use of single microorganisms or plants. Qu et al. (2018) evaluated the atrazine degradation and its detoxification by Myriophyllum spicatum in combination with the bacterial community present in the lake sediments. The results of the study indicated that M. spicatum and possibly the predominant sediment bacteria (Nitrospirae and Acidobacteria) degraded atrazine to biuret over a 60-day incubation period. Jiang et al. (2020) investigated the effects of Pseudomonas chlororaphis strain PAS18, a type of plant growth-promoting bacterium (PGPB), on the growth and physiological responses of Pennisetum americanum (L.) K. Schum seedlings under three different levels of atrazine (0, 20 and 100 mg kg–1) in a pot experiment. The results suggest that strain PAS18 could alleviate atrazine-induced growth and stress in P. americanum by enhancing photosystem II repair and antioxidant defence capacity, as well as balancing Ca2+ influx. Yang et al. (2021) showed the influence of co-inoculation of Trichoderma harzianum LTR-2 and A. ureafaciens DnL1-1 on wheat treated with atrazine. Strains LTR-2 and DnL1-1 caused significant increases in shoot biomass, root biomass and root/shoot ratio and significantly decreased the amount of atrazine and its degradation products.
Plant microbial remediation presents a wide range of application and research prospects, due to the low cost of application, low energy consumption and possible large-scale application to remediate contaminated environments (He et al. 2019).
3.4 Material-Microbial Remediation
Microorganisms used in bioremediation processes constitute an attractive methodology for the recovery of contaminated environments. Some of the strategies used to achieve that goal include on-site introduction of nutrients and O2, which stimulate native microbial strains (biostimulation) or on-site inoculation with competent microorganisms (bioaugmentation) (Nzila et al. 2016). However, an important factor to be considered in a bioaugmentation process is using the correct inoculation system to ensure the successful adaptation of the inoculated microorganisms. Immobilisation on biological or polymeric supports can be an appropriate inoculation strategy during bioaugmentation. The advantages of microbial immobilisation are (a) microbial protection from adverse environmental conditions or possible predators (protozoa, parasites, etc.) that threaten microbial survival, (b) nutritional stress prevention by allowing the exchange of nutrients and waste products, (c) the introduction of a higher density of degrading bacteria to the environment, (d) a major stability of microorganisms in sites with high concentrations of contaminants, (e) cell viability preservation in the long term by offering the possibility of repeated inoculation of cells, (f) allows microbial metabolism to remain relatively constant over time and (g) the easy recovery of the already decontaminated solution (Hsieh et al. 2008; Khondee et al. 2012; Angelim et al. 2013). In addition, immobilised cells are easier to handle, thus minimising the risks of contamination during transport, application and storage (Park and Chang 2000).
There is a wide range of substrates used as cell immobilisation matrices, which usually show an appropriate ability to be applied for removing organic pollutants. Synthetic polymers include polyacrylamide, polyethylene glycol, polyvinyl alcohol and polyurethane (Wang et al. 2012; Tong et al. 2014; Zhang et al. 2020) whereas natural substrates include alginate, carrageenan, agar, collagen, chitin, chitosan and biochar (Morgante et al. 2010; Banerjee and Ghoshal 2011; Lu et al. 2012; Sun et al. 2020b; Yu et al. 2020). Desitti et al. (2017) encapsulated Pseudomonas sp. strain ADP in core-shell electrospun microtubes and then used it for atrazine removal in a reactor. Besides, the sol-gel process was employed to immobilise ADP strains in thin silica layers, which were coated onto carrier materials (Pannier et al. 2014). Currently, there is an increasing focus of interest to find low-cost, more efficient and easier-to-handle support matrices. Khatoon and Rai (2018) investigated the potential of sugarcane bagasse as an immobilising support for Bacillus badius strain ABP6 for atrazine biodegradation. The results showed that the cells were strongly adsorbed and completely dispersed on the bagasse surface after immobilisation, removing 85.3% of atrazine at 14 days of assay.
Biochar produced from biological sources is an important source of C and with high stability to chemical and microbial degradation (Kupryianchyk et al. 2016). The biochar has been studied for remediation of pesticide-contaminated sites, due to its multiple advantages including the possibility of trapping bioremediation bacteria in the biochar micropores (Morillo and Villaverde 2017). Yu et al. (2019) evaluated the immobilisation of Arthrobacter sp. strain ZXY-2 on mushroom pellet biochar (Aspergillus niger Y3). The self-immobilised biomixture was capable to remove 50 mg L–1 of atrazine in 1 h. Tao et al. (2020) evaluated the immobilisation of four phosphate-solubilising bacteria and one atrazine-degrading bacterium (Acinetobacter lwoffii DNS32) on BC550 straw-based biochar. The results indicated that the combined immobilisation showed 49% higher capacity to degrade 100 mg L–1 atrazine in 24 h and 27% higher capacity to degrade 20 mg kg–1 atrazine after 3 days in liquid and soil, respectively.
4 Strategies for Bioremediation of Atrazine Under Laboratory and Field Conditions
As was previously highlighted, atrazine is a persistent herbicide frequently found in agricultural areas, surface water as well as groundwater (by infiltration through soils) (Alonso et al. 2018; Bachetti et al. 2021), and different alternatives have been proposed for its remediation. Among them, microbial degradation is one of the most efficient strategies from an economical and environmental point of view. However, increasing the efficiency of biodegradation processes is a challenge that researchers have to solve. Selection of suitable microorganisms, the increase of pollutant uptake and degradation, the immobilisation of microorganisms in adequate matrices and/or performing an appropriate bio-formulation are some of the approaches used to improve tolerance and atrazine removal (Desitti et al. 2017; Chen et al. 2019; Zhu et al. 2019; Herrera-Gallardo et al. 2021). More recently, computational models have been used to make simulations in order to design proper media not only for growth optimisation but also for enhancing atrazine biodegradation (Ofaim et al. 2020). Other strategies include optimisation of certain agronomic factors; the management of soils and microbial communities, including rhizospheric bacteria, fungi and endophytic microorganisms; and their selection and improvement through different techniques to generate a beneficial effect on plants or modify atrazine bioavailability (Liu et al. 2021; James and Singh 2021). For instance, it was reported that an epiphytic root bacteria Pseudomonas spp. strains AACB and TTLB and Arthrobacter spp. strain PPKB isolated from emergent hydrophytes could decontaminate atrazine at different pH and temperatures. Due to these strains also exhibiting PGP properties, they could be successfully applied as bioinoculants, for the phyto/rhizoremediation of terrestrial and aquatic ecosystems (James and Singh 2021). In the same way, intracellular crude enzyme extracts from these Pseudomonas strains were applied for atrazine detoxification and could be proposed as an alternative remediation technique (James and Singh 2021).
Despite several bacterial strains being able to display a great catabolic potential under laboratory conditions, they could fail to behave similarly in natural surroundings. This could be due to suboptimal growth environmental conditions, such as variations in pH, temperature and nutrient sources; the competition with well-adapted indigenous organisms and, also, the possible presence of environmental traits that might repress the genes responsible for the catabolic activities. These aspects are often not taken into account and are causes of failure of strains in the fields because to date, a high number of available results of atrazine decontamination by different microorganisms have been carried out under controlled laboratory conditions. On the other hand, it is interesting to consider that the experimental design should not only include synthetic solutions but the results must also be validated using more complex matrices such as soil, sediments, surface waters, etc. since many times certain microorganisms can degrade atrazine with high efficiency in a simple matrix but not in more real environments. In this sense, although it is widely recognised that Pseudomonas sp. ADP is able to degrade atrazine with high efficiency in synthetic solutions, the same did not occur in bioaugmentation experiments carried out in a liquid phase of sediment slurries, as well as in water circulating in columns filled with sediments. Besides, concomitant biostimulation with Na-citrate did not affect atrazine degradation in these experimental conditions (Liu et al. 2020). This and many other examples have been described in the literature showing diverse experimental devices for bioremediation studies. Such preliminary tests are fundamental to address in an appropriate way this highly complex subject. However, assays at microcosm and mesocosm levels are also needed. These systems constitute experimental devices that are interesting to make an approximation to reality, since they allow evaluating bioavailability and the effects of temperature, radiation, etc., which may have a decisive role on the observed responses. In addition, they allow to study the optimal conditions for implementing a biotechnological process. Microcosms can also be used to analyse certain specific target genes, related with degradation activities, and these studies could serve to predict the results of certain bioremediation strategies (Sagarkar et al. 2013). The literature provides many examples of studies related to bioremediation of atrazine using microcosms and mesocosms as experimental systems that were performed in the last years (Liu et al. 2021; Sagarkar et al. 2014, 2013; Urseler et al. 2021). For instance, bioaugmentation with Arthrobacter sp. strain AAC22 improved atrazine removal avoiding its lixiviation, being almost complete (>99%) after 8 days of treatment in a microcosm system using an agricultural soil. A bioassay indicated that toxic by-products were not detected after this treatment, demonstrating that AAC22 could be an efficient biotechnological tool for remediating atrazine-polluted soils (Urseler et al. 2021). Other research works suggest the existence of complex regulatory pathways for atrazine degradation in agricultural soils, which may be affected by the presence of N (Govantes et al. 2009).
Simulating field conditions, several bioremediation strategies have been proved at mesocosm level, in order to scale-up the process. For instance, a bacterial consortium with atrazine-degrading capabilities was used and this process was monitored at biochemical and genetic level (Sagarkar et al. 2014). Nonetheless, it is very important to follow a gradient of work scales and finally to confirm the behaviour of these systems on a larger scale, for example, in contaminated sites, since atrazine bioavailability as well as climatic traits could generate greater discrepancies between laboratory and field conditions. As it can be easily deduced, one of the main challenges is still the utilisation of these technologies to larger extensions, in the field and/or in the aquifers. In this sense, an analysis of the actual situation shows that there is some experience at the level of basic studies but its implementation in the field is very limited. Thus, future studies should be focused on reducing the gaps between atrazine bioremediation at laboratory and at field scales to find a proper and adaptable strategy to remediate extensive polluted sites.
5 Conclusion and Perspectives
Pollution caused by herbicides, such as atrazine, is considered among the top ten environmental hazards, which require the contribution of several disciplines to find strategies that allow mitigating its effects on ecosystems and also on human/animal health. As it was highlighted in this chapter, bioremediation is a cost-effective, efficient and clean strategy as against other common detoxifying methods, which has to be continuously improved. In the last years, several microorganisms have demonstrated to be efficient for atrazine removal. However, microbial consortia frequently showed more advantages because they can better withstand different environmental conditions. Despite an extensive knowledge now available in this regard, the continuous selection of more suitable and effective microorganisms is still an area of great interest.
Another key aspect is the understanding of the complex biodegradation processes of atrazine in natural environments. In this context, the use of new computational platforms together with the integration of different omic approaches (genomics, metagenomics, proteomics, metabolomics, etc.) is likely to strongly contribute to this aim and will promote the progress and the improvement of novel bioremediation strategies to enhance atrazine remediation applicability. Besides, the study of microbial communities by molecular tools could provide new knowledge related on catabolic potential and diversity of microorganisms. Thus, this information could improve treatments against pollution in addition to encouraging a better comprehension of these complex biological communities and opening new ways for biotechnological advances. Surely, some of these powerful approaches could help for the development of eco-friendly and efficient strategies for atrazine biodegradation.
On the other hand, a well-formulated and designed strategy is needed in order to implement an efficient bioremediation technology, by taking into consideration several aspects that influence the process. In this sense, the search and selection of new and low-cost matrices and nanomaterials would be useful to make this process more economical and environmentally viable. In fact, the development of new biomaterials, which possess good performance, is a new and promising research line in the area of pollutant remediation. However, they sometimes have certain disadvantages related to the entrapment material, the preparation of the biomaterial and the toxicity of certain components, among others. These and other aspects have to be solved previous to their large-scale application and constitute the aims of future research lines related to the use of such new technologies.
It is expected that atrazine bioremediation at different environments can be improved in the coming years not only by integrating recent findings but also by combining various strategies simultaneously in a safe way, by means of a synergistic action. This is one of the approaches that require more investigation in the future to test the efficiency. It should also deepen all those aspects related to the successful application of these technologies on a larger scale and with low environmental impact, which constitutes a challenge for the scientific community.
References
Abdulelah SA, Crile KG, Almouseli A, Awali S, Tutwiler AY, Tien EA, Manzo VJ, Hadeed MN, Belanger RM (2020) Environmentally relevant atrazine exposures cause DNA damage in cells of the lateral antennules of crayfish (Faxonius virilis). Chemosphere 239:124786. https://doi.org/10.1016/j.chemosphere.2019.124786
Abhilash PC, Powell JR, Singh HB, Singh BK (2012) Plant-microbe interactions: novel applications for exploitation in multipurpose remediation technologies. Trends Biotechnol 30:416–420. https://doi.org/10.1016/j.tibtech.2012.04.004
Acayaba RD, de Albuquerque AF, Ribessi RL, de Umbuzeiro GA, Montagner CC (2021) Occurrence of pesticides in waters from the largest sugar cane plantation region in the world. Environ Sci Pollut Res 28:9824–9835. https://doi.org/10.1007/s11356-020-11428-1
Adeyemi JA, Da Cunha MA, Barbosa F (2015) Teratogenicity, genotoxicity and oxidative stress in zebrafish embryos (Danio rerio) co-exposed to arsenic and atrazine. Comp Biochem Physiol Part C Toxicol Pharmacol 172–173:7–12. https://doi.org/10.1016/j.cbpc.2015.04.001
Aguiar LM, dos Santos JB, Barroso GM, Ferreira EA, Cabral CM, Costa MR, Vieira ERD, Zanuncio JC (2020) Phytoremediation by Eremanthus crotonoides and Inga striata decay atrazine and clomazone residues in the soil. Int J Phytoremediation 22:827–833. https://doi.org/10.1080/15226514.2019.1710818
Aislabie J, Bej AK, Ryburn J, Lloyd N, Wilkins A (2005) Characterization of Arthrobacter nicotinovorans HIM, an atrazine-degrading bacterium, from agricultural soil New Zealand. FEMS Microbiol Ecol 52:279–286. https://doi.org/10.1016/j.femsec.2004.11.012
Ali N, Khan S, Khan MA, Waqas M, Yao H (2019) Endocrine disrupting pesticides in soil and their health risk through ingestion of vegetables grown in Pakistan. Environ Sci Pollut Res 26:8808–8820. https://doi.org/10.1007/s11356-019-04287-y
Almasi H, Takdastan A, Jaafarzadeh N, Babaei AA, Tahmasebi Birgani Y, Cheraghian B, Saki A, Jorfi S (2020) Spatial distribution, ecological and health risk assessment and source identification of atrazine in Shadegan international wetland, Iran. Mar Pollut Bull 160:111569. https://doi.org/10.1016/j.marpolbul.2020.111569
Almberg KS, Turyk ME, Jones RM, Rankin K, Freels S, Stayner LT (2018) Atrazine contamination of drinking water and adverse birth outcomes in community water systems with elevated atrazine in Ohio, 2006–2008. Int J Environ Res Public Health 15:12–15. https://doi.org/10.3390/ijerph15091889
Alonso LL, Demetrio PM, Agustina Etchegoyen M, Marino DJ (2018) Glyphosate and atrazine in rainfall and soils in agroproductive areas of the pampas region in Argentina. Sci Total Environ 645:89–96. https://doi.org/10.1016/j.scitotenv.2018.07.134
Angelim AL, Costa SP, Farias BCS, Aquino LF, Melo VMM (2013) An innovative bioremediation strategy using a bacterial consortium entrapped in chitosan beads. J Environ Manag 127:10–17. https://doi.org/10.1016/j.jenvman.2013.04.014
Aparicio V, De Gerónimo E, Guijarro KH, Perez D, Portocarrero R, Vidal C (2015) Los plaguicidas agregados al suelo y su destino en el ambiente. INTA Ediciones, Buenos Aires, pp 1–74
Arbeli Z, Fuentes C (2010) Prevalence of the gene trzN and biogeographic patterns among atrazine-degrading bacteria isolated from 13 Colombian agricultural soils. FEMS Microbiol Ecol 73:611–623. https://doi.org/10.1111/j.1574-6941.2010.00905.x
Asemoloye MD, Jonathan SG, Jayeola AA, Ahmad R (2017) Mediational influence of spent mushroom compost on phytoremediation of black-oil hydrocarbon polluted soil and response of Megathyrsus maximus Jacq. J Environ Manag 200:253–262. https://doi.org/10.1016/j.jenvman.2017.05.090
Asemoloye MD, Jonathan SG, Ahmad R (2019) Synergistic plant-microbes interactions in the rhizosphere: a potential headway for the remediation of hydrocarbon polluted soils. Int J Phytoremediation 21:71–83. https://doi.org/10.1080/15226514.2018.1474437
Bachetti RA, Urseler N, Morgante V, Damilano G, Porporatto C, Agostini E, Morgante C (2021) Monitoring of atrazine pollution and its spatial-seasonal variation on surface water sources of an agricultural river basin. Bull Environ Contam Toxicol 106:929–935. https://doi.org/10.1007/s00128-021-03264-x
Bai X, Sun C, Xie J, Song H, Zhu Q, Su Y, Qian H, Fu Z (2015) Effects of atrazine on photosynthesis and defense response and the underlying mechanisms in Phaeodactylum tricornutum. Environ Sci Pollut Res 22:17499–17507. https://doi.org/10.1007/s11356-015-4923-7
Banerjee A, Ghoshal AK (2011) Phenol degradation performance by isolated Bacillus cereus immobilized in alginate. Int Biodeterior Biodegrad 65:1052–1060. https://doi.org/10.1016/j.ibiod.2011.04.011
Barchanska H, Rusek M, Szatkowska A (2012) New procedures for simultaneous determination of mesotrione and atrazine in water and soil. Comparison of the degradation processes of mesotrione and atrazine. Environ Monit Assess 184:321–334. https://doi.org/10.1007/s10661-011-1970-5
Barriuso E, Houot S (1996) Rapid mineralization of the s-triazine ring of atrazine in soils in relation to soil management. Soil Biol Biochem 28:1341–1348. https://doi.org/10.1016/S0038-0717(96)00144-7
Battaglin WA, Smalling KL, Anderson C, Calhoun D, Chestnut T, Muths E (2016) Potential interactions among disease, pesticides, water quality and adjacent land cover in amphibian habitats in the United States. Sci Total Environ 566–567:320–332. https://doi.org/10.1016/j.scitotenv.2016.05.062
Battaglin WA, Bradley PM, Iwanowicz L, Journey CA, Walsh HL, Blazer VS (2018) Pharmaceuticals, hormones, pesticides, and other bioactive contaminants in water, sediment, and tissue from Rocky Mountain National Park, 2012–2013. Sci Total Environ 643:651–673. https://doi.org/10.1016/j.scitotenv.2018.06.150
Baxter L, Brain RA, Lissemore L, Solomon KR, Hanson ML, Prosser RS (2016) Influence of light, nutrients, and temperature on the toxicity of atrazine to the algal species Raphidocelis subcapitata: implications for the risk assessment of herbicides. Ecotoxicol Environ Saf 132:250–259. https://doi.org/10.1016/j.ecoenv.2016.06.022
Bazhanov DP, Yang K, Li H, Li C, Li J, Chen X, Yang H (2017) Colonization of plant roots and enhanced atrazine degradation by a strain of Arthrobacter ureafaciens. Appl Microbiol Biotechnol 101:6809–6820. https://doi.org/10.1007/s00253-017-8405-3
Beker Akbulut G, Yigit E (2010) The changes in some biochemical parameters in Zea mays cv. “Martha F1” treated with atrazine. Ecotoxicol Environ Saf 73:1429–1432. https://doi.org/10.1016/j.ecoenv.2010.05.023
Benimeli CS, Fuentes MS, Abate CM, Amoroso MJ (2008) Bioremediation of lindane-contaminated soil by Streptomyces sp. M7 and its effects on Zea mays growth. Int Biodeterior Biodegrad 61:233–239. https://doi.org/10.1016/j.ibiod.2007.09.001
Bini C (2009) From soil contamination to land restoration. In: Steimberg RV (ed) Contaminated soils: environmental impact, disposal and treatment. Nova Science Publishers, Inc., New York, pp 97–137
Blahova J, Dobsikova R, Enevova V, Modra H, Plhalova L, Hostovsky M, Marsalek P, Mares J, Skoric M, Vecerek V, Svobodova Z (2020) Comprehensive fitness evaluation of common carp (Cyprinus carpio L.) after twelve weeks of atrazine exposure. Sci Total Environ 718:135059. https://doi.org/10.1016/j.scitotenv.2019.135059
Bohn T, Cocco E, Gourdol L, Guignard C, Hoffmann L (2011) Determination of atrazine and degradation products in luxembourgish drinking water: Origin and fate of potential endocrine-disrupting pesticides. Food Addit Contam. https://doi.org/10.1080/19440049.2011.580012
Boundy-Mills KL, De Souza ML, Mandelbaum RT, Wackett LP, Sadowsky MJ (1997) The atzB gene of Pseudomonas sp. strain ADP encodes the second enzyme of a novel atrazine degradation pathway. Appl Environ Microbiol 63:916–923. https://doi.org/10.1128/aem.63.3.916-923.1997
Bouquard C, Ouazzani J, Prome J, Michel-Briand Y, Plesiat P (1997) Dechlorination of atrazine by a Rhizobium sp. isolate. Appl Environ Microbiol 63:862–866. https://doi.org/10.1128/aem.63.3.862-866.1997
Brasil VLM, Ramos Pinto MB, Bonan RF, Kowalski LP, da Cruz Perez DE (2018) Pesticides as risk factors for head and neck cancer: a review. J Oral Pathol Med 47:641–651. https://doi.org/10.1111/jop.12701
Cai B, Han Y, Liu B, Ren Y, Jiang S (2003) Isolation and characterization of an atrazine-degrading bacterium from industrial wastewater in China. Lett Appl Microbiol 36:272–276. https://doi.org/10.1046/j.1472-765X.2003.01307.x
Caldas SS, Arias JLO, Rombaldi C, Mello LL, Cerqueira MBR, Martins AF, Primel EG (2019) Occurrence of pesticides and PPCPs in surface and drinking water in southern Brazil: Data on 4-year monitoring. J Braz Chem Soc 30:71–80. https://doi.org/10.21577/0103-5053.20180154
Çanakci-Gülengül S, Karabulut F (2020) The biochemical changes caused by metolachlor and atrazine on wheat (Triticum aestivum L.) varieties. Prog Nutr 22:8336. https://doi.org/10.23751/pn.v22i3.8336
Cao B, Zhang Y, Wang Z, Li M, Yang F, Jiang D, Jiang Z (2018) Insight into the variation of bacterial structure in atrazine-contaminated soil regulating by potential phytoremediator: Pennisetum americanum (L.). Front Microbiol 9:864. https://doi.org/10.3389/fmicb.2018.00864
Cao D, He S, Li X, Shi L, Wang F, Yu S, Xu S, Ju C, Fang H, Yu Y (2021) Characterization, genome functional analysis, and detoxification of atrazine by Arthrobacter sp. C2. Chemosphere 264:128514. https://doi.org/10.1016/j.chemosphere.2020.128514
CASAFE, Cámara de Sanidad Agropecuaria y Fertilizantes (2013) Agrochemicals market report. Argentina. http://www.casafe.org/publicaciones/estadisticas/. Accessed June 2021
Chen Y, Jiang Z, Wu D, Wang H, Li J, Bi M, Zhang Y (2019) Development of a novel bio-organic fertilizer for the removal of atrazine in soil. J Environ Manag 233:553–560. https://doi.org/10.1016/j.jenvman.2018.12.086
Cheng G, Shapir N, Sadowsky MJ, Wackett LP (2005) Allophanate hydrolase, not urease, functions in bacterial cyanuric acid metabolism. Appl Environ Microbiol 71:4437–4445. https://doi.org/10.1128/AEM.71.8.4437-4445.2005
Chevrier C, Limon G, Monfort C, Rouget F, Garlantézec R, Petit C, Durand G, Cordier S (2011) Urinary biomarkers of prenatal atrazine exposure and adverse birth outcomes in the pelagie birth cohort. Environ Health Perspect 119:1034–1041. https://doi.org/10.1289/ehp.1002775
Chokejaroenrat C, Watcharenwong A, Sakulthaew C, Rittirat A (2020) Immobilization of atrazine using oxidized lignite amendments in agricultural soils. Water Air Soil Pollut 231:1–15. https://doi.org/10.1007/s11270-020-04608-9
Cleary JA, Tillitt DE, vom Saal FS, Nicks DK, Claunch RA, Bhandari RK (2019) Atrazine induced transgenerational reproductive effects in medaka (Oryzias latipes). Environ Pollut 251:639–650. https://doi.org/10.1016/j.envpol.2019.05.013
Climent MJ, Herrero-Hernández E, Sánchez-Martín MJ, Rodríguez-Cruz MS, Pedreros P, Urrutia R (2019) Residues of pesticides and some metabolites in dissolved and particulate phase in surface stream water of Cachapoal River basin, central Chile. Environ Pollut 251:90–101. https://doi.org/10.1016/j.envpol.2019.04.117
Cook A (1987) Biodegradation of s-triazine xenobiotics. FEMS Microbiol Lett 46:93–116. https://doi.org/10.1016/0378-1097(87)90059-0
Cook AM, Hütter R (1981) s-Triazines as nitrogen sources for bacteria. J Agric Food Chem 29:1135–1143. https://doi.org/10.1021/jf00108a009
Cosselman KE, Navas-Acien A, Kaufman JD (2015) Environmental factors in cardiovascular disease. Nat Rev Cardiol 12:627–642. https://doi.org/10.1038/nrcardio.2015.152
Cycoń M, Mrozik A, Piotrowska-Seget Z (2017) Bioaugmentation as a strategy for the remediation of pesticide-polluted soil: a review. Chemosphere 172:52–71. https://doi.org/10.1016/j.chemosphere.2016.12.129
Dani VD, Simpson AJ, Simpson MJ (2018) Analysis of earthworm sublethal toxic responses to atrazine exposure using 1H nuclear magnetic resonance (NMR)-based metabolomics. Environ Toxicol Chem 37:473–480. https://doi.org/10.1002/etc.3978
de Albuquerque FP, de Oliveira JL, Moschini-Carlos V, Fraceto LF (2020) An overview of the potential impacts of atrazine in aquatic environments: perspectives for tailored solutions based on nanotechnology. Sci Total Environ 700:134868. https://doi.org/10.1016/j.scitotenv.2019.134868
de Araújo FR, dos Santos JB, Ferreira EA, Cabral CM, Laia M, Silva DV, de Freitas SM (2019) Selection of arboreal species to compose and remedy riparian forests next to agricultural areas. Ecol Eng 131:9–15. https://doi.org/10.1016/j.ecoleng.2019.02.023
De Gerónimo E, Aparicio VC, Bárbaro S, Portocarrero R, Jaime S, Costa JL (2014) Presence of pesticides in surface water from four sub-basins in Argentina. Chemosphere 107:423–431. https://doi.org/10.1016/j.chemosphere.2014.01.039
De Souza ML, Wackett LP, Boundy-Mills KL, Mandelbaum RT, Sadowsky MJ (1995) Cloning, characterization, and expression of a gene region from Pseudomonas sp. strain ADP involved in the dechlorination of atrazine. Appl Environ Microbiol 61:3373–3378. https://doi.org/10.1128/aem.61.9.3373-3378.1995
De Souza ML, Sadowsky MJ, Wackett LP (1996) Atrazine chlorohydrolase from Pseudomonas sp. strain ADP: Gene sequence, enzyme purification, and protein characterization. J Bacteriol 178:4894–4900. https://doi.org/10.1128/jb.178.16.4894-4900.1996
De Souza ML, Seffernick J, Martinez B, Sadowsky MJ, Wackett LP (1998) The atrazine catabolism genes atzABC are widespread and highly conserved. J Bacteriol 180:1951–1954. https://doi.org/10.1128/jb.180.7.1951-1954.1998
DeLorenzo ME, Scott GI, Ross PE (2001) Toxicity of pesticides to aquatic microorganisms: a review. Environ Toxicol Chem 20:84–98. https://doi.org/10.1002/etc.5620200108
Desitti C, Beliavski M, Tarre S, Avrahami R, Zussman E, Green M (2017) Durable electrospun microtubes for encapsulation of bacteria in atrazine bioremediation. J Water Process Eng 19:205–211. https://doi.org/10.1016/j.jwpe.2017.08.004
Devers M, Rouard N, Martin-Laurent F (2007) Genetic rearrangement of the atzAB atrazine-degrading gene cassette from pADP1:Tn5 to the chromosome of Variovorax sp. MD1 and MD2. Gene 392:1–6. https://doi.org/10.1016/j.gene.2006.09.015
Dong J, Wang L, Ma F, Yang J, Qi S, Zhao T (2016) The effect of Funnelliformis mosseae inoculation on the phytoremediation of atrazine by the aquatic plant Canna indica L. var. flava Roxb. RSC Adv 6:22538–22549. https://doi.org/10.1039/C5RA23583A
Douglass JF, Radosevich M, Tuovinen OH (2016) Biomineralization of atrazine and analysis of 16S rRNA and catabolic genes of atrazine degraders in a former pesticide mixing site and a machinery washing area. J Soils Sediments 16:2263–2274. https://doi.org/10.1007/s11368-016-1416-3
Douglass JF, Radosevich M, Tuovinen OH (2017) Microbial attenuation of atrazine in agricultural soils: Biometer assays, bacterial taxonomic diversity, and catabolic genes. Chemosphere 176:352–360. https://doi.org/10.1016/j.chemosphere.2017.02.102
El Sebaï T, Devers-Lamrani M, Changey F, Rouard N, Martin-Laurent F (2011) Evidence of atrazine mineralization in a soil from the Nile Delta: isolation of Arthrobacter sp. TES6, an atrazine-degrading strain. Int Biodeterior Biodegrad 65(8):1249–1255. https://doi.org/10.1016/j.ibiod.2011.05.011
Ensz AP, Knapp CW, Graham DW (2003) Influence of autochthonous dissolved organic carbon and nutrient limitation on alachlor biotransformation in aerobic aquatic systems. Environ Sci Technol 37:4157–4162. https://doi.org/10.1021/es0303790
Esperanza M, Houde M, Seoane M, Cid Á, Rioboo C (2017) Does a short-term exposure to atrazine provoke cellular senescence in Chlamydomonas reinhardtii? Aquat Toxicol 189:184–193. https://doi.org/10.1016/j.aquatox.2017.06.015
Esquirol L, Peat TS, Sugrue E, Balotra S, Rottet S, Warden AC, Wilding M, Hartley CJ, Jackson CJ, Newman J, Scott C (2020) Bacterial catabolism of s-triazine herbicides: biochemistry, evolution and application. Adv Microb Physiol 76:129–186. https://doi.org/10.1016/bs.ampbs.2020.01.004
EU (2004) Commission decision of March 2004. Off J Eur Union
Fernandes AFT, Wang P, Staley C, Moretto JAS, Altarugio LM, Campanharo SC, Stehling EG, Sadowsky MJ (2020) Impact of atrazine exposure on the microbial community structure in a Brazilian tropical latosol soil. Microbes Environ 35:1–7. https://doi.org/10.1264/jsme2.ME19143
Fernandez MV, Gardinali PR (2016) Risk assessment of triazine herbicides in surface waters and bioaccumulation of Irgarol and M1 by submerged aquatic vegetation in Southeast Florida. Sci Total Environ 541:1556–1571. https://doi.org/10.1016/j.scitotenv.2015.09.035
Fernández LA, Valverde C, Gómez MA (2013) Isolation and characterization of atrazine-degrading Arthrobacter sp. strains from Argentine agricultural soils. Ann Microbiol 63:207–214. https://doi.org/10.1007/s13213-012-0463-2
Fingler S, Mendaš G, Dvoršćak M, Stipičević S, Vasilić DV (2017) Herbicide micropollutants in surface, ground and drinking waters within and near the area of Zagreb, Croatia. Environ Sci Pollut Res 24:11017–11030. https://doi.org/10.1007/s11356-016-7074-6
Foradori CD, Hinds LR, Hanneman WH, Handa RJ (2009) Effects of atrazine and its withdrawal on gonadotropin-releasing hormone neuroendocrine function in the adult female wistar rat. Biol Reprod 81:1099–1105. https://doi.org/10.1095/biolreprod.109.077453
Foradori CD, Healy JE, Zimmerman AD, Kemppainen RJ, Jones MA, Read CC, White BD, Yi KD, Hinds LR, Lacagnina AF, Quihuis AM, Breckenridge CB, Handa RJ (2018) Characterization of activation of the hypothalamic-pituitary-adrenal axis by the herbicide atrazine in the female rat. Endocrinology 159:3378–3388. https://doi.org/10.1210/en.2018-00474
Gaffar S, Dattamudi S, Baboukani AR, Chanda S, Novak JM, Watts DW, Wang C, Jayachandran K (2021) Physiochemical Characterization of biochars from six feedstocks and their effects on the sorption of atrazine in an organic soil. Agronomy 11:716. https://doi.org/10.3390/agronomy11040716
Gao QT, Wong YS, Tam NFY (2011) Removal and biodegradation of nonylphenol by immobilized Chlorella vulgaris. Bioresour Technol 102:10230–10238. https://doi.org/10.1016/j.biortech.2011.08.070
Gao Y, Fang J, Li W, Wang X, Li F, Du M, Fang J, Lin F, Jiang W, Jiang Z (2019) Effects of atrazine on the physiology, sexual reproduction, and metabolism of eelgrass (Zostera marina L.). Aquat Bot 153:8–14. https://doi.org/10.1016/j.aquabot.2018.10.002
García-González V, Govantes F, Shaw LJ, Burns RG, Santero E (2003) Nitrogen control of atrazine utilization in Pseudomonas sp. strain ADP. Appl Environ Microbiol 69:6987–6993. https://doi.org/10.1128/AEM.69.12.6987-6993.2003
García-González V, Govantes F, Porrúa O, Santero E (2005) Regulation of the Pseudomonas sp. strain ADP cyanuric acid degradation operon. J Bacteriol 187:155–167. https://doi.org/10.1128/JB.187.1.155-167.2005
Gely-Pernot A, Hao C, Becker E, Stuparevic I, Kervarrec C, Chalmel F, Primig M, Jégou B, Smagulova F (2015) The epigenetic processes of meiosis in male mice are broadly affected by the widely used herbicide atrazine. BMC Genomics 16:1–22. https://doi.org/10.1186/s12864-015-2095-y
Gentry TJ, Rensing C, Pepper IL (2004) New approaches for bioaugmentation as a remediation technology. Crit Rev Environ Sci Technol 34:447–494. https://doi.org/10.1080/10643380490452362
Gerhardt KE, Huang XD, Glick BR, Greenberg BM (2009) Phytoremediation and rhizoremediation of organic soil contaminants: potential and challenges. Plant Sci 176:20–30. https://doi.org/10.1016/j.plantsci.2008.09.014
Glinski DA, Purucker ST, Van Meter RJ, Black MC, Henderson WM (2018) Analysis of pesticides in surface water, stemflow, and throughfall in an agricultural area in South Georgia, USA. Chemosphere 209:496–507. https://doi.org/10.1016/j.chemosphere.2018.06.116
Gonzalez-Rey M, Tapie N, Le Menach K, Dévier MH, Budzinski H, Bebianno MJ (2015) Occurrence of pharmaceutical compounds and pesticides in aquatic systems. Mar Pollut Bull 96:384–400. https://doi.org/10.1016/j.marpolbul.2015.04.029
Govantes F, Porrúa O, García-González V, Santero E (2009) Atrazine biodegradation in the lab and in the field: enzymatic activities and gene regulation. Microb Biotechnol 2:178–185. https://doi.org/10.1111/j.1751-7915.2008.00073.x
Graceli JB, Dettogni RS, Merlo E, Niño O, da Costa CS, Zanol JF, Ríos Morris EA, Miranda-Alves L, Denicol AC (2020) The impact of endocrine-disrupting chemical exposure in the mammalian hypothalamic-pituitary axis. Mol Cell Endocrinol 518:110997. https://doi.org/10.1016/j.mce.2020.110997
Hansen AM, Treviño-Quintanilla LG, Márquez-Pacheco H, Villada-Canela M, González-Márquez LC, Guillén-Garcés RA, Hernández-Antonio A (2013) Atrazina: Un herbicida polémico. Rev Int Contam Ambiental 29:65–84
He H, Liu Y, You S, Liu J, Xiao H, Tu Z (2019) A review on recent treatment technology for herbicide atrazine in contaminated environment. Int J Environ Res Public Health 16:5129. https://doi.org/10.3390/ijerph16245129
Hedayatirad M, Mirvaghefi A, Nematollahi MA, Forsatkar MN, Brown C (2020) Transgenerational disrupting impacts of atrazine in zebrafish: Beneficial effects of dietary spirulina. Comp Biochem Physiol Part C Toxicol Pharmacol 230:1–7. https://doi.org/10.1016/j.cbpc.2019.108685
Hernández M, Villalobos P, Morgante V, González M, Reiff C, Moore E, Seeger M (2008) Isolation and characterization of a novel simazine-degrading bacterium from agricultural soil of central Chile, Pseudomonas sp. MHP41. FEMS Microbiol Lett 286:184–190. https://doi.org/10.1111/j.1574-6968.2008.01274.x
Herrera-Gallardo BE, Guzmán-Gil R, Colín-Luna JA, García-Martínez JC, León-Santiesteban HH, González-Brambila OM, González-Brambila MM (2021) Atrazine biodegradation in soil by Aspergillus niger. Can J Chem Eng 99:932–946. https://doi.org/10.1002/cjce.23924
Hirano LQL, Alves S, Menezes-Reis LT, Mendonça J, Simões K, Santos AL, Vieira LG (2019) Effects of egg exposure to atrazine and/or glyphosate on bone development in Podocnemis unifilis (Testudines, Podocnemididae). Ecotoxicol Environ Saf 182:109400. https://doi.org/10.1016/j.ecoenv.2019.109400
Hsieh FM, Huang C, Lin TF, Chen YM, Lin JC (2008) Study of sodium tripolyphosphate-crosslinked chitosan beads entrapped with Pseudomonas putida for phenol degradation. Process Biochem 43:83–92. https://doi.org/10.1016/j.procbio.2007.10.016
Huang Y, Liu Z, He Y, Zeng F, Wang R (2013) Quantifying effects of primary parameters on adsorption-desorption of atrazine in soils. J Soils Sediments 13:82–93. https://doi.org/10.1007/s11368-012-0572-3
Jablonowski ND, Hamacher G, Martinazzo R, Langen U, Köppchen S, Hofmann D, Burauel P (2010) Metabolism and persistence of atrazine in several field soils with different atrazine application histories. J Agric Food Chem 58:12869–12877. https://doi.org/10.1021/jf103577j
Jablonowski ND, Schäffer A, Burauel P (2011) Still present after all these years: persistence plus potential toxicity raise questions about the use of atrazine. Environ Sci Pollut Res 18:328–331. https://doi.org/10.1007/s11356-010-0431-y
James A, Singh DK (2021) Atrazine detoxification by intracellular crude enzyme extracts derived from epiphytic root bacteria associated with emergent hydrophytes. J Environ Sci Health Part B 56:577–586. https://doi.org/10.1080/03601234.2021.1922043
James A, Singh DK, Khankhane PJ (2018) Enhanced atrazine removal by hydrophyte-bacterium associations and in vitro screening of the isolates for their plant growth-promoting potential. Int J Phytoremediation 20:89–97. https://doi.org/10.1080/15226514.2017.1337068
Jiang Z, Jiang D, Zhou Q, Zheng Z, Cao B, Meng Q, Qu J, Wang Y, Zhang Y (2020) Enhancing the atrazine tolerance of Pennisetum americanum (L.) K. Schum by inoculating with indole-3-acetic acid producing strain Pseudomonas chlororaphis PAS18. Ecotoxicol Environ Saf 202:110854. https://doi.org/10.1016/j.ecoenv.2020.110854
Jowa L, Howd R (2011) Should atrazine and related chlorotriazines be considered carcinogenic for human health risk assessment? J Environ Sci Health 29:91–144. https://doi.org/10.1080/10590501.2011.577681
Kapsi M, Tsoutsi C, Paschalidou A, Albanis T (2019) Environmental monitoring and risk assessment of pesticide residues in surface waters of the Louros River (N.W. Greece). Sci Total Environ 650:2188–2198. https://doi.org/10.1016/j.scitotenv.2018.09.185
Khatoon H, Rai JPN (2018) Augmentation of atrazine biodegradation by two Bacilli immobilized on α-Fe2O3 magnetic nanoparticles. Sci Rep 8:1–12. https://doi.org/10.1038/s41598-018-36296-1
Khondee N, Tathong S, Pinyakong O, Powtongsook S, Chatchupong T, Ruangchainikom C, Luepromchai E (2012) Airlift bioreactor containing chitosan-immobilized Sphingobium sp. P2 for treatment of lubricants in wastewater. J Hazard Mater 213–214:466–473. https://doi.org/10.1016/j.jhazmat.2012.02.018
Kidd PS, Prieto-Fernández A, Monterroso C, Acea MJ (2008) Rhizosphere microbial community and hexachlorocyclohexane degradative potential in contrasting plant species. Plant Soil 302:233–247. https://doi.org/10.1007/s11104-007-9475-2
Kirsten KS, Canova R, Soveral LF, Friedrich MT, Frandoloso R, Kreutz LC (2017) Reduced expression of selective immune-related genes in silver catfish (Rhamdia quelen) monocytes exposed to atrazine. Fish Shellfish Immunol 64:78–83. https://doi.org/10.1016/j.fsi.2017.03.006
Köck-Schulmeyer M, Postigo C, Farré M, Barceló D, López de Alda M (2019) Medium to highly polar pesticides in seawater: analysis and fate in coastal areas of Catalonia (NE Spain). Chemosphere 215:515–523. https://doi.org/10.1016/j.chemosphere.2018.10.049
Kolić NU, Hršak D, Begonja Kolar A, Petrić I, Stipičevic S, Soulas G, Martin-Laurent F (2007) Combined metabolic activity within an atrazine-mineralizing community enriched from agrochemical factory soil. Int Biodeterior Biodegrad 60:299–307. https://doi.org/10.1016/j.ibiod.2007.05.004
Komsky-Elbaz A, Roth Z (2017) Effect of the herbicide atrazine and its metabolite DACT on bovine sperm quality. Reprod Toxicol 67:15–25. https://doi.org/10.1016/j.reprotox.2016.11.001
Krastanov A, Alexieva Z, Yemendzhiev H (2013) Microbial degradation of phenol and phenolic derivatives. Eng Life Sci 13:76–87. https://doi.org/10.1002/elsc.201100227
Krutz LJ, Shaner DL, Accinelli C, Zablotowicz RM, Henry WB (2008) Atrazine dissipation in s-triazine adapted and nonadapted soil from Colorado and Mississippi: Implications of enhanced degradation on atrazine fate and transport parameter. J Environ Qual 37:848–857. https://doi.org/10.2134/jeq2007.0448
Kupryianchyk D, Hale SE, Breedveld GD, Cornelissen G (2016) Treatment of sites contaminated with perfluorinated compounds using biochar amendment. Chemosphere 142:35–40. https://doi.org/10.1016/j.chemosphere.2015.04.085
Lammertyn S, Masín CE, Zalazar CS, Fernandez ME (2021) Biomarkers response and population biological parameters in the earthworm Eisenia fetida after short term exposure to atrazine herbicide. Ecol Indic 121:107–173. https://doi.org/10.1016/j.ecolind.2020.107173
Le Coadou L, Le Ménach K, Labadie P, Dévier MH, Pardon P, Augagneur S, Budzinski H (2017) Quality survey of natural mineral water and spring water sold in France: monitoring of hormones, pharmaceuticals, pesticides, perfluoroalkyl substances, phthalates, and alkylphenols at the ultra-trace level. Sci Total Environ 603–604:651–662. https://doi.org/10.1016/j.scitotenv.2016.11.174
Lewis KA, Tzilivakis J, Warner DJ, Green A (2016) An international database for pesticide risk assessments and management. Hum Ecol Risk Assess 22:1050–1064. https://doi.org/10.1080/10807039.2015.1133242
Li Q, Li Y, Zhu X, Cai B (2008) Isolation and characterization of atrazine-degrading Arthrobacter sp. AD26 and use of this strain in bioremediation of contaminated soil. J Environ Sci 20:1226–1230. https://doi.org/10.1016/S1001-0742(08)62213-5
Li XN, Lin J, Xia J, Qin L, Zhu SY, Li JL (2017) Lycopene mitigates atrazine-induced cardiac inflammation via blocking the NF-κB pathway and NO production. J Funct Foods 29:208–216. https://doi.org/10.1016/j.jff.2016.12.029
Li H, Feng Y, Li X, Zeng D (2018) Analytical confirmation of various herbicides in drinking water resources in sugarcane production regions of Guangxi, China. Bull Environ Contam Toxicol 100:815–820. https://doi.org/10.1007/s00128-018-2324-6
Li MR, Zhan FD, Chen JJ, Zu YQ, Li Y (2020) Atrazine degradation pathway and genes of Arthrobacter sp. Fm326. Pol J Environ Stud 29:3683–3689. https://doi.org/10.15244/pjoes/115326
Liu Y, Fan X, Zhang T, He W, Song F (2020) Effects of the long-term application of atrazine on soil enzyme activity and bacterial community structure in farmlands in China. Environ Pollut 262:114–264. https://doi.org/10.1016/j.envpol.2020.114264
Liu J, Zhou JH, Guo QN, Ma LY, Yang H (2021) Physiochemical assessment of environmental behaviors of herbicide atrazine in soils associated with its degradation and bioavailability to weeds. Chemosphere 262:1–13. https://doi.org/10.1016/j.chemosphere.2020.127830
Lu D, Zhang Y, Niu S, Wang L, Lin S, Wang C, Ye W, Yan C (2012) Study of phenol biodegradation using Bacillus amyloliquefaciens strain WJDB-1 immobilized in alginate-chitosan-alginate (ACA) microcapsules by electrochemical method. Biodegradation 23:209–219. https://doi.org/10.1007/s10532-011-9500-2
Lyon DY, Vogel TM (2013) Bioaugmentation for groundwater remediation: an overview. In: Stroo H, Leeson A, Ward C (eds) Bioaugmentation for groundwater remediation. SERDP ESTCP environmental remediation technology. Springer, New York. https://doi.org/10.1007/978-1-4614-4115-1_1
Ma L, Chen S, Yuan J, Yang P, Liu Y, Stewart K (2017) Rapid biodegradation of atrazine by Ensifer sp. strain and its degradation genes. Int Biodeterior Biodegrad 116:133–140. https://doi.org/10.1016/j.ibiod.2016.10.022
Mac Loughlin TM, Peluso L, Marino DJG (2017) Pesticide impact study in the peri-urban horticultural area of Gran La Plata, Argentina. Sci Total Environ 598:572–580. https://doi.org/10.1016/j.scitotenv.2017.04.116
Mahler BJ, Van Metre PC, Burley TE, Loftin KA, Meyer MT, Nowell LH (2017) Similarities and differences in occurrence and temporal fluctuations in glyphosate and atrazine in small Midwestern streams (USA) during the 2013 growing season. Sci Total Environ 579:149–158. https://doi.org/10.1016/j.scitotenv.2016.10.236
Mandelbaum RT, Allan DL, Wackett LP (1995) Isolation and characterization of a Pseudomonas sp. that mineralizes the s-triazine herbicide atrazine. Appl Environ Microbiol 61:1451–1457. https://doi.org/10.1128/aem.61.4.1451-1457.1995
Marchi G, Marchi ECS, Guimarães TG (2008). Herbicidas: mecanismos de ação e uso. Embrapa Cerrados-Documentos (INFOTECA-E) (in Portuguese)
Martinez B, Tomkins J, Wackett LP, Wing R, Sadowsky M (2001) Complete nucleotide sequence and organization of the atrazine catabolic plasmid pADP-1 from Pseudomonas sp. strain ADP. J Bacteriol 183:5684–5697. https://doi.org/10.1128/JB.183.19.5684-5697.2001
Martins EC, de Freitas MV, Bohone JB, Abate G (2018) Sorption and desorption of atrazine on soils: the effect of different soil fractions. Geoderma 322:131–139. https://doi.org/10.1016/j.geoderma.2018.02.028
Mas LI, Aparicio VC, De Gerónimo E, Costa JL (2020) Pesticides in water sources used for human consumption in the semiarid region of Argentina. SN Appl Sci 2:1–18. https://doi.org/10.1007/s42452-020-2513-x
Masoud AA, Abdel-Wahab Arafa NA, El-Bouraie M (2018) Patterns and trends of the pesticide pollution of the shallow Nile Delta aquifer (Egypt). Water Air Soil Pollut 229:1–23. https://doi.org/10.1007/s11270-018-3802-5
Mauffret A, Baran N, Joulian C (2017) Effect of pesticides and metabolites on groundwater bacterial community. Sci Total Environ 576:879–887. https://doi.org/10.1016/j.scitotenv.2016.10.108
Menchen A, Heras JD, Alday JJ (2017) Pesticide contamination in groundwater bodies in the Júcar River European Union Pilot Basin (SE Spain). Environ Monit Assess 189:146. https://doi.org/10.1007/s10661-017-5827-4
Merini LJ, Bobillo C, Cuadrado V, Corach D, Giulietti AM (2009) Phytoremediation potential of the novel atrazine tolerant Lolium multiflorum and studies on the mechanisms involved. Environ Pollut 157:3059–3063. https://doi.org/10.1016/j.envpol.2009.05.036
Metcalfe CD, Helm P, Paterson G, Kaltenecker G, Murray C, Nowierski M, Sultana T (2019) Pesticides related to land use in watersheds of the Great Lakes basin. Sci Total Environ 648:681–692. https://doi.org/10.1016/j.scitotenv.2018.08.169
Montagner CC, Sodré FF, Acayaba RD, Vidal C, Campestrini I, Locatelli MA, Pescara IC, Albuquerque AF, Umbuzeiro GA, Jardim WF (2019) Ten years-snapshot of the occurrence of emerging contaminants in drinking, surface and ground waters and wastewaters from São Paulo State, Brazil. J Braz Chem Soc 30:614–632. https://doi.org/10.21577/0103-5053.20180232
Montiel-León JM, Munoz G, Vo Duy S, Do DT, Vaudreuil MA, Goeury K, Guillemette F, Amyot M, Sauvé S (2019a) Widespread occurrence and spatial distribution of glyphosate, atrazine, and neonicotinoids pesticides in the St. Lawrence and tributary rivers. Environ Pollut 250:29–39. https://doi.org/10.1016/j.envpol.2019.03.125
Montiel-León JM, Vo Duy S, Munoz G, Bouchard MF, Amyot M, Sauvé S (2019b) Quality survey and spatiotemporal variations of atrazine and desethylatrazine in drinking water in Quebec, Canada. Sci Total Environ 671:578–585. https://doi.org/10.1016/j.scitotenv.2019.03.228
Montoya JC, Porfiri C, Roberto ZE, Viglizzo EF (2019) Assessing the vulnerability of groundwater resources in semiarid lands of central Argentina. Sustain Water Resour Manage 5:1419–1434. https://doi.org/10.1007/s40899-018-0246-4
Morales-Pérez AA, Arias C, Ramírez-Zamora RM (2016) Removal of atrazine from water using an iron photo catalyst supported on activated carbon. Adsorption 22:49–58. https://doi.org/10.1007/s10450-015-9739-8
Morgante V, López-López A, Flores C, González M, González B, Vásquez M, Rosselló-Mora R, Seeger M (2010) Bioaugmentation with Pseudomonas sp. strain MHP41 promotes simazine attenuation and bacterial community changes in agricultural soils. FEMS Microbiol Ecol 71:114–126. https://doi.org/10.1111/j.1574-6941.2009.00790.x
Morillo E, Villaverde J (2017) Advanced technologies for the remediation of pesticide-contaminated soils. Sci Total Environ 586:576–597. https://doi.org/10.1016/j.scitotenv.2017.02.020
Mudhoo A, Garg VK (2011) Sorption, transport and transformation of atrazine in soils, minerals and composts: a review. Pedosphere 21:11–25. https://doi.org/10.1016/S1002-0160(10)60074-4
Mukherjee PK, Sondhia S, Singh P, Sagar RL (2019) Atrazine use to control weeds and its residue determination in fodder crops of maize and sorghum. Indian J Weed Sci 51:163. https://doi.org/10.5958/0974-8164.2019.00035.2
Muratova A, Golubev S, Wittenmayer L, Dmitrieva T, Bondarenkova A, Hirche F, Merbach W, Turkovskaya O (2009) Effect of the polycyclic aromatic hydrocarbon phenanthrene on root exudation of Sorghum bicolor (L.) Moench. Environ Exp Bot 66:514–521. https://doi.org/10.1016/j.envexpbot.2009.03.001
Nguyen LN, Hai FI, Yang S, Kang J, Leusch FDL, Roddick F, Price WE, Nghiem LD (2014) Removal of pharmaceuticals, steroid hormones, phytoestrogens, UV-filters, industrial chemicals and pesticides by Trametes versicolor: role of biosorption and biodegradation. Int Biodeterior Biodegrad 88:169–175. https://doi.org/10.1016/j.ibiod.2013.12.017
Niti C, Sunita S, Kamlesh K, Rakesh K (2013) Bioremediation: an emerging technology for remediation of pesticides. Res J Chem Environ 17:88–105
Nödler K, Licha T, Voutsa D (2013) Twenty years later-atrazine concentrations in selected coastal waters of the Mediterranean and the Baltic Sea. Mar Pollut Bull 70:112–118. https://doi.org/10.1016/j.marpolbul.2013.02.018
Nzila A, Razzak SA, Zhu J (2016) Bioaugmentation: an emerging strategy of industrial wastewater treatment for reuse and discharge. Int J Environ Res Public Health 13:846. https://doi.org/10.3390/ijerph13090846
Ofaim S, Zarecki R, Porob S, Gat D, Lahav T, Kashi Y, Aly R, Eizenberg H, Ronen Z, Freilich S (2020) Genome-scale reconstruction of Paenarthrobacter aurescens TC1 metabolic model towards the study of atrazine bioremediation. Sci Rep 10:13019. https://doi.org/10.1038/s41598-020-69509-7
Ortiz-Hernández ML, Monterrosas-Brisson M, Yánez-Ocampo G, Sánchez-Salinas E (2001) Biodegradation of methyl-parathion by bacteria isolated of agricultural soil. Rev Int Contam Ambient 17:147–155
Ouyang W, Zhang Y, Gu X, Tysklind M, Lin C, Wang B, Xin M (2019) Occurrence, transportation, and distribution difference of typical herbicides from estuary to bay. Environ Int 130:104858. https://doi.org/10.1016/j.envint.2019.05.052
Owolabi OD, Omotosho JS (2017) Atrazine-mediated oxidative stress responses and lipid peroxidation in the tissues of Clarias gariepinus. Iran J Toxicol 11:29–38. https://doi.org/10.29252/arakmu.11.2.29
Pan L, Feng X, Cao M, Zhang S, Huang Y, Xu T, Jing J, Zhang H (2019) Determination and distribution of pesticides and antibiotics in agricultural soils from northern China. RSC Adv 9:15686–15693. https://doi.org/10.1039/c9ra00783k
Pannier A, Lehrer T, Vogel M, Soltmann U, Böttcher H, Tarre S, Green M, Raff J, Pollmann K (2014) Long-term activity of biohybrid coatings of atrazine-degrading bacteria Pseudomonas sp. RSC Adv 4:19970–19979. https://doi.org/10.1039/c4ra02928c
Park JK, Chang HN (2000) Microencapsulation of microbial cells. Biotechnol Adv 18:303–319. https://doi.org/10.1016/S0734-9750(00)00040-9
Peng J, Lu X, Jiang X, Zhang Y, Chen Q, Lai B, Yao G (2018) Degradation of atrazine by persulfate activation with copper sulfide (CuS): Kinetics study, degradation pathways and mechanism. Chem Eng J 354:740–752. https://doi.org/10.1016/j.cej.2018.08.038
Philp JC, Atlas RM (2005) Bioremediation of contaminated soils and aquifers. In: Atlas RM, Philip J (eds) Bioremediation. Springer, New York. https://doi.org/10.1128/9781555817596.ch5
Piratoba ARA, Miranda MS, Marulanda NME, Pereira GAM, Lima CF, Silva AA (2021) Sorption and desorption of atrazine in horizons of the red-yellow latosol. Adv Weed Sci 39:1–7. https://doi.org/10.51694/advweedsci/2021;39:00003
Piutti S, Semon E, Landry D, Hartmann A, Dousset S, Lichtfouse E, Topp E, Soulas G, Martin-Laurent F (2003) Isolation and characterisation of Nocardioides sp. SP12, an atrazine-degrading bacterial strain possessing the gene trzN from bulk- and maize rhizosphere soil. FEMS Microbiol Lett 221:111–117. https://doi.org/10.1016/S0378-1097(03)00168-X
Prado B, Duwig C, Hidalgo C, Müller K, Mora L, Raymundo E, Etchevers JD (2014) Transport, sorption and degradation of atrazine in two clay soils from Mexico: Andosol and Vertisol. Geoderma 232–234:628–639. https://doi.org/10.1016/j.geoderma.2014.06.011
Prosen H, Zupančič-Kralj L (2005) Evaluation of photolysis and hydrolysis of atrazine and its first degradation products in the presence of humic acids. Environ Pollut 133:517–529. https://doi.org/10.1016/j.envpol.2004.06.015
Qu M, Li N, Li H, Yang T, Liu W, Yan Y, Feng X, Zhu D (2018) Phytoextraction and biodegradation of atrazine by Myriophyllum spicatum and evaluation of bacterial communities involved in atrazine degradation in lake sediment. Chemosphere 209:439–448. https://doi.org/10.1016/j.chemosphere.2018.06.055
Radosevich M, Traina SJ, Hao YL, Tuovinen OH (1995) Degradation and mineralization of atrazine by a soil bacterial isolate. Appl Environ Microbiol 61:297–302. https://doi.org/10.1128/aem.61.1.297-302.1995
Rajendran K, Pujari L, Ethiraj K (2021) Biodegradation and bioremediation of s-triazine herbicides. In: Gothandam KM, Ranjan S, Dasgupta N, Lichtfouse E (eds) Environmental biotechnology vol. 3. Environmental chemistry for a sustainable world. Springer, Cham. https://doi.org/10.1007/978-3-030-48973-1_2
Religia P, Kato Y, Fukushima EO, Matsuura T, Muranaka T, Watanabe H (2019) Atrazine exposed phytoplankton causes the production of non-viable offspring on Daphnia magna. Mar Environ Res 145:177–183. https://doi.org/10.1016/j.marenvres.2019.02.007
Rimayi C, Odusanya D, Weiss JM, de Boer J, Chimuka L (2018) Seasonal variation of chloro-s-triazines in the Hartbeespoort Dam catchment, South Africa. Sci Total Environ 613–614:472–482. https://doi.org/10.1016/j.scitotenv.2017.09.119
Rinsky JL, Hopenhayn C, Golla V, Browning S, Bush HM (2012) Atrazine exposure in public drinking water and preterm birth. Public Health Rep 127:72–80. https://doi.org/10.1177/003335491212700108
Rousseaux S, Hartmann A, Soulas G (2001) Isolation and characterisation of new gram-negative and gram-positive atrazine degrading bacteria from different French soils. FEMS Microbiol Ecol 36:211–222. https://doi.org/10.1016/S0168-6496(01)00135-0
Rousseaux S, Soulas G, Hartmann A (2002) Plasmid localisation of atrazine-degrading genes in newly described Chelatobacter and Arthrobacter strains. FEMS Microbiol Ecol 41:69–75. https://doi.org/10.1016/S0168-6496(02)00269-6
Sadowsky MJ, Tong Z, De Souza M, Wackett LP (1998) AtzC is a new member of the amidohydrolase protein superfamily and is homologous to other atrazine-metabolizing enzymes. J Bacteriol 180:152–158. https://doi.org/10.1128/jb.180.1.152-158.1998
Sagarkar S, Mukherjee S, Nousiainen A, Björklöf K, Purohit HJ, Jorgensen KS, Kapley A (2013) Monitoring bioremediation of atrazine in soil microcosms using molecular tools. Environ Pollut 172:108–115. https://doi.org/10.1016/j.envpol.2012.07.048
Sagarkar S, Nousiainen A, Shaligram S, Björklöf K, Lindström K, Jørgensen KS, Kapley A (2014) Soil mesocosm studies on atrazine bioremediation. J Environ Manag 139:208–216. https://doi.org/10.1016/j.jenvman.2014.02.016
Sagarkar S, Bhardwaj P, Storck V, Devers-Lamrani M, Martin-Laurent F, Kapley A (2016) s-triazine degrading bacterial isolate Arthrobacter sp. AK-YN10, a candidate for bioaugmentation of atrazine contaminated soil. Appl Microbiol Biotechnol 100:903–913. https://doi.org/10.1007/s00253-015-6975-5
Sajjaphan K, Shapir N, Wackett LP, Palmer M, Blackmon B, Tomkins J, Sadowsky MJ (2004) Arthrobacter aurescens TC1 atrazine catabolism genes trzN, atzB, and atzC are linked on a 160-kilobase region and are functional in Escherichia coli. Appl Environ Microbiol 70:4402–4407. https://doi.org/10.1128/AEM.70.7.4402-4407.2004
Salazar-Ledesma M, Prado B, Zamora O, Siebe C (2018) Mobility of atrazine in soils of a wastewater irrigated maize field. Agric Ecosyst Environ 255:73–83. https://doi.org/10.1016/j.agee.2017.12.018
Sánchez V, López-Bellido FJ, Cañizares P, Rodríguez L (2017) Assessing the phytoremediation potential of crop and grass plants for atrazine-spiked soils. Chemosphere 185:119–126. https://doi.org/10.1016/j.chemosphere.2017.07.013
Sánchez V, López-Bellido FJ, Rodrigo MA, Fernández FJ, Rodríguez L (2020) A mesocosm study of electrokinetic-assisted phytoremediation of atrazine-polluted soils. Sep Purif Technol 233:116044. https://doi.org/10.1016/j.seppur.2019.116044
Sassine L, Le Salle C, Khaska M, Verdoux P, Meffre P, Benfodda Z, Roig B (2017) Spatial distribution of triazine residues in a shallow alluvial aquifer linked to groundwater residence time. Environ Sci Pollut Res 24:6878–6888. https://doi.org/10.1007/s11356-016-7224-x
Seffernick JL, Johnson G, Sadowsky MJ, Wackett LP (2000) Substrate specificity of atrazine chlorohydrolase and atrazine-catabolizing bacteria. Appl Environ Microbiol 66:4247–4252. https://doi.org/10.1128/AEM.66.10.4247-4252.2000
Seffernick JL, Shapir N, Schoeb M, Johnson G, Sadowsky MJ, Wackett LP (2002) Enzymatic degradation of chlorodiamino-s-triazine. Am Soc Microbiol 68:4672–4675. https://doi.org/10.1128/AEM.68.9.4672
Seffernick JL, Erickson JS, Cameron SM, Cho S, Dodge AG, Richman JE, Sadowsky MJ, Wackett LP (2012) Defining sequence space and reaction products within the cyanuric acid hydrolase (AtzD)/barbiturase protein family. J Bacteriol 194:4579–4588. https://doi.org/10.1128/JB.00791-12
Shapir N, Rosendahl C, Johnson G, Andreina M, Sadowsky MJ, Wackett LP (2005) Substrate specificity and colorimetric assay for recombinant trzN derived from Arthrobacter aurescens TC1. Appl Environ Microbiol 71:2214–2220. https://doi.org/10.1128/AEM.71.5.2214-2220.2005
Shapir N, Pedersen C, Gil O, Strong L, Seffernick J, Sadowsky MJ, Wackett LP (2006) TrzN from Arthrobacter aurescens TC1 is a zinc amidohydrolase. J Bacteriol 188:5859–5864. https://doi.org/10.1128/JB.00517-06
Shimp J, Tracy J, Davis L, Lee E, Huang W, Erickson L, Schnoor J (1993) Beneficial effects of plants in the remediation of soil and groundwater contaminated with organic materials. Crit Rev Environ Sci Technol 23:41–77. https://doi.org/10.1080/10643389309388441
Simpkins JW, Swenberg JA, Weiss N, Brusick D, Eldridge JC, Stevens JT, Handa RJ, Hovey RC, Plant TM, Pastoor TP, Breckenridge CB (2011) Atrazine and breast cancer: a framework assessment of the toxicological and epidemiological evidence. Toxicol Sci 123:441–459. https://doi.org/10.1093/toxsci/kfr176
Singh SN, Jauhari N (2017) Degradation of atrazine by plants and microbes. In: Singh S (ed) Microbe-induced degradation of pesticides. Environmental science and engineering. Springer, Cham. https://doi.org/10.1007/978-3-319-45156-5_10
Siripattanakul S, Wirojanagud W, McEvoy JM, Casey FXM, Khan E (2009) Atrazine removal in agricultural infiltrate by bioaugmented polyvinyl alcohol immobilized and free Agrobacterium radiobacter J14a: A sand column study. Chemosphere 74:308–313. https://doi.org/10.1016/j.chemosphere.2008.09.005
Smith D, Alvey S, Crowley DE (2005) Cooperative catabolic pathways within an atrazine-degrading enrichment culture isolated from soil. FEMS Microbiol Ecol 53:265–275. https://doi.org/10.1016/j.femsec.2004.12.011
Soltanian S (2016) Effect of atrazine on immunocompetence of red-eared slider turtle (Trachemys scripta). J Immunotoxicol 13:804–809. https://doi.org/10.1080/1547691X.2016.1195463
Song Y, Jia ZC, Chen JY, Hu JX, Zhang LS (2014) Toxic effects of atrazine on reproductive system of male rats. Biomed Environ Sci 27:281–288. https://doi.org/10.3967/bes2014.050
Souza PMS, Lobo FA, Rosa AH, Fraceto LF (2012) Desenvolvimento de nanocápsulas de poli-ε-caprolactona contendo o herbicida atrazina. Quim Nova 35:132–137. https://doi.org/10.1590/S0100-40422012000100024
Stara A, Kouba A, Velisek J (2018) Biochemical and histological effects of sub-chronic exposure to atrazine in crayfish Cherax destructor. Chem Biol Interact 291:95–102. https://doi.org/10.1016/j.cbi.2018.06.012
Stayner LT, Almberg K, Jones R, Graber J, Pedersen M, Turyk M (2017) Atrazine and nitrate in drinking water and the risk of preterm delivery and low birth weight in four Midwestern states. Environ Res 152:294–303. https://doi.org/10.1016/j.envres.2016.10.022
Strong LC, Rosendahl C, Johnson G, Sadowsky MJ, Wackett LP (2002) Arthrobacter aurescens TC1 metabolizes diverse s-triazine ring compounds. Appl Environ Microbiol 68:5973–5980. https://doi.org/10.1128/AEM.68.12.5973-5980.2002
Struthers JK, Jayachandran K, Moorman TB (1998) Biodegradation of atrazine by Agrobacterium radiobacter J14a and use of this strain in bioremediation of contaminated soil. Appl Environ Microbiol 64:3368–3375. https://doi.org/10.1128/aem.64.9.3368-3375.1998
Sui Y, Yang H (2013) Bioaccumulation and degradation of atrazine in several Chinese ryegrass genotypes. Environ Sci Process Impacts 15:2338–2344. https://doi.org/10.1039/c3em00375b
Sun JT, Pan LL, Zhan Y, Tsang DCW, Zhu LZ, Li XD (2017) Atrazine contamination in agricultural soils from the Yangtze River Delta of China and associated health risks. Environ Geochem Health 39:369–378. https://doi.org/10.1007/s10653-016-9853-x
Sun X, Liu F, Shan R, Fan Y (2019) Spatiotemporal distributions of Cu, Zn, metribuzin, atrazine, and their transformation products in the surface water of a small plain stream in eastern China. Environ Monit Assess 191:1–13. https://doi.org/10.1007/s10661-019-7556-3
Sun C, Xu Y, Hu N, Ma J, Sun S, Cao W, Klobučar G, Hu C, Zhao Y (2020a) To evaluate the toxicity of atrazine on the freshwater microalgae Chlorella sp. using sensitive indices indicated by photosynthetic parameters. Chemosphere 244:125514. https://doi.org/10.1016/j.chemosphere.2019.125514
Sun T, Miao J, Saleem M, Zhang H, Yang Y, Zhang Q (2020b) Bacterial compatibility and immobilization with biochar improved tebuconazole degradation, soil microbiome composition and functioning. J Hazard Mater 398:122941. https://doi.org/10.1016/j.jhazmat.2020.122941
Tanimoto E (2005) Regulation of root growth by plant hormones - roles for auxin and gibberellin. CRC Crit Rev Plant Sci 24:249–265. https://doi.org/10.1080/07352680500196108
Tao Y, Han S, Zhang Q, Yang Y, Shi H, Akindolie MS, Jiao Y, Qu J, Jiang Z, Han W, Zhang Y (2020) Application of biochar with functional microorganisms for enhanced atrazine removal and phosphorus utilization. J Clean Prod 257:120535. https://doi.org/10.1016/j.jclepro.2020.120535
Thueson LE, Emmons TR, Browning DL, Kreitinger JM, Shepherd DM, Wetzel SA (2015) In vitro exposure to the herbicide atrazine inhibits T cell activation, proliferation, and cytokine production and significantly increases the frequency of Foxp3+ regulatory T cells. Toxicol Sci 143:418–429. https://doi.org/10.1093/toxsci/kfu242
Tong HW, Mutlu BR, Wackett LP, Aksan A (2014) Manufacturing of bioreactive nanofibers for bioremediation. Biotechnol Bioeng 111:1483–1493. https://doi.org/10.1002/bit.25208
Topp E, Mulbry WM, Zhu H, Nour SM, Cuppels D (2000a) Characterization of s-triazine herbicide metabolism by a Nocardioides sp. isolated from agricultural soils. Appl Environ Microbiol 66:3134–3141. https://doi.org/10.1128/AEM.66.8.3134-3141.2000
Topp E, Zhu H, Nour SM, Houot S, Lewis M, Cuppels D (2000b) Characterization of an atrazine-degrading Pseudaminobacter sp. isolated from Canadian and French agricultural soils. Appl Environ Microbiol 66:2773–2782. https://doi.org/10.1128/AEM.66.7.2773-2782.2000
Tyagi M, da Fonseca MM, de Carvalho CC (2011) Bioaugmentation and biostimulation strategies to improve the effectiveness of bioremediation processes. Biodegradation 22:231–241. https://doi.org/10.1007/s10532-010-9394-4
Udiković N, Hršak D, Mendaš G, Filipčić D (2003) Enrichment and characterization of atrazine degrading bacterial communities. Food Technol Biotechnol 41:211–217
Udiković-Kolić N, Scott C, Martin-Laurent F (2012) Evolution of atrazine-degrading capabilities in the environment. Appl Microbiol Biotechnol 96:1175–1189. https://doi.org/10.1007/s00253-012-4495-0
Urseler N, Bachetti R, Morgante V, Agostini E, Morgante C (2021) Atrazine behavior in an agricultural soil: adsorption-desorption, leaching and bioaugmentation with Arthrobacter sp. strain AAC22. J Soils Sediments. https://doi.org/10.1007/s11368-021-03045-3
US EPA (2007) Atrazine - chemical summary. Publicacions Matem. https://doi.org/10.5565/PUBLMAT_Introduction
Vaishampayan PA, Kanekar PP, Dhakephalkar PK (2007) Isolation and characterization of Arthrobacter sp. strain MCM B-436, an atrazine-degrading bacterium, from rhizospheric soil. Int Biodeterior Biodegrad 60:273–278. https://doi.org/10.1016/j.ibiod.2007.05.001
Verrhiest GJ, Clément B, Volat B, Montuelle B, Perrodin Y (2002) Interactions between a polycyclic aromatic hydrocarbon mixture and the microbial communities in a natural freshwater sediment. Chemosphere 46:187–196. https://doi.org/10.1016/S0045-6535(01)00064-9
Vibber LL, Pressler MJ, Colores GM (2007) Isolation and characterization of novel atrazine-degrading microorganisms from an agricultural soil. Appl Microbiol Biotechnol 75:921–928. https://doi.org/10.1007/s00253-007-0871-6
Viegas CA, Chelinho S, Moreira-santos M, Costa C, Gil FN, Silva C, Lima D, Ribeiro R, Sousa JP, Fialho AM (2012) Bioremediation of soils contaminated with atrazine and other s-triazine herbicides: current state and prospects. Adv Environ Res 6:1–49
Viegas CA, Silva VP, Varela VM, Correia V, Ribeiro R, Moreira-Santos M (2019) Evaluating formulation and storage of Arthrobacter aurescens strain TC1 as a bioremediation tool for terbuthylazine contaminated soils: Efficacy on abatement of aquatic ecotoxicity. Sci Total Environ 668:714–722. https://doi.org/10.1016/j.scitotenv.2019.02.355
Wang D, Shin JY, Cheney MA, Sposito G, Spiro TG (1999) Manganese dioxide as a catalyst for oxygen-independent atrazine dealkylation. Environ Sci Technol 33:3160–3165. https://doi.org/10.1021/es990419t
Wang J, Van Tittelboom K, De Belie N, Verstraete W (2012) Use of silica gel or polyurethane immobilized bacteria for self-healing concrete. Constr Build Mater 26:532–540. https://doi.org/10.1016/j.conbuildmat.2011.06.054
Wang H, Liu Y, Li J, Lin M, Hu X (2016) Biodegradation of atrazine by Arthrobacter sp. C3, isolated from the herbicide-contaminated corn field. Int J Environ Sci Technol 13:257–262. https://doi.org/10.1007/s13762-015-0860-8
Wang F, Yang QW, Zhao WJ, Du QY, Chang ZJ (2019) Effects of short-time exposure to atrazine on miRNA expression profiles in the gonad of common carp (Cyprinus carpio). BMC Genomics 20:1–15. https://doi.org/10.1186/s12864-019-5896-6
Wang A, Hu X, Wan Y, Mahai G, Jiang Y, Huo W, Zhao X, Liang G, He Z, Xia W, Xu S (2020) A nationwide study of the occurrence and distribution of atrazine and its degradates in tap water and groundwater in China: assessment of human exposure potential. Chemosphere 252:126533. https://doi.org/10.1016/j.chemosphere.2020.126533
WHO (2011) Atrazine and its metabolites in drinking-water. In: Background document for development of WHO guidelines for drinking-water quality. World Health Organization Press, Geneva
Xian-Zhu D, Jian-Dong J, Li-Feng G, Rong-Qing P, Shun-Peng L (2007) Study on the atrazine-degrading genes in Arthrobacter sp. AG1. Chin J Biotechnol 23:789–793. https://doi.org/10.1016/S1872-2075(07)60049-1
Xie H, Wang X, Chen J, Li X, Jia G, Zou Y, Zhang Y, Cui Y (2019) Occurrence, distribution and ecological risks of antibiotics and pesticides in coastal waters around Liaodong Peninsula, China. Sci Total Environ 656:946–951. https://doi.org/10.1016/j.scitotenv.2018.11.449
Xie J, Lin L, Sánchez OF, Bryan C, Freeman JL, Yuan C (2021) Pre-differentiation exposure to low-dose of atrazine results in persistent phenotypic changes in human neuronal cell lines. Environ Pollut 271:116379. https://doi.org/10.1016/j.envpol.2020.116379
Xing H, Wang Z, Gao X, Chen D, Wang L, Li S, Xu S (2015) Atrazine and chlorpyrifos exposure induces liver autophagic response in common carp. Ecotoxicol Environ Saf 113:52–58. https://doi.org/10.1016/j.ecoenv.2014.11.027
Xue Y, Zhang Z-M, Zhang R-R, Li Y-Q, Sun A-L, Shi X-Z, Chen J, Song S (2021) Aquaculture-derived distribution, partitioning, migration, and transformation of atrazine and its metabolites in seawater, sediment, and organisms from a typical semi-closed mariculture bay. Environ Pollut 271:116362. https://doi.org/10.1016/j.envpol.2020.116362
Yang C, Li Y, Zhang K, Wang X, Ma C, Tang H, Xu P (2010) Atrazine degradation by a simple consortium of Klebsiella sp. A1 and Comamonas sp. A2 in nitrogen enriched medium. Biodegradation 21:97–105. https://doi.org/10.1007/s10532-009-9284-9
Yang K, Li H, Li L, Hu J, Wei Y, Yang H, Denton MD, Zhou Y, Li J (2021) Soil metabolomics reveal complex interactions between Arthrobacter ureafaciens and Trichoderma harzianum when co-inoculated on wheat. Pedobiologia 85–86:150–723. https://doi.org/10.1016/j.pedobi.2021.150723
Yanze-Kontchou C, Gschwind N (1994) Mineralization of the herbicide atrazine as a carbon source by a Pseudomonas strain. Appl Environ Microbiol 60:4297–4302. https://doi.org/10.1128/aem.60.12.4297-4302.1994
Ye JY, Zhang JB, Gao JG, Li HT, Liang D, Liu RM (2016) Isolation and characterization of atrazine-degrading strain Shewanella sp. YJY4 from cornfield soil. Lett Appl Microbiol 63:45–52. https://doi.org/10.1111/lam.12584
Yu T, Wang L, Ma F, Yang J, Bai S, You J (2019) Self-immobilized biomixture with pellets of Aspergillus niger Y3 and Arthrobacter sp. ZXY-2 to remove atrazine in water: a bio-functions integration system. Sci Total Environ 689:875–882. https://doi.org/10.1016/j.scitotenv.2019.06.313
Yu T, Wang L, Ma F, Wang Y, Bai S (2020) A bio-functions integration microcosm: self-immobilized biochar-pellets combined with two strains of bacteria to remove atrazine in water and mechanisms. J Hazard Mater 384:121326. https://doi.org/10.1016/j.jhazmat.2019.121326
Yue L, Ge CJ, Feng D, Yu H, Deng H, Fu B (2017) Adsorption–desorption behavior of atrazine on agricultural soils in China. J Environ Sci 57:180–189. https://doi.org/10.1016/j.jes.2016.11.002
Zhang Y, Jiang Z, Cao B, Hu M, Wang Z, Dong X (2011) Metabolic ability and gene characteristics of Arthrobacter sp. strain DNS10, the sole atrazine-degrading strain in a consortium isolated from black soil. Int Biodeterior Biodegrad 65:1140–1144. https://doi.org/10.1016/j.ibiod.2011.08.010
Zhang Y, Ge S, Jiang M, Jiang Z, Wang Z, Ma B (2014) Combined bioremediation of atrazine-contaminated soil by Pennisetum and Arthrobacter sp. strain DNS10. Environ Sci Pollut Res 21:6234–6238. https://doi.org/10.1007/s11356-013-2410-6
Zhang JJ, Gao S, Xu JY, Lu YC, Lu FF, Ma LY, Su XN, Yang H (2017) Degrading and phytoextracting atrazine residues in rice (Oryza sativa) and growth media intensified by a phase II mechanism modulator. Environ Sci Technol 51:11258–11268. https://doi.org/10.1021/acs.est.7b02346
Zhang B, Ni Y, Liu J, Yan T, Zhu X, Li QX, Hua R, Pan D, Wu X (2020) Bead-immobilized Pseudomonas stutzeri Y2 prolongs functions to degrade s-triazine herbicides in industrial wastewater and maize fields. Sci Total Environ 731:139183. https://doi.org/10.1016/j.scitotenv.2020.139183
Zhao X, Wang L, Du L, Yang J, Dong J, Ma F (2017) Optimization of culturing conditions for isolated: Arthrobacter sp. ZXY-2, an effective atrazine-degrading and salt-adaptive bacterium. RSC Adv 7:33177–33184. https://doi.org/10.1039/c7ra04661h
Zhao F, Li Y, Huang L, Gu Y, Zhang H, Zeng D, Tan H (2018) Individual and combined toxicity of atrazine, butachlor, halosulfuron-methyl and mesotrione on the microalga Selenastrum capricornutum. Ecotoxicol Environ Saf 148:969–975. https://doi.org/10.1016/j.ecoenv.2017.11.069
Zhu J, Fu L, Jin C, Meng Z, Yang N (2019) Study on the isolation of two atrazine-degrading bacteria and the development of a microbial agent. Microorganisms 7:80. https://doi.org/10.3390/microorganisms7030080
Acknowledgements
The authors want to thank CONICET, SECyT-UNRC; SECyT UNVM, MINCyT Córdoba. EA is a member of the research career from Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET) (Argentina). NU has a scholarship from CONICET.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Urseler, N., Bachetti, R., Morgante, C., Agostini, E. (2022). Bioremediation Strategies to Mitigate the Impact of Atrazine on the Environment: Recent Advances and Prospects. In: Naeem, M., Bremont, J.F.J., Ansari, A.A., Gill, S.S. (eds) Agrochemicals in Soil and Environment. Springer, Singapore. https://doi.org/10.1007/978-981-16-9310-6_21
Download citation
DOI: https://doi.org/10.1007/978-981-16-9310-6_21
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-16-9309-0
Online ISBN: 978-981-16-9310-6
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)