Abstract
The onset of nineteenth century along with anthropogenic pressure paved the way for global climatic variation which is a major factor for global undernourishment, malnutrition and endangered food security. The major upshot of climate change is abiotic stress like salinity, flood and drought that declines the agricultural productivity. Abiotic stress hampers the survival of the plants and restricts their growth and development. Each abiotic stress confers negative impact on plants by altering its physiology, morphology and metabolism. Production of reactive oxygen species during stress condition alters the structure and metabolic function in plants and restricts its growth. Drought is one of the serious threats to crop productivity among the abiotic stress that imposes multidimensional effects on plants. Drought alters physiology and anatomy of the plants and is the main reason for economic loss in terms of livestock and grain yield in both developed and developing countries. Plants adapt several resistance mechanisms to cope up with the drastic impact of stress. Main tolerance mechanisms are alteration in membrane structure, escaping the unfavourable conditions, activation of antioxidant defense system, production of compatible solutes for maintaining osmotic balance of the cell. Present manuscript focuses on the outcome of major abiotic stress in plants and their tolerance strategies against the variable environmental conditions.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
5.1 Introduction
Contemporary civilization and industrialization have led to global climate change whose consequences like flood, drought, high temperature and global warming are quite perceptible and hazardous for soil heath, microbial diversity and sustainable crop production. Climate fluctuation also threatens the vulnerable and endangered organisms in the ecosystem that are incompetent to endure such environmental changes in their habitat. Climate variability has immense impact on food security. It hits the underdeveloped regions of the world causing yield and economic losses. It has been estimated that 26% of total loss was in agriculture sector due to calamitous events from 2006 to 2016 (FAO 2018). Figure 5.1a, b illustrates the severity of disastrous events in the past years along with its impact on agricultural sector. There has been a dire inflation in the number of undernourished people across the world from 806 million in 2016 to 821 million in 2017 as an outcome of climate shock (FAO 2018). In view of the above facts, the foremost challenge is to provide food security to the projected global population of about 9.7 billion in 2050 under climate fluctuation and dwindling crop productivity.
Abiotic stress is an aftermath of climatologically variation that ceases plant growth, development and productivity below optimum levels. A surfeit or scant in energetic, chemical or physical condition in the nature paved the origin of abiotic stress in plants. The utmost abiotic stress comprises of drought, salinity, flooding and oxidative stress that abolishes metabolic equilibrium of plants thereby threatening food security (Fedoroff et al. 2010). Economic forfeiture in the developing countries in terms of crop and livestock production was discerned up to USD 96 billion from 2005 to 2015 with a 2.6% decrement in national agricultural value added growth due to occurrence of each climatic hazards (FAO 2018). An alarming upshot of climate shock is forecasted to decline agriculture productivity and influence five billion people by 2050 (UNESCO 2018; Watts 2018).
5.2 Abiotic Stress in Plants
5.2.1 Salt Stress
One of the most detrimental climate shocks is salinity stress which is prevalent in arid and semiarid regions in the world as depicted in Fig. 5.2. Approximately 33% of the world’s irrigated land and 20% of overall cultivated land are devastated by salinity stress (Shrivastava and Kumar 2015). It has been forecasted that by 2050 around 50% of arable land would be influenced by salinity (Jamil et al. 2011). In India seven million hectares of land is sheathed by saline soil (Patel and Dave 2011). An electrical conductivity of precisely 4 dSm−1 of saturation extract from root zone at 25 °C marks the saline soil (Jamil et al. 2011). Salinity damages the agricultural productivity as the major food crops are susceptible to it. A remarkable yield loss of about 65% in wheat was evident as an outcome of salinity stress (Ahmad et al. 2013). Yield reduction of about 55% in corn and cotton was observed in case of salinity in the range of about 8–10 dSm−1 and 18 dSm−1, respectively (Satir and Berberoglu 2016; Zörb et al. 2019).
Salt affected regions of the world. Adopted from FAO (2016)
5.2.2 Impact of Salinity Stress in Plants
Salinity stress devastates the plants by two ways: (a) Osmotic effect that diminishes plant potential for water uptake due to excess salt in soil and (b) ion excessive effect that occurs due to accumulation of excess salt in the leaf tissues (Greenway and Munns 1980). Salinity amends the major physiological processes like photosynthesis, transpiration, nutrient regulation and osmotic balance as an outcome and affects plant growth and yield. Influence of salinity stress in plants has been illustrated in Table 5.1.
5.2.3 Plant Tolerance Strategies against Salinity Stress
Resistance strategies of plants to salinity rely on environmental conditions and plant species. Plants mainly endure salt stress by three ways:
-
(a)
Endurance to osmotic stress
-
(b)
Efflux of sodium from leaf blades
-
(c)
Tissue tolerance
5.2.3.1 Osmotic Stress Endurance
Osmotic stress is marked by generation of osmolytes like glycine betaine, proline, polyols, etc. to maintain the osmotic balance. Osmotic tolerance also aids in balancing stomatal conductance and leaf expansion during salinity (Rajendran et al. 2009). Osmolytes stabilize the membrane integrity, proteins and pigments in the cell and also alleviate reactive oxygen species (Krasensky and Jonak 2012; Gupta and Huang 2014). It was evident that proline accumulation improved the salinity tolerance in maize (Tang et al. 2019). Likewise, trehalose accumulation also assisted in boosting salinity endurance in wheat (Sadak 2019).
5.2.3.2 Sodium Ion Efflux from Leaf Blades
Outcome of salinity stress in plants comprises ion toxicity due to sodium ion (Na+) accumulation inside the cell. This can be overcome by down regulating the expression of ionic channels and Na+ transporters (Munns and Tester 2008; Rajendran et al. 2009). An antiporter (AtNHX1) confined in tonoplast of Arabidopsis maintains Na+ balance in the cell under salinity condition by directing the influx of Na+ ions from cytosol to vacuole (Apse et al. 1999). Likewise NAX1gene in durum wheat impedes Na+ translocation in the shoots and leaf sheath from roots, whereas NAX2 gene confers high potassium ion (K+)translocation in leaf (Flagella et al. 2006). Sodium exclusion theory for salt resistance has been validated in rice (Ren et al. 2005) and durum wheat (James et al. 2006). It was studied that potassium transporter (PpHKT1) gene from rootstock of almond also enhanced salt tolerance in transgenic Arabidopsis by balancing ion homeostasis (Kaundal et al. 2019).
5.2.3.3 Tissue Tolerance
For hindering the senescence of leaves and enhancing its survival rate, tissue tolerance is a salient attribute during stress. Generation of compatible solutes that balances the osmotic homeostasis in the cell and antioxidant defense system that scavenges the reactive oxygen species under stress condition is an attribute of tolerance of plants to stress condition (Hasegawa et al. 2000; Zhu 2001; Sakamoto and Murata 2002). Tissue resistance also incites the ion compartmentalization in the cell to diminish toxicity of ion (Munns and Tester 2008). A notable rise in osmolyte production like proline up to 14.87 μmol g−1 DW and an increment in antioxidants like ascorbate peroxidase up to 77%, catalase up to 15% and glutathione reductase up to 138% were recorded in chickpea as a tolerance response to salinity stress (Arefian et al. 2018).
5.3 Flooding Stress
Prolonged submergence and soil waterlogging result in hypoxic condition that constitutes flood stress in plants. One of the key limitations to crop productivity in high rainfall areas of the world is flood stress. Climate change promotes submergence to be more recurring and drastic (Pachauri et al. 2014). Approximately 10% of irrigated land in India, China, Russia and 16% of cultivable land in the USA are damaged by waterlogging (FAO 2015). Figure 5.3 explains the ubiquity of flood hazard in the world. According to statistical approximates of food and agriculture organization of United Nations, flood stress upshots 17% of total loss and damage in agriculture (FAO 2018).
Global flood hazard prevalence. Adopted from SEDAC (n.d.)
It was discerned that annually 20–50% production loss in wheat occurred under waterlogging condition (Hossain and Uddin 2011). Overall 15% of maize production in South Asia is damaged by floods. India accounts for 25–30% maize production loss every year on account of flood stress (Bailey-Serres et al. 2012).
5.3.1 Outcome of Flood Stress in Plants
The prime consequence of flood stress in plant comprises anoxia or oxygen limitation. Necrosis, senescence, low nitrogen fixation and ultimately plant death are the consequences of flood stress (Hasanuzzaman et al. 2012). Overall effects of flood stress in plants are outlined in Table 5.2.
5.3.2 Adaptation of Plants to Flood Stress
5.3.2.1 Morphological Alterations
For escaping anoxic conditions under flood stress, plants showcase morphological adaptations. Submerged leaves have low stomatal counts and are often dissected and filamentous (Sculthorpe 1967). Vigorous adventitious root growth is one of the prominent adaptation as evident in Sesbania javanica (Jackson 2006) and Chrysanthemum (Yin et al. 2009). Decrement in root hairs and diameter, rapid shoot elongation are some of the evident variations (Jackson 2008). Under submergence, formation of leaf gas film improves carbohydrate supply, generation of adventitious roots and aeration of roots. In rice, resistance to radial oxygen loss is enhanced by development of effective barriers. This aids in shielding the plants from toxins generated from anoxic environment and boosts root tip aeration under flood stress (Kirk et al. 2014; Yamauchi et al. 2018).
5.3.2.2 Anatomical Alterations
To hinder the loss of radial oxygen from submerged roots, plants show formation of lateral diffusion barrier (Sauter 2013). Lenticels development is also evident in flood affected plants as this aids in oxygen diffusion in downward direction (Parelle et al. 2006). Aerenchyma formation is one of the unique characters of plants under flood or submerged condition as these gas spaces allow oxygen transport from roots to shoots (Shiono et al. 2008). Exodermis thickness was improved by 23.70% compared to control in Garcinia brasiliensis under flood state in addition to increment in root xylem vessels and phloem width (de Souza et al. 2013). In flood tolerant barley cultivar (TX9425 and Yerong), more well developed intercellular spaces in leaves and mesophyll cells were observed that enhanced photosynthesis due to quick gaseous exchange Zhang et al. 2016). In rice seedlings, rapid coleoptile elongation during germination is a marker for submergence tolerance that facilitates aeration of developing embryo (Guru and Dwivedi 2018).
5.3.2.3 Biochemical Alterations
Under flood stress, switch in the metabolism of plants from aerobic to anaerobic condition with high activity of alcohol dehydrogenase, a key enzyme in anaerobic fermentation is observed (Sairam et al. 2008). Proline content in Casuarina was high in tolerant cultivar as it acts as a signal molecule, an osmolyte and showcases antioxidant defense response under stress condition (Bajpai and Chandra 2015). Enhanced activity of alpha aminobutyric acid has been evident in soybean nodules in flooded conditions as this metabolite balances intercellular pH, acts as nitrogen reservoir under flood condition and also serves as antioxidant (Souza et al. 2016). Elevated activity of enzymes like phosphoenolpyruvate carboxylase, glutamate dehydrogenase and alcohol dehydrogenase was observed in flooded nodules of Sesbania herbacea (Krishnan et al. 2019).
5.4 Heavy Metal Stress
One of the major aftermaths of industrial revolution in the twenty-first century is heavy metal pollution in the environment. Heavy metals are potentially noxious elements and present in trace amount in soil. They are comprised of metalloids, lanthanoides and transition metals with atomic density ˃6 g cm−3except selenium, boron and arsenic (Park et al. 2011). The root cause of heavy metals in the environment encompasses usage of pesticides and fertilizers, combustion of fuels, mining, sewage sludge, oil spills, etc. Heavy metals are diverse in their functioning. Metals like iron, zinc, copper, nickel, manganese, chromium impart proper functioning of plant’s metabolic processes contrarily lead, arsenic, mercury have negative effect on plant growth and productivity (Pierart et al. 2015). These noxious elements enter the food chain and attack human, animal and soil health (Augustsson et al. 2015).
Uptake of heavy metal by plants interrupts the chief metabolic processes like photosynthesis, nitrogen fixation and nutrients uptake and results in biomass and yield reduction and also death of the plants (Buendía-González et al. 2010). In China, about 20,000,000 acres of farmland is polluted by heavy metal with an annual crop loss of 1,000,000 million tons per year (Wu et al. 2010). About 12% of world’s agricultural land is estimated to be affected by heavy metal pollution (Dziubanek et al. 2015). One of the most detrimental heavy metal for human health is lead. Lead exposure resulted in loss in economic productivity of about $977 billion annually in low to middle-income count (Attina and Trasande 2013). Statistical estimates of Institute for Health Metrics and Evaluation disclosed that lead exposure caused nearly 0.5 million deaths and 9.3 million life years lost among adults of 15 years and older (Landrigan et al. 2018).
5.4.1 Outcome of heavy metal stress in plants
Heavy metal stress deteriorates soil and plant health. Metal toxicity alters the morphology and physiology of plants at different growth stage as a consequence there is decline in crop productivity. Table 5.3 outlines the impact of heavy metal stress in plants.
5.4.2 Plant tolerance to heavy metal stress
5.4.2.1 Antioxidant enzymes
To escape heavy metal toxicity plants evolve many alterations. Exclusion of metal ions from the cell or binding with ligand prevents entry of metal ions to the roots is the first line of defense mechanism (Zeng et al. 2011). Metal ions also make complexes with carboxyl group of pectin in the cell wall thereby restricting its entry in the cell (Jiang and Liu 2010). Secondary response for metal detoxification is generation of antioxidant enzymes like superoxide dismutase, catalase and peroxidase. Compounds like glutathione, proline, cysteine, ascorbic acid also serve as scavengers of reactive oxygen species (Shahid et al. 2012; Pourrut et al. 2013).
Glutathione is a tripeptide antioxidant with thiol group and forms mercaptide bond with metals due to its nucleophilic thiol group and its reduced form also scavenges peroxide radicals. Glutathione conjugates with compounds of heavy metals that are translocated to vacuole to protect cell from its harmful effects (Klein et al. 2006; Yazaki 2006). It was also studied that in response to cadmium toxicity there is a modulation in the activity of antioxidants like guaiacol peroxidase, ascorbate peroxidase and catalase in tolerant plants (Alves et al. 2017; Borges et al. 2018). In Brassica juncea L. such kind of high efficiency antioxidative defense system was evident during caesium toxicity (Lai and Luo 2019).
5.4.2.2 Metallothioneins and Phytochelatins
Vacuolar sequestration of heavy metals is an important response of plants to metal stress. Complex formation of metal ion with ligand reduces the toxicity of heavy metals. Metallothioneins and phytochelatins rich in cysteine are natural metal chelators in plants that reduce metal toxicity by forming mercaptide bonds with metals and sequester them to vacuoles (Gupta et al. 2010; Jiang and Liu 2010). In Brassica napus, cadmium toxicity results in strong expression of MT4 metallothionein in germinating seeds as a defense mechanism (Mierek-Adamska et al. 2018). Likewise in Oryza sativa, phytochelatin synthase genes (OsPCS5 and OsPCS15) were profoundly induced under cadmium stress as tolerance mechanism. These genes encode phytochelatins that bind heavy metals in cytosol and detoxify them in the vacuoles (Park et al. 2019).
5.5 Low Temperature Stress
Chilling or low temperature stress amends the plant morphology, physiology and metabolism. Occurrence of chilling stress at temperature 0 °C to 15 °C whereas freezing stress at ambient temperature for ice crystal formation causes cellular injury and osmotic dehydration in the cell (Bhatla 2018).
5.5.1 Impact of low temperature stress in plants
Chilling in plants is advantageous for breaking seed dormancy, for vernalization and cold acclimation. In contrast, chilling in susceptible crops has many aftermaths as listed in Table 5.4.
5.5.2 Adaptations to low temperature stress
5.5.2.1 Cold acclimation
Increment in phospholipid proportion and decrement in ceramide and sugar containing lipids in plasma membrane results in reduction in expansion induced lysis. Expression of sugar producing enzymes like acid invertase, galactinol synthase and sucrose synthase to improve membrane stability is induced in response to cold acclimation (Turhan and Ergin 2012). Accumulation of compatible solutes like hydroxyl proline and arabinose in the cell maintains osmotic balance during cold acclimation (Bhatla 2018). Expression of hydrophilic and LEA (late embryogenesis abundance protein) proteins declines the denaturation of proteins under cold and drought stress. In Brassica napus, dehydrin proteins, a class of LEA proteins was highly expressed under cold stress in tolerant cultivar. It is used as biomarker for selection of cold tolerant lines (Maryan et al. 2019).
5.5.2.2 Modification in gene expression
Expression of cold regulated genes (COR) confers cold tolerance in plants by stabilizing the plasma membrane during low temperature stress. Hydrophilic proteins encoded by COR genes expressed during low temperature stress are involved in the production of anthocyanin and play indirect role in cold acclimatization. Extracellular production of antifreeze proteins (AFP) prevents ice crystal formation during low temperature stress. Molecular chaperons like HSP90 restrict denaturation of proteins during chilling stress. Other proteins like MAP (mitogen activated protein) kinase and calmodulin related proteins are encoded by COR genes and impart role in cold temperature signalling in plants (Bhatla 2018). A chromatin remodelling factor PICKLE (PKL) was found responsible for cold stress tolerance in Arabidopsis. It also modulates expression of other cold responsive genes like RD29A and COR15A (Zhao et al. 2019). In Brassica rapa L., genes such as temperature-induced lipocalin-1, zinc finger protein ZAT12, dehydrin ERD10 and adenosylhomocysteinase 2 were analysed and found to be involved in cold stress tolerance (Ma et al. 2019).
5.6 Drought Stress
One of the most disastrous outcomes of climate change is drought that restricts plant growth and development in both developed and developing countries. The utmost driver of undernourishment and hunger is geographical and global drought that declines agricultural productivity (FAO 2017).US, a technologically advanced country encounters an annual loss of $ 6 billion in agricultural productivity due to drought and such losses are more severe in underdeveloped and developing countries (CIA 2017). Around 20% of world’s cultivable land is hit by drought and more than 80% damage in livestock and agricultural production is because of drought stress (FAO 2018). Major drought exposed regions in the world have been delineated in Fig. 5.4.
Global map of drought affected regions. Adapted from Carrao et al. (2016)
Drought is a climatic variation with perpetuate periods of rainfall scarcity that results in hunger, malnutrition, deficit food productivity and ultimately food insecurity. Plants encounter severe drought when water loss via transpiration is high enough or when roots are deficient in water supply (Anjum et al. 2011). A severe drought can threaten the national food availability and results in pervasiveness of undernourishment. Approximate global yield loss of 21% in wheat and 40% in maize was observed from 1980 to 2015 (Daryanto et al. 2016). Table 5.5 illustrates susceptible and resistant crops under major abiotic stress.
Drought alters photosynthesis, water balance, nutrient acquisition, oxidative metabolism and assimilates partitioning in plants. This modification in plants depends on species, growth stage of the plants and environmental conditions. Apparent symptoms of drought in plants are depicted in Fig. 5.5. Reduction in grain filling rate, harvest index and solar use efficiency are some key drivers responsible for diminished yield under drought stress. Table 5.6 describes deleterious impact of drought stress in plants ranging from morphological, biochemical to molecular level and is discernible at every phenological stage of crop growth at any period of time (Farooq et al. 2009; Deepak et al. 2019).
5.6.1 Outcome of Drought Stress on Morphological Attributes
The foremost impact of drought stress on morphology of plant is restricted seed germination and seedling growth (Farooq et al. 2009). Drought negatively alters seed vigour, seedling growth and also results in poor seed quality as reported in Medicago sativa (Zhang et al. 2019a, b), Oryza sativa (Bhavyasree et al. 2019), Glycine max (Reddy et al. 2019), Zea mays and Sorghum (Queiroz et al. 2019).
Drought impedes mitosis and cell enlargement because of restricted water flow from xylem to neighbouring cells (Hussain et al. 2008). Reduction in plant size and biomass is one of the major morphological alterations in plants during drought stress. Fifty percent reduction in biomass in wheat was observed as compared to control under drought stress (Paul et al. 2019). Dry weight and fresh weight of root and shoot as well as root shoot ratio tend to cut down in drought induced Brassica napus L. (Khan et al. 2019).
Decrement in stomatal aperture, leaf rolling, cutinization of leaf surfaces are some observable drought stress induced morphological parameters (Hussain et al. 2008). Drought hampers the leaf size, grain yield in crops, for instance, yield parameters like panicle length, seed setting rate and grain weight declined in Oryza sativa L. under drought (Wei et al. 2017). Likewise in wheat, 62% of grain loss was observed in drought condition compared to well-watered conditions (Paul et al. 2019).
5.6.2 Physiological and Biochemical Alterations Under Drought
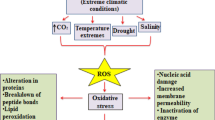
Physiological attributes like transpiration rate, stomatal conduction, leaf water potential and relative water content are adversely affected by drought. Low water potential, transpiration rate, relative water content and enhanced leaf temperature were observed in Oryza sativa under drought (Fahad et al. 2017). Drought stress induces the formation of reactive oxygen species (ROS) along with enhanced generation of osmolytes like proline, glycine betaine. Production of ROS leads to oxidative stress that ultimately results in cell death in plants (Silva et al. 2019).
Another important trait influenced by drought is photosynthesis. Drought stress induces reduction in leaf expansion, damages the photosynthetic apparatus and activity of photosynthetic enzymes. Reduction in phosphorylation and disruption in ATP synthesis are key drivers for impaired photosynthesis under drought (Fahad et al. 2017). Reduction in thylakoid membrane proteins and PSII activity under PEG induced drought stress have been evident in many studies (Wang et al. 2019).
Activity of rubisco, a motor enzyme of photosynthesis is declined drastically under drought. Reduction in amount of substrate, i.e. ribulose 1, 5 bisphosphate for carboxylation, modification in ultrastructure of rubisco and chloroplast and acidification of chloroplast stroma are the driving factors for decrement in rubisco activity. Other enzymes like fructose-1, 6-bisphosphatase, NADP-glyceraldehyde phosphate dehydrogenase, phosphoribulokinase, phosphoenol pyruvate carboxylase and pyruvate orthophosphate dikinase also decline under drought stress (Farooq et al. 2009).
Drought restricts nutrient movement from soil to plants. Under drought, root growth and proliferation are hindered that limit nutrient translocation in plants. Limited flow of oxygen, carbon and nitrogen assimilation in root nodules impedes nitrogen fixing ability of legumes under drought (Ladrera et al. 2007). Nitrate reduction in leaves was limited by declined photosynthesis, which supplies reducing power, generated during photosynthetic via electron transport for the process of nitrate reduction (Chen et al. 2018).
5.6.3 Adaptation of Plants to Drought Stress
5.6.3.1 Escape
Drought escape is characterized by short life cycle that enables plants to flower early before onset of drought. Growing season is dependent on environmental variation and plant genotype. Drought escape is enhanced when development of plant synergizes with duration when soil moisture is available. Although drought escape helps in plants survival during stress yet there is a decrement in the yield (Farooq et al. 2009). High grain yield in Lens culinaris was recorded under early drought as a result of drought escape. Maximum leaf area, stomatal conductance, high stomatal density, early flowering with short life cycle were also observed as an outcome of drought escape strategy in lentil (Sanchez-Gomez et al. 2019).
5.6.3.2 Avoidance
Avoidance is marked by maintaining the water potential of plants during drought. It is also characterized by reduced water deprivation through transpiration and balancing the water uptake from soil. For efficient water uptake, a deep and extensive root system is required along with thick cuticle whose formation requires high input of energy that ultimately results in low yield. Therefore, plants with avoidance strategies are generally of compact size (Lisar et al. 2012). In Sorghum, aquaporin genes PIP2;3 were strongly expressed in roots under drought compared to roots under watered conditions. Aquaporins are the membrane proteins that allow water and solute passage through the membrane into cell and their expression under drought is an adaptive strategy (Schulze et al. 2019).
5.6.3.3 Tolerance
Tolerance to drought stress is perceptible by limited area and number of leaves during water deficit condition. Plants show traits like formation of trichomes and hairy leaves which reduce leaf temperature during drought. Root architecture like root density, root proliferation, root growth rate are some key alterations in plants during drought. Reduction in stomata number, increment in root shoot ratio, accumulation of osmolytes and induction of antioxidant defense system are other drought tolerance traits in plants (Sapeta et al. 2013). In sorghum, leaf rolling, leaf waxiness, stay green trait, root architecture, abscisic acid metabolism, ion transporter, transpiration efficiency and osmotic adjustments are drought tolerant traits that enable it to survive under drought stress (Badigannavar et al. 2018).
5.7 Conclusion
Major constraint for sustainable agricultural productivity and global food security is abiotic stress which is an outcome of global climate change. Extreme weather conditions attack morphological, physiological and biochemical attributes of the plants and threat their survival in the environment. Multiple abiotic stresses like salinity, flood, heavy metal and drought impact the crop yield. Drought is the most devastating abiotic stress that declines the global crop productivity. Drought hampers seed germination, reduces the stomatal frequency and diminishes leaf area and water potential. At physiological level, drought restricts photosynthesis, thylakoid structure and enzyme production. All these modifications alter the metabolism and limit growth and development of plants. Despite such abiotic constraints, plants have inherent tolerance mechanisms that enable them to cope up and survive under extreme conditions. Activation of antioxidant defense system, changes in the membrane lipid composition and enzyme production as well as morphological alterations in plants are some of the tolerance traits in plants. In spite of such adaptive strategies, improvements in the tolerance of plants against the extreme calamities are required. Strategies like alteration in expression of stress responsive genes and transcription factors are potential candidates to develop stress tolerant crops. More emphases should be there on molecular research for exploiting the stress tolerance traits and minimizing the aftermath of stress in plants.
References
Ahmad M, Shahzad A, Iqbal M, Asif M, Hirani AH (2013) Morphological and molecular genetic variation in wheat for salinity tolerance at germination and early seedling stage. Aust J Crop Sci 7:66–74
Alamri SA, Siddiqui MH, Al-Khaishany MY, Nasir Khan M, Ali HM, Alaraidh IA, Alsahli AA, Al-Rabiah H, Mateen M (2018) Ascorbic acid improves the tolerance of wheat plants to lead toxicity. J Plant Interact 13:409–419
Alves LR, Monteiro CC, Carvalho RF, Ribeiro PC, Tezotto T, Azevedo RA, Gratão PL (2017) Cadmium stress related to root-to-shoot communication depends on ethylene and auxin in tomato plants. Environ Exp Bot 134:102–115
Amjad M, Ameen N, Murtaza B, Imran M, Shahid M, Abbas G, Naeem MA, Jacobsen SE (2019) Comparative physiological and biochemical evaluation of salt and nickel tolerance mechanisms in two contrasting tomato genotypes. Physiol Plant 168:27–37
Anjum SA, Wang LC, Farooq M, Hussain M, Xue LL, Zou CM (2011) Brassinolide application improves the drought tolerance in maize through modulation of enzymatic antioxidants and leaf gas exchange. J Agron Crop Sci 197:177–185
Apse MP, Aharon GS, Snedden WA, Blumwald E (1999) Salt tolerance conferred by overexpression of a vacuolar Na+/H+ antiport in Arabidopsis. Science 285:1256–1258
Arefian M, Vessal S, Shafaroudi SM, Bagheri A (2018) Comparative analysis of the reaction to salinity of different chickpea (Cicer aretinum L.) genotypes: a biochemical, enzymatic and transcriptional study. J. Plant Growth Regul 37:391–402
Asch F, Dingkuhn M, Dörffling K, Miezan K (2000) Leaf K/Na ratio predicts salinity induced yield loss in irrigated rice. Euphytica 113:109–118
Ashraf M (2009) Biotechnological approach of improving plant salt tolerance using antioxidants as markers. Biotechnol Adv 27:84–93
Attina TM, Trasande L (2013) Economic costs of childhood lead exposure in low-and middle-income countries. Environ Health Perspect 121:1097–1102
Augustsson AL, Uddh-Söderberg TE, Hogmalm KJ, Filipsson ME (2015) Metal uptake by homegrown vegetables–the relative importance in human health risk assessments at contaminated sites. Environ Res 138:181–190
Badigannavar A, Teme N, de Oliveira AC, Li G, Vaksmann M, Viana VE, Ganapathi TR, Sarsu F (2018) Physiological, genetic and molecular basis of drought resilience in sorghum [Sorghum bicolor (L.) Moench]. Indian J Plant Physiol 23(4):670–688
Bailey-Serres J, Lee SC, Brinton E (2012) Waterproofing crops: effective flooding survival strategies. Plant Physiol 160:1698–1709
Bajpai S, Chandra R (2015) Effect of waterlogging stress on growth characteristics and sod gene expression in sugarcane. Int J Sci Res Publ 5:1–8
Bakhshandeh S, Corneo PE, Yin L, Dijkstra FA (2019) Drought and heat stress reduce yield and alter carbon rhizodeposition of different wheat genotypes. J Agron Crop Sci 205:157–167
Banavath JN, Chakradhar T, Pandit V, Konduru S, Guduru KK, Akila CS, Podha S, Puli CO (2018) Stress inducible overexpression of AtHDG11 leads to improved drought and salt stress tolerance in peanut (Arachis hypogaea L.). Front Chem 6:1–24
Bar Y, Apelbaum A, Kafkafi U, Goren R (1997) Relationship between chloride and nitrate and its effect on growth and mineral composition of avocado and citrus plants. J Plant Nutr 20:715–731
Barber SA (1995) Soil nutrient bioavailability: a mechanistic approach, 2nd edn. New York, Wiley
Batra NG, Kumari N, Sharma V (2018) Chlorophyll fluorescence based assessment of low temperature stress in different varieties of Vigna radiata (L.). Int J Plant Res 31:146–155
Bhatla SC (2018) Abiotic stress. In: Plant physiology, development and metabolism. Springer, Singapore, pp 969–1028
Bhavyasree RK, Joel AJ, Jeyaprakash P, Raveendran M (2019) The effect of induced drought stress on seedling vigour and antioxidant enzymes in wild and cultivated Oryza species. Int J Chem Stud 7:1078–1081
Bilska-Kos A, Solecka D, Dziewulska A, Ochodzki P, Jończyk M, Bilski H, Sowiński P (2017) Low temperature caused modifications in the arrangement of cell wall pectins due to changes of osmotic potential of cells of maize leaves (Zea mays L.). Protoplasma 254:713–724
Borges KLR, Salvato F, Alcântara BK, Nalin RS, Piotto FA, Azevedo RA (2018) Temporal dynamic responses of roots in contrasting tomato genotypes to cadmium tolerance. Ecotoxicology 27:245–258
Buendía-González L, Orozco-Villafuerte J, Cruz-Sosa F, Barrera-Díaz CE, Vernon-Carter EJ (2010) Prosopis laevigata a potential chromium (VI) and cadmium (II) hyperaccumulator desert plant. Bioresour Technol 101:5862–5867
Camejo D, Jiménez A, Alarcón JJ, Torres W, Gómez JM, Sevilla F (2006) Changes in photosynthetic parameters and antioxidant activities following heat-shock treatment in tomato plants. Funct Plant Biol 33:177–187
Carrao H, Naumann G, Barbosa P (2016) Mapping global patterns of drought risk: an empirical framework based on sub-national estimates of hazard, exposure and vulnerability. Glob Environ Chang 39:108–124
Carter AY, Ottman MJ, Curlango-Rivera G, Huskey DA, D’Agostini BA, Hawes MC (2019) Drought-tolerant barley: II. Root tip characteristics in emerging roots. Agronomy 9:1–10
Carvalho MEA, Piotto FA, Franco MR, Borges KLR, Gaziola SA, Castro PRC, Azevedo RA (2018) Cadmium toxicity degree on tomato development is associated with disbalances in B and Mn status at early stages of plant exposure. Ecotoxicology 27:1293–1302
Chatterjee C, Dube BK, Sinha P, Srivastava P (2004) Detrimental effects of lead phytotoxicity on growth, yield, and metabolism of rice. Commun Soil Sci Plant Anal 35:255–265
Chen H, Zhang Q, Lu Z, Xu F (2018) Accumulation of ammonium and reactive oxygen mediated drought-induced rice growth inhibition by disturbed nitrogen metabolism and photosynthesis. Plant Soil 431:107–117
Chiang PN, Chiu CY, Wang MK, Chen BT (2011) Low-molecular-weight organic acids exuded by Millet (Setaria italica (L.) Beauv.) roots and their effect on the remediation of cadmium-contaminated soil. Soil Sci 176:33–38
Chigbo C, Batty L (2013) Effect of combined pollution of chromium and benzo (a) pyrene on seed growth of Lolium perenne. Chemosphere 90:164–169
CIA (2017) The world factbook. https://www.cia.gov/library/publications/the-world-factbook/. Accessed 23 Sept 2019
Daryanto S, Wang L, Jacinthe PA (2016) Global synthesis of drought effects on maize and wheat production. PLoS One 11:1–15
Dawood T, Rieu I, Wolters-Arts M, Derksen EB, Mariani C, Visser EJ (2014) Rapid flooding-induced adventitious root development from preformed primordia in Solanum dulcamara. AoB Plants 6:1–13
Deepak SB, Thakur A, Singh S, Bakshi M, Bansal S (2019) Changes in crop physiology under drought stress: a review. J Pharmacogn Phytochem 8:1251–1253
de Souza TC, dos Santos SE, Dousseau S, de Castro EM, Magalhães PC (2013) Seedlings of Garcinia brasiliensis (Clusiaceae) subjected to root flooding: physiological, morphoanatomical, and antioxidant responses to the stress. Aquat Bot 111:43–49
Devi SR, Prasad MNV (1999) Membrane lipid alterations in heavy metal exposed plants. In: Prasad MNV (ed) Heavy metal stress in plants. Springer, Berlin, Heidelberg, pp 99–116
Dhawan SS, Mishra A, Gupta P, Bahl JR, Bansal RP (2018) Phylogentic relationship of cold tolerant Mentha arvensis variety ‘CIM Kranti’ with some released varieties as assessed through physiological and molecular analysis. J Appl Res Med Aromat Plants 10:67–74
Din J, Khan SU, Ali I, Gurmani AR (2011) Physiological and agronomic response of canola varieties to drought stress. J Anim Plant Sci 21:78–82
Dolatabadi N, Toorchi M, Valizadeh M, Bandehagh A (2019) The proteome response of salt-sensitive rapeseed (Brassica napus L.) genotype to salt stress. Not Bot Horti Agrobot Cluj Napoca 47:17–23
Dwivedi SK, Kumar S, Bhakta N, Srivastava AK, Mishra JS, Kumar V, Kumara BH, Bhatt BP, Singh S (2018) Physiological mechanism and nutrient management strategies for flood tolerance in rice grown in lowland flood prone ecosystem. J Crop Sci Biotechnol 21:321–331
Dziubanek G, Piekut A, Rusin M, Baranowska R, Hajok I (2015) Contamination of food crops grown on soils with elevated heavy metals content. Ecotoxicol Environ Saf 118:183–189
Ezaki B, Nagao E, Yamamoto Y, Nakashima S, Enomoto T (2008) Wild plants, Andropogon virginicus L. and Miscanthus sinensis Anders, are tolerant to multiple stresses including aluminum, heavy metals and oxidative stresses. Plant Cell Rep 27:951–961
Ezin V, Pena RDL, Ahanchede A (2010) Flooding tolerance of tomato genotypes during vegetative and reproductive stages. Braz J Plant Physiol 22:131–142
Fahad S, Bajwa AA, Nazir U, Anjum SA, Farooq A, Zohaib A, Sadia S, Nasim W, Adkins S, Saud S, Ihsan MZ (2017) Crop production under drought and heat stress: plant responses and management options. Front Plant Sci 8:1–16
FAO (2015) Food and Agriculture Organization of the United Nations. http://www.fao.org/3/a-bc600e.pdf. Accessed 1 Oct 2016
FAO (2016) Food and Agriculture Organization of the United Nations. http://www.fao.org/3/a-i6471e.pdf. Accessed 1 Oct 2017
FAO (2017) How close we are to zero Hunger. http://www.fao.org/state-of-food-securitynutrition/en/geos/kz.html. Accessed 1 Mar 2019
FAO (2018) The state of food security and nutrition in the world. http://www.fao.org/state-of-food-security-nutrition/en/. Accessed 15 Sept 2019
Farahbakhsh H, Pasandi Pour A, Reiahi N (2017) Physiological response of henna (Lawsonia inermise L.) to salicylic acid and salinity. Plant Prod Sci 20:237–247
Farooq M, Wahid A, Kobayashi N, Fujita D, Basra SMA (2009) Plant drought stress: effects, mechanisms and management. In: Lichtfouse E, Navarrete M, Debaeke P, Véronique S, Alberola C (eds) Sustainable agriculture. Springer, Dordrecht, pp 153–188
Fedoroff NV, Battisti DS, Beachy RN, Cooper PJ, Fischhoff DA, Hodges CN, Knauf VC, Lobell D, Mazur BJ, Molden D, Reynolds MP (2010) Radically rethinking agriculture for the 21st century. Science 327:833–834
Flagella Z, Trono D, Pompa M, Di Fonzo N, Pastore D (2006) Seawater stress applied at germination affects mitochondrial function in durum wheat (Triticum durum) early seedlings. Funct Plant Biol 33:357–366
Ghoulam C, Foursy A, Fares K (2002) Effects of salt stress on growth, inorganic ions and proline accumulation in relation to osmotic adjustment in five sugar beet cultivars. Environ Exp Bot 47:39–50
Gomes-Filho E, Lima CRFM, Costa JH, da Silva ACM, Lima MDGS, de Lacerda CF, Prisco JT (2008) Cowpea ribonuclease: properties and effect of NaCl-salinity on its activation during seed germination and seedling establishment. Plant Cell Rep 27:147–157
Gonzalez JA, Gallardo M, Hilal M, Rosa M, Prado FE (2009) Physiological responses of quinoa (Chenopodium quinoa Willd.) to drought and waterlogging stresses: dry matter partitioning. Bot Stud 50:35–42
Greenway H, Munns R (1980) Mechanisms of salt tolerance in nonhalophytes. Annu Rev Plant Physiol 31:149–190
Grzesiak S, Hura T, Grzesiak MT, Pieńkowski S (1999) The impact of limited soil moisture and waterlogging stress conditions on morphological and anatomical root traits in maize (Zea mays L.) hybrids of different drought tolerance. Acta Physiol Plant 21:305–315
Guala SD, Vega FA, Covelo EF (2010) The dynamics of heavy metals in plant–soil interactions. Ecol Model 221:1148–1152
Gupta B, Huang B (2014) Mechanism of salinity tolerance in plants: physiological, biochemical, and molecular characterization. Int J Genom 2014:1–18
Gupta DK, Huang HG, Yang XE, Razafindrabe BHN, Inouhe M (2010) The detoxification of lead in Sedum alfredii H. is not related to phytochelatins but the glutathione. J Hazard Mater 177:437–444
Guru A, Dwivedi P (2018) Physiological, biochemical and molecular mechanism of submergence tolerance in rice (Oryza sativa L.). J Pharmacogn Phytochem 7:1116–1121
Han Y, Yin S, Huang L, Wu X, Zeng J, Liu X, Qiu L, Munns R, Chen ZH, Zhang G (2018) A sodium transporter HvHKT1; 1 confers salt tolerance in barley via regulating tissue and cell ion homeostasis. Plant Cell Physiol 59:1976–1989
Han Y, Yang H, Wu M, Yi H (2019) Enhanced drought tolerance of foxtail millet seedlings by sulfur dioxide fumigation. Ecotoxicol Environ Saf 178:9–16
Hasanuzzaman M, Hossain MA, da Silva JAT, Fujita M (2012) Plant response and tolerance to abiotic oxidative stress: antioxidant defense is a key factor. In: Tuteja N, Gill SS (eds) Crop stress and its management: perspectives and strategies. Springer, Dordrecht, pp 261–315
Hasegawa PM, Bressan RA, Zhu JK, Bohnert HJ (2000) Plant cellular and molecular responses to high salinity. Annu Rev Plant Biol 51:463–499
Hossain MA, Uddin SN (2011) Mechanisms of waterlogging tolerance in wheat: morphological and metabolic adaptations under hypoxia or anoxia. Aust J Crop Sci 5:1094–1101
Hu Y, Schmidhalter U (1997) Interactive effects of salinity and macronutrient level on wheat. II. Composition. J Plant Nutr 20:1169–1182
Hussain M, Malik MA, Farooq M, Ashraf MY, Cheema MA (2008) Improving drought tolerance by exogenous application of glycinebetaine and salicylic acid in sunflower. J Agron Crop Sci 194:193–199
Jackson MB (2006) Plant survival in wet environments: resilience and escape mediated by shoot systems. In: Bobbink R, Beltman B, Verhoeven JTA, Whigham DE (eds) Wetlands: functioning, biodiversity conservation, and restoration. Springer, Berlin, Heidelberg, pp 15–36
Jackson MB (2008) Ethylene-promoted elongation: an adaptation to submergence stress. Ann Bot 101:229–248
Jaleel CA, Manivannan P, Wahid A, Farooq M, Al-Juburi HJ, Somasundaram R, Panneerselvam R (2009) Drought stress in plants: a review on morphological characteristics and pigments composition. Int J Agric Biol 11:100–105
James RA, Davenport RJ, Munns R (2006) Physiological characterization of two genes for Na+ exclusion in durum wheat, Nax1 and Nax2. Plant Physiol 142:1537–1547
Jamil A, Riaz S, Ashraf M, Foolad MR (2011) Gene expression profiling of plants under salt stress. Crit Rev Plant Sci 30:435–458
Jeschke WD (1984) K+-Na+ exchange at cellular membranes, intracellular compartmentation of cations, and salt tolerance. In: Staples RC, Toenniessen GH (eds) Salinity tolerance in plants strategies for crop improvement. Wiley, Toronto, pp 37–66
Jiang W, Liu D (2010) Pb-induced cellular defense system in the root meristematic cells of Allium sativum L. BMC Plant Biol 10:40–48
Kalai T, Khamassi K, Teixeira da Silva JA, Gouia H, Bettaieb Ben-Kaab L (2014) Cadmium and copper stress affect seedling growth and enzymatic activities in germinating barley seeds. Arch Agron Soil Sci 60:765–783
Kalaji HM, Loboda T (2007) Photosystem II of barley seedlings under cadmium and lead stress. Plant Soil Environ 53:511–516
Kaloki P, Devasirvatham V, Tan DK (2019) Chickpea abiotic stresses: combating drought, heat and cold. In: Alexandre DO (ed) Abiotic and biotic stress in plants. IntechOpen, London, pp 725–729
Kaundal A, Sandhu D, Duenas M, Ferreira JF (2019) Expression of the high-affinity K+ transporter 1 (PpHKT1) gene from almond rootstock ‘Nemaguard’ improved salt tolerance of transgenic Arabidopsis. PLoS One 14:e0214473
Khan MA, Gul B (2006) Halophyte seed germination. In: Khan MA, Weber DJ (eds) Ecophysiology of high salinity tolerant plants. Springer, Dordrecht, pp 11–30
Khan MN, Zhan J, Luo T, Liu J, Ni F, Rizwan M, Fahad S, Hu L (2019) Morpho-physiological and biochemical responses of tolerant and sensitive rapeseed cultivars to drought stress during early seedling growth stage. Acta Physiol Plant 41:1–13
Khatun S, Rizzo CA, Flowers TJ (1995) Genotypic variation in the effect of salinity on fertility in rice. Plant Soil 173:239–250
Kim JY, Mahé A, Brangeon J, Prioul JL (2000) A maize vacuolar invertase, IVR2, is induced by water stress. Organ/tissue specificity and diurnal modulation of expression. Plant Physiol 124:71–84
Kirk GJD, Greenway H, Atwell BJ, Ismail AM, Colmer TD (2014) Adaptation of rice in flooded soil. In: Lüttge U, Beyschlag W, Cushman J (eds) Progress in botany. Springer, Berlin, pp 215–253
Klein M, Burla B, Martinoia E (2006) The multidrug resistance-associated protein (MRP/ABCC) subfamily of ATP-binding cassette transporters in plants. FEBS Lett 580:1112–1122
Kranner I, Colville L (2011) Metals and seeds: biochemical and molecular implications and their significance for seed germination. Environ Exp Bot 72:93–105
Krasensky J, Jonak C (2012) Drought, salt, and temperature stress-induced metabolic rearrangements and regulatory networks. J Exp Bot 63:1593–1608
Krishnan HB, Oehrle NW, Alaswad AA, Stevens WG, Maria John KM, Luthria DL, Natarajan SS (2019) Biochemical and anatomical investigation of Sesbania herbacea (Mill.) McVaugh nodules grown under flooded and non-flooded conditions. Int J Mol Sci 20:1–20
Kulichikhin K, Yamauchi T, Watanabe K, Nakazono M (2014) Biochemical and molecular characterization of rice (Oryza sativa L.) roots forming a barrier to radial oxygen loss. Plant Cell Environ 37:2406–2420
Kumar P, Pal M, Joshi R, Sairam RK (2013) Yield, growth and physiological responses of mung bean [Vigna radiata (L.) Wilczek] genotypes to waterlogging at vegetative stage. Physiol Mol Biol Plants 19:209–220
Kuzminov FI, Brown CM, Fadeev VV, Gorbunov MY (2013) Effects of metal toxicity on photosynthetic processes in coral symbionts, Symbiodinium spp. J Exp Mar Biol Ecol 446:216–227
Ladrera R, Marino D, Larrainzar E, González EM, Arrese-Igor C (2007) Reduced carbon availability to bacteroids and elevated ureides in nodules, but not in shoots, are involved in the nitrogen fixation response to early drought in soybean. Plant Physiol 145:539–546
Lahaye PA, Epstein E (1971) Calcium and salt toleration by bean plants. Physiol Plant 25:213–218
Lai JL, Luo XG (2019) High-efficiency antioxidant system, chelating system and stress-responsive genes enhance tolerance to cesium ionotoxicity in Indian mustard (Brassica juncea L.). Ecotoxicol Environ Saf 181:491–498
Landrigan PJ, Fuller R, Acosta NJ, Adeyi O, Arnold R, Baldé AB, Bertollini R, Bose-O’Reilly S, Boufford JI, Breysse PN, Chiles T (2018) The lancet commission on pollution and health. Lancet 391:462–512
Lawas LMF, Shi W, Yoshimoto M, Hasegawa T, Hincha DK, Zuther E, Jagadish SK (2018) Combined drought and heat stress impact during flowering and grain filling in contrasting rice cultivars grown under field conditions. Field Crops Res. 229:66–77
Lea-Cox JD, Syvertsen JP (1993) Salinity reduces water use and nitrate-N-use efficiency of citrus. Ann Bot 72:47–54
Lisar SY, Motafakkerazad R, Hossain MM, Rahman IM (2012) Water stress in plants: causes, effects and responses. In: Ismail MD, Mofizur R, Hiroshi H (eds) Water stress. IntechOpen, London, pp 1–15
Ma L, Coulter JA, Liu L, Zhao Y, Chang Y, Pu Y, Zeng X, Xu Y, Wu J, Fang Y, Bai J (2019) Transcriptome analysis reveals key cold-stress-responsive genes in winter rapeseed (Brassica rapa L.). Int J Mol Sci 20:1–19
Mahmood T, Mustafa HSB, Aftab M, Ali Q, Malik A (2019) Super canola: newly developed high yielding, lodging and drought tolerant double zero cultivar of rapeseed (Brassica napus L.). Genet Mol Res 18:gmr16039951
Makino T, Murakami M, Ishikawa S, Abe T (2019) Regulations for cadmium in Rice and soil in Japan and countermeasures to reduce the concentrations. In: Himeno S, Aoshima K (eds) Cadmium toxicity. Springer, Singapore, pp 103–114
Manickavelu A, Nadarajan N, Ganesh SK, Gnanamalar RP, Babu RC (2006) Drought tolerance in rice: morphological and molecular genetic consideration. Plant Growth Regul 50:121–138
Marchiol L, Assolari S, Sacco P, Zerbi G (2004) Phytoextraction of heavy metals by canola (Brassica napus) and radish (Raphanus sativus) grown on multicontaminated soil. Environ Pollut 132:21–27
Maryan KE, Lahiji HS, Farrokhi N, Komeleh HH (2019) Analysis of Brassica napus dehydrins and their co-expression regulatory networks in relation to cold stress. Gene Expr Patterns 31:7–17
Maxwell K, Johnson GN (2000) Chlorophyll fluorescence—a practical guide. J Exp Bot 51:659–668
Mensah JK, Obadoni BO, Eruotor PG, Onome-Irieguna F (2006) Simulated flooding and drought effects on germination, growth, and yield parameters of sesame (Sesamum indicum L.). Afr J Biotechnol 5:1249–1253
Mierek-Adamska A, Dąbrowska GB, Blindauer CA (2018) The type 4 metallothionein from Brassica napus seeds folds in a metal-dependent fashion and favours zinc over other metals. Metallomics 10:1430–1443
Munns R (2002a) Comparative physiology of salt and water stress. Plant Cell Environ 25:239–250
Munns R (2002b) Salinity, growth and phytohormones. In: Läuchli A, Lüttge U (eds) Salinity: environment-plants-molecules. Springer, Dordrecht, pp 271–290
Munns R (2005) Genes and salt tolerance: bringing them together. New Phytol 167:645–663
Munns R, Tester M (2008) Mechanisms of salinity tolerance. Annu Rev Plant Biol 59:651–681
Ohnishi N, Wacera F, Sakamoto W (2019) Photosynthetic responses to high temperature and strong light suggest potential post-flowering drought tolerance of Sorghum Japanese landrace Takakibi. Plant Cell Physiol 60:2086–2099
Pachauri RK, Allen MR, Barros VR, Broome J, Cramer W, Christ R, Church JA, Clarke L, Dahe Q, Dasgupta P, Dubash NK (2014) Climate change 2014: synthesis report. In: Pachauri R, Meyer L (eds) Contribution of working groups I, II and III to the fifth assessment report of the intergovernmental panel on climate change. Ipcc, Geneva, p 151
Pan L, Yu X, Shao J, Liu Z, Gao T, Zheng Y, Chen C (2019) Transcriptomic profiling and analysis of differentially expressed genes in asparagus bean (Vigna unguiculata ssp. sesquipedalis) under salt stress. PLoS One 14:e0219799
Panozzo A, Dal Cortivo C, Ferrari M, Vicelli B, Varotto S, Vamerali T (2019) Morphological changes and expressions of AOX1A, CYP81D8 and putative PFP genes in a large set of commercial maize hybrids under extreme waterlogging. Front Plant Sci 10:1–14
Panuccio MR, Jacobsen SE, Akhtar SS, Muscolo A (2014) Effect of saline water on seed germination and early seedling growth of the halophyte quinoa. AoB Plants 6:plu047
Parelle J, Roudaut JP, Ducrey M (2006) Light acclimation and photosynthetic response of beech (Fagus sylvatica L.) saplings under artificial shading or natural Mediterranean conditions. Ann For Sci 63:257–266
Parent C, Capelli N, Berger A, Crèvecoeur M, Dat JF (2008) An overview of plant responses to soil waterlogging. Plant Stress 2:20–27
Park JH, Lamb D, Paneerselvam P, Choppala G, Bolan N, Chung JW (2011) Role of organic amendments on enhanced bioremediation of heavy metal (loid) contaminated soils. J Hazard Mater 185:549–574
Park HC, Hwang JE, Jiang Y, Kim YJ, Kim SH, Nguyen XC, Kim CY, Chung WS (2019) Functional characterisation of two phytochelatin synthases in rice (Oryza sativa cv. Milyang 117) that respond to cadmium stress. Plant Biol 21:854–861
Parra-Almuna L, Diaz-Cortez A, Ferrol N, de la Luz Mora M (2018) Aluminium toxicity and phosphate deficiency activates antioxidant systems and up-regulates expression of phosphate transporters gene in ryegrass (Lolium perenne L.) plants. Plant Physiol Biochem 130:445–454
Patel BB, Dave RS (2011) Studies on infiltration of saline-alkali soils of several parts of Mehsana and Patan districts of North Gujarat. J Appl Technol Environ Sanit 1:87–92
Paul K, Pauk J, Kondic-Spika A, Grausgruber H, Allahverdiyev T, Sass L, Vass I (2019) Co-occurrence of mild salinity and drought synergistically enhances biomass and grain retardation in wheat. Front Plant Sci 10:1–15
Pekcan V, Evci G, Yilmaz MI, Nalcaiyi ASB, Erdal SÇ, Cicek N, Ekmekci Y, Kaya Y (2015) Drought effects on yield traits of some sunflower inbred lines. Poljopr Sumar 61:101–107
Pierart A, Shahid M, Sejalon-Delmas N, Dumat C (2015) Antimony bioavailability: knowledge and research perspectives for sustainable agricultures. J Hazard Mater 289:219–234
Popović V, Miladinović J, Vidić M, Vučković S, Dolijanović Ž, Ikanović J, Zivanović LJ, Kolarić L (2015) Drought–limiting factors in soybean production. The effect of irrigation on yield of soybean [Glycine max (L.) Merr.]. Proceedings. Institute of PKB Agroekonomik, Belgrade, pp 11–21
Pourrut B, Shahid M, Douay F, Dumat C, Pinelli E (2013) Molecular mechanisms involved in lead uptake, toxicity and detoxification in higher plants. In: Gupta DK (ed) Heavy metal stress in plants. Springer, Berlin, Heidelberg, pp 121–147
Qadir M, Schubert S (2002) Degradation processes and nutrient constraints in sodic soils. Land Degrad Dev 13:275–294
Queiroz MS, Oliveira CE, Steiner F, Zuffo AM, Zoz T, Vendruscolo EP, Silva MV, Mello BFFR, Cabra RC, Menis FT (2019) Drought stresses on seed germination and early growth of maize and sorghum. J Agric Sci 11:310–318
Rajendran K, Tester M, Roy SJ (2009) Quantifying the three main components of salinity tolerance in cereals. Plant Cell Environ 32:237–249
Rathaur P, Ramteke PW, Raja W, John SA (2012) Isolation and characterization of nickel and cadmium tolerant plant growth promoting rhizobacteria from rhizosphere of Withania somnifera. J Environ Sci 6:253–261
Reddy MD, Mittra BN (1985) Effect of complete plant submergence at different growth stages on grain yield, yield components and nutrient content of rice. Plant Soil 86:379–386
Reddy KR, Krutz L, Gao W, Bellaloui N (2019) Poor seed quality, reduced germination, and decreased seedling vigor in soybean is linked to exposure of the maternal lines to drought stress. BioRxiv:590059 (in press)
Ren ZH, Gao JP, Li LG, Cai XL, Huang W, Chao DY, Zhu MZ, Wang ZY, Luan S, Lin HX (2005) A rice quantitative trait locus for salt tolerance encodes a sodium transporter. Nat Genet 37:1141–1146
Romero-Puertas MC, Rodríguez-Serrano M, Corpas FJ, Gomez MD, Del Rio LA, Sandalio LM (2004) Cadmium-induced subcellular accumulation of O2·− and H2O2 in pea leaves. Plant Cell Environ 27:1122–1134
Ronga D, Rizza F, Badeck FW, Milc J, Laviano L, Montevecchi G, Pecchioni N, Francia E (2018) Physiological responses to chilling in cultivars of processing tomato released and cultivated over the past decades in Southern Europe. Sci Hortic 231:118–125
Rozeff N (1995) Sugarcane and salinity—a review paper. Sugarcane 5:8–19
Sadak MS (2019) Physiological role of trehalose on enhancing salinity tolerance of wheat plant. Bull Natl Res Cent 43:1–10
Saha P, Chatterjee P, Biswas AK (2010) NaCl pretreatment alleviates salt stress by enhancement of antioxidant defense system and osmolyte accumulation in mungbean (Vigna radiata L. Wilczek). Indian J. Exp Biol 48:593–600
Sairam RK, Kumutha D, Ezhilmathi K, Deshmukh PS, Srivastava GC (2008) Physiology and biochemistry of waterlogging tolerance in plants. Biol Plant 52:401
Sakamoto A, Murata N (2002) The role of glycine betaine in the protection of plants from stress: clues from transgenic plants. Plant Cell Environ 25:163–171
Sánchez-Gómez D, Cervera MT, Escolano-Tercero MA, Vélez MD, de María N, Diaz L, Sánchez-Vioque R, Aranda I, Guevara MA (2019) Drought escape can provide high grain yields under early drought in lentils. Theor Exp Plant Physiol 31(2):273–286
Sapeta H, Costa JM, Lourenco T, Maroco J, Van der Linde P, Oliveira MM (2013) Drought stress response in Jatropha curcas: growth and physiology. Environ Exp Bot 85:76–84
Satir O, Berberoglu S (2016) Crop yield prediction under soil salinity using satellite derived vegetation indices. Field Crops Res 192:134–143
Sauter M (2013) Root responses to flooding. Curr Opin Plant Biol 16:282–286
Schulz E, Tohge T, Zuther E, Fernie AR, Hincha DK (2016) Flavonoids are determinants of freezing tolerance and cold acclimation in Arabidopsis thaliana. Sci Rep 6:1–14
Schulze ED, Beck E, Buchmann N, Clemens S, Müller-Hohenstein K, Scherer-Lorenzen M (2019) Water deficiency (Drought). In: Schulze ED, Beck E, Buchmann N (eds) Plant Ecology. Springer, Heidelberg, pp 162–202
Sculthorpe CD (1967) The biology of aquatic vascular plants. Edward Arnold Press, London, p 610
SEDAC (n.d.) Global flood hazard frequency and distribution, v1 (1985 – 2003). http://sedac.ciesin.columbia.edu/data/set/ndh-flood-hazard-frequency-distribution. Accessed 15 Oct 2019
Selpair S (2018) Analyzing reproductive capabilities of chickpea in cold environment. http://210.212.34.21/handle/32116/1952. Accessed 2 Mar 2018
Shahid M, Arshad M, Kaemmerer M, Pinelli E, Probst A, Baque D, Pradere P, Dumat C (2012) Long-term field metal extraction by Pelargonium: phytoextraction efficiency in relation to plant maturity. Int J Phytoremediation 14:493–505
Sharma N, Bisht SS, Gupta S, Panda AK, Rana M (2018) Analysis of proteomic diversity and calcium binding protein(s) in seeds of horse gram (Macrotyloma uniflorum) cultivars from Uttarakhand. Int J Pharm Sci Res 9:3274–3280
Shawon RA, Kang DH, Ryu CS, Kim DE, Lee SY, Bae JH, Kim YO, Ku YG (2017) Physiological responses and antioxidative enzyme activities in pepper (Capsicum annuum) seedlings under low temperature stress. J Agric Life Sci 51:67–77
Shiono K, Takahashi H, Colmer TD, Nakazono M (2008) Role of ethylene in acclimations to promote oxygen transport in roots of plants in waterlogged soils. Plant Sci 175:52–58
Shrivastava P, Kumar R (2015) Soil salinity: a serious environmental issue and plant growth promoting bacteria as one of the tools for its alleviation. Saudi J Biol Sci 22:123–131
Silva EN, Silveira JA, Aragão RM, Vieira CF, Carvalho FE (2019) Photosynthesis impairment and oxidative stress in Jatropha curcas exposed to drought are partially dependent on decreased catalase activity. Acta Physiol Plant 41:1–12
Singh S, Singh SP, Pathak AD, Pandey N (2019) Assessment of waterlogging induced physiobiochemical changes in sugarcane varieties and its association with waterlogging tolerance. J Environ Biol 40:384–392
Sinha S, Raxwal VK, Joshi B, Jagannath A, Katiyar-Agarwal S, Goel S, Kumar A, Agarwal M (2015) De novo transcriptome profiling of cold-stressed siliques during pod filling stages in Indian mustard (Brassica juncea L.). Front Plant Sci 6:1–17
Solaiman Z, Colmer TD, Loss SP, Thomson BD, Siddique KHM (2007) Growth responses of cool-season grain legumes to transient waterlogging. Aust J Agric Res 58:406–412
Souza SC, Mazzafera P, Sodek L (2016) Flooding of the root system in soybean: biochemical and molecular aspects of N metabolism in the nodule during stress and recovery. Amino Acids 48:1285–1295
Stvolinskaya NS (2000) Viability of Taraxacum officinale Wigg. in populations of the city of Moscow in relation to motor transport pollution. Russ J Ecol 31:129–131
Sudhir P, Murthy SDS (2004) Effects of salt stress on basic processes of photosynthesis. Photosynthetica 42:481–486
Suhayda CG, Giannini JL, Briskin DP, Shannon MC (1990) Electrostatic changes in Lycopersicon esculentum root plasma membrane resulting from salt stress. Plant Physiol 93:471–478
Taiz L, Zeiger E (2006) Plant physiology, 4th edn. Sinauer Associates, Sunderland, MA, pp 345–376
Tang H, Niu L, Wei J, Chen X, Chen Y (2019) Phosphorus limitation improved salt tolerance in maize through tissue mass density increase, osmolytes accumulation, and Na+ uptake inhibition. Front Plant Sci 10:1–10
Tarekegne A, Bennie ATP, Labuschagne MT (2000) Effects of soil waterlogging on the concentration and uptake of selected nutrients in wheat genotypes differing in tolerance. In: The eleventh regional wheat workshop for eastern, central and southern Africa, Addis Abeba, Ethiopia. CIMMYT, Addis Ababa, pp 253–263
Tewari S, Arora NK (2016) Soybean production under flooding stress and its mitigation using plant growth-promoting microbes. In: Mohammad M (ed) Environmental stresses in soybean production: soybean production, vol 2. Academic press, San Diego, pp 23–40
Turhan E, Ergin S (2012) Soluble sugars and sucrose-metabolizing enzymes related to cold acclimation of sweet cherry cultivars grafted on different rootstocks. Sci World J 2012:1–7
Turner NC, Wright GC, Siddique KHM (2001) Adaptation of grain legumes (pulses) to water-limited environments. Adv Agron 71:194–233
UNESCO (2018) Climate change and water security. https://en.unesco.org/themes/addressing-climate-change/climate-change-and-water-security. Accessed 12 Mar 2018
Vandoorne B, Descamps C, Mathieu AS, Van den Ende W, Vergauwen R, Javaux M, Lutts S (2014) Long term intermittent flooding stress affects plant growth and inulin synthesis of Cichorium intybus (var. sativum). Plant Soil 376:291–305
Wang Y, Zhang B, Jiang D, Chen G (2019) Silicon improves photosynthetic performance by optimizing thylakoid membrane protein components in rice under drought stress. Environ Exp Bot 158:117–124
Watts J (19 March 2018) Water shortages could affect 5bn people by 2050, UN report warns. The Guardian. https://www.theguardian.com/environment/2018/mar/19/water-shortages-could-affect-5bn-people-by-2050-un-report-warns. Accessed 31 Oct 2019
Wei H, Chen C, Ma X, Zhang Y, Han J, Mei H, Yu S (2017) Comparative analysis of expression profiles of panicle development among tolerant and sensitive rice in response to drought stress. Front Plant Sci 8:1–10
Williams G, Vanniarajan C, Vetriventhan M, Thiageshwari S, Anandhi K, Rajagopal B (2019) Genetic variability for seedling stage salinity tolerance in barnyard millet [Echinochloa frumentacea (Roxb.) Link]. Electron J Plant Breed 10:552–558
Wise RR, Naylor AW (1987) Chilling-enhanced photooxidation: the peroxidative destruction of lipids during chilling injury to photosynthesis and ultrastructure. Plant Physiol 83(2):272–277
Wise RR, Olson AJ, Schrader SM, Sharkey TD (2004) Electron transport is the functional limitation of photosynthesis in field-grown Pima cotton plants at high temperature. Plant Cell Environ 27:717–724
Wu G, Kang H, Zhang X, Shao H, Chu L, Ruan C (2010) A critical review on the bio-removal of hazardous heavy metals from contaminated soils: issues, progress, eco-environmental concerns and opportunities. J Hazard Mater 174:1–8
Wu GQ, Wang JL, Feng RJ, Li SJ, Wang CM (2018) iTRAQ-based comparative proteomic analysis provides insights into molecular mechanisms of salt tolerance in sugar beet (Beta vulgaris L.). Int J Mol Sci 19:1–20
Xiong ZT, Zhao F, Li MJ (2006) Lead toxicity in Brassica pekinensis Rupr.: effect on nitrate assimilation and growth. Environ Toxicol 21:147–153
Yadav P, Kaur R, Kanwar MK, Bhardwaj R, Sirhindi G, Wijaya L, Alyemeni MN, Ahmad P (2018) Ameliorative role of castasterone on copper metal toxicity by improving redox homeostasis in Brassica juncea L. J Plant Growth Regul 37:575–590
Yamauchi T, Colmer TD, Pedersen O, Nakazono M (2018) Regulation of root traits for internal aeration and tolerance to soil waterlogging-flooding stress. Plant Physiol 176:1118–1130
Yazaki K (2006) ABC transporters involved in the transport of plant secondary metabolites. FEBS Lett 580:1183–1191
Yin D, Chen S, Chen F, Guan Z, Fang W (2009) Morphological and physiological responses of two chrysanthemum cultivars differing in their tolerance to waterlogging. Environ Exp Bot 67:87–93
Yu B, Zhao CY, Li J, Li JY, Peng G (2015) Morphological, physiological, and biochemical responses of Populus euphratica to soil flooding. Photosynthetica 53:110–117
Zeng F, Ali S, Zhang H, Ouyang Y, Qiu B, Wu F, Zhang G (2011) The influence of pH and organic matter content in paddy soil on heavy metal availability and their uptake by rice plants. Environ Pollut 159:84–91
Zhang X, Wan S, Hao J, Hu J, Yang T, Zong X (2016) Large-scale evaluation of pea (Pisum sativum L.) germplasm for cold tolerance in the field during winter in Qingdao. Crop J 4:377–383
Zhang C, Bai T, Xie D, Wang Y, Zhang H, Chen Y, Ni Z (2019a) Effects of PEG-6000 simulation drought stress on seed germination of mango (Mangiferca indica L.). Nanfang Nongye Xuebao 50:600–606 (in Chinese)
Zhang W, Wang S, Yu F, Tang J, Shan X, Bao K, Yu L, Wang H, Fei Z, Li J (2019b) Genome-wide characterization and expression profiling of SWEET genes in cabbage (Brassica oleracea var. capitata L.) reveal their roles in chilling and clubroot disease responses. BMC Genomics 20:1–16
Zhao C, Yang R, Hong Y, Ren Z, Tang K, Zhang H, Zhu JK (2019) A role for PICKLE in the regulation of cold and salt stress tolerance in Arabidopsis. Front Plant Sci 10:1–11
Zheng C, Jiang D, Liu F, Dai T, Jing Q, Cao W (2009) Effects of salt and waterlogging stresses and their combination on leaf photosynthesis, chloroplast ATP synthesis, and antioxidant capacity in wheat. Plant Sci 176:575–582
Zhou M (2010) Improvement of plant waterlogging tolerance. In: Mancuso S, Shabala S (eds) Waterlogging signalling and tolerance in plants. Springer, Heidelberg, pp 267–285
Zhu JK (2001) Plant salt tolerance. Trends Plant Sci 6:66–71
Zinselmeier C, Jeong BR, Boyer JS (1999) Starch and the control of kernel number in maize at low water potentials. Plant Physiol 121:25–36
Zörb C, Geilfus CM, Dietz KJ (2019) Salinity and crop yield. Plant Biol 21:31–38
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Rawat, P., Shankhdhar, D., Shankhdhar, S.C. (2021). Abiotic Stress: Its Outcome and Tolerance in Plants. In: Sharma, A. (eds) Microbes and Signaling Biomolecules Against Plant Stress. Rhizosphere Biology. Springer, Singapore. https://doi.org/10.1007/978-981-15-7094-0_5
Download citation
DOI: https://doi.org/10.1007/978-981-15-7094-0_5
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-15-7093-3
Online ISBN: 978-981-15-7094-0
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)