Abstract
Drought is a major threat to agriculture globally and improving crop yield under drought conditions is a major challenge of plant breeding. Many QTLs have been identified for drought stress response, and researchers are striving hard to comprehend and dissect plant tolerance mechanisms related to drought stress. Unravelling the biochemical regulation of drought tolerance and molecular breeding and transgenic approaches can help us manage drought stress in plants. Recent advances achieved regarding genomic tools and genetic techniques in addition to precise phenotyping and advanced breeding methodologies will enable exhibiting metabolic pathways and candidate genes underlying drought tolerance in rice. Taken altogether, new horizons have been opened for the breeders to utilize markers for QTLs, signaling cascade, hormonal cross talk, or gene transformation in plants to develop a drought resistant genotype.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Among cereal crops, rice (Oryza sativa) is a highly significant crop included in the family of grasses called poaceae, and after maize production, it is the second most cultivated crop worldwide. The tremendously increasing population has enhanced the need for rice yields worldwide (Liang et al. 2014), and rice demand will increase by up to 50% by 2025 (Khush 2001). Rainfed ecosystems provide only 25% of the global water sources. Rice is critically influenced by drought stress because it can reduce production by 15–50% (Srividhya et al. 2011). At the time of reproduction, rice is particularly sensitive to drought stress, leading to decreased grain production (Venuprasad et al. 2008).
Rice (Oryza sativa) can be categorized into three subspecies, specifically japonica, javanica, and indica (Mae 1997). Because of its adaptability and extensive distribution, rice serves as a tremendous model structure for investigating evolutionary plant genomics in a wide series of developmental, physiological, and morphological varieties located in both rice and its extensively distributed wild lines, Oryza nivara and Oryza rufipogon (Zhao et al. 2010).
For drought tolerance, conventional breeding, including the selection of diversified landraces under conditions of drought, has facilitated crop improvement (Kumar et al. 2008). Similarly, molecular regulation of candidate genes, viz., regulatory genes and transcription factors, and their expression analysis have helped us to develop drought tolerant plants (Todaka et al. 2015).
2 Mechanisms of Drought Resistance
Drought resistance is described as the capacity of a plant to produce its highest commercial product under water stress conditions and its relation to water control free management. It is a complex feature whose influence depends on the actions and relations regarding various biochemical, physiological, and morphological responses (Kooyers 2015). Under drought stress, rice mostly responds by stomata closure, leaf rolling, and enhanced abscisic acid (ABA) production to reduce the water deficit (Price et al. 2002). Research regarding physiology recommends that drought resistance in rice mostly relies upon water use efficiency (WUE) that tolerates the lowest utilization of water for the highest production and osmotic adjustment that allows plants to conserve the meristem and sustain turgor (Nguyen et al. 2004).
There are several processes for the adjustment of plants under drought stress, including drought tolerance, escape, and avoidance; they are modified based on the cause of their morphological, physiological, and molecular alteration (Fukai and Cooper 1995).
2.1 Drought Escape
Drought escape is defined as the capacity of a plant to accomplish its life cycle before the advancement of critical water deficiency in soil. It includes two distinct processes: plasticity development and quick phenological improvement. In quick phenological enhancement, plants are competent to produce flowers with the lowest vegetative development allowing them to make seeds with an inadequate amount of water. In plasticity development, plants show plenty of vegetative growth, flowers, and seeds during abundant rain. This allows the desert ephemerals to drought escape and survive for long periods without rain. In drought susceptible zones of the southern US, drought escape is a significant process that enables rice to produce seeds regardless of restricted water accessibility (Kumar et al. 2008).
2.2 Drought Avoidance
Drought avoidance is described as the capacity of plants to sustain maximum water contents in tissue in spite of a soil humidity deficiency. The tissue of plants has two possibilities to retain a maximum water status under maximum needs of evaporation and improving water deficiency in soil, to either decrease the loss of water or sustain the water supply. Rice cultivars that can sustain water potential through biosynthesis of ABA, further biochemical processes, or modified root structure arise under different processes of drought avoidance. These cultivars are competent to reduce the loss of yield in drought stress (Singh et al. 2012). Methods exist for increasing water use efficiency (WUE) and minimizing loss of water to achieve drought avoidance. Rice varieties that drought escape generally have bristle-like and deep roots with a maximum capacity for penetration in soil and branching, maximum root to shoot proportion, early stomata closure, and maximum resistance in cuticle and leaf rolling elasticity (Wang et al. 2006).
2.3 Drought Tolerance
Drought tolerance is the capacity of plants to live with minimum water potential in tissue (Fleury et al. 2010). In genetically modified rice, drought tolerance is a composite attribute that is regulated by polygenic impact and includes complicated physiological and morphological processes (Li et al. 2017); for instance, balancing of turgor pressure by osmotic adaptation, cell size reduction, improved cell elasticity, and dehydration tolerance via protoplasmic resistance. The reaction of plants regarding water contents in tissue control their status under drought tolerance (Mitra 2001) and the qualities linked with such processes are studied as secondary attributes. Secondary attributes, including RWC, osmotic adaptation, stomatal conductivity, and leaf rolling, have been utilized for selection (Kato et al. 2006).
3 Assessing Drought Tolerance Performance
Plant efficiency is an important constituent that needs to be measured while abiotic stress resistance rice is cultivated. Plants efficiency is extremely disturbed with drought stress in the field, which fluctuates in harsh periods, and is also supplemented by synchronized stresses such as low and high temperature. Therefore, analyses of drought tolerance attained from glasshouse conditions is not necessarily equivalent to field conditions. Thus, experiments in the field are essential for the appropriate assessment of drought stress challenges.
Drought stress has been considered in lowland and upland conditions with various levels of stress, such as moderate, severe, and mild environment stress. In the field, drought stress measurement is completed by applying unceasing stress for 10 or more days before proceeding, the resulting International Rice Research Institute’s typical measures include observing modifications in soil humidity and diffusion resistance in a stressed field via penetrometer and gravimetric methods (Bengough et al. 2011). RWC is measured after leaf necrosis and rolling have initiated (Jeong et al. 2012; Raman et al. 2012).
The plant reactions under drought stress are further categorized into moderate, severe, and mild depending on the reduction of yield percentage contrasted with grain yield from four non-stress trials (Kumar et al. 2008). In lowland conditions, stress trials revealing a 30% or less reduction of yield are called mild stress, reductions of 31–65% are called moderate stress, and above 65% reduction of yield are denoted as severe stress. At the time of a non-stress upland part of this trial, the reduction percentage of yield contrasted with the average yield of a non-stress lowland trial was utilized to categorize the trial as mild stress upland treatments (Dixit et al. 2012).
The field plots are watered by sprayers two times a week during the development and premature vegetative phase (Kumar et al. 2014). After this period, drought stress is applied via cover-up irrigation and the field is watered only while the water stretches in soil exits under 50 kPa at 30 cm depth of soil. At this water content in soil, the maximum number of rice plants wither and show leaf desiccation. This kind of repeated stress is measured to be competent regarding rice population selection for drought tolerant containing varieties that have the highest growth in this period (Lafitte et al. 2004), and it confirms the capability of the entire plant to tolerate stress during reproductive growth. Similar agricultural trials are useful in non-stress upland field plots, excluding when irrigation is constant two times a week more than 10 days before yield. In the lowland field conditions, uprooted field plots are pumped out after 30 days of uprooting and irrigation is pending to impose drought stress during the reproductive period (Kumar et al. 2014). Drought stress is constant till severe leaf rolling. For the plant’s continued existence, irrigation is supplied subsequently through flooding, and water is pumped out after 24 h to apply the next rotation of drought stress. The water table penetration is calculated by installing a 1.1 m polyvinyl chloride (PVC) pipeline in the field plots to ensure constant time intervals. Pipelines are generally installed to a depth of 1.0 m, and 10 cm of pipeline is leftover above the soil surface. Reduction in the water table is constantly calculated through a meter ruler every day after the beginning of the stress (Xiao et al. 2009; Wang et al. 2014).
4 Quantitative Trait Loci for Drought Tolerance in Rice
The process for drought tolerance is complicated, due todifferences in plant phenology and measured by numerous quantitative trait loci (QTLs). Plant reactions are complex and difficult to realize without a detailed study of physiological and molecular sources of how these reactions are managed. Neither conventional breeding nor advanced genetics can successfully expand the drought tolerance in plants if the genetic phenomena associated with production of seed strength are not understood (Lisei-de-Sá et al. 2017). Advances in systematic plant phenotyping, plant physiology, and genomics lead to innovative findings in drought tolerant crops. Consequently, yield breeders will be capable of boosting yields of crops utilizing the modern gene system data and approaches for plant development (Tuberosa 2012). Advancing plant physiology improves our information regarding the complicated system of drought tolerance and its link with various traits, molecular biology, and effective genomics tools, and improved choice productivity will result in the recognition of candidate QTLs and genes associated with these qualities. Tools for molecular breeding can be utilized to develop QTLs for chop enhancement; therefore, candidate genes are a major focus of genetic engineering and production of genetically modified crops (Varshney et al. 2011).
In various abiotic stresses, the recognition of candidate genes reliable for plant resistance, laterally with the utilization of the most appropriate promoters linked with these procedures, is important to produce genetically modified crops with increasing drought tolerance (Varshney et al. 2011). While tools for genetic engineering include high regulatory events and adverse public observations for biosafety, targeted induced local lesions in genome (TILLING) and molecular breeding crops are broadly identified (Varshney et al. 2011). The qualitative traits loci are normally fragments of linkage mapping or linkage analysis-based QTL mapping (Appels et al. 2018). Conventional QTLs mapping contains populations mapping in which qualities associated with tolerance of drought are separating; classifying polymorphic markers that are used for genotyping mapping populations; making genetic maps; correctly phenotyping depends on the drought tolerance associated qualities; and ultimately, according to phenotyping and genotyping QTL mapping data. For drought tolerance in many crops, numerous linkage mapping considerations have been accomplished (Fleury et al. 2010).
Due to intrinsic restrictions connected to populations mapping, linkage analysis-based QTL mapping cannot suggest comprehensive data related to QTLs. These restrictions comprise the following: the recognized QTLs are generally connected with large chromosomal fragments or genomic areas because of insufficient time for hybridization, insufficient phenotypic difference connected to occurring qualities in populations mapping, and the separation of diverse QTLs connected with identical qualities in various populations mapping (Liu et al. 2012).
Linkage disequilibrium (LD) depends on association mapping (AM), which has been utilized in human inheritances, and is recommended as a QTL mapping tool to affect some of these limitations in different crop species (Bolger et al. 2014). The AM technique contains five phases: choosing many distinct panels or groups from a wild pool of the population or germplasm; obtaining perfect phenotypic information on every panel; fascinating candidates genes have high-density sequencing or the markers for a genotyping panel; observing the genetic variation level among panels in the specific population and understanding the factors between distinct pairs in the population; and evaluating the suggestion mapping consequences corresponding to information attained regarding population assembly, relationship, and the association of phenotypic and haplotypic or genotypic information. There are several benefits of the mapping method over biparental mapping linkage, as follows: advanced determination due to use of all recombination actions during the evolutionary history of a particular crop species; avoiding the growth of a specific population mapping and the facility of a wild germplasm selection for a particular crop to decrease the mandatory period for QTL recording; utilizing similar genotyping information and panel of AM for various mapping qualities, assembling it into a practical tool; removing casually hybrid hereditary lines which express a deficient agronomic category from the system of populations; and being competent to model and nearby several alleles for each locus comparative for mapping linkage. In brief, in rice, several QTLs associated with drought tolerance have been recognized. Although, to date, only some QTL reports have been described regarding the influence of grain quality on drought; for instance, a main QTL (qDTY1.1), which has stabilizer influence on grain yield (GY) at the reproductive phase in drought stress, was identified on rice chromosomal 1 and lined through RM431 and RM11943 in three populations: N22/IR64, N22/MTU1010, and N22/Swarna (Vikram et al. 2015). Maximum QTLs have been reported based on an extensive series of significant characteristics, including (1) mechanisms of yield, (2) physiological reactions, for example, osmotic adjustment, osmotic potential, RWC, chlorophyll contents, index of rolling flag leaf, osmotic potential of leaf, ratio of carbon isotope, water soluble carbohydrates, and different isotopes of carbon in grain, and (3) root characteristics.
5 Identification of Candidate Drought Tolerance Genes in Model Plant Systems
For drought tolerance, a substantial number of candidate genes can be recognized by significant developments in traditional plant species. Thus far, genomes of several classical plants and the main species of plants have been sequenced (Feuillet et al. 2011). Functional genomics, molecular physiology, and annotation of genome reports have been accomplished in numerous main and model crops to detect candidate genes included under drought tolerance. Under drought stress, these candidate genes contain a large family of excluded genes. Various protein expressions via drought stress linked candidate genes carry out important functions regarding (1) protection of cells, for instance, modification in structure, osmotic adjustment, detoxification, degradation, and repair and (2) positive relations with additional transcription factors and proteins, for example, MYB, protein kinases, DREB, and bZIP that are included in the reaction of plant under drought conditions via controlling further reactive genes, for instance, those which participated in the protection of cells, to survive under drought stress (Basu and Roychoudhury 2014; Banerjee and Roychoudhury 2017). For various main and model plants, detecting drought tolerance (DT) genes is important to consider the practical role of the DT system and its utilization downstream, incorporating proof by MAS over molecular breeding. From distinct plant species, various DT candidate genes involved in transcriptome reactions have been estimated and categorized.
Candidate genes have been verified using various methods; for instance, qRT-PCR, QTL maps penetration, expression analysis, TILLING, linkage mapping, mining of allele, and utilization of these methods have been previously studied (Bevan et al. 2017). Recently, several functional genomic and transcriptomic reports have been accomplished to recognize the stress processes in various crop plants. Prevalent methods that efficiently separate candidate genes associated with drought stress under drought resistant varieties produce expressed sequence tags (ESTs) from complementary DNA libraries of tissues accumulated in drought. To date, various reactive genes for drought have been recognized from numerous crop species, including from seedlings of rice-controlled cDNA libraries to the classification of several genes related to drought tolerance, which were extremely expressed in drought (Hadiarto and Tran 2010).
Another method to detect the candidate genes is transcriptional profiling, and it contains the variance expression of genes in plant tissues during various periods after the beginning of drought stress, as well as between drought tolerant and drought prone cultivars (Hampton et al. 2010). In spite of this, choosing the accurate tissue category, tissue phase, and stress action, such as strength and effectiveness, are important to regulate the minimal drought stress necessary to extract RNA for transcriptomics reports (Du et al. 2017). For hereditary materials, near-isogenic lines (NILs) are the system; they differ only in the particular attributes between cultivars with different genetic conditions. Therefore, concentrating on NILs can offer maximum declaration consequences that are particular and are attained from differentially expressed genes associated with the focus on qualities. Furthermore, miRNAs have been established to be involved in the reaction to drought, and tolerance in several crops (rice and soybean) can enhance drought tolerance (Kulcheski et al. 2011).
For transcriptional profiling, there are several programs which can be used, including cDNA-AFLP (Ridout and Donini 1999), DDRT-PCR (Liang and Pardee 1992), cDNA and SSH (Sahebi et al. 2015), cDNA and microarrays oligonucleotide (Sreenivasulu et al. 2010), and EST depends on digital expression analysis (Raju et al. 2010). Super-SAGE is another method that can be effectively utilized in various crops under stress conditions (Matsumura et al. 2010). Because next generation sequencing (NGS) tools offer accurate real time and digital study of sequences dependent on transcriptomes, other approaches, for example, microarrays, will have a tendency to be replaced with NGS in upcoming years. In analysis of gene expression, the utilization of NGS has contributed to different methods, for instance, RNA-seq (Nagalakshmi et al. 2008), DGE-TAG (Moore 2015) and Deep SAGE (Nielsen et al. 2006).
Using RNA-Seq with NGS tools has numerous benefits in the analysis of transcriptome assembly, for instance, the detection of particular expression of allele and unit intersections (Malone and Oliver 2011), while NGS may offer maximum sequencing quantity results directly from RNA of stress faced tissues from various cultivars. Drought sensitive and drought tolerant transcriptional profiling can recognize candidate genes associated with drought tolerance besides in grouping with genetic/QTL maps and can play a role in markers that are used for molecular genetics (Hiremath et al. 2011).
Numerous candidate genes recognized by the above methods may be correlated with QTLs used for drought tolerance characteristics. Consequently, a genomic and inheritance technique that permits the measurable study of transcriptional profiling may segregate the QTLs (expression QTLs) used for drought tolerance characteristics (Varshney et al. 2005). Therefore, in cis situations, if QTLs expression are identified, the targets genes depending on molecular markers will play a role as screening markers for specific traits (Potokina et al. 2008). Transcriptional profiling depending on NGS tools will likely be utilized to detect candidate DT genes from most important crop species and then applied in genomic, genetic, and molecular breeding.
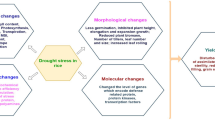
6 The Morphological Responses of Rice Under Drought Stress
During drought stress, plants suffer maximum transcription rates and deficiency of required water adjacent to the roots. Extensive drought harms the development, yield, and growth of rice. When water is deficient, normally rice growth is reduced or ceased (Zhu et al. 2002). Reduction in development and growth of rice under drought stress have demonstrated a selection of premature morphological modifications in rice (Wakim et al. 2008). Cell growth is reduced by an imbalance in turgor under drought stress (Saddique et al. 2018). Drought effects both elongation and expansion growth (Miller et al. 2010) and hinders increasing cell division (Jaleel et al. 2009). Furthermore, it inhibits the germination of rice seedlings (Swain et al. 2014) and reduces the quantity of tillers (Muhammad 2012) and height of the plant (Sokoto and Muhammad 2014). Drought stress greatly reduces biomass production (Farooq et al. 2010). Numerous reports have discovered a decrease in the fresh and dry biomass of shoots and roots during drought stress. In rice, there is a reduction of photosynthetic and biochemical mechanisms due to the decreasing length and height of the fresh biomass of shoots and roots (Basu et al. 2010a, b; Usman et al. 2013).
7 Drought Stress Omics
Under drought stress, a dataset involving modifications in gene expression, metabolites, and profiling of proteins discovered in plants should be obtained via genomics techniques. Under abiotic stress, dicot and monocot plants utilize similar transcription factors (Yamaguchi-Shinozaki and Shinozaki 2006). In drought tolerant plants, the molecular tools involve (1) a signal transduction force monitored via transcriptional regulation and initiation, (2) protein fortification with support of several proteins, for instance, dehydrin for late embryogenesis or chaperones such as HS proteins (Banerjee and Roychoudhury 2016), (3) increasing of osmolytes, comprising glycine, myoinositol, proline, mannitol, trehalose, and betaine (Roychoudhury et al. 2015), (4) the initiation of antioxidants, for example, ascorbic acid and glutathione (Roychoudhury and Basu 2012), and (5) decreasing of ROS toxicity through glutathione S-transferase and superoxide dismutase. Various processes may include numerous identical genes that can be recognized by transcriptomic trials on rice families cultivated under standard and drought conditions. Numerous regulatory methods for drought tolerance occur in several species; however, the molecular reaction under drought stress has been analytically exposed (Aprile et al. 2009) to be less consistent due to alterations in growing stage, tissue analysis, and stress dynamics.
8 The Role of Photosynthesis and Photosynthetic Pigments in the Drought Response
For several metabolic mechanisms, photosynthesis is an important complicated mechanism under water stress. The most significant aspects that control photosynthesis are diffusional limitation in carbon dioxide due to reduction in photosynthesis, early stomata closure, and the reduced productivity of PSII photochemical development and the biochemical mechanisms associated with triose phosphate production. The photosynthetic rate is disturbed by changes in any of these processes. During drought stress, uptake of carbon dioxide is reduced in stomatal conductance (gs) and mesophyll conductance (gm) (Chakhchar et al. 2017; Banerjee and Roychoudhury 2018). Therefore, during drought stress, the gm value exhibits the tolerance in rice under water deficit (Lauteri et al. 2014). Energy and ATP are provided by the activity of PSII. If PSII movement increases, an extreme reduction in ETC in the photosynthetic mechanism may occur, thus promoting ROS production. Therefore, an equilibrium between the need for photochemical and photo assimilates activity should be essential. In rice flag leaves, the PSII activity is damaged expressively by drought stress (Pieters and El Souki 2005). This mechanism occurs because drought stimulates the D1 polypeptide degradation, triggering the inactivation of the reaction center of PSII. Photosynthesis is limited by the drought stress because the efficiency of the enzyme present in the Calvin cycle and the performance of Rubisco have been reduced (Zhou et al. 2007). However, the quantity of Rubisco activase stimulates conformational modification in ATP-dependent, releases Rubisco positions from dead-end blockage, and can enhance the defensive process in drought stress. At this time, enzymes included in photosynthesis of C4 plants have been initiated in rice crops to change the photosynthesis rate and increase plant efficiency against the stress reaction. Enzymes that are required for photosynthesis of C4 overexpressed in genetically modified rice crops, for instance, pyruvate orthophosphate dikinase and phosphoenolpyruvate carboxylase, are extremely drought tolerant (Gu et al. 2013). Under the metabolic role, different modifications occur owing to drought in which the most significant is loss and decrease of synthesis of pigments in photosynthesis, thus decreasing energy and light harvesting, which play important roles in photosynthesis.
Yield and biomass have been closely related to alteration in the quantity of photosynthetic pigments (Miller et al. 2010).
Chlorophyll uptake the energy and translocate it into the reaction center of photosynthesis. In higher plants, chlorophyll a and b are generally located and are related to soil dehydration. Furthermore, supplementary pigments such as carotenoids have several roles in the chloroplast photosystem structure, photoprotection, and light harvesting and can moderately stimulate resistance in crops against drought. Fv/Fm (PSII quantum yield) increases and reductions in chlorophyll content have been identified in numerous reports in rice crop against drought stress (Hà 2014). Reductions in chlorophyll content and Fv/Fm have been detected lowest in autotetraploid families compared to diploid lineages in drought stress; therefore, it is suggested that autotetraploid rice is significantly tolerant of drought stress (Yang et al. 2014). Reductions in chlorophyll contents due to the degradation of biosynthesis systems of pigments resulting from stress and the concentration of lipid peroxidation are enhanced and the membrane of chloroplast is degraded.
9 The Biochemical Response of Rice Under Drought
In cytosol the osmotic potential is decreased due to the deposition of organic and inorganic solutes that balance the turgor in drought stress (Rhodes and Samaras 1994). The biochemical method is a kind of osmotic adjustment that is significantly based on water deficit potential. Osmotic adjustment occurs by the deposition of proline, sucrose, and glycine betaine, and in cytoplasm, other solutes stimulate the absorption of water during water shortage. Amino acids such as proline have been extensively studied because they have many roles in alleviating stress under harsh conditions. Deposition of carbohydrates are also induced by water deficiency (Shehab et al. 2010).
9.1 The Role of Proline Under Drought
As declared above, in plants under different unfavorable conditions, the proline works as an osmolyte (Verbruggen and Hermans 2008). In 1954, MacPherson and Kemble presented the first report on the function of proline after their work on rye grass where they observed the deposition of free proline reduced stress (Kemble and Macpherson 1954). In rice, the deposition of proline in stress and non-stress conditions have been investigated (Lum et al. 2014). Furthermore, proline reveals three major function under stress conditions, i.e., as antioxidant responsive molecules, metal chelators, and signaling molecules (Hayat et al. 2012; Roychoudhury et al. 2015). In drought stress, the deposition of proline may help restore the destruction via enhancing the activity of antioxidants (Fahramand et al. 2014). Therefore, the contents of proline can be utilized as a marker to select the rice genotypes for drought tolerance.
9.2 The Role of Antioxidants Under Drought
The difference between reducing and production of reactive oxygen species is the general process in drought stress (Faize et al. 2011). The ROS peroxide and protein are denatured by them, and they cause mutations in DNA, imbalance homeostasis in cells, oxidative cell damage, and lipid peroxidation. The antioxidant mechanism is complicated, comprising non-enzymatic and enzymatic molecules that defend plants alongside the unfavorable result of ROS. Ascorbate (AsA) and glutathione (GSH) serve as nonenzymatic antioxidants in the cell. Enzymatic antioxidants contain glutathione reductase (GR), catalase (CAT), guaiacol peroxidase (GPX), ascorbate peroxidase (APX), superoxide dismutase (SOD), dehydro ascorbate reductase (DHAR), and monodehydroascorbate reductase (MDHAR) (Noctor and Foyer 1998; Das and Roychoudhury 2014). In crops, these antioxidants have crucial reducing ROS mechanisms, and in rice, their regulations are enhanced for drought tolerance (Wang et al. 2005). In rice, due to enhanced drought stress, these antioxidants molecules have also increased AsA, APX, GR, SOD, phenylalanine ammonia lyase, CAT, MDHAR, DHAR, and GSH (Shehab et al. 2010). The tendency of these antioxidant protection enzymes to enhance their activity reveals their defensive action to respond to the oxidative damage stimulated by drought stress in rice. The actions of SOD, CAT, and POD can efficiently reduce ROS, and thus reduce the adverse effect of drought on rice (Lum et al. 2014; Yang et al. 2014).
9.3 The Role of Polyamines Under Drought
Polyamines (PAs) are partially positively charged molecules (Takahashi and Kakehi 2009) that play a role in the reaction of drought tolerance in plants (Calzadilla et al. 2014). In plants, the PAs contains putrescine (Put), spermidine (Spd), and spermine (Spm). Polyamines can intermingle with various signaling systems. Moreover, they control osmotic potential, membranes stabilization, and ionic homeostasis. In plants, enhanced PA concentration during drought is directly associated with reduced loss of water, enhanced limits of photosynthesis, and upgraded osmotic adjustment and detoxification (Roychoudhury and Das 2014), although the full mechanisms are poorly recognized. The functions of PAs include regulating gene expression by upholding ion equilibrium, helping the binding of transcription factors with DNA, stabilizing membranes, scavenging radicals, and inhibiting senescence by conformational change in protein phosphorylation and DNA (Martin-Tanguy 2001). A current report has confirmed that rice can stimulate the biosynthesis of PA, particularly Spd and Spm, and in leaves, unite them into an insoluble state before suppression in drought stress (Yang et al. 2007). Spray of PAs can decrease drought stress. Their utilization enhances WUE, accumulation of free proline, water potential in leaves, soluble phenolics, anthocyanins, net photosynthesis, and reduces oxidative destruction in cell membranes (Farooq et al. 2009).
10 Potential Solutions for Enhanced Yield Stability Under Drought Stress
Increasing productivity is an important part of rice cultivation. Previously, scientists determined that the helpful relations central to crop production and tolerance of drought in drought stress conditions may affects the maximum yield production (Guan et al. 2010). Additionally, several processes respond to numerous drought conditions by adaptive stress approaches, which are the important cause of differences for screening in drought stress (Kamoshita et al. 2008). For specific adaptation, plants need water accessibility, and it is suggested that escape from drought conditions is essential to minimize lethal stress and increase yield from the vegetative to reproductive stages (Blum 2005).
Formerly, studies have shown that plants display the morphology of desiccation avoidance in addition to a short leaf area and reduction of plant size because of the direct selection for yield under stress conditions and the plant ability is significantly reduced (Gu et al. 2012). The enhancement in productivity of yield and production of breeding tools is hindered due to complications regarding GY caused by genetic regulation and many other main features such as raceme, number of tillers, and grains reproductivity (Dixit et al. 2014).
11 Molecular Analysis of the DRO1 Gene for Improved Root Traits
The ability of Oryza sativa to originate under stress conditions is possible because of deep root expansion that enables a method for the water supplies and avoidance processes. The population for breeding is attained via the cross of shallow roots of IR64 and deep roots of Kinandang Patong, which have the ability to hold under drought stress, and deeper rooting 1 (DRO1) QTL is detected in the population (Uga et al. 2013). In the DRO1 gene, the 1 bps altered at the four-coding sequence can change into an antisense codon and result in smaller protein synthesis, diminishing the root angle; hence, contradictory to the Kinandang Patong allele, resulting in the descending growth expansion via DRO1kp in harsh drought conditions. The improvement in the yield implementation and sensitive characteristics of roots can be demonstrated as supportive to enhance the drought avoidance via the cross between DRO1-kp and IR64 genes in contrast to inheritor parents in drought conditions. The auxin stimulates the root angel, root elongation, and gravitropism for maximum yield, while the DRO1 gene is too adversely regulated by auxin.
12 Genetic Engineering for Drought Tolerance
Genetic engineering can successfully improve yield by genome editing rice to enhance drought tolerant mechanisms. The genetically modified rice and transformation of external genes in rice and promoters for efficient productivity are mentioned in Table 1. The target genes were identified by studying the response meantime of various stress conditions, recognized as possible genes that play a role in tolerance under stress during flowering.
12.1 AP2/ERF Transcription Factors
In Oryza sativa, the photosynthetic process has a multifaceted influence on yield enhancement under stress conditions because it depends on three important steps of resistance: the layer of mesophyll, the stomata (Lauteri et al. 2014), and the ability of the plant to use sunlight. The system for regulation of genes and the biochemistry of the photosynthetic carbon process are associated with transcriptional factors such as maximum rice yield and AP2/ERF. The photosynthetic process can be maximum due to additional expression of the HYR gene in nippobare because of regulation via CaMV355 promoters to locate the physiological and structural processes that also play a part in improving grain yield under harsh conditions. The essential role of the HYR gene is identified as the activation of gene and highly impacts on PSII, which is connected to the carbon metabolic processes (Ambavaram et al. 2014). The additional AP2/ERF and OSP37 gene expression stimulates drought tolerance in genetically modified plants in vegetative stages. Transgenic plants compared to wild plants are more effective at displaying the maximum yield after displaying a deficiency of water conditions; essentially, this is due to improved grain filling. Consequently, it is suggested that grain yield can be improved by OSAP37 during the period of stress and there is no risk of undesirable expression of growth (Oh et al. 2009). DREB/CBF are identified as the transcriptional factors that switch the stress stimulating genes according to the support of CRT/DRE factors and also regulate the minimum disturbance in photo-oxidative and maximum accumulation of ions that control the osmotic potential resulting from overexpression under drought tolerance (Datta et al. 2012).
The overexpression of DREBIA of genetically modified plants of Arabidopsis and rice displays maximum drought resistance at the vegetative phase with maximum efficiency spikelet in contrast with non-transgenic crop (Datta et al. 2012; Ravikumar et al. 2014). A1DREBIC transgenic plants show maximum drought tolerance at both reproductive and vegetative phases and have maximum sink ability and improve yield productivity compared to normal drought treated plants (Ishizaki et al. 2013).
12.2 bZIP Transcription Factors
Studies have exposed that binding of responsive factors such as AREB/ABRE/ABI with bZIP transcriptional factors seriously influences ABA biosynthesis under drought tolerance (Fukao and Xiong 2013; Banerjee and Roychoudhury 2017). In genetically modified rice, the OSb21P23 overexpression gene is a maximum functional for ABA biosynthesis, it stimulates the drought tolerance during all the reproductive and vegetative phases under ideal conditions, and the GY/plant was maximum compared to transformed and non-mutant plants. The microarray method releases maximum consecutive genes alongside their role, approximately 30, which have a putative mission associated with dehydrins and transcriptional factors (Xiang et al. 2008). OsbZIP46 is an important transcriptional factor that has consecutive ranges of amino acids such as 122–129 residues that ultimately influences challenging the genes of activation at various locus. Drought tolerance was observed along with GY by eliminating one more gene from over expression in transgenic rice than local. ABF is phosphorylated in Arabidopsis with SnRK2 and Osb21p46 (Todaka et al. 2015). OsbZ1023 and Osb21p46 play vital roles as positive regulators in the ABA pathway. Over expression of Osb21p71 leads to enhanced drought tolerance in CaMV355 along with better seed setting. Furthermore, drought tolerance can also be achieved by gene stacking of COR413-IMI, OSNHX1, and OsMYB4 (Liu et al. 2014). Research reveals that bZIP transcriptional factors could be the best choice to obtain high yield by gene editing under drought stress conditions. The mechanism of tolerance is regulated by signaling of ABA, which is exhibited by the maximum deposition of ABRE and ABA at the promotor positions of responsive genes (Maruyama et al. 2011, 2014).
12.3 NAC Transcription Factors
NAC is studied as a restricted class of transcriptional factors taking the various c-terminal positions that have been identified as carrying out the vital role in stress conditions to control the molecular and metabolism mechanisms (Nuruzzaman et al. 2010). The overexpression of SNACI in genetically modified rice displays improvement under drought tolerance at reproductive and vegetative phases and displays maximum spikelet and seeds productivity compared with non-mutant plants having no yield. Expression factor OSROIc is utilized to produce the transformed plant that has maximum resilience under drought conditions and to protect the loss of water by stomata closure, but the photosynthetic mechanism remains constant (You et al. 2012). Under ideal conditions, the production of maximum yield and resistance under drought stress at the sexual stage is only supported by some factors such as OSNAC5, OSNAC9, and OSNAC10 overexpression in rice having promotors for particular roots. The microarray method described the consecutive function of gene encoding associated with p450 zinc-finger, transporting Ca-ATPase, and non-regulated HAK5 and NCED (Jeong et al. 2010; Redillas et al. 2012). Another transcriptional factor of NAC is OSNAP that transports due to inorganic components such as stress, ABA, and salt. In rice, overexpression stimulates drought resistance, reduces the loss of water and growth at the vegetative stage, and improves yield and increases the number of spikelets at reproductive stages. OSNAP functions similar to the other transcriptional factors such as OSAP37, OSMYB2, and OSDREBIA and some dissimilar genes, for example, OSPP2C68 and OSPP2C66/OSAB12 (Chen et al. 2014).
12.4 Other Transcription Factors
Studies of transcriptional factors in mutant rice plants have shown improved maximum drought resistance and maximum yield potentials. Transcription factor homodomain leucine is coded by the EDT1/HDG11 gene, and in rice, the gene id is transformed by the Arabidopsis gene, resulting in its overexpression of genes exhibiting maximum drought resistance. Drought tolerance is related to efficient root systems, improvement in WUE, and minimal stomatal opening to accommodate the water deficit conditions. The enhancement of grain yield in transformed plants is only probable via the healthier seed formation and maximum rate of photosynthesis (Yu et al. 2013). The Zhengsheng 97B and IRAT109 upland are hybridized to grow QTL drought resistant in the fourth position of the chromosome that remains on OSGRAS23, which is related to the GRAS family (Zou et al. 2005). Inorganic aspects such as temperature and salt stress are related to the expression of OSGRA523 and support reducing the accumulation of hydrogen peroxide. The transformed plants exhibited the maximum horizon rate of seed and enhanced the yield under drought stress (Xu et al. 2015). OSWRKY47 transforms under drought resistant conditions and increases the harvest. OSNRKY47 expression also controls the status of drought resistance through the support of controlling genes CaMBP and CRRSP (Raineri et al. 2015).
13 Drought Protective Genes
13.1 Protein Kinases
In the relationship of the CIPK class with OSCIPK23, a portion of CBL most perspicuous in the pollen and ovule is promoted by the support of hormones such as ABA and abiotic factors (Pandey et al. 2015; Banerjee and Roychoudhury 2017). RNAi regulate the OSCIPK23 in rice and show the prone response under drought conditions at the vegetative growth stage and reduce the formation of seeds at the reproductive stage, exhibiting an important function under fertilization and drought tolerance (Yang et al. 2008). Reporter enzyme GUDK works in cytosol for phosphorylation and is identified from the estimation of drought sensitive mutant genes that show the prone response in inorganic stress. GUDK can be the main reason for reduced biomass and photosynthesis at the proliferation stage. It also reduces the quantity of yield at the reproductive stage under drought stress.
According to phosphoproteome analysis, GUDK is the main regulator of OSAP37, which is responsible for the GY parameter in rice (Ramegowda et al. 2014). Mitogen is triggered by the enzyme, which is reflected to be an essential factor in the plant growth adjustment and consequences of stress (Sinha et al. 2011). DSMI-RNA lineages are recognized as considerably functional in drought during the development of flowering contributing to a lateral decrease in the number of spikelets and consequently a yield reduction. Scavenging and ROS are utilized to develop the overexpression lineages of maximum drought resistance (Ning et al. 2010). The estimated T-DNA transformed are utilized to recognize the salt receptive genes that code for inositol 1,3,4 triphosphate 516 kinase. Under drought conditions, accumulation of osmotic ions, productivity of spikelets, and yield is reduced because of transformed genes. Identical expressions have been discovered associated with the overexpression lineages. To determine the entire result for the essential drought tolerance, the precise expression conditions are required (Du et al. 2011).
13.2 LEA Protein
Late embryogenesis abundant protein constitutes a significant portion of hydrophilic proteins that are involved in functions during stress conditions (Todaka et al. 2015). The OSEA 3-1 gene exhibited maximum expression in rice whilst drought tolerance hinders its regulation under stress inducing factors. It also improves the amount of yield and spikelet efficiency compared to non-mutant plants (Xiao et al. 2007).
13.3 Osmoprotectant Synthesis
The drought producing radicals can be countered with the support of osmolytes and antioxidants. For instance, proline is accumulated as a defensive mechanism as the consequence of loss due to initiation of stress. OSOAT, a kind of pyridoxal-5′-phosphate, is dependent on the biochemistry of arginine and proline. Abiotic factors and plant hormones can trigger the production of OSOAT. An extreme quantity of OSOAT results in extreme tolerance regarding storing water and oxidative stress and improves the seed setting. A significant portion of the GY attribute in transformed plants is not protected from drought stress. OSAT is targeted by SNAC2. The scavenging of ROS and improvement of proline and antioxidant enzymes lead to overexpression of OSOAT (You et al. 2012). APX is the anti-oxidant that helps to maintain ROS and triggers the dismutation regarding water and oxygen. OSAPX overexpression increases productivity in rice in contrast to the native plant under stress conditions (Zhang et al. 2013).
13.4 Other Genes
At the posttranslational level, Famesy transferase contains two subdivisions as a result of improvement in protein with the support of stimulating the femesyle group at the thiol S–H group of cysteine. The synthesis of isoprenoid based on SOS occurs in ER and helps to trigger the development of catabolism where 2-femsyle pyrophosphate is transformed into squalene, which directly contributes to the production of sterol. RNAi bridges the gap between maize SOS and rice SOS and boosts the yields over natural plants. Drought tolerance might be improved by excluding loss of water and fewer open stomata (Manavalan et al. 2011). Lipid transferase triggers the mechanism of phospholipid transferring in membrane and has some significance concerning the protection of plant and formation of embryo (Kader 1996). LIP OSDIL have more expression characteristics that display maximum tolerance during proliferation stages with the decline of alternative nutritious tissues at the reproductive stage where ABA decreases the enzymes that stimulate the synthesis of ABA and tolerate the level of self-expression for downregulating the OSDIL gene (Guo et al. 2013). SKIP proteins are formed from the crossing of onco (cancer) and yeast genes, while V-SKI exhibits the traits such as fragments of spliceosome and supports the regulation of transcription OSKIPa. In humans, SKIP proteins are similar to rice proteins. This protein is competently relevant to alter the hormones of plant and stress initiating features. During the production of seeds, the plant shows maximum self-expression of OSKIPa, with enhanced spikelet efficiency and amount of yield at reproductive stage. In transformed rice, OSKIPs influences the drought tolerance that is related to tremendous ROS scavenged mechanisms and stressed regulated genes (Hou et al. 2009). In rice crop, the SAP protein has domain A20/ANI that significantly contributes to drought tolerance. OSAP8 protein exhibits its expression to cause drought resistance during the production of flowering lacking any reduction of yield in contrast to non-stressed transformed plants (Kanneganti and Gupta 2008). The plants exhibit enhanced response in drought stress due to degradation or synthesis of RNA by regulating post transcriptional activities.
The role of RBP during the stress tolerance process and modification of the protein is identified by the AtGRP2 gene overexpression, and transformed plants have the sequence of protein for maximum RBP glycine. The genes that are sensitive to stress are DB21, DIP, and OSE2, where DIP controls improvement in seeds via the ATGRP overexpression in mutant plants (Yang et al. 2014). Seven genes are responsible for promoting the stress resistance in which kinase enzymes are coded by the transcriptional factors SOS2, CBF3/DREBIA, and AP2ERF, while synthesis of ABA occurs by LOS5 and NCED2 genes. Kinase enzymes are also coded by NPK1 and ZAT10 that work similar to a Zn-transcriptional factor. Overexpression of NHX1 antiporters Na+/H+ are regulated by the promotors that are stress inducers. The spikelet efficiency and maximum yield are displayed by the PVC tubes in transformed plants. The transformed plants both in field and PVC tubes demonstrate better overexpression of ZAT10 and LOS5 in contrast with the seven genes (Xiao et al. 2009).
14 Plant Hormones Affecting Drought Tolerance
In plants, the signals that transmit the stimulus for better growth and response against abiotic stress are regulated by phytohormones that effectively create a complex network structure and establish signaling molecules (Kohli et al. 2013). In drought stress, different hormones such as GAs, CKs, ethylene, and ABA have a significant role in plants (Wilkinson et al. 2012). In plant body, different mechanisms take place such as elongation of shoot and root during the vegetative stage, morphology, translating phytohormone signals, modification in gases, and relation of water contents, which have been described with detailed methods (Peleg et al. 2011). Moreover, the plant hormones are limited for controlling the traits that are related to yield. The study impact on various hormones, relationships, and protections during the period of sexual stress is a maximum developing trend at this time; mostly, GY is inclined. Drought can induce sterility in pollen at the pre-flowering phase, which can be terminated, and post-flowering stage sink-source association influences the filling of grain (Xing and Zhang 2010).
14.1 Abscisic Acid
Closing and opening of stomata are regulated by ABA hormones, which help to store water in drought stress during the vegetative stage (Schroeder et al. 2001). Under drought stress, the amount of ABA is increased, and it can inhibit the transcription by stomata closure that induces the complication at sexual stage stress (Roychoudhury and Paul 2012). To achieve the improvement in the GY via ABA remains a challenge because stomata closure reduces the uptake of C and ABA causes abortion and sterility (Ji et al. 2011). In wheat, the concentration of ABA was shown to remain at anthers during sexual stress. The species of wheat that have the enhancing enzyme that degrades the ABA effect resulting in minimum accumulation of ABA in anther show the first feedback in drought stress (Ji et al. 2011).
Still, maximum production of ABA may inhibit the production of invertase enzymes that are essential to maintain the tendency of sink resulting in the spoiling of pollens. In sexual stress, ABA can be exploited to improve the GY, except controlling of stomata. For instance, ABA can alter the process of sap in plant by improving the aquaporin competences, and osmotic adjustment measures the sustained growth in the propagative stress (Tardieu et al. 2010; Travaglia et al. 2010). However, the ABA play a significant role in the flowering developing stage. Earlier reports investigate whether grain and ABA are directly linked to each other. The applications of ABA expose that there is a similarity between the ABA and drought stimulating the storage of C in seeds and filling of grain (Yang et al. 2004). Deposition of ABA in leaves and roots is connected with the reprocess of deposited carbon in drought (Yang and Zhang 2010). More studies are required to improve the drought resistant value with the support of ABA maintenance through making some adjustments in ABA metabolism; however, thus far the researchers have not yet been able to produce the maximum improvement in plants that directly influence GY. For instance, ABA regulated genes OSNAC5 and OSNAP and falling of leaves produce some improvement in drought at the anthesizing phase (Chen et al. 2014; Liang et al. 2014). Overexpression is increased in some genes such as OSPP2C68, OSPP2C06/OSAP12, and OSPP09, which are responsible for signaling of ABA. Signaling of ABA can improve the interaction between source and sink in the period of grain stuffing. It is expected that sequences of hydrolases of B-carotene are coded by the DSM gene, which controls the synthesis of zeaxanthin, which is a precursor of biogenesis of ABA. Mutant T-DNA shows the sensitivity against drought stress due to its minimum photosynthesis and reductions in yield in stress. DSM2 shows overexpression to offer the interference for maximum drought tolerance by boosting the pigment of xanthophyll in plants for improvement of spikelet efficiency and seed arrangement (Du et al. 2010).
14.2 Cytokinin
Cytokinin is another phytohormone, which is responsible for differentiation and division of cells in plants. Additionally, it has improved the time of maturity. Under water deficit, it increases the range of aging and death in young leaves. This quality is more efficient in filling of grain and reproductive stress. Concentration of cytokines improves the rice growth for a short time during the filling in grain phase (Yang et al. 2001). The worth of transformed rice cultivars and improvement in GY are increased by the spray of 6-benzylaminopurine, a type of cytokinin (Pan et al. 2013). When biogenesis of CKs occur, the plant becomes more resistant to stress (Kuppu et al. 2013). Expression of IPT in drought and promotors MI improve the GY after and before stress of flowering because it increases the feedback of stress (Peleg and Blumwald 2011). Transformed plants displayed the maximum expression of brassinosteroids (BRs) in consistent gene and reduced expression of gene in JA jasmonate (JA). When expressed, cytokine alleviates the adverse effect of jasmonate in maximum yield productivity.
14.3 Gibberellic Acid
Under drought stress conditions, development of plants is maintained by GA (Colebrook et al. 2014), and DELLl protein releases severe signaling of GA (Davière and Achard 2013). It is accepted that in the pre-filling of grain stage there is a maximum deposition of GA in contrast with the post-filling stage. This promotes the healthy development of embryo, which explains the important function of GA in the development of embryo. In addition, a deficit of water significantly reduced the synthesis of GA. There is an adverse relationship between components of GA and filling of grain (Yang et al. 2004). SNPs have been utilized to examine the adverse relationship between filling of grain and components of GA in propagative stress by utilizing the crossed lines (IR64 × Cabacu) that describe the position adjacent to the sd1 locus and functional sd1 gene GA 20-oxidase influence on GA and GY (Trijatmiko et al. 2014). Through the support of drought resilient alleles, the partial short green rotation with the sd1-inherited structure is formed and positions of qDTY1.1 regarding GY and QTL under drought conditions resist and collapse via hybridization (Vikram et al. 2015).
14.4 Auxin
Auxin is classified as an essential hormone involved in plant body development, including shoots and roots, along with the CKs phytohormone (Su et al. 2011). Meanwhile, in rice, filling of grain depends on the production of auxin, where maximum auxin leads to early filling of grain, while minimum production of auxin leads to less filling of grain; therefore, it is recommended that the concentration of auxin should be maximum early in the filling of grain phase to maintain its quantity (Tamaki et al. 2015). Under drought conditions, at the time of biogenesis, auxin downregulation is linked with genes significances in the minimum assembly of auxin constituents (Du et al. 2011). During the signaling, several genes are convoluted for auxin biosynthesis and are supposed to connect under stress, but its influence on GY remains unclear (Peleg and Blumwald 2011; Zhang et al. 2012). The genes that regulate root elongation are not associated with the progressive regulation by auxin, while it is predictable at the DRO1/QTL position. DRO1 controls drought avoidance in fewer root crops and high yield productivity under stress conditions (Uga et al. 2013).
14.5 Ethylene
Ethylene is an extremely important phytohormone that is responsible for falling of leaves, maturity, and response in waterlogging conditions (Perata and Voesenek 2007). Additionally, during drought stress, root elongation will be stopped by the maximum production of ethylene, and it will also reduce leaf enlargement and other physiology mechanisms such as photosynthesis (Sharp 2002). Excess concentration of ethylene promotes the abortion of embryo and reduces the degree of grain contents (Tamaki et al. 2015). During the early grain stuffing stage, ethylene can promote improvement in rice seeds (Yang et al. 2004). The ethylene displays an inverse relation to grain filling percentage. It is proposed that ABA and ethylene both have an antagonistic function regarding each other, and their effects depend on equilibrium between production of ethylene and ABA to advance the grain stuffing percentage (Zhu et al. 2011).
14.6 Jasmonic Acid
This plant hormone has implications associated with development and defense reactions (Turner et al. 2002). However, its functions and physiognomies are not recognized against stress. Endogenous of JA may be increased in stress (Du et al. 2011). Under propagative stress, the MeJA level is upgraded 19-fold in rice and produces a minimum GY in rice (Kim et al. 2009). The AtJMT gene is related to the advancement of the MeJA level in flowering bunches in Arabidopsis. Lower spikelet efficiency and filling percentage stimulate the reduction in Ubil:AtJMT via GY. Transformed plants comprise the number of spikelets relating to the lower part of flowers, ovule and anther. All these features are the replication of the maximum MeJA level, and it reduces the development of spikelet.
15 Future Trends in Research
Accomplishing improvements in drought resilient crops is a very complex achievement that requires detailed information regarding the formation of phenotype, biochemical, physiological, morphological characteristics, as well as the genotype of plants. A number of drought tolerant crops are produced for survival under drought stress; water status should influence profound rooting, moderation in metabolism should occur, and vital antioxidant and osmotic ions are essential. Undesirable signals are interfered with by signaling mechanisms of phytohormones. Therefore, an optimum and sustained yield is the chief aim of investigations to produce drought resistant crops. However, the integrative plan to link physiology, quantitative genetics, and omics, as recommended above, has been followed by only a few research programs. Rice cultivars are selected for their current sensitive characteristics to produce maximum drought resistance from current QTL to help identify gene areas associated with drought tolerance under stress conditions. Exact phenotyping is very critical to identify the superior collections/core mapping populations to identify the accurate QTLs and isolate genes of interest to use in plant breeding. Detecting the yield productivity under stress is a complex phenomenon, which would be the consequence of several characteristics and it would also be obliged to exhibit its self-expression to identify the primary provider involved in selecting the position under stress conditions; yet it is not a suitable method for production of maximum yield of drought resistance crops. Under several environmental conditions, QTL mapping effects on GY have been realized through molecular methods results. However, the molecular environment is also being utilized, with the editing of genome and functional genes being exposed in the highest production of crops under stress conditions. To recognize the various reaction mechanisms and control field yields, the studies should be sustained on a widespread level. Recent advancements in marker development genome analysis sequencing have motivated the rethinking of the mechanisms of creating populations suitable for, as well as to challenge the precise players in, drought tolerance. Consequently, to obtain the maximum yield production and information regarding the plant breeding section, genotype and physiology at the genetic level is essential. Hence, development of cheap and rapid measures to explore drought response characteristics will effectively enhance genetic resolution by integrating molecular genetics, crop physiology, and breeding approaches.
References
Ahmad I, Mian A, Maathuis FJM (2016) Overexpression of the rice AKT1 potassium channel affects potassium nutrition and rice drought tolerance. J Exp Bot 67(9):2689–2698. https://doi.org/10.1093/jxb/erw103
Ambavaram MM, Basu S, Krishnan A, Ramegowda V, Batlang U, Rahman L et al (2014) Coordinated regulation of photosynthesis in rice increases yield and tolerance to environmental stress. Nat Commun 5:5302
Appels R, Eversole K, Stein N, Feuillet C, Keller B, Rogers J et al (2018) Shifting the limits in wheat research and breeding using a fully annotated reference genome. Science 361(6403):eaar7191. https://doi.org/10.1126/science.aar7191
Aprile A, Mastrangelo AM, De Leonardis AM, Galiba G, Roncaglia E, Ferrari F et al (2009) Transcriptional profiling in response to terminal drought stress reveals differential responses along the wheat genome. BMC Genomics 10(1):279. https://doi.org/10.1186/1471-2164-10-279
Banerjee A, Roychoudhury A (2016) Group II late embryogenesis abundant (LEA) proteins: structural and functional aspects in plant abiotic stress. Plant Growth Regul 79:1–17
Banerjee A, Roychoudhury A (2017) Abscisic-acid-dependent basic leucine zipper (bZIP) transcription factors in plant abiotic stress. Protoplasma 254:3–16
Banerjee A, Roychoudhury A (2018) Regulation of photosynthesis under salinity and drought stress. In: Singh VP, Singh S, Singh R, Prasad SM (eds) Environment and photosynthesis: a future prospect. Studium Press, India, pp 134–144
Basu S, Roychoudhury A (2014) Expression profiling of abiotic stress-inducible genes in response to multiple stresses in rice (Oryza sativa L.) varieties with contrasting level of stress tolerance. BioMed Res Int 2014:706890
Basu S, Roychoudhury A, Saha PP, Sengupta DN (2010a) Differential antioxidative responses of indica rice cultivars to drought stress. Plant Growth Regul 60:51–59
Basu S, Roychoudhury A, Saha PP, Sengupta DN (2010b) Comparative analysis of some biochemical responses of three indica rice varieties during polyethylene glycol-mediated water stress exhibits distinct varietal differences. Acta Physiol Plant 32:551–563
Bengough AG, McKenzie BM, Hallett PD, Valentine TA (2011) Root elongation, water stress, and mechanical impedance: a review of limiting stresses and beneficial root tip traits. J Exp Bot 62(1):59–68. https://doi.org/10.1093/jxb/erq350
Bevan MW, Uauy C, Wulff BBH, Zhou J, Krasileva K, Clark MD (2017) Genomic innovation for crop improvement. Nature 543(7645):346–354. https://doi.org/10.1038/nature22011
Blum A (2005) Drought resistance, water-use efficiency, and yield potential—are they compatible, dissonant, or mutually exclusive? Aust J Agric Res 56(11):1159–1168
Bolger ME, Weisshaar B, Scholz U, Stein N, Usadel B, Mayer KFX (2014) Plant genome sequencing—applications for crop improvement. Curr Opin Biotechnol 26:31–37. https://doi.org/10.1016/j.copbio.2013.08.019
Cai R, Zhao Y, Wang Y, Lin Y, Peng X, Li Q et al (2014) Overexpression of a maize WRKY58 gene enhances drought and salt tolerance in transgenic rice. Plant Cell Tissue Organ Cult 119(3):565–577. https://doi.org/10.1007/s11240-014-0556-7
Cai S, Jiang G, Ye N, Chu Z, Xu X, Zhang J, Zhu G (2015) A key ABA catabolic gene, OsABA8ox3, is involved in drought stress resistance in rice. PLoS One 10(2):e0116646. https://doi.org/10.1371/journal.pone.0116646
Calzadilla PI, Gazquez A, Maiale SJ, Ruiz OA, Bernardina MA (2014) Polyamines as indicators and modulators of the abiotic stress in plants. In: Plant adaptation to environmental change: significance of amino acids and their derivatives. CABI, Wallingford, UK, pp 109–128
Chakhchar A, Haworth M, El Modafar C, Lauteri M, Mattioni C, Wahbi S, Centritto M (2017) An assessment of genetic diversity and drought tolerance in argan tree (Argania spinosa) populations: potential for the development of improved drought tolerance. Front Plant Sci 8:276. https://doi.org/10.3389/fpls.2017.00276
Chen X, Wang Y, Lv B, Li J, Luo L, Lu S et al (2014) The NAC family transcription factor OsNAP confers abiotic stress response through the ABA pathway. Plant Cell Physiol 55(3):604–619
Chen G, Liu C, Gao Z, Zhang Y, Jiang H, Zhu L et al (2017) OsHAK1, a high-affinity potassium transporter, positively regulates responses to drought stress in rice. Front Plant Sci 8:1885. https://doi.org/10.3389/fpls.2017.01885
Chen J, Qi T, Hu Z, Fan X, Zhu L, Iqbal MF et al (2019) OsNAR2.1 positively regulates drought tolerance and grain yield under drought stress conditions in rice. Front Plant Sci 10:197. https://doi.org/10.3389/fpls.2019.00197
Colebrook EH, Thomas SG, Phillips AL, and Hedden P (2014) The role of gibberellin signallingin plant responses to abiotic stress. J Exp Biol 217(1):67–75. https://doi.org/10.1242/jeb.089938
Cui Y, Wang M, Zhou H, Li M, Huang L, Yin X et al (2016) OsSGL, a novel DUF1645 domain-containing protein, confers enhanced drought tolerance in transgenic rice and Arabidopsis. Front Plant Sci 7:2001. https://doi.org/10.3389/fpls.2016.02001
Das K, Roychoudhury A (2014) Reactive oxygen species (ROS) and response of antioxidants as ROS-scavengers during environmental stress in plants. Front Environ Sci 2:53
Datta K, Baisakh N, Ganguly M, Krishnan S, Yamaguchi Shinozaki K, Datta SK (2012) Overexpression of Arabidopsis and rice stress genes’ inducible transcription factor confers drought and salinity tolerance to rice. Plant Biotechnol J 10(5):579–586
Davière J-M, Achard P (2013) Gibberellin signaling in plants. Development 140(6):1147–1151
Dixit S, Mallikarjuna Swamy BP, Vikram P, Bernier J, Sta Cruz MT, Amante M et al (2012) Increased drought tolerance and wider adaptability of qDTY 12.1 conferred by its interaction with qDTY 2.3 and qDTY 3.2. Mol Breed 30(4):1767–1779. https://doi.org/10.1007/s11032-012-9760-5
Dixit S, Singh A, Kumar A (2014) Rice breeding for high grain yield under drought: a strategic solution to a complex problem. Int J Agron 2014:863683
Dou M, Fan S, Yang S, Huang R, Yu H, Feng X (2016) Overexpression of AmRosea1 gene confers drought and salt tolerance in rice. Int J Mol Sci 18(1):2. https://doi.org/10.3390/ijms18010002
Du H, Wang N, Cui F, Li X, Xiao J, Xiong L (2010) Characterization of the β-carotene hydroxylase gene DSM2 conferring drought and oxidative stress resistance by increasing xanthophylls and abscisic acid synthesis in rice. Plant Physiol 154(3):1304–1318
Du H, Liu L, You L, Yang M, He Y, Li X, Xiong L (2011) Characterization of an inositol 1,3,4-trisphosphate 5/6-kinase gene that is essential for drought and salt stress responses in rice. Plant Mol Biol 77(6):547–563
Du H, Wu N, Cui F, You L, Li X, Xiong L (2014) A homolog of ETHYLENE OVERPRODUCER, OsETOL1, differentially modulates drought and submergence tolerance in rice. Plant J 78(5):834–849. https://doi.org/10.1111/tpj.12508
Du Q, Zhao M, Gao W, Sun S, Li W-X (2017) microRNA/microRNA* complementarity is important for the regulation pattern of NFYA5 by miR169 under dehydration shock in Arabidopsis. Plant J 91(1):22–33. https://doi.org/10.1111/tpj.13540
El-Esawi M, Alayafi A (2019) Overexpression of rice Rab7 gene improves drought and heat tolerance and increases grain yield in rice (Oryza sativa L.). Genes 10(1):56. https://doi.org/10.3390/genes10010056
Fahramand M, Mahmoody M, Keykha A, Noori M, Rigi K (2014) Influence of abiotic stress on proline, photosynthetic enzymes and growth. Int Res J Appl Basic Sci 8(3):257–265
Faize M, Burgos L, Faize L, Piqueras A, Nicolas E, Barba-Espin G et al (2011) Involvement of cytosolic ascorbate peroxidase and Cu/Zn-superoxide dismutase for improved tolerance against drought stress. J Exp Bot 62(8):2599–2613
Farooq M, Wahid A, Lee D-J (2009) Exogenously applied polyamines increase drought tolerance of rice by improving leaf water status, photosynthesis and membrane properties. Acta Physiol Plant 31(5):937–945
Farooq M, Kobayashi N, Ito O, Wahid A, Serraj R (2010) Broader leaves result in better performance of indica rice under drought stress. J Plant Physiol 167(13):1066–1075. https://doi.org/10.1016/j.jplph.2010.03.003
Ferrero-Serrano Á, Assmann SM (2016) The α-subunit of the rice heterotrimeric G protein, RGA1, regulates drought tolerance during the vegetative phase in the dwarf rice mutant d1. J Exp Bot 67(11):3433–3443. https://doi.org/10.1093/jxb/erw183
Feuillet C, Leach JE, Rogers J, Schnable PS, Eversole K (2011) Crop genome sequencing: lessons and rationales. Trends Plant Sci 16(2):77–88. https://doi.org/10.1016/j.tplants.2010.10.005
Fleury D, Jefferies S, Kuchel H, Langridge P (2010) Genetic and genomic tools to improve drought tolerance in wheat. J Exp Bot 61(12):3211–3222. https://doi.org/10.1093/jxb/erq152
Fu J, Wu H, Ma S, Xiang D, Liu R, Xiong L (2017) OsJAZ1 attenuates drought resistance by regulating JA and ABA signaling in rice. Front Plant Sci 8:2108. https://doi.org/10.3389/fpls.2017.02108
Fukai S, Cooper M (1995) Development of drought-resistant cultivars using physiomorphological traits in rice. Field Crop Res 40(2):67–86. https://doi.org/10.1016/0378-4290(94)00096-u
Fukao T, Xiong L (2013) Genetic mechanisms conferring adaptation to submergence and drought in rice: simple or complex? Curr Opin Plant Biol 16(2):196–204
Gao T, Wu Y, Zhang Y, Liu L, Ning Y, Wang D et al (2011) OsSDIR1 overexpression greatly improves drought tolerance in transgenic rice. Plant Mol Biol 76(1–2):145–156. https://doi.org/10.1007/s11103-011-9775-z
Gu J, Yin X, Stomph T-J, Wang H, Struik PC (2012) Physiological basis of genetic variation in leaf photosynthesis among rice (Oryza sativa L.) introgression lines under drought and well-watered conditions. J Exp Bot 63(14):5137–5153
Gu J-F, Qiu M, Yang J-C (2013) Enhanced tolerance to drought in transgenic rice plants overexpressing C4 photosynthesis enzymes. Crop J 1(2):105–114. https://doi.org/10.1016/j.cj.2013.10.002
Guan Y, Serraj R, Liu S, Xu J, Ali J, Wang W et al (2010) Simultaneously improving yield under drought stress and non-stress conditions: a case study of rice (Oryza sativa L.). J Exp Bot 61(15):4145–4156
Guo C, Ge X, Ma H (2013) The rice OsDIL gene plays a role in drought tolerance at vegetative and reproductive stages. Plant Mol Biol 82(3):239–253
Hà PTT (2014) Physiological responses of rice seedlings under drought stress. J Sci Dev 12(5):635–640
Hadiarto T, Tran L-SP (2010) Progress studies of drought-responsive genes in rice. Plant Cell Rep 30(3):297–310. https://doi.org/10.1007/s00299-010-0956-z
Hampton M, Xu WW, Kram BW, Chambers EM, Ehrnriter JS, Gralewski JH et al (2010) Identification of differential gene expression in Brassica rapa nectaries through expressed sequence tag analysis. PLoS One 5(1):e8782. https://doi.org/10.1371/journal.pone.0008782
Hayat S, Hayat Q, Alyemeni MN, Wani AS, Pichtel J, Ahmad A (2012) Role of proline under changing environments: a review. Plant Signal Behav 7(11):1456–1466
Hiremath PJ, Farmer A, Cannon SB, Woodward J, Kudapa H, Tuteja R et al (2011) Large-scale transcriptome analysis in chickpea (Cicer arietinum L.), an orphan legume crop of the semi-arid tropics of Asia and Africa. Plant Biotechnol J 9(8):922–931. https://doi.org/10.1111/j.1467-7652.2011.00625.x
Hong Y, Zhang H, Huang L, Li D, Song F (2016) Overexpression of a stress-responsive NAC transcription factor gene ONAC022 improves drought and salt tolerance in rice. Front Plant Sci 7:4. https://doi.org/10.3389/fpls.2016.00004
Hou X, Xie K, Yao J, Qi Z, Xiong L (2009) A homolog of human ski-interacting protein in rice positively regulates cell viability and stress tolerance. Proc Natl Acad Sci U S A 106(15):6410–6415
Hu T, Zhu S, Tan L, Qi W, He S, Wang G (2016) Overexpression of OsLEA4 enhances drought, high salt and heavy metal stress tolerance in transgenic rice (Oryza sativa L.). Environ Exp Bot 123:68–77. https://doi.org/10.1016/j.envexpbot.2015.10.002
Huang L, Hong Y, Zhang H, Li D, Song F (2016) Rice NAC transcription factor ONAC095 plays opposite roles in drought and cold stress tolerance. BMC Plant Biol 16(1):203. https://doi.org/10.1186/s12870-016-0897-y
Huang L, Wang Y, Wang W, Zhao X, Qin Q, Sun F et al (2018) Characterization of transcription factor gene OsDRAP1 conferring drought tolerance in rice. Front Plant Sci 9:94. https://doi.org/10.3389/fpls.2018.00094
Ishizaki T, Maruyama K, Obara M, Fukutani A, Yamaguchi-Shinozaki K, Ito Y, Kumashiro T (2013) Expression of Arabidopsis DREB1C improves survival, growth, and yield of upland New Rice for Africa (NERICA) under drought. Mol Breed 31(2):255–264
Jaleel CA, Manivannan P, Wahid A, Farooq M, Al-Juburi HJ, Somasundaram R, Panneerselvam R (2009) Drought stress in plants: a review on morphological characteristics and pigments composition. Int J Agric Biol 11(1):100–105
Jeong JS, Kim YS, Baek KH, Jung H, Ha S-H, Do Choi Y et al (2010) Root-specific expression of OsNAC10 improves drought tolerance and grain yield in rice under field drought conditions. Plant Physiol 153(1):185–197. https://doi.org/10.1104/pp.110.154773
Jeong JS, Kim YS, Redillas MCFR, Jang G, Jung H, Bang SW et al (2012) OsNAC5 overexpression enlarges root diameter in rice plants leading to enhanced drought tolerance and increased grain yield in the field. Plant Biotechnol J 11(1):101–114. https://doi.org/10.1111/pbi.12011
Ji X, Dong B, Shiran B, Talbot MJ, Edlington JE, Hughes T et al (2011) Control of abscisic acid catabolism and abscisic acid homeostasis is important for reproductive stage stress tolerance in cereals. Plant Physiol 156(2):647–662
Jiang Y, Qiu Y, Hu Y, Yu D (2016) Heterologous expression of AtWRKY57 confers drought tolerance in Oryza sativa. Front Plant Sci 7:145. https://doi.org/10.3389/fpls.2016.00145
Joo J, Lee YH, Song SI (2014) Overexpression of the rice basic leucine zipper transcription factor OsbZIP12 confers drought tolerance to rice and makes seedlings hypersensitive to ABA. Plant Biotechnol Rep 8(6):431–441. https://doi.org/10.1007/s11816-014-0335-2
Joo J, Oh N-I, Nguyen NH, Lee YH, Kim Y-K, Song SI, Cheong J-J (2017) Intergenic transformation of AtMYB44 confers drought stress tolerance in rice seedlings. Appl Biol Chem 60(4):447–455. https://doi.org/10.1007/s13765-017-0297-5
Jung H, Lee D-K, Choi YD, Kim J-K (2015) OsIAA6, a member of the rice Aux/IAA gene family, is involved in drought tolerance and tiller outgrowth. Plant Sci 236:304–312. https://doi.org/10.1016/j.plantsci.2015.04.018
Kader J-C (1996) Lipid-transfer proteins in plants. Annu Rev Plant Biol 47(1):627–654
Kamoshita A, Babu RC, Boopathi NM, Fukai S (2008) Phenotypic and genotypic analysis of drought-resistance traits for development of rice cultivars adapted to rainfed environments. Field Crop Res 109(1–3):1–23
Kanneganti V, Gupta AK (2008) Overexpression of OsiSAP8, a member of stress associated protein (SAP) gene family of rice confers tolerance to salt, drought and cold stress in transgenic tobacco and rice. Plant Mol Biol 66(5):445–462
Kato Y, Abe J, Kamoshita A, Yamagishi J (2006) Genotypic variation in root growth angle in rice (Oryza sativa L.) and its association with deep root development in upland fields with different water regimes. Plant Soil 287(1–2):117–129. https://doi.org/10.1007/s11104-006-9008-4
Kemble A, Macpherson HT (1954) Liberation of amino acids in perennial rye grass during wilting. Biochem J 58(1):46
Khong GN, Pati PK, Richaud F, Parizot B, Bidzinski P, Mai CD et al (2015) OsMADS26 negatively regulates resistance to pathogens and drought tolerance in rice. Plant Physiol 169(4):2935–2949. https://doi.org/10.1104/pp.15.01192
Khush GS (2001) Green revolution: the way forward. Nat Rev Genet 2(10):815–822. https://doi.org/10.1038/35093585
Kim EH, Park S-H, Kim J-K (2009) Methyl jasmonate triggers loss of grain yield under drought stress. Plant Signal Behav 4(4):348–349
Kim H, Lee K, Hwang H, Bhatnagar N, Kim D-Y, Yoon IS et al (2014) Overexpression of PYL5 in rice enhances drought tolerance, inhibits growth, and modulates gene expression. J Exp Bot 65(2):453–464. https://doi.org/10.1093/jxb/ert397
Kohli A, Sreenivasulu N, Lakshmanan P, Kumar PP (2013) The phytohormone crosstalk paradigm takes center stage in understanding how plants respond to abiotic stresses. Plant Cell Rep 32(7):945–957
Kooyers NJ (2015) The evolution of drought escape and avoidance in natural herbaceous populations. Plant Sci 234:155–162. https://doi.org/10.1016/j.plantsci.2015.02.012
Kulcheski FR, de Oliveira LFV, Molina LG, Almerão MP, Rodrigues FA, Marcolino J et al (2011) Identification of novel soybean microRNAs involved in abiotic and biotic stresses. BMC Genomics 12(1):307. https://doi.org/10.1186/1471-2164-12-307
Kumar A, Bernier J, Verulkar S, Lafitte HR, Atlin GN (2008) Breeding for drought tolerance: direct selection for yield, response to selection and use of drought-tolerant donors in upland and lowland-adapted populations. Field Crop Res 107(3):221–231. https://doi.org/10.1016/j.fcr.2008.02.007
Kumar A, Dixit S, Ram T, Yadaw RB, Mishra KK, Mandal NP (2014) Breeding high-yielding drought-tolerant rice: genetic variations and conventional and molecular approaches. J Exp Bot 65(21):6265–6278. https://doi.org/10.1093/jxb/eru363
Kuppu S, Mishra N, Hu R, Sun L, Zhu X, Shen G et al (2013) Water-deficit inducible expression of a cytokinin biosynthetic gene IPT improves drought tolerance in cotton. PLoS One 8(5):e64190
Lafitte HR, Price AH, Courtois B (2004) Yield response to water deficit in an upland rice mapping population: associations among traits and genetic markers. Theor Appl Genet 109(6):1237–1246. https://doi.org/10.1007/s00122-004-1731-8
Lauteri M, Haworth M, Serraj R, Monteverdi MC, Centritto M (2014) Photosynthetic diffusional constraints affect yield in drought stressed rice cultivars during flowering. PLoS One 9(10):e109054
Lee D-K, Kim HI, Jang G, Chung PJ, Jeong JS, Kim YS et al (2015a) The NF-YA transcription factor OsNF-YA7 confers drought stress tolerance of rice in an abscisic acid independent manner. Plant Sci 241:199–210. https://doi.org/10.1016/j.plantsci.2015.10.006
Lee SS, Park HJ, Yoon DH, Kim B-G, Ahn JC, Luan S, Cho HS (2015b) Rice cyclophilin OsCYP18-2 is translocated to the nucleus by an interaction with SKIP and enhances drought tolerance in rice and Arabidopsis. Plant Cell Environ 38(10):2071–2087. https://doi.org/10.1111/pce.12531
Lee D-K, Jung H, Jang G, Jeong JS, Kim YS, Ha S-H et al (2016) Overexpression of the OsERF71 transcription factor alters rice root structure and drought resistance. Plant Physiol 172(1):575–588. https://doi.org/10.1104/pp.16.00379
Lee D-K, Chung PJ, Jeong JS, Jang G, Bang SW, Jung H et al (2017) The rice OsNAC6 transcription factor orchestrates multiple molecular mechanisms involving root structural adaptions and nicotianamine biosynthesis for drought tolerance. Plant Biotechnol J 15(6):754–764. https://doi.org/10.1111/pbi.12673
Lee H, Cha J, Choi C, Choi N, Ji H-S, Park SR et al (2018) Rice WRKY11 plays a role in pathogen defense and drought tolerance. Rice 11(1):5. https://doi.org/10.1186/s12284-018-0199-0
Li J, Li Y, Yin Z, Jiang J, Zhang M, Guo X et al (2016) OsASR5 enhances drought tolerance through a stomatal closure pathway associated with ABA and H2O2 signalling in rice. Plant Biotechnol J 15(2):183–196. https://doi.org/10.1111/pbi.12601
Li M, Wang W-S, Pang Y-L, Domingo JR, Ali J, Xu J-L et al (2017) Characterization of salt-induced epigenetic segregation by genome-wide loss of heterozygosity and its association with salt tolerance in rice (Oryza sativa L.). Front Plant Sci 8:977. https://doi.org/10.3389/fpls.2017.00977
Liang P, Pardee A (1992) Differential display of eukaryotic messenger RNA by means of the polymerase chain reaction. Science 257(5072):967–971. https://doi.org/10.1126/science.1354393
Liang C, Wang Y, Zhu Y, Tang J, Hu B, Liu L et al (2014) OsNAP connects abscisic acid and leaf senescence by fine-tuning abscisic acid biosynthesis and directly targeting senescence-associated genes in rice. Proc Natl Acad Sci U S A 111(27):10013–10018
Lisei-de-Sá ME, Monteiro Arraes FB, Brito GG, Beneventi MA, Lourenço-Tessutti IT, Basso AMM et al (2017) AtDREB2A-CA influences root architecture and increases drought tolerance in transgenic cotton. Agric Sci 08(10):1195–1225. https://doi.org/10.4236/as.2017.810087
Liu W, Reif JC, Ranc N, Porta GD, Würschum T (2012) Comparison of biometrical approaches for QTL detection in multiple segregating families. Theor Appl Genet 125(5):987–998. https://doi.org/10.1007/s00122-012-1889-4
Liu C, Mao B, Ou S, Wang W, Liu L, Wu Y et al (2013) OsbZIP71, a bZIP transcription factor, confers salinity and drought tolerance in rice. Plant Mol Biol 84(1–2):19–36. https://doi.org/10.1007/s11103-013-0115-3
Liu C, Mao B, Ou S, Wang W, Liu L, Wu Y et al (2014) OsbZIP71, a bZIP transcription factor, confers salinity and drought tolerance in rice. Plant Mol Biol 84(1–2):19–36
Lou D, Wang H, Liang G, Yu D (2017) OsSAPK2 confers abscisic acid sensitivity and tolerance to drought stress in rice. Front Plant Sci 8:993. https://doi.org/10.3389/fpls.2017.00993
Lum M, Hanafi M, Rafii Y, Akmar A (2014) Effect of drought stress on growth, proline and antioxidant enzyme activities of upland rice. J Anim Plant Sci 24(5):1487–1493
Mae T (1997) Physiological nitrogen efficiency in rice: nitrogen utilization, photosynthesis, and yield potential. In: Plant nutrition for sustainable food production and environment. Springer, Netherlands, pp 51–60
Malone JH, Oliver B (2011) Microarrays, deep sequencing and the true measure of the transcriptome. BMC Biol 9(1):34. https://doi.org/10.1186/1741-7007-9-34
Manavalan LP, Chen X, Clarke J, Salmeron J, Nguyen HT (2011) RNAi-mediated disruption of squalene synthase improves drought tolerance and yield in rice. J Exp Bot 63(1):163–175
Martin-Tanguy J (2001) Metabolism and function of polyamines in plants: recent development (new approaches). Plant Growth Regul 34(1):135–148
Maruyama K, Todaka D, Mizoi J, Yoshida T, Kidokoro S, Matsukura S et al (2011) Identification of cis-acting promoter elements in cold- and dehydration-induced transcriptional pathways in Arabidopsis, rice, and soybean. DNA Res 19(1):37–49
Maruyama K, Urano K, Yoshiwara K, Morishita Y, Sakurai N, Suzuki H et al (2014) Integrated analysis of the effects of cold and dehydration on rice metabolites, phytohormones, and gene transcripts. Plant Physiol 164(4):1759–1771
Matsumura H, Yoshida K, Luo S, Kimura E, Fujibe T, Albertyn Z et al (2010) High-throughput superSAGE for digital gene expression analysis of multiple samples using next generation sequencing. PLoS One 5(8):e12010. https://doi.org/10.1371/journal.pone.0012010
Miller GAD, Suzuki N, Ciftci-Yilmaz S, Mittler RON (2010) Reactive oxygen species homeostasis and signalling during drought and salinity stresses. Plant Cell Environ 33(4):453–467. https://doi.org/10.1111/j.1365-3040.2009.02041.x
Mitra J (2001) Genetics and genetic improvement of drought resistance in crop plants. Curr Sci 80:758–763
Moore MJ (2015) Twenty years of technology. RNA 21(4):697–698. https://doi.org/10.1261/rna.051052.115
Muhammad A (2012) Breeding potential of the basmati rice germplasm under water stress condition. Afr J Biotechnol 11(25). https://doi.org/10.5897/ajb11.3698
Nagalakshmi U, Wang Z, Waern K, Shou C, Raha D, Gerstein M, Snyder M (2008) The transcriptional landscape of the yeast genome defined by RNA sequencing. Science 320(5881):1344–1349. https://doi.org/10.1126/science.1158441
Nguyen TTT, Klueva N, Chamareck V, Aarti A, Magpantay G, Millena ACM et al (2004) Saturation mapping of QTL regions and identification of putative candidate genes for drought tolerance in rice. Mol Gen Genomics 272(1):35–46. https://doi.org/10.1007/s00438-004-1025-5
Nielsen KL, Høgh AL, Emmersen J (2006) DeepSAGE—digital transcriptomics with high sensitivity, simple experimental protocol and multiplexing of samples. Nucleic Acids Res 34(19):e133–e133. https://doi.org/10.1093/nar/gkl714
Ning J, Li X, Hicks LM, Xiong L (2010) A Raf-like MAPKKK gene DSM1 mediates drought resistance through reactive oxygen species scavenging in rice. Plant Physiol 152(2):876–890
Noctor G, Foyer CH (1998) Ascorbate and glutathione: keeping active oxygen under control. Annu Rev Plant Biol 49(1):249–279
Nuruzzaman M, Manimekalai R, Sharoni AM, Satoh K, Kondoh H, Ooka H, Kikuchi S (2010) Genome-wide analysis of NAC transcription factor family in rice. Gene 465(1–2):30–44
Oh S-J, Kim YS, Kwon C-W, Park HK, Jeong JS, Kim J-K (2009) Overexpression of the transcription factor AP37 in rice improves grain yield under drought conditions. Plant Physiol 150(3):1368–1379
Pan S, Rasul F, Li W, Tian H, Mo Z, Duan M, Tang X (2013) Roles of plant growth regulators on yield, grain qualities and antioxidant enzyme activities in super hybrid rice (Oryza sativa L.). Rice 6(1):9
Pandey GK, Kanwar P, Singh A, Steinhorst L, Pandey A, Yadav AK et al (2015) Calcineurin B-like protein-interacting protein kinase CIPK21 regulates osmotic and salt stress responses in Arabidopsis. Plant Physiol 169(1):780–792
Peleg Z, Blumwald E (2011) Hormone balance and abiotic stress tolerance in crop plants. Curr Opin Plant Biol 14(3):290–295
Peleg Z, Reguera M, Tumimbang E, Walia H, Blumwald E (2011) Cytokinin-mediated source/sink modifications improve drought tolerance and increase grain yield in rice under water-stress. Plant Biotechnol J 9(7):747–758
Perata P, Voesenek LA (2007) Submergence tolerance in rice requires Sub1A, an ethylene-response-factor-like gene. Trends Plant Sci 12(2):43–46
Pieters AJ, El Souki S (2005) Effects of drought during grain filling on PS II activity in rice. J Plant Physiol 162(8):903–911. https://doi.org/10.1016/j.jplph.2004.11.001
Potokina E, Druka A, Luo Z, Wise R, Waugh R, Kearsey M (2008) Gene expression quantitative trait locus analysis of 16 000 barley genes reveals a complex pattern of genome-wide transcriptional regulation. Plant J 53(1):90–101. https://doi.org/10.1111/j.1365-313x.2007.03315.x
Price AH, Steele KA, Moore BJ, Jones RGW (2002) Upland rice grown in soil-filled chambers and exposed to contrasting water-deficit regimes. Field Crop Res 76(1):25–43. https://doi.org/10.1016/s0378-4290(02)00010-2
Quan R, Hu S, Zhang Z, Zhang H, Zhang Z, Huang R (2010) Overexpression of an ERF transcription factor TSRF1 improves rice drought tolerance. Plant Biotechnol J 8(4):476–488. https://doi.org/10.1111/j.1467-7652.2009.00492.x
Raineri J, Wang S, Peleg Z, Blumwald E, Chan RL (2015) The rice transcription factor OsWRKY47 is a positive regulator of the response to water deficit stress. Plant Mol Biol 88(4–5):401–413
Raju NL, Gnanesh BN, Lekha P, Jayashree B, Pande S, Hiremath PJ et al (2010) The first set of EST resource for gene discovery and marker development in pigeon pea (Cajanus cajan L.). BMC Plant Biol 10(1):45. https://doi.org/10.1186/1471-2229-10-45
Raman A, Verulkar SB, Mandal NP, Variar M, Shukla VD, Dwivedi JL et al (2012) Drought yield index to select high yielding rice lines under different drought stress severities. Rice 5(1):31. https://doi.org/10.1186/1939-8433-5-31
Ramegowda V, Basu S, Krishnan A, Pereira A (2014) Rice GROWTH UNDER DROUGHT KINASE is required for drought tolerance and grain yield under normal and drought stress conditions. Plant Physiol 166(3):1634–1645
Ravikumar G, Manimaran P, Voleti S, Subrahmanyam D, Sundaram R, Bansal K et al (2014) Stress-inducible expression of AtDREB1A transcription factor greatly improves drought stress tolerance in transgenic indica rice. Transgenic Res 23(3):421–439
Redillas MC, Jeong JS, Kim YS, Jung H, Bang SW, Choi YD et al (2012) The overexpression of OsNAC9 alters the root architecture of rice plants enhancing drought resistance and grain yield under field conditions. Plant Biotechnol J 10(7):792–805
Rhodes D, Samaras Y (1994) Genetic control of osmoregulation in plants. In: Cellular and molecular physiology of cell volume regulation. CRC Press, Boca Raton, FL, p 416
Ridout CJ, Donini P (1999) Use of AFLP in cereals research. Trends Plant Sci 4(2):76–79. https://doi.org/10.1016/s1360-1385(98)01363-6
Roychoudhury A, Basu S (2012) Ascorbate-glutathione and plant tolerance to various abiotic stresses. In: Anjum NA, Umar S, Ahmad A (eds) Oxidative stress in plants: causes, consequences and tolerance. IK International Publishers, New Delhi, pp 177–258
Roychoudhury A, Das K (2014) Functional role of polyamines and polyamine-metabolizing enzymes during salinity, drought and cold stresses. In: Anjum NA, Gill SS, Gill R (eds) Plant adaptation to environmental change: significance of amino acids and their derivatives. CAB International Publishers, Wallingford, UK, pp 141–156
Roychoudhury A, Paul A (2012) Abscisic acid-inducible genes during salinity and drought stress. In: Berhardt LV (ed) Advances in medicine and biology, vol 51. Nova Science Publishers, New York, pp 1–78
Roychoudhury A, Banerjee A, Lahiri V (2015) Metabolic and molecular-genetic regulation of proline signaling and its cross-talk with major effectors mediates abiotic stress tolerance in plants. Turk J Bot 39:887–910
Saddique MAB, Ali Z, Khan AS, Rana IA, Shamsi IH (2018) Inoculation with the endophyte Piriformospora indica significantly affects mechanisms involved in osmotic stress in rice. Rice 11(1):34. https://doi.org/10.1186/s12284-018-0226-1
Sahebi M, Hanafi MM, Azizi P, Hakim A, Ashkani S, Abiri R (2015) Suppression subtractive hybridization versus next-generation sequencing in plant genetic engineering: challenges and perspectives. Mol Biotechnol 57(10):880–903. https://doi.org/10.1007/s12033-015-9884-z
Schroeder JI, Allen GJ, Hugouvieux V, Kwak JM, Waner D (2001) Guard cell signal transduction. Annu Rev Plant Biol 52(1):627–658
Sharp R (2002) Interaction with ethylene: changing views on the role of abscisic acid in root and shoot growth responses to water stress. Plant Cell Environ 25(2):211–222
Shehab GG, Ahmed OK, El-Beltagi HS (2010) Effects of various chemical agents for alleviation of drought stress in rice plants (Oryza sativa L.). Notulae Botanicae Horti Agrobotanici Cluj-Napoca 38(1):139–148
Shim JS, Oh N, Chung PJ, Kim YS, Choi YD, Kim J-K (2018) Overexpression of OsNAC14 improves drought tolerance in rice. Front Plant Sci 9:310. https://doi.org/10.3389/fpls.2018.00310
Singh S, Pradhan S, Singh A, Singh O (2012) Marker validation in recombinant inbred lines and random varieties of rice for drought tolerance. Aust J Crop Sci 6(4):606
Singha DL, Tuteja N, Boro D, Hazarika GN, Singh S (2017) Heterologous expression of PDH47 confers drought tolerance in indica rice. Plant Cell Tissue Organ Cult 130(3):577–589. https://doi.org/10.1007/s11240-017-1248-x
Sinha AK, Jaggi M, Raghuram B, Tuteja N (2011) Mitogen-activated protein kinase signaling in plants under abiotic stress. Plant Signal Behav 6(2):196–203
Sokoto MB, Muhammad A (2014) Response of rice varieties to water stress in Sokoto, Sudan Savannah, Nigeria. J Biosci Med 02(01):68–74. https://doi.org/10.4236/jbm.2014.21008
Sreenivasulu N, Sunkar R, Wobus U, Strickert M (2010) Array platforms and bioinformatics tools for the analysis of plant transcriptome in response to abiotic stress, Methods in molecular biology. Humana, New York, pp 71–93
Srivastava AK, Zhang C, Caine RS, Gray J, Sadanandom A (2017) Rice SUMO protease Overly Tolerant to salt 1 targets the transcription factor, OsbZIP23 to promote drought tolerance in rice. Plant J 92(6):1031–1043. https://doi.org/10.1111/tpj.13739
Srividhya A, Vemireddy LR, Sridhar S, Jayaprada M, Ramanarao PV, Hariprasad AS et al (2011) Molecular mapping of QTLs for yield and its components under two water supply conditions in rice (Oryza sativa L.). J Crop Sci Biotechnol 14(1):45–56. https://doi.org/10.1007/s12892-010-0023-x
Su Y-H, Liu Y-B, Zhang X-S (2011) Auxin–cytokinin interaction regulates meristem development. Mol Plant 4(4):616–625
Swain P, Anumalla M, Prusty S, Marndi BC, Rao GJN (2014) Characterization of some Indian native land race rice accessions for drought tolerance at seedling stage. Aust J Crop Sci 8(3):324
Takahashi T, Kakehi J-I (2009) Polyamines: ubiquitous polycations with unique roles in growth and stress responses. Ann Bot 105(1):1–6
Takeuchi K, Hasegawa H, Gyohda A, Komatsu S, Okamoto T, Okada K et al (2016) Overexpression of RSOsPR10, a root-specific rice PR10 gene, confers tolerance against drought stress in rice and drought and salt stresses in bentgrass. Plant Cell Tissue Organ Cult 127(1):35–46. https://doi.org/10.1007/s11240-016-1027-0
Tamaki H, Reguera M, Abdel-Tawab YM, Takebayashi Y, Kasahara H, Blumwald E (2015) Targeting hormone-related pathways to improve grain yield in rice: a chemical approach. PLoS One 10(6):e0131213
Tang N, Ma S, Zong W, Yang N, Lv Y, Yan C et al (2016) MODD mediates deactivation and degradation of OsbZIP46 to negatively regulate ABA signaling and drought resistance in rice. Plant Cell 28(9):2161–2177. https://doi.org/10.1105/tpc.16.00171
Tardieu F, Parent B, Simonneau T (2010) Control of leaf growth by abscisic acid: hydraulic or non-hydraulic processes? Plant Cell Environ 33(4):636–647
Todaka D, Shinozaki K, Yamaguchi-Shinozaki K (2015) Recent advances in the dissection of drought-stress regulatory networks and strategies for development of drought-tolerant transgenic rice plants. Front Plant Sci 6:84. https://doi.org/10.3389/fpls.2015.00084
Travaglia C, Reinoso H, Cohen A, Luna C, Tommasino E, Castillo C, Bottini R (2010) Exogenous ABA increases yield in field-grown wheat with moderate water restriction. J Plant Growth Regul 29(3):366–374
Trijatmiko KR, Prasetiyono J, Thomson MJ, Cruz CMV, Moeljopawiro S, Pereira A (2014) Meta-analysis of quantitative trait loci for grain yield and component traits under reproductive-stage drought stress in an upland rice population. Mol Breed 34(2):283–295
Tuberosa R (2012) Phenotyping for drought tolerance of crops in the genomics era. Front Physiol 3:347. https://doi.org/10.3389/fphys.2012.00347
Turner JG, Ellis C, Devoto A (2002) The jasmonate signal pathway. Plant Cell 14(suppl 1):S153–S164
Uga Y, Sugimoto K, Ogawa S, Rane J, Ishitani M, Hara N et al (2013) Control of root system architecture by DEEPER ROOTING 1 increases rice yield under drought conditions. Nat Genet 45(9):1097
Usman M, Raheem Z, Ahsan T, Iqbal A, Sarfaraz ZN, Haq Z (2013) Morphological, physiological and biochemical attributes as indicators for drought tolerance in rice (Oryza sativa L.). Eur J Biol Sci 5(1):23–28
Varshney R, Graner A, Sorrells M (2005) Genomics-assisted breeding for crop improvement. Trends Plant Sci 10(12):621–630. https://doi.org/10.1016/j.tplants.2005.10.004
Varshney RK, Bansal KC, Aggarwal PK, Datta SK, Craufurd PQ (2011) Agricultural biotechnology for crop improvement in a variable climate: hope or hype? Trends Plant Sci 16(7):363–371. https://doi.org/10.1016/j.tplants.2011.03.004
Venuprasad R, Sta Cruz MT, Amante M, Magbanua R, Kumar A, Atlin GN (2008) Response to two cycles of divergent selection for grain yield under drought stress in four rice breeding populations. Field Crop Res 107(3):232–244. https://doi.org/10.1016/j.fcr.2008.02.004
Verbruggen N, Hermans C (2008) Proline accumulation in plants: a review. Amino Acids 35(4):753–759
Vikram P, Swamy BM, Dixit S, Singh R, Singh BP, Miro B et al (2015) Drought susceptibility of modern rice varieties: an effect of linkage of drought tolerance with undesirable traits. Sci Rep 5:14799
Wakim LM, Waithman J, van Rooijen N, Heath WR, Carbone FR (2008) Dendritic cell-induced memory T cell activation in nonlymphoid tissues. Science 319(5860):198–202. https://doi.org/10.1126/science.1151869
Wang F-Z, Wang Q-B, Kwon S-Y, Kwak S-S, Su W-A (2005) Enhanced drought tolerance of transgenic rice plants expressing a pea manganese superoxide dismutase. J Plant Physiol 162(4):465–472
Wang H, Inukai Y, Yamauchi A (2006) Root development and nutrient uptake. Crit Rev Plant Sci 25(3):279–301. https://doi.org/10.1080/07352680600709917
Wang Y, Zhang Q, Zheng T, Cui Y, Zhang W, Xu J, Li Z (2014) Drought-tolerance QTLs commonly detected in two sets of reciprocal introgression lines in rice. Crop Pasture Sci 65(2):171. https://doi.org/10.1071/cp13344
Wilkinson S, Kudoyarova GR, Veselov DS, Arkhipova TN, Davies WJ (2012) Plant hormone interactions: innovative targets for crop breeding and management. J Exp Bot 63(9):3499–3509
Xiang Y, Tang N, Du H, Ye H, Xiong L (2008) Characterization of OsbZIP23 as a key player of the basic leucine zipper transcription factor family for conferring abscisic acid sensitivity and salinity and drought tolerance in rice. Plant Physiol 148(4):1938–1952
Xiang J, Chen X, Hu W, Xiang Y, Yan M, Wang J (2018) Overexpressing heat-shock protein OsHSP50.2 improves drought tolerance in rice. Plant Cell Rep 37(11):1585–1595. https://doi.org/10.1007/s00299-018-2331-4
Xiao B, Huang Y, Tang N, Xiong L (2007) Over-expression of a LEA gene in rice improves drought resistance under the field conditions. Theor Appl Genet 115(1):35–46
Xiao B-Z, Chen X, Xiang C-B, Tang N, Zhang Q-F, Xiong L-Z (2009) Evaluation of seven function-known candidate genes for their effects on improving drought resistance of transgenic rice under field conditions. Mol Plant 2(1):73–83. https://doi.org/10.1093/mp/ssn068
Xing Y, Zhang Q (2010) Genetic and molecular bases of rice yield. Annu Rev Plant Biol 61:421–442
Xiong H, Li J, Liu P, Duan J, Zhao Y, Guo X et al (2014) Overexpression of OsMYB48-1, a novel MYB-related transcription factor, enhances drought and salinity tolerance in rice. PLoS One 9(3):e92913. https://doi.org/10.1371/journal.pone.0092913
Xiong H, Yu J, Miao J, Li J, Zhang H, Wang X et al (2018) Natural variation in OsLG3 increases drought tolerance in rice by inducing ROS scavenging. Plant Physiol 178(1):451–467. https://doi.org/10.1104/pp.17.01492
Xu K, Chen S, Li T, Ma X, Liang X, Ding X et al (2015) OsGRAS23, a rice GRAS transcription factor gene, is involved in drought stress response through regulating expression of stress-responsive genes. BMC Plant Biol 15(1). https://doi.org/10.1186/s12870-015-0532-3
Yamaguchi-Shinozaki K, Shinozaki K (2006) Transcriptional regulatory networks in cellular responses and tolerance to dehydration and cold stresses. Annu Rev Plant Biol 57(1):781–803. https://doi.org/10.1146/annurev.arplant.57.032905.105444
Yang J, Zhang J (2010) Crop management techniques to enhance harvest index in rice. J Exp Bot 61(12):3177–3189
Yang J, Zhang J, Wang Z, Zhu Q, Liu L (2001) Water deficit–induced senescence and its relationship to the remobilization of pre-stored carbon in wheat during grain filling. Agron J 93(1):196–206
Yang J, Zhang J, Wang Z, Xu G, Zhu Q (2004) Activities of key enzymes in sucrose-to-starch conversion in wheat grains subjected to water deficit during grain filling. Plant Physiol 135(3):1621–1629
Yang J, Zhang J, Liu K, Wang Z, Liu L (2007) Involvement of polyamines in the drought resistance of rice. J Exp Bot 58(6):1545–1555
Yang W, Kong Z, Omo-Ikerodah E, Xu W, Li Q, Xue Y (2008) Calcineurin B-like interacting protein kinase OsCIPK23 functions in pollination and drought stress responses in rice (Oryza sativa L.). J Genet Genomics 35(9):531–S532
Yang PM, Huang QC, Qin GY, Zhao SP, Zhou JG (2014) Different drought-stress responses in photosynthesis and reactive oxygen metabolism between autotetraploid and diploid rice. Photosynthetica 52(2):193–202. https://doi.org/10.1007/s11099-014-0020-2
Yin XM, Huang LF, Zhang X, Wang ML, Xu GY, Xia XJ (2015) OsCML4 improves drought tolerance through scavenging of reactive oxygen species in rice. J Plant Biol 58(1):68–73. https://doi.org/10.1007/s12374-014-0349-x
Yoon S, Lee D-K, Yu IJ, Kim YS, Choi YD, Kim J-K (2017) Overexpression of the OsbZIP66 transcription factor enhances drought tolerance of rice plants. Plant Biotechnol Rep 11(1):53–62. https://doi.org/10.1007/s11816-017-0430-2
You J, Zong W, Li X, Ning J, Hu H, Li X et al (2012) The SNAC1-targeted gene OsSRO1c modulates stomatal closure and oxidative stress tolerance by regulating hydrogen peroxide in rice. J Exp Bot 64(2):569–583
You J, Zong W, Hu H, Li X, Xiao J, Xiong L (2014) A STRESS-RESPONSIVE NAC1-regulated protein phosphatase gene rice protein phosphatase18 modulates drought and oxidative stress tolerance through abscisic acid-independent reactive oxygen species scavenging in rice. Plant Physiol 166(4):2100–2114. https://doi.org/10.1104/pp.114.251116
Yu L, Chen X, Wang Z, Wang S, Wang Y, Zhu Q et al (2013) Arabidopsis enhanced drought tolerance1/HOMEODOMAIN GLABROUS11 confers drought tolerance in transgenic rice without yield penalty. Plant Physiol 162(3):1378–1391
Yu J, Lai Y, Wu X, Wu G, Guo C (2016a) Overexpression of OsEm1 encoding a group I LEA protein confers enhanced drought tolerance in rice. Biochem Biophys Res Commun 478(2):703–709. https://doi.org/10.1016/j.bbrc.2016.08.010
Yu Y, Yang D, Zhou S, Gu J, Wang F, Dong J, Huang R (2016b) The ethylene response factor OsERF109 negatively affects ethylene biosynthesis and drought tolerance in rice. Protoplasma 254(1):401–408. https://doi.org/10.1007/s00709-016-0960-4
Zhang Q, Li J, Zhang W, Yan S, Wang R, Zhao J et al (2012) The putative auxin efflux carrier OsPIN3t is involved in the drought stress response and drought tolerance. Plant J 72(5):805–816
Zhang Z, Zhang Q, Wu J, Zheng X, Zheng S, Sun X et al (2013) Gene knockout study reveals that cytosolic ascorbate peroxidase 2 (OsAPX2) plays a critical role in growth and reproduction in rice under drought, salt and cold stresses. PLoS One 8(2):e57472
Zhao K, Wright M, Kimball J, Eizenga G, McClung A, Kovach M et al (2010) Genomic diversity and introgression in O. sativa reveal the impact of domestication and breeding on the rice genome. PLoS One 5(5):e10780. https://doi.org/10.1371/journal.pone.0010780
Zhou Y, Lam HM, Zhang J (2007) Inhibition of photosynthesis and energy dissipation induced by water and high light stresses in rice. J Exp Bot 58(5):1207–1217. https://doi.org/10.1093/jxb/erl291
Zhou L, Liu Z, Liu Y, Kong D, Li T, Yu S et al (2016) A novel gene OsAHL1 improves both drought avoidance and drought tolerance in rice. Sci Rep 6(1):30264. https://doi.org/10.1038/srep30264
Zhu J, Gong Z, Zhang C, Song C-P, Damsz B, Inan G et al (2002) OSM1/SYP61: a syntaxin protein in Arabidopsis controls abscisic acid–mediated and non-abscisic acid–mediated responses to abiotic stress. Plant Cell 14(12):3009–3028. https://doi.org/10.1105/tpc.006981
Zhu G, Ye N, Yang J, Peng X, Zhang J (2011) Regulation of expression of starch synthesis genes by ethylene and ABA in relation to the development of rice inferior and superior spikelets. J Exp Bot 62(11):3907–3916
Zou G, Mei H, Liu H, Liu G, Hu S, Yu X et al (2005) Grain yield responses to moisture regimes in a rice population: association among traits and genetic markers. Theor Appl Genet 112(1):106–113
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Rehman, A., Almas, H.I., Akbar, F., Ali, Q., Du, X. (2020). An Integrated Approach for Drought Tolerance Improvement in Rice. In: Roychoudhury, A. (eds) Rice Research for Quality Improvement: Genomics and Genetic Engineering. Springer, Singapore. https://doi.org/10.1007/978-981-15-4120-9_12
Download citation
DOI: https://doi.org/10.1007/978-981-15-4120-9_12
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-15-4119-3
Online ISBN: 978-981-15-4120-9
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)