Abstract
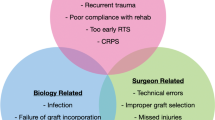
Anterior cruciate ligament (ACL) tears continue to be one of the most common sports injuries. ACL reconstruction (ACLR) are also common, and the number of ACL reconstruction (ACLR) has increased over the last years [1, 2]. Despite improved methods and techniques for primary ACLR, graft re-rupture rates have been reported between 0 and 5.6% [3]. Subsequently, the number of ACL revision surgeries has increased [4]. Outcomes after revision ACLR are generally inferior to those after primary ACLR. Only 43% of patients have been reported to return to their previous activity level, a significantly lower figure than that reported for primary ACLR [5]. In addition, the rates of re-revision ACLR after revision ACLR are higher than after primary ACLR (6.5% at 5 years and 9.0% at 8 years) [6]. The exact reasons why revision ACLR results in inferior outcomes are not completely known. However, it is a common perception for surgeons that revision ACLR is a challenging operation.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
21.1 Introduction
Anterior cruciate ligament (ACL) tears continue to be one of the most common sports injuries. ACL reconstruction (ACLR) are also common, and the number of ACL reconstruction (ACLR) has increased over the last years [1, 2]. Despite improved methods and techniques for primary ACLR, graft re-rupture rates have been reported between 0 and 5.6% [3]. Subsequently, the number of ACL revision surgeries has increased [4]. Outcomes after revision ACLR are generally inferior to those after primary ACLR. Only 43% of patients have been reported to return to their previous activity level, a significantly lower figure than that reported for primary ACLR [5]. In addition, the rates of re-revision ACLR after revision ACLR are higher than after primary ACLR (6.5% at 5 years and 9.0% at 8 years) [6]. The exact reasons why revision ACLR results in inferior outcomes are not completely known. However, it is a common perception for surgeons that revision ACLR is a challenging operation.
21.2 Preoperative Planning
Preoperative planning, including comprehensive clinical and radiological evaluation, is the first and most important step. It is necessary to adequately understand the patient’s symptoms such as instability, swelling, locking, stiffness, and pain. The history of previous injuries, how it occurred, pre-injured activity level (the competition and its level), the postoperative course, information on the mechanism and timing of reinjuries, is also crucial. Blood tests provide surgeons with information about possible infections as a routine preoperative examination. It is prerequisite to know the complete details of the primary surgery, the presence of the concomitant lesions and their treatment, and the type of graft and fixation methods.
Routine clinical examinations are conducted to assess anterior instability, rotational instability, and associated complications as before the primary surgery. Imaging evaluation should include X-ray, MRI, and CT scan. X-ray is a comprehensive imaging test that is very useful for understanding the overall condition. It is important to carefully observe the alignment evaluation, especially varus alignment and/or increased posterior slope of the tibia. Those conditions can provoke ACL instability and the risk of graft failure [7]. In those cases, it should be considered to perform proximal tibial osteotomy concurrently in order to obtain suitable alignment. The position of the bone tunnel and previous device may also be observed, and if there is tunnel lysis or expansion, the decision of the revision surgical procedure will be affected. The CT scan can provide more detailed information related to tunnel position, size of tunnels, miserable tunnel expansion, and the presence of hardware. The MRI is useful to perceive the status of the graft, cartilage, meniscal lesion, and the surrounding soft tissues.
21.3 Graft Choice
The graft choice for revision surgery depends upon the graft used for primary ACLR and the placement of the tunnel and tunnel size. The surgeon’s preferences and concepts are also influenced by the decision. Which of the graft is superior for primary or revision ACLR is still controversial [8, 9]. However, recent large cohort studies of revision ACLR reported that autograft is superior to allografts in terms of lower re-revision rate, lower postoperative laxity, patient reported outcome (IKDC and KOOS) at 2-year postoperatively, and the rate of return to sports (RTS) [10,11,12,13,14,15]. On the other hand, there is no clear consensus on what type of autograft should be selected in the revision ACLR, which depends mainly on experience and local conditions. Our most frequent graft choice in primary or revision is quadriceps tendon (QT). In certain cases, hamstring tendon (HT) or bone-patellar tendon-bone (BPTB) may also be considered, and surgeons need to master several techniques. Table 21.1 summarizes our algorithm for autograft management in revision ACLR.
Graft choice for revision ACLR was quite variable between large cohorts in France, Norway, and North America (Table 21.2) [9]. In the Multicenter ACL Revision study (MARS) cohort, allografts (49.4%) and autografts (47.9%) were used with similar frequency (hybrid were used for the remaining 2.6%), with BPTB autografts (26.2%) used slightly more than HT autografts (20.1%). In the Norwegian Knee Ligament Registry (NKLR), HT autograft (56.0%) was the most commonly used graft followed by BPTB autograft (32.4%). Other graft types were rare. In the Société Française d’Arthroscopie (SFA) survey, BPTB autograft (55.9%) was most common, followed by hamstring autograft (38.6%). QT autografts were used in 1–2% of cases in each cohort. However, interestingly, the analysis of MARS cohort [16] demonstrated that extrinsic factors (age, gender, previous graft choice) significantly influence graft choice, but finally the most important factor in revision ACL R graft choice is the surgeon.
21.4 Quadriceps Tendon (QT)
Recently, the quadriceps tendon (QT) autograft (Fig. 21.1a.) has been discussed as a potential alternative graft for ACLR. Although the QT autograft is the least studied and least used autograft, its use is expected to increase [17]. In 2010, 2.5% of all anatomic ACLR were performed with QT autografts [18], but increased to 11% in 2014 [19]. The biomechanical study showed that QT graft has a clear advantage because it provided a thicker graft with more advantageous tensile properties compared with BPTB and HT grafts [20,21,22,23]. Several studies found that the cross-sectional area of the QT was nearly twice that of the BPTB [20,21,22,23]. Shani et al. [21]. reported that ultimate load to failure (2186 vs. 1581 N) and stiffness (466 vs. 278 N/mm) were significantly higher for the QT graft compared with BPTB, respectively. Harris et al. [20] demonstrated that the load to failure of the QT was 1.36 times higher than that of a BPTB graft of comparable width. The most recent meta-analysis reported that QT autograft had comparable clinical and functional outcomes and graft survival rate compared with BPTB and HT autografts, with QT autograft showing significantly less harvest site pain compared with BPTB autograft and better functional outcome scores (Lysholm) compared with HT autograft [24].
In terms of cosmetic aspects, Bartlett et al. [25] noted that taking QT can cause unsightly wounds. In addition, Kim et al. [26] in his series, 7 patients out of 21 treated with ACLR using QT autografts required cosmetic surgery for scars that impair the appearance. However, Cavaignac et al. [27] reported that the appearance of the scar and the length of the incision were significantly better in QT versus BPTB and tended to be better without reaching the significant threshold of 2.5% in QT versus HT patients. Moreover, in this study, the harvesting of QT autograft for ACLR produces less area of hypoesthesia than both BPTB and HT autografts with statistical significance [27].
While many primary clinical outcomes have been reported, there are not many reports on the use of QT for revision ACLR. Wright et al. [28] reported that the systematic review of 766 patients showed autograft was used in 89.4% (685) and consisted of BPTB in 48%, HT in 40%, and QT in 12%. Barié et al. [29] reported that revision ACLR using QT autografts provides good objective knee stability in over 90% of the patients. This level of stability is comparable with that after primary ACLR, as indicated in previous studies. It is able to restore the function to a good or very good level in over 50% of the patients as previously reported in the literature. Nonetheless, there is still little data on the use of QT in revision ACLR and further research is required.
21.5 Bone-Patellar Tendon-Bone (BPTB)
BPTB (Fig. 21.1b) is one of the most commonly harvested autologous tissues used as grafts for primary or revision ACLR [9, 28]. In primary reconstruction, some authors suggest that BPTB autograft is the most favorable graft choice because of faster graft incorporation [30] a higher proportion of patients returning to preinjury activity levels [31], and potentially a lower risk of graft rupture [32]. A recent large Scandinavian registry study [33] reported a higher risk of graft rupture with HT than BPTB. The most recent meta-analysis found that patients undergoing primary ACLR with BPTB autograft were less likely to experience graft rupture and/or revision ACLR than patients treated with HT autograft [34].
In terms of revision surgery using BPTB, good results have also been reported in the literatures [35, 36]. Keizer et al. [37] reported that there was a significant difference in rate of return to sports (RTS) type in favor of using an ipsilateral BPTB autograft over a patellar tendon allograft (43.3% versus 75.0%, respectively) in patients undergoing revision ACLR after a minimum follow-up of 2 years.
However, disadvantages of using a BPTB autograft tendon might include anterior knee pain [38], donor site morbidity, quadriceps weakness [39, 40], and therefore a lower knee extensor moment [41].
21.6 Hamstring Tendon (HT)
HT (Fig. 21.1c) is also one of the common grafts used for both primary and revision surgery as well as BPTB [9]. Denti et al. [42] reported similar outcomes between HT and BPTB autografts in revision surgery. On the other hand, Grassi et al. [12] reported that HT autografts had better outcomes than BPTB autografts in revision ACLR, with IKDC score, Lysholm score, Tegner score and lower rates of complications and re-operations, while HT and BPTB autografts had similar outcomes in terms of laxity and pivot-shift. Legnani et al. [13] demonstrated that the use of contralateral HT autografts for ACL revision surgery produced similar subjective and objective outcomes at 5.2 years follow-up compared to revision with allograft patellar or Achilles tendon. With regard to RTS, patients undergoing revision surgery with autografts experienced a quicker RTS compared to patients who underwent allograft revision surgery.
Nevertheless, there are many reports that HT is higher risk of graft failure than BPTB in primary ACLR [32,33,34], therefore it is important to consider graft choice according to the individual characteristics.
21.7 Allograft
An allograft is still the preferred graft used in North America for revision ACLR [9, 43]. An allograft has the greatest advantage of being able to avoid donor site morbidity [43]. In addition, it can provide a large bone block that helps to fill the bone gap [44]. However, consideration should be given to infection, disease transmission risk, late re-cellularization, possibility of late failure, and the increased cost with this option [43,44,45,46,47].
However, a recent systematic review of graft type and the outcomes of revision ACLR reported autografts to have better results than allografts with lower postoperative laxity, and lower rates of reoperation and complications [12]. Some previous studies described allograft is inferior to autograft in terms of RTS [13, 36]. The Danish registry [14, 15] also showed that the re-revision rate was significantly higher for allograft compared with autograft (12.7% vs. 5.4%; P < 0.001), leading to a hazard ratio for re-revision of 2.2 (95% CI, 1.4–3.4) for allografts compared with autografts when corrected for age. Otherwise, Condello et al. [48] reported that the use of allografts for ACL revision can be regarded as a safe and effective approach: data from several studies have shown that the infection and overall complication rate, is similar with respect to primary procedures with autografts [49], and also clinical outcomes are satisfactory in terms of durable knee stability after revision and RTS [5, 7]. Additionally, Kay et al. [35] reported that there was no significant influence of graft choice on the rate of RTS after revision anterior cruciate ligament reconstruction identified between BPTB autografts (67%), HT autografts (55%), and allografts (64%).
21.8 One-Stage or Two-Stage Surgery?
Once all the necessary information has been obtained, the surgeon can choose a one- or a two-stage revision. One-stage revision can be considered when graft healing and fixation will not be influenced by previous surgery. Therefore, one-stage revision can be performed in a patient in whom previous tunnel did not interfere with the new tunnel, the tunnel did not significantly expand, no tunnel lysis or void bony defect, no associated injuries (malalignments, meniscal and chondral lesion, unless treated simultaneously), and no hardware removal problem. The two-stage procedure is performed in 6–9% of all ACL revision cases [48]. Even if the previous tunnel is correctly placed, if there is a significant tunnel enlargement or a void bony defect (Fig. 21.2), a two-stage revision is recommended [50]. The amount of tunnel enlargement required for two-stage surgery is still contentious. In general, it is accepted that two-stage surgery may be required when tunnel enlargement greater than 15–16 mm or 100% larger than the original tunnel [51, 52]. Even a 10–15 mm anatomically correct tunnel may require two-stage surgery depending on the shape of the tunnel or anticipated graft choice, while tunnels measuring less than 10 mm usually may be reused without grafting (permitting single-stage surgery) [52] (Fig. 21.3).
The radiological evaluation of bone tunnel placement. (a) X-ray shows a posteriorly placed tibial tunnel placed. In this case, we decided to perform staged surgery in order be able to safely place the tibial tunnel and to assure a good fixation. (b) CT image after first step surgery for bone grafting. In this particular case, you should await 6 months before performing the second stage surgery. (c) Postoperative X-ray shows that previous bone tunnel was well filled up with grafted bone and allows for a proper placement of the tibial tunnel and a primary fixation with interference screw + post fixation as a safe backup
If a two-stage revision is deemed necessary, the first stage consists of removal of the old graft, and the metalwork from the primary repair. Some authors now advocate that metalwork should be left in situ if possible because screw and graft removal can leave large defect obtained between 3 and 6 months (3–4 months for autograft, 4–6 months for allograft) to assess the bone quality and the degree of graft incorporation. If these are deemed satisfactory, the second stage of the procedure consists of a standard graft repair.
Other indications for a two-stage revision are loss of range of motion, and concomitant surgery such as repair for locked bucket-handle meniscal tear or proximal tibial osteotomy that would slow down the postoperative recovery [48].
21.9 Fixation
The fixation method should be considered during planning, along with the suspected reason of previous graft failure. A variety of devices for bone and soft tissue fixation must be available aiming to not restrict the choice of the graft and use the best bone support [53]. The role of graft fixation in ACLR is to maintain sufficient graft stability in the early stages of healing to allow incorporation. The graft is completely reliant on the fixation device for its strength until it becomes incorporated into the tunnel wall. There are multiple types of fixation devices (interference screw, staples, buttons, post-screw, etc.) for the femur or tibia, and in some difficult revision cases these methods may be combined to increase primary resistance [52, 53].
One of the commonest methods is the interference screw technique [52], and we also prefer this technique. This may be metallic or biodegradable and works on the “press fit” principle, jamming the graft tightly against the tunnel wall so it may be eccentrically placed within the tunnels. The disadvantage of this technique was that interference screw tends to slip, especially with HT grafts. This slippage might provoke the graft laxity or graft migration. The EndoButton is also useful technique, which has advantage to adjust the length of the graft easily and keep bone stock. However, tunnel widening is commoner following the use of EndoButton fixation devices, and some authors have postulated that the delay in incorporation of these types of fixation method allows for micromotion to erode the adjacent tunnel wall during knee motion [54].
These techniques for graft fixation are mainly dependent on the remaining bone stock and the chosen graft [53, 55], thus, surgeons need to be familiar with each of these types of characteristics and must be trained in their use.
21.10 Associated Lesions
The most commonly reported associated injuries were meniscal tears and chondral lesions [36, 37, 56]. These associated lesions are very important. Revision procedures are able to restore knee stability, but symptoms and pain reduce knee function, resulting in poorer subjective outcome. As suggested by a French study, this can most likely be explained by concomitant meniscal and cartilage injuries in the revision reconstruction patients [57]. In the MARS ACL revision cohort, meniscal and cartilage injuries were seen in 90% of patients, and meniscal injury at the primary reconstruction resulted in an increased risk of cartilage deterioration at the time of revision surgery [58,59,60]. Wu et al. [61] obtained similar findings in 63 patients who had undergone ACLR and meniscectomy. These patients had significantly lower subjective function scores and ability to perform a single-leg hop test compared with patients with intact menisci at an average 10.4 years of follow-up. Therefore, it is extremely important to provide appropriate treatment for these combined injuries since the approach for meniscal injuries and/or chondral lesions should affect outcome of revision ACLR.
Recently, the presence of ramp lesion and posterolateral corner (PLC) injuries associated with ACL injury has been reported. Their incidence in primary ACLR is reported as high as 23.9% [62] to 19.7% [63].
Ramp lesion is a specific type of meniscal tear, which were defined as longitudinal tears of the peripheral attachment of the posterior horn of the medial meniscus (Fig. 21.4a). The posterior horn is recognized as a critical stabilizer in the ACL-deficient knee [64]. In a more recent cadaveric study, sectioning of the posteromedial meniscocapsular junction in an ACL- deficient knee resulted in a significant increase in anterior tibial translation and external rotation [65].
Also, many studies have established that missing the diagnosis of posterolateral corner (PLC) injury can increase the varus load on the ACL graft and the risk of graft failure [63, 66, 67]. The anatomy of the PLC is complicated, and injury is overlooked in many cases (50–76%) [63, 68, 69]. Thus, PLC-deficient condition after ACLR may be one of reason of graft failure or poor outcome. Furthermore, dysfunction of PLC should be overlooked again in revision surgery so that it is quite important to familiar with this pathology and strive to repair in both primary and revision surgery. We perform either mini-open or arthroscopic technique for these abnormal conditions which may lead to knee laxity (Fig. 21.4b, c).
Both ramp lesions and PLC injuries play a role in disrupting an important secondary stabilizer of the knee, and arthroscopic examination of the posterior structures and PLC of the knee and repair of these lesions must always be undertaken when revision ACL surgery is performed.
21.11 Summary
-
The preoperative planning is the first and most important step.
-
Autografts are superior to allograft for revision ACL surgery in many aspects.
-
The evaluation of bone tunnel position and its enlargement is very important to determine whether to indicate one- or two-stage surgery.
-
Don’t overlook the associated lesions, especially ramp lesion and PLC injury.
References
Buller LT, Best MJ, Baraga MG, Kaplan LD. Trends in anterior cruciate ligament reconstruction in the United States. Orthop J Sports Med. 2015;3:2325967114563664.
Sanders TL, Maradit Kremers H, Bryan AJ, Larson DR, Dahm DL, Levy BA, et al. Incidence of anterior cruciate ligament tears and reconstruction: a 21-year population-based study. Am J Sports Med. 2016;44:1502–7.
Hurley ET, Calvo-Gurry M, Withers D, Farrington SK, Moran R, Moran CJ. Quadriceps tendon autograft in anterior cruciate ligament reconstruction: a systematic review. Arthroscopy. 2018;34(5):1690–8.
Hettrich CM, Dunn WR, Reinke EK, MOON Group, Spindler KP. The rate of subsequent surgery and predictors after anterior cruciate ligament reconstruction: two- and 6-year follow-up results from a multicenter cohort. Am J Sports Med. 2013;41:1534–40.
Andriolo L, Filardo G, Kon E, Ricci M, Della Villa F, Della Villa S, Zaffagnini S, Marcacci M. Revision anterior cruciate ligament reconstruction: clinical outcome and evidence for return to sport. Knee Surg Sports Traumatol Arthrosc. 2015;23:2825–45.
Vap AR, Persson A, Fenstad AM, Moatshe G, LaPrade RF, Engebretsen L. Re-revision anterior cruciate ligament reconstruction: an evaluation from the Norwegian knee ligament registry. Arthroscopy. 2019;35(6):1695–701.
Crawford MD, Diehl L, Amendola A. Surgical management and treatment of the anterior cruciate ligament–deficient knee with malalignment. Clin Sports Med. 2017;36(1):119–33.
Kim HS, Seon JK, Jo AR. Current trends in anterior cruciate ligament reconstruction. Knee Surg Relat Res. 2013;25(4):165–73.
Magnussen RA, Trojani C, Granan LP, Neyret P, Colombet P, Engebretsen L, Wright RW, Kaeding CC, MARS Group; SFA Revision ACL Group. Patient demographics and surgical characteristics in ACL revision: a comparison of French, Norwegian, and North American cohorts. Knee Surg Sports Traumatol Arthrosc. 2015;8:2339–48.
Wright RW, Huston LJ, Haas AK, Spindler KP, Nwosu SK, et al.; MARS Group. Effect of graft choice on the outcome of revision anterior cruciate ligament reconstruction in the multicenter ACL revision study (MARS) cohort. Am J Sports Med. 2014;42(10):2301–10.
Niki Y, Matsumoto H, Enomoto H, Toyama Y, Suda Y. Single-stage anterior cruciate ligament revision with bone-patellar tendon-bone: a case-control series of revision of failed synthetic anterior cruciate ligament reconstructions. Arthroscopy. 2010;26(8):1058–65.
Rosenberg TD, Franklin JL, Baldwin GN, Nelson KA. Extensor mechanism function after patellar tendon graft harvest for anterior cruciate ligament reconstruction. Am J Sports Med. 1992;20(5):519–26.
Denti M, Lo Vetere D, Bait C, et al. Revision anterior cruciate ligament reconstruction: causes of failure, surgical technique, and clinical results. Am J Sports Med. 2008;36:1896–902.
Pascual-Garrido C, Carbo L, Makino A. Revision of anterior cruciate ligament reconstruction with allografts in patients younger than 40 years old: a 2 to 4 year results. Knee Surg Sports Traumatol Arthrosc. 2014;22(5):1106–11.
Cerulli G, Placella G, Sebastiani E, Tei MM, Speziali A, Manfreda F. ACL reconstruction: choosing the graft. Joints. 2013;1(1):18–24.
MARS Group. Factors influencing graft choice in revision ACL reconstructions: a propensity analysis of the MARS cohort. J Knee Surg. 2016;29(6):458–63.
Lubowitz JH. Editorial commentary: quadriceps tendon autograft use for anterior cruciate ligament reconstruction predicted to increase. Arthroscopy. 2016;32(1):76–7.
Van Eck CF, Schreiber VM, Mejia HA, et al. “Anatomic” anterior cruciate ligament reconstruction: a systematic review of surgical techniques and reporting of surgical data. Arthroscopy. 2010;26(9 suppl):S2–S12.
Middleton KK, Hamilton T, Irrgang JJ, et al. Anatomic anterior cruciate ligament (ACL) reconstruction: a global perspective, part 1. Knee Surg Sports Traumatol Arthrosc. 2014;22(7):1467–82.
Harris NL, Smith DA, Lamoreaux L. Central quadriceps tendon for anterior cruciate ligament reconstruction, part I: morphometric and biomechanical evaluation. Am J Sports Med. 1997;25(1):23–8.
Shani RH, Umpierez E, Nasert M. Biomechanical comparison of quadriceps and patellar tendon grafts in anterior cruciate ligament reconstruction. Arthroscopy. 2016;32(1):71–5.
Stäubli HU, Schatzmann L, Brunner P. Mechanical tensile properties of the quadriceps tendon and patellar ligament in young adults. Am J Sports Med. 1999;27(1):27–34.
Hersekli MA, Akpinar S, Ozalay M, et al. Tunnel enlargement after arthroscopic anterior cruciate ligament reconstruction: comparison of bone-patellar tendon-bone and hamstring autografts. Adv Ther. 2004;21(2):123–31.
Mouarbes D, Menetrey J, Vincent Marot V, Courtot L, Berard E, Cavaignac E. Anterior cruciate ligament reconstruction. A systematic review and meta-analysis of outcomes for quadriceps tendon autograft versus bone–patellar tendon–bone and hamstring-tendon autografts. Am J Sports Med. 2019;21:363546518825340. [Epub ahead of print].
Bartlett RJ, Clatworthy MG, Nguyen TNV. Graft selection in reconstruction of the anterior cruciate ligament. J Bone Joint Surg Br. 2001;83-B(5):625–3.
Kim SJ, Kumar P, Oh KS. Anterior cruciate ligament reconstruction: autogenous quadriceps tendon- bone compared with bone-patellar tendon-bone grafts at 2-year follow-up. Arthroscopy. 2009;25(2):137–44.
Cavaignac E, Mouarbes D, Chiron P, Peque E, Dagneaux L, Matthieu O. Cutaneous and neurological complications of anterior cruciate ligament (ACL) reconstruction with quadriceps tendon (QT), bone patellar-tendon bone (BPTB) or Hamstring tendon (HT). 2019. In press.
Wright RW, Gill CS, Chen L, Brophy RH, Matava MJ, Smith MV, Mall NA. Outcome of revision anterior cruciate ligament reconstruction: a systematic review. J Bone Joint Surg Am. 2012;94(6):531–6.
Barié A, Ehmann Y, Jaber A, Huber J, Streich NA. Revision ACL reconstruction using quadriceps or hamstring autografts leads to similar results after 4 years: good objective stability but low rate of return to pre-injury sport level. Knee Surg Sports Traumatol Arthrosc. 2019;27:3527–35. https://doi.org/10.1007/s00167-019-05444-z. [Epub ahead of print].
Papageorgiou CD, Ma CB, Abramowitch SD, Clineff TD, Woo SL. A multidisciplinary study of the healing of an intraarticular anterior cruciate ligament graft in a goat model. Am J Sports Med. 2001;29:620–6.
Xie X, Liu X, Chen Z, Yu Y, Peng S, Li Q. A meta-analysis of bone-patellar tendon-bone autograft versus four-strand hamstring tendon autograft for anterior cruciate ligament reconstruction. Knee. 2015;22:100–10.
Mohtadi NG, Chan DS, Dainty KN, Whelan DB. Patellar tendon versus hamstring tendon autograft for anterior cruciate ligament rupture in adults. Cochrane Database Syst Rev. 2011;9:CD005960.
Gifstad T, Foss OA, Engebretsen L, Lind M, Forssblad M, Albrektsen G, Drogset JO. Lower risk of revision with patellar tendon autografts compared with hamstring autografts: a registry study based on 45,998 primary ACL reconstructions in Scandinavia. Am J Sports Med. 2014;42:2319–28.
Samuelsen BT, Webster KE, Johnson NR, Hewett TE, Krych AJ. Hamstring autograft versus patellar tendon autograft for ACL reconstruction: is there a difference in graft failure rate? A meta-analysis of 47,613 patients. Clin Orthop Relat Res. 2017;475(10):2459–68.
Kay J, Naji L, Simunovic N, Peterson D, Samuelsson K, Musahl V, Ayeni OR. Graft choice has no significant influence on the rate of return to sport at the preinjury level after revision anterior cruciate ligament reconstruction: a systematic review and meta-analysis. J ISAKOS. 2016;2:21. https://doi.org/10.1136/jisakos-2016-000113.
Keizer MNJ, Hoogeslag RAG, van Raay JJAM, Otten E, Brouwer RW. Superior return to sports rate after patellar tendon autograft over patellar tendon allograft in revision anterior cruciate ligament reconstruction. Knee Surg Sports Traumatol Arthrosc. 2018;26(2):574–81.
Victor J, Bellemans J, Witvrouw E, Govaers K, Fabry G. Graft selection in anterior cruciate ligament reconstruction-pro- spective analysis of patellar tendon autografts compared with allografts. Int Orthop. 1997;21(2):93–7.
Hart JM, Turman KA, Diduch DR, Hart JA, Miller MD. Quadriceps muscle activation and radiographic osteoarthritis following ACL revision. Knee Surg Sports Traumatol Arthrosc. 2011;19(4):634–40.
Victor J, Bellemans J, Witvrouw E, Govaers K, Fabry G. Graft selection in anterior cruciate ligament reconstruction-prospective analysis of patellar tendon autografts compared with allografts. Int Orthop. 1997;21(2):93–7.
Grassi A, Kim C, Marcheggiani MGM, Zaffagnini S, Amendola A. What is the mid-term failure rate of revision ACL reconstruction? A systematic review. Orthop Relat Res. 2017;475(10):2484–99.
Legnani C, Zini S, Borgo E, Ventura A. Can graft choice affect return to sport following revision anterior cruciate ligament reconstruction surgery? Arch Orthop Trauma Surg. 2016;136(4):527–31.
Arianjam A, Inacio MCS, Funahashi TT, Maletis GB. Analysis of 2019 patients undergoing revision anterior cruciate ligament reconstruction from a community-based registry. Am J Sports Med. 2017;45(7):1574–80.
Thomas N, Carmichael J. Failure in ACL reconstruction: etiology, treatment, and results. In: Bonnin M, Amendola A, Bellemans J, MacDonald S, Ménétrey J, editors. The knee joint. Paris: Springer; 2012. p. 343–53.
Prodromos CC, Joyce BT. Allograft complications and risk factors. In: Prodromos C, Brown C, Fu FH, et al., editors. The anterior cruciate ligament reconstruction and basic science. Philadelphia: Saunders-Elsevier; 2008. p. 561–4.
Pereira H, Sevivas N, Pereira R, Espregueira-Mendes J, et al. Systematic approach from Porto School. In: Siebold R, Dejour D, Zaffagnini S, editors. Anterior cruciate ligament reconstruction: a practical surgical guide. Heidelberg: Springer; 2014. p. 367–86.
Lind M, Menhert F, Pedersen AB. Incidence and outcome after revision anterior cruciate ligament reconstruction: results from the Danish registry for knee ligament reconstructions. Am J Sports Med. 2012;40(7):1551–7.
Nissen KA, Eysturoy NH, Nielsen TG, Lind M. Allograft use results in higher re-revision rate for revision anterior cruciate ligament reconstruction. Orthop J Sports Med. 2018;6(6):2325967118775381.
Condello V, Zdanowicz U, Di Matteo B, et al. Allograft tendons are a safe and effective option for revision ACL reconstruction: a clinical review. Knee Surg Sports Traumatol Arthrosc. 2019;27(6):1771–81.
Maletis GB, Inacio MC, Reynolds S, Desmond JL, Maletis MM, Funahashi TT. Incidence of postoperative anterior cruciate ligament reconstruction infections: graft choice makes a difference. Am J Sports Med. 2013;41:1780–5.
Erickson BJ, Cvetanovich G, Waliullah K, Khair M, Smith P, Bach B Jr, Sherman S. Two-stage revision anterior cruciate ligament reconstruction. Orthopedics. 2016;39(3):e456–64.
Rizer M, Foremny GB, Rush A, Singer AD, Baraga M, Kaplan LD, Jose J. Anterior cruciate ligament reconstruction tunnel size: causes of tunnel enlargement and implications for single versus two-stage revision reconstruction. Skelet Radiol. 2017;46(2):161–9.
Groves C, Chandramohan M, Chew C, Subedi N. Use of CT in the management of anterior cruciate ligament revision surgery. Clin Radiol. 2013;68(10):e552–9.
Harvey A. Fixation of the graft in reconstruction of the anterior cruciate ligament. J Bone Joint Surg Br. 2005;87:593–603.
Cheung P, Chan WL, Yen CH, et al. Femoral tunnel widening after quadrupled hamstring anterior cruciate ligament reconstruction. J Orthop Surg (Hong Kong). 2010;18:198–202.
Thomas NP. Revision anterior cruciate ligament. In: Prodromos C, Brown C, Fu FH, Georgoulis AD, Gobbi A, Howell SM, Johson DL, Paulos LE, Shelbourne KD, editors. The anterior cruciate ligament: reconstruction and basic science. Philadelphia: Saunders Elsevier; 2008. p. 443–57.
Garofalo R, Djahangiri A, Siegrist O. Revision anterior cruciate ligament reconstruction with quadriceps tendon-patellar bone autograft. Arthroscopy. 2006;22(2):205–14.
Trojani C, Sbihi A, Djian P, et al. Causes for failure of ACL reconstruction and influence of meniscectomies after revision. Knee Surg Sports Traumatol Arthrosc. 2011;19(2):196–201.
Borchers JR, Kaeding CC, Pedroza AD, Huston LJ, Spindler KP, Wright RW. Intra-articular findings in primary and revision anterior cruciate ligament reconstruction surgery: a comparison of the MOON and MARS study groups. Am J Sports Med. 2011;39(9):1889–93.
Wright R, Spindler K, Huston L, et al. Revision ACL reconstruction outcomes: MOON cohort. J Knee Surg. 2011;24(4):289–94.
Wright RW, Huston LJ, Spindler KP, et al. Descriptive epidemiology of the multicenter ACL revision study (MARS) cohort. Am J Sports Med. 2010;38(10):1979–86.
Wu WH, Hackett T, Richmond JC. Effects of meniscal and articular surface status on knee stability, function, and symptoms after anterior cruciate ligament reconstruction a long-term prospective study. Am J Sports Med. 2002;30(6):845–50.
Sonnery-Cottet B, Praz C, Rosenstiel N, Blakeney WG, Ouanezar H, Kandhari V, Vieira TD, Saithna A. Epidemiological evaluation of meniscal ramp lesions in 3214 anterior cruciate ligament-injured knees from the SANTI study group database: a risk factor analysis and study of secondary meniscectomy rates following 769 ramp repairs. Am J Sports Med. 2018;46(13):3189–97.
Temponi EF, de Carvalho Júnior LH, Saithna A, Thaunat M, Sonnery-Cottet B. Incidence and MRI characterization of the spectrum of posterolateral corner injuries occurring in association with ACL rupture. Skelet Radiol. 2017;46(8):1063–70.
Musahl V, Citak M, O’Loughlin PF, Choi D, Bedi A, Pearle AD. The effect of medial versus lateral meniscectomy on the stability of the anterior cruciate ligament-deficient knee. Am J Sports Med. 2010;38(8):1591–7.
Stephen JM, Halewood C, Kittl C, Bollen SR, Williams A, Amis AA. Posteromedial meniscocapsular lesions increase tibiofemoral joint laxity with anterior cruciate ligament deficiency, and their repair reduces laxity. Am J Sports Med. 2016;44(2):400–8.
Shon OJ, Park JW, Kim BJ. Current concepts of posterolateral corner injuries of the knee. Knee Surg Relat Res. 2017;29(4):256–68.
LaPrade RF, Resig S, Wentorf F, et al. The effects of grade III posterolateral knee complex injuries on anterior cruciate ligament graft force. A biomechanical analysis. Am J Sports Med. 1999;27:469–75.
Pacheco RJ, Ayre CA, Bollen SR. Posterolateral corner injuries of the knee: a serious injury commonly missed. J Bone Joint Surg Br. 2011;93:194–7.
LaPrade RF, Wentorf FA, Fritts H, et al. A prospective magnetic resonance imaging study of the incidence of posterolateral and multiple ligament injuries in acute knee injuries presenting with a hemarthrosis. Arthroscopy. 2007;23:1341–7.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 ESSKA
About this chapter
Cite this chapter
Goto, K., Menetrey, J. (2020). Surgical Technique: What We Would Do in Different Situations—Graft Choice, One or Two Steps, Fixation, Associated Lesions. In: Hirschmann, M., Kon, E., Samuelsson, K., Denti, M., Dejour, D. (eds) ESSKA Instructional Course Lecture Book . Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-662-61264-4_21
Download citation
DOI: https://doi.org/10.1007/978-3-662-61264-4_21
Published:
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-662-61263-7
Online ISBN: 978-3-662-61264-4
eBook Packages: MedicineMedicine (R0)