Abstract
Plants face a multitude of abiotic and biotic stresses with varying severity throughout their life, and these stresses can result in varying changes to the ecosystem services provided by the plants. Climate change involves modification of several environmental drivers, and it is predicted to increase the frequency and severity of various abiotic and biotic stresses, including rising temperatures, increasingly uneven distribution of precipitation, and more frequent outbreaks of herbivore and pathogen attacks. As any stress reduces plant CO2 fixation, enhanced stress frequency and severity are expected to lead to faster rise of atmospheric CO2 concentration, thereby further exacerbating climate change. On the other hand, plants can importantly modify their own life environment by release of volatile organic compounds (BVOC). The plant-generated volatiles modify the oxidative status of the ambient atmosphere by enhancing the rate of ozone formation in atmospheres polluted by mono-nitrogen oxides (NOx). From this perspective, plant emissions can be considered as ecosystem “disservice.” Plant-emitted volatiles also importantly participate in aerosol and cloud formation in both polluted and non-polluted atmospheres, thereby reducing solar radiation penetration and ambient temperature. Plant-facilitated cooling can partly counteract global warming, and thus, plant emissions provide an important global regulatory ecosystem service. Apart from constitutive volatile emissions that are present in only some species and are expected to decrease under stress, especially under severe stress, all plants respond to stresses by inducing BVOC emissions that serve as signal molecules eliciting stress response pathways and leading to plant acclimation. These induced BVOC emissions, the plant “talk,” also contribute to atmospheric processes and can potentially reduce the stress severity, and, accordingly, stress-driven reductions in CO2 uptake. Thus, the stress responses and acclimation of vegetation to future environmental stresses can importantly modify the speed and magnitude of climate change.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
- Constitutive Emissions
- Climate Change
- Induced Emissions
- Global Feedbacks
- Plant-Biosphere Relationships
- Stress
- Terpenoids
- Volatile Organic Compounds
1 Introduction What Are Plant Volatiles?
Plant-released organic volatiles constitute a vast spectrum of compounds, more than 30,000 different compounds with a certain capacity to escape into the gas phase from a liquid or solid (Niinemets et al. 2004). In common with the compounds characteristically called volatiles is that they have normal pressure boiling points between ca. 30 and 250 °C and, thus, support a relatively high vapor partial pressure at ambient temperatures (between ca. 101 and 105 Pa at 25 °C) (Fuentes et al. 2000; Copolovici and Niinemets 2005; Kosina et al. 2013). In addition, studies on plant volatiles also often consider semivolatiles that support a much lower vapor pressure (partial pressure ca. 10−6 and 100 at 25 °C) (Helmig et al. 2003; Widegren and Bruno 2010; Kosina et al. 2013). Semivolatiles have a large capacity for partitioning into liquid and solid phases and, once released from plants, play a major role in atmospheric particle formation (Ehn et al. 2014).
All plants emit a plethora of volatiles that are synthesized in different subcellular compartments involving multiple biochemical pathways (Fig. 1), and the emissions can be further tissue- and organ-specific. The volatiles emitted can be intermediates of normal plant metabolic activity and are released from plant tissues because the metabolic pathways are “leaky.” Emissions of such compounds can be enhanced under certain periods of plant life. For instance, plants emit methanol as the result of demethylation of cell wall pectins (Nemecek-Marshall et al. 1995; Fall and Benson 1996). Methanol emissions occur at low level from all physiologically active plant tissues, but the emissions are strongly enhanced in growing tissues due to relaxation and rigidification of cell walls during tissue expansion growth (Harley et al. 2007; Hüve et al. 2007). Analogously, root zone hypoxia during flooding leads to ethanol formation in the roots and its transportation to the leaves with the transpiration stream (Bracho Nunez et al. 2009; Kreuzwieser and Rennenberg 2013). Ethanol that reaches the leaves can be further enzymatically oxidized to acetic acid via acetaldehyde, and enter into the primary metabolism, but some ethanol and acetaldehyde escape leaves due to limited alcohol and aldehyde dehydrogenase activities (Kreuzwieser et al. 2000, 2001; Rottenberger et al. 2008; Kreuzwieser and Rennenberg 2013).
Main volatiles emitted by plants are formed in different subcellular compartments and their synthesis involves a variety of biochemical pathways . Methanol and lipoxygenase (LOX) pathway volatiles (also called green leaf volatiles, dominated by various C6 aldehydes and alcohols) are ubiquitous volatiles that can be released from all plant tissues as the result of constitutive activity of key enzymes of their synthesis pathways, while volatile isoprenoids (hemiterpenes like isoprene, monoterpenes, and sesquiterpenes) and phenylpropanoids and benzenoids are specialized volatiles and are emitted as the result of induction of expression of genes coding for specific rate-limiting synthases, although in several species, certain specialized volatiles can be emitted constitutively. Methanol is released as the result of demethylation of pectins in cell walls in growing tissues or upon different biotic and abiotic stresses, whereas green leaf volatiles are formed from free polyunsaturated fatty acids released from membrane lipids upon membrane damage characteristic to exposure to severe stresses (Liavonchanka and Feussner 2006; Andreou and Feussner 2009). Emissions of phenolic compounds typically originate from cytosol, but isoprenoid emissions can originate from plastids or cytosol, depending on the compound emitted (Niinemets et al. 2013; Rosenkranz and Schnitzler 2013; Pazouki and Niinemets 2016). There is evidence that some terpenoids can be potentially also released from mitochondria (not shown in the figure, see Tholl and Lee 2011; Dong et al. 2016), but the possible mitochondrial release of volatiles is much less studied
These examples demonstrate how normal physiological processes in plant life, and the ecosystem services they provide, can be associated with major leakage of organic compounds due to relatively high vapor pressure of these compounds. Apart from metabolic intermediates, a large number of organic compounds are specifically made to be volatile, including several volatile benzenoids and phenylpropanoids and volatile isoprenoids such as the hemiterpene isoprene (C5), monoterpenes (C10), and sesquiterpenes (C15) (Fig. 1, Peñuelas and Llusià 2004; Fineschi et al. 2013; Guenther 2013a; Portillo-Estrada et al. 2015). These specialized volatiles constitute the plant “talk,” fulfilling a plethora of biological and ecological functions from communication among plant organs, other plants, and other organisms to altering the plant stress resistance. Furthermore, all volatiles participate in multiple landscape- to global-scale processes, altering ambient air oxidative status, atmospheric particle condensation, and cloud cover (Peñuelas and Staudt 2010; Kulmala et al. 2013). Both the specific biological and broad-scale atmospheric roles of volatiles result in a number of key ecosystem services beneficial to humans. As discussed in this chapter, these services can be local to regional scale services such as preservation of ecosystem integrity under biotic and abiotic stresses and, thus, the preservation of the capacity to provide the “standard,” well-perceived, ecosystem services to humans, e.g., wood production of forest stands. These services also include regional to global-scale services such as environmental cooling and dampening the global climate change .
In the current chapter, I first provide a short overview of key types of biological volatile emission and of the environmental controls on volatile emission and modification of emissions by abiotic and biotic stresses. Then I focus on the biological roles of volatiles, on the roles of volatiles in large-scale biosphere-atmosphere processes , and on ecological services provided by plant volatiles. I demonstrate that the trace gas release is a key vegetation characteristic that contributes a number of unique ecosystem services that alter the performance of ecosystems in current and future climates with major implications for human life. Quantitative significance of plant volatiles in Earth system processes is still poorly understood, and the role of plant volatiles in climate has been largely neglected in the last report of the Intergovernmental Panel on Climate Change (IPCC, Field et al. 2014; Stocker et al. 2014). In this chapter, I show that the evidence is accumulating that plant trace gas exchange participates in multiple feedback loops that can potentially play major roles in Earth system processes and argue that biosphere-atmosphere interactions mediated by plant volatiles need to be included in Earth system models intending to predict future climate.
2 Plant Volatile Diversity, Environmental Controls on Emission and Emission Capacities
2.1 Sites of Volatile Synthesis and Diversity
Synthesis of volatile phenolics typically occurs in cytosol, while volatile isoprenoids can be synthesized in plastids or cytosol, depending on the compound emitted (Niinemets et al. 2013; Pazouki and Niinemets 2016). Hemiterpene and monoterpene synthesis is considered to occur in the plastids where corresponding C5 and C10 terminal enzymes responsible for terpene synthesis, terpene synthases, are located, while sesquiterpene synthases are located in the cytosol (Fig. 1, Chen et al. 2011). However, there is also recent evidence of mixed substrate specificity of some terpene synthases (Pazouki and Niinemets 2016), suggesting that product profiles can potentially vary depending on substrate availability in different subcellular compartments. Furthermore, there is evidence that some mono- and sesquiterpenes can be synthesized in mitochondria (Tholl and Lee 2011; Dong et al. 2016), further complicating the picture.
The diversity of emitted volatiles varies for different volatile compound classes. In the case of hemiterpenes, in addition to isoprene, plants also emit the oxygenated hemiterpene 2-methyl-3-buten-2-ol (Gray et al. 2006, 2011), but mono- and sesquiterpene-emitters typically release a wide spectrum of compounds. Often more than 20 different terpenoids are observed in the emissions from a single species (e.g., Niinemets et al. 2002a; Winters et al. 2009). Such a high diversity reflects the presence of multiple terpene synthases in the given emitting species (e.g., Falara et al. 2011; Jiang et al. 2016a) but also the specific reaction mechanism of terpene synthesis. In particular, terpene synthesis involves formation of a highly reactive carbocation intermediate, and depending on the extent to which the carbocation can be stabilized, the product specificity of terpene synthases strongly varies (e.g., Christianson 2008). In fact, most terpene synthases catalyze formation of multiple products, and only some terpene synthases form single terpenes, explaining the huge chemical diversity of emitted volatile terpenoids.
2.2 Constitutive and Stress-Induced Volatile Emissions
2.2.1 Constitutive Emissions of Specialized Volatiles
While all plant species can emit metabolic intermediates, only some species emit specialized volatiles, in particular, volatile isoprenoids under typical non-stressed physiological conditions, being, thus, constitutive emitters (Peñuelas and Llusià 2004; Fineschi et al. 2013). The capacity for constitutive emission of certain volatiles requires that the specific synthesis pathways are constitutively active, although the degree of activation can vary with environmental conditions, sometimes several-fold (e.g., Niinemets et al. 2010b). The constitutive emissions can result from emission of compounds stored in specialized storage structures such as oil glands in Citrus species, resin ducts in conifers, and glandular trichomes in species from Labiatae or Solanaceae. Typically, volatiles stored in these structures are mono- and sesquiterpenes, but sometimes benzenoids are also stored (Loreto et al. 2000; Jiang et al. 2016a). Filling up the storage structures takes typically multiple days to months, and thus, the release of the volatiles from the storage is uncoupled from the synthesis of these compounds. Thus, the rate of constitutive emissions from storage structures depends on the rate of compound evaporation and diffusion and therefore scales exponentially with temperature (Niinemets et al. 2010b; Grote et al. 2013).
The hemiterpenes, isoprene and 2-methyl-3-buten-2-ol, cannot be stored due to high volatility. In addition, several constitutive monoterpene emitters such as the Mediterranean evergreen oaks Quercus ilex and Q. suber and the broad-leaved deciduous temperate tree Fagus sylvatica lack specialized storage structures (Staudt et al. 2004; Dindorf et al. 2006). In hemiterpene and non-storage monoterpene emitters, the volatile emissions result from de novo compound synthesis, and the emissions respond to environmental drivers similarly to photosynthesis, increasing asymptotically with light intensity and scaling positively with temperature up to an optimum temperature and decreasing thereafter (Niinemets et al. 2010b; Monson et al. 2012). Emissions of de novo synthesized volatiles also respond to immediate changes in ambient CO2 concentration. In particular, isoprene emissions decrease with increasing CO2 concentration (e.g., Wilkinson et al. 2009), but the CO2 response is less clear for monoterpene emissions (Sun et al. 2012 for a discussion). However, the CO2 sensitivity of isoprene is gradually lost at higher temperatures (Rasulov et al. 2010; Li and Sharkey 2013), and the emission response to longer-term changes in ambient CO2 concentration can be different from the immediate response due to acclimation of isoprene synthesis pathway to long-term ambient CO2 concentration (Sun et al. 2012).
2.2.2 Induction of Volatiles Upon Abiotic and Biotic Stresses
In field environments, plants are often exposed to various abiotic and biotic stresses . Although only some plant species can emit volatiles constitutively, all species can be triggered to release volatiles upon biotic and abiotic stresses (Fig. 2). Among the volatiles triggered are ubiquitous stress compounds such as lipoxygenase (LOX) pathway volatiles (also called green leaf volatiles). Green leaf volatiles, typically dominated by various C6 aldehydes and alcohols, are rapidly formed when free polyunsaturated fatty acids are released from membrane lipids (Beauchamp et al. 2005; Copolovici et al. 2011). This typically occurs upon membrane-level damage characteristic to exposure to severe stresses such as mechanic wounding during biotic stresses but also upon exposure to severe heat, frost, and ozone stress (Beauchamp et al. 2005; Copolovici et al. 2011). Methanol , which can be released from non-stressed tissues constitutively, also serves as another ubiquitous stress volatile; major rapid methanol bursts are associated with both severe biotic and abiotic stresses (Beauchamp et al. 2005; von Dahl et al. 2006).
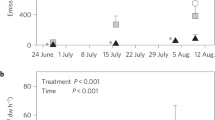
Induction of monoterpene emissions in broad-leaved temperate deciduous tree Alnus incana upon feeding by larvae of the moth Cabera pusaria (a), and correlation of the degree of elicitation of emissions with the amount of leaf area consumed on the fourth day since the start of feeding (b). Alnus incana does not significantly emit volatiles in non-stressed conditions and is therefore considered a non-emitter species, but exposure to different stresses results in significant emissions of green leaf volatiles, mono-, sesqui-, and homoterpenes (Modified from Copolovici et al. 2011)
Apart from the ubiquitous compounds emitted from virtually all plant species, induction of synthesis of specialized compounds requires the presence of given stress-elicited synthase genes in plant genomes. For instance, glucosinolate pathway volatiles (short-chained S- and/or N-containing volatiles resulting from breakdown of glucosylated amino acids and their derivatives) are only released upon stress from the species in the order Brassicales (Kask et al. 2016). On the other hand, all plants do include terpene synthase genes in their genomes, and expression of these genes is typically activated upon stress. Studies have demonstrated elicitation of emissions of mono- and sesquiterpenes upon biotic stresses such as herbivory (Fig. 2, Blande et al. 2007; Copolovici et al. 2014a, 2011; Farré-Armengol et al. 2015); pathogen infections such as powdery mildew (Fig. 3), leaf rust (Fig. 4), and canker fungus (Achotegui-Castells et al. 2015) infections; and upon abiotic stresses such as ozone stress (Beauchamp et al. 2005) and heat stress (Copolovici et al. 2012; Kask et al. 2016). However, the number of terpenoid synthase genes strongly varies among species from as few as only one synthase gene to more than 80 genes (Rajabi Memari et al. 2013), implying that the diversity of induced terpenoid emission responses can also be variable. Furthermore, different stresses can trigger emissions of different volatiles (Dicke et al. 2009; Zhang et al. 2009), but so far, understanding of the overall stress-dependent emission diversity and variation of emission profiles under different stresses is very limited.
Oak powdery mildew (Erysiphe alphitoides) is a major pathogen infesting pedunculate oak (Quercus robur) all over Europe. The visual damage symptoms can be detected through the growing season in young oak trees (a), and in late summer and autumn in old plants (b). Erysiphe alphitoides infections are associated with reduction of constitutive emissions of isoprene (c) and elicitation of emissions of monoterpenes (d), sesqui- and homoterpenes (data not shown) and green leaf volatiles (e) ((c–e) Modified from Copolovici et al. 2014b). Open symbols denote non-infected leaves
Poplar (Populus spp.) is the telial host for the widespread rust fungus Melampsora larici-populina, infections of which are initially associated with diffusely spread yellow spots (a), followed by extensive leaf yellowing and necrosis and premature leaf senescence encompassing large parts of the canopy (encircled area in b). Melampsora larici-populina infection reduces constitutive emissions of isoprene (c) and induces emissions of monoterpenes (d), sesqui- and homoterpenes (data not shown) and green leaf volatiles (e) in an infection-dependent manner (Jiang et al. 2016b). Open symbols refer to emissions from leaves without visible signs of infection. Pictures (a) and (b) are for P. laurifolia and data (c–e) for P. balsamifera var. suaveolens
Differently from LOX volatiles and methanol , emissions of which are triggered rapidly due to the presence of a certain constitutive activity of lipoxygenases (Liavonchanka and Feussner 2006) and pectin methylesterases (Micheli 2001), elicitation of terpenoid emissions is more time-consuming because it requires gene expression and protein synthesis to reach a certain terpene synthase activity. Typically, emissions of terpenoids can be detected hours after the stress impact and the emissions peak 24–48 h after the impact (Fig. 2, Pazouki et al. 2016). On the other hand, when the stress is relieved, the emissions gradually decrease, reaching the initial non-induced level in a few days after the stress relief (Fig. 2a). This reduction is a characteristic feature to induced emissions and contrasts to constitutive emissions. Although the rate of constitutive emissions can also be affected by stress, typically negatively the level of constitutive emissions almost never reaches zero.
2.3 Relationships Among Constitutive and Stress-Induced Emissions
As noted above, constitutive emissions are present only in several plant species, while induced emissions can be elicited in all species, including the constitutive emitters . Typically, the composition of induced emissions is different from constitutive emissions. For instance, in constitutively isoprene-emitting deciduous oak Quercus robur, infection by oak powdery mildew (Erysiphe alphitoides) results in emissions of mono- and sesquiterpenes (Fig. 3). Analogously, infection of the constitutively isoprene-emitting poplar (Populus spp.) with the rust fungus Melampsora larici-populina, results in mono- and sesquiterpene emissions (Fig. 4).
In the case of constitutive monoterpene emitters , an environmental or a biotic stress often results in elicitation of emissions of monoterpenes different from constitutively emitted monoterpenes. In particular, typical stress-elicited monoterpenes are ocimenes, linalool, and 1,8-cineole, while constitutive emissions are characteristically dominated by limonene and pinenes (Staudt and Bertin 1998; Niinemets et al. 2002b). Importantly, in constitutive storage emitters, stress-induced monoterpene emissions reflect de novo synthesis of volatiles and scale similarly with light and temperature as the emissions in non-storage emitters (Niinemets et al. 2010a, b). In addition to monoterpenes, stress often results in elicitation of emissions of sesquiterpenes and homoterpenes 4,8-dimethylnona-1,3,7-triene (DMNT) and 4,8,12-trimethyltrideca-1,3,7,11-tetraene (TMTT) that are not observed in constitutive emissions (e.g., Niinemets et al. 2010b; Staudt and Lhoutellier 2011; Tholl et al. 2011).
In both of the biotic stress case studies highlighted here, the constitutive isoprene emissions decreased in pathogen-infected leaves (Figs. 3 and 4), and analogous negative relationships between induced and constitutive emissions have been demonstrated in the case of other stresses as well (e.g., Kleist et al. 2012 for heat stress). Overall, positive stress dose vs. induced emission relationships have been observed for several abiotic stresses such as frost and heat stress (Copolovici et al. 2012) and ozone stress (Beauchamp et al. 2005). Although biotic impacts have been considered to be hard to quantify (Niinemets et al. 2013), quantitative stress dose vs. induced emission responses have been observed for several biotic stresses such as herbivory (Fig. 2) and fungal pathogen infections (Figs. 3 and 4). The key issue with the biotic stresses seems to be how to quantify the severity of biotic stress (Copolovici and Niinemets 2016), but once the biotic stress severity has been properly characterized, it becomes clear that the rate of emission of volatile organic compounds scales with stress severity similarly to abiotic stresses (Niinemets et al. 2013; Copolovici and Niinemets 2016).
3 Ecosystem Services and Impacts of Plant Volatiles
3.1 Biological Role of Plant Volatiles in Ecological Processes
The biological role of constitutive emissions of isoprene and monoterpenes is not yet fully clear, but there is increasing evidence that these compounds have antioxidative and/or membrane-stabilizing properties (Sharkey et al. 2001; Loreto et al. 2004; Vickers et al. 2009; Peñuelas and Staudt 2010), and it has been postulated that they play an important role in enhancing the abiotic stress resistance in species emitting them (Vickers et al. 2009; Possell and Loreto 2013). Furthermore, volatiles released from constitutively emitting species can be taken up from the ambient air by neighboring non-emitting species (Copolovici et al. 2005; Noe et al. 2008; Himanen et al. 2010). This can enhance the stress resistance of non-emitters, resulting in an overall increase in ecosystem stress resistance. Such an increase of ecosystem stress resistance is the key ecological service which allows for maintenance of all other well-recognized ecosystem services provided by healthy ecosystems.
While the constitutive emissions can provide certain protection against chronic mild abiotic stresses that do not lead to induction of stress volatiles, once induced, chemically similar induced volatiles can also directly participate in stress protection during short-term severe stresses that trigger their emission. However, more importantly, stress-elicited volatiles play various functions in communication among plants and plants and other organisms (Dicke and Baldwin 2010; Holopainen et al. 2013; Blande et al. 2014). In plants, stress-induced volatiles serve as infochemicals eliciting stress response pathways, leading to plant acclimation to the altered environmental conditions and priming defenses against herbivore and pathogen attacks in leaves and neighboring not yet stressed plants (Dicke and Baldwin 2010; Peng et al. 2011). Such a defense priming can again augment the whole ecosystem resistance to both abiotic and biotic stresses. Furthermore, herbivory-induced volatiles serve as infochemicals for the enemies of herbivores, and thus, the release of attractants to predatory and parasitic insects can importantly reduce the spread of herbivores (D’Alessandro et al. 2006; Dicke and Baldwin 2010) and, thus, provides a further important means for enhancing the resilience of ecosystems.
3.2 Plant Volatiles in Broad-Scale Ecological Processes
Apart from the biological role of plant-emitted volatiles, plant-generated volatiles play important roles in large-scale regional and global processes. The global amount of emitted biogenic volatiles has been estimated to be roughly 1.1 Pg yr−1 (equivalent to ca. 0.84 Pg C yr−1) (Guenther et al. 2012). Thus, the biogenic release of trace gases exceeds the anthropogenic release by more than a factor of ten (Guenther et al. 2012). Despite only certain species are capable of constitutive isoprenoid emissions , many of these species are widely distributed, often being the dominating plant species in given ecosystems. In fact, global plant emissions are currently derived using species-specific emission potentials obtained by extensive screening studies (e.g., Karlik and Winer 2001; Simon et al. 2006; Keenan et al. 2009; Llusià et al. 2010, 2014). The emphasis in these screening studies has been on constitutive emissions, and thus, global emission estimates are mainly based on constitutive emitters (Guenther et al. 2012).
Indeed, constitutively emitted isoprene is the most important plant-generated volatile compound with the global source strength predicted to be ca. 550 Tg yr−1 by different models (Arneth et al. 2008; Guenther et al. 2012; Guenther 2013b). While different global emission models based on profoundly different emission algorithms converge to a similar value of global isoprene emission, due to uncertainties in the share of storage vs. de novo emissions and constitutive vs. stress-induced emissions , the model estimates are more variable for mono- and sesquiterpenes than for isoprene (Arneth et al. 2008). Based on empirical model approaches, total emissions of ca. 160 Tg yr−1 for mono- and ca. 30 Tg yr−1 for sesquiterpenes have been estimated (Guenther et al. 2012). The rest of global BVOC emission source strength of ca. 0.4 Pg yr−1 is mainly made up of oxygenated compounds, dominated by methanol , ethanol, acetone, and acetaldehyde (Guenther et al. 2012).
The largest uncertainty in the global volatile emission estimates seems to be the lack of proper consideration of stress-induced emissions. As shown above, exposure to stress conditions can turn a constitutively non-emitter or moderate emitter species into a strong emitter of mono- and sesquiterpenes. This might mean that the overall capacity of vegetation to emit volatiles has been strongly underestimated. Of course, the stress-elicited emissions occur only when there is a stress and emissions return to background levels when the stress is relieved (e.g., Fig. 2), but it is relevant to consider that these relatively short-termed emission peaks might not only importantly contribute to the total emission amount, but alter the timing of peak atmospheric concentrations of volatiles with major consequences for large-scale physiological and atmospheric processes.
3.2.1 Role of Volatiles in Altering Atmospheric Reactivity
Due to the large emissions, biogenic volatiles play major roles in biosphere-atmosphere processes . The chemical reactivity of non-oxygenated non-saturated compounds, in particular, non-oxygenated isoprenoids, is much larger than the reactivity of oxygenated volatiles, and thus, the role of different compound classes, oxygenated vs. non-oxygenated non-saturated compounds, in large-scale processes is different. Highly reactive compounds play a major role in ozone formation and quenching reactions in the troposphere. In particular, in a human-polluted air enriched with NOx (sum of NO and NO2), plant-generated volatiles contribute to photochemical ozone production and, in fact, control the rate of ozone formation in the atmosphere (Chameides et al. 1992; Fehsenfeld et al. 1992; Fuentes et al. 2000). In contrast, in non-polluted air with low NOx, reactive hydrocarbons contribute to a reduction of ozone concentrations (Lerdau and Slobodkin 2002; Atkinson and Arey 2003; Loreto and Fares 2007).
The influence of elevated ozone concentration on photosynthetic productivity and volatile emissions can be different over the short- and the long-term. Increases in ozone concentration can strongly reduce plant photosynthetic productivity, but constitutive volatile emission itself can initially increase under moderately elevated atmospheric ozone concentration (Calfapietra et al. 2013 for a review). This might have global consequences as a reduction in photosynthetic CO2 fixation can speed up elevation of atmospheric CO2 concentration, and this in turn can increase the rate of temperature increase (Sitch et al. 2007). Due to the positive effect of temperature on volatile emissions, this is further expected to enhance volatile release and ozone formation and reduce carbon gain even more (Lerdau 2007; Sitch et al. 2007). Furthermore, the concentration of ozone, significantly driven by the concentration of reactive volatiles, is itself an important greenhouse gas that can contribute to global warming (Shindell et al. 2006), amplifying these patterns.
A severe ozone stress results in a reduction of constitutive volatile emissions as well (Calfapietra et al. 2013 for a review), but it also leads to elicitation of induced emissions in both constitutive emitters and non-emitters (Beauchamp et al. 2005; Hartikainen et al. 2009). Among the induced volatiles, mono- and sesquiterpenes are typically more reactive in ozone formation reactions than isoprene (Calogirou et al. 1996) and, thus, could temporarily even speed up ozone formation, especially because these emissions are induced in all species in vegetation. So far, the understanding of ozone-dependent modifications in constitutive and induced emissions is only rudimentary, limiting quantitative assessment of species physiological responses in ozone formation potential of vegetation . Nevertheless, the available evidence suggests that the use of static emission factors estimated from screening studies that have considered only constitutive emitters and omission of physiological modifications in volatile emissions driven by ozone can lead to major uncertainties in predicting tropospheric ozone formation and quenching.
3.2.2 Volatiles in Altering Solar Radiation Scattering and Penetration and Ambient Temperature
Upon oxidation, the volatility of isoprenoids dramatically decreases, implying that they partition much more strongly to the liquid and solid phases than to the gas phase, creating secondary organic aerosols (SOA) (Kulmala et al. 2004a; Chen and Hopke 2009; Mentel et al. 2009; Kirkby et al. 2016). The presence of SOA decreases atmospheric clearness, thereby potentially reducing solar radiation penetration, but also increasing light scattering and, thus, the diffuse to total solar radiation ratio (Fig. 5, Malm et al. 1994; Farquhar and Roderick 2003; Misson et al. 2005). Because diffuse radiation penetrates deeper into the plant canopies and results in a more uniform distribution of solar radiation (Cescatti and Niinemets 2004), increases in diffuse to total solar radiation ratio enhance vegetation productivity (Gu et al. 2002, 2003; Mercado et al. 2009). Due to strong light effects on constitutive de novo volatile emissions , greater canopy light interception is expected to directly enhance these emissions (Fig. 5). In fact, the emissions are expected to increase even more than the rate of photosynthesis, because de novo volatile emissions are more light-limited than photosynthetic carbon fixation, typically saturating at higher light intensities than photosynthesis (Niinemets et al. 1999, 2015). Although the connection between photosynthesis and storage emissions is less straightforward, enhanced carbon availability can also enhance the emissions in the storage emitters by increasing the size of the storage pools (Blanch et al. 2007, 2009, 2011).
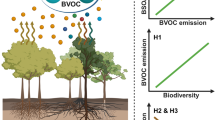
Regional and global feedback relationships between constitutive biogenic volatile organic compound (BVOC) emissions and climatic drivers . The rates of BVOC emission are controlled by environmental drivers, whereas environmental drivers, in turn, are modified by BVOC emission generating feedback loops. There are two main types of BVOC emitters, the storage emitters and the de novo emitters (Grote et al. 2013; Copolovici and Niinemets 2016). In the storage emitters, BVOC is released from large storage pools, and the emissions depend only on temperature, increasing exponentially with increasing temperature (Niinemets et al. 2010b; Grote et al. 2013). In the case of de novo emitters, the emissions rely on immediately synthesized BVOCs and depend both on light intensity and temperature. These emissions increase asymptotically with increasing light intensity and increase exponentially with temperature up to an optimum temperature and decline thereafter (Niinemets et al. 2010b; Grote et al. 2013). For simplicity, the figure shows only the environmental controls on BVOC emission for de novo emitters. In the case of light, light interception by plant canopies depends on total solar radiation flux and the distribution of solar radiation between diffuse and direct components. Diffuse light drives canopy photosynthesis more efficiently because of its penetration to deeper canopy layers and resulting greater uniformity of radiation field and total light interception (Gu et al. 2003). Atmospheric volatiles enhance concentration of secondary organic aerosols (SOA) that increase the radiation scattering, but SOA also moderately reduce total radiation penetration through atmosphere (Spracklen et al. 2008; Chen and Hopke 2009; Kulmala et al. 2013). Increasing concentrations of SOA, in turn, enhance the concentration of cloud condensation nuclei (CCN) thereby contributing to enhanced cloudiness and thus, increasing radiation scattering too, but CNN more strongly reduce total solar radiation flux penetrating through the atmosphere to the vegetation (Roderick et al. 2001; Huff Hartz et al. 2005; Spracklen et al. 2008; Kulmala et al. 2013). The other key environmental driver, temperature, depends on total solar radiation flux, but can also directly depend on CCN as the result of reduced thermal radiation losses to the space, but this effect is more relevant at night and is therefore not shown in the figure. In addition to the feedbacks shown, more complex feedbacks can operate through modifications in atmospheric CO2 and water vapor concentrations (e.g., Kulmala et al. 2013) as well as through induction of BVOC emissions upon abiotic and biotic stresses, severity of which depends on environmental drivers (section “Constitutive and Stress-Induced Volatile Emissions”)
Once formed in the atmosphere, the size of SOA particles increases in time due to condensation of atmospheric organics on particle surface (Kulmala et al. 2004a; Kirkby et al. 2016). These bigger particles can also serve as cloud condensation nuclei (CCN) , especially if their hygroscopicity also increases as the result of further compound oxidations on particle surface or as more hydrophilic compounds condense onto the particle surface (Engelhart et al. 2008; Kulmala et al. 2013). Greater CCN concentrations imply a greater condensation sink and potentially higher cloudiness that can dramatically increase the diffuse to total solar radiation ratio but also strongly reduce the total solar radiation penetration (Spracklen et al. 2008; Still et al. 2009; Kulmala et al. 2013). Thus, although radiation penetration into deeper canopy layers is increased by enhanced cloudiness, the reduction in total radiation intensity reaching to the top of the vegetation is the dominating factor, ultimately reducing the vegetation productivity and the rate of volatile emission (Fig. 5). Furthermore, cooling due to increased cloudiness also directly reduces both de novo and storage volatile emissions (Fig. 5).
As the result of volatile effects on SOA and CCN concentrations , multiple feedback loops operate between solar radiation, temperature, volatile emission, and productivity at regional and global scales, and the overall effect of volatiles on climate depends on the relative significance of these loops (Fig. 5, Kulmala et al. 2013). In particular, both rising SOA concentrations and greater cloudiness can reduce the surface temperature, and this can directly reduce volatile formation due to the physiological controls on the emission rates, and this, in turn, is expected to inhibit further SOA and CCN formation (Fig. 5, Kulmala et al. 2013). On the other hand, enhanced SOA concentrations increase the fraction of diffuse radiation, thereby increasing the rate of volatile release and further enhancing SOA and CCN formation (Fig. 5, Kulmala et al. 2013). In contrast, dramatic reductions in total solar radiation by enhanced cloudiness are expected to lead to decreased volatile formation, thereby feedback-inhibiting SOA and CCN formation . Testing the quantitative significance of these feedback loops requires a combination of long-term data with regional- and global-scale modeling. The first such modeling exercise based on 15 years of measurements of vegetation carbon fixation fluxes and 6 years of measurements of emission fluxes of volatile organic compounds in a boreal forest ecosystem suggests that such feedback loops do indeed operate in nature (Kulmala et al. 2013). Due to both physiological and acclimation responses of volatile emissions to changes in environmental drivers, quantitative prediction of the feedback responses is complex, and clearly, more such case studies are needed to scale up to other regions and the globe and to quantitatively evaluate the role of volatile emissions on solar radiation penetration and surface temperature.
3.2.3 Volatiles as Modifiers of the Lifetime of Greenhouse Gases
As demonstrated above, reactive plant volatiles in polluted atmospheres can exacerbate the plant abiotic stress due to enhancing the key atmospheric pollutant, ozone, concentrations, while reactive volatiles in non-polluted atmospheres can reduce ozone concentrations. Thus reactive plant hydrocarbons can alter the rate of global warming by changes in vegetation CO2 fixation capacity and, thus, by long-term modifications in atmospheric CO2 concentration. Furthermore, ozone itself is a strong greenhouse gas (section “Constitutive Emissions of Specialized Volatiles”, Shindell et al. 2006). In addition, alteration in solar radiation intensity, diffuse to total solar radiation ratio, and ambient temperature due to modifications in SOA and CCN concentrations directly affect the vegetation carbon fixation capacity and, thus, can strongly alter atmospheric CO2 concentration as well, creating further major feedback loops (Kulmala et al. 2004b, 2013).
Apart from the highly reactive compounds, less reactive oxygenated hydrophilic compounds such as aldehydes, alcohols, and ketones, including, for instance, lipoxygenase pathway volatiles, but also saturated oxygenated volatiles, can also partition to particle phase and participate in SOA and CCN formation (Mentel et al. 2009). In addition, several of these volatiles can react with atmospheric OH radicals, reducing atmospheric OH radical concentration (Fall 2003; Sinha et al. 2010; Nölscher et al. 2012). Given that the reaction with OH radicals is the primary process reducing the atmospheric concentrations of the key greenhouse gas methane , a reduction of OH concentration due to biogenic volatiles increases the methane lifetime in the atmosphere (Jacob et al. 2005; Ashworth et al. 2013; Voulgarakis et al. 2013), thereby significantly contributing to global warming. In particular, plant-generated emissions of methanol , the oxygenated volatile with the greatest global source strength (Stavrakou et al. 2011; Guenther et al. 2012), can potentially contribute to the greatest degree to the increases in methane lifetime.
3.3 Trace-Gas-Driven Ecosystem Services
The vegetation capacity to emit volatiles has not generally been considered as an ecosystem service, and overall, the atmosphere is often not considered as part of ecosystem services (Cooter et al. 2013). In fact, due to the contribution of volatiles to ozone formation in NOx-polluted atmospheres, volatile emission has even been considered an ecosystem “disservice” (Russo et al. 2016). However, from a biological perspective, plants can provide several key ecosystem services due to their trace gas emission. Although these ecosystem services are little-recognized in the community, they can have profound impacts on ecosystem performance. Among these biological services are:
-
Direct enhancement of plant abiotic stress resistance by directly quenching reactive oxidative species generated in plant membranes upon abiotic stress (Vickers et al. 2009) or more specifically, improving heat stress tolerance by increasing membrane stability (Velikova et al. 2012; Sun et al. 2013)
-
Increases in ecosystem resilience through communication among plants and with other organisms (e.g., plants and herbivore enemies), thereby ameliorating the biotic and abiotic stress impacts
-
Enhancement of ecosystem capacity to uphold diversity, in particular, to maintain the integrity of multitrophic interactions
Although often “hidden,” these services are crucial for stability and performance of ecosystems, and impairing some of these services can result in drastic deterioration of other ecosystem services. For instance, the carbon gain of chemically less diverse ecosystems such as monospecific tree plantations or crop fields can be much more vulnerable to deleterious pest attacks than that of more diverse ecosystems (Lerdau and Slobodkin 2002; Altieri and Nicholls 2004; Tooker and Frank 2012). This is highly relevant from the perspective of the “traditionally” perceived ecosystem services as pulpwood production of tree plantations or yield of crop fields, both of which are directly dependent on vegetation carbon gain.
From the perspective of atmospheric chemistry and large-scale biosphere-atmosphere processes , the balance between ecosystem service and “disservice” of volatile emissions importantly depends on the relative extent to which different atmospheric processes are affected by volatiles. The balance between different processes strongly varies regionally and depends on human effects on atmosphere. While in urban NOx-polluted atmospheres, volatiles emitted by vegetation contribute to elevated ozone concentration and photochemical smog, in remote non-polluted atmospheres with low NOx levels, plant-generated volatiles are expected to reduce atmospheric oxidant concentrations, including ozone concentrations (Lerdau and Slobodkin 2002). Analogously, reduction in atmospheric clearness and alteration of ambient air particle concentration by volatile contribution to SOA formation could be considered a disservice in urban habitats (Cooter et al. 2013), although it might also contribute to cooling of urban environments (Arneth et al. 2009).
From a global perspective, plant volatiles can provide three key ecosystem services:
-
Improvement of ecosystem capacity to fix carbon by altering diffuse/direct radiation due to SOA formation
-
Cooling of environment through production of SOA and CCN
-
Reduction of the rate of global climate change through improved carbon gain and reduced transmission of solar radiation
The ultimate significance of these services depends on the relative magnitude of different environmental changes and volatile emissions as connected through multiple feedback loops (Fig. 5). It is, furthermore, highly likely that globally changing environmental drivers and CO2 concentration alter the quantitative significance of the feedback loops due to the modifications in plant stress status, carbon fixation, and trace gas release as discussed in the next section.
3.4 Plant Stress, Volatile Emissions, and Trace-Gas-Dependent Ecosystem Services in Changing Climates
Climate change is expected to result in more severe heat stress worldwide (Kirtman et al. 2013; Field et al. 2014). In addition, climate change alters the distribution of precipitation with some areas predicted to become drier and some areas wetter (Kirtman et al. 2013; Field et al. 2014). Thus, the overall abiotic stress level is expected to increase in the future, but prediction of how enhanced stress level modifies volatile emission and ecosystem services is complicated. Increases in temperature can initially result in enhanced emissions of constitutive de novo emissions until the physiological optimum is exceeded (Loreto et al. 1998; Staudt and Bertin 1998; Rasulov et al. 2015). Beyond the physiological optimum, the rate of constitutive emissions decreases, but the stress volatile emissions are induced (Staudt and Bertin 1998; Kleist et al. 2012). These emissions under heat stress have been detected at the ecosystem scale (Karl et al. 2008), demonstrating that the stress emissions do contribute to large-scale atmospheric processes. Given that the stress emissions are induced in all plant species, more frequent heat waves in future climates can strongly enhance the overall release of volatiles. However, the quantitative information on the kinetics of elicitation of emissions under heat stress and interspecific variability of the capacity for heat-dependent release of stress volatiles is currently very limited, hindering scaling up from case studies to whole ecosystems, regions, and globe.
Global changes in environmental drivers have to be tempered in light of simultaneous modifications of ambient CO2 concentrations. Elevation of atmospheric CO2 concentration itself can improve plant carbon gain in drier climates due to reduction of the diffusion limitations on the CO2 pathway from the ambient air through stomata and mesophyll to the chloroplasts where photosynthesis takes place (Niinemets et al. 2011; Flexas et al. 2016). In addition, elevated CO2 can protect leaves from the heat stress, possibly by increasing leaf sugar concentrations that enhance the heat stability of cell and chloroplast membranes (Darbah et al. 2010; Sun et al. 2013). In the case of constitutive isoprene emissions , however, several studies have demonstrated that the capacity for isoprene emission decreases with a long-term increase in ambient CO2 concentration similarly to the response of isoprene emissions to rapid changes in CO2 concentration (Niinemets et al. 2010a; Possell and Hewitt 2011). Such an acclimatory response would mean that the elevation of CO2 concentration impairs the heat stress protection by isoprene. However, in other studies, plants grown under elevated CO2 had greater isoprene emission potential and improved heat stress resistance (Peñuelas and Staudt 2010; Sun et al. 2012, 2013). These controversial responses are evident when comparing model projections of emissions, which diverge greatly between models under future climate change (Keenan et al. 2009). This implies that prediction of constitutive emissions in future atmospheres is subject to large uncertainties and calls for more work on acclimation responses of constitutive emissions.
Apart from abiotic stresses, global environmental change is predicted to result in increasingly more frequent and severe pest and pathogen attacks (DeLucia et al. 2008; Chakraborty 2013). Such a greater biotic stress pressure can result from a reduction of constitutive defenses of vegetation due to reduced photosynthetic carbon availability under more severe abiotic stresses, shorter life cycles of current pests, and pathogens in hotter climates as well as invasion of new pests and pathogens facilitated by global trade and travel (Fig. 6, Vanhanen 2008; Huang et al. 2010; Gutierrez and Ponti 2014). Although biotic stress itself typically elicits volatile emissions for a relatively short period of time (Fig. 2), as the result of greater biotic stress pressure, the frequency of multiple sequential and simultaneous biotic attacks is likely to increase (Fig. 6). Thus, in the future climates , the emissions from biotic stresses are expected to continue longer and contribute to a larger extent to the overall emission of plant-produced volatiles.
Illustration of single and multiple biotic infections of A. incana leaves in the field. Mass infestations by the alder leaf beetle (Agelastica alni, a) and alder rust (Melampsoridium hiratsukanum, b, c) are frequently observed in European alder stands, and one might often also encounter combined infestations by both A. alni and M. hiratsukanum (d). In particular, the eastern Asian rust M. hiratsukanum that was first observed to lead to mass alder infestation in the mid-1990s in northern Europe (Põldmaa 1997; Hantula et al. 2009) has spread over much of the Europe by now, and infestations involving all alder trees in a given stand are common. Typically, the signs of infestations, orange urediniospores on the lower leaf surface, are observed in late summer and ultimately result in premature leaf senescence, extensive necrosis, and the leaf drying out with characteristic inward rolling of leaf margins (c)
The available evidence collectively suggests that global change enhances emissions induced by both abiotic and biotic stresses and might reduce constitutive emissions. Given that induced emissions occur in all plant species, vegetation trace gas emissions are expected to increase. However, due to stochastic nature of stress events, the emission kinetics of induced emissions is inherently much less predictable than the kinetics of constitutive emissions (Arneth and Niinemets 2010). Such stochasticity poses a challenge for model approaches trying to evaluate the influence of climate change on the feedback loops between constitutive and stress-induced volatile emissions, SOA , CCN , plant photosynthetic production, diffuse/direct radiation ratio, and warming and elevated CO2 (Fig. 5). Future experimental work should fill the gaps in quantitative understanding of how stress-elicited volatile emissions scale with the severity of different abiotic and biotic stresses, how stress and altered atmospheric CO2 modify constitutive emissions, and what is the biological variability in these responses. Armed with this knowledge, the community can start targeting the key research questions on the extent to which plant trace gas release can reduce the effects of climate change on vegetation and the extent to which it can reduce the rate of climate change.
4 Concluding Perspectives
Several widespread plant species are strong constitutive emitters of volatile isoprenoids such as isoprene and monoterpenes, and all plants can be induced to release volatiles upon abiotic and biotic stresses. These emissions play a variety of biological and biogeochemical roles, overall improving directly or/and indirectly the stress resistance of vegetation and altering the ambient environment at local, regional, and global scales. From a local perspective, release of volatiles can be considered both ecosystem service or ecosystem disservice depending on the human impact on atmospheric composition. In atmospheres polluted with nitrogen mono-oxides, plant volatiles contribute to ozone formation in the atmosphere, and thus, volatile release adversely affects the environment. In clean atmospheres, however, plant volatiles reduce atmospheric ozone levels and thus contribute to atmospheric cleansing. Furthermore, by enhancing vegetation stress resistance, the volatiles contribute to the maintenance of ecosystem integrity and, thus, contribute to all the well-perceived ecosystem services such as the capacity of forest ecosystems to provide wood and agroecosystems to provide agricultural products. In addition to controlling atmospheric ozone levels, plant volatiles provide a number of other key regulating services of local to global importance. In particular, plant volatiles alter atmospheric clearness due to the effects of volatiles on atmospheric particle concentrations and cloudiness. Modifications in atmospheric clearness in turn alter the ratio of diffuse to total solar radiation and atmospheric temperature with ultimate impacts on global vegetation productivity, rate of change in atmospheric CO2 concentration, and rate of global change. In future hotter more stressful environments, stress-induced volatile emissions can be particularly relevant in driving the global feedbacks between volatile production, modifications in atmospheric oxidative status, clearness, and global change. Given that the role of volatiles in global coupled vegetation-climate models is still largely unaccounted, I argue that the plant/atmosphere interface should be a high priority research target in future climate change studies.
References
Achotegui-Castells A, Danti R, Llusià J, Della Rocca G, Barberini S, Peñuelas J (2015) Strong induction of minor terpenes in Italian cypress, Cupressus sempervirens, in response to infection by the fungus Seiridium cardinale. J Chem Ecol 41:224–243
Altieri M, Nicholls C (2004) Biodiversity and pest management in agroecosystems, 2nd edn. Food Products Press, New York
Andreou A, Feussner I (2009) Lipoxygenases – structure and reaction mechanism. Phytochemistry 70:1504–1510
Arneth A, Niinemets Ü (2010) Induced BVOCs: how to bug our models? Trends Plant Sci 15:118–125
Arneth A, Monson RK, Schurgers G, Niinemets Ü, Palmer PI (2008) Why are estimates of global isoprene emissions so similar (and why is this not so for monoterpenes)? Atmos Chem Phys 8:4605–4620
Arneth A, Unger N, Kulmala M, Andreae MO (2009) Clean the air, heat the planet? Science 326:672–673
Ashworth K, Boissard C, Folberth G, Lathière J, Schurgers G (2013) Global modeling of volatile organic compound emissions. In: Niinemets Ü, Monson RK (eds) Biology, controls and models of tree volatile organic compound emissions, Tree physiology, vol 5. Springer, Berlin, pp 451–487
Atkinson R, Arey J (2003) Gas-phase tropospheric chemistry of biogenic volatile organic compounds: a review. Atmos Environ 37:197–219
Beauchamp J, Wisthaler A, Hansel A, Kleist E, Miebach M, Niinemets Ü, Schurr U, Wildt J (2005) Ozone induced emissions of biogenic VOC from tobacco: relations between ozone uptake and emission of LOX products. Plant Cell Environ 28:1334–1343
Blanch J-S, Peñuelas J, Llusià J (2007) Sensitivity of terpene emissions to drought and fertilization in terpene-storing Pinus halepensis and non-storing Quercus ilex. Physiol Plant 131:211–225
Blanch J-S, Peñuelas J, Sardans J, Llusià J (2009) Drought, warming and soil fertilization effects on leaf volatile terpene concentrations in Pinus halepensis and Quercus ilex. Acta Physiol Plant 31:207–218
Blanch J-S, Llusià J, Niinemets Ü, Noe SM, Peñuelas J (2011) Instantaneous and historical temperature effects on α-pinene emissions in Pinus halepensis and Quercus ilex. J Environ Biol 32:1–6
Blande JD, Tiiva P, Oksanen E, Holopainen JK (2007) Emission of herbivore-induced volatile terpenoids from two hybrid aspen (Populus tremula x tremuloides) clones under ambient and elevated ozone concentrations in the field. Glob Chang Biol 13:2538–2550
Blande JD, Holopainen JK, Niinemets Ü (2014) Plant volatiles in polluted atmospheres: stress responses and signal degradation. Plant Cell Environ 37:1892–1904
Bracho Nunez A, Knothe N, Liberato MAR, Schebeske G, Ciccioli P, Piedade MTF, Kesselmeier J (2009) Flooding effects on plant physiology and VOC emissions from Amazonian tree species from two different flooding environments: Varzea and Igapo. Geophys Res Abstr 11:EGU2009–EGU1497
Calfapietra C, Pallozzi E, Lusini I, Velikova V (2013) Modification of BVOC emissions by changes in atmospheric [CO2] and air pollution. In: Niinemets Ü, Monson RK (eds) Biology, controls and models of tree volatile organic compound emissions, Tree physiology, vol 5. Springer, Berlin, pp 253–284
Calogirou A, Larsen BR, Brussol C, Duane M, Kotzias D (1996) Decomposition of terpenes by ozone during sampling on Tenax. Anal Chem 68:1499–1506
Cescatti A, Niinemets Ü (2004) Sunlight capture. Leaf to landscape. In: Smith WK, Vogelmann TC, Chritchley C (eds) Photosynthetic adaptation. Chloroplast to landscape, Ecological studies, vol 178. Springer, Berlin, pp 42–85
Chakraborty S (2013) Migrate or evolve: options for plant pathogens under climate change. Glob Chang Biol 19:1985–2000
Chameides WL, Fehsenfeld F, Rodgers MO, Cardelino C, Martinez J, Parrish D, Lonneman W, Lawson DR, Rasmussen RA, Zimmerman P, Greenberg J, Middleton P, Wang T (1992) Ozone precursor relationships in the ambient atmosphere. J Geophys Res 97:6037–6055
Chen X, Hopke PK (2009) A chamber study of secondary organic aerosol formation by linalool ozonolysis. Atmos Environ 43:3935–3940
Chen F, Tholl D, Bohlmann J, Pichersky E (2011) The family of terpene synthases in plants: a mid-size family of genes for specialized metabolism that is highly diversified throughout the kingdom. Plant J 66:212–229
Christianson DW (2008) Unearthing the roots of the terpenome. Curr Opin Chem Biol 12:141–150
Cooter EJ, Rea A, Bruins R, Schwede D, Dennis R (2013) The role of the atmosphere in the provision of ecosystem services. Sci Total Environ 448:197–208
Copolovici LO, Niinemets Ü (2005) Temperature dependencies of Henry’s law constants and octanol/water partition coefficients for key plant volatile monoterpenoids. Chemosphere 61:1390–1400
Copolovici L, Niinemets Ü (2016) Environmental impacts on plant volatile emission. In: Blande J, Glinwood R (eds) Deciphering chemical language of plant communication, Signaling and communication in plants. Springer International Publishing, Berlin, pp 35–59
Copolovici LO, Filella I, Llusià J, Niinemets Ü, Peñuelas J (2005) The capacity for thermal protection of photosynthetic electron transport varies for different monoterpenes in Quercus ilex. Plant Physiol 139:485–496
Copolovici L, Kännaste A, Remmel T, Vislap V, Niinemets Ü (2011) Volatile emissions from Alnus glutinosa induced by herbivory are quantitatively related to the extent of damage. J Chem Ecol 37:18–28
Copolovici L, Kännaste A, Pazouki L, Niinemets Ü (2012) Emissions of green leaf volatiles and terpenoids from Solanum lycopersicum are quantitatively related to the severity of cold and heat shock treatments. J Plant Physiol 169:664–672
Copolovici L, Kännaste A, Remmel T, Niinemets Ü (2014a) Volatile organic compound emissions from Alnus glutinosa under interacting drought and herbivory stresses. Environ Exp Bot 100:55–63
Copolovici L, Väärtnõu F, Portillo Estrada M, Niinemets Ü (2014b) Oak powdery mildew (Erysiphe alphitoides)-induced volatile emissions scale with the degree of infection in Quercus robur. Tree Physiol 34:1399–1410
D’Alessandro M, Held M, Triponez Y, Turlings TCJ (2006) The role of indole and other shikimic acid derived maize volatiles in the attraction of two parasitic wasps. J Chem Ecol 32:2733–2748
Darbah JNT, Sharkey TD, Calfapietra C, Karnosky DF (2010) Differential response of aspen and birch trees to heat stress under elevated carbon dioxide. Environ Pollut 158:1008–1014
DeLucia EH, Casteel CL, Nabity PD, O’Neill BF (2008) Insects take a bigger bite out of plants in a warmer, higher carbon dioxide world. Proc Natl Acad Sci U S A 105:1781–1782
Dicke M, Baldwin IT (2010) The evolutionary context for herbivore-induced plant volatiles: beyond the ‘cry for help’. Trends Plant Sci 15:167–175
Dicke M, van Loon JJA, Soler R (2009) Chemical complexity of volatiles from plants induced by multiple attack. Nat Chem Biol 5:317–324
Dindorf T, Kuhn U, Ganzeveld L, Schebeske G, Ciccioli P, Holzke C, Köble R, Seufert G, Kesselmeier J (2006) Significant light and temperature dependent monoterpene emissions from European beech (Fagus sylvatica L.) and their potential impact on the European volatile organic compound budget. J Geophys Res Atmos 111:D16305
Dong L, Jongedijk E, Bouwmeester H, Van Der Krol A (2016) Monoterpene biosynthesis potential of plant subcellular compartments. New Phytol 209:679–690
Ehn M, Thornton JA, Kleist E, Sipilä M, Junninen H, Pullinen I, Springer M, Rubach F, Tillmann R, Lee B, Lopez-Hilfiker F, Andres S, Acir I-H, Rissanen M, Jokinen T, Schobesberger S, Kangasluoma J, Kontkanen J, Nieminen T, Kurtén T, Nielsen LB, Jørgensen S, Kjaergaard HG, Canagaratna M, Dal Maso M, Berndt T, Petäjä T, Wahner A, Kerminen V-M, Kulmala M, Worsnop DR, Wildt J, Mentel TF (2014) A large source of low-volatility secondary organic aerosol. Nature 506:476–479
Engelhart GJ, Asa-Awuku A, Nenes A, Pandis SN (2008) CCN activity and droplet growth kinetics of fresh and aged monoterpene secondary organic aerosol. Atmos Chem Phys 8:3937–3949
Falara V, Akhtar TA, Nguyen TTH, Spyropoulou EA, Bleeker PM, Schauvinhold I, Matsuba Y, Bonini ME, Schilmiller AL, Last RL, Schuurink RC, Pichersky E (2011) The tomato terpene synthase gene family. Plant Physiol 157:770–789
Fall R (2003) Abundant oxygenates in the atmosphere: a biochemical perspective. Chem Rev 103:4941–4952
Fall R, Benson AA (1996) Leaf methanol – the simplest natural product from plants. Trends Plant Sci 1:296–301
Farquhar GD, Roderick ML (2003) Pinatubo, diffuse light, and the carbon cycle. Science 299:1997–1998
Farré-Armengol G, Filella I, Llusià J, Primante C, Peñuelas J (2015) Enhanced emissions of floral volatiles by Diplotaxis erucoides (L.) in response to folivory and florivory by Pieris brassicae (L.) Biochem Syst Ecol 63:51–58
Fehsenfeld F, Calvert J, Fall R, Goldan P, Guenther AB, Hewitt CN, Lamb B, Liu S, Trainer M, Westberg H, Zimmerman P (1992) Emissions of volatile organic compounds from vegetation and the implications for atmospheric chemistry. Glob Biogeochem Cycles 6:389–430
Field CB, Barros VR, Dokken DJ, Mach KJ, Mastrandrea MD, Bilir TE, Chatterjee M, Ebi KL, Estrada YO, Genova RC, Girma B (eds) (2014) Climate change 2014: impacts, adaptation, and vulnerability. Part A: global and sectoral aspects. Contribution of Working Group II to the fifth assessment report of the Intergovernmental Panel on Climate Change. Cambridge University Press, Cambridge, UK/New York, USA
Fineschi S, Loreto F, Staudt M, Peñuelas J (2013) Diversification of volatile isoprenoid emissions from trees: evolutionary and ecological perspectives. In: Niinemets Ü, Monson RK (eds) Biology, controls and models of tree volatile organic compound emissions, Tree physiology, vol 5. Springer, Berlin, pp 1–20
Flexas J, Díaz-Espejo A, Conesa MA, Coopman R, Douthe C, Gago J, Gallé A, Galmés J, Medrano H, Ribas-Carbo M, Tomàs M, Niinemets Ü (2016) Mesophyll conductance to CO2 and Rubisco as targets for improving intrinsic water use efficiency in C3 plants. Plant Cell Environ 39:965–982
Fuentes JD, Lerdau M, Atkinson R, Baldocchi D, Bottenheim JW, Ciccioli P, Lamb B, Geron C, Gu L, Guenther A, Sharkey TD, Stockwell W (2000) Biogenic hydrocarbons in the atmospheric boundary layer: a review. Bull Am Meteorol Soc 81:1537–1575
Gray DW, Goldstein AH, Lerdau M (2006) Thermal history regulates methylbutenol basal emission rate in Pinus ponderosa. Plant Cell Environ 29:1298–1308
Gray DW, Breneman SR, Topper LA, Sharkey TD (2011) Biochemical characterization and homology modeling of methylbutenol synthase and implications for understanding hemiterpene synthase evolution in plants. J Biol Chem 286:20582–20590
Grote R, Monson RK, Niinemets Ü (2013) Leaf-level models of constitutive and stress-driven volatile organic compound emissions. In: Niinemets Ü, Monson RK (eds) Biology, controls and models of tree volatile organic compound emissions, Tree physiology, vol 5. Springer, Berlin, pp 315–355
Gu L, Baldocchi D, Verma SB, Black TA, Vesala T, Falge EM, Dowty PR (2002) Advantages of diffuse radiation for terrestrial ecosystem productivity. J Geophys Res 107. https://doi.org/10.1029/2001JD001242
Gu L, Baldocchi DD, Wofsy SC, Munger JW, Michalsky JJ, Urbanski SP, Boden TA (2003) Response of a deciduous forest to the Mount Pinatubo eruption: enhanced photosynthesis. Science 299:2035–2038
Guenther A (2013a) Biological and chemical diversity of biogenic volatile organic emissions into the atmosphere. ISRN Atmos Sci 2013:786290
Guenther A (2013b) Upscaling biogenic volatile compound emissions from leaves to landscapes. In: Niinemets Ü, Monson RK (eds) Biology, controls and models of tree volatile organic compound emissions, Tree physiology, vol 5. Springer, Berlin, pp 391–414
Guenther AB, Jiang X, Heald CL, Sakulyanontvittaya T, Duhl T, Emmons LK, Wang X (2012) The model of emissions of gases and aerosols from nature version 2.1 (MEGAN2.1): an extended and updated framework for modeling biogenic emissions. Geosci Model Dev 5:1471–1492
Gutierrez AP, Ponti L (2014) Analysis of invasive insects: links to climate change. In: Ziska LH, Dukes JS (eds) Invasive species and global climate change. CABI Publishing, Wallingford, pp 45–61
Hantula J, Kurkela T, Hendry S, Yamaguchi T (2009) Morphological measurements and ITS sequences show that the new alder rust in Europe is conspecific with Melampsoridium hiratsukanum in eastern Asia. Mycologia 101:622–631
Harley P, Greenberg J, Niinemets Ü, Guenther A (2007) Environmental controls over methanol emission from leaves. Biogeosciences 4:1083–1099
Hartikainen K, Nerg A-M, Kivimäenpää M, Kontunen-Sopplea S, Mäenpää M, Oksanen E, Rousi M, Holopainen T (2009) Emissions of volatile organic compounds and leaf structural characteristics of European aspen (Populus tremula) grown under elevated ozone and temperature. Tree Physiol 29:1163–1173
Helmig D, Revermann T, Pollmann J, Kaltschmidt O, Hernandez AJ, Bocquet F, David D (2003) Calibration system and analytical considerations for quantitative sesquiterpene measurements in air. J Chromatogr A 1002:193–211
Himanen SJ, Blande JD, Klemola T, Pulkkinen J, Heijari J, Holopainen JK (2010) Birch (Betula spp.) leaves adsorb and re-release volatiles specific to neighbouring plants – a mechanism for associational herbivore resistance? New Phytol 186:722–732
Holopainen JK, Nerg A-M, Blande JD (2013) Multitrophic signalling in polluted atmospheres. In: Niinemets Ü, Monson RK (eds) Biology, controls and models of tree volatile organic compound emissions, Tree physiology, vol 5. Springer, Berlin, pp 285–314
Huang S-H, Cheng C-H, Wu W-J (2010) Possible impacts of climate change on rice insect pests and management tactics in Taiwan. Crop Environ Bioinform 7:269–279
Huff Hartz KE, Rosenørn T, Ferchak SR, Raymond TM, Bilde M, Donahue NM, Pandis SN (2005) Cloud condensation nuclei activation of monoterpene and sesquiterpene secondary organic aerosol. J Geophys Res Atmos 110:D14208, https://doi.org/10.11029/12004JD005754
Hüve K, Christ MM, Kleist E, Uerlings R, Niinemets Ü, Walter A, Wildt J (2007) Simultaneous growth and emission measurements demonstrate an interactive control of methanol release by leaf expansion and stomata. J Exp Bot 58:1783–1793
Jacob DJ, Field BD, Li Q, Blake DR, de Gouw J, Warneke C, Hansel A, Wisthaler A, Singh HB, Guenther A (2005) Global budget of methanol: constraints from atmospheric observations. J Geophys Res Atmos 110:D08303, https://doi.org/10.01029/02004JD005172
Jiang Y, Ye J, Li S, Niinemets Ü (2016a) Regulation of floral terpenoid emission and biosynthesis in sweet basil (Ocimum basilicum). J Plant Growth Regul 35:921–935
Jiang Y, Ye J, Veromann L-L, Niinemets Ü (2016b) Scaling of photosynthesis and constitutive and induced volatile emissions with severity of leaf infection by rust fungus (Melampsora larici-populina) in Populus balsamifera var. suaveolens. Tree Physiol 38:856–872
Karl T, Guenther A, Turnipseed A, Patton EG, Jardine K (2008) Chemical sensing of plant stress at the ecosystem scale. Biogeosciences 5:1287–1294
Karlik JF, Winer AM (2001) Measured isoprene emission rates of plants in California landscapes: comparison to estimates from taxonomic relationships. Atmos Environ 35:1123–1131
Kask K, Kännaste A, Talts E, Copolovici L, Niinemets Ü (2016) How specialized volatiles respond to chronic and short-term physiological and shock heat stress in Brassica nigra. Plant Cell Environ 39:2027–2042
Keenan T, Niinemets Ü, Sabate S, Gracia C, Peñuelas J (2009) Process based inventory of isoprenoid emissions from European forests: model comparisons, current knowledge and uncertainties. Atmos Chem Phys 9:4053–4076
Kirkby J, Duplissy J, Sengupta K, Frege C, Gordon H, Williamson C, Heinritzi M, Simon M, Yan C, Almeida J, Tröstl J, Nieminen T, Ortega IK, Wagner R, Adamov A, Amorim A, Bernhammer A-K, Bianchi F, Breitenlechner M, Brilke S, Chen X, Craven J, Dias A, Ehrhart S, Flagan RC, Franchin A, Fuchs C, Guida R, Hakala J, Hoyle CR, Jokinen T, Junninen H, Kangasluoma J, Kim J, Krapf M, Kürten A, Laaksonen A, Lehtipalo K, Makhmutov V, Mathot S, Molteni U, Onnela A, Peräkylä O, Piel F, Petäjä T, Praplan AP, Pringle K, Rap A, Richards NAD, Riipinen I, Rissanen MP, Rondo L, Sarnela N, Schobesberger S, Scott CE, Seinfeld JH, Sipilä M, Steiner G, Stozhkov Y, Stratmann F, Tomé A, Virtanen A, Vogel AL, Wagner AC, Wagner PE, Weingartner E, Wimmer D, Winkler PM, Ye P, Zhang X, Hansel A, Dommen J, Donahue NM, Worsnop DR, Baltensperger U, Kulmala M, Carslaw KS, Curtius J (2016) Ion-induced nucleation of pure biogenic particles. Nature 533:521–526
Kirtman B, Power SB, Adedoyin JA, Boer GJ, Bojariu R, Camilloni I, Doblas-Reyes FJ, Fiore AM, Kimoto M, Meehl GA, Prather M, Sarr A, Schär C, Sutton R, van Oldenborgh GJ, Vecchi G, Wang HJ (2013) Near-term climate change: projections and predictability. In: Stocker TF et al (eds) Climate change 2013: the physical science basis. Contribution of Working Group I to the fifth assessment report of the Intergovernmental Panel on Climate Change. Cambridge University Press, Cambridge, UK/New York, USA, pp 953–1028
Kleist E, Mentel TF, Andres S, Bohne A, Folkers A, Kiendler-Scharr A, Rudich Y, Springer M, Tillmann R, Wildt J (2012) Irreversible impacts of heat on the emissions of monoterpenes, sesquiterpenes, phenolic BVOC and green leaf volatiles from several tree species. Biogeosciences 9:5111–5123
Kosina J, Dewulf J, Viden I, Pokorska O, Van Langenhove H (2013) Dynamic capillary diffusion system for monoterpene and sesquiterpene calibration: quantitative measurement and determination of physical properties. Int J Environ Anal Chem 93:637–649
Kreuzwieser J, Rennenberg H (2013) Flooding-driven emissions from trees. In: Niinemets Ü, Monson RK (eds) Biology, controls and models of tree volatile organic compound emissions, Tree physiology, vol 5. Springer, Berlin, pp 237–252
Kreuzwieser J, Kühnemann F, Martis A, Rennenberg H, Urban W (2000) Diurnal pattern of acetaldehyde emission by flooded poplar trees. Physiol Plant 108:79–86
Kreuzwieser J, Harren FJM, Laarhoven LJJ, Boamfa I, te Lintel HS, Scheerer U, Huglin C, Rennenberg H (2001) Acetaldehyde emission by the leaves of trees – correlation with physiological and environmental parameters. Physiol Plant 113:41–49
Kulmala M, Laakso L, Lehtinen KEJ, Riipinen I, Dal Maso M, Anttila T, Kerminen V-M, Hõrrak U, Vana M, Tammet H (2004a) Initial steps of aerosol growth. Atmos Chem Phys 4:2553–2560
Kulmala M, Suni T, Lehtinen KEJ, Dal Maso M, Boy M, Reissell A, Rannik Ü, Aaalto P, Keronen P, Hakola H, Bäck J, Hoffmann T, Vesala T, Hari P (2004b) A new feedback mechanism linking forests, aerosols, and climate. Atmos Chem Phys 4:557–562
Kulmala M, Nieminen T, Chellapermal R, Makkonen R, Bäck J, Kerminen V-M (2013) Climate feedbacks linking the increasing atmospheric CO2 concentration, BVOC emissions, aerosols and clouds in forest ecosystems. In: Niinemets Ü, Monson RK (eds) Biology, controls and models of tree volatile organic compound emissions, Tree physiology, vol 5. Springer, Berlin, pp 489–508
Lerdau M (2007) A positive feedback with negative consequences. Science 316:212–213
Lerdau M, Slobodkin K (2002) Trace gas emissions and species-dependent ecosystem services. Trends Ecol Evol 17:309–312
Li Z, Sharkey TD (2013) Molecular and pathway controls on biogenic volatile organic compound emissions. In: Niinemets Ü, Monson RK (eds) Biology, controls and models of tree volatile organic compound emissions, Tree physiology, vol 5. Springer, Berlin, pp 119–151
Liavonchanka A, Feussner N (2006) Lipoxygenases: occurrence, functions and catalysis. J Plant Physiol 163:348–357
Llusià J, Peñuelas J, Sardans J, Owen SM, Niinemets Ü (2010) Measurement of volatile terpene emissions in 70 dominant vascular plant species in Hawaii: aliens emit more than natives. Glob Ecol Biogeogr 19:863–874
Llusià J, Sardans J, Niinemets Ü, Owen SM, Peñuelas J (2014) A screening study of leaf terpene emissions of 43 rainforest species in Danum Valley Conservation Area (Borneo) and their relationships with chemical and morphological leaf traits. Plant Biosyst 148:307–317
Loreto F, Fares S (2007) Is ozone flux inside leaves only a damage indicator? Clues from volatile isoprenoid studies. Plant Physiol 143:1096–1100
Loreto F, Förster A, Dürr M, Csiky O, Seufert G (1998) On the monoterpene emission under heat stress and on the increased thermotolerance of leaves of Quercus ilex L. fumigated with selected monoterpenes. Plant Cell Environ 21:101–107
Loreto F, Nascetti P, Graverini A, Mannozzi M (2000) Emission and content of monoterpenes in intact and wounded needles of the Mediterranean pine, Pinus pinea. Funct Ecol 14:589–595
Loreto F, Pinelli P, Manes F, Kollist H (2004) Impact of ozone on monoterpene emissions and evidence for an isoprene-like antioxidant action of monoterpenes emitted by leaves. Tree Physiol 24:361–367
Malm WC, Gebhart KA, Molenar J, Cahill T, Eldred R, Huffman D (1994) Examining the relationship between atmospheric aerosols and light extinction at Mount Rainier and North Cascades National Parks. Atmos Environ 28:347–360
Mentel TF, Wildt J, Kiendler-Scharr A, Kleist E, Tillmann R, Dal Maso M, Fisseha R, Hohaus T, Spahn H, Uerlings R, Wegener R, Griffiths PT, Dinar E, Rudich Y, Wahner A (2009) Photochemical production of aerosols from real plant emissions. Atmos Chem Phys 9:4387–4406
Mercado LM, Bellouin N, Sitch S, Boucher O, Huntingford C, Wild M, Cox PM (2009) Impact of changes in diffuse radiation on the global land carbon sink. Nature 458:1014–1017
Micheli F (2001) Pectin methylesterases: cell wall enzymes with important roles in plant physiology. Trends Plant Sci 6:414–419
Misson L, Lunden M, McKay M, Goldstein AH (2005) Atmospheric aerosol light scattering and surface wetness influence the diurnal pattern of net ecosystem exchange in a semi-arid ponderosa pine plantation. Agric For Meteorol 129:69–83
Monson RK, Grote R, Niinemets Ü, Schnitzler J-P (2012) Tansley review. Modeling the isoprene emission rate from leaves. New Phytol 195:541–559
Nemecek-Marshall M, MacDonald RC, Franzen JJ, Wojciechowski CL, Fall R (1995) Methanol emission from leaves. Enzymatic detection of gas-phase methanol and relation of methanol fluxes to stomatal conductance and leaf development. Plant Physiol 108:1359–1368
Niinemets Ü, Tenhunen JD, Harley PC, Steinbrecher R (1999) A model of isoprene emission based on energetic requirements for isoprene synthesis and leaf photosynthetic properties for Liquidambar and Quercus. Plant Cell Environ 22:1319–1336
Niinemets Ü, Hauff K, Bertin N, Tenhunen JD, Steinbrecher R, Seufert G (2002a) Monoterpene emissions in relation to foliar photosynthetic and structural variables in Mediterranean evergreen Quercus species. New Phytol 153:243–256
Niinemets Ü, Reichstein M, Staudt M, Seufert G, Tenhunen JD (2002b) Stomatal constraints may affect emission of oxygenated monoterpenoids from the foliage of Pinus pinea. Plant Physiol 130:1371–1385
Niinemets Ü, Loreto F, Reichstein M (2004) Physiological and physicochemical controls on foliar volatile organic compound emissions. Trends Plant Sci 9:180–186
Niinemets Ü, Arneth A, Kuhn U, Monson RK, Peñuelas J, Staudt M (2010a) The emission factor of volatile isoprenoids: stress, acclimation, and developmental responses. Biogeosciences 7:2203–2223
Niinemets Ü, Monson RK, Arneth A, Ciccioli P, Kesselmeier J, Kuhn U, Noe SM, Peñuelas J, Staudt M (2010b) The leaf-level emission factor of volatile isoprenoids: caveats, model algorithms, response shapes and scaling. Biogeosciences 7:1809–1832
Niinemets Ü, Flexas J, Peñuelas J (2011) Evergreens favored by higher responsiveness to increased CO2. Trends Ecol Evol 26:136–142
Niinemets Ü, Kännaste A, Copolovici L (2013) Quantitative patterns between plant volatile emissions induced by biotic stresses and the degree of damage. Front Plant Sci Front Plant-Microbe Interact 4:262
Niinemets Ü, Sun Z, Talts E (2015) Controls of the quantum yield and saturation light of isoprene emission in different-aged aspen leaves. Plant Cell Environ 38:2707–2720
Noe SM, Copolovici L, Niinemets Ü, Vaino E (2008) Foliar limonene uptake scales positively with leaf lipid content: “non-emitting” species absorb and release monoterpenes. Plant Biol 10:129–137
Nölscher AC, Williams J, Sinha V, Custer T, Song W, Johnson AM, Axinte R, Bozem H, Fischer H, Pouvesle N, Phillips G, Crowley JN, Rantala P, Rinne J, Kulmala M, Gonzales D, Valverde-Canossa J, Vogel A, Hoffmann T, Ouwersloot HG, Vilà-Guerau de Arellano J, Lelieveld J (2012) Summertime total OH reactivity measurements from boreal forest during HUMPPA-COPEC 2010. Atmos Chem Phys 12:8257–8270
Pazouki L, Niinemets Ü (2016) Multi-substrate terpenoid synthases: their occurrence and physiological significance. Front Plant Sci 7:1019
Pazouki L, Kanagendran A, Li S, Kännaste A, Rajabi Memari H, Bichele R, Niinemets Ü (2016) Mono- and sesquiterpene release from tomato (Solanum lycopersicum) leaves upon mild and severe heat stress and through recovery: from gene expression to emission responses. Environ Exp Bot 132:1–15
Peng J, van Loon JJ, Zheng S, Dicke M (2011) Herbivore-induced volatiles of cabbage (Brassica oleracea) prime defence responses in neighbouring intact plants. Plant Biol 13:276–284
Peñuelas J, Llusià J (2004) Plant VOC emissions: making use of the unavoidable. Trends Ecol Evol 19:402–404
Peñuelas J, Staudt M (2010) BVOCs and global change. Trends Plant Sci 15:133–144
Põldmaa K (1997) Explosion of Melampsoridium sp. on Alnus incana. Folia Cryptogamica Estonica 31:48–51
Portillo-Estrada M, Kazantsev T, Talts E, Tosens T, Niinemets Ü (2015) Emission timetable and quantitative patterns of wound-induced volatiles across different damage treatments in aspen (Populus tremula). J Chem Ecol 41:1105–1117
Possell M, Hewitt CN (2011) Isoprene emissions from plants are mediated by atmospheric CO2 concentrations. Glob Chang Biol 17:1595–1610
Possell M, Loreto F (2013) The role of volatile organic compounds in plant resistance to abiotic stresses: responses and mechanisms. In: Niinemets Ü, Monson RK (eds) Biology, controls and models of tree volatile organic compound emissions, Tree physiology, vol 5. Springer, Berlin, pp 209–235
Rajabi Memari H, Pazouki L, Niinemets Ü (2013) The biochemistry and molecular biology of volatile messengers in trees. In: Niinemets Ü, Monson RK (eds) Biology, controls and models of tree volatile organic compound emissions, Tree physiology, vol 5. Springer, Berlin, pp 47–93
Rasulov B, Hüve K, Bichele I, Laisk A, Niinemets Ü (2010) Temperature response of isoprene emission in vivo reflects a combined effect of substrate limitations and isoprene synthase activity: a kinetic analysis. Plant Physiol 154:1558–1570
Rasulov B, Bichele I, Hüve K, Vislap V, Niinemets Ü (2015) Acclimation of isoprene emission and photosynthesis to growth temperature in hybrid aspen: resolving structural and physiological controls. Plant Cell Environ 38:751–766
Roderick ML, Farquhar GD, Berry SL, Noble IR (2001) On the direct effect of clouds and atmospheric particles on the productivity and structure of vegetation. Oecologia 129:21–30
Rosenkranz M, Schnitzler J-P (2013) Genetic engineering of BVOC emissions from trees. In: Niinemets Ü, Monson RK (eds) Biology, controls and models of tree volatile organic compound emissions, Tree physiology, vol 5. Springer, Berlin, pp 95–118
Rottenberger S, Kleiss B, Kuhn U, Wolf A, Piedade MTF, Junk W, Kesselmeier J (2008) The effect of flooding on the exchange of the volatile C2-compounds ethanol, acetaldehyde and acetic acid between leaves of Amazonian floodplain tree species and the atmosphere. Biogeosciences 5:1085–1100
Russo A, Escobedo FJ, Zerbe S (2016) Quantifying the local-scale ecosystem services provided by urban treed streetscapes in Bolzano, Italy. AIMS Environ Sci 3:58–76
Sharkey TD, Chen XY, Yeh S (2001) Isoprene increases thermotolerance of fosmidomycin-fed leaves. Plant Physiol 125:2001–2006
Shindell D, Faluvegi G, Lacis A, Hansen J, Ruedy R, Aguilar E (2006) Role of tropospheric ozone increases in 20th-century climate change. J Geophys Res Atmos 111:D08302, 083. https://doi.org/10.01029/02005JD006348
Simon V, Dumergues L, Ponche J-L, Torres L (2006) The biogenic volatile organic compounds emission inventory in France. Application to plant ecosystems in the Berre-Marseilles area (France). Sci Total Environ 372:164–182
Sinha V, Williams J, Lelieveld J, Ruuskanen TM, Kajos MK, Patokoski J, Hellen H, Hakola H, Mogensen D, Boy M, Rinne J, Kulmala M (2010) OH reactivity measurements within a boreal forest: evidence for unknown reactive emissions. Environ Sci Technol 44:6614–6620
Sitch S, Cox PM, Collins WJ, Huntingford C (2007) Indirect radiative forcing of climate change through ozone effects on the land-carbon sink. Nature 448:791–794
Spracklen DV, Bonn B, Carslaw KS (2008) Boreal forests, aerosols and the impacts on clouds and climate. Philos Trans R Soc Lond A 366:4613–4626
Staudt M, Bertin N (1998) Light and temperature dependence of the emission of cyclic and acyclic monoterpenes from holm oak (Quercus ilex L.) leaves. Plant Cell Environ 21:385–395
Staudt M, Lhoutellier L (2011) Monoterpene and sesquiterpene emissions from Quercus coccifera exhibit interacting responses to light and temperature. Biogeosciences 8:2757–2771
Staudt M, Mir C, Joffre R, Rambal S, Bonin A, Landais D, Lumaret R (2004) Isoprenoid emissions of Quercus spp. (Q. suber and Q. ilex) in mixed stands contrasting in interspecific genetic introgression. New Phytol 163:573–584
Stavrakou T, Guenther A, Razavi A, Clarisse L, Clerbaux C, Coheur P-F, Hurtmans D, Karagulian F, De Maziére M, Vigouroux C, Amelynck C, Schoon N, Laffineur Q, Heinesch B, Aubinet M, Rinsland C, Müller J-F (2011) First space-based derivation of the global atmospheric methanol emission fluxes. Atmos Chem Phys 11:4873–4898
Still CJ, Riley WJ, Biraud SC, Noone DC, Buenning NH, Randerson JT, Torn MS, Welker JM, White JWC, Vachon R, Farquhar GD, Berry JA (2009) Influence of clouds and diffuse radiation on ecosystem-atmosphere CO2 and CO18O exchanges. J Geophys Res Biogeosci 114:G01018
Stocker TF, Qin D, Plattner G-K, Tignor M, Allen SK, Boschung J, Nauels A, Xia Y, Bex V, Midgley PM (eds) (2014) Climate change 2014: the physical science basis. Contribution of Working Group I to the fifth assessment report of the Intergovernmental Panel on Climate Change. Cambridge University Press, Cambridge, UK/New York, USA
Sun Z, Niinemets Ü, Hüve K, Noe SM, Rasulov B, Copolovici L, Vislap V (2012) Enhanced isoprene emission capacity and altered light responsiveness in aspen grown under elevated atmospheric CO2 concentration. Glob Chang Biol 18:3423–3440
Sun Z, Hüve K, Vislap V, Niinemets Ü (2013) Elevated [CO2] magnifies isoprene emissions under heat and improves thermal resistance in hybrid aspen. J Exp Bot 64:5509–5523
Tholl D, Lee S (2011) Terpene specialized metabolism in Arabidopsis thaliana. Arabidopsis Book 9:e0143
Tholl D, Sohrabi R, Huh J-H, Lee S (2011) The biochemistry of homoterpenes – common constituents of floral and herbivore-induced plant volatile bouquets. Phytochemistry 72:1635–1646
Tooker JF, Frank SD (2012) Genotypically diverse cultivar mixtures for insect pest management and increased crop yields. J Appl Ecol 49:974–985
Vanhanen H (2008) Invasive insects in Europe – the role of climate change and global trade. Dissertationes Forestales 57. Faculty of Forest Sciences, University of Joensuu, 33 pages
Velikova V, Sharkey TD, Loreto F (2012) Stabilization of thylakoid membranes in isoprene-emitting plants reduces formation of reactive oxygen species. Plant Signal Behav 7:139–141
Vickers CE, Gershenzon J, Lerdau MT, Loreto F (2009) A unified mechanism of action for volatile isoprenoids in plant abiotic stress. Nat Chem Biol 5:283–291
von Dahl C, Hävecker M, Schlögl R, Baldwin IT (2006) Caterpillar-elicited methanol emission: a new signal in plant-herbivore interaction? Plant J 46:948–960
Voulgarakis A, Naik V, Lamarque J-F, Shindell DT, Young PJ, Prather MJ, Wild O, Field RD, Bergmann D, Cameron-Smith P, Cionni I, Collins WJ, Dalsøren SB, Doherty RM, Eyring V, Faluvegi G, Folberth GA, Horowitz LW, Josse B, MacKenzie IA, Nagashima T, Plummer DA, Righi M, Rumbold ST, Stevenson DS, Strode SA, Sudo K, Szopa S, Zeng G (2013) Analysis of present day and future OH and methane lifetime in the ACCMIP simulations. Atmos Chem Phys 13:2563–2587
Widegren JA, Bruno TJ (2010) Vapor pressure measurements on low-volatility terpenoid compounds by the concatenated gas saturation method. Environ Sci Technol 44:388–393
Wilkinson MJ, Monson RK, Trahan N, Lee S, Brown E, Jackson RB, Polley HW, Fay PA, Fall R (2009) Leaf isoprene emission rate as a function of atmospheric CO2 concentration. Glob Chang Biol 15:1189–1200
Winters AJ, Adams MA, Bleby TM, Rennenberg H, Steigner D, Steinbrecher R, Kreuzwieser J (2009) Emissions of isoprene, monoterpene and short-chained carbonyl compounds from Eucalyptus spp. in southern Australia. Atmos Environ 43:3035–3043
Zhang P-J, Zheng S-J, van Loona JJA, Boland W, David A, Mumm R, Dicke M (2009) Whiteflies interfere with indirect plant defense against spider mites in lima bean. Proc Natl Acad Sci U S A 106:21202–21207
Acknowledgments
I thank Prof. Josep Peñuelas (Global Ecology Unit CREAF-CSIC-UAB, Barcelona, Catalonia) and Dr. Trevor F. Keenan (Earth and Environmental Sciences, Lawrence Berkeley National Lab, USA) for insightful comments on the MS. My work on plant volatiles has been supported by the Estonian Ministry of Science and Education (institutional grant IUT-8-3) and the European Commission through the European Research Council (advanced grant 322603, SIP-VOL+) and the European Regional Development Fund (Centre of Excellence EcolChange, TK 131).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer International Publishing AG, part of Springer Nature
About this chapter
Cite this chapter
Niinemets, Ü. (2018). What Are Plant-Released Biogenic Volatiles and How They Participate in Landscape- to Global-Level Processes?. In: Perera, A., Peterson, U., Pastur, G., Iverson, L. (eds) Ecosystem Services from Forest Landscapes. Springer, Cham. https://doi.org/10.1007/978-3-319-74515-2_3
Download citation
DOI: https://doi.org/10.1007/978-3-319-74515-2_3
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-74514-5
Online ISBN: 978-3-319-74515-2
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)