Abstract
Structure and functions of Na+/K+-ATPase and SERCA are described with details on their subunits, isoforms, and intracellular localization. Main regulatory mechanisms are summarized. Molecular mechanisms of cell death and heart failure are explained with the analysis of the role of Na+/K+-ATPase and SERCA in these processes. Facts are considered from a cytological, pathological, and clinical perspective with an accent to new therapeutic strategies. The aim of this contribution is an overview of functional results in a structural context.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Na+/K+-ATPase
- SERCA
- Cardiomyocytes
- Apoptosis
- Necrosis
- Cell injury
- Ischemia
- Hypertrophy
- Heart failure
- Cardiomyopathy
1 Introduction
Various forms of ATPase have the utmost significance in the understanding of major principles of cardiovascular physiology and molecular mechanisms of cardiovascular diseases. There is ample scope of literature dealing with this problem with new results obtained. This fact promises that our knowledge will be amplified for the benefit of our patients. However, plenty of new results always open new dilemmas and controversies, suggesting that we should recapitulate what we know for a fact and what we have recently learned. Our intention in this chapter is to summarize molecular, cytological, pathological, and clinical aspects of Na+/K+-ATPase and SERCA functions. We focus on their role in cardiomyocyte cell death and heart failure. We start with the principles that we have already known and provide new information from a cytological and pathological point of view.
2 Na+/K+-ATPase: Structure and Function
Danish researcher, Jeans C. Skou was the first to suggest a link between transport of Na+ and K+ across the plasma membrane and ATPase activity in 1950s. For the discovery of the Na+/K+-ATPase, Jeans C. Skou was awarded by the Nobel Prize in Chemistry 1997.
2.1 Structure of Na+/K+-ATPase
Na+/K+-ATPase is a membrane-embedded protein complex, a hetero-oligomer composed of α and β subunits, in a 1:1 ratio [1, 2]. The large catalytic α-subunit (~110 kDa) contains binding sites for Na+ and K+ ions, ATP and for cardiac glycoside ouabain [1, 2]. Catalytic subunit has conserved aspartate369 residue where terminal phosphate of ATP can be attached [3, 4]. The α-subunit has ten transmembrane domains and two large intracellular loops. The ATP binding site is located in the larger cytoplasmic loop [2]. Both amino and carboxyl ends of this molecule are located intracellularly [4].
The smaller and highly glycosylated β-subunit (~35–55 kDa) acts as a chaperone required for proper folding and localization of Na+/K+-ATPase subunits to the plasma membrane [1, 2, 5]. Beta subunit is composed from one transmembrane segment, short cytoplasmic tail and large glycosylated extracellular segment [6, 7]. In vitro studies suggest that separation of α and β subunits results in a lack of measurable enzyme activity [8].
2.1.1 Isoforms of α Subunit
Four isoforms of α-subunit have been described (α1, α2, α3, and α4) [1, 9, 10]. While the α1-isoform is expressed ubiquitously and is a housekeeping form, α2 is expressed largely in the brain, muscle, adult heart, and number of other tissues [2, 11]. The α3-isoform is found in ovaries, neurons, fetal and adult hearts, and white blood cells [1, 11, 12]. The α4-isoform is localized in the testis, and this isoform is specifically synthesized at the spermatogonia stage. The main role of α4-isoform is in the sperm motility [13]. Various α isoforms are tissue-specific, and they share a high degree of sequence identity (ca. ~85 % identity) [1, 12, 14]. Different α subunit isoforms could be localized in different regions of the same cell [15] and are capable of carrying out specific functions.
2.1.2 Isoforms of β Subunit
There are three isoforms of the β-subunit (β1, β2, and β3) [1, 2]. Beta1 isoform is, like α1, ubiquitously expressed suggesting a housekeeping role for the α1-β1 Na+/K+-ATPase in most cells [16]. Beta 2 isoform is expressed mainly in the brain and muscle [17], while the β3 isoform is predominantly expressed in the skeletal muscle, brain, lung, testis, and liver [16, 18]. In human heart, α1, α2, and α3 are expressed together with β1 and very low levels of β2 in a region-specific manner [19, 20]. Alpha and β subunit isoforms of Na+/K+-ATPase have been encoded by different genes. They are synthesized independently of each other in the endoplasmic reticulum and assembly very soon after the synthesis [21–24]. Isoforms combine to form a number of Na+/K+-ATPase isoenzymes expressed in a tissue- and cell-specific manner [24].
2.1.3 The Role of FXYD Proteins
FXYD proteins (also referred to as γ-subunits) have functions in stabilization or attenuation of Na+/K+-ATPase [25] and form an auxiliary subunit of Na+/K+-ATPase. The FXYD proteins are a family of seven small regulatory proteins (FXYD1–7). They are transmembrane proteins and have conserved FXYD amino acid motif located in their extracellular domain [25, 26]. N-terminal part of FXYD is extracellular, while C-terminus is cytoplasmic [25]. FXYD proteins are expressed in a tissue specific fashion. FXYD1 (phospholemman) is expressed in the heart and the skeletal muscle [27–29].
2.2 Functions of Na+/K+-ATPase
Na+/K+-ATPase is universally expressed in all animal cells. The primary function of the Na+/K+-ATPase is generation and maintenance of electrochemical Na+ and K+ gradients across the cell membrane. The Na+/K+-ATPase is responsible for the low intracellular Na+ and high intracellular K+ concentrations required for normal cellular functions. Activity of this enzyme occurs in several steps and is dependent on ATP hydrolysis [1, 2]. Following binding of ATP to the enzyme, three Na+ ions from the cytoplasm associate with the active site of Na+/K+-ATPase. Phosphorylation of the Na+/K+-ATPase (at aspartate residue) results in its conformational change. As a consequence of this change, three bound Na+ ions are released out of the cell. Thereafter, two extracellular K+ ions bind along with dephosphorylation process and are transported into the cell [1, 2, 30].
The Na+/K+-ATPase is the specific target for the action of ouabain, digitalis and endogenous cardiac glycosides. They regulate cardiac contractility by indirect way (positive inotropy) [31, 32]. By binding to the extracellular part of Na+/K+-ATPase, cardiac glycosides inhibit its activity [2, 32] and increase Na+ concentration. These actions precede increased intracellular Ca2+ concentration [31, 32] which, on the other hand, enhances heart contraction [31, 33]. This mechanism is the basis for the usage of cardiac glycosides (digoxin) in the therapy of congestive heart failure. Number of Na+/K+-ATPase molecules decreases in heart failure, but also in some other disorders as for example, obesity [34, 35]. Cardiomyocytes of patients with heart failure are more sensitive to effects of cardiac glycosides [34, 36–39]. Rathore et al. [40] reported that higher serum digoxin concentrations are associated with increased mortality in patients diagnosed with heart failure. That is why, as explained in further discussion, cardiac glycosides are known for their narrow therapeutic window.
2.3 Regulation of Na+/K+-ATPase Function in Physiological and Pathophysiological Conditions
Na+/K+-ATPase activity can be regulated by hormones and environmental factors through: gene expression, trafficking of the newly synthesized enzyme subunits, and phosphorylation. The first mechanism affects de novo Na+/K+-ATPase synthesis or degradation through regulation of gene transcription [1, 41–45]. Second critically important step in regulatory process is the synthesis of subunits of Na+/K+-ATPase and their translocation to the plasma membrane from intracellular stores [22, 46, 47].
Regulation of Na+/K+-ATPase activity can be achieved as well through direct effects on the kinetic behavior of the enzyme located in the membrane. This is a short-term regulation, accomplished within minutes to hours. It is realized via protein kinase A (PKA), protein kinase C (PKC), protein kinase B (PKB), or cGMP-dependent protein kinase (PKG) phosphorylation [1, 39, 48–50]. Activation of PKA and PKC leads to modulation of Na+/K+-ATPase activity in a tissue- and species-specific manner. PKA of the cardiomyocytes is one of the principle molecules involved in sympathetic innervations. After β adrenergic receptors activation, signal is transduced through cardiac-specific isoform of phosphoinositide 3-kinase (PI-3 kinase), p100γ [49]. PKA is associated with its intracellular domain. It breaks down cAMP molecule and controls the activity of Na+/K+-ATPase through phospholemman phosphorylation. However, regulation of Na+/K+-ATPase through this third mechanism is an issue of intensive research with, currently, very controversial results, conclusions and opinions [38, 39, 50, 51].
Phospholemman (FXYD1 protein) is regulated through activity of PKA and PKC. Dynamic interaction between protein kinases, phospholemman, and Na+/K+-ATPase controls the intracellular concentration of Na+. Unphosphorylated phospholemman inhibits Na+/K+-ATPase, while phosphorylated phospholemman relieves this inhibition. It is an immediate response, activated by sympathetic innervations, the fact fully coherent with previously mentioned detail that PKA is involved in short-term regulation. Hyperphosphorylation of phospholemman through PKA activity enhances the activity of Na+/K+-ATPase and prevents Na+ overload during normal, physiological response to increased heart rate, as for example in stress [52]. On the other hand, it was shown that blockade of β adrenergic receptor has beneficial effect for patients, owing to reduction in PKA activity. One of the supposed regulatory mechanisms is almost completely opposite to previously described one. Namely, it is suggested, by using animal models and isolated cardiomyocytes that Na+/K+-ATPase can be regulated by glutathionylation. It is a form of a reversible oxidative modification in which the glutathione (GSH) forms a disulfide bond with β1 subunit of Na+/K+-ATPase and subsequently inhibits it [53]. This is caused by PKA activation and “mediated by the activation of PKC and nicotinamide adenine dinucleotide phosphate (NADPH) oxidase in a downstream pathway shared with angiotensin II (Ang II)” [53]. Blockage of β adrenergic receptors, decreases the activity of PKA in this model, reduces glutathionylation and stimulates the activity of Na+/K+-ATPase [53].
2.4 Na+/K+-ATPase and Intracellular Signaling
Although Na+/K+-ATPase is generally considered a non-receptor membrane molecule, it can serve and act in certain pathways as a signal transducer [54]. Lower doses of ouabain (non-inhibitory doses) after binding to Na+/K+-ATPase trigger signaling that involves inositol 1,4,5-trisphosphate receptor (IP3R) [54–56]. Activation of IP3R results in increase of intracellular Ca2+ [54, 55] and activates the pleiotropic transcriptional factor nuclear factor kappa B (NF-κB) [56]. NF-κB has dual potential effect on cardiomyocytes. As a part of TNF-α pathway, it mediates detrimental effects of TNF-α on cardiomyocytes. On the other hand, data on pharmacologic inhibition of NF-κB indicate that it could be involved in cardioprotective mechanisms during ischemic preconditioning of the myocardium [57]. Another common intracellular pathway activated by ouabain-related inhibition of Na+/K+-ATPase is PI3K. PI3K phosphorylates membrane phospholipid and generates the Akt kinase (protein kinase B). PI3K/Akt signaling pathway has antiapoptotic activity in cardiomyocytes. However, the net effect of proapoptotic and antiapoptotic properties of ouabain remains to be elucidated.
From the pathological and clinical point of view, control of Na+/K+-ATPase is of essential importance for the therapy of heart failure, one of the leading causes of death in modern world. Inhibition of Na+/K+-ATPase exerts the positive inotropic effect, but at the same time is associated with known mechanisms of cardiomyocytes cell death, which is the major concern in the application of cardiac glycosides. New studies offer promising and exciting results. It was shown that inhibition of Na+/K+-ATPase could be achieved simultaneously with the activation of another ATPase important for cardiomyocytes preservation and contractility, SERCA (isoform SERCA2a) [58]; SERCA structure, function, and role of Na+/K+-ATPase and SERCA in heart failure are explained in the following sections.
3 SERCA Proteins: Structure, Isoforms, and Function
3.1 Structure of SERCA
Sarcoplasmic reticulum Ca2+-ATPase (SERCA) is a 110 kDa integral membrane protein and belongs to P-type family of ion pumps, which also includes plasma membrane Ca2+-ATPase (PMCA), Na+/K+-ATPase, H+-ATPase, and K+-ATPase [59, 60]. It is the only active transporting pump located on the membranes of the sarcoplasmic reticulum (SR) [61]. In vertebrates there are three main different forms of SERCA pumps (SERCA1, 2, and 3), encoded by ATP2A1, ATP2A2, and ATP2A3 genes, located on three different chromosomes (chromosome 16, 12, and 17). These genes produce more than ten isoforms by alternative splicing [60, 62–64]. These isoforms are highly conserved in structure, with 75 % or more homology between them [60]. Besides its role in coding the SERCA2 protein, the ATP2A2 gene is associated with certain conditions such as Darier’s disease and Acrokeratosis verruciformis [65].
3.2 Isoforms of SERCA
SERCA1 has two main isoforms, SERCA1a and 1b, expressed in adult fast-twitch skeletal muscles (1a) and fetal tissues (1b). The main form of SERCA in cardiac muscle cells is the SERCA2 which has four isoforms (a, b, c, and d) [62]. SERCA2a, also known as “muscle specific isoform”, is expressed in cardiac muscle cells, slow-twitch skeletal muscles and smooth muscle cells. SERCA2b is a ubiquitous form present in muscle, but also in non-muscle cells [60]. The existence of SERCA2c protein was demonstrated in the heart [66] and its mRNA was also found in hematopoietic cells [67]. In humans, SERCA3 includes five isoforms (SERCA3b, 3c, 3d, 3e, and 3f) in addition to the species-unspecific SERCA3a, making a total of six isoforms. These isoforms are mostly expressed in non-muscle cells [60, 62], but recent evidence suggest cardiac expression of certain isoforms, such as SERCA3d and SERCA 3f, which are only present in humans [62, 68]. In summary, in human cardiomyocytes six isoforms have been detected so far: SERCA2a, SERCA2b, and SERCA2c, as well as SERCA3a, SERCA3d, and SERCA3f with specific intracellular localization [61].
3.3 Functions of SERCA
Contraction is the main characteristic of muscle cells and it is mediated by calcium ions. The main intracellular depot of Ca2+ is the sarcoplasmic reticulum (SR), a membranous network present in muscle cells, able to sequester and store millimolar amounts of calcium [60]. By releasing Ca2+ from its cisternae into the cytosol, it initiates the process of muscle cell contraction. Most of the Ca2+ responsible for muscle contraction comes from the SR and it is released during cardiac systole by the process of Ca2+ induced Ca2+ release, where an increase of cytoplasmic Ca2+ concentration produced by the L-type Ca2+ current is the main trigger for Ca2+ from SR [69]. The ryanodine receptor (RyR) located on SR, serves as a Ca2+ release channel through which Ca2+ enters the cytosol. A key factor that controls the level of SR Ca2+ levels is the activity of SERCA. By reuptaking the Ca2+ back to SR, it lowers the cytosolic calcium levels and replenishes the Ca2+ stores in SR, thus enabling the relaxation and new contraction of muscle cells in a repeated contraction-relaxation cycles [60]. SERCA and the role of Ca2+ ions in the muscle contraction was firstly described almost simultaneously by two different laboratories in two different articles: that of Ebashi and Lipmann in 1962 and Hasselbach and Makinose in 1961 [70, 71].
3.4 Regulation of SERCA Function in Physiological and Pathophysiological Conditions
The activity of SERCA pumps is regulated by certain proteins such as sarcolipin and phospholamban. Sarcolipin, which is mainly localized in the atrium, inhibits the expression of SERCA1a and SERCA2b, while phospholamban suppresses the function of SERCA protein and is highly expressed in ventricular muscle. Thus, these two intrinsic membrane proteins expressed in the SR, lower the SERCA affinity for Ca2+ [64, 72]. Phospholamban is in dynamic interaction to SERCA2a. It is a 52 amino acid protein of the sarcoplasmic reticulum and exists in monomeric and pentameric form. The monomeric form is a key regulator of the SERCA activity.
The SERCA pumps represent key elements that are necessary for the normal contractility of the human myocardium. Contractile dysfunction which is present in certain conditions such as cardiac hypertrophy and heart failure can be attributed to the reductions in SERCA activity [73]. Reduced expression of SERCA2a (predominant cardiac isoform) contributes to the abnormal contractility of the myocardium and is present in several cardiac diseases including ischemic heart disease and certain forms of cardiomyopathies, which tend to progress to heart failure [72]. Patients with end stage heart failure of different etiologies show decreased levels of SERCA2a mRNA and SERCA2a protein, but certain studies reported unchanged levels of SERCA, while others only reported alterations in phospholamban status.
The important role of SERCA pumps has been studied extensively in numerous studies on transgenic animals that overexpress SERCA or are deficient in SERCA pump isoforms in cardiomyocytes [60]. Overexpression of the predominant cardiac SERCA isoform (SERCA2a) in experimental transgenic animals resulted in increased maximal rates of contraction and relaxation of the heart, without any structural or functional abnormalities and with a normal life span.
4 Cardiomyocytes Ultrastructure and Localization of ATP-ases
The further discussion on ATPases and their functional, clinical, and pathological significance should be preceded by brief resume of cardiomyocytes structure. Cardiomyocytes are principal, contractile cells of the myocardium and working force of the cardiac pump. On a light microscopy level, cardiomyocytes resemble striated muscle with alteration of dark (A) and light (I) bands. Cardiomyocytes consist of even smaller subunits called myofibrils which consist, further, of series of basic structural, organizational and functional units—sarcomeres (Fig. 7.1). Each sarcomere contains thick myofilaments made of myosin II in the central region, and thin F-actin myofilaments associated with Z lines, at the end of sarcomeres (Fig. 7.1). Besides F-actin, thin filaments contain troponin and tropomyosin. Organization of thick and thin filaments within sarcomeres and association of sarcomeres into myofibrils form a morphological pattern of cardiac muscle as a cross-striated muscle. A bands are formed of myosin filaments with portions of actin filaments in-between, while I bands contain actin myofilaments. Running through the midlines of I bands are Z lines. The position of myofilaments is controlled and preserved during each myocardial cycle by a network of cytoskeletal and cytoskeleton-associated proteins. These proteins form supportive mesh that protects sarcomeres from mechanical stress.
Structure of cardiomyocytes (Transmission Electron Microscopy (TEM); original magnification 3500×, bar = 10 μm). Dark bands—A bands, contain myosin and actin (A); light bands—I bands with actin myofilaments (I); Z lines (black arrowheads); H band and M line—white arrowhead (H band is a space between ends of actin filaments inserted in between myosin filaments and it contains creatine kinase; M line contains myomesin that links myosin filaments); IC intercalated disks (Courtesy of Aleksandar Djordjevic, Department of Chemistry, Faculty of Sciences, University of Novi Sad and NanoBiomedicine team)
Accessory proteins, components of this network are titin, nebulin, α actinin, myomesin, desmin, αB-kristalin, plectin, dystrophin, tropomodulin, and ankyrin. Desmin is an intermediate filament that forms lattice surrounding the Z lines of sarcomeres. It is linked to Z lines via plectin and stabilizes myofibrils and anchors them to sarcolemma. Ankyrin, αB-kristalin, dystrophin, dystroglycan complex, and sarcoglycan complex form the attachment area of actin filaments to sarcolemma and laminin of the external (basal) lamina at the specific structural unit called costamere [74]. Association of costameric proteins with Na+/K+-ATPase is important for survival of cardiomyocytes as explained in following sections.
Myofibrils are surrounded with cisternae of smooth endoplasmic reticulum—sarcoplasmic reticulum. Sarcoplasmic reticulum is a network of longitudinal and transversal cisternae. Longitudinal cisternae parallel myofibrils, while transversally oriented cisternae (terminal cisternae) are in a form of membranous sacs in close proximity and laterally to T tubules. T tubules are invaginations of the plasma membrane—sarcolemma. Association of terminal cisternae and T tubules form diads. Diads are located at the level of Z lines.
Cardiomyocytes are interconnected with intercalated disks, junctional complexes that consist of fasciae adherentes, desmosomes, and gap junctions. Intercalated disks are located at the level of Z lines. Z lines consist of α-actinin which anchors actin to Z lines. Z lines also contain desmin, nebulin, titin, and plectin. Sarcoglycan, dystroglycan, and dystrophin are also components of intercalated disks. For more details on cardiomyocytes structure, readers are referred to excellent literature [75–81].
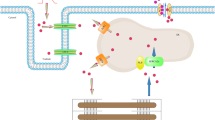
4.1 Localization of Na+/K+-ATPase and SERCA
Na+/K+-ATPase is a transmembrane protein, and hence its primary localization is sarcolemma (Fig. 7.2). However, the distribution of Na+/K+-pump isoforms in cardiomyocytes is not equal: α2- and α3-subunits are located mainly in T tubules (in close proximity to sarcoplasmic reticulum), while α1-subunits are more uniformly distributed throughout the sarcolemma [76, 77]. Alpha1, α2 and β1 subunit of Na+/K+-ATPase are present at the level of intercalated disks [76, 78]. In muscle cells, SERCA pumps are localized in the SR (SERCA1, SERCA2a). Immunohistochemical studies on other cells showed that SERCA2 and SERCA3 are mostly localized inside the network of endoplasmic reticulum, but also in the outer membrane of the nuclear envelope [79]. Additional and more detailed studies on human cardiomyocytes showed that SERCA2a and SERCA2b have a uniform pattern of distribution in the sarcoplasmic reticulum, although certain differences between them still exist. For example, SERCA2a is in the regions located close to the T-tubules and to longitudinal sarcoplasmic reticulum, while SERCA2b appears to be mostly localized in the regions close to T-tubules only. SERCA2c isoform is in close proximity to the sarcolemma and in intercalated disks, as well as SERCA3a. SERCA3d and SERCA3f proteins are located in perinuclear and subplasmalemmal regions of human cardiomyocytes [62].
5 ATP-ases and Mechanisms of Cardiomyocyte Cell Death
The insufficiency of membrane ionic transport systems is associated with the increased rate of cell death. The first reason for the Na+/K+-ATPase and SERCA insufficiency is the lack of ATP and derangement of the aerobic respiration or mitochondrial oxidative phosphorylation. The most frequent cause of cardiomyocytes injury due to a lack aerobic metabolism and ATP deficiency is ischemic heart disease with its main entities: angina pectoris, acute myocardial infarction, chronic ischemic heart disease and sudden cardiac death. Disruption of the oxidative phosphorylation itself provokes series of mutually connected intracellular events leading to disturbance of other three processes vital for the preservation of the cell, namely: protein synthesis, cell membrane integrity and genetic material preservation. The oxidative phosphorylation blockage with the decreased ATP reserves and insufficiency of Na+/K+-ATPase leads to changes in the concentration gradient of Na+ and K+ ions. There is an efflux of K+ ions, and influx of Na+ ions. Since water isoosmotically follows potassium, intracellular edema and hydropic swelling are progredient.
These changes are reversible up to a certain point. Hydropic swelling of the cell is a reversible change [80]. Dilatation of cisternae of sarcoplasmic reticulum and mitochondria due to lack of ionic and osmotic disbalance are main morphological characteristic of reversible injury and illustrate the fact that the disruption of the structural integrity and influx of water is not an exclusive characteristic of the plasma membrane, but membranes of organelles are affected as well, hence the dilatation of sarcoplasmic reticulum and mitochondria.
Reduced cellular respiration is associated with the decrease of intracellular ATP, but also with the increase of AMP and the activation of anaerobic glycolysis and enzymes phosphofructokinase and phosphorylase. Lactic acid, the side product of this metabolic pathway, is formed. A presence of lactic acid leads to intracellular acidosis. Low pH values interfere with the normal functioning of intracellular enzymes essential for the synthesis of structural proteins and phospholipids, which ultimately leads to further disruption of cell membranes and membranes of the organelles.
The control of Ca2+ ions influx is affected by disruption of membranes and energy depletion. Namely, the Ca2+ concentrations inside cardiomyocytes cytoplasm are regulated at very persistent level of 30–100 nM during the diastole. This is achieved by the activity of sarcolemmal Ca2+-ATP-ase and Na+/Ca2+ exchanger as well as the activity of SERCA and Ca2+ buffering molecules [81, 82]. Decreased amount of ATP is followed by insufficiency of the Ca2+–Mg2+-ATP-ase and sarcolemmal Na+/Ca2+ exchanger (NCX).
Namely, metabolic acidosis initiates Na+/H+ exchange also, with transport of H+ out of the cell. H+ is exchanged for Na+, transported into the cell [80]. High concentration of sodium activates NCX, contributing to increase in intracellular Ca2+. Calcium ions are also released from sarcoplasmic reticulum. We have already described this process in the section on SERCA structure and function. Namely, the ryanodine receptor (RyR) located on SR, serves as a Ca2+ release channel through which Ca2+ enters the cytosol.
Recently, a new specific form of transient intracytoplasmic influx of Ca2+ was described, called Ca2+ sparks. Ca2+ sparks occur when RyR opens spontaneously and release a small amount of Ca2+ locally [82, 83]. Ca2+ sparks are present in both systole and diastole. During diastole Ca2+ sparks are independent from Ca2+ influx through sarcolemma associated molecules [82]. During excitation-contraction coupling and systole, openings of L-type Ca2+ channel induces release of Ca2+ into cytosol which resemble thousands of Ca2+ sparks triggered by this process [82].
Insufficiency of Na+/K+-ATPase and NCX provoke ample and spontaneous diastolic Ca2+ sparks and cause arrhythmias and sudden cardiac death [84]. On the other hand, control of calcium ions through SERCA mechanism and their transport back to lumens of the SR cisternae is insufficient due to the lack of ATP.
Increase of the free cytosolic calcium ions activate enzymes leaking through the damaged organelles membranes. Activated phospholipases (phospholipase A2) and proteases further damage the membrane and cytoskeleton. Costameric ankyrin dissociates from its attachment [84] with severe disturbance of cytoskeleton and degeneration of myofibrils. At the same time, α1 and α2 subunits of Na+/K+-ATPase are detached from cytoskeleton, namely ankyrin-B. These events are directly provoked by influx of Ca2+ and activation of calpain in the setting of complex intracellular changes and metabolic acidosis [85–87] and are proved to augment Na+/K+-ATPase insufficiency.
Influx of calcium ions, also, leads to irregular and dissociated contraction of the cardiomyocytes with hypercontraction of sarcomeres and contraction band necrosis (Fig. 7.3). This phenomenon called the calcium paradox is a consequence of uncontrolled influx of calcium ions into energy-depleted cardiomyocytes. It is associated with the ischemic damage of the cell. That is why it could be found at infarction area and at the periphery of infarcted areas where it is a part of the ischemia-reperfusion sequence [85–88].
Nevertheless, it is not unique for the ischemic damage, but other forms of injury agents provoke it too by mechanisms similar to aforementioned mechanisms. It is also observed in the donors’ hearts [89]. It is observed in damages activated through ROS generation, for example lipid peroxidation associated with doxorubicin application [90].
In summary, defects in oxidative phosphorylation and ATP deprivation induce complex intracellular alterations with ionic and osmotic disbalance, hydropic swelling of the cell, and dissociated contraction. Balances of sodium, potassium and calcium ions are interconnected and lead to a same result.
Such observations are recently confirmed in a sophisticated model of a knock-in mouse in which the phospholemman residues phosphorylated by PKC and PKA have been mutated with reduction of Na+/K+-ATPase. This exacerbated Na+ overload and resulted in profound contractile dysfunction [52].
Disruption of lysosome membranes leads to leakage of lysosomal enzymes and their activation in the conditions of low intracellular pH and results in tissue digestion and necrosis.
Mitochondrial damage leads to further reduction or blockage of the oxidative phosphorylation, switch from aerobic into anaerobic metabolism and ceased synthesis of phospholipids, which subsequently afflicts integrity of all membranes. During ischemia there is an increase of reactive oxygen species (ROS). They cause lipid peroxidation of cardiolipin and damage the electronic transport chain, harming further oxidative phosphorylation.
During the mitochondrial injury, cytochrome c is released from the disrupted inner membrane of mitochondria, through mitochondrial permeability transition pores, which resembles potential for the activation of the inner apoptotic pathway. Endonucleases are also activated by uncontrolled calcium ions influx during the cell injury. In concordance with this observation is the fact, that all these changes are followed with the translocation of annexin A5 to the sarcolemma [91] and activation of caspases 3 and 8 [80, 92] which is consistent with apoptotic cell death. Mitochondrial injury has a potential to activate autophagy [93]. Regardless of a specific form of cell death, all these changes are followed by nuclear degeneration: karyolysis, karyopiknosis, and karyorrhexis, indicating irreversible changes. These changes are observed in endothelial cells as well, so the myocardial tissue is deprived of oxygen and glycose with progression of defects and absence of protective mechanisms. Once started, cellular injury is obviously a vicious circle that could hardly be stopped. After this summary, the first question is what type of cell death is actually provoked with this sequence of processes?
According to contemporary opinions on cardiac muscle cell death [89, 90, 94], two concurrent mechanisms of myocardial cell death persist in different models: apoptosis and necrosis. However, considering the presented mechanism of cellular injury, it is clear that each cellular damage has the potential for both necrosis and apoptosis, and intermediate forms of cell death could also be expected.
There are at least two more aspects that deserve to be mentioned. Reduction of the content of Na+/K+-ATPase itself is proved to have similar effect as ATP depletion. Decrease in synthesis, expression of mRNA, quantity or activity of Na+/K+-ATPase or its subunits is observed in cardiovascular diseases including heart failure, various forms of cardiomyopathies, hypertension, hypertrophy or obesity. Nevertheless, in the analysis of this particular question, we should always keep in mind that the decrease may be primary, but also a consequence of serial changes in the synthetic potential of the already injured cell, where the injury is provoked by hypoxia, ischemia, volume- or pressure-overload, reactive oxygen species (ROS), or complex genetic disorders.
Reduction of α1 subunit in an animal model of hypertension induced increased rate of myocardial cell death. However, the number of cardiomyocytes was higher. It was confirmed that although the reduction of α1 subunit induced increase in cell death, it stimulates expression of proliferative marker Ki67 in cardiomyocytes as well as increase in number of c-kit positive progenitor cells [95]. High glucose level is associated with reduced Na+/K+-ATPase activity and increased proapoptotic machinery: raised caspase-3 activity and Bax, as well as down-regulated Bcl-2 expression [96]. On the opposite, insulin has a protective effect and prevents apoptosis of cardiomyocytes as confirmed in the setting of digoxin treated heart failure [97].
Resveratrol, active principle of grape and red wine, has a cardioprotective effect in the model of ischemia-reperfusion injury. It increases the activity of both Na+/K+-ATPase and Ca2+-ATPase, increasing at the same time viability of cardiomyocytes. It reduces apoptotic cell death by increase in Bcl2 and decrease of Bax and caspase-3, reduces intracellular calcium and balance the activity of reactive oxygen species (ROS) [98]. Stable expression of Na+/K+-ATPase is essential for the survival of cardiomyocytes after ischemia-reperfusion injury in ouabain-preconditioning model [99].
Third question is relation of ouabain and ouabain-like cardiac glycosides, including digoxin, to cell death phenomenon. Cardiac glycosides have been used for the treatment of heart failure because of their capabilities of inhibiting Na+/K+ ATPase. This inhibition raises intracellular Na+ and attenuates Ca2+ extrusion via the Na+/Ca2+ exchanger, causing intracellular Ca2+ elevation and empowering contractile strength of the heart [100]. The question is if the therapeutic doses of ouabain and digoxin induce and increase cardiomyocyte cell death by inhibition of Na+/K+-ATPase in addition to positive inotropic effect?
It was shown that ouabain induces increased frequency of cardiomyocyte cell death by both apoptosis and necrosis, and reduced cell viability. It simultaneously activates antiapoptotic mechanism of PI3K/Akt, which is, however, insufficient to block effects of proapoptotic ouabain activity [101]. In addition to positive inotropic effect and elevated oxygen consumption, inhibition of Na+/K+-ATPase by glycosides, impair mitochondrial energetics and cause oxidative stress especially in conditions of increased workload.
The studies on guinea pig cardiomyocytes showed that elevated intracellular Na+ induced by glycosides treatment caused mitochondrial Ca2+ deficiency by activating the mitochondrial Na+/Ca2+ exchanger, the major mitochondrial Ca2+ efflux pathway. Furthermore, it is associated with significantly decreased NADH level and increased reactive oxygen species (ROS) accumulation [102–105].
Li et al. proposed that Na+/K+-ATPase inhibition such as by cardiac glycosides causes mitochondrial oxidative stress and increased ROS production through two separate pathways. In first way, Na+/K+-ATPase inhibition blunt mitochondrial Ca2+ accumulation, that reduces NADH production and therefore ROS removal. And in second way, Na+/K+-ATPase inhibition increasing intracellular Ca2+ and ATP hydrolysis, which produces a large amount of ADP that stimulates mitochondrial respiration and therefore ROS production [100].
The interrelation of described processes causes that many different sources of cell injury produces similar changes in myocardial tissue. Intracellular edema, swelling, vacuolization and hypercontraction of sarcomeres are common signs of injury in ischemic heart disease. They are present in reversible form during extracorporeal long-term preservation of donors’ hearts in the procedure of heart transplantation [94]. Lipid peroxidation of membranes in doxorubicin toxicity model has the same effect [90]. Damage of intracellular membranes by advanced glycation end products and ROS, due to NADH oxidase activity, in diabetes has also the same effect. As resumed in following section, hypertrophy and heart failure of different origin include the same sequence of processes.
6 Molecular Mechanisms of Heart Failure
Heart failure (HF) remains a major cause of morbidity and mortality in the developed world. In the population under the age of 65, HF prevalence approaches 1 % [106]. Over the last decade, important progress has been made in understanding of various intracellular and molecular mechanisms of HF. Heart failure (congestive heart failure—CHF) is a complex, chronic and progressive disorder, although acute forms of heart failure persist in association with sudden volume overload, acute myocardial infarction, valvular dysfunction or compromised ventricular filling and retention of blood in peripheral circulation. It is a common endpoint of several pathophysiological pathways included in different forms of cardiomyopathies, ischemic heart disease, hypertension or valvular heart disease.
Heart failure includes forward and backward component, systolic and diastolic insufficiency. Systolic dysfunction comes from reduced myocardial contractility most frequently due to ischemic heart disease, hypertension or dilated cardiomyopathy. Diastolic dysfunction is caused by inability of ventricles to relax and accept sufficient volume of blood during diastole in conditions like hypertrophy of left ventricle, myocardial fibrosis or constrictive pericarditis. The end result of systolic and diastolic dysfunction is reduced cardiac output, forward component of heart failure, as well as retention of blood in venous circulation—backward component.
Heart failure due to conditions with pressure or volume overload is preceded by hypertrophy. Hypertrophy is a compensatory mechanism aimed to enhance contractile strength of the myocardium. On a molecular level, hypertrophy is achieved trough intensive synthesis of new myofilaments and division of mitochondria, as well as accumulation of other organelles. Extensive synthesis of myofilaments’ proteins is followed with their intensive association into new sarcomeres, and hence the size of cardiomyocytes is raised. Two forms of hypertrophy exist. Concentric hypertrophy develops with pressure overload (systemic hypertension, chronic pulmonary hypertension or valvular disease, i.e., aortic stenosis). In this form of hypertrophy, new sarcomeres are arranged parallel, in a fashion that augments transversal cardiomyocyte diameter [107].
Volume overload stimulates another form of hypertrophy with deposition of newly synthesized sarcomeres along the longitudinal axis of cardiomyocytes and dilatation of ventricles. The gene expression in hypertrophy is changed towards activation of c-myc, c-fos, jun, and EGR1 as well as towards the activations of fetal programs. For example, β-myosin heavy chains are synthesized instead adult forms of α-myosin heavy chains [57]. Atrial natriuretic peptide (factor) (ANP) is synthesized in ventricular cardiomyocytes in addition to physiological synthesis in atrial myoendocrine cells. These changes have compensatory effect up to a certain point. For example, Ca2+/calmodulin-dependent protein phosphatase (calcineurin) is shown to have a role in inducing hypertrophy. It is activated to dephosphorylate the transcription factor NF-A3, enabling its translocation to the nucleus and enhance hypertrophic remodeling [108]. However, calcineurin, in this setting, activates apoptotic signaling pathways at the same time and is responsible for the increased rate of cardiomyocytes’ cell death.
More important, the enlargement of cardiomyocytes is not followed by the proper extension of capillary network. The capillary-to-cardiomyocyte ratio, which should be at least 1:1 in normal myocardium, is reduced, while the distance between capillaries and cardiomyocytes is increased. Hypertrophic myocardium is in increased need for oxygen consumption and there is a profound discrepancy between the oxygen and nutrients available and oxygen and nutrients needed. That is why most cardiomyocytes stay without nutritive support and enter some form of reversible injury or cell death as described above through the aforementioned mechanisms. This is followed by the decrease in the number of functional cardiomyocytes and fibrosis, since the regeneration of cardiomyocytes from circulating and cardiac progenitor cells or dividing cardiomyocytes is suppressed and cell death prevailed [57, 90, 109]. With the development of this irreversible damage of cardiomyocytes and fibrosis, cardiac hypertrophy loses its compensatory potential, sliding to heart failure with contractile dysfunction and dilatation of chambers.
In viable cardiomyocytes, synthesis of all proteins including contractile proteins and enzymes is altered, leading to reduced synthesis, synthesis of dysfunctional proteins, structurally anomalous proteins or nonspecific and less functional isoforms of proteins [110, 111]. Besides, duration of heart failure in each patient, nature and severity of the injury that caused heart failure, and dynamics of heart failure development could affect findings of molecular and cytological research. These facts should be kept in mind in the interpretation of results on Na+/K+-ATPase and SERCA expression, presented in the following chapters.
7 NA+/K+-ATP-ase and Heart Failure
Before the consideration on a linkage between Na+/K+-ATPase and heart failure, we will summarize briefly functions of Na+/K+-ATPase. Na+/K+-ATPase (Na+/K+-pump) is an enzyme located in the cell membrane which transports three sodium ions outside of the cell and two potassium ions into the cell, generating ion gradients necessary for the maintenance of the membrane potential [112]. Na+/K+-pump has been found in almost all animal tissues, including human myocardium. For its proper functioning, this enzyme uses the energy of the hydrolysis of ATP molecule. Na+/K+-ATPase indirectly modulates the myocardial contractility by controlling the function and driving force for Na+/Ca2+ exchanger (NCX) [113]. NCX functions as a membrane transporter for extrusion of Ca2+ outside of the cardiomyocytes, at the same time transferring three Na+ into the cell [114]. Activity of Na+/Ca2+ exchanger greatly depends on intracellular Na+ concentration, and thus Na+/K+-ATPase activity: even a slightly elevated Na+ concentration limits the function of NCX which extrudes less Ca2+, resulting in higher intracellular Ca2+ concentration. Cardiac glycosides, which are used in the treatment of heart failure, act as Na+/K+-ATPase inhibitors by the aforementioned mechanism to evince their inotropic effect [115].
Na+/K+-ATPase is composed of three subunits: α, β and γ-subunit. Na+/K+-ATPase α-subunit contains the binding sites for ions (Na+ and K+), ATP and cardiac glycosides, and also has catalytic ability for ATP molecule. Smaller, β-subunit is important for the transport of synthesized Na+/K+-ATPase to the plasma membrane, while it also modulates ATPase activity [29]. Na+/K+-ATPase γ-subunit (also called FXYD) is the latest subunit discovered. FXYD represents a family of proteins associated with Na+/K+-ATPase which modulates the function of this enzyme [26]. Cardiomyocytes contain only one form of FXYD protein called phospholemman, which regulates the function of Na+/K+-ATPase. Namely, in unphosphorylated state, phospholemman inhibits Na+/K+-ATPase by reducing the affinity for intracellular Na+, while phosphorylated phospholemman disinhibits Na+/K+-ATPase, returning it to its active state [116, 117].
Cardiomyocytes contain three α (α1–α3) and two β (β1–β2) isoforms of Na+/K+-ATPase subunits. Measurement showed that Na+/K+-ATPase α1-subunit is predominantly present in cardiomyocytes, while Na+/K+-ATPase α2- and α3-subunits are expressed to a lesser extent [118]. Early measurements of Na+/K+-ATPase quantity showed that the concentration of Na+/K+-pump in normal human ventricular myocardium was approximately 700 pmol/g wet weight [119]. Heart failure is characterized by the elevation of Na+ concentration in human cardiomyocytes but also in animal models [120, 121]. There are two possible explanations for this finding: reduced Na+ extrusion (which implies changes in Na+/K+-ATPase) or larger Na+ influx (implying changes in other Na-pumps, such as Na+/H+ exchanger) [122].
Different studies have shown that the reduced Na+ extrusion could be the result of Na+/K+-ATPase alterations [20, 34]. It was demonstrated that the expression of certain Na+/K+-ATPase subunits was diminished in cardiomyocytes obtained from human heart failure material. Particularly, α1-subunit expression was found to be lower by 38 %, α3-subunit by 30 %, β1-subunit by 30 % and overall Na+/K+-ATPase activity was lower by 42 % in heart failure cardiomyocytes, with unchanged levels of mRNA [20, 34].
Animal heart failure models showed different results when observing expression and activity of Na+/K+-ATPase subunits. Rabbit heart failure model pointed to similar results as human heart failure cardiomyocytes, with all α-subunits having lower protein expression [123], whereas in rat heart failure model Na+/K+-ATPase α1-isoform expression was unchanged, with α2-subunit expression reduced and α3-subunit expression increased [124]. Different structural or functional mechanisms underlie the lower Na+/K+-pump activity. Therefore, heart failure in rabbit was characterized only by reduced Na+/K+-pump expression with normal Na+/K+-ATPase pumping ability [125], while certain rat heart failure models showed decreased Na+/K+-ATPase pumping ability with normal Na+ affinity [124].
Nevertheless, it is still unclear whether Na+/K+-ATPase dysfunction is one of the reasons for heart failure, or perhaps compensatory mechanism functioning similarly to cardiac glycosides. In their animal model of heart failure, Zahler at al. [126] indicated that it is more probable that Na+/K+-ATPase expression is reduced during early development of myocardial dysfunction and asymptomatic phase of heart failure, and not being its compensatory mechanism. Since heart failure is a condition principally characterized by reduced left ventricle ejection fraction (LVEF), Norgaard et al. [35] showed correlation between decreased LVEF and decreased Na+/K+-ATPase concentration in cardiomyocytes.
Cardiac glycosides have been in use for the treatment of heart failure for more than two centuries. Due to serious toxic effect and narrow therapeutic window, number of indications for the use of cardiac glycosides has been minimized. In patients with heart failure and atrial fibrillation, cardiac glycosides are recommended for the control of ventricular rate in patients intolerable to beta-blocker therapy [127]. Cardiac glycosides, such as digoxin, exert their positive inotropic effect by inhibiting Na+/K+-ATPase and increasing Na+ intracellular concentration, which in turn stimulates Na+/Ca2+ exchanger to transport Ca2+ inside the cell, elevating Ca2+ availability for muscle contraction and thus improving cardiomyocyte contraction force [128].
During therapeutic digitalization, not all Na+/K+-ATPase receptors are occupied by digoxin; it is estimated that the percentage of occupied Na+/K+-ATPase receptors is 24–35 % in the human heart [119, 129]. Cardiac glycosides bind to Na+/K+-ATPase α-subunit consequently blocking this enzyme. It was generally believed that cardiac glycosides had similar affinity for different Na+/K+-ATPase α-subunits. However, studies comparing ouabain’s affinity for Na+/K+-pump showed that its affinity for α2β1-isoform was two times greater than for α1β1- or α3β1-isoforms [130, 131]. Moreover, these studies showed different Na+ affinity for Na+/K+-ATPase isoforms; in one study Na+ affinity was similar for all enzyme isoforms [131], while the other study showed that the highest affinity was for α1β1-isoform and the lowest for α3β1 Na+/K+-ATPase heterodimer [130].
Cardiac glycosides may have additional useful roles in the treatment of heart failure, which do not include Na+/K+-ATPase inhibition, yet their neurohumoral effects. Except inhibiting Na+/K+-ATPase in cardiomyocytes, cardiac glycosides also inhibit extracardiac Na+/K+-ATPase, thus possibly improving overall hemodynamic and restoring bar receptor activity in heart failure patients [132, 133]. Also, in recent years it has been proposed that cardiac glycosides may have hormone-like functions, by acting on different Na+/K+-ATPase isoforms which in that case may have receptor-like functions [134]. Such presumption has been supported by the evidence of potential antitumorous effect of cardiac glycosides [135]. Also, it has been shown that cardiac glycosides act by inhibiting cell growth and division, stimulate programmed cell death and release endothelin-1 from endothelial cells [134].
During the ongoing search for new effective, well tolerable and less toxic inotropic drug, an antibody was developed against the L7/8 extracellular domain of the Na+/K+-ATPase α-subunit. This antibody showed not only the inhibition of Na+/K+-pump, but also several cardioprotective effects against ischemia and reperfusion through PI3K/Akt signaling cascade [136]. This finding may open new therapeutic approach in heart failure patients, and also shed a new light on Na+/K+-ATPase physiologic function.
8 Downregulation of SERCA in Heart Failure
One of the key abnormalities in both human heart failure and experimental models of heart failure is abnormal intracellular calcium ion (Ca2+) handling. Before further consideration, we will summarize briefly facts on SERCA function.
SERCA plays a pivotal role in both myocardial contraction and relaxation. The predominant SERCA isoform in the heart is SERCA2a (97.5 %), although low levels of SERCA2b (2.5 %) are also found [137].
SERCA functions to pump Ca2+ into the sarcoplasmic reticulum (SR). It returns the intracellular Ca2+ concentration ([Ca2+]i) to resting levels causing relaxation of the cardiomyocyte and replenishment of the SR Ca2+ store for the next cycle [138]. Alterations in SERCA level affect Ca2+ homeostasis and cardiac contractility by influencing Ca2+ reuptake as well as the expression and activity of other Ca2+ handling proteins [139].
The reduced function of SERCA in heart failure is caused by reduced synthesis, activity or gene expression. Reduced SERCA-to-phospholamban ratio has the same effect. Phospholamban as a major regulator of SERCA2a activity is directly involved in development of cardiac disease, including heart failure [140].
SERCA2 activity is reversibly regulated by phospholamban through cAMP dependent phosphorylation [137, 140–143]. Dephosphorylated phospholamban is an inhibitor of SERCA, while phosphorylation of phospholamban relieves this inhibition.
Phospholamban activity, itself, is regulated by two phosphoproteins, the inhibitor-1 of protein phosphatase 1 (PP1) and the small heat shock protein 20. PKA and PKC are molecules actively involved in phosphorylation of phospholamban as well. In general, the whole Ca2+ transporting machinery including L-type Ca channels and the ryanodine receptor (RyR) is under control of PKA and PKC [49, 53].
Recently, two new molecules have been identified as regulators of SERCA activity: SUMO, S100 and the histidine-rich Ca2+-binding protein [140] and regucalcin [108]. It is also established that previously described Ca2+ sparks or precisely controlled diastolic sparks are essential for the normal balance of SERCA activity [82, 83].
On experimental heart failure model, it was established that out of six isoforms of SERCA expressed in heart, there is decreased expression of transcripts of SERCA2a, SERCA3b and 3c, while the expression of SERCA2b and 3a transcripts remains unchanged. Although SERCA2a is downregulated in heart failure, it is still predominant isoform in cardiomyocytes.
In heart failure in humans, there is a reduction of SERCA2a and an increase of SERCA3f. SERCA3f is proved to induce protein synthesis anomalies, endoplasmic reticulum stress, and apoptosis in cell cultures [60, 61].
Disturbances in the regulatory function of SERCA/phospholamban have also been implicated as important contributors to heart failure pathogenesis. Phospholamban is less phosphorylated in heart failure due to increase in global phosphatase expression in cardiomyocytes of patients with heart failure, resulting in even greater SERCA inhibition.
Interesting also is the observation that functional PLB-null genotype in humans causes heart failure, whereas the gene-targeted PLB-knockout mice have no apparent cardiac problems [144].
Molecules that control phospholamban activity are implicated in heart failure as well. Heart failure and downregulation of adrenergic receptors are associated with reduced cAMP-dependent protein kinase (PKA). This mechanism leads to the inactivation of inhibitor-1 with consequently increased activity of PP1. Hyperactive PP1 leads to the dephosphorylation of phospholamban and inhibition of SERCA2a thus reducing the calcium uptake [145].
Inhibition of SERCA activity leads to profound disturbance in concentration of calcium ions which along with other molecular mechanisms of heart failure described previously deepen the morphological and physiological disturbances. In concordance with this is the finding that preserved SERCA activity and controlled calcium ions turnover inhibits calcineurin associated apoptotic pathway [61].
8.1 Potentials for New Therapeutic Approaches
Kranias et al. recognized SERCA/phospholamban complex and its role in cardiac contractility, and indentified the potential for new therapeutic approach by targeting this complex [140].
Normalization of SERCA2a function has been shown to increase contractility in failing human cardiomyocytes and to improve hemodynamics along with survival in rodent and large animal models of heart failure [146–148].
The overexpression of SERCA2a has also been found to restore energetic supply and to decrease ventricular arrhythmias in a model of ischemia/reperfusion injury [149–151]. Therefore, SERCA2a is one of the most promising targets for the treatment of HF.
Ferrandi et al. [152] showed that istaroxime represents the first example of a small molecule that exerts a luso-inotropic effect in the failing human heart through the stimulation of SERCA2a activity and the enhancement of Ca2+ uptake into the SR by relieving the phospholamban inhibitory effect on SERCA2a in a cAMP/PKA independent way. We previously mentioned that istaroxime inhibits Na+/K+-ATPase with positive inotropic effect simultaneously with the activation of SERCA2a [58]. In such a way, it brings together positive properties of Na+/K+-ATPase inhibition with better control of calcium ions concentration. The consequence of this potential therapeutic strategy is achievement of enhanced contractility (inotropy) with facilitated relaxation (lusotropy).
Advances in the understanding of the molecular basis of myocardial dysfunction together with the evolution of gene transfer technology has placed congestive heart failure as a separate task within reach of gene-based therapy [61, 153–155]. Lipskaia et al. focused on gene therapy using SERCA2a or molecules regulating SERCA2a activity to treat heart failure. New data show that SERCA2a gene transfer improves contractile function and restores electric stability of the failing cardiomyocytes [156].
9 Downregulation of SERCA in Aging Heart
Aging is associated with alteration in cardiac structure and function, while the most prominent feature is increased left ventricular mass (left ventricular hypertrophy), impaired diastolic function and preservation of systolic function [157]. The characteristic cellular changes in aging myocardium include myocyte hypertrophy, interstitial fibrosis, and impaired myocyte relaxation. Many studies suggest that calcium dysregulation contributes to impaired function of cardiomyocytes in the aging process [158–166]. The intracellular calcium transient is regulated by a family of proteins including sarcoplasmic reticulum (SR) calcium ATPase (SERCA), its inhibitory protein phospholamban (PLB), the calcium storage protein, calsequestrin, and the SR calcium release channel (ryanodine receptor) [159]. SERCA plays a particularly important role in maintaining intracellular calcium through its ability to pump cytosolic calcium into SR during myocardial relaxation [166]. Several studies have shown that SERCA activity is decreased in aging heart [159, 165]. In some cases, this decrease in activity has been related to a decrease in SERCA protein level or a decrease in the ratio of SERCA to PLB [159]. Other studies have also demonstrated age-associated decreases in the amount of calcium/calmodulin-dependent protein kinase (CaMK), endogenous CaMK-mediated phosphorylation of SERCA and PLB, and the phosphorylation-dependent stimulation of SR calcium sequestration [159]. Besides changes in amounts and isoforms of calcium regulation proteins in heart tissue, recent studies suggest that alterations in the function of SERCA can be regulated by means of oxidative posttranslational modifications [167, 168]. It is well known that reactive oxygen species (ROS) and oxidative stress are increased in aging myocardium [169–171].
In cardiac myocytes in vitro, it has been shown that oxidants (e.g., nitroxyl or peroxynitrite) in low, “physiologic” levels cause reversible S-glutathiolation of SERCA at cysteine 674 (C674) leading to its activation. In contrast, higher levels of oxidants (e.g., H2O2 or peroxynitrite) that may be associated with pathologic conditions lead to irreversible oxidation of SERCA at one or more sites, including sulfonation at C674. Irreversible oxidation of C674 may inhibit basal enzyme activity and further prevent activation via S-glutathiolation. Studies in aging myocardium have further demonstrated irreversible oxidation of SERCA cysteines and nitration of tyrosines [172]. It was also observed that myocardial levels of 3-nitrotyrosine and 4-HNE indicative of oxidative stress and sulfonation of SERCA at C674 are markedly increased in aging hearts and that these increases are prevented in transgenic mice with catalase overexpression. Furthermore, catalase overexpression prevents decreased SERCA activity, and impaired diastolic function in myocytes from aging hearts [172, 173]. These studies suggest that reactive oxygen species such as H2O2 contribute to impaired diastolic function in cardiac aging, at least in part via oxidative modification of SERCA, and in particular, via sulfonation at C674 [172–174]. The recent study in male animal aging model indicates that aging reduces cell shortening, which is associated with a decrease in the amplitude of the systolic Ca2+ transient. This may be occurring due to a decrease in peak L-type Ca2+ current. The same study has shown that SR Ca2+ load appears to be maintained during normal aging but evidence suggests that SR function is disrupted, such that the rate of sarco/endoplasmic reticulum Ca2+-ATPase (SERCA)-mediated Ca2+ removal is reduced and the properties of SR Ca2+ release in terms of Ca2+ sparks are altered [175]. Besides, it was shown that there are male–female differences in the way the heart ages at the cellular level. The data on aging and gender-base differences have important clinical implications. Although aging is a physiological process, many of its aspects, including alterations in Ca2+ homeostasis, make the myocardium prone to disease [175].
Improved comprehension of cellular mechanisms of aging will help us to understand susceptibility to different cardiovascular diseases during aging. Ultimately, we will be able to identify new targets for intervention in the treatment of these diseases.
10 ATP-ases and Cardiomyopathy
By definition, cardiomyopathy is a primary, intrinsic defect of the myocardium. Before the advance in genetics, cardiomyopathies were mostly characterized as idiopathic. Due to progress in molecular biology techniques, numerous gene abnormalities were identified as specific causes of cardiomyopathies.
Ischemic cardiomyopathy is a term used in clinical practice to describe clinical and morphological findings in patients with heart failure caused by ischemic heart disease and by strict definition it should not be a part of cardiomyopathy entity.
Cardiomyopathies are classified as dilated, hypertrophic, and restrictive. Characteristic of dilated cardiomyopathy is progressive cardiac dilatation and systolic dysfunction, while hypertrophic cardiomyopathy is defined by myocardial hypertrophy and mostly diastolic dysfunction [111].
Ischemic heart disease and consequent heart failure resemble dilated cardiomyopathy, while hypertensive heart disease bears a resemblance to dilated or hypertrophic cardiomyopathy depending on a phase in the disease progression.
There are changes in ATPases activities in different forms of cardiomyopathy. As showed in the study of Norgaard et al., among 24 patients with idiopathic dilated cardiomyopathy, 19 had impaired LV function and lower Na+/K+-ATPase concentration whereas 5 patients had normal LV function and higher Na+/K+-ATPase concentrate [35].
In previous discussion we interpreted results applicable mostly on ischemic cardiomyopathy. Semb et al. studied expression of Na+/K+-ATPase subunits in the post-infraction rat model of congestive heart failure (CHF) [176] and found that expression of α2 and α3 isoforms was affected whereas expression of the α1- and β1-subunits (mRNA and protein) was not significantly different than in controls. Expression of α2 isoform at the level of mRNA and protein were lower in CHF hearts and the α3 isoform mRNA was higher.
We will review changes of ATPases expression and activity in cardiomyocytes in diabetes mellitus. Diabetic cardiomyopathy is the term used for cytological and pathological cardiac alterations developed through the course of diabetes, with clinical manifestations. Myocardium is affected due to effects of several mechanisms, including metabolic disturbances: hyperglycemia and advanced glycation end products (AGEs) generation, as well as prolonged activation of PKC. Enhanced atherosclerosis, especially of coronary arteries, and diabetic microangiopathy provoke further ischemic injury of cardiomyocytes [177].
Overall analysis of the available literature showed that ionic balance due to activity of Na+/K+-ATPase and SERCA is disturbed in cardiomyocytes during diabetic cardiomyopathy. At the level of sarcoplasmic reticulum, there is a deficiency of both Ca2+ release as well as Ca2+ uptake [178]. SERCA2a, RyR and phospholamban molecules are equally affected and although there are some discrepancies among different studies (Table 7.1), conclusions suggest disruption of Ca2+ transport and increase of intracellular Ca2+ [179–181]. Phosphorylation of phospholamban is also reduced mostly due to activity of protein phosphatase 1 [182]. The changes of SERCA2a and its regulatory proteins are associated with Na+/K+-ATPase activity decrease along with increased activity of Na+/H+ and Na+/Ca2+ exchanger as explained in previous sections. Complex cell injury ensues with deepening of metabolic disturbances and increased intracellular Ca2+. One of the consequences is activation of calpain induced apoptosis of cardiomyocytes as previously referred.
11 Conclusions
Na+/K+-ATPase and SERCA insufficiency and their interrelation with multiple intracellular functions start the cascade of events that represent almost a universal model of injury associated with heart failure of different origin [90, 183]. The analysis of Na+/K+-ATPase and SERCA is even more complex because they are direct targets for the orthodox therapy of heart failure, as well as for new therapeutical strategies [136, 152, 156]. That is why it is of essential importance to know their roles in all aspects of normal and pathological functioning of cardiomyocytes, to be able to understand future findings yet to come.
References
Therien AG, Blostein R (2000) Mechanisms of sodium pump regulation. Am J Physiol Cell Physiol 279:C541–C566
Kaplan JH (2002) Biochemistry of Na, K-ATPase. Annu Rev Biochem 71:511–535
Pedersen PA, Rasmussen JH, Jøorgensen PL (1996) Expression in high yield of pig alpha 1 beta 1 Na, K-ATPase and inactive mutants D369N and D807N in Saccharomyces cerevisiae. J Biol Chem 271:2514–2522
Ziegelhoffer A, Kjeldsen K, Bundgaard H et al (2000) Na, K-ATPase in the myocardium: molecular principles, functional and clinical aspects. Gen Physiol Biophys 19:9–47
Kawamura M, Noguchi S (1991) Possible role of the beta-subunit in the expression of the sodium pump. Soc Gen Physiol Ser 46:45–61
Beggah AT, Jaunin P, Geering K et al (1997) Role of glycosylation and disulfide bond formation in the beta subunit in the folding and functional expression of Na, K-ATPase. J Biol Chem 272:10318–10326
Morth JP, Pedersen BP, Toustrup-Jensen MS et al (2007) Crystal structure of the sodium-potassium pump. Nature 450:1043–1049
Xie Z, Wang Y, Liu G et al (1996) Similarities and differences between the properties of native and recombinant Na+/K+-ATPases. Arch Biochem Biophys 330:153–162
Juhaszova M, Blaustein MP (1997) Na+ pump low and high ouabain affinity alpha subunit isoforms are differently distributed in cells. Proc Natl Acad Sci U S A 94:1800–1805
Segall L, Daly SE, Blostein R (2001) Mechanistic basis for kinetic differences between the rat alpha 1, alpha 2, and alpha 3 isoforms of the Na, K-ATPase. J Biol Chem 276:31535–31541
Dostanic-Larson I, Lorenz JN, Van Huysse JW et al (2006) Physiological role of the alpha1- and alpha2-isoforms of the Na+-K+-ATPase and biological significance of their cardiac glycoside binding site. Am J Physiol Regul Integr Comp Physiol 290:R524–R528
Jewell EA, Lingrel JB (1991) Comparison of the substrate dependence properties of the rat Na, K-ATPase alpha 1, alpha 2, and alpha 3 isoforms expressed in HeLa cells. J Biol Chem 266:16925–16930
Woo AL, James PF, Sánchez G et al (2000) Sperm motility is dependent on a unique isoform of the Na, K-ATPase. J Biol Chem 275:20693–20699
Blanco G, Mercer RW (1998) Isozymes of the Na-K-ATPase: heterogeneity in structure, diversity in function. Am J Physiol 275(5 Pt 2):F633–F650
James PF, Grupp IL, Grupp G et al (1999) Identification of a specific role for the Na, K-ATPase alpha 2 isoform as a regulator of calcium in the heart. Mol Cell 3:555–563
Tokhtaeva E, Clifford RJ, Kaplan JH et al (2012) Subunit isoform selectivity in assembly of Na, K-ATPase alpha-beta heterodimers. J Biol Chem 287:26115–26125
Avila J, Alvarez de la Rosa D, González-Martínez LM et al (1998) Structure and expression of the human Na, K-ATPase beta 2-subunit gene. Gene 208:221–227
Appel C, Gloor S, Schmalzing G et al (1996) Expression of a Na, K-ATPase beta 3 subunit during development of the zebrafish central nervous system. J Neurosci Res 46:551–564
Schwinger RH, Bundgaard H, Müller-Ehmsen J et al (2003) The Na, K-ATPase in the failing human heart. Cardiovasc Res 57:913–920
Schwinger RH, Wang J, Frank K et al (1999) Reduced sodium pump alpha1, alpha3, and beta1-isoform protein levels and Na+, K+-ATPase activity but unchanged Na+-Ca2+ exchanger protein levels in human heart failure. Circulation 99:2105–2112
Geering K, Beggah A, Good P et al (1996) Oligomerization and maturation of Na, K-ATPase: functional interaction of the cytoplasmic NH2 terminus of the beta subunit with the alpha subunit. J Cell Biol 133:1193–1204
Efendiev R, Das-Panja K, Cinelli AR et al (2007) Localization of intracellular compartments that exchange Na, K-ATPase molecules with the plasma membrane in a hormone-dependent manner. Br J Pharmacol 151:1006–1013
Lingrel JB, Kuntzweiler T (1994) Na+, K(+)-ATPase. J Biol Chem 269:19659–19662
Jorgensen PL, Hakansson KO, Karlish SJ (2003) Structure and mechanism of Na, K-ATPase: functional sites and their interactions. Annu Rev Physiol 65:817–849
Garty H, Karlish SJ (2006) Role of FXYD proteins in ion transport. Annu Rev Physiol 68:431–459
Sweadner KJ, Rael E (2000) The FXYD gene family of small ion transport regulators or channels: cDNA sequence, protein signature sequence, and expression. Genomics 68:41–56
Chen EP, Bittner HB, Tull F et al (1997) An adult canine model of chronic pulmonary hypertension for cardiopulmonary transplantation. J Heart Lung Transplant 16:538–547
Lifshitz Y, Lindzen M, Garty H et al (2006) Functional interactions of phospholemman (PLM) (FXYD1) with Na+, K+-ATPase. Purification of alpha1/beta1/PLM complexes expressed in Pichia pastoris. J Biol Chem 281:15790–15799
Geering K (2008) Functional roles of Na, K-ATPase subunits. Curr Opin Nephrol Hypertens 17:526–532
Shinoda T, Ogawa H, Cornelius F et al (2009) Crystal structure of the sodium-potassium pump at 2.4 A resolution. Nature 459:446–450
Bagrov AY, Shapiro JI, Fedorova OV et al (2009) Endogenous cardiotonic steroids: physiology, pharmacology, and novel therapeutic targets. Pharmacol Rev 61:9–38
Lingrel JB (2010) The physiological significance of the cardiotonic steroid/ouabain-binding site of the Na, K-ATPase. Annu Rev Physiol 72:395–412
Grupp I, Im WB, Lee CO et al (1985) Relation of sodium pump inhibition to positive inotropy at low concentrations of ouabain in rat heart muscle. J Physiol 360:149–160
Shamraj OI, Grupp IL, Grupp G et al (1993) Characterisation of Na/K-ATPase, its isoforms, and the inotropic response to ouabain in isolated failing human hearts. Cardiovasc Res 27:2229–2237
Norgaard A, Bagger JP, Bjerregaard P et al (1988) Relation of left ventricular function and Na, K-pump concentration in suspected idiopathic dilated cardiomyopathy. Am J Cardiol 61:1312–1315
Liu C, Bai Y, Chen Y et al (2012) Reduction of Na/K-ATPase potentiates marinobufagenin-induced cardiac dysfunction and myocyte apoptosis. J Biol Chem 287:16390–16398
Schwinger RH, Bohm M, Erdmann E (1990) Effectiveness of cardiac glycosides in human myocardium with and without “downregulated” beta-adrenoceptors. J Cardiovasc Pharmacol 15:692–697
Obradovic M, Bjelogrlic P, Rizzo M et al (2013) Effects of obesity and estradiol on Na+/K+-ATPase and their relevance to cardiovascular diseases. J Endocrinol 218:R13–R23
Obradovic M, Stewart AJ, Pitt SJ et al (2014) In vivo effects of 17beta-estradiol on cardiac Na(+)/K(+)-ATPase expression and activity in rat heart. Mol Cell Endocrinol 388:58–68
Rathore SS, Curtis JP, Wang Y et al (2003) Association of serum digoxin concentration and outcomes in patients with heart failure. JAMA 289:871–878
Bonvalet JP (1998) Regulation of sodium transport by steroid hormones. Kidney Int Suppl 65:S49–S56
Hundal HS, Marette A, Mitsumoto Y et al (1992) Insulin induces translocation of the alpha 2 and beta 1 subunits of the Na+/K(+)-ATPase from intracellular compartments to the plasma membrane in mammalian skeletal muscle. J Biol Chem 267:5040–5043
Al-Khalili L, Yu M, Chibalin A (2003) Na+, K+-ATPase trafficking in skeletal muscle: insulin stimulates translocation of both alpha 1- and alpha 2-subunit isoforms. FEBS Lett 536:198–202
Isenovic ER, Jacobs DB, Kedees MH et al (2004) Angiotensin II regulation of the Na+ pump involves the phosphatidylinositol-3 kinase and p42/44 mitogen-activated protein kinase signaling pathways in vascular smooth muscle cells. Endocrinology 145:1151–1160
Isenovic ER, Meng Y, Jamali N et al (2004) Ang II attenuates IGF-1-stimulated Na+, K(+)-ATPase activity via PI3K/Akt pathway in vascular smooth muscle cells. Int J Mol Med 13:915–922
Bertorello AM, Katz AI (1993) Short-term regulation of renal Na-K-ATPase activity: physiological relevance and cellular mechanisms. Am J Physiol 265(6 Pt 2):F743–F755
Li WD, Reed DR, Lee JH et al (1999) Sequence variants in the 5′ flanking region of the leptin gene are associated with obesity in women. Ann Hum Genet 63(Pt 3):227–234
Sudar E, Velebit J, Gluvic Z et al (2008) Hypothetical mechanism of sodium pump regulation by estradiol under primary hypertension. J Theor Biol 251:584–592
Perino A, Ghigo A, Ferrero E et al (2011) Integrating cardiac PIP3 and cAMP signaling through a PKA anchoring function of p110γ. Mol Cell 42:84–95
Chibalin AV, Pedemonte CH, Katz AI et al (1998) Phosphorylation of the catalytic alpha-subunit constitutes a triggering signal for Na+, K+-ATPase endocytosis. J Biol Chem 273:8814–8819
Fuller W, Tulloch LB, Shattock MJ et al (2013) Regulation of the cardiac sodium pump. Cell Mol Life Sci 70:1357–1380
Boguslavskyi A, Pavlovic D, Aughton K et al (2014) Cardiac hypertrophy in mice expressing unphosphorylatable phospholemman. Cardiovasc Res 104:72–82
Galougahi KK, Liu CC, Garcia A et al (2013) Protein kinase-dependent oxidative regulation of the cardiac Na+–K+ pump: evidence from in vivo and in vitro modulation of cell signaling. J Physiol 591:2999–3015
Aperia A (2007) New roles for an old enzyme: Na, K-ATPase emerges as an interesting drug target. J Intern Med 261:44–52
Aizman O, Uhlen P, Lal M et al (2001) Ouabain, a steroid hormone that signals with slow calcium oscillations. Proc Natl Acad Sci U S A 98:13420–13424
Khodus GR, Kruusmagi M, Li J et al (2011) Calcium signaling triggered by ouabain protects the embryonic kidney from adverse developmental programming. Pediatr Nephrol 26:1479–1482
Hilfiker-Kleiner D, Landmesser U, Drexler H (2006) Molecular mechanisms in heart failure focus on cardiac hypertrophy, inflammation, angiogenesis, and apoptosis. J Am Coll Cardiol 48(Suppl A):55–66
Gheorghiade M, Ambrosy AP, Ferrandi M et al (2011) Combining SERCA2a activation and Na-K ATPase inhibition: a promising new approach to managing acute heart failure syndromes with low cardiac output. Discov Med 12(63):141–151
Sweadner KJ, Donnet C (2001) Structural similarities of Na, K-ATPase and SERCA, the Ca(2+)-ATPase of the sarcoplasmic reticulum. Biochem J 356(Pt 3):685–704
Periasamy M, Kalyanasundaram A (2007) SERCA pump isoforms: their role in calcium transport and disease. Muscle Nerve 35:430–442
Lipskaia L, Keuylian Z, Blirando K et al (2014) Expression of sarco (endo) plasmic reticulum calcium ATPase (SERCA) system in normal mouse cardiovascular tissues, heart failure and atherosclerosis. Biochim Biophys Acta 1843:2705–2718
Dally S, Corvazier E, Bredoux R et al (2010) Multiple and diverse coexpression, location, and regulation of additional SERCA2 and SERCA3 isoforms in nonfailing and failing human heart. J Mol Cell Cardiol 48:633–644
Fajardo VA, Bombardier E, Vigna C et al (2013) Co-expression of SERCA isoforms, phospholamban and sarcolipin in human skeletal muscle fibers. PLoS One 8:e84304
Chemaly ER, Bobe R, Adnot S et al (2013) Sarco (endo) plasmic reticulum calcium ATPases (SERCA) isoforms in the normal and diseased cardiac, vascular and skeletal muscle. J Cardiovasc Dis Diagn 1:113
Dhitavat J, Macfarlane S, Dode L et al (2003) Acrokeratosis verruciformis of Hopf is caused by mutation in ATP2A2: evidence that it is allelic to Darier’s disease. J Invest Dermatol 120:229–232
Dally S, Bredoux R, Corvazier E et al (2006) Ca2+-ATPases in non-failing and failing heart: evidence for a novel cardiac sarco/endoplasmic reticulum Ca2+-ATPase 2 isoform (SERCA2c). Biochem J 395:249–258
Gélébart P, Martin V, Enouf J et al (2003) Identification of a new SERCA2 splice variant regulated during monocytic differentiation. Biochem Biophys Res Commun 303:676–684
Dally S, Monceau V, Corvazier E et al (2009) Compartmentalized expression of three novel sarco/endoplasmic reticulum Ca2+ ATPase 3 isoforms including the switch to ER stress, SERCA3f, in non-failing and failing human heart. Cell Calcium 45:144–154
Bode EF, Briston SJ, Overend CL et al (2011) Changes of SERCA activity have only modest effects on sarcoplasmic reticulum Ca2+ content in rat ventricular myocytes. J Physiol (Lond) 589(Pt 19):4723–4729
Ebashi S, Lipmann F (1962) Adenosine triphosphate-linked concentration of calcium ions in a particulate fraction of rabbit muscle. J Cell Biol 14:389–400
Hasselbach W, Makinose M (1961) Imaginie calciumpumpe der “erschlaffungsgrana” des muskels und ihre abhangigkeit von der ATP-spaltung. Biochem Z 333:518–528
Shareef MA, Anwer LA, Poizat C (2014) Cardiac SERCA2A/B: therapeutic targets for heart failure. Eur J Pharmacol 724:1–8
Trafford AW, Lederer WJ, Sobie EA (2009) Keeping the beat: life without SERCA—is it possible? J Mol Cell Cardiol 47:171–173
Di Mauro D, Gaeta R, Arco A et al (2009) Distribution of costameric proteins in normal human ventricular and atrial cardiac muscle. Folia Histochem Cytobiol 47:605–608
Kierszenbaum AL (2007) Muscle Tissue. In: Kierszenbaum AL (ed) Histology and cell biology, 2nd edn. Mosby Elsevier, Philadelphia, pp 197–221
Swift F, Tovsrud N, Enger UH et al (2007) The Na+/K+-ATPase α2-isoform regulates cardiac contractility in rat cardiomyocytes. Cardiovasc Res 75:109–117
Berry RG, Despa S, Fuller W et al (2007) Differential distribution and regulation of mouse cardiac Na+/K+-ATPase alpha1 and alpha2 subunits in T-tubule and surface sarcolemmal membranes. Cardiovasc Res 73:92–100
Hu L-YR, Kontrogianni-Konstantopoulos A (2013) The kinase domains of obscurin interact with intercellular adhesion proteins. FASEB J 27:2001–2012
Misquitta CM, Mack DP, Grover AK (1999) Sarco/endoplasmic reticulum Ca2+ (SERCA)-pumps: link to heart beats and calcium waves. Cell Calcium 25(4):277–290
Strayer DS, Rubin E (2014) Cell Adaptation, Injury and Death. In: Strayer DS, Rubin E (eds) Rubin’s pathology, 7th edn. Wolters Kluwer, Philadelphia, pp 3–55
Ross MH, Pawlina W (2011) In: Ross MH, Pawlina W (eds) Histology, 6th edn. Wolters Kluwer/Lippincott Williams and Wilkins, Philadelphia, pp 310–338
Guatimosim S, Guatimosim C, Song LS (2011) Imaging calcium sparks in cardiac myocytes. Methods Mol Biol 689:205–214
Walker MA, Williams GSB, Kohl T et al (2014) Superresolution modeling of calcium release in the heart. Biophys J 107:3018–3029
Camors E, Mohler PJ, Bers DM et al (2012) Ankyrin-B reduction enhances Ca spark-mediated SR release promoting cardiac myocyte arrhythmic activity. J Mol Cell Cardiol 52:1240–1248
Inserte J, Garcia-Dorado D, Hernando V et al (2005) Calpain-mediated impairment of Na/K–ATPase activity during early reperfusion contributes to cell death after myocardial ischemia. Circ Res 97:465–473
Alto LE, Singal PK, Dhalla NS (1980) Calcium paradox. Dependence of reperfusion-induced changes on the extracellular calcium concentration. Adv Myocardiol 2:177–185
Stoica SC, Satchithananda DK, Dunning J et al (2001) Two-decade analysis of cardiac storage for transplantation. Eur J Cardiothorac Surg 20:792–798
Garcia-Dorado D, Rodriguez-Sinovas A, Ruiz-Meana M et al (2006) The end-effectors of preconditioning protection against myocardial cell death secondary to ischemia–reperfusion. Cardiovasc Res 70:274–285
Aupperle H, Garbade J, Ullman C et al (2007) Comparing the ultrastructural effects of two different cardiac preparation- and perfusion-techniques in a porcine model of extracorporeal long-term preservation. Eur J Cardiothorac Surg 31:214–221
Labudović Borović M, Ičević I, Kanački Z et al (2014) Effects of fullerenol C60(OH)24 nanoparticles on a single dose doxorubicin-induced cardiotoxicity in pigs—an ultrastructural study. Ultrastruct Pathol 38:150–163
Monceau V, Belikova Y, Kratassiouk G et al (2006) Myocyte apoptosis during acute A5 in border zone. Am J Physiol Heart Circ Physiol 291:H965–H971
Wencker D, Chandra M, Nguyen K et al (2003) A mechanistic role for cardiac myocyte apoptosis in heart failure. J Clin Invest 111(10):1497–1504
Lu L, Wu W, Yan J et al (2009) Adriamycin-induced autophagic cardiomyocyte death plays a pathogenic role in a rat model of heart failure. Int J Cardiol 134:82–90
Arbustini E, Brega A, Narula J (2008) Ultrastructural definition of apoptosis in heart failure. Heart Fail Rev 13:121–135
Drummond CA, Sayed M, Evans KL et al (2014) Reduction of Na/K-ATPase affects cardiac remodeling and increases c-kit cell abundance in partial nephrectomized mice. Am J Physiol Heart Circ Physiol 306:H1631–H1643
Chan CY, Mong MC, Liu WH et al (2014) Three pentacyclic triterpenes protect H9c2 cardiomyoblast cells against high-glucose-induced injury. Free Radic Res 48:402–411
Oubaassine R, Weckering M, Kessler L et al (2012) Insulin interacts directly with Na+/K+ATPase and protects from digoxin toxicity. Toxicology 299:1–9
Shen M, Wu RX, Zhao L, Li J et al (2012) Resveratrol attenuates ischemia/reperfusion injury in neonatal cardiomyocytes and its underlying mechanism. PLoS One 7:e51223
Belliard A, Sottejeau Y, Duan Q et al (2013) Modulation of cardiac Na+, K+ ATPase cell surface abundance by simulated ischemia-reperfusion and ouabain preconditioning. Am J Physiol Heart Circ Physiol 304:H94–H103
Li Q, Pogwizd SM, Prabhu SD et al (2014) Inhibiting Na+/K+ ATPase can impair mitochondrial energetics and induce abnormal Ca2+ cycling and automaticity in guinea pig cardiomyocytes. PLoS One 9:e93928
Sapia L, Palomeque J, Mattiazzi A et al (2010) Na+/K+-ATPase inhibition by ouabain induces CaMKII-dependent apoptosis in adult rat cardiac myocytes. J Mol Cell Cardiol 49:459–468
Liu T, Brown DA, O’Rourke B (2010) Role of mitochondrial dysfunction in cardiac glycoside toxicity. J Mol Cell Cardiol 49:728–736
Liu T, O’Rourke B (2008) Enhancing mitochondrial Ca2+ uptake in myocytes from failing hearts restores energy supply and demand matching. Circ Res 103:279–288
Maack C, Cortassa S, Aon MA et al (2006) Elevated cytosolic Na+ decreases mitochondrial Ca2+ uptake during excitation-contraction coupling and impairs energetic adaptation in cardiac myocytes. Circ Res 99:172–182
Kojic ZZ, Flögel U, Schrader J et al (2003) Endothelial NO formation does not control myocardial O2 consumption in mouse heart. Am J Physiol Heart Circ Physiol 285:H392–H397
Rosamond W, Flegal K, Furie K, Go A et al (2008) Heart disease and stroke statistics—2008 update: a report from the American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Circulation 117:25–146
Obradovic M, Sudar E, Zafirovic S et al (2015) Estradiol in vivo induces changes in cardiomyocytes size in obese rats. Angiology 66:25–35
Yamaguchi M (2014) Regulatory role of regucalcin in heart calcium signaling: insight into cardiac failure (review). Biomed Rep 2:303–308
Bergmann O, Bhardwaj RD, Bernard S et al (2009) Evidence for cardiomyocyte renewal in humans. Science 324:98–102
Buja M, Vela D (2008) Cardiomyocyte and renewal in the normal and diseased heart. Cardiovasc Pathol 17:349–374
Schoen FJ (2005) The Heart. In: Kumar V, Abbas AK, Fausto N (eds) Robbins and Cotran pathologic basis of disease, 7th edn. Elsevier Saunders, Philadelphia, pp 555–619
Skou JC (1965) Enzymatic basis for active transport of Na and K across cell membrane. Physiol Rev 45:596–617
Bers DM, Barry WH, Despa S (2003) Intracellular Na+ regulation in cardiac myocytes. Cardiovasc Res 57:897–912
Bers DM (2008) Calcium cycling and signaling in cardiac myocytes. Annu Rev Physiol 70:23–49
Smith TW (1988) Digitalis. Mechanisms of action and clinical use. N Engl J Med 318:358–365
Crambert G, Fuzesi M, Garty H et al (2002) Phospholemman (FXYD1) associates with Na, K-ATPase and regulates its transport properties. Proc Natl Acad Sci U S A 99:11476–11481
Bibert S, Roy S, Schaer D et al (2008) Phosphorylation of phospholemman (FXYD1) by protein kinases A and C modulates distinct Na, K-ATPase isozymes. J Biol Chem 283:476–486
Zahler R, Gilmore-Hebert M, Baldwin JC et al (1993) Expression of alpha isoforms of the Na, K-ATPase in human heart. Biochim Biophys Acta 1149:189–194
Schmidt TA, Allen PD, Colucci WS et al (1993) No adaptation to digitalization as evaluated by digitalis receptor (Na, K-ATPase) quantification in explanted hearts from donors without heart disease and from digitalized recipients with end-stage heart failure. Am J Cardiol 71:110–114
Despa S, Islam MA, Weber CR et al (2002) Intracellular Na+ concentration is elevated in heart failure but Na/K pump function is unchanged. Circulation 105:2543–2548
Schillinger W, Teucher N, Christians C et al (2006) High intracellular Na+ preserves myocardial function at low heart rates in isolated myocardium from failing hearts. Eur J Heart Fail 8:673–680
Baartscheer A, Schumacher CA, Borren MMGJ et al (2003) Increased Na+/H+-exchange activity is the cause of increased [Na+]i and underlies disturbed calcium handling in the rabbit pressure and volume overload heart failure model. Cardiovasc Res 57:1015–1024
Bossuyt J, Ai X, Moorman JR, Pogwizd SM et al (2005) Expression and phosphorylation of the Na-pump regulatory subunit phospholemman in heart failure. Circ Res 97:558–565
Verdonck F, Volders PGA, Vos MA et al (2003) Intracellular Na+ and altered Na+ transport mechanisms in cardiac hypertrophy and failure. J Mol Cell Cardiol 35:5–25
Swift F, Birkeland JAK, Tovsrud N et al (2008) Altered Na+/Ca2+-exchanger activity due to downregulation of Na+/K+-ATPase alpha2-isoform in heart failure. Cardiovasc Res 78:71–78
Zahler R, Gilmore-Hebert M, Sun W et al (1996) Na, K-ATPase isoform gene expression in normal and hypertrophied dog heart. Basic Res Cardiol 91:256–266
McMurray JJV, Adamopoulos S, Anker SD et al (2012) ESC guidelines for the diagnosis and treatment of acute and chronic heart failure 2012: The Task Force for the Diagnosis and Treatment of Acute and Chronic Heart Failure 2012 of the European Society of Cardiology. Developed in collaboration with the Heart Failure Association (HFA) of the ESC. Eur J Heart Fail 14:803–869
Lee CO, Abete P, Pecker M et al (1985) Strophanthidin inotropy: role of intracellular sodium ion activity and sodium-calcium exchange. J Mol Cell Cardiol 17:1043–1053
Schmidt TA (1997) Human myocardial and skeletal Na, K-ATPase in relation to digoxin treatment of heart failure. Dan Med Bull 44:499–521
Crambert G, Hasler U, Beggah AT et al (2000) Transport and pharmacological properties of nine different human Na, K-ATPase Isozymes. J Biol Chem 275:1976–1986
Müller-Ehmsen J, Juvvadi P, Thompson CB et al (2001) Ouabain and substrate affinities of human Na(+)-K(+)-ATPase alpha(1)beta(1), alpha(2)beta(1), and alpha(3)beta(1) when expressed separately in yeast cells. Am J Physiol Cell Physiol 281:C1355–C1364
Gheorghiade M, Ferguson D (1991) Digoxin. A neurohormonal modulator in heart failure? Circulation 84:2181–2186
Kirch W (2001) Pressor and vascular effects of cardiac glycosides. Eur J Clin Invest 31(Suppl 2):5–9
Schoner W, Scheiner-Bobis G (2007) Endogenous and exogenous cardiac glycosides: their roles in hypertension, salt metabolism, and cell growth. Am J Physiol Cell Physiol 293:C509–C536
Calderón-Montaño JM, Burgos-Morón E, Orta ML et al (2014) Evaluating the cancer therapeutic potential of cardiac glycosides. Biomed Res Int 2014:794930
Zheng J, Koh X, Hua F, Li G et al (2011) Cardioprotection induced by Na(+)/K(+)-ATPase activation involves extracellular signal-regulated kinase 1/2 and phosphoinositide 3-kinase/Akt pathway. Cardiovasc Res 89:51–59
Periasamy M, Huke S (2001) SERCA pump level is a critical determinant of Ca2+ homeostasis and cardiac contractility. J Mol Cell Cardiol 33:1053–1063
Elliott EB, Kelly A, Smith GL et al (2012) Isolated rabbit working heart function during progressive inhibition of myocardial SERCA activity. Circ Res 110:1618–1627
Vangheluwe P, Raeymaekers L, Dode L et al (2005) Modulating sarco(endo)plasmic reticulum Ca2+-ATPase 2 (SERCA2) activity: cell biological implications. Cell Calcium 38:291–302
Kranias EG, Hajjar RJ (2012) Modulation of cardiac contractility by the phospholamban/SERCA2a regulatome. Circ Res 110:1646–1660
James P, Inui M, Tada M, Chiesi M et al (1989) Nature and site of phospholamban regulation of the Ca2+ pump of sarcoplasmic reticulum. Nature 342:90–92
Kim HW, Steenaart NA, Ferguson DG, Kranias EG (1990) Functional reconstitution of the cardiac sarcoplasmic reticulum Ca2+-ATPase with phospholamban in phospholipid vesicles. J Biol Chem 265:1702–1709
Kranias EG (1985) Regulation of calcium transport by protein phosphotase activity associated with cardiac sarcoplasmic reticulum. J Biol Chem 260:11006–11010
MacLennan DH, Kranias EG (2003) Phospholamban: a crucial regulator of cardiac contractility. Nat Rev Mol Cell Biol 4:566–577
Champion HC (2005) Targeting protein phosphatase 1 in heart failure. Circ Res 96:708–710
Lipskaia L, Ly H, Kawase Y, Hajjar R et al (2008) Treatment of heart failure by calcium cycling gene therapy. Future Cardiol 3:413–423
Del Monte F, Hajjar RJ (2008) Intracellular devastation in heart failure. Heart Fail Rev 13:151–162
Kawase Y, Hajjar RJ (2008) The cardiac sarcoplasmic/endoplasmic reticulum calcium ATPase: a potent target for cardiovascular diseases. Nat Clin Pract Cardiovasc Med 5:554–565
Sakata S, Lebeche D, Sakata N et al (2007) Restoration of mechanical and energetic function in failing aortic-banded rat hearts by gene transfer of calcium cycling proteins. J Mol Cell Cardiol 42:852–861
Sakata S, Lebeche D, Sakata Y et al (2006) Mechanical and metabolic rescue in a type II diabetes model of cardiomyopathy by targeted gene transfer. Mol Ther 13:987–996
Del Monte F, Lebeche D, Guerrero JL et al (2004) Abrogation of ventricular arrhythmias in a model of ischemia and reperfusion by targeting myocardial calcium cycling. Proc Natl Acad Sci U S A 101:5622–5627
Ferrandi M, Barassi P, Tadini-Buoninsegni F et al (2013) Istaroxime stimulates SERCA2a and accelerates calcium cycling in heart failure by relieving phospholamban inhibition. Br J Pharmacol 169:1849–1861
Vinge LE, Raake PW, Koch WJ (2008) Gene therapy in heart failure. Circ Res 102:1458–1470
Lyon AR, Sato M, Hajjar RJ et al (2008) Gene therapy: targeting the myocardium. Heart 94:89–99
Lipskaia L, Chemaly E, Hadri L et al (2010) Sarcoplasmic reticulum Ca2+ ATPase as a therapeutic target for heart failure. Expert Opin Biol Ther 10:29–41
Cutler MJ, Wan X, Plummer BN, Liu H et al (2012) Targeted sarcoplasmic reticulum Ca2+ ATPase 2a gene delivery to restore electrical stability in the failing heart. Circulation 126:2095–2104
Oxenham H, Sharpe N (2003) Cardiovascular aging and heart failure. Eur J Heart Fail 5:427–434
Bernhard D, Laufer G (2008) The aging cardiomyocyte: a mini-review. Gerontology 54:24–31
Janczewski AM, Lakatta EG (2010) Modulation of sarcoplasmic reticulum Ca(2+) cycling in systolic and diastolic heart failure associated with aging. Heart Fail Rev 15(5):431–445
Dai DF, Santana LF, Vermulst M et al (2009) Overexpression of catalase targeted to mitochondria attenuates murine cardiac aging. Circulation 119:2789–2797
Davies CH, Davia K, Bennett JG et al (1995) Reduced contraction and altered frequency response of isolated ventricular myocytes from patients with heart failure. Circulation 92:2540–2549
Houck WV, Pan LC, Kribbs SB et al (1999) Effects of growth hormone supplementation on left ventricular morphology and myocyte function with the development of congestive heart failure. Circulation 100:2003–2009
Kinugawa S, Tsutsui H, Ide T et al (1999) Positive inotropic effect of insulin-like growth factor-1 on normal and failing cardiac myocytes. Cardiovasc Res 43:157–164
Zarain-Herzberg A (2006) Regulation of the sarcoplasmic reticulum Ca2+ − ATPase expression in the hypertrophic and failing heart. Can J Physiol Pharmacol 84:509–521
Lim C, Liao R, Varma N et al (1999) Impaired myocardial relaxation in the senescent mouse heart correlates with age-related alterations in calcium handling proteins. Biophys J 76:A309
Ren J, Li Q, Wu S et al (2007) Cardiac overexpression of antioxidant catalase attenuates aging-induced cardiomyocyte relaxation dysfunction. Mech Ageing Dev 128:276–285
Lancel S, Qin FZ, Lennon SL et al (2010) Oxidative posttranslational modifications mediate decreased SERCA activity and myocyte dysfunction in G alpha q-overexpressing mice. Circ Res 107:228–232
Adachi T, Weisbrod RM, Pimentel DR et al (2004) S-Glutathiolation by peroxynitrite activates SERCA during arterial relaxation by nitric oxide. Nat Med 10:1200–1207
Li SY, Du M, Dolence EK et al (2005) Aging induces cardiac diastolic dysfunction, oxidative stress, accumulation of advanced glycation endproducts and protein modification. Aging Cell 4:57–64
Rueckschloss U, Villmow M, Klockner U (2010) NADPH oxidase-derived superoxide impairs calcium transients and contraction in aged murine ventricular myocytes. Exp Gerontol 45:788–796
Wang MY, Zhang J, Walker SJ et al (2010) Involvement of NADPH oxidase in age-associated cardiac remodeling. J Mol Cell Cardiol 48:765–772
Kuster GM, Lancel S, Zhang JM et al (2010) Redox-mediated reciprocal regulation of SERCA and Na(+)-Ca(2+) exchanger contributes to sarcoplasmic reticulum Ca(2+) depletion in cardiac myocytes. Free Radic Biol Med 48:1182–1187
Qin F, Siwik DA, Lancel S et al (2013) Hydrogen peroxide mediated SERCA cysteine 674 oxidation contributes to impaired cardiac myocyte relaxation in senescent mouse heart. J Am Heart Assoc 2:e000184
Periasamy M, Bhupathy P, Babu GJ (2008) Regulation of sarcoplasmic reticulum Ca2+ ATPase pump expression and its relevance to cardiac muscle physiology and pathology. Cardiovasc Res 77:265–273
Feridooni HA, Dibb KM, Howlett SE (2015) How cardiomyocyte excitation, calcium release and contraction become altered with age. J Mol Cell Cardiol 83:62–72
Semb SO, Lunde PK, Holt E et al (1998) Reduced myocardial Na+, K(+)-pump capacity in congestive heart failure following myocardial infarction in rats. J Mol Cell Cardiol 30:1311–1328
Maitra A, Abbas AK (2005) The Endocrine Pancreas. In: Kumar V, Abbas AK, Fausto N (eds) Robbins and Cotran pathologic basis of disease, 7th edn. Elsevier Saunders, Philadelphia, pp 1190–1205
Dhalla NS, Rangi S, Zieroth S et al (2012) Alterations in sarcoplasmic reticulum and mitochondrial functions in diabetic cardiomyopathy. Exp Clin Cardiol 17:115–120
Xu YJ, Elimban V, Takeda S et al (1996) Cardiac sarcoplasmic reticulum function and gene expression in chronic diabetes. Cardiovasc Pathobiol 1:89–96
Zhou BQ, Hu SJ, Wang GB (2006) The analysis of ultrastructure and gene expression of sarco/endoplasmic reticulum calcium handling proteins in alloxan-induced diabetic rat myocardium. Acta Cardiol 61:21–27
Zarain-Herzberg A, Yano K, Elimban V et al (1994) Cardiac sarcoplasmic reticulum Ca2+ ATPase expression in streptozotocin-induced diabetic rat heart. Biochem Biophys Res Commun 203:113–120
Vasanji Z, Dhalla NS, Netticadan T (2004) Increased inhibition of SERCA2 by phospholamban in the type 1 diabetic heart. Mol Cell Biochem 261:245–249
Haidara MA, Assiri AS, Yassin HZ et al (2015) Heart failure models: traditional and novel therapy. Curr Vasc Pharmacol Feb 12 [Epub ahead of print]
Acknowledgements
The research activities were supported by grants No. 173033, 175005, 175015, 175061, III41002, III41022, III41027, and III45005 from the Ministry of Education, Science and Technological Development of Republic of Serbia and by the MedSAP Research Network Project, granted by the U.S. Embassy in Belgrade (Award Number: SRB100-15-GR-355).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Borović, M.M.L. et al. (2016). Myocardial Na+ K+-ATPase and SERCA: Clinical and Pathological Significance From a Cytological Perspective. In: Chakraborti, S., Dhalla, N. (eds) Regulation of Membrane Na+-K+ ATPase. Advances in Biochemistry in Health and Disease, vol 15. Springer, Cham. https://doi.org/10.1007/978-3-319-24750-2_7
Download citation
DOI: https://doi.org/10.1007/978-3-319-24750-2_7
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-24748-9
Online ISBN: 978-3-319-24750-2
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)