Abstract
Osteoarthritis (OA) is a common disease defined as degenerative arthritis or joint disease involving degradation of articular cartilage and subchondral bone, and it could potentially affect the quality of life of elderly populations worldwide. The management of OA remains challenging and controversial. Although there are several clinical options for the treatment of OA, regeneration of the damaged articular cartilage has proven difficult due to the limited healing capacity. With the advancements in tissue engineering approaches including cell-based technologies and development of biomaterial scaffolds over the past decade, new therapeutic options for patients with osteochondral lesions potentially exist. This chapter will focus on the feasibility of tissue-engineered biomaterial scaffolds, which can mimic the native osteochondral complex, for osteochondral repair and highlight the recent development of these techniques toward tissue regeneration, which will contribute to osteochondral repair for the patients who are involved with an incurable OA treated by traditional procedures. Moreover, basic anatomy, strategy for osteochondral repair, and the design and fabrication methods of scaffolds as well as the choice of cells, growth factor, and materials will be discussed. Specifically, we focus on the latest preclinical animal studies using large animals and clinical trials with high clinical relevance. Accordingly, this will contribute to an understanding of the latest trends in osteochondral repair and future application of such clinical therapies in patients with OA.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
FormalPara Learning OutcomesAfter you have studied this chapter, you will have an understanding of (1) the basic anatomy of osteochondral tissue; (2) the strategy for osteochondral repair; (3) the design and fabrication methods of scaffolds; (4) the choice of cells, growth factor, and materials; and (5) the latest preclinical animal studies and clinical trials.
FormalPara Terminology- Osteoarthritis:
-
Degenerative arthritis or joint disease involving degradation of articular cartilage and subchondral bone.
- Scaffold:
-
An artificial material supporting 3D tissue formation, in which cells are usually seeded.
- Growth factor:
-
A protein or hormone stimulating cellular growth, proliferation, and cellular differentiation.
- Chondrocyte:
-
Cells found in cartilage.
- Mesenchymal stem cell:
-
Multipotent stromal cells that can differentiate into a variety of cell types, including osteoblasts (bone cells), chondrocytes (cartilage cells), and adipocytes (fat cells).
- Induced pluripotent stem (iPS) cell:
-
A type of pluripotent stem cell that can be generated directly from adult cells. Typically, adult cells can be converted into pluripotent stem cells by the introduction of four specific transcription factors.
- Chondrogenesis:
-
The process by which the cartilage is developed.
- Osteogenesis:
-
The process by which the bone is developed.
Is a biological joint replacement available in the near future?
A 73-year-old female patient came in the outpatient clinic with symptoms of knee pain. The X-ray of this patient showed a severe OA (Fig. 23.3b). As conservative treatments including physiotherapy and pharmacologic therapies were failed, a total knee replacement was performed for the patient. At 6 months postoperatively, the patient was pain-free and has acquired normal gait. Today, the joint replacement should be a gold standard treatment for the patients with terminal OA, but there exist several risks and complications including infection, loss of motion, and revision surgeries [90, 98]. Also, patients may lose their normal joint movements and structures such as the cartilage, bone, and ligaments after the joint replacement. With the recent advancements in tissue engineering approaches, there have been many promising scaffolds developed for osteochondral repair, some of which are already at the stage of clinical trials [29, 55]. Therefore, the application of these technologies to osteochondral lesions could be expected in the near future, and it will contribute to restore normal structures and functions by biologically replacing the damaged tissues.
Introduction
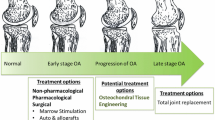
Osteoarthritis (OA) is a common disease causing joint pain, joint deformity, and functional disability. Globally, approximately 250 million people are potentially affected with OA [120]. Overall, OA affects 13.9 % of adults aged 25 years and older and 33.6 % of those 65 and older in the United States, consequently affecting the quality of life of elderly populations [26, 32, 58, 66]. Also, OA is associated with an extremely high economic burden. The costs due to hospital expenditures of total knee and hip joint replacements were estimated at $28.5 billion and $13.7 billion, respectively [78]. Current treatment strategies can be divided into nonsurgical (conservative) and surgical therapies according to the severity of OA [125–127]. In the early stage of OA, pharmacologic and/or physical therapies as conservative treatments are typically selected for the purpose of reducing pain and, in some cases, attempting to delay the progressive structural deterioration in affected joints. Surgical therapies such as joint replacement and osteotomy are available for patients who fail to respond to more conservative measures. These treatments are well established and effective for reducing pain and improving quality of life. Regardless of these therapeutic options, however, there is no method available that facilitates complete healing of the articular cartilage [6, 7, 15, 44, 47, 108]. Recently, several biological approaches, such as the use of tissue-engineered materials, have been tested to overcome such potential problems (Fig. 23.1). This chapter will focus on the feasibility of tissue-engineered materials in osteochondral repair and highlight recent advances in the biological repair of osteochondral lesions.
Anatomy of the Cartilage, Subchondral Bone, and Their Interface
The osteochondral complex consists of both the articular cartilage and underlying subchondral bone. Biochemically, cartilage tissue is largely comprised of water, chondrocytes, type II collagen, and proteoglycan [50, 81, 122]. The cartilage can be differentiated into four distinct zones: the superficial, middle, deep, and calcified cartilage zones (Fig. 23.2) [65]. Each zone is defined by a particular composition and organization of cells and extracellular matrix (ECM) molecules. The differential proportions in ECM composition influence the mechanical properties of each zone of the cartilage. For example, the superficial zone is strong in tension along the alignment of its collagen fibrils, thereby assisting in the resistance of shear forces at the surface. By comparison, the deep zone has more compressive strain.
Subchondral bone is a complex tissue consisting of water, collagen type I, and hydroxyapatite, with the two latter components providing the tissue’s stiffness and compressive strength [9, 81, 122]. The compressive modulus of subchondral bone is higher than that of the cartilage. The different morphological compositions and mechanical properties of subchondral bone and cartilage indicate the complexity of the tissue interface.
The osteochondral interface is described by the interaction of calcified cartilage and the underlying subchondral bone [16]. Structurally, collagen fibers extend from the deep zone to calcified cartilage through a wavy tidemark, which enables the dispersal of force through the vertical orientation of collagen fibrils [83]. However, despite the fact that calcified cartilage is mineralized tissue, its mechanical strength is lower than that of the subchondral bone [72]. Calcified cartilage is interdigitated with subchondral bone, but fibers do not extend across the zone into the bone [23, 83]. The wavy tidemark and vertically oriented fibers at the tidemark, as well as interdigitations present at the interface, may allow for reducing stress concentrations, as well as better integration with the underlying subchondral bone [81, 83].
Characteristic of Osteoarthritic Joint
An osteoarthritic joint is characterized by degenerative changes, such as articular cartilage loss, subchondral bone thickening, and osteophyte formation [12, 39, 64, 77, 96]. The primary morphologic changes include thinning, fissuring, and fragmentation of articular cartilage. With the progression of the disease comes a continuous loss of articular cartilage, accompanied with the decrease of collagen type II and aggrecan [31, 76], leading to exposure of subchondral bone. Secondary changes are frequently seen in the underlying bone, such as sclerosis, cystic change, and new bone formation (Fig. 23.3a, b). These changes are considered to be triggered by a multitude of factors, including aging, trauma, obesity, mechanical overload, congenital disorder, and infection, which do not heal spontaneously once damaged.
Strategy for Osteochondral Repair
For an ideal repair of osteochondral lesions, it is important to regenerate subchondral bone and to facilitate zonal restoration of the cartilage and subchondral bone, layer by layer, mimicking the natural articular structure [37, 46, 53, 74, 85, 86, 102]. As a strategy to regenerate these structures in a layer-by-layer fashion, biphasic or triphasic constructs have been developed due to both mechanical and biological reasons, including the acquisition of initial mechanical strength, mimicking a natural articulate structure, a uniform tidemark at the osteochondral junction, and an integration of the biphasic implant with host tissue to sustain biological function (Fig. 23.4a–d) [2, 4, 18, 36, 43, 47, 69, 82, 84, 107, 110]. For satisfying the biological requirements, an osteochondral implant should ideally have a rigid osseous layer to support the overlying cartilage and integrate with the native bone and a chondral layer to allow the seeding and proliferation of chondrocytes or mesenchymal stem cells (MSCs) and subsequent deposition of cartilaginous ECM. Also, for the successful osteochondral repair, the integration between the implant and host tissue should be one of crucial factors. Our previous study showed that the tissue integration to native surrounding osteochondral tissue after the implantation of biphasic construct could influence the quality and maturation of repair tissue [109]. In case of the failure of integration with the adjacent host cartilage, the repair tissue might evolve into a pathological condition such as OA due to mechanically unstable condition, which potentially raises concerns regarding the long-term durability of the repair tissue.
Design and Fabrication of Biomaterial Scaffold
In general, osteochondral tissue engineering strategies can be categorized into monophasic (Fig. 23.4b) and biphasic (Fig. 23.4c) depending on the biological and biomechanical characteristics of the scaffold. As mentioned above, a successful tissue engineering approach for osteochondral repair involves the design of a biphasic scaffold with the potential to regenerate both the cartilage and subchondral bone. The fabrication of the majority of scaffolds is performed through independent processes, whereby different scaffolds for the two sides are created and then combined, or via a simultaneous process through which a single scaffold is created and cultured simultaneously for both sides [67, 81]. A biphasic construct developed independently allows the cultivation of both chondrogenic and osteogenic cells in separate media and environmental conditions. However, these constructs must be hybridized into a single composite graft by connecting the two layers together. The potential disadvantage of this approach might be the difficulty in achieving a secure biological and mechanical integration between the two layers [67]. On the other hand, when the two layers are hybridized prior to culture, a complicated system will be required to promote osteo- and chondral differentiation separately in each layer. Due to the difficulty of two different cell cultures simultaneously, such pre-developed biphasic constructs are mainly used as a cell-free scaffold [53].
Some research groups have raised the importance of an intermediate layer between the cartilage and subchondral bone layers to represent the tidemark or calcified cartilage; triphasic scaffolds were therefore developed (Fig. 23.4d) [53, 69]. However, the intermediate layer has unique osteochondral characteristics owing to the infiltration of the blood vessels, and thus, it may be difficult to mimic the unique structure with currently available biomaterial technologies. In fact, the superiority of triphasic scaffolds over biphasic for osteochondral repair has not yet been demonstrated and requires further investigation.
Most scaffolds have the pore structures inside, which would affect the regulation of cell invasion, vascularization, and tissue maturation [105]. The pore size and porosity should be controlled suitable for tissue engineering in the fabrication process of porous scaffolds. Regarding the effects of pore size on osteogenesis, the scaffold structure composed of porosity higher than 50 % and pores larger than 300 μm is recommended to achieve direct osteogenesis with enhanced vascularization [48]. On the contrary, the scaffold with smaller pores have been suggested for favorable chondrogenesis on 90–120 μm pores, in which MSCs proliferate and promote chondrogenesis in the scaffold [51].
Recent advances in computer-aided tissue engineering including three-dimensional (3D) printing technique enable the fabrication of multifunctional scaffolds that meet the microstructural, mechanical, and nutritional requirements based on optimized models [59, 63]. Moreover, these techniques will be expected to generate the custom-shaped engineered grafts from clinical imaging data with the use of CT or MRI, which fit the specific defect [79]. Therefore, the bioprinting technology should be a powerful tool for building tissues at cellular and organ levels.
Choice of Cells and Growth Factors
The most direct cell source may be the biopsy specimens taken from the patients, from which mature osteoblasts and chondrocytes may be obtained. However, as the number of cells obtained is usually limited, it is typically not enough to allow seeding onto the scaffolds. Also, expansion of primary cells may result in a loss of differentiation capacity; for example, the expansion of articular chondrocytes can lead to dedifferentiation into fibroblast [13, 104, 115]. To overcome such potential problems with respect to dedifferentiation, three-dimensional (3D) culture can be used to retain the cellular phenotype and avoid dedifferentiation [116]. The most common method is the use of various scaffolds to produce a 3D culture condition [68, 128] and may be combined with the supplementation of growth factors [22], the use of bioreactor [35], the mechanical stimulation of the cells [33, 49], and the use of low oxygen tension [57] during cultivation. Also, even if chondrocytes lose their differentiated phenotype, dedifferentiated chondrocytes can regain their differentiated phenotype through the redifferentiation process of cultivation in a 3D scaffold combined with growth factors [3, 60].
As an additional option, stem cells may represent promising alternatives [113]. Specifically, mesenchymal stem cells (MSCs) have the capability to differentiate into a variety of connective tissue cell types, including the bone, cartilage, tendon, muscle, and adipose tissue [27, 108]. These cells may be isolated from various tissues, such as bone marrow, skeletal muscle, synovial membrane, adipose tissue, and umbilical cord blood [6, 7, 27, 52, 71, 99]. Moreover, allogeneic MSCs [25, 108] or induced pluripotent stem (iPS) cells [114, 119] may also be considered. However, there have not been much evidence using these cells forthcoming in terms of preclinical and clinical safety, and thus, further studies with such cells are likely necessary.
In addition, the use of a growth factor or its cocktail (combination), including insulin-like growth factor 1 (IGF-1), transforming growth factor beta 1 (TGF-β1), fibroblast growth factor 2 (FGF-2), and bone morphogenetic proteins (BMP-2, BMP-7), may support tissue maturation for the cartilage [10, 41, 87, 111]. Similar to the cartilage, the bone also possesses a large variety of growth factors that are involved in the regenerative process, including TGF-β; BMP-2, 4, 6, and 7; IGF-1 and 2; and platelet-derived growth factor (PDGF) [91, 97, 100].
On the other hand, some researchers have tested an acellular approach using a scaffold alone [29, 53]. Considering the time and cost-effectiveness, as well as safety issues associated with cell culture, this approach could represent a reasonable strategy in tissue engineering. Scaffolds should be developed to meet requirements such as the recruitment of enough tissue progenitor cells from the host tissue.
Choice of Materials
Several methods have been proposed to develop biphasic scaffolds with the hybridization of two distinct biomaterials, each of which being adequate to integrate with the respective surrounding tissue [67]. Many specific material types have been developed for both cartilage and bone regeneration, which are typically made of biocompatible and biodegradable polymers. For the cartilage layer, natural or synthetic polymer-based scaffolds are commonly used. More recently, scaffold-free implants have been developed and the potential feasibility tested. On the other hand, for a scaffold of the subchondral bone layer, it is important to choose materials with initial mechanical strength, good bone ingrowth, and integration of native surrounding bone. Ceramics, glasses, and metallic materials are commonly used. Also, natural or synthetic polymers, similar to cartilage layer, could be used alone or combined with ceramics [4, 18, 28, 54, 123, 124].
Natural Polymers
The materials of naturally derived polymers could provide a naturally occurring environment for the cells and tissues and thereby potentially facilitate cell proliferation and differentiation [42, 118]. Moreover, natural polymers usually contain specific molecular domains that can support and guide cells at various stages of their development [67, 81]; thus, biological interaction of the scaffold with the host tissue can be enhanced. However, they are, in general, biomechanically weak and less stiff than other materials [81]. As a source of materials, collagen, gelatin, glycosaminoglycan, chitosan, starch, hyaluronic acid, alginate, and bacterial-sourced polymers (hydroxyalkanoates) are commonly used.
Synthetic Polymers
Biodegradable synthetic polymers offer several advantages over other materials for developing scaffolds in tissue engineering. The main advantages are being able to control mechanical properties (i.e., strength and stiffness) and degradation speed [38]. Synthetic polymers are also attractive because they can be fabricated into various shapes with a desired pore according to the speed of cell migration or tissue ingrowth [30]. Moreover, the progression of current techniques such as electrospinning methods and the 3D printer has enabled the simple design and fabrication of scaffolds, which mimic the original tissue structure [61–63]. On the other hand, synthetic polymers have limitations in bioactivity due to their hydrophobic surface not supporting cell attachment and proliferation [14, 89, 101, 106]. Surface treatment with chondroitin sulfate [17], silicate [20], and alkaline [89] could increase hydrophilicity and provide a suitable scaffold for tissue engineering. Also, these polymers, incorporated with growth factors such as TGF-β and BMP, would be helpful and convenient to support cell proliferation and differentiation, stimulating the repair of damaged tissue [93, 94]. As a source of biodegradable synthetic polymers, polyglycolic acid (PGA), poly(D,L-lactic-co-glycolic acid) (PLGA), poly-L-lactic acid (PLLA), polycaprolactone (PCL), and polyethylene glycol (PEG) have been commonly used.
Scaffold-Free Biomaterials
Polymer-based scaffolds have been reported to contribute to good osteochondral repair in vivo [2, 4, 43, 69, 84, 107]. Despite this, there remain several concerns associated with the long-term safety of these constructs due to the involvement of chemical or animal-derived materials. To overcome such potential problems, we have developed a scaffold-free three-dimensional tissue-engineered construct (TEC) composed of MSCs derived from the synovium and ECMs synthesized by the cells (Fig. 23.5a) [6, 7]. The feasibility of the resultant TEC to facilitate cartilage repair was demonstrated in a preclinical large animal model [5, 6, 108], and we have now proceeded clinical studies under the auspices of an approved first-in-man protocol [80]. These TECs are developed without an artificial scaffold, and thus, their implantation could eliminate or minimize the risk of potential side effects induced by extrinsic chemical or biological materials. Furthermore, such TEC are highly adherent to cartilage matrix and secure integration of the TEC until adjacent cartilage tissue is observed following implantation. Therefore, combined constructs of TEC and several materials for the subchondral bone layer may effectively repair an osteochondral lesion with zonal restoration, and TEC could be one of the strong candidates for a cartilage bioimplant. In our animal study, we have demonstrated that the combined bioimplant of TEC and ceramic-based artificial bone significantly accelerated and improved osteochondral repair (Fig. 23.5b, c) [109].
(a) In vitro generated scaffold-free tissue-engineered construct (TEC) composed of MSCs derived from the synovium and ECMs synthesized by the cells. (b, c) Toluidine blue staining of repair tissues in osteochondral defect (b) untreated or (c) treated with the biphasic implant made of TEC and ceramic artificial bone using a rabbit model. The untreated repair tissue showed insufficient cartilage and subchondral bone repair with fibrous or fibrocartilaginous repair at 2 months postoperatively. The repair tissue treated with the implant showed complete osteochondral repair at 2 months postoperatively. Bar = 1 mm
Ceramics and Glasses
Ceramics, such as hydroxyapatite (HA), or other calcium phosphates, such as tricalcium phosphate (TCP), and bioactive glasses, such as Bioglass®, are widely used for bone tissue engineering [45, 70, 117, 121]. These materials promote the formation of a bone-like tissue and enhance integration of the scaffold to the host tissue due to excellent osteoconductivity and osteoinductivity. Also, inclusion of growth factors in the scaffolds may be an interesting concept to explore and contribute to the maturation of bone tissue. Notably, the inclusion of BMP-2 in an HA-based scaffold was reported to promote subchondral bone as well as cartilage repair [117]. On the other hand, these scaffolds have low structural integrity being brittle and unsuitable for applications under mechanical stress, although they exhibit suitable stiffness [81]. The degradation behavior of these scaffolds can be controlled by changes in the porous structures, which can be tailored in terms of their degradation kinetics appropriate for bone tissue engineering. It is also well known that increasing porosity impairs further the mechanical properties of bioceramic scaffolds. This problem can be solved by modifying any porous scaffolds with infiltration or coating by biodegradable polymers [19, 73, 92].
Metallic Materials
Metals are widely used in orthopedic implants such as titanium, titanium alloys, stainless steels, and cobalt-chromium alloy. As an application of osteochondral bone repair, metallic materials withhold the capability of withstanding mechanical loading when used in the subchondral bone layer. On the other hand, the lack of degradation over time and the possibility of wear particle release or corrosion are disadvantages. As one of the examples, porous tantalum was reported to induce subchondral bone growth and showed integration to adjacent host bone in an in vivo rabbit study [11].
Preclinical Study and Clinical Trial
There have been many therapeutic procedures investigated to biologically repair damaged cartilage, some of which are already at the stage of clinical application. On the contrary, considering the higher incidence of OA, which involves subchondral bone pathology, by comparison to isolated chondral injury [8, 24, 26, 40, 88, 126], there is an urgent need to develop novel therapeutic methods for osteochondral repair with clinical relevance. In this regard, the number of animal experiments and clinical trials to treat osteochondral lesions has been recently increased. We focus on the latest preclinical animal studies using large animal and clinical studies.
At first, in order to evaluate the feasibility and safety of new implants with clinical relevance, the selection of appropriate animal models is important. Due to the differences in matrix structure and composition, as well as in the natural osteochondral healing response and technical difficulty in creating the lesions of consistent size and location, the use of small animals such as rabbits, rats, and mice may not be appropriate [1, 21, 95]. Rather, in consideration of clinical relevance, it is preferable to utilize larger animal models, such as pigs, sheep, goats, and horses. Marquass et al. used an MSC-seeded combined implant with a collagen I hydrogel and β-TCP in an ovine osteochondral defect model and showed comparable repair quality to osteochondral autografts in terms of histology and biomechanical testing [69]. Miot et al. prepared engineered cartilage, which was generated from autologous chondrocytes cultured in hyaluronic acid scaffolds of different pre-culture periods, and implanted the engineered cartilage above the hydroxyapatite/hyaluronic acid sponges into goat osteochondral defects. They concluded that 2 weeks pre-culture of engineered cartilage achieved a suitable compromise between tissue maturity and structural/integrative properties of the repair tissue. These data demonstrate that the stage of development of engineered cartilage is an important parameter to be considered in designing cartilage repair strategies [75]. Kon et al. used an aragonite/hyaluronate biphasic scaffold for osteochondral defects in a goat model and showed that mechanical modification with drilled channels in the cartilage phase and impregnation of HA within the coral pores enhanced the scaffold’s cartilage regenerative potential [56]. Schleicher et al. compared two biphasic scaffolds of either hydroxylapatite/collagen or allogenous sterilized bone/collagen and tested their integration in a sheep model. They showed that the latter scaffold proved to be stable and sufficiently integrated in the short term [103]. Sosio et al. treated the osteochondral lesions with a biphasic scaffold made of collagen type I and hydroxyapatite in a pig model, in which they compared the scaffold seeded with autologous chondrocyte with the unseeded scaffold [112]. Surprisingly, they showed that the unseeded scaffold exhibited better macroscopic and histological results than the cell-seeded scaffold. Kon et al. developed an acellular three-gradient multilayer scaffold made of collagen type I and nanoparticles of hydroxyapatite and tested the scaffold with or without autologous chondrocytes in sheep osteochondral defect model. They concluded that the scaffold contributed to the process of the bone and hyaline-like cartilage regeneration, regardless of the use of chondrocytes [54]. Also, they treated 27 patients with chondral or osteochondral lesions using an acellular scaffold [34, 53, 55] and demonstrated the safety and potential clinical benefit of the graded biomimetic osteochondral scaffold in promoting the bone and cartilage tissue with good clinical and magnetic resonance imaging results until the 5-year follow-up. Dhollander et al. treated 27 for cartilage lesions with an acellular osteochondral plug, which is composed of polylactide-co-glycolide copolymer, calcium sulfate, polyglycolide fibers, and surfactant (TruFit plug; Smith and Nephew, Andover, MA) [29]. In this clinical pilot study, a modest clinical improvement became apparent at 12 months of follow-up. Also, MRI data showed no deterioration of the repair tissue. However, 20 % of the patients had persistent clinical symptoms after surgery and had an additional surgery such as removal of the osteochondral plug remnants. The two latter studies were Level IV study, and further studies, which would be compared with conventional treatment such as bone marrow stimulation and osteochondral transplantation, are necessary. Also, in contrast with cell-free scaffolds, no clinical trial using cell-seeded scaffold has been reported, and these studies should be expected in the near future.
Future Directions
The management of OA remains challenging and controversial. Considering the steady progression of tissue engineering and cell-based technologies over the past decade, we may have new therapeutic options for osteochondral repair in clinical practice. In this chapter, we have focused on biomaterial scaffold for osteochondral repair, including the concept, scaffold fabrication, in addition to the selection of cells and materials. There have been many promising scaffolds developed, some of which contribute to good osteochondral repair in vivo. Moreover, some of them are already at the stage of preclinical, large animal studies, as well as clinical trials. In addition, the recent work has been focused on not only investigating the effectiveness of materials or cells but also applying several new concepts and techniques such as mechanical [56], microstructural [30], and local microenvironment modification [94] for the design and fabrication of scaffolds. Therefore, the application of additional new implants to osteochondral lesions could be expected in the near future. On the other hand, the most suitable biomaterials for the cartilage or subchondral bone layers have not been fully investigated, while there are many biomaterials available for osteochondral repair. Therefore, the comparison of these materials should be performed to ultimately determine the ideal material. Further studies will be needed and should be conducted in a methodologically rigorous fashion.
References
Ahern BJ, Parvizi J, Boston R, Schaer TP (2009) Preclinical animal models in single site cartilage defect testing: a systematic review. Osteoarthritis Cartilage 17(6):705–713. doi:10.1016/j.joca.2008.11.008, S1063-4584(08)00353-1 [pii]
Ahn JH, Lee TH, Oh JS, Kim SY, Kim HJ, Park IK, Choi BS, Im GI (2009) Novel hyaluronate-atelocollagen/beta-TCP-hydroxyapatite biphasic scaffold for the repair of osteochondral defects in rabbits. Tissue Eng Part A 15(9):2595–2604. doi:10.1089/ten.TEA.2008.0511, 10.1089/ten.TEA.2008.0511 [pii]
Albrecht C, Schlegel W, Bartko P, Eckl P, Jagersberger T, Vecsei V, Marlovits S (2010) Changes in the endogenous BMP expression during redifferentiation of chondrocytes in 3D cultures. Int J Mol Med 26(3):317–323
Alhadlaq A, Mao JJ (2005) Tissue-engineered osteochondral constructs in the shape of an articular condyle. J Bone Joint Surg Am 87(5):936–944. doi:10.2106/JBJS.D.02104, 87/5/936 [pii]
Ando W, Fujie H, Moriguchi Y, Nansai R, Shimomura K, Hart DA, Yoshikawa H, Nakamura N (2012) Detection of abnormalities in the superficial zone of cartilage repaired using a tissue engineered construct derived from synovial stem cells. Eur Cell Mater 24:292–307. doi:vol024a21 [pii]
Ando W, Tateishi K, Hart DA, Katakai D, Tanaka Y, Nakata K, Hashimoto J, Fujie H, Shino K, Yoshikawa H, Nakamura N (2007) Cartilage repair using an in vitro generated scaffold-free tissue-engineered construct derived from porcine synovial mesenchymal stem cells. Biomaterials 28(36):5462–5470. doi:10.1016/j.biomaterials.2007.08.030, S0142-9612(07)00672-2 [pii]
Ando W, Tateishi K, Katakai D, Hart DA, Higuchi C, Nakata K, Hashimoto J, Fujie H, Shino K, Yoshikawa H, Nakamura N (2008) In vitro generation of a scaffold-free tissue-engineered construct (TEC) derived from human synovial mesenchymal stem cells: biological and mechanical properties and further chondrogenic potential. Tissue Eng Part A 14(12):2041–2049. doi:10.1089/ten.tea.2008.0015
Aroen A, Loken S, Heir S, Alvik E, Ekeland A, Granlund OG, Engebretsen L (2004) Articular cartilage lesions in 993 consecutive knee arthroscopies. Am J Sports Med 32(1):211–215
Arvidson K, Abdallah BM, Applegate LA, Baldini N, Cenni E, Gomez-Barrena E, Granchi D, Kassem M, Konttinen YT, Mustafa K, Pioletti DP, Sillat T, Finne-Wistrand A (2011) Bone regeneration and stem cells. J Cell Mol Med 15(4):718–746. doi:10.1111/j.1582-4934.2010.01224.x
Babensee JE, McIntire LV, Mikos AG (2000) Growth factor delivery for tissue engineering. Pharm Res 17(5):497–504
Bal BS, Rahaman MN, Jayabalan P, Kuroki K, Cockrell MK, Yao JQ, Cook JL (2010) In vivo outcomes of tissue-engineered osteochondral grafts. J Biomed Mater Res B Appl Biomater 93(1):164–174. doi:10.1002/jbm.b.31571
Barr RJ, Gregory JS, Reid DM, Aspden RM, Yoshida K, Hosie G, Silman AJ, Alesci S, Macfarlane GJ (2012) Predicting OA progression to total hip replacement: can we do better than risk factors alone using active shape modelling as an imaging biomarker? Rheumatology (Oxford) 51(3):562–570. doi:10.1093/rheumatology/ker382, ker382 [pii]
Benya PD, Shaffer JD (1982) Dedifferentiated chondrocytes reexpress the differentiated collagen phenotype when cultured in agarose gels. Cell 30(1):215–224. doi:0092-8674(82)90027-7 [pii]
Bhattarai SR, Bhattarai N, Viswanathamurthi P, Yi HK, Hwang PH, Kim HY (2006) Hydrophilic nanofibrous structure of polylactide; fabrication and cell affinity. J Biomed Mater Res A 78(2):247–257. doi:10.1002/jbm.a.30695
Brittberg M, Lindahl A, Nilsson A, Ohlsson C, Isaksson O, Peterson L (1994) Treatment of deep cartilage defects in the knee with autologous chondrocyte transplantation. N Engl J Med 331(14):889–895. doi:10.1056/NEJM199410063311401
Castro NJ, Hacking SA, Zhang LG (2012) Recent progress in interfacial tissue engineering approaches for osteochondral defects. Ann Biomed Eng 40(8):1628–1640. doi:10.1007/s10439-012-0605-5
Chang KY, Cheng LW, Ho GH, Huang YP, Lee YD (2009) Fabrication and characterization of poly(gamma-glutamic acid)-graft-chondroitin sulfate/polycaprolactone porous scaffolds for cartilage tissue engineering. Acta Biomater 5(6):1937–1947. doi:10.1016/j.actbio.2009.02.002, S1742-7061(09)00063-4 [pii]
Chen J, Chen H, Li P, Diao H, Zhu S, Dong L, Wang R, Guo T, Zhao J, Zhang J (2011) Simultaneous regeneration of articular cartilage and subchondral bone in vivo using MSCs induced by a spatially controlled gene delivery system in bilayered integrated scaffolds. Biomaterials 32(21):4793–4805. doi:10.1016/j.biomaterials.2011.03.041, S0142-9612(11)00310-3 [pii]
Chen QZ, Boccaccini AR (2006) Poly(D,L-lactic acid) coated 45S5 bioglass-based scaffolds: processing and characterization. J Biomed Mater Res A 77(3):445–457. doi:10.1002/jbm.a.30636
Chouzouri G, Xanthos M (2007) In vitro bioactivity and degradation of polycaprolactone composites containing silicate fillers. Acta Biomater 3(5):745–756. doi:10.1016/j.actbio.2007.01.005, S1742-7061(07)00016-5 [pii]
Chu CR, Szczodry M, Bruno S (2010) Animal models for cartilage regeneration and repair. Tissue Eng Part B Rev 16(1):105–115. doi:10.1089/ten.TEB.2009.0452
Chubinskaya S, Segalite D, Pikovsky D, Hakimiyan AA, Rueger DC (2008) Effects induced by BMPS in cultures of human articular chondrocytes: comparative studies. Growth Factors 26(5):275–283. doi:10.1080/08977190802291733, 795281645 [pii]
Clark JM, Huber JD (1990) The structure of the human subchondral plate. J Bone Joint Surg Br 72(5):866–873
Curl WW, Krome J, Gordon ES, Rushing J, Smith BP, Poehling GG (1997) Cartilage injuries: a review of 31,516 knee arthroscopies. Arthroscopy 13(4):456–460
Dashtdar H, Rothan HA, Tay T, Ahmad RE, Ali R, Tay LX, Chong PP, Kamarul T (2011) A preliminary study comparing the use of allogenic chondrogenic pre-differentiated and undifferentiated mesenchymal stem cells for the repair of full thickness articular cartilage defects in rabbits. J Orthop Res 29(9):1336–1342. doi:10.1002/jor.21413
Dawson J, Linsell L, Zondervan K, Rose P, Randall T, Carr A, Fitzpatrick R (2004) Epidemiology of hip and knee pain and its impact on overall health status in older adults. Rheumatology (Oxford) 43(4):497–504. doi:10.1093/rheumatology/keh086, keh086 [pii]
De Bari C, Dell’Accio F, Tylzanowski P, Luyten FP (2001) Multipotent mesenchymal stem cells from adult human synovial membrane. Arthritis Rheum 44(8):1928–1942. doi:10.1002/1529-0131(200108)44:8, <1928::AID-ART331>3.0.CO;2-P
Deng T, Lv J, Pang J, Liu B, Ke J (2012) Construction of tissue-engineered osteochondral composites and repair of large joint defects in rabbit. J Tissue Eng Regen Med. doi:10.1002/term.1556 [doi]
Dhollander AA, Liekens K, Almqvist KF, Verdonk R, Lambrecht S, Elewaut D, Verbruggen G, Verdonk PC (2012) A pilot study of the use of an osteochondral scaffold plug for cartilage repair in the knee and how to deal with early clinical failures. Arthroscopy 28(2):225–233. doi:10.1016/j.arthro.2011.07.017, S0749-8063(11)00994-7 [pii]
Duan P, Pan Z, Cao L, He Y, Wang H, Qu Z, Dong J, Ding J (2013) The effects of pore size in bilayered poly(lactide-co-glycolide) scaffolds on restoring osteochondral defects in rabbits. J Biomed Mater Res A. doi:10.1002/jbm.a.34683 [doi]
Duan Y, Hao D, Li M, Wu Z, Li D, Yang X, Qiu G (2012) Increased synovial fluid visfatin is positively linked to cartilage degradation biomarkers in osteoarthritis. Rheumatol Int 32(4):985–990. doi:10.1007/s00296-010-1731-8
Dunlop DD, Manheim LM, Song J, Chang RW (2001) Arthritis prevalence and activity limitations in older adults. Arthritis Rheum 44(1):212–221. doi:10.1002/1529-0131(200101)44:1, <212::AID-ANR28>3.0.CO;2-Q
El-Ayoubi R, DeGrandpre C, DiRaddo R, Yousefi AM, Lavigne P (2011) Design and dynamic culture of 3D-scaffolds for cartilage tissue engineering. J Biomater Appl 25(5):429–444. doi:10.1177/0885328209355332, 0885328209355332 [pii]
Filardo G, Kon E, Di Martino A, Busacca M, Altadonna G, Marcacci M (2013) Treatment of knee osteochondritis dissecans with a cell-free biomimetic osteochondral scaffold: clinical and imaging evaluation at 2-year follow-up. Am J Sports Med 41(8):1786–1793. doi:10.1177/0363546513490658, 0363546513490658 [pii]
Forsey RW, Tare R, Oreffo RO, Chaudhuri JB (2012) Perfusion bioreactor studies of chondrocyte growth in alginate-chitosan capsules. Biotechnol Appl Biochem 59(2):142–152. doi:10.1002/bab.1009
Gao J, Dennis JE, Solchaga LA, Goldberg VM, Caplan AI (2002) Repair of osteochondral defect with tissue-engineered two-phase composite material of injectable calcium phosphate and hyaluronan sponge. Tissue Eng 8(5):827–837. doi:10.1089/10763270260424187
Gomoll AH, Madry H, Knutsen G, van Dijk N, Seil R, Brittberg M, Kon E (2010) The subchondral bone in articular cartilage repair: current problems in the surgical management. Knee Surg Sports Traumatol Arthrosc 18(4):434–447. doi:10.1007/s00167-010-1072-x
Gunatillake PA, Adhikari R (2003) Biodegradable synthetic polymers for tissue engineering. Eur Cell Mater 5:1–16, discussion 16. doi:vol005a01 [pii]
Haverkamp DJ, Schiphof D, Bierma-Zeinstra SM, Weinans H, Waarsing JH (2011) Variation in joint shape of osteoarthritic knees. Arthritis Rheum 63(11):3401–3407. doi:10.1002/art.30575
Hjelle K, Solheim E, Strand T, Muri R, Brittberg M (2002) Articular cartilage defects in 1,000 knee arthroscopies. Arthroscopy 18(7):730-734. doi:S0749806302000257 [pii]
Holland TA, Bodde EW, Cuijpers VM, Baggett LS, Tabata Y, Mikos AG, Jansen JA (2007) Degradable hydrogel scaffolds for in vivo delivery of single and dual growth factors in cartilage repair. Osteoarthritis Cartilage 15(2):187–197. doi:10.1016/j.joca.2006.07.006, S1063-4584(06)00231-7 [pii]
Hsu FY, Hung YS, Liou HM, Shen CH (2010) Electrospun hyaluronate-collagen nanofibrous matrix and the effects of varying the concentration of hyaluronate on the characteristics of foreskin fibroblast cells. Acta Biomater 6(6):2140–2147. doi:10.1016/j.actbio.2009.12.023, S1742-7061(09)00564-9 [pii]
Hung CT, Lima EG, Mauck RL, Takai E, LeRoux MA, Lu HH, Stark RG, Guo XE, Ateshian GA (2003) Anatomically shaped osteochondral constructs for articular cartilage repair. J Biomech 36(12):1853–1864. doi:S0021929003002136 [pii]
Hunziker EB (2002) Articular cartilage repair: basic science and clinical progress. A review of the current status and prospects. Osteoarthritis Cartilage 10(6):432–463. doi:10.1053/joca.2002.0801, S1063458402908010 [pii]
Hutmacher DW (2000) Scaffolds in tissue engineering bone and cartilage. Biomaterials 21(24):2529–2543. doi:S0142961200001216 [pii]
Jiang CC, Chiang H, Liao CJ, Lin YJ, Kuo TF, Shieh CS, Huang YY, Tuan RS (2007) Repair of porcine articular cartilage defect with a biphasic osteochondral composite. J Orthop Res 25(10):1277–1290. doi:10.1002/jor.20442
Kandel RA, Grynpas M, Pilliar R, Lee J, Wang J, Waldman S, Zalzal P, Hurtig M, Team CI-BoST (2006) Repair of osteochondral defects with biphasic cartilage-calcium polyphosphate constructs in a sheep model. Biomaterials 27(22):4120–4131. doi:10.1016/j.biomaterials.2006.03.005, S0142-9612(06)00214-6 [pii]
Karageorgiou V, Kaplan D (2005) Porosity of 3D biomaterial scaffolds and osteogenesis. Biomaterials 26(27):5474–5491. doi:10.1016/j.biomaterials.2005.02.002
Kawanishi M, Oura A, Furukawa K, Fukubayashi T, Nakamura K, Tateishi T, Ushida T (2007) Redifferentiation of dedifferentiated bovine articular chondrocytes enhanced by cyclic hydrostatic pressure under a gas-controlled system. Tissue Eng 13(5):957–964. doi:10.1089/ten.2006.0176
Keeney M, Pandit A (2009) The osteochondral junction and its repair via bi-phasic tissue engineering scaffolds. Tissue Eng Part B Rev 15(1):55–73. doi:10.1089/ten.teb.2008.0388, 10.1089/ten.teb.2008.0388 [pii]
Kim K, Yeatts A, Dean D, Fisher JP (2010) Stereolithographic bone scaffold design parameters: osteogenic differentiation and signal expression. Tissue Eng Part B Rev 16(5):523–539. doi:10.1089/ten.TEB.2010.0171
Koga H, Shimaya M, Muneta T, Nimura A, Morito T, Hayashi M, Suzuki S, Ju YJ, Mochizuki T, Sekiya I (2008) Local adherent technique for transplanting mesenchymal stem cells as a potential treatment of cartilage defect. Arthritis Res Ther 10(4):R84. doi:10.1186/ar2460, ar2460 [pii]
Kon E, Delcogliano M, Filardo G, Busacca M, Di Martino A, Marcacci M (2011) Novel nano-composite multilayered biomaterial for osteochondral regeneration: a pilot clinical trial. Am J Sports Med 39(6):1180–1190. doi:10.1177/0363546510392711, 0363546510392711 [pii]
Kon E, Delcogliano M, Filardo G, Fini M, Giavaresi G, Francioli S, Martin I, Pressato D, Arcangeli E, Quarto R, Sandri M, Marcacci M (2010) Orderly osteochondral regeneration in a sheep model using a novel nano-composite multilayered biomaterial. J Orthop Res 28(1):116–124. doi:10.1002/jor.20958
Kon E, Filardo G, Di Martino A, Busacca M, Moio A, Perdisa F, Marcacci M (2013a) Clinical results and MRI evolution of a nano-composite multilayered biomaterial for osteochondral regeneration at 5 years. Am J Sports Med. doi:0363546513505434 [pii] 10.1177/0363546513505434 [doi]
Kon E, Filardo G, Robinson D, Eisman JA, Levy A, Zaslav K, Shani J, Altschuler N (2013b) Osteochondral regeneration using a novel aragonite-hyaluronate bi-phasic scaffold in a goat model. Knee Surg Sports Traumatol Arthrosc. doi:10.1007/s00167-013-2467-2 [doi]
Kurz B, Domm C, Jin M, Sellckau R, Schunke M (2004) Tissue engineering of articular cartilage under the influence of collagen I/III membranes and low oxygen tension. Tissue Eng 10:1277–1286. doi:10.1089/ten.2004.10.1277
Lawrence RC, Felson DT, Helmick CG, Arnold LM, Choi H, Deyo RA, Gabriel S, Hirsch R, Hochberg MC, Hunder GG, Jordan JM, Katz JN, Kremers HM, Wolfe F (2008) Estimates of the prevalence of arthritis and other rheumatic conditions in the United States. Part II. Arthritis Rheum 58(1):26–35. doi:10.1002/art.23176
Lee M, Wu BM (2012) Recent advances in 3D printing of tissue engineering scaffolds. Methods Mole Biol (Clifton, NJ) 868:257–267. doi:10.1007/978-1-61779-764-4_15
Levett PA, Melchels FP, Schrobback K, Hutmacher DW, Malda J, Klein TJ (2013) Chondrocyte redifferentiation and construct mechanical property development in single-component photocrosslinkable hydrogels. J Biomed Mater Res A. doi:10.1002/jbm.a.34924 [doi]
Li WJ, Cooper JA Jr, Mauck RL, Tuan RS (2006) Fabrication and characterization of six electrospun poly(alpha-hydroxy ester)-based fibrous scaffolds for tissue engineering applications. Acta Biomater 2(4):377–385. doi:10.1016/j.actbio.2006.02.005, S1742-7061(06)00021-3 [pii]
Li WJ, Mauck RL, Cooper JA, Yuan X, Tuan RS (2007) Engineering controllable anisotropy in electrospun biodegradable nanofibrous scaffolds for musculoskeletal tissue engineering. J Biomech 40(8):1686–1693. doi:10.1016/j.jbiomech.2006.09.004, S0021-9290(06)00318-6 [pii]
Lin H, Zhang D, Alexander PG, Yang G, Tan J, Cheng AW, Tuan RS (2013) Application of visible light-based projection stereolithography for live cell-scaffold fabrication with designed architecture. Biomaterials 34(2):331–339. doi:10.1016/j.biomaterials.2012.09.048, S0142-9612(12)01056-3 [pii]
Lynch JA, Parimi N, Chaganti RK, Nevitt MC, Lane NE, Study of Osteoporotic Fractures Research G (2009) The association of proximal femoral shape and incident radiographic hip OA in elderly women. Osteoarthritis Cartilage 17(10):1313–1318. doi:10.1016/j.joca.2009.04.011, S1063-4584(09)00114-9 [pii]
Madry H, van Dijk CN, Mueller-Gerbl M (2010) The basic science of the subchondral bone. Knee Surg Sports Traumatol Arthrosc 18(4):419–433. doi:10.1007/s00167-010-1054-z
Mannoni A, Briganti MP, Di Bari M, Ferrucci L, Costanzo S, Serni U, Masotti G, Marchionni N (2003) Epidemiological profile of symptomatic osteoarthritis in older adults: a population based study in Dicomano, Italy. Ann Rheum Dis 62(6):576–578
Mano JF, Reis RL (2007) Osteochondral defects: present situation and tissue engineering approaches. J Tissue Eng Regen Med 1(4):261–273. doi:10.1002/term.37
Marcacci M, Berruto M, Brocchetta D, Delcogliano A, Ghinelli D, Gobbi A, Kon E, Pederzini L, Rosa D, Sacchetti GL, Stefani G, Zanasi S (2005) Articular cartilage engineering with Hyalograft C: 3-year clinical results. Clin Orthop Relat Res 435:96–105. doi:00003086-200506000-00015 [pii]
Marquass B, Somerson JS, Hepp P, Aigner T, Schwan S, Bader A, Josten C, Zscharnack M, Schulz RM (2010) A novel MSC-seeded triphasic construct for the repair of osteochondral defects. J Orthop Res 28(12):1586–1599. doi:10.1002/jor.21173
Martin I, Miot S, Barbero A, Jakob M, Wendt D (2007) Osteochondral tissue engineering. J Biomech 40(4):750–765. doi:10.1016/j.jbiomech.2006.03.008, S0021-9290(06)00096-0 [pii]
Martin MJ, Muotri A, Gage F, Varki A (2005) Human embryonic stem cells express an immunogenic nonhuman sialic acid. Nat Med 11(2):228–232. doi:10.1038/nm1181, nm1181 [pii]
Mente PL, Lewis JL (1994) Elastic modulus of calcified cartilage is an order of magnitude less than that of subchondral bone. J Orthop Res 12(5):637–647. doi:10.1002/jor.1100120506
Miao X, Tan DM, Li J, Xiao Y, Crawford R (2008) Mechanical and biological properties of hydroxyapatite/tricalcium phosphate scaffolds coated with poly(lactic-co-glycolic acid). Acta Biomater 4(3):638–645. doi:10.1016/j.actbio.2007.10.006, S1742-7061(07)00172-9 [pii]
Minas T, Gomoll AH, Rosenberger R, Royce RO, Bryant T (2009) Increased failure rate of autologous chondrocyte implantation after previous treatment with marrow stimulation techniques. Am J Sports Med 37(5):902–908. doi:10.1177/0363546508330137, 0363546508330137 [pii]
Miot S, Brehm W, Dickinson S, Sims T, Wixmerten A, Longinotti C, Hollander AP, Mainil-Varlet P, Martin I (2012) Influence of in vitro maturation of engineered cartilage on the outcome of osteochondral repair in a goat model. Eur Cell Mater 23:222–236, vol023a17 [pii]
Mobasheri A (2012) Osteoarthritis year 2012 in review: biomarkers. Osteoarthritis Cartilage 20(12):1451–1464. doi:10.1016/j.joca.2012.07.009, S1063-4584(12)00897-7 [pii]
Mosher TJ, Walker EA, Petscavage-Thomas J, Guermazi A (2013) Osteoarthritis year 2013 in review: imaging. Osteoarthritis Cartilage 21(10):1425–1435. doi:10.1016/j.joca.2013.07.010, S1063-4584(13)00895-9 [pii]
Murphy L, Helmick CG (2012) The impact of osteoarthritis in the United States: a population-health perspective. Am J Nurs 112(3 Suppl 1):S13–S19. doi:10.1097/01.naj.0000412646.80054.21
Murphy SV, Atala A (2014) 3D bioprinting of tissues and organs. Nat Biotechnol 32(8):773–785. doi:10.1038/nbt.2958
Nakamura N, Hui J, Koizumi K, Yasui Y, Nishii T, Lad D, Karnatzikos G, Gobbi A (2014) Stem cell therapy in cartilage repair – culture-free and cell culture–based methods. Oper Tech Orthop 24(1):54–60. doi:10.1053/j.oto.2014.02.006
Nooeaid P, Salih V, Beier JP, Boccaccini AR (2012) Osteochondral tissue engineering: scaffolds, stem cells and applications. J Cell Mol Med 16(10):2247–2270. doi:10.1111/j.1582-4934.2012.01571.x
O’Shea TM, Miao X (2008) Bilayered scaffolds for osteochondral tissue engineering. Tissue Eng Part B Rev 14(4):447–464. doi:10.1089/ten.teb.2008.0327
Oegema TR Jr, Carpenter RJ, Hofmeister F, Thompson RC Jr (1997) The interaction of the zone of calcified cartilage and subchondral bone in osteoarthritis. Microsc Res Tech 37(4):324–332. doi:10.1002/(SICI)1097-0029(19970515)37:4<324::AID-JEMT7>3.0.CO;2-K, 10.1002/(SICI)1097-0029(19970515)37:4<324::AID-JEMT7>3.0.CO;2-K [pii]
Oliveira JM, Rodrigues MT, Silva SS, Malafaya PB, Gomes ME, Viegas CA, Dias IR, Azevedo JT, Mano JF, Reis RL (2006) Novel hydroxyapatite/chitosan bilayered scaffold for osteochondral tissue-engineering applications: scaffold design and its performance when seeded with goat bone marrow stromal cells. Biomaterials 27(36):6123–6137. doi:10.1016/j.biomaterials.2006.07.034, S0142-9612(06)00659-4 [pii]
Orth P, Cucchiarini M, Kohn D, Madry H (2013) Alterations of the subchondral bone in osteochondral repair – translational data and clinical evidence. Eur Cell Mater 25:299–316, discussion 314–296. doi:vol025a21 [pii]
Orth P, Meyer HL, Goebel L, Eldracher M, Ong MF, Cucchiarini M, Madry H (2013) Improved repair of chondral and osteochondral defects in the ovine trochlea compared with the medial condyle. J Orthop Res 31(11):1772–1779. doi:10.1002/jor.22418
Panseri S, Russo A, Cunha C, Bondi A, Di Martino A, Patella S, Kon E (2012) Osteochondral tissue engineering approaches for articular cartilage and subchondral bone regeneration. Knee Surg Sports Traumatol Arthrosc 20(6):1182–1191. doi:10.1007/s00167-011-1655-1
Peat G, McCarney R, Croft P (2001) Knee pain and osteoarthritis in older adults: a review of community burden and current use of primary health care. Ann Rheum Dis 60(2):91–97
Pena J, Corrales T, Izquierdo-Barba I, Serrano MC, Portoles MT, Pagani R, Vallet-Regi M (2006) Alkaline-treated poly(epsilon-caprolactone) films: degradation in the presence or absence of fibroblasts. J Biomed Mater Res A 76(4):788–797. doi:10.1002/jbm.a.30547
Ravi B, Escott B, Shah PS, Jenkinson R, Chahal J, Bogoch E, Kreder H, Hawker G (2012) A systematic review and meta-analysis comparing complications following total joint arthroplasty for rheumatoid arthritis versus for osteoarthritis. Arthritis Rheum 64(12):3839–3849. doi:10.1002/art.37690
Re’em T, Witte F, Willbold E, Ruvinov E, Cohen S (2012) Simultaneous regeneration of articular cartilage and subchondral bone induced by spatially presented TGF-beta and BMP-4 in a bilayer affinity binding system. Acta Biomater 8(9):3283–3293. doi:10.1016/j.actbio.2012.05.014, S1742-7061(12)00218-8 [pii]
Ren J, Zhao P, Ren T, Gu S, Pan K (2008) Poly (D,L-lactide)/nano-hydroxyapatite composite scaffolds for bone tissue engineering and biocompatibility evaluation. J Mater Sci Mater Med 19(3):1075–1082. doi:10.1007/s10856-007-3181-8
Reyes R, Delgado A, Sanchez E, Fernandez A, Hernandez A, Evora C (2012) Repair of an osteochondral defect by sustained delivery of BMP-2 or TGFbeta1 from a bilayered alginate-PLGA scaffold. J Tissue Eng Regen Med. doi:10.1002/term.1549 [doi]
Reyes R, Delgado A, Solis R, Sanchez E, Hernandez A, San Roman J, Evora C (2013) Cartilage repair by local delivery of TGF-beta1 or BMP-2 from a novel, segmented polyurethane/polylactic-co-glycolic bilayered scaffold. J Biomed Mater Res A. doi:10.1002/jbm.a.34769 [doi]
Rodrigues MT, Gomes ME, Reis RL (2011) Current strategies for osteochondral regeneration: from stem cells to pre-clinical approaches. Curr Opin Biotechnol 22(5):726–733. doi:10.1016/j.copbio.2011.04.006, S0958-1669(11)00068-1 [pii]
Roemer FW, Guermazi A (2012) Osteoarthritis year 2012 in review: imaging. Osteoarthritis Cartilage 20(12):1440–1446. doi:10.1016/j.joca.2012.07.008, S1063-4584(12)00896-5 [pii]
Rose FR, Hou Q, Oreffo RO (2004) Delivery systems for bone growth factors – the new players in skeletal regeneration. J Pharm Pharmacol 56(4):415–427. doi:10.1211/0022357023312
Sadoghi P, Liebensteiner M, Agreiter M, Leithner A, Bohler N, Labek G (2013) Revision surgery after total joint arthroplasty: a complication-based analysis using worldwide arthroplasty registers. J Arthroplasty 28(8):1329–1332. doi:10.1016/j.arth.2013.01.012
Sakaguchi Y, Sekiya I, Yagishita K, Muneta T (2005) Comparison of human stem cells derived from various mesenchymal tissues: superiority of synovium as a cell source. Arthritis Rheum 52(8):2521–2529. doi:10.1002/art.21212
Salgado AJ, Coutinho OP, Reis RL (2004) Bone tissue engineering: state of the art and future trends. Macromol Biosci 4(8):743–765. doi:10.1002/mabi.200400026
Sarasam AR, Krishnaswamy RK, Madihally SV (2006) Blending chitosan with polycaprolactone: effects on physicochemical and antibacterial properties. Biomacromolecules 7(4):1131–1138. doi:10.1021/bm050935d
Schek RM, Taboas JM, Segvich SJ, Hollister SJ, Krebsbach PH (2004) Engineered osteochondral grafts using biphasic composite solid free-form fabricated scaffolds. Tissue Eng 10(9–10):1376–1385. doi:10.1089/ten.2004.10.1376
Schleicher I, Lips KS, Sommer U, Schappat I, Martin AP, Szalay G, Hartmann S, Schnettler R (2013) Biphasic scaffolds for repair of deep osteochondral defects in a sheep model. J Surg Res 183(1):184–192. doi:10.1016/j.jss.2012.11.036, S0022-4804(12)01910-5 [pii]
Schnabel M, Marlovits S, Eckhoff G, Fichtel I, Gotzen L, Vecsei V, Schlegel J (2002) Dedifferentiation-associated changes in morphology and gene expression in primary human articular chondrocytes in cell culture. Osteoarthritis Cartilage 10(1):62–70. doi:10.1053/joca.2001.0482, S1063458401904820 [pii]
Seo SJ, Mahapatra C, Singh RK, Knowles JC, Kim HW (2014) Strategies for osteochondral repair: focus on scaffolds. J Tissue Eng 5:2041731414541850. doi: 10.1177/2041731414541850
Shafiee A, Soleimani M, Chamheidari GA, Seyedjafari E, Dodel M, Atashi A, Gheisari Y (2011) Electrospun nanofiber-based regeneration of cartilage enhanced by mesenchymal stem cells. J Biomed Mater Res A 99(3):467–478. doi:10.1002/jbm.a.33206
Sherwood JK, Riley SL, Palazzolo R, Brown SC, Monkhouse DC, Coates M, Griffith LG, Landeen LK, Ratcliffe A (2002) A three-dimensional osteochondral composite scaffold for articular cartilage repair. Biomaterials 23(24):4739–4751. doi:S0142961202002235 [pii]
Shimomura K, Ando W, Tateishi K, Nansai R, Fujie H, Hart DA, Kohda H, Kita K, Kanamoto T, Mae T, Nakata K, Shino K, Yoshikawa H, Nakamura N (2010) The influence of skeletal maturity on allogenic synovial mesenchymal stem cell-based repair of cartilage in a large animal model. Biomaterials 31(31):8004–8011. doi:10.1016/j.biomaterials.2010.07.017, S0142-9612(10)00858-6 [pii]
Shimomura K, Moriguchi Y, Ando W, Nansai R, Fujie H, Hart DA, Gobbi A, Kita K, Horibe S, Shino K, Yoshikawa H, Nakamura N (2014a) Osteochondral repair using a scaffold-free tissue-engineered construct derived from synovial mesenchymal stem cells and a hydroxyapatite-based artificial bone. Tissue Eng Part A. doi:10.1089/ten.tea.2013.0414 [doi]
Shimomura K, Moriguchi Y, Murawski CD, Yoshikawa H, Nakamura N (2014b) Osteochondral tissue engineering with biphasic scaffold: current strategies and techniques. Tissue Eng Part B Rev. doi:10.1089/ten.TEB.2013.0543 [doi]
Shintani N, Hunziker EB (2007) Chondrogenic differentiation of bovine synovium: bone morphogenetic proteins 2 and 7 and transforming growth factor beta1 induce the formation of different types of cartilaginous tissue. Arthritis Rheum 56(6):1869–1879. doi:10.1002/art.22701
Sosio C, Di Giancamillo A, Deponti D, Gervaso F, Scalera F, Melato M, Campagnol M, Boschetti F, Nonis A, Domeneghini C, Sannino A, Peretti GM (2014) Osteochondral repair by a novel interconnecting collagen-hydroxyapatite substitute: a large-animal study. Tissue Eng Part A. doi:10.1089/ten.TEA.2014.0129
Sundelacruz S, Kaplan DL (2009) Stem cell- and scaffold-based tissue engineering approaches to osteochondral regenerative medicine. Semin Cell Dev Biol 20(6):646–655. doi:10.1016/j.semcdb.2009.03.017, S1084-9521(09)00069-X [pii]
Takahashi K, Yamanaka S (2006) Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 126(4):663–676. doi:10.1016/j.cell.2006.07.024
Takahashi N, Knudson CB, Thankamony S, Ariyoshi W, Mellor L, Im HJ, Knudson W (2010) Induction of CD44 cleavage in articular chondrocytes. Arthritis Rheum 62(5):1338–1348. doi:10.1002/art.27410
Takahashi T, Ogasawara T, Asawa Y, Mori Y, Uchinuma E, Takato T, Hoshi K (2007) Three-dimensional microenvironments retain chondrocyte phenotypes during proliferation culture. Tissue Eng 13(7):1583–1592. doi:10.1089/ten.2006.0322
Tamai N, Myoui A, Hirao M, Kaito T, Ochi T, Tanaka J, Takaoka K, Yoshikawa H (2005) A new biotechnology for articular cartilage repair: subchondral implantation of a composite of interconnected porous hydroxyapatite, synthetic polymer (PLA-PEG), and bone morphogenetic protein-2 (rhBMP-2). Osteoarthritis Cartilage 13(5):405–417. doi:10.1016/j.joca.2004.12.014, S1063-4584(04)00289-4 [pii]
Tan W, Twomey J, Guo D, Madhavan K, Li M (2010) Evaluation of nanostructural, mechanical, and biological properties of collagen-nanotube composites. IEEE Trans Nanobioscience 9(2):111–120. doi:10.1109/TNB.2010.2043367
Tsumaki N, Okada M, Yamashita A (2014) iPS cell technologies and cartilage regeneration. Bone. doi:10.1016/j.bone.2014.07.011
Vos T, Flaxman AD, Naghavi M, Lozano R, Michaud C, Ezzati M, Shibuya K, Salomon JA, Abdalla S, Aboyans V, Abraham J, Ackerman I, Aggarwal R, Ahn SY, Ali MK, Alvarado M, Anderson HR, Anderson LM, Andrews KG, Atkinson C, Baddour LM, Bahalim AN, Barker-Collo S, Barrero LH, Bartels DH, Basanez MG, Baxter A, Bell ML, Benjamin EJ, Bennett D, Bernabe E, Bhalla K, Bhandari B, Bikbov B, Bin Abdulhak A, Birbeck G, Black JA, Blencowe H, Blore JD, Blyth F, Bolliger I, Bonaventure A, Boufous S, Bourne R, Boussinesq M, Braithwaite T, Brayne C, Bridgett L, Brooker S, Brooks P, Brugha TS, Bryan-Hancock C, Bucello C, Buchbinder R, Buckle G, Budke CM, Burch M, Burney P, Burstein R, Calabria B, Campbell B, Canter CE, Carabin H, Carapetis J, Carmona L, Cella C, Charlson F, Chen H, Cheng AT, Chou D, Chugh SS, Coffeng LE, Colan SD, Colquhoun S, Colson KE, Condon J, Connor MD, Cooper LT, Corriere M, Cortinovis M, de Vaccaro KC, Couser W, Cowie BC, Criqui MH, Cross M, Dabhadkar KC, Dahiya M, Dahodwala N, Damsere-Derry J, Danaei G, Davis A, De Leo D, Degenhardt L, Dellavalle R, Delossantos A, Denenberg J, Derrett S, Des Jarlais DC, Dharmaratne SD, Dherani M, Diaz-Torne C, Dolk H, Dorsey ER, Driscoll T, Duber H, Ebel B, Edmond K, Elbaz A, Ali SE, Erskine H, Erwin PJ, Espindola P, Ewoigbokhan SE, Farzadfar F, Feigin V, Felson DT, Ferrari A, Ferri CP, Fevre EM, Finucane MM, Flaxman S, Flood L, Foreman K, Forouzanfar MH, Fowkes FG, Franklin R, Fransen M, Freeman MK, Gabbe BJ, Gabriel SE, Gakidou E, Ganatra HA, Garcia B, Gaspari F, Gillum RF, Gmel G, Gosselin R, Grainger R, Groeger J, Guillemin F, Gunnell D, Gupta R, Haagsma J, Hagan H, Halasa YA, Hall W, Haring D, Haro JM, Harrison JE, Havmoeller R, Hay RJ, Higashi H, Hill C, Hoen B, Hoffman H, Hotez PJ, Hoy D, Huang JJ, Ibeanusi SE, Jacobsen KH, James SL, Jarvis D, Jasrasaria R, Jayaraman S, Johns N, Jonas JB, Karthikeyan G, Kassebaum N, Kawakami N, Keren A, Khoo JP, King CH, Knowlton LM, Kobusingye O, Koranteng A, Krishnamurthi R, Lalloo R, Laslett LL, Lathlean T, Leasher JL, Lee YY, Leigh J, Lim SS, Limb E, Lin JK, Lipnick M, Lipshultz SE, Liu W, Loane M, Ohno SL, Lyons R, Ma J, Mabweijano J, MacIntyre MF, Malekzadeh R, Mallinger L, Manivannan S, Marcenes W, March L, Margolis DJ, Marks GB, Marks R, Matsumori A, Matzopoulos R, Mayosi BM, McAnulty JH, McDermott MM, McGill N, McGrath J, Medina-Mora ME, Meltzer M, Mensah GA, Merriman TR, Meyer AC, Miglioli V, Miller M, Miller TR, Mitchell PB, Mocumbi AO, Moffitt TE, Mokdad AA, Monasta L, Montico M, Moradi-Lakeh M, Moran A, Morawska L, Mori R, Murdoch ME, Mwaniki MK, Naidoo K, Nair MN, Naldi L, Narayan KM, Nelson PK, Nelson RG, Nevitt MC, Newton CR, Nolte S, Norman P, Norman R, O’Donnell M, O’Hanlon S, Olives C, Omer SB, Ortblad K, Osborne R, Ozgediz D, Page A, Pahari B, Pandian JD, Rivero AP, Patten SB, Pearce N, Padilla RP, Perez-Ruiz F, Perico N, Pesudovs K, Phillips D, Phillips MR, Pierce K, Pion S, Polanczyk GV, Polinder S, Pope CA 3rd, Popova S, Porrini E, Pourmalek F, Prince M, Pullan RL, Ramaiah KD, Ranganathan D, Razavi H, Regan M, Rehm JT, Rein DB, Remuzzi G, Richardson K, Rivara FP, Roberts T, Robinson C, De Leon FR, Ronfani L, Room R, Rosenfeld LC, Rushton L, Sacco RL, Saha S, Sampson U, Sanchez-Riera L, Sanman E, Schwebel DC, Scott JG, Segui-Gomez M, Shahraz S, Shepard DS, Shin H, Shivakoti R, Singh D, Singh GM, Singh JA, Singleton J, Sleet DA, Sliwa K, Smith E, Smith JL, Stapelberg NJ, Steer A, Steiner T, Stolk WA, Stovner LJ, Sudfeld C, Syed S, Tamburlini G, Tavakkoli M, Taylor HR, Taylor JA, Taylor WJ, Thomas B, Thomson WM, Thurston GD, Tleyjeh IM, Tonelli M, Towbin JA, Truelsen T, Tsilimbaris MK, Ubeda C, Undurraga EA, van der Werf MJ, van Os J, Vavilala MS, Venketasubramanian N, Wang M, Wang W, Watt K, Weatherall DJ, Weinstock MA, Weintraub R, Weisskopf MG, Weissman MM, White RA, Whiteford H, Wiersma ST, Wilkinson JD, Williams HC, Williams SR, Witt E, Wolfe F, Woolf AD, Wulf S, Yeh PH, Zaidi AK, Zheng ZJ, Zonies D, Lopez AD, Murray CJ, AlMazroa MA, Memish ZA (2012) Years lived with disability (YLDs) for 1160 sequelae of 289 diseases and injuries 1990–2010: a systematic analysis for the global burden of disease study 2010. Lancet 380(9859):2163–2196. doi:10.1016/s0140-6736(12)61729-2
Yamasaki N, Hirao M, Nanno K, Sugiyasu K, Tamai N, Hashimoto N, Yoshikawa H, Myoui A (2009) A comparative assessment of synthetic ceramic bone substitutes with different composition and microstructure in rabbit femoral condyle model. J Biomed Mater Res B Appl Biomater 91(2):788–798. doi:10.1002/jbm.b.31457
Yang PJ, Temenoff JS (2009) Engineering orthopedic tissue interfaces. Tissue Eng Part B Rev 15(2):127–141. doi:10.1089/ten.teb.2008.0371
Zhang S, Chen L, Jiang Y, Cai Y, Xu G, Tong T, Zhang W, Wang L, Ji J, Shi P, Ouyang HW (2013) Bi-layer collagen/microporous electrospun nanofiber scaffold improves the osteochondral regeneration. Acta Biomater 9(7):7236–7247. doi:10.1016/j.actbio.2013.04.003, S1742-7061(13)00175-X [pii]
Zhang W, Chen J, Tao J, Hu C, Chen L, Zhao H, Xu G, Heng BC, Ouyang HW (2013) The promotion of osteochondral repair by combined intra-articular injection of parathyroid hormone-related protein and implantation of a bi-layer collagen-silk scaffold. Biomaterials 34(25):6046–6057. doi:10.1016/j.biomaterials.2013.04.055, S0142-9612(13)00529-2 [pii]
Zhang W, Moskowitz RW, Nuki G, Abramson S, Altman RD, Arden N, Bierma-Zeinstra S, Brandt KD, Croft P, Doherty M, Dougados M, Hochberg M, Hunter DJ, Kwoh K, Lohmander LS, Tugwell P (2007) OARSI recommendations for the management of hip and knee osteoarthritis, part I: critical appraisal of existing treatment guidelines and systematic review of current research evidence. Osteoarthritis Cartilage 15(9):981–1000. doi:10.1016/j.joca.2007.06.014, S1063-4584(07)00234-8 [pii]
Zhang W, Moskowitz RW, Nuki G, Abramson S, Altman RD, Arden N, Bierma-Zeinstra S, Brandt KD, Croft P, Doherty M, Dougados M, Hochberg M, Hunter DJ, Kwoh K, Lohmander LS, Tugwell P (2008) OARSI recommendations for the management of hip and knee osteoarthritis, part II: OARSI evidence-based, expert consensus guidelines. Osteoarthritis Cartilage 16(2):137–162. doi:10.1016/j.joca.2007.12.013, S1063-4584(07)00397-4 [pii]
Zhang W, Nuki G, Moskowitz RW, Abramson S, Altman RD, Arden NK, Bierma-Zeinstra S, Brandt KD, Croft P, Doherty M, Dougados M, Hochberg M, Hunter DJ, Kwoh K, Lohmander LS, Tugwell P (2010) OARSI recommendations for the management of hip and knee osteoarthritis: part III: changes in evidence following systematic cumulative update of research published through January 2009. Osteoarthritis Cartilage 18(4):476–499. doi:10.1016/j.joca.2010.01.013, S1063-4584(10)00046-4 [pii]
Zheng MH, Willers C, Kirilak L, Yates P, Xu J, Wood D, Shimmin A (2007) Matrix-induced autologous chondrocyte implantation (MACI): biological and histological assessment. Tissue Eng 13(4):737–746. doi:10.1089/ten.2006.0246
Acknowledgments
This study was supported by a grant from the New Energy and Industrial Technology Development Organization, Japan, Grant-in-Aid for Scientific Research (B), and Japan Society for the Promotion of Science, Japan.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Shimomura, K. et al. (2016). Current Strategies in Osteochondral Repair with Biomaterial Scaffold. In: Korkusuz, F. (eds) Musculoskeletal Research and Basic Science. Springer, Cham. https://doi.org/10.1007/978-3-319-20777-3_23
Download citation
DOI: https://doi.org/10.1007/978-3-319-20777-3_23
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-20776-6
Online ISBN: 978-3-319-20777-3
eBook Packages: MedicineMedicine (R0)