Abstract
Despite remarkable advances in our understanding of the pathogenesis and treatment of osteoporosis, the disease is still underdiagnosed and undertreated, even in patients who clearly warrant pharmacological therapy. However, several gaps in the osteoporotic patients’ care and treatment have been identified. Failure to prevent secondary fracture is one of the largest gaps in the practice of evidence-based medicine. Fewer than 20% of patients presenting with low trauma fractures to hospitals or general practices are investigated or treated for osteoporosis, whereas, nearly 35% of patients with a prior diagnosis of osteoporosis had not consulted a GP or a specialist in the previous 12 months for their condition. On another front, among those who are prescribed medicines, poor adherence to osteoporosis treatment is a leading cause of repeat fractures and hospitalisation, with patients either not taking their treatment as prescribed (poor compliance) or discontinuing therapy within 6 months (lack of persistence). Patient concerns about rare side effects of osteoporosis medicines and the perception of lack of clear evidence in support of their long-term efficacy are commonly reported reasons for low rates of patient adherence to and persistence with osteoporosis treatment. This chapter will start by discussing the need to treat in osteoporosis, followed by highlighting the gaps in osteoporotic patients’ care as well as treatment. The chapter will expand to analyse and describe approaches to close these gaps and lastly, the chapter will present the most recent guidelines for the management of osteoporosis.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Case finding
- Disease awareness
- Fragility fracture
- Osteoporosis
- Policy
- Primary prevention
- Prioritisation
- Secondary prevention
Introduction
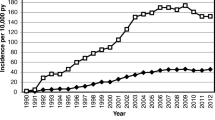
Osteoporosis is a major public health threat in the United States and around the globe. As of 2010, 10.2 million adults have osteoporosis and another 43.4 million have low bone mass, a figure expected to rise nearly 30% by the year 2030 (Fig. 21.1). Osteoporosis is the major cause of fragility fractures, which are from low trauma not likely to occur in healthy bone, in the population age 50 and above. Taking the American population as an example, as osteoporosis prevalence increases parallel with aging, the number of fragility fractures may increase from two million in 2005 to three million in 2025 [1, 2]. Patients who sustain fragility fractures will, most likely, experience the morbidity/comorbidity consequent upon fracturing. Furthermore, fragility fractures cause substantial pain and severe disability, often leading to a reduced quality of life. In addition, hip and vertebral fractures are associated with decreased life expectancy. Patients who have had any one fracture have an increased risk of subsequent fractures. The US Preventive Services Task Force recommends osteoporosis screening and treatment after a first fracture due to increased risk of future fractures, including a 20-fold greater risk for a clinically serious hip or spine fracture [3, 4]. The National Osteoporosis Foundation considers all postmenopausal women and men older than 50 years with prior hip or vertebral fracture as candidates for osteoporosis treatment [5].
Fortunately, osteoporosis is a preventable disease that can be diagnosed and managed before any fracture occurs. Previous studies suggest that sex, age, race, education level, insurance type, baseline calcium use, fracture site, prior osteoporosis diagnosis, previous fracture, chronic comorbidities, and history of cigarette smoking are predictors of the use of osteoporosis medication for secondary prevention of further fractures [6,7,8,9,10,11]. In patients who have already experienced a fracture, the appropriate use of available therapies can effectively decrease the risk of future fractures by up to 50%. Yet osteoporosis is underdiagnosed and undertreated worldwide, and secondary fracture risk is poorly addressed in patients who have sustained a first fracture [12,13,14,15,16,17,18,19,20,21].
This chapter will start by discussing the need to treat in osteoporosis, followed by highlighting the gaps in osteoporotic patients’ care as well as treatment. The chapter will expand to analyse and describe approaches to close these gaps and lastly and present the most recent guidelines for the management of osteoporosis.
Osteoporosis: The Need to Treat
Many guidelines for the assessment and treatment of osteoporosis recommend that intervention be considered in men and women who have sustained a fragility fracture [22]. Guidelines in North America [23, 24] specifically refer to a prior hip fracture, as well as spine fracture, as mandatory indication for treatment because of the marked effect of fractures at these sites on both morbidity and mortality. In addition, hip fractures have large economic consequences. For example, hip fractures account for 17% of all osteoporotic fractures in Europe but comprise 54% of the direct cost of fractures [22]. The need for treatment arises because of the increased risk of a second fracture [25], which is particularly acute in the immediate post-fracture period when fracture rates are substantially increased [26,27,28].
Despite a number of advances, particularly in the diagnosis of osteoporosis, the assessment of fracture risk, the development of interventions that reduce the risk of fractures, and the production of practice guidelines, many surveys indicate that a minority of men and women at high fracture risk actually receive treatment [29,30,31,32,33,34,35]. According to the 2008 Joint Commission Report, Improving and Measuring Osteoporosis Management, only 20% of patients with low-impact fractures in the general population are ever tested or treated for osteoporosis or receive therapies to reduce the risk of future fracture within the year following the fracture [13, 36,37,38,39].
In another large observational cohort study, only 6.6% of hip fracture patients received calcium and vitamin D after surgery [12]. Paradoxically, the therapeutic care gap may be particularly wide in the elderly in whom the importance and impact of treatment is high; studies have shown that as few as 10% of older women with fragility fractures receive any osteoporosis therapy (oestrogens not considered) [40, 41]. Furthermore, treatment rates following a fracture are lower for those individuals who reside in long-term care [33]. This contrasts with the situation following myocardial infarction, for which condition a significant care gap has been overcome in the past 15 years: 75% of such individuals now receive beta blockers to help prevent recurrent myocardial infarction [42].
Osteoporosis is a silent disease that progresses without the evidence of symptoms until a fracture occurs. Fragility fractures are responsible for considerable pain and suffering, severe disability, reduced quality of life, and use of long-term care and rehabilitation resources [43]. The leading cause of the loss of independence in men or women 70 years of age and older are fragility fractures due to falls at home [44]. Most patients do not regain their pre-fracture functionality or independence and many are permanently limited in mobility, ability to fulfil social roles, and performance of activities of daily living and self-care [45]. Psychological consequences have also been noted, such as loss of self-efficacy, depression, and anxiety [46].
The economic burden due to osteoporotic fractures is high and will escalate as the population ages. Overall, the medical cost of osteoporosis and related fractures is estimated to be $20 billion per year. The annual cost in the United States of caring for osteoporotic-related fractures alone parallels or exceeds the annual cost for myocardial infarction, breast cancer, and/or cerebrovascular accidents [47]. Direct costs are predicted to escalate to $25 billion by 2025 and $50 billion by 2050 due to the increase in the incidence of osteoporotic fractures [48].
In the United States, two million osteoporotic fractures occur every year. One of two women and one of five men will sustain an osteoporotic fracture in their lifetime [49]. For women over 50 years, the lifetime risk of a fracture is higher than the combined risk of developing cervical, uterine or breast cancer, while men over 50 it is higher than risk of developing prostate cancer [50].The incidence rates of fragility fracture due to osteoporosis at all skeletal sites increase with advancing age in both women and men, with those 85 years and older at highest risk [51].
Furthermore, an initial fragility fracture increases the absolute risk of sustaining future fractures for both men and women [52]. An individual who sustains a fracture is 86% more likely to sustain a fracture of another type [53]. For men, although their risk for an initial fracture is lower than that of women, once they sustain an initial fracture, their risk for additional fractures escalates to the same level of risk for subsequent fracture as women in their age group. For women, an initial fracture increases their risk for subsequent fraction as high as or higher than the initial fracture risk carried by women in the 10-year age group above theirs. Research has demonstrated increased risk for future fracture applies to virtually all clinical fracture sites, is highest immediately after the initial event, and persists for up to 10 years [54].
Premature mortality associated with fracture, particularly following hip and vertebral fractures, is well documented [55, 56], and evidence of elevated mortality risk following other types of osteoporotic fractures is mounting [55]. Risk of death is most pronounced in the first three to 6 months after sustaining a fracture and the risk increases substantially with subsequent fractures [57]. The cumulative incidence of adverse outcomes following all low-trauma fractures leads to the death in 39% women and 51% men within 5 years and excess mortality related to fracture can extend up to 10 year. These mortality rates far exceed that expected for an age- and sex-matched population (24% in women and 27% in men) [58].
Osteoporosis: Gaps in Care
Osteoporosis is a preventable disease that physicians can diagnose and manage in the early stages of low bone mass. For reducing morbidity and mortality associated with osteoporosis-related fractures, it is imperative to recognize individuals at risk for osteoporosis. Yet, contrary to recommendations for universal screening and treatment, osteoporosis is vastly underdiagnosed and undertreated worldwide. Considering the reasons linked to such paradox, three gaps in patient care could be identified challenges facing healthcare professionals and policymakers responsible for providing care to populations in relation to bone health.
Gap 1: Failure to Follow Guidelines for Screening for Osteoporosis
Based on the analysis of medical claims data collected from a large American cohort between 2008 and 2014, screening rates among privately insured women ages 50+ were persistently low. Only 26.5% women in the age group 65–79 and 12.8% women 80 years and older had measurement for their bone mass. Even lower utilization rates were seen among non-Hispanic black women and women of low socioeconomic status [59].
There is also evidence that physicians who do screen may not be following recommended diagnostic guidelines and may be making the treatment decisions based on incorrect assumptions. The analysis of 5 years of electronic health and radiological records at a regional health care system in United States revealed two-thirds of women receiving new medication prescriptions for osteoporosis therapy did not need treatment. In fact, one half of the women being treated may not have qualified for screening at all, because they were of younger age and had no risk factors for osteoporosis [60]. Another study found that family physicians order bone densitometry and try to manage osteoporosis appropriately but lack a rationale for testing [61]. Surveys on physicians’ learning needs indicate the majority (66.8–83.2%) want to be informed about criteria for ordering and the interpretation of densitometry reports and T-scores and the frequency of testing [62, 63].
In conclusion, the first gap in the patients’ care lies in the hands of the physicians who need information regarding who and when to test, guideline-based diagnostic criteria and indications for testing, and information on how to interpret tests.
Gap 2: Failure of Secondary Fracture Prevention
Secondary fracture prevention is an obvious first step in the development of a systematic approach to prevention of all fragility fractures caused by osteoporosis. Early aggressive treatment intervention after a first low-trauma fracture, especially in those with low bone density, can reduce the risk of additional fractures and associated premature mortality. Since the 1980s, it has been reported that up to one half of hip fracture patients have already sustained a previous fracture [64,65,66,67]. Meta-analyses have shown that individuals who have sustained a fracture are at approximately double the risk of sustaining subsequent fractures, as compared to their fracture-free peers [68, 69]. However, data show that the percentage of patients receiving a treatment for osteoporosis, even after sustaining a hip fracture, has declined in the United States from 41% in 2001 to 21% in 2011. These numbers demonstrate a low participation of physicians in their patients’ secondary fracture prevention.
The effectiveness of the broad range of currently available osteoporosis treatments has been comprehensively reviewed [70]. Cochrane Collaboration systematic reviews have evaluated most of the anti-osteoporotic medications for secondary fracture prevention. The outcomes documented the effectiveness of the available osteoporosis treatments to reduce future fracture risk. Therefore, it is of great concern that a pervasive and persistent secondary prevention care gap is evident throughout the world. The International Osteoporosis Foundation (IOF) Capture the Fracture® program website provides an up-to-date bibliography of all PubMed cited secondary prevention audits and surveys, undertaken internationally, nationally, regionally, and locally [71].
In response to this widely documented care gap, models of care have been developed in many countries to ensure that fragility fracture patients receive secondary preventive care – which includes both osteoporosis management and intervention to prevent falls – in a consistent and reliable fashion. The most common models are referred to as orthogeriatrics services and fracture liaison services (FLS) [72].
In conclusion, physicians need information regarding the range of anti-osteoporotic agents available for treatment and how to select the appropriate one for each patient, drug safety profiles, dosing instructions, timing of initiation of medication, and how to treat patients at moderate risk for fracture. The implementation of services such as orthogeriatrics and fracture liaison service does help in secondary fracture prevention.
Gap 3: Patient-Physician Communication Failure
This lack of patient awareness and action is coupled with a lack of healthcare professional (HCP) awareness and intervention. Multiple studies demonstrate that patients diagnosed to have osteoporosis tend to underestimate their risk of becoming osteoporotic and are less concerned about the consequences of osteoporosis than other diseases. Earlier studies assessing osteoporotic patients who have multiple FRAX risk factors, one-third did not believe they were at an increased risk for future fracture [73, 74]. Even when patients have had fragility fractures, more than half do not link their fractures with osteoporosis even when told they have the disease, nor do they appear to understand they are at increased risk for future fracture [75].
Patient education on low bone mass and osteoporosis is imperative for long-term management of osteoporosis and fracture prevention. Therefore, it is crucial for physicians to communicate to patients that a diagnosis of osteoporosis, increasing age, or a fragility fracture increases the risk of future fracture. However, surveys and focus groups indicate primary care physicians feel there are barriers to communicating with elderly patients about the complexity of osteoporosis risk and fracture prevention, which include time constraints, the complexity of their other health problems, and their reluctance to add new medications to long lists of prescribed therapies [61, 63].
In conclusion, better approaches to deliver the message such as informative video clips, adverts, or information leaflets to highlight the magnitude of the issue to the patients, educate and empower them to actively participate in shared decision-making, and support self-care and medication adherence as well as persistence. Physicians need training to enable them to provide clear physician-patient communication and patient education that can help patients understand their risk and agree to adhere to a management pathway.
Osteoporosis: Mind the Treatment Gap
Despite the increasing number of effective drugs to treat osteoporosis, discouraging evidence suggests that there is a growing gap in treatment options. This has been evidenced by the finding that many patients who should receive pharmacological treatment are either not being offered these drugs or, when prescribed, not taking them [76]. This has also been reported in patients recovering from hip fracture, for whom there is universal agreement of the importance of pharmacological therapy [77]. Although many reasons exist for this gap in osteoporosis treatment, perhaps the two most important reasons are fear of rare side effects and concerns regarding long-term efficacy.
Fear of Rare Side Effects
In an article published earlier in the New York Times Gina Kolata [78], patient concerns with side effects, particularly atypical femur fractures, were highlighted as an important contributor to the lack of appropriate treatment for osteoporosis. Although these side effects have only been clearly associated with bisphosphonates, patient perceptions about these risks are extending to all osteoporosis drugs, which is particularly concerning because atypical femur fractures are extremely rare. So although the relative risk of atypical femur fractures in patients taking bisphosphonates is increased, the absolute risk ranges from 3.2 to 50 cases per 100,000 person-years [79]. When used in patients who are at high risk of fracture, these drugs are estimated to prevent 80–5000 fragility fractures for each atypical femur fracture possibly induced by treatment [80]. Several steps can be taken to address this problem [81], such as improved patient and doctor education regarding both the risk-benefit ratio of these drugs and the prodromal symptoms (e.g., groin or hip pain) of atypical femur fractures; potential use of dual x-ray energy absorptiometry to monitor patients on therapy specifically for features of atypical femur fractures [82]; identification of high-risk patients using femur geometrical characteristics and other risk factors for atypical femur fractures [83]; and the development of pharmacogenomic markers identifying patients at increased risk of atypical femur fractures.
A second rare side effect of bisphosphonate use is osteonecrosis of the jaw, which was initially described in the setting of high-dose bisphosphonate use in patients with metastatic cancer. This side effect is extremely rare in patients treated at doses recommended for osteoporosis, with an estimated incidence of 0.001–0.01% [84]. Again, better education of patients, doctors, and dental practitioners, along with maintenance of good oral hygiene and dental health, are key to overcoming this barrier to treatment.
Concerns Regarding Long-Term Efficacy
As highlighted by a position statement from the FDA [85], data regarding the anti-fracture efficacy of bisphosphonates after 5 years of use is scarce and perhaps conflicting. This assessment, combined with the observation that the risk of the rare side effects of atypical femur fractures and osteonecrosis of the jaw increases with duration of therapy [79, 84], has led to legitimate concerns about the long-term (>5 years) treatment of patients with bisphosphonates or other anti-resorptive agents, such as denosumab. However, data from the Fracture Intervention Trial Long-term Extension (FLEX) [86] showed that postmenopausal women with low hip T scores (−2.0 to −2.5) who continued treatment with alendronate for 10 years had fewer clinical vertebral fractures than women receiving placebo after 5 years had. Similarly, in the HORIZON extension study of zoledronic acid [87], women with T scores less than −2.5 had fewer morphometric vertebral fractures after six annual infusions than women who received only 3 years of treatment had. On the basis of these studies, current recommendations are to treat patients who warrant therapy with a bisphosphonate for 5 years and then reassess, basing subsequent treatment on the level of fracture risk and potentially considering a so-called drug holiday for a variable period of time, albeit in the absence of data showing the efficacy of this approach [88]. Long-term treatment (up to 10 years) with denosumab in an open-label extension of the FREEDOM trial has been shown to have a persistent benefit by reducing non-vertebral fractures [89].
Osteoporosis: Closing the Gaps
Osteoporosis is a preventable disease that physicians can diagnose and manage in the early stages of low bone mass. Recognition and management of individuals at risk for osteoporosis is imperative for reducing morbidity and mortality associated with osteoporosis-related fractures. This part of the chapter will provide a comprehensive overview of the state of osteoporosis care for individuals at high risk of suffering fragility fractures and how to close the gaps in both the patients’ care and treatment. The different ‘gaps’ identified are listed in Table 21.1; to facilitate their discussion and approach to handling, they have been clustered into four major themes. Given current projections indicating that the burden of fragility fracture is heavy and expected to grow over the coming few decades, it is imperative that governments, key opinion leaders, and national patient societies work together now to ensure that epidemiological data are available to inform policy development. There is much to be done. Therefore, the task now is to ensure the dissemination and adoption of these best practice examples, adapted for local considerations, in order to tackle the current, and future, burden of fragility fractures worldwide.
Closing Gap 1: Secondary Fracture Prevention
In 2012, the IOF issued a report on the World Osteoporosis Day devoted to the global Capture the Fracture® Campaign [64, 90]. Approximately half of the patients admitted with hip fracture suffered a prior fragility fracture in the months or few years before breaking their hip [64, 65, 67, 91], representing an obvious opportunity and, indeed, imperative for assessment and intervention to be carried out to prevent future fractures. The report also cited numerous audits undertaken across the world to establish what proportion of fracture patients received the osteoporosis care that they needed: in the absence of a systematic approach, less than a fifth received such care. Whilst some exciting progress has been made to close this care gap, many publications and initiatives since 2012 highlight that there is still a huge amount of work to be done throughout the world. This was reviewed in a dedicated publication by the international osteoporosis foundation [92]. Clinically effective models of care, namely, orthogeriatrics and fracture liaison services, have been developed in many countries to close the secondary prevention care gap in a highly cost-effective manner.
Models of Care: Orthogeriatrics Services and Fracture Liaison Services
In response to the well-documented secondary fracture prevention care gap, innovators throughout the world have developed models of care designed to ensure that health systems respond to the first fracture to prevent second and subsequent fractures.
-
Orthogeriatrics services (OGS): The need for effective orthopaedic–geriatric co-care of patients admitted to hospital with hip fractures is well recognised in professional guidance [93,94,95]. Such models of care focus on expediting surgery, ensuring optimal management of the acute phase through adherence to a care plan overseen by senior orthopaedic and geriatrician/internal medicine personnel, and delivery of secondary fracture prevention through osteoporosis management and falls prevention.
-
Fracture liaison services (FLS): The fracture liaison service (FLS) model of care has also been adopted in many countries. The purpose of an FLS is to ensure that all patients aged 50 years or over, who present to urgent care services with a fragility fracture, undergo fracture risk assessment and receive treatment in accordance with prevailing national clinical guidelines for osteoporosis. The FLS also ensures that falls risk is addressed among older patients through referral to appropriate local falls prevention services.
These two service models are entirely complementary. As the adoption of orthogeriatrics services for hip fracture sufferers becomes more widespread, orthogeriatrics services are increasingly likely to deliver secondary preventive care for these patients. As hip fractures constitute approximately 20% of all clinically apparent fragility fractures, in health systems which have implemented orthogeriatrics services, FLS will provide secondary preventive care for the other 80% of fragility fracture sufferers who have experienced fractures of the wrist, humerus, spine, pelvis, and other sites. This ‘division of labour’ is illustrated in the falls and fractures pyramid in Fig. 21.2, which was first presented in policy developed by the Department of Health for England in 2009 [96]. A similar approach has been advocated in Australia [97], Canada [98], New Zealand [99], and the United States [100, 101].
Orthogeriatrics services and fracture liaison services as components of a systematic approach. (Quoted from reference [96] under open access scheme)
For the vital role of secondary fracture prevention, as well as both orthogeriatrics services and FLS as a reliable means to deliver this care to fracture patients, this has been addressed and featured in a growing number of clinical guidelines and government policies.
Closing Gap 2: Medication-Induced Osteoporosis
Many widely used medicines have been associated with decreases in bone mineral density and/or increased fracture incidence, although these links have not been proven as causal in every case. A 2014 review described the potential pathogenesis of bone loss associated with all of these classes of medicines [102, 103]. Table 21.2 shows a list of these medications. This section will focus on three very commonly used agents: glucocorticoids for a range of conditions, androgen deprivation therapy for treatment of prostate cancer in men, and aromatase inhibitors for the treatment of hormone receptor-positive breast cancer in women.
Steroids-Induced Osteoporosis
Steroids are very commonly used to control inflammation in the setting of a broad range of conditions including autoimmune dermatological and respiratory diseases, as well as malignancies and organ transplants. Estimates suggest that 1 in 13 adults aged 18 years and over have been prescribed oral steroids at some stage of their life [103]. Up to 30–50% of patients receiving chronic glucocorticoid therapy experience clinically apparent fragility fractures and/or asymptomatic vertebral fractures, making steroid-induced osteoporosis the leading cause of secondary osteoporosis [104]. Meta-analysis has shown previous steroid use to be associated with a relative risk of 2 for any fracture at the age of 50 years and 1.7 at the age of 85 years [105]. For osteoporotic fracture, the range of relative risk is 2.6 and 1.7; and for hip fracture 4.4 and 2.5 for the same age groups.
Steroids have a direct impact on the bones as it affects both the function and numbers of the three major types of bone cells [106,107,108,109,110] (Fig. 21.3):
-
Osteoclasts: Stimulation by steroids results in prolonged survival of osteoclasts, leading to excessive bone resorption, particularly in trabecular bone in the spine.
-
Osteoblasts: By reducing the recruitment of the precursors to osteoblasts, the number of mature osteoblasts is reduced, resulting in decreased bone formation.
-
Osteocytes: Osteocyte apoptosis (cell death) is triggered by steroids and may contribute to an increase in fracture risk prior to a reduction in bone mineral density (BMD).
Direct effects of glucocorticoids on bone. PPARγ2: Peroxisome proliferator-activated receptor gamma receptor 2 (favoring the differentiation of pluripotent precursor cells to adipocytes in preference to osteoblasts). Sclerostin: It binds to the co-receptors for frizzled, Lrp4 and Lrp5, resulting in the inhibition of Wnt sigaling, leading to reduced differentiation of osteoblast precursors to mature osteoblasts and increased osteoblast and osteocyte apoptosis. M-CSF macrophage colony stimulating factor, RANKL receptor activator of nuclear factor-κB ligand, OPG osteoprotegerin. (Increased M-CSF and RANKL and decreasing production of OPG by osteoblastic cells and osteocytes result in an increase in both the number and activity of osteoclasts. This effect diminishes with time, possibly as a result of the reduction in number of osteoblasts and osteocytes)
Steroids’ indirect effects on bone: Other mechanisms that may contribute to glucocorticoid-induced bone loss through indirect effects on bone include hypogonadism, reduced physical activity, increased renal and intestinal losses of calcium, and reduced production of growth hormone, insulin-like growth factor 1 (IGF1), and IGF1 binding protein (IGF-BP) [111]. In addition, the underlying diseases for which glucocorticoid therapy is administered are often associated with increased inflammation, which contributes to bone loss through increased production of pro-inflammatory, pro-resorptive cytokines. Whilst glucocorticoids suppress inflammation and hence should mitigate the adverse effects of inflammation, disease relapse despite therapy is associated with episodes of increased bone resorption. Finally, glucocorticoid excess has adverse effects on muscle mass and function, leading to myopathy and increased risk of falls [112, 113].
To close this gap, clinical guidelines for the prevention and treatment of glucocorticoid-induced osteoporosis are already available in several countries. The European League Against Rheumatism (EULAR) [114] and a Joint Guideline Working Group of IOF and the European Calcified Tissue Society (ECTS) [115] have produced internationally relevant guidance. Whilst the detail of individual guidelines varies somewhat, the common theme is that individuals receiving chronic steroids therapy are at increased risk of fracture on account of taking steroids, and, in a significant proportion, the risk is great enough to warrant the offer of preventive treatment. An organized programme of care – GIOP (Glucocorticoid-Induced Osteoporosis Program) – has been implemented in the United States in order to improve preventive care for members [116]. The programme goals are to identify patients at risk of fracture, provide education, redesign and implement new pathways of care, and monitor outcomes.
Androgen Deprivation Therapy-Induced Osteoporosis
Pathogenesis: Androgen deprivation therapy (ADT) , in the form of gonadotropin-releasing hormone agonists (GnRHs), limits the production of testosterone and estradiol, leading to chemical castration [102]. GnRHs elicit this effect by reducing the secretion of luteinizing hormone and follicle-stimulating hormone. This is a consequence of GnRHs binding to GnRH receptors in the pituitary gland and downregulating the gonadotropin producing cells.
The beneficial clinical effects of ADT in men with symptomatic metastatic prostate cancer are rapid and dramatic [117]. ADT is universally accepted as the first-line treatment of symptomatic metastatic prostate cancer [118]. Prostate cancer is the most common non-cutaneous malignancy in men, with 1 in 6 men being diagnosed during their lifetime [119]. Approximately half of men diagnosed with prostate cancer will receive ADT at some stage after diagnosis [120]. A meta-analysis of relevant studies reported that between 9% and 53% of survivors had osteoporosis166. A rapid decline in BMD is observed during the first year of ADT treatment [121]. A cohort study based on medical claims data from Medicare beneficiaries in the United States compared fracture rates for men with non-metastatic prostate cancer who initiated GnRH agonist treatment against a comparison group who did not receive GnRH agonist treatment [122]. The men treated with GnRHs had statistically significantly higher rates of any clinical fracture (relative risk [RR]: 1.2), vertebral fractures (RR: 1.5), and hip/femur fractures (RR: 1.3). Longer duration of treatment also conferred greater fracture risk.
To close this gap, clinical guidelines relating to the prevention and treatment of ADT-induced osteoporosis have been published in several countries. Local clinical leaders in osteoporosis care should explore opportunities for collaboration with colleagues in urology departments to establish what proportion of ADT treated patients have undergone osteoporosis assessment and received guideline-based care.
Aromatase Inhibitor-Induced Osteoporosis
Pathogenesis: Aromatase inhibitors (AIs) reduce oestrogen levels by the inhibition of the peripheral conversion of androgens to oestrogens. This results in lower oestrogen levels with a consequent increase in bone turnover and bone loss.
Aromatase inhibitor use and fracture incidence: Breast cancer is the most common neoplasm and primary cause of cancer-related mortality in women, affecting 1 in 8 women worldwide. Aromatase Inhibitors currently represent the gold standard adjuvant treatment for postmenopausal women with hormone receptor-positive breast cancer [123]. The annual rate of bone loss observed for women taking Aromatase Inhibitors of around 2.5%. This figure is elevated compared to healthy postmenopausal women who lose about 1–2% per year [124].
The analysis of the Women’s Health Initiative Observational Study compared fracture rates among breast cancer survivors with women with no history of breast cancer at baseline [125]. After adjustment for factors related to hormone levels , risk of falls, prior fracture history, medication use, comorbidity, and lifestyle, the increased risk for all fractures studied among survivors was 15%. Studies comparing two commonly used aromatase inhibitors, anastrozole [126] and letrozole [127], with tamoxifen have reported significant increases in fracture risk for the aromatase inhibitor–treated patients. A comparative study of anastrozole with exemestane showed similar fracture rates [128]. A position paper from the European Society from Clinical and Economical Aspects of Osteoporosis (ESCEO) has comprehensively documented studies on the skeletal effects of aromatase inhibitors [124].
The care gap for aromatase inhibitor–induced osteoporosis has not been documented as comprehensively as the secondary fracture prevention and steroid-induced osteoporosis care gaps have been discussed. Clinical guidelines relating to the prevention and treatment of aromatase inhibitor–induced osteoporosis are available in several countries. Local clinical leaders in osteoporosis care should explore opportunities for collaboration with colleagues in oncology departments to establish what proportion of aromatase inhibitor–treated patients have undergone osteoporosis assessment and received guideline-based care.
Closing Gap 3: Diseases Associated with Osteoporosis
There are many health problems which can increase an individual’s risk of developing osteoporosis and suffering fragility fractures [129]. These include a broad array of disorders: autoimmune, digestive and gastrointestinal, endocrine and hormonal, hematologic, neurological, mental illness, cancer, and AIDS/HIV. These include also malabsorption, anorexia nervosa, primary or secondary hypogonadism, dementia, and diabetes.
In many of these conditions, there is lack of specific guidelines for management of osteoporosis, whereas clinical guidelines relating to the prevention and treatment of associated osteoporosis is available in some countries for some of these conditions. Working Group comprised of clinical experts in the field of COPD and fracture prevention published a 5-step approach which includes case finding, risk evaluation, differential diagnosis, therapy, and follow-up [130]. Clinical guidelines relating to the prevention and treatment of osteoporosis in celiac disease are available. Similarly, guidelines relating to the prevention and treatment of osteoporosis in inflammatory bowel disease (IBD) have been published [92].
The estimated number of people living with dementia in 2013 was estimated to be 44.4 million, a figure set to increase to 75.6 million and 135.5 million by 2030 and 2050, respectively. The largest increases in the projected number of dementia sufferers will be in East Asia and Sub-Saharan African regions. By 2050, the proportion living in what are currently low- and middle-income countries will increase to 71%, compared to 62% in 2013. In 2010, the global societal cost of dementia was US$604 billion, representing 1% of global GDP [131], and 486,000 people died as a result of dementia worldwide [132].
A significant overlap exists between sufferers of dementia and older people at high risk of injurious falls and fractures; this is particular evident amongst patients presenting with hip fracture. A UK study published in 2009 found that during a 12-month period, 66% of participants with dementia had a fall compared with 36% of age-matched controls [133]. Furthermore, the incidence of falls in dementia was nine times higher than that observed among a control group. The incidence of hip fracture among patients with Alzheimer’s disease has been reported to be almost three times higher than amongst cognitively healthy peers [134]. In a meta-analysis, the prevalence of dementia amongst older hip fracture patients was estimated to be 19% [135]. The prevalence of cognitive impairment was estimated at 42%. In 2007, the Scottish Hip Fracture Audit reported on the prevalence of dementia amongst hip fracture patients [136]. Over a quarter (28%) of patients had a documented past medical history of dementia, which the authors indicated was likely to be a significant underestimate of actual prevalence on account of the poor diagnosis rates for dementia documented at that time.
In 2011, a monograph on the subject of dementia, falls, and fractures summarised the current evidence [137]:
-
Persons with dementia suffer more falls, more fractures, and higher post-fracture mortality than those without dementia, yet they are under-assessed for falls risk factors and are less likely to receive treatment for osteoporosis.
-
Falls and fracture patients have a high prevalence of dementia and cognitive impairment, yet do not routinely receive cognitive assessment and, consequently, frequently miss an opportunity for a diagnosis of dementia to be made.
Subsequent studies from Canada [138], Finland [139], the United Kingdom [121], and the United States have added to the evidence that osteoporosis is infrequently diagnosed and treated in people living with dementia.
As the population of dementia sufferers is set to grow spectacularly in the coming decades, evidence-based guidelines for the management of osteoporosis – and falls risk – in dementia must be drafted and implemented as soon as possible.
Closing Gap 4: Primary Fracture Prevention
Whilst the prevention of secondary fractures remains a priority, in the long term, the ultimate goal would be the prevention of the first fracture. Advances in the fracture risk assessment during the last decade provide a platform for development of clinically effective and, crucially, cost-effective approaches for the identification of those individuals at high risk of a primary fracture. In order to ensure that a primary fracture prevention programme has the potential to be cost effective, consideration must be given to which first fragility fracture is to be prevented. Primary prevention of hip fracture is likely to be more cost-effective than primary prevention of wrist fracture, because hip fractures cost considerably more to manage than wrist fractures. In this regard, consideration must be given to what proportion of all hip fractures occur as an individual’s first fragility fracture at any skeletal site [93].
Whilst definitive data are not available to give an accurate estimate of the primary hip fracture incidence, the following illustration is consistent with the current evidence-base:
-
Approximately 50% of hip fracture patients have suffered clinically apparent fragility fracture(s) prior to breaking their hip, which was usually a non-vertebral fracture [64, 65, 67, 91].
-
Conservative interpretation of studies from Spain and Japan suggests that a further 10% [140] to 25% [141] of hip fracture patients may have suffered previous vertebral fractures – the majority of which are not recognised or diagnosed as such [142] – but have not suffered clinically apparent non-vertebral fractures.
-
Therefore, 25–40% of hip fracture patients may have suffered the hip fracture as their first overt fragility fracture at any skeletal site.
This analysis highlights the challenge faced by efforts to proactively case-find the relatively small proportion of individuals who are likely to suffer a hip fracture as their first fragility fracture. It should also be noted that fragility fractures at sites other than the hip impose a significant burden on older people.
Vertebral fractures lead to many adverse consequences for sufferers, including [143]:
-
Back pain, loss of height, deformity, immobility and increased number of hospital bed days [144, 145]
-
Reduced quality of life resulting from the loss of self-esteem, distorted body image, and depression [146,147,148,149]
-
A significant negative impact on activities of daily living [150, 151]
Studies from Australia [152], Canada [153], and the international Global Longitudinal Study of Osteoporosis in Women (GLOW) [154] have all reported significant reductions in health-related quality of life among individuals who have suffered fragility fractures at all skeletal sites. Accordingly, a robust clinical case exists for primary prevention of all major osteoporosis fractures, defined as hip, clinical vertebral, wrist or proximal humerus fractures.
To close this gap, pragmatic approaches to case-finding individuals at high risk of suffering these fractures as their first fracture should be adopted, these include:
-
Gap 2: Osteoporosis induced by medicines: systematic case finding of individuals at high fracture risk in this group
-
Gap 3: Diseases associated with osteoporosis: systematic case finding of individuals at high fracture risk in this group
-
Absolute fracture risk calculation: systematic application of tools such as FRAX® to risk stratify the older population
This is supported by most clinical guidelines which cover both secondary and primary fracture prevention
Closing Gap 5: Adherence to Therapy
Similar to other chronic, asymptomatic diseases, adherence to osteoporosis therapies is poor. The reasons for suboptimal adherence are multiple but include fear of possible side effects, dosing requirements, and an unwillingness to take a medication for a “silent” disease. Two measures of adherence to treatment are commonly used in studies:
-
Persistence: Defined as either the time to treatment discontinuation or as the proportion of patients that at a certain time point still fill prescriptions without a gap in refills longer than an allowed period of time (e.g., 30, 60 or 90 days).
-
Compliance: Defined as the ability of a patient to adhere to the dosing, timing, and conditions described by the prescriber or in accordance with the medicine’s patient information leaflet. One measure of compliance is the medication possession ratio (MPR). MPR is usually defined as the number of days of medication available to the patient, divided by the number of days of observation.
In routine clinical practice, both persistence and compliance with osteoporosis treatment are sub-optimal, a phenomenon previously reported for other classes of widely used medicines including antihypertensives [155] and statins [156]. Approximately half of patients initiated on osteoporosis treatment do not follow their prescribed treatment regimen and/or discontinue treatment within a year [157]. This is particularly notable on account of the flexibility of dosing options of widely available osteoporosis treatments, which can be taken as daily, weekly, or monthly tablets, or as daily, quarterly, six-monthly, or annual injections. Intravenous or sub-cutaneous routes of administration provide a means to ensure 100% adherence with treatment, as long as a robust system is in place to administer the initial injection and reliably arrange follow-up injections at appropriate intervals. It has been estimated that improved adherence in the United States would reduce fracture rates by 25%, equating to approximately 300,000 fewer fractures per year and generate savings of US$3 billion [158].
In a trial to close the gap, in 2013, the Medication Adherence and Persistence Special Interest Group of the International Society for Pharmacoeconomics and Outcomes Research (ISPOR) undertook a systematic literature review of interventions to improve osteoporosis medication adherence [159]. Interventions identified which may improve adherence were:
-
Simplification of dosing regimens
-
Electronic prescriptions
-
Patients’ decision aids
-
Patient education
Patients were most persistent with medications which had the least frequent dosing regimens [160,161,162]. The use of electronic prescriptions in combination with verbal counselling was associated with a 2.6-fold improvement in short-term compliance compared to verbal counselling alone [163]. A study from the United States evaluated the use of a patient decision aid in combination with usual primary care practice compared to usual primary care practice alone [164]. While adherence at 6 months was similar for both groups, the proportion with more than 80% adherence was significantly higher with the decision aid. With regard to the impact of patient education, it should be noted that the largest and least biased studies reviewed showed only marginal improvement in adherence [165,166,167,168].
The impact of FLS on adherence has been evaluated in several studies [169,170,171,172,173]. Among patients managed by an FLS after fracture, between 74% and 88% remained on treatment at 12 months, and between 64% and 75% at 24 months. These data reinforce the notion that a ‘teachable moment’ exists after individuals have suffered a fragility fracture which can be capitalized upon by an FLS to improve adherence to treatment.
Closing Gap 6: Public Awareness of Osteoporosis and Fracture Risk
Over the past 2 decades, a number of studies have been undertaken to characterise the awareness of osteoporosis and fracture risk among older people. In 2008, investigators from a non-profit Health Maintenance Organization (HMO) in the Northwest United States sought to evaluate key stakeholder perspectives on osteoporosis care after a fracture [174]. These stakeholders included fracture patients, quality and other healthcare managers, primary care physicians, and orthopaedic clinicians as well as staff. Both patients and primary care physicians commented that confusion of osteoporosis with osteoarthritis was common. Furthermore, this confusion led to the perception that osteoporosis is a benign consequence of ageing.
In 2010, Canadian investigators evaluated osteoporosis knowledge among older fracture patients who were treated by orthopaedic surgeons at two major teaching hospitals in Ontario [175]. Fractured patients were asked two questions:
-
1.
Do you know what osteoporosis is?
-
2.
If yes, what do you think it is?
The overwhelming majority of respondents (91%, 115/127) said they knew what osteoporosis was. Among these individuals, 75% gave responses that were considered to be correct. Almost 40% of the interview participants completed a ‘Facts on Osteoporosis Quiz’. Notably, less than half (41%) of those who took the quiz knew that a person who had suffered a spine fracture was at increased risk of suffering a fracture in the future as compared to a fracture-free individual.
The international GLOW study compared self-perception of fracture risk with actual risk among more than 60,000 postmenopausal women in 10 countries in Europe, North America, and Australia [176]. Key findings included:
-
Among women reporting a diagnosis of osteopenia or osteoporosis, only 25% and 43%, respectively, thought their risk was increased.
-
Among women whose actual risk was increased based on the presence of any one of seven fracture risk factors, the proportion who recognized their increased risk ranged from 19% for smokers to 39% for current users of glucocorticoid medication.
-
Only 33% of those with at least 2 risk factors perceived themselves as being at higher risk.
To close the gap, efforts to improve awareness need to provide clear, evidence-based messages.
Disease awareness campaigns (DACs) such as 2Million2Many from the NBHA in the United States provided an innovative example of implementing this approach [177]. The key messages for 2Million2Many are very simple and compelling:
-
Every year, there are two million bone breaks that are no accident (in the USA).
-
They are the signs of osteoporosis in people as young as 50.
-
But only 2 out of 10 get a simple follow-up assessment.
-
Together we can break osteoporosis before it breaks us. But we must speak up. Remember: Break a bone, request a test.
The main target of the disease awareness campaigns is to drive awareness throughout the population of the world that fracture begets fracture. If all individuals aged 50 years or over know that suffering a first fragility fracture significantly increases their risk of suffering second and subsequent fractures, up to one half of all people who will suffer hip fractures in the future could be aware of that risk, and be proactive in taking steps to lower it.
Closing Gap 7: Public Awareness of Benefits Versus Risks of Osteoporosis Treatment
Numerous RCTs and Cochrane Collaboration systematic reviews have demonstrated the efficacy and safety of treatments for osteoporosis. However, in the last decade use of these treatments among individuals at high risk of fracture has been significantly impacted by reports relating to rare side effects, including osteonecrosis of the jaw (ONJ), atrial fibrillation (AF), and atypical femur fracture (AFF).
Earlier studies documented that the risk-benefit calculation for the treatment of osteoporosis among individuals who are at high risk of suffering fragility fractures, including life-changing and life-threatening hip fractures, significantly favours treatment [80, 178, 179]. Patients at risk of osteoporotic fractures should not be discouraged from initiating bisphosphonates, because clinical trials have documented that these medicines can substantially reduce the incidence of typical hip fractures. The increased risk of atypical fractures should be taken into consideration when continuing bisphosphonates beyond 5 years [180].
To close the gap, public awareness of osteoporosis must be increased dramatically throughout the world. Effective disease awareness campaigns are needed to ensure that when an older person sustains a fragility fracture, their first thought – and that of their family and friends – is: ‘Did that bone break because of osteoporosis?’ Health professionals and their organisations, national patient societies, health system leaders, and regulatory agencies must work together to craft clear, balanced communications concerning the benefits and risks of treatments. In concordance, both clinicians and patients need to be able to objectively discuss and evaluate the risk-benefit calculation for the patient’s individual circumstances when making collaborative treatment decisions. Having ready access to absolute fracture risk calculation tools such as FRAX® can make such discussions far more tailored – and meaningful – to individual patients. It requires all those involved in the care of osteoporosis patients to ensure clear, balanced communication of these issues – to – individual patients and more widely when opportunities arise.
Closing the Gap 8: Access and Reimbursement for Osteoporosis Assessment and Treatment
During the last decade, IOF has undertaken a series of regional audits throughout the world [181,182,183,184,185,186]. These audits have evaluated epidemiology, costs, and the burden of osteoporosis in the regions, and have included an overview of access and reimbursement to treatment. Some countries had a very good reimbursement policy for diagnostic tools and therapies, while in other countries there was absolutely no reimbursement available and patients had to pay for all diagnostic tests and treatment.
In the United States reimbursement for treatment varies greatly depending on each patient’s health plan. Health care reform is evolving from fee for service to supporting improved quality, prevention, and care coordination with financial incentives (or penalties) to encourage healthcare professionals and health systems to report on and improve patient outcomes. There are a number of quality measures focused on osteoporosis and post-fracture care but performance around these measures remains low compared to other major chronic diseases. Further, a major drop in reimbursement for DXAs performed in the office setting has led to a drop in the number of providers and more than one million less DXAs performed [93].
Closing the Gap 9: Prioritization of Fragility Fracture Prevention in National Policy
The IOF regional audits provide comprehensive information on the level of priority afforded to fragility fracture prevention by governments throughout the world [181,182,183,184,185,186].
Osteoporosis guidelines have been endorsed by several governments all over the world; however, there has been quite variation regarding the designation of osteoporosis as a national health priority. The majority of EU states (18/27), as well as most of the developing world, did not recognize osteoporosis or musculoskeletal diseases as a national health priority (NHP).
In the United States, despite a landmark report by the Surgeon General in 2004 and the specific recommendations from key national and scientific societies intended to prioritize and improve osteoporosis and fracture prevention [187,188,189], implementation has been poor. Many patients are not given the necessary information about prevention and are not receiving appropriate testing to diagnose osteoporosis or establish osteoporosis risk. Most importantly, a majority of patients who have osteoporosis-related fractures are not being diagnosed with osteoporosis and are not receiving any of the Food and Drug Administration (FDA)–approved, effective therapies.
To close this gap, the provision of robust epidemiological estimates of fracture incidence throughout Asia-Pacific, Central Asia, Latin America, the Middle East, and Africa will be a critical step towards supporting development of fracture prevention policies for these rapidly aging populations.
Closing the Gap 10: The Burden of Osteoporosis in the Developing World
The developing world is set to bear the brunt of the burden of osteoporosis as the world’s population rapidly ages during the first half of this century. Accordingly, it is ironic that few data on fracture rates exist in many developing countries. The IOF regional audits provide valuable insights in this regard [181,182,183,184,185,186].
In Asia, there is an urgent need at the national level to accurately quantify osteoporosis and fracture prevalence in many countries of this region. In eastern Europe and central Asia the lack of solid epidemiological and economic data on the costs and burden of the disease has been linked to the under recognition of osteoporosis status on the side of both the governments and healthcare professionals. Similarly, in Latin America, regional Audit identified a major lack of data on fracture incidence in the region. In the middle east and Africa, The IOF Middle East and Africa Regional Audit identified a major lack of data on fracture incidence in the region in 2011 [186]. Only 6 of the 17 countries in the audit had published hip fracture incidence data. Further, prevalence rates for vertebral fractures were available for only 3 countries.
To close this gap, all governments need to establish osteoporosis as a national health priority, with commensurate human and financial resources to ensure that best practice is delivered for all.
patients in their jurisdictions. Where the current disease burden is not known, studies to close such evidence gaps must be commissioned forthwith.
Guidelines
There has been significant development in the guidelines published for the management of osteoporosis. In general, guideline provides recommendations based on current evidence for best practice in the management of osteoporosis and prevention of fractures. It addresses risk factors for fracture, commonly used tools for fracture risk assessment, approaches to targeting therapy, pharmacological, and non-pharmacological treatments to reduce fracture risk in different patient groups, treatment of painful vertebral fractures and systems of care. Sometimes, the assessment and prevention of falls and surgical management of fractures is included.
The most recent guidelines published is the guideline update of the pharmacological management of osteoporosis in postmenopausal women published by the Endocrine Society, USA [190]. The Guideline Update is a document that permits rapid and focused communication to guideline stakeholders in response to new developments that substantially impact the recommendations of an existing clinical practice guideline (e.g., important new drug approval or withdrawal, important new risks or harms). This Guideline Update is published in response to the recent approval of romosozumab by the United States Food and Drug Administration (FDA), the European Medicines Agency, Health Canada, and other agencies; and it represents a formal amendment to the Endocrine Society’s recently published clinical practice guideline regarding the pharmacological management of postmenopausal osteoporosis [191].
The guideline for the management of postmenopausal osteoporosis is designed to provide the clinician with an evidence-based approach to the management of this condition. The guidelines (Fig. 21.4) stratify the patients according to their risk of fracture, using that FRAX algorithm, into four risk categories:
-
1.
Low risk: It includes no prior hip or spine fractures, a BMD T-score at the hip and spine both above −1.0, a 10-year hip fracture risk <3%, and 10-year risk of major osteoporotic fractures <20%.
-
2.
Moderate risk: It includes no prior hip or spine fractures, a BMD T-score at the hip and spine both above −2.5, and 10-year hip fracture risk <3% or risk of major osteoporotic fractures <20%.
-
3.
High risk: It includes a prior spine or hip fracture, or a BMD T-score at the hip or spine of −2.5 or below, or 10-year hip fracture risk ≥3%, or risk of major osteoporotic fracture risk ≥20%.
-
4.
Very high risk: It includes multiple spine fractures and a BMD T-score at the hip or spine of −2.5 or below.
Updated algorithm for the management of postmenopausal osteoporosis. (Quoted with permission from the American endocrine society update [190])
NOGG (UK) provided another approach to stratification of osteoporotic fracture risk combining FRAX risk assessment and BMD measurement (Fig. 21.5). The intervention threshold is set at a risk equivalent to that associated with a prior fracture. Two bounds around the intervention threshold where the assessment of BMD will help to determine whether the individual close to the threshold either exceeds that bound or lies below the intervention threshold. These are called assessment threshold for bones. Very high risk is identified as the risk lying above the upper assessment threshold, whereas high risk lies between the intervention threshold and the upper assessment threshold. On the other hand, low risk is reported when the risk lies below the intervention threshold. Figure 21.6 shows a suggested algorithm for the management of postmenopausal women adopting the recent recommendations. Recommendation regarding drug holiday and further assessment including repeat FRAX measurement has also been outlined in the guidelines.
Infographic outlining of the characterisation of fracture risk by FRAX major osteoporotic fracture probability in postmenopausal women. FRAX probability in the intermediate (orange) zone should be followed by BMD assessment and recalculation of FRAX probability including femoral neck BMD. After recalculation, risk located in the red zone, above the intervention threshold (2), is identified as “very high risk”, orange zone above the intervention threshold (1) is identified as “high risk,” whereas risk below the intervention threshold (1) or in the green zone is identified as “low risk”
Case finding and treatment pathways according to the categorisation of fracture risk: updated algorithm for the management of postmenopausal osteoporosis. The determination of fracture risk was carried out based on fracture risk score calculation (e.g., FRAX) and the measurement of lumbar spine and hip BMD. *Stratification of osteoporotic fracture risk can be based on NOGG (UK) as shown in the figures. The intervention threshold is set at a risk equivalent to that associated with a prior fracture. Two intervention thresholds are identified based on FRAX calculation based on BMD assessment. The treatment modality is suggested based on whether the individual either exceeds the intervention threshold or lies below it. Alternatively, using FRAX score alone, the fracture risks can be defined as follows: (1) low risk includes no prior hip or spine fractures, a BMD T-score at the hip and spine both above −1.0, a 10-year hip fracture risk <3%, and 10-year risk of major osteoporotic fractures <20%; (2) moderate risk includes no prior hip or spine fractures, a BMD T-score at the hip and spine both above −2.5, and 10-year hip fracture risk <3% or risk of major osteoporotic fractures <20%; (3) high risk includes a prior spine or hip fracture, or a BMD T-score at the hip or spine of −2.5 or below, or 10-year hip fracture risk ≥3%, or risk of major osteoporotic fracture risk ≥20%; and (4) very high risk includes multiple spine fractures and a BMD T-score at the hip or spine of −2.5 or below [190]. **Continue treatment up to 3 years (IV zoledronate) or 5 years (oral bisphosphonate / denosumab), reassess fracture risk: 1. if low or low-moderate risk, consider drug holiday. Reassess fracture risk every 2–4 years; if bone loss, fracture occurs or patient becomes high risk consider restarting therapy. 2. If high risk, continue therapy after checking for adherence or switch to another therapy. ***After the completion of the anabolic therapy course, consider giving bisphosphonate, then stopping for a drug holiday. Reassess fracture risk every 1–3 years. If bone loss, fracture occurs, or patient becomes high risk, consider restarting therapy
In conclusion, this chapter has outlined a stepwise approach to case finding individuals who are at high risk of sustaining fragility fractures. By first closing the secondary fracture prevention care gap, up to half of individuals who would otherwise fracture their hip could be treated to prevent this debilitating and costly injury. Integration of bone health and falls risk assessments into the management of individuals who take medicines which have adverse effects on bone must become standard practice. Similarly, individuals who are diagnosed with diseases which feature osteoporosis as a common comorbidity need to receive care that will minimise their fracture risk. When the needs of these obviously high-risk groups have been addressed, we must turn our attention to the development of cost-effective strategies to prevent the first major osteoporotic fracture.
References
Johnell O, Kanis JA. An estimate of the worldwide prevalence and disability associated with osteoporotic fractures. Osteoporos Int. 2006;17(12):1726–33.
Blume SW, Curtis JR. Medical costs of osteoporosis in the elderly Medicare population. Osteoporos Int. 2011;22(6):1835–44.
Nelson HD, Haney EM, Dana T, et al. Screening for osteoporosis: an update for the U.S. preventive services task force. Ann Intern Med. 2010;153:99–111.
Cummings SR, Black DM, Nevitt MC, et al. Bone density at various sites for prediction of hip fractures. The study of osteoporotic fractures research group. Lancet. 1993;341(8837):72–5.
National Osteoporosis Foundation. Clinician’s guide to prevention and treatment of osteoporosis. Washington, DC: National Osteoporosis Foundation; 2010.
Greenspan SL, Wyman A, Hooven FH, et al. Predictors of treatment with osteoporosis medications after recent fragility fractures in a multinational cohort of postmenopausal women. J Am Geriatr Soc. 2012;60(3):455–61.
Solomon DH, Johnston SS, Boytsov NN, et al. Osteoporosis medication use after hip fracture in U.S. patients between 2002 and 2011. J Bone Miner Res. 2014;29(9):1929–37.
Liu Z, Weaver J, de Papp A, et al. Disparities in osteoporosis treatments. Osteoporos Int. 2016;27(2):509–19.
Cuddihy M, Gabriel SE, Crowson CS, et al. Osteoporosis intervention following distal forearm fractures: a missed opportunity? Arch Intern Med. 2002;162:421–6.
Andrade SE, Majumdar SR, Chan KA, et al. Low frequency of treatment of osteoporosis among postmenopausal women following a fracture. Arch Intern Med. 2003;163(17):2052–7.
Freedman KB, Kaplan FS, Bilker WB, et al. Treatment of osteoporosis: are physicians missing an opportunity? J Bone Joint Surg Am. 2000;82:1063–70.
Jennings LA, Auerbach AD, Maselli J, et al. Missed opportunities for osteoporosis treatment in patients hospitalized for hip fracture. J Am Geriatr Soc. 2010;58:650–7.
The Joint Commission. Improving and measuring osteoporosis management. Oakbrook Terrace: The Joint Commission; 2007.
Elliot-Gibson V, Bogoch ER, Jamal SA, et al. Practice patterns in the diagnosis and treatment of osteoporosis after a fragility fracture: a systematic review. Osteoporos Int. 2004;15:767–78.
Torgerson DJ, Dolan P. Prescribing by general practitioners after an osteoporotic fracture. Ann Rheum Dis. 1998;57:378–9.
Giangregorio L, Papaioannou A, Cranney A, et al. Fragility fractures and the osteoporosis care gap: an international phenomenon. Semin Arthritis Rheum. 2006;35(5):293–305.
Bessette L, Ste-Marie LG, Jean S, et al. The care gap in diagnosis and treatment of women with a fragility fracture. Osteoporos Int. 2008;19:79–86.
Kamel HK, Hussain MS, Tariq S, et al. Failure to diagnose and treat osteoporosis in elderly patients hospitalized with hip fracture. Am J Med. 2000;109:326–8.
Rabenda V, Vanoverloop J, Fabri V, et al. Low incidence of antiosteoporosis treatment after hip fracture. J Bone Joint Surg Am. 2008;90(10):2142–8.
Balasubramanian A, Tosi LL, Lane JM, Dirschi DR, Ho PR, O’Malley CD. Declining rates of osteoporosis management following fragility fractures in the U.S., 2000 through 2009. J Bone Joint Surg Am. 2014;96(7):e52.
Kim SC, Kim MS, Sanfélix-Gimeno G, et al. Use of osteoporosis medications after hospitalization for hip fracture: a cross-national study. Am J Med. 2015;128(5):519–5.
Hernlund E, Svedbom A, Ivergård M, et al. Osteoporosis in the European Union: Medical Management, Epidemiology and Economic Burden. A report prepared in collaboration with the International Osteoporosis Foundation (IOF) and the European Federation of Pharmaceutical Industry Associations (EFPIA). Arch Osteoporos. 2013;8:136.
National Osteoporosis Foundation. Clinician’s guide to prevention and treatment of osteoporosis. Washington, DC: National Osteoporosis Foundation; 2014. Available from: http://nof.org/files/nof/public/content/file/2791/upload/919.pdf.
Papaioannou A, Morin S, Cheung AM, et al. Scientific Advisory Council of Osteoporosis Canada. 2010 clinical practice guidelines for the diagnosis and management of osteoporosis in Canada: summary. CMAJ. 2010;182:1864–73.
Klotzbuecher CM, Ross PD, Landsman PB, Abbott TA III, Berger M. Patients with prior fractures have an increased risk of future fractures: a summary of the literature and statistical synthesis. J Bone Miner Res. 2004;15:721–39.
Johnell O, Oden A, Caulin F, Kanis JA. Acute and long term increase in fracture risk after hospitalization for vertebral fracture. Osteoporos Int. 2001;12:207–14.
Johnell O, Kanis JA, Odén A, et al. Fracture risk following an osteoporotic fracture. Osteoporos Int. 2004;15:175–9.
Lindsay R, Silverman SL, Cooper C, et al. Risk for new vertebral fracture in the year following a fracture. JAMA. 2001;285:320–3.
Díez-Pérez A, Hooven FH, Adachi JD, et al. Regional differences in treatment for osteoporosis. The global longitudinal study of osteoporosis in women (GLOW). Bone. 2011;49:493–8.
Guggina P, Flahive J, Hooven FH, et al. Characteristics associated with anti-osteoporosis medication use: data from the Global Longitudinal Study of Osteoporosis in Women (GLOW) USA cohort. Bone. 2012;51:975–80.
Jennings LA, Auerbach AD, Maselli J, Pekow PS, Lindenauer PK, Lee SJ. Missed opportunities for osteoporosis treatment in patients hospitalized for hip fracture. J Am Geriatr Soc. 2010;58:650–7.
Freedman KB, Kaplan FS, Bilker WB, Strom BL, Lowe RA. Treatment of osteoporosis: are physicians missing an opportunity? J Bone Joint Surg Am. 2000;82-A:1063–70.
Giangregorio L, Papaioannou A, Cranney A, Zytaruk N, Adachi JD. Fragility fractures and the osteoporosis care gap: an international phenomenon. Semin Arthritis Rheum. 2006;35:293–305.
Nayak S, Roberts MS, Greenspan SL. Factors associated with diagnosis and treatment of osteoporosis in older adults. Osteoporos Int. 2009;20:1963–7.
Vaile J, Sullivan L, Bennett C, Bleasel J. First fracture project: addressing the osteoporosis care gap. Intern Med J. 2007;37:717–20.
Papaioannou A, Giangregorio L, Kvern B, Boulos P, Ioannidis G, Adachi JD. The osteoporosis care gap in Canada. BMC Musculoskelet Disord. 2004;5:11.
Bessette L, Ste-Marie LG, Jean S, et al. Recognizing osteoporosis and its consequences in Quebec (ROCQ): background, rationale, and methods of an anti-fracture patient health-management programme. Contemp Clin Trials. 2008;29:194–210.
Elliot-Gibson V, Bogoch ER, Jamal SA, Beaton DE. Practice patterns in the diagnosis and treatment of osteoporosis after a fragility fracture: a systematic review. Osteoporos Int. 2004;15:767–78.
Haaland DA, Cohen DR, Kennedy CC, Khalidi NA, Adachi JD, Papaioannou A. Closing the osteoporosis care gap: increased osteoporosis awareness among geriatrics and rehabilitation teams. BMC Geriatr. 2009;9:28.
Swedish National Board of Health and Welfare. Quality and efficiency in Swedish health care. Stockholm: Swedish Association of Local Authorities and Regions, Swedish National Board of Health and Welfare; 2009.
Borgstrom F, Johnell O, Kanis JA, Jonsson B, Rehnberg C. At what hip fracture risk is it cost-effective to treat? International intervention thresholds for the treatment of osteoporosis. Osteoporos Int. 2006;17:1459–71.
Austin P, Tu J, Ko D, Alter D. Factors associated with the use of evidence-based therapies after discharge among elderly patients with myocardial infarction. CMAJ. 2008;179:901–8.
Office of the Surgeon General (US). Bone health and osteoporosis: a report of the surgeon general. Rockville: Office of the Surgeon General (US); 2004.
Ambrose AF, Cruz L, Paul G. Falls and fractures: a systematic approach to screening and prevention. Maturitas. 2015;82(1):85–93.
Magaziner J, Hawkes W, Hebel JR, et al. Recovery from hip fracture in eight areas of function. J Gerontol A Biol Sci Med Sci. 2000;55(9):M498–507.
Lenze EJ, Munin MC, Skidmore ER, et al. Onset of depression in elderly persons after hip fracture: implications for prevention and early intervention of late-life depression. J Am Geriatr Soc. 2007;55(1):81–6.
Singer A, Exuzides A, Spangler L, O’Malley C, Colby C, Johnston K, Agodoa I, Baker J, Kagan R. Burden of illness for osteoporotic fractures compared with other serious diseases among postmenopausal women in the United States. Mayo Clin Proc. 2015;90(1):53–62.
Burge R, Dawson-Hughes B, Solomon DH, Wong JB, King A, Tosteson A. Incidence and economic burden of osteoporosis-related fractures in the United States, 2005–2025. J Bone Miner Res. 2007;1;22(3):465–75.
Johansson H, Siggeirsdóttir K, Harvey NC, Odén A, Gudnason V, McCloskey E, Sigurdsson G, Kanis JA. Imminent risk of fracture after fracture. Osteoporos Int. 2016;27:1–6.
Johnell O, Kanis J. Epidemiology of osteoporotic fractures. Osteoporos Int. 2005;16(2):S3–7.
Holt G, Smith R, Duncan K, et al. Changes in population demographics and the future incidence of hip fracture. Injury. 2009;40(7):722–6.
Langsetmo L, Goltzman D, Kovacs CS, Adachi JD, Hanley DA, Kreiger N, Josse R, Papaioannou A, Olszynski WP, Jamal SA. Repeat low-trauma fractures occur frequently among men and women who have osteopenic BMD. J Bone Miner Res. 2009;24(9):1515–22.
Kanis JA, Johnell O, De Laet C, Johansson H, Oden A, Delmas P, Eisman J, Fujiwara S, Garnero P, Kroger H, McCloskey EV, Mellstrom D, Melton LJ, Pols H, Reeve J, Silman A, Tenenhouse A. A meta-analysis of previous fracture and subsequent fracture risk. Bone. 2004;35(2):375–82. Epub 2004 Jul 23
Center JR, Bliuc D, Nguyen TV, Eisman JA. Risk of subsequent fracture after low-trauma fracture in men and women. JAMA. 2007;297(4):387–94.
Haentjens P, Magaziner J, Colón-Emeric CS, Vanderschueren D, Milisen K, Velkeniers B, Boonen S. Meta-analysis: excess mortality after hip fracture among older women and men. Ann Intern Med. 2010;152(6):380–90.
Kanis JA, Oden A, Johnell O, De Laet C, Jonsson B. Excess mortality after hospitalization for vertebral fracture. Osteoporos Int. 2004;15(2):108–12.
Bliuc D, Nguyen ND, Milch VE, Nguyen TV, Eisman JA, Center JR. Mortality risk associated with low-trauma osteoporotic fracture and subsequent fracture in men and women. JAMA. 2009;301(5):513–21.
Bliuc D, Nguyen ND, Nguyen TV, Eisman JA, Center JR. Compound risk of high mortality following osteoporotic fracture and refracture in elderly women and men. J Bone Miner Res. 2013;28(11):2317–24.
Gillespie CW, Morin PE. Trends and disparities in osteoporosis screening among women in the United States, 2008-2014. Am J Med. 2017;130(3):306–16.
Fenton JJ, Robbins JA, Amarnath AL, Franks P. Osteoporosis overtreatment in a regional health care system. JAMA Intern Med. 2016;176(3):391–3.
Jaglal SB, Carroll J, Hawker G, McIsaac WJ, Jaakkimainen L, Cadarette SM, Cameron C, Davis D. How are family physicians managing osteoporosis? Qualitative study of their experiences and educational needs. Can Fam Physician. 2003;49(4):462–8.
Ritchard J, Karampatos S, Ioannidis G, Adachi J, Thabane L, Nash L, Mehan U, Kozak J, Feldman S, Hirsch S, Jovaisas AV. Osteoporosis guideline implementation in family medicine using electronic medical records survey of learning needs and barriers. Can Fam Physician. 2016;62(6):e326–33.
Jaglal SB, McIsaac WJ, Hawker G, Carroll J, Jaakkimainen L, Cadarette SM, Cameron C, Davis D. Information needs in the management of osteoporosis in family practice: an illustration of the failure of the current guideline implementation process. Osteoporos Int. 2003;14(8):672–6.
Gallagher JC, Melton LJ, Riggs BL, Bergstrath E. Epidemiology of fractures of the proximal femur in Rochester, Minnesota. Clin Orthop Relat Res. 1980;150:163–71.
Port L, Center J, Briffa NK, Nguyen T, Cumming R, Eisman J. Osteoporotic fracture: missed opportunity for intervention. Osteoporos Int. 2003;14:780–4.
McLellan A, Reid D, Forbes K, Reid R, Campbell C, Gregori A, Raby N, Simpson A. Effectiveness of strategies for the secondary prevention of osteoporotic fractures in Scotland (CEPS 99/03). Glasgow: NHS Quality Improvement Scotland; 2004.
Edwards BJ, Bunta AD, Simonelli C, Bolander M, Fitzpatrick LA. Prior fractures are common in patients with subsequent hip fractures. Clin Orthop Relat Res. 2007;461:226–30.
Klotzbuecher CM, Ross PD, Landsman PB, Abbott TA 3rd, Berger M. Patients with prior fractures have an increased risk of future fractures: a summary of the literature and statistical synthesis. J Bone Miner Res Off J Am Soc Bone Miner Res. 2000;15:721–39.
Kanis JA, Johnell O, De Laet C, et al. A meta-analysis of previous fracture and subsequent fracture risk. Bone. 2004;35:375–82.
Kanis JA, EV MC, Johansson H, Cooper C, Rizzoli R, Reginster JY, Scientific Advisory Board of the European Society for C, Economic Aspects of O, Osteoarthritis, the Committee of Scientific Advisors of the International Osteoporosis F. European guidance for the diagnosis and management of osteoporosis in postmenopausal women. Osteoporos Int. 2013;24:23–57.
International Osteoporosis Foundation. Capture the Fracture® program website: audits & surveys page. http://www.capturethefracture.org/audits-surveys. Accessed 14 Mar 2020.
Harvey N, McCloskey E, Mitchell P, Dawson-Hughes B, Pierroz D, Reginster J, Rizzoli R, Cooper C, Kanis J. Mind the (treatment) gap: a global perspective on current and future strategies for prevention of fragility fractures. Osteoporos Int. 2017;28:1507–29.
Hsieh C, Novielli KD, Diamond JJ, Cheruva D. Health beliefs and attitudes toward the prevention of osteoporosis in older women. Menopause. 2001;8:372–6.
Siris ES, Gehlbach S, Adachi JD, Boonen S, Chapurlat RD, Compston JE, Cooper C, Delmas P, Diez-Perez A, Hooven FH, LaCroix AZ. Failure to perceive increased risk of fracture in women 55 years and older: the Global Longitudinal Study of Osteoporosis in Women (GLOW). Osteoporos Int. 2011;22(1):27–35.
Giangregorio L, Papaioannou A, Thabane L, Cranney A, Dolovich L, Adili A, Adachi JD. Do patients perceive a link between a fragility fracture and osteoporosis? BMC Musculoskelet Disord. 2008;9(1):38.
Khosla S, Shane E. A crisis in the treatment of osteoporosis. J Bone Miner Res. 2016;31:1485–7.
Eisman JA, Bogoch ER, Dell R, et al. for the ASBMR Task Force on Secondary Fracture Prevention. Making the first fracture the last fracture: ASBMR Task Force on Secondary Fracture Prevention. J Bone Miner Res. 2012;27:2039–46.
Kolata, G. Accessed 6 Apr 2017. New York Times. 1 Jun 2016.; http://www.nytimes.com/2016/06/02/health/osteoporosis-drugs-bones.html?_r=0
Shane E, Burr D, Abrahamsen B, et al. Atypical subtrochanteric and diaphyseal femoral fractures: second report of a task force of the American Society for Bone Mineral Research. J Bone Miner Res. 2014;29:1–23.
Black DM, Rosen CJ. Clinical practice. Postmenopausal osteoporosis. N Engl J Med. 2016;374:254–62.
Khosla S, Cauley JA, Compston J, et al. Addressing the crisis in the treatment of osteoporosis: a path forward. J Bone Miner Res. 2017;32:424–30.
McKenna MJ, van der Kamp S, Heffernan E, Hurson C. Incomplete atypical femoral fractures: assessing the diagnostic utility of DXA by extending femur length. J Clin Densitom. 2013;16:579–83.
Mahjoub Z, Jean S, Leclerc JT, et al. Incidence and characteristics of atypical femoral fractures: clinical and geometrical data. J Bone Miner Res. 2013;31:767–76.
Khosla S, Burr D, Cauley J, et al. Bisphosphonate-associated osteonecrosis of the jaw: report of a task force of the American Society for Bone and Mineral Research. J Bone Miner Res. 2007;22:1479–91.
Whitaker M, Guo J, Kehoe T, Benson G. Bisphosphonates for osteoporosis—where do we go from here? N Engl J Med. 2012;366:2048–51.
Black DM, Schwartz AV, Ensrud KE, et al. Effects of continuing or stopping alendronate after 5 years of treatment The Fracture Intervention Trial Long-Term Extension (FLEX): a randomized trial. JAMA. 2006;296:2927–38.
Black DM, Reid IR, Boonen S, et al. The effect of 3 versus 6 years of zoledronic acid treatment of osteoporosis: a randomized extension to the HORIZON-Pivotal Fracture Trial (PFT). J Bone Miner Res. 2012;27:243–54.
Adler RA, El-Hajj Fuleihan G, Bauer DC, et al. Managing osteoporosis in patients on long-term bisphosphonate treatment: report of a task force of the American Society for Bone and Mineral Research. J Bone Miner Res. 2016;31:16–35.
Bone HG, Wagman RB, Brandi ML, et al. 10 years of denosumab treatment in postmenopausal women with osteoporosis: results from the phase 3 randomised FREEDOM trial and open-label extension. Lancet Diabetes Endocrinol. 2017;5:513–23.
International Osteoporosis Foundation. Capture the Fracture® Programme website. http://www.capture-thefracture.org/. Accessed 14 Mar 2020.
McLellan A, Reid D, Forbes K, et al. Effectiveness of strategies for the secondary prevention of osteoporotic fractures in Scotland (CEPS 99/03). Glasgow: NHS Quality Improvement Scotland; 2004.
Gaps and Solutions in Bone Health. 2016. http://share.iofbonehealth.org/WOD/2016/thematic-report/2016TR-key-messages.pdf. Accessed on 15 Mar 2020.
British Orthopaedic Association, British Geriatrics Society. The care of patients with fragility fracture. 2007.
Australian and New Zealand Hip Fracture Registry (ANZHFR) Steering Group. Australian and New Zealand guideline for hip fracture care: improving outcomes in hip fracture management of adults. Sydney: Australian and New Zealand Hip Fracture Registry Steering Group; 2014.
Kates SL, Mears SC, Sieber F, et al. A guide to improving the care of patients with fragility fractures. Geriatr Orthop Surg Rehabil. 2011;2(1):5–37.
Department of Health. Falls and fractures: Effective interventions in health and social care. In: Department of Health, editor. 2009.
New South Wales Agency for Clinical Innovation Musculoskeletal Network. NSW Model of Care for Osteoporotic Refracture Prevention. Chatswood, NSW; 2011.
Osteoporosis Canada. Make the FIRST break the LAST with fracture liaison services 2013.
Osteoporosis New Zealand. Bone care 2020: a systematic approach to hip fracture care and prevention for New Zealand. Wellington: Osteoporosis New Zealand; 2012.
National Osteoporosis Foundation. The Surgeon General’s Report: 10 years later. Washington, DC 2014.
Mitchell PJ, Cooper C, Dawson-Hughes B, Gordon CM, Rizzoli R. Life-course approach to nutrition. Osteoporos Int. 2015;26(12):2723–42.
Panday K, Gona A, Humphrey MB. Medication-induced osteoporosis: screening and treatment strategies. Ther Adv Musculoskelet Dis. 2014;6(5):185–202.
Henneicke H, Gasparini SJ, Brennan-Speranza TC, Zhou H, Seibel MJ. Glucocorticoids and bone: local effects and systemic implications. Trends Endocrinol Metab. 2014;25(4):197–211.
Lbaum JM, Youn S, Levesque LE, Gershon AS, Cadarette SM. Osteoporosis management among chronic glucocorticoid users: a systematic review. J Popul Ther Clin Pharmacol. 2014;21(3):e486–504.
Canalis E, Mazziotti G, Giustina A, Bilezikian JP. Glucocorticoid-induced osteoporosis: pathophysiology and therapy. Osteoporos Int. 2007;18(10):1319–28.
Wu Z, Bucher NLR, Farmer SR. Induction of peroxisome proliferator-activated receptor γ during the conversion of 3T3 fibroblasts into adipocytes is mediated by C/EBPh, C/EBPy, and glucocorticoids. Mol Cell Biol. 1996;16:4128–36.
Weinstein RS, Jilka RL, Parfitt AM, Manolagas SC. Inhibition of osteoblastogenesis and promotion of apoptosis of osteoblasts and osteocytes by glucocorticoids—potential mechanisms of their deleterious effects on bone. J Clin Invest. 1998;102:274–82.
Ohnaka K, Tanabe M, Kawate H, Nawata H, Takayanagi R. Glucocorticoid suppresses the canonical Wnt signal in cultured human osteoblasts. Biochem Biophys Res Commun. 2005;329:177–81.
Swanson C, Lorentzon M, Conaway HH, Lerner UH. Glucocorticoid regulation of osteoclast differentiation and expression of receptor activator of nuclear factor-kappaB (NF-kappaB) ligand, osteoprotegerin, and receptor activator of NF-kappaB in mouse calvarial bones. Endocrinology. 2006;147(7):3613–22.
Hofbauer LC, Gori F, Riggs BL, Lacey DL, Dunstan CR, Spelsberg TC, Khosla S. Stimulation of osteoprotegerin ligand and inhibition of osteoprotegerin production by glucocorticoids in human osteoblasts: potential paracrine mechanisms of glucocorticoid-induced osteoporosis. Endocrinology. 1999;140:4382–9.
Mazziotti G, Formenti AM, Adler RA, Bilezikian JP, Grossman A, Sbardella E, Minisola S, Giustina A. Glucocorticoid induced osteoporosis: pathophysiological role of GH/IGF-I and PTH/VITAMIN D axes, treatment options and guidelines. Endocrine. 2016;54(3):603–11.
Sato AY, Richardson D, Cregor M, Davis HM, Au ED, McAndrews K, Zimmers TA, Organ JM, Peacock M, Plotkin LI, Bellido T. Glucocorticoids induce bone and muscle atrophy by tissue-specific mechanisms upstream of E3 ubiquitin ligases. Endocrinology. 2017;158(3):664–77.
Compston J. Glucocorticoid-induced osteoporosis: an update. Endocrine. 2018;61:7–16.
Duru N, van der Goes MC, Jacobs JW, et al. EULAR evidence-based and consensus-based recommendations on the management of medium to high-dose glucocorticoid therapy in rheumatic diseases. Ann Rheum Dis. 2013;72(12):1905–13.
Ekamwasam S, Adachi JD, Agnusdei D, et al. A framework for the development of guidelines for the management of glucocorticoid-induced osteoporosis. Osteoporos Int. 2012;23(9):2257–76.
Newman ED, Matzko CK, Olenginski TP, et al. Glucocorticoid-Induced Osteoporosis Program (GIOP): a novel, comprehensive, and highly successful care program with improved outcomes at 1 year. Osteoporos Int. 2006;17(9):1428–34.
Huggins C, Hodges CV. Studies on prostatic cancer II: the effects of castration on advanced carcinoma of the prostate gland. Arch Surg. 1941;43:209–23.
Perlmutter MA, Lepor H. Androgen deprivation therapy in the treatment of advanced prostate cancer. Rev Urol. 2007;9(Suppl 1):S3–8.
Stangelberger A, Waldert M, Djavan B. Prostate cancer in elderly men. Rev Urol. 2008;10(2):111–9.
Damji AN, Bies K, Alibhai SM, Jones JM. Bone health management in men undergoing ADT: examining enablers and barriers to care. Osteoporos Int. 2015;26(3):951–9.
Mittan D, Lee S, Miller E, Perez RC, Basler JW, Bruder JM. Bone loss following hypogonadism in men with prostate cancer treated with GnRH analogs. J Clin Endocrinol Metab. 2002;87(8):3656–61.
Smith MR, Lee WC, Brandman J, Wang Q, Botteman M, Pashos CL. Gonadotropin-releasing hormone agonists and fracture risk: a claims-based cohort study of men with nonmetastatic prostate cancer. J Clin Oncol. 2005;23(31):7897–903.
Dowsett M, Cuzick J, Ingle J, et al. Meta-analysis of breast cancer outcomes in adjuvant trials of aromatase inhibitors versus tamoxifen. J Clin Oncol. 2010;28(3):509–18.
Rizzoli R, Body JJ, DeCensi A, et al. Guidance for the prevention of bone loss and fractures in postmenopausal women treated with aromatase inhibitors for breast cancer: an ESCEO position paper. Osteoporos Int. 2012;23(11):2567–76.
Chen Z, Maricic M, Bassford TL, et al. Fracture risk among breast cancer survivors: results from the women’s health initiative observational study. Arch Intern Med. 2005;165(5):552–8.
Cuzick J, Sestak I, Baum M, et al. Effect of anastrozole and tamoxifen as adjuvant treatment for early-stage breast cancer: 10-year analysis of the ATAC trial. Lancet Oncol. 2010;11(12):1135–41.
Coates AS, Keshaviah A, Thurlimann B, et al. Five years of letrozole compared with tamoxifen as initial adjuvant therapy for postmenopausal women with endocrine-responsive early breast cancer: update of study BIG 1-98. J Clin Oncol. 2007;25(5):486–92.
Goss PE, Ingle JN, Pritchard KI, et al. Exemestane versus anastrozole in postmenopausal women with early breast cancer: NCIC CTG MA.27--a randomized controlled phase III trial. J Clin Oncol. 2013;31(11):1398–404.
National Osteoporosis Foundation. Diseases and conditions that may cause bone loss. http://nof.org/articles/5. Accessed 14 Mar 2020.
Romme EA, Geusens P, Lems WF, et al. Fracture prevention in COPD patients; a clinical 5-step approach. Respir Res. 2015;16:32.
Wimo A, Prince M. World Alzheimer report 2010: the global economic impact of dementia. London: Alzheimer’s Disease International; 2010.
Lozano R, Naghavi M, Foreman K, et al. Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: a systematic analysis for the global burden of disease study 2010. Lancet. 2012;380(9859):2095–128.
Allan LM, Ballard CG, Rowan EN, Kenny RA. Incidence and prediction of falls in dementia: a prospective study in older people. PLoS One. 2009;4(5):e5521.
Baker NL, Cook MN, Arrighi HM, Bullock R. Hip fracture risk and subsequent mortality among Alzheimer’s disease patients in the United Kingdom, 1988-2007. Age Ageing. 2011;40(1):49–54.
Seitz DP, Adunuri N, Gill SS, Rochon PA. Prevalence of dementia and cognitive impairment among older adults with hip fractures. J Am Med Dir Assoc. 2011;2(8):556–64.
NHS National Services Scotland. Scottish hip fracture audit rehabilitation report 2007. 2007.
Mitchell PJ, Bateman K. Dementia, falls and fractures: integrated approaches to improve quality and reduce costs. Camberley: Novartis UK Ltd; 2012.
Knopp-Sihota JA, Cummings GG, Newburn-Cook CV, Homik J, Voaklander D. Dementia diagnosis and osteoporosis treatment propensity: a population-based nested case control study. Geriatr Gerontol Int. 2014;14(1):121–9.
Tiihonen M, Taipale H, Tanskanen A, Tiihonen J, Hartikainen S. Incidence and duration of cumulative bisphosphonate use among community-dwelling persons with or without Alzheimer’s disease. J Alzheimers Dis. 2016;52(1):127–32.
Sosa Henriquez M, Saavedra Santana P. grupo de trabajo en osteoporosis de la Sociedad Espanola de Medicina I. [Prevalence of vertebral fractures in hip fracture patients]. Rev Clin Esp. 2007;207(9):464–8.
Imai N, Endo N, Hoshino T, Suda K, Miyasaka D, Ito T. Mortality after hip fracture with vertebral compression fracture is poor. J Bone Miner Metab. 2016;34(1):51–4.
Delmas PD, van de Langerijt L, Watts NB, et al. Underdiagnosis of vertebral fractures is a worldwide problem: the IMPACT study. J Bone Miner Res. 2005;20(4):557–63.
Mitchell PJ, Dolan L, Sahota O, et al. Breaking point report 2015. London: Breaking Point Editorial Board; 2015.
Nevitt MC, Ettinger B, Black DM, et al. The association of radiographically detected vertebral fractures with back pain and function: a prospective study. Ann Intern Med. 1998;128(10):793–800.
Lips P, Cooper C, Agnusdei D, et al. Quality of life in patients with vertebral fractures: validation of the Quality of Life Questionnaire of the European Foundation for Osteoporosis (QUALEFFO). Working Party for Quality of Life of the European Foundation for Osteoporosis. Osteoporos Int. 1999;10(2):150–60.
Gold DT. The nonskeletal consequences of osteoporotic fractures. Psychologic and social outcomes. Rheum Dis Clin N Am. 2001;27(1):255–62.
Robbins J, Hirsch C, Whitmer R, Cauley J, Harris T. The association of bone mineral density and depression in an older population. J Am Geriatr Soc. 2001;49(6):732–6.
Lyles KW. Osteoporosis and depression: shedding more light upon a complex relationship. J Am Geriatr Soc. 2001;49(6):827–8.
Osteson AN, Gabriel SE, Grove MR, Moncur MM, Kneeland TS, Melton LJ 3rd. Impact of hip and vertebral fractures on quality-adjusted life years. Osteoporos Int. 2001;12(12):1042–9.
Hall SE, Criddle RA, Comito TL, Prince RL. A case-control study of quality of life and functional impairment in women with long-standing vertebral osteoporotic fracture. Osteoporos Int. 1999;9(6):508–15.
Adachi JD, Ioannidis G, Olszynski WP, et al. The impact of incident vertebral and non-vertebral fractures on health related quality of life in postmenopausal women. BMC Musculoskelet Disord. 2002;3:11.
Abimanyi-Ochom J, Watts JJ, Borgstrom F, et al. Changes in quality of life associated with fragility fractures: Australian arm of the International Cost and Utility Related to Osteoporotic Fractures Study (AusICUROS). Osteoporos Int. 2015;26(6):1781–90.
Tarride JE, Burke N, Leslie WD, et al. Loss of health related quality of life following low-trauma fractures in the elderly. BMC Geriatr. 2016;16(1):84.
Roux C, Wyman A, Hooven FH, et al. Burden of non-hip, non-vertebral fractures on quality of life in postmenopausal women: the Global Longitudinal study of Osteoporosis in Women (GLOW). Osteoporos Int. 2012;23(12):2863–71.
Burnier M. Medication adherence and persistence as the cornerstone of effective antihypertensive therapy. Am J Hypertens. 2006;19(11):1190–6.
De Vera MA, Bhole V, Burns LC, Lacaille D. Impact of statin adherence on cardiovascular disease and mortality outcomes: a systematic review. Br J Clin Pharmacol. 2014;78(4):684–98.
Seeman E, Compston J, Adachi J, et al. Non-compliance: the Achilles’ heel of anti-fracture efficacy. Osteoporos Int. 2007;18(6):711–9.
Siris ES, Selby PL, Saag KG, Borgstrom F, Herings RM, Silverman SL. Impact of osteoporosis treatment adherence on fracture rates in North America and Europe. Am J Med. 2009;122(2 Suppl):S3–13.
Hiligsmann M, Salas M, Hughes DA, et al. Interventions to improve osteoporosis medication adherence and persistence: a systematic review and literature appraisal by the ISPOR Medication Adherence & Persistence Special Interest Group. Osteoporos Int. 2013;24(12):2907–18.
Cooper A, Drake J, Brankin E, Investigators P. Treatment persistence with once-monthly ibandronate and patient support vs. once-weekly alendronate: results from the PERSIST study. Int J Clin Pract. 2006;60(8):896–905.
Freemantle N, Satram-Hoang S, Tang ET, et al. Final results of the DAPS (Denosumab adherence preference satisfaction) study: a 24-month, randomized, crossover comparison with alendronate in postmenopausal women. Osteoporos Int. 2012;23(1):317–26.
Kendler DL, McClung MR, Freemantle N, et al. Adherence, preference, and satisfaction of postmenopausal women taking denosumab or alendronate. Osteoporos Int. 2011;22(6):1725–35.
Hill DA, Cacciatore M, Lamvu GM. Electronic prescribing influence on calcium supplementation: a randomized controlled trial. Am J Obstet Gynecol. 2010;202(3):236. e231-235.
Montori VM, Shah ND, Pencille LJ, et al. Use of a decision aid to improve treatment decisions in osteoporosis: the osteoporosis choice randomized trial. Am J Med. 2011;124(6):549–56.
Nielsen D, Ryg J, Nielsen W, Knold B, Nissen N, Brixen K. Patient education in groups increases knowledge of osteoporosis and adherence to treatment: a two-year randomized controlled trial. Patient Educ Couns. 2010;81(2):155–60.
Shu AD, Stedman MR, Polinski JM, et al. Adherence to osteoporosis medications after patient and physician brief education: post hoc analysis of a randomized controlled trial. Am J Manag Care. 2009;15(7):417–24.
Solomon DH, Iversen MD, Avorn J, et al. Osteoporosis telephonic intervention to improve medication regimen adherence: a large, pragmatic, randomized controlled trial. Arch Intern Med. 2012;172(6):477–83.
Yuksel N, Majumdar SR, Biggs C, Tsuyuki RT. Community pharmacist-initiated screening program for osteoporosis: randomized controlled trial. Osteoporos Int. 2010;21(3):391–8.
Boudou L, Gerbay B, Chopin F, Ollagnier E, Collet P, Thomas T. Management of osteoporosis in fracture liaison service associated with long-term adherence to treatment. Osteoporos Int. 2011;22(7):2099–106.
Eekman DA, van Helden SH, Huisman AM, et al. Optimizing fracture prevention: the fracture liaison service, an observational study. Osteoporos Int. 2014;25(2):701–9.
Ganda K, Schaffer A, Pearson S, Seibel MJ. Compliance and persistence to oral bisphosphonate therapy following initiation within a secondary fracture prevention program: a randomised controlled trial of specialist vs. non-specialist management. Osteoporos Int. 2014;25(4):1345–55.
Dehamchia-Rehailia N, Ursu D, Henry-Desailly I, Fardellone P, Paccou J. Secondary prevention of osteoporotic fractures: evaluation of the Amiens University Hospital’s fracture liaison service between January 2010 and December 2011. Osteoporos Int. 2014;25(10):2409–16.
Chandran M, Cheen M, Ying H, Lau TC, Tan M. Dropping the ball and falling off the care wagon. Factors correlating with nonadherence to secondary fracture prevention programs. J Clin Densitom. 2016;19(1):117–24.
Feldstein AC, Schneider J, Smith DH, et al. Harnessing stakeholder perspectives to improve the care of osteoporosis after a fracture. Osteoporos Int. 2008;19(11):1527–40.
Giangregorio L, Thabane L, Cranney A, et al. Osteoporosis knowledge among individuals with recent fragility fracture. Orthop Nurs. 2010;29(2):99–107.
Siris ES, Gehlbach S, Adachi JD, et al. Failure to perceive increased risk of fracture in women 55 years and older: the Global Longitudinal Study of Osteoporosis in Women (GLOW). Osteoporos Int. 2011;22(1):27–35.
National Bone Health Alliance. 2 Million 2 Many. http://www.2million2many.org/. Accessed 15 Mar 2020.
Black DM, Kelly MP, Genant HK, et al. Bisphosphonates and fractures of the subtrochanteric or diaphyseal femur. N Engl J Med. 2010;362(19):1761–71.
Abrahamsen B. Older women who use bisphosphonate for longer than 5 years may have increased odds of a subtrochanteric or femoral shaft fracture, but absolute risk is low. Evid Based Med. 2011;16(6):168–9.
Reyes C, Hitz M, Prieto-Alhambra D, Abrahamsen B. Risks and benefits of bisphosphonate therapies. J Cell Biochem. 2016;117(1):20–8.
International Osteoporosis Foundation. The Asia-Pacific regional audit: epidemiology, costs and burden of osteoporosis in 2013. Nyon; 2013.
International Osteoporosis Foundation. The Eastern European & Central Asian Regional Audit: epidemiology, costs & burden of osteoporosis in 2010 2011.
Kanis JA, Borgstrom F, Compston J, et al. SCOPE: a scorecard for osteoporosis in Europe. Arch Osteoporos. 2013;8(1-2):144.
Svedbom A, Hernlund E, Ivergard M, et al. Osteoporosis in the European Union: a compendium of country-specific reports. Arch Osteoporos. 2013;8(1-2):137.
International Osteoporosis Foundation. The Latin America regional audit: epidemiology, costs & burden of osteoporosis in 2012. Nyon; 2012.
International Osteoporosis Foundation. The Middle East & Africa regional audit: epidemiology, costs & burden of osteoporosis in 2011 2011.
National Osteoporosis Society. Fracture Liaison service implementation toolkit. https://www.nos.org.uk/healthprofessionals/fracture-liaison-service/implementation-toolkit. Accessed 8 Mar 2020.
Watts NB, Adler RA, Bilezikian JP, et al. Osteoporosis in men: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2012;97(6):1802–22.
Cosman F, de Beur SJ, LeBoff MS, et al. Clinician’s guide to prevention and treatment of osteoporosis. Osteoporos Int. 2014;25(10):2359–81.
Shoback D, Rosen C, Black D, Cheung A, Murad M, Eastell R. Pharmacological management of osteoporosis in postmenopausal women: an endocrine society guideline update. J Clin Endocrinol Metab. 2020;105(3):1–8.
Eastell R, Rosen CJ, Black DM, Cheung AM, Murad MH, Shoback D. Pharmacological management of osteoporosis in postmenopausal women: an endocrine society* clinical practice guideline. J Clin Endocrinol Metab. 2019;104(5):1595–622.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 Springer Nature Switzerland AG
About this chapter
Cite this chapter
El Miedany, Y. (2022). Osteoporosis Management: Gaps in Patients’ Care and Treatment. In: El Miedany, Y. (eds) New Horizons in Osteoporosis Management. Springer, Cham. https://doi.org/10.1007/978-3-030-87950-1_21
Download citation
DOI: https://doi.org/10.1007/978-3-030-87950-1_21
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-87949-5
Online ISBN: 978-3-030-87950-1
eBook Packages: MedicineMedicine (R0)