Abstract
The enzymatic gelation of milk is one of the most studied topics in food colloids, as the reaction is based on the gradual hydrolysis of a polyelectrolyte layer on the surface of the casein micelles. The loss of the polyelectrolyte layer causes the destabilization of these protein particles in milk, and results in aggregates and a gel network. This chapter discusses the mechanisms of rennet-induced gelation of milk, with particular attention to the release of caseinomacropeptide from κ-casein and the rennet-induced aggregation of casein micelles. These two phases are strictly dependent on temperature, pH and the presence of calcium and other components in milk. The recent findings on the effects of heating, high pressure and concentration on enzymatic coagulation are also described.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
11.1 Introduction
The process of cheese making is based on a simple, specific, proteolytic reaction that causes the destabilization of protein particles in milk, which aggregate and form a gel network. The final structure of the gel will be a function of other factors, including, but not limited to, pH, calcium concentration and milk processing history. By modulating the early stages of structure formation or manipulating the cheese curd it is possible to obtain the large variety of cheese products available today.
Cheese curd is a protein-based gel in which the hydrolyzed casein micelles form a continuous network containing fat globules. The reaction is initiated by the enzyme chymosin (in most rennets), which hydrolyzes specifically κ-casein, a protein present on the surface of the casein micelles. Upon hydrolysis of this protein, the overall free energy of association between the micelles decreases and the casein particles stick to one another forming a three-dimensional gel.
The present chapter will outline the current understanding of the mechanism of rennet-induced gelation of the proteins in milk. The reader is also encouraged to consult previous reviews for fundamental aspects related to the kinetics of the enzymatic reaction as well as the models of rennet coagulation, as they have been well described (Hyslop, 2003; Horne and Banks, 2004). This chapter will focus on the preliminary stages of aggregation, as well as the recent findings on the effect of environmental and processing conditions on the enzymatic gelation of casein micelles. The effect of extrinsic factors on the aggregation of caseins in milk continues to be an important topic, as manufacturing practices evolve, and products are no longer obtained only using traditional methods, but other ingredients and more advanced processing technologies are put in place for increasing yields, modifying texture, creating novel products, responding to the demands of a very competitive market.
A number of experimental approaches have been developed over the years to follow the primary stages of rennet-induced aggregation of the casein micelles, as these initial coagulation steps are thought to be critical to the development of the final gel network. However, much less is understood, from a fundamental standpoint, about the rearrangements of the protein network after gelation, as studies at the molecular level become really challenging at this point. This phase is critical and affected by intrinsic and extrinsic factors, such as for example, temperature, salt concentration, time of cutting, or manipulations of the gel. These manipulations after gelation as well as ripening will not be discussed in this chapter, as they are beyond the scope of this review; however, they ultimately affect the final properties of the cheese (Fox et al., 2000, 2004).
11.2 Rennets
Rennet coagulation of milk is a key step in the preparation of several dairy products. Traditionally, the separation of milk into curds and whey is caused by the addition of an extract from the abomasum (the fourth and last compartment of the stomach of ruminants). The most common rennet extract derives from unweaned calves; however, in the Mediterranean areas, rennet paste from small ruminants (lambs or kids) is also used to produce cheeses. In this case, the final products are characterized by complex flavor profiles due to the presence of other enzymes, for example, pre-gastric and gastric lipases in the rennet extracts. These lipolytic enzymes are absent from calf rennet or are present at much lower concentrations.
Animal rennet extracts are mainly comprised of three enzymes, belonging to the pepsin-like family of aspartic proteinases: chymosin (EC 3.4.23.4), pepsin A (EC 3.4.23.1), and gastricsin (EC 3.4.23.3). The proportion of these enzymes depends on the feeding regime and the age of the animal at slaughter. In particular, the proportion of pepsin increases in adult cattle, whilst chymosin is reduced. Chymosin is the specific milk-clotting enzyme contained in rennet, and it cleaves the Phe105-Met106 bond of κ-casein, while pepsin A and gastricsin are generic proteases, which exhibit less specific activity.
For cheese production, it is critical to have a weak general proteolytic activity and narrow specificity. High proteolytic activity will result in the production of small and medium-sized peptides, which will impart bitter or unpleasant notes to the cheese and reduce yield. The clotting activity, expressed as International Milk-Clotting Units (IMCU), refers to the specific ability of the enzyme to cleave the Phe105-Met106 bond of κ-casein, starting the coagulation process, while the general proteolytic activity is linked to the hydrolysis of peptide bonds elsewhere in the casein system. A low ratio of clotting (C)/proteolytic (P) activity may cause a reduction of cheese yield, losses of fat and protein in the whey and the onset of defects, such as bitterness, during cheese ripening. The residues of the enzyme in cheese curd can lead to the formation of peptides responsible for bitter taste during ripening, and also have a role in cheese texture development.
There are other sources of coagulant enzymes generally indicated as rennet substitutes, and may have different origins (recombinant, animal, microbial and vegetable). The performances of calf rennet and fermentation-produced chymosins in terms of yield and C/P ratio are higher than for other enzymes (Barbano and Rasmussen, 1992; Crabbe, 2004).
Nowadays, most chymosin is produced by fermentation. Calf chymosin (genetic variant B) is widely produced through recombinant DNA technology by microorganisms such as Aspergillus niger and Kluyveromyces lactis (Jaros et al., 2008). Camel chymosin is also commercially available, produced by Aspergillus niger. This enzyme has 85 % sequence identity with bovine chymosin and when added to cow’s milk, it has a reduced generic proteolytic ability with 70 % higher specificity compared to bovine chymosin (Kappeler et al., 2006). Bovine chymosin, on the other hand, cannot cause gelation of camel milk (Kappeler et al., 2006). These differences are attributed to the differences in the amino acid residues of the substrate binding site, causing a better association between camel chymosin and κ-casein in bovine milk (Jensen et al., 2013). Other recombinant chymosins have been obtained from goat and buffalo. In particular, goat chymosin seems to have the highest catalytic specificity compared to the other enzymes (Vallejo et al., 2012).
Rennet substitutes from other sources, such as animals, and plants or microorganisms have been used for cheese production. The substitutes of animal origin are extracts of pepsin A from pig or chicken stomachs. Microbial coagulants, containing proteinases (EC 3.4.23.6) are commercially extracted from three microorganisms: Rhizomucor miehei, Rhizomucor pusillus and Cryphonectria parasitica. Plant proteinases are extensively used in the production of raw sheep and goat milk cheeses from Spain and Portugal. These coagulants (mainly two aspartic proteinases such as cardosin A and B) are extracted from the flowers of Cynara cardunculus, and are characterized by a high specific milk-clotting activity, but also by a higher proteolytic activity throughout cheese ripening compared to chymosin or calf rennet (Galán et al., 2008, 2012; Pino et al., 2009). The higher proteolytic activity of plant enzymes compared to bovine chymosin allows optimal sensory characteristics for cheeses to be obtained in shorter times, causing an acceleration of the ripening process. Mathematical models have been proposed to relate the concentration of these enzymes with milk clotting time, and to evaluate their proteolytic activity on different milk samples (Silva and Malcata, 2005).
11.2.1 Chymosin
Chymosin belongs to the family of aspartic proteinases, which are widely distributed in many organisms and tissues. The structure-function relationships of various aspartic proteinases have been extensively reviewed (Chitpinityol and Crabbe;, 1998; Crabbe, 2004). Bovine chymosin is a globular protein consisting of 323 residues and presenting a 55 % sequence homology with pepsin. Natural calf chymosin is found in three isoforms: A, B and C. The difference between A and B forms is related to a different amino acid residue in the position 243 of the molecular chain, which is an Asp in the A form and a Gly in the B form (Chitpinityol and Crabbe, 1998). The isoform B is the most abundant form and most stable, while chymosin C is a degradation product of the A isoform (Danley and Geoghegan, 1988).
The three-dimensional structure and active site geometry of aspartic proteinases have been widely studied using X-ray crystallography (Gilliland et al., 1990; Sielecki et al., 1990; Newman et al., 1991, 1993). A high resolution (1.8 Å) study has been recently published using X-ray synchrotron data (Jensen et al., 2013). The major differences among aspartic proteinases are related to a basic region for the substrate binding specificity, the so-called flap region, the movement of which is essential for enzyme-substrate binding. In bovine chymosin, this flap corresponds to amino acid residues 73–85.
The substrate-binding pockets of chymosin for κ-casein have been previously described (Gilliland et al., 1990; Newman et al., 1991; Chitpinityol and Crabbe, 1998). Both bovine and camel chymosins have positively charged patches on their surface, which are responsible for interactions with the negatively charged κ-casein (Jensen et al., 2013). Bovine chymosin has two similar β-barrel domains, with an active site consisting of Asp-Thr-Gly amino acid residues in N- and C-terminal lobes of chymosin (Newman et al., 1991). An interdomain region held together by hydrogen bridges is present between the two domains, often referred to as “fireman’s grip”. This area consists of two catalytic aspartic residues containing an activated water molecule (Gilliland et al., 1990; Sielecki et al., 1990; Newman et al., 1991). The specificity of chymosin for κ-casein arises from the amino acid sequence in the region 98–102 of the substrate molecule. This sequence consists of alternating His and Pro residues, acting as an allosteric regulator, converting the self-inhibited form of chymosin to the active form (Visser et al., 1987; Gutchina et al., 1996). This amino acid cluster binds to the C-terminal domain of chymosin (Palmer et al., 2010). The two lysine residues at positions 111 and 112 contribute to form positively charged clusters on the surface of κ-casein, that facilitate the docking of the chymosin enzyme at the cleavage site (Farrell et al., 1999). Camel chymosin differs from bovine chymosin by two amino acid residues: Lys221 and Val223 in bovine chymosin are substituted with Val221 and Phe223 in the camel enzyme. This difference, as already mentioned, affects the geometric and charge characteristics of the substrate binding site, improving its specificity for κ-casein (Palmer et al., 2010; Sørensen et al., 2011; Jensen et al., 2013).
11.3 The Initial Stages of Milk Renneting
κ-casein molecules are not free in solution, but are part of a supramolecular structure, the casein micelle. The location of κ-casein on the casein micelles is of particular relevance to rennet-induced gelation of milk. The cleavage of κ-casein causes destabilization of the micelles, and precipitation of the proteins. Casein micelles are highly hydrated particles (about 3.5 g of water per g of protein) which are composed of four different caseins (αs1-,αs2-, β-and κ-casein), with a diameter ranging between 80 and 400 nm. More details on the structure of these particles can be found elsewhere (see for example, Dalgleish and Corredig, 2012; de Kruif et al., 2012). The caseins are held together by non-covalent forces; hydrogen bonding, electrostatic interactions, van der Waals, hydrophobic interactions, and salt bridges between phosphoserine residues and colloidal calcium phosphate, are all present in the micelles to form small clusters (about 800 of them) of 2 nm in size (de Kruif et al., 2012). Figure 11.1 shows the microstructure of casein micelles as analyzed using cryo-transmission electron microscopy (cryo-TEM). The micelles appear spherical in nature with a diameter around 140 nm, and with a rugged surface. The darker areas in the images have been attributed to the calcium phosphate nanoclusters (Marchin et al., 2007). κ-Casein lacks phosphate centers and cannot participate in the formation of the nanoclusters; hence, while β-casein and αs-caseins are predominantly found in the interior, κ-casein is present on the surface of the casein micelles (Dalgleish et al., 1989). The protein extends in solution forming a polyelectrolyte layer (“the hairy layer”). This layer creates steric and charge repulsion between the protein particles, providing stability (de Kruif and Zhulina, 1996). The gelation of casein micelles by renneting is then the result of two important reactions: the hydrolysis of the polyelectrolyte layer of the casein micelles and the destabilization and aggregation of the casein micelles.
During the enzymatic reaction, the hydrodynamic diameter of the casein micelles, measured by dynamic light scattering, decreases by about 10 nm (Walstra et al., 1981; Horne and Davidson, 1993). The viscosity of the milk also shows a decrease, which has been attributed to the change in the hydrodynamic radius of the micelles (Scott Blair and Oosthuizen, 1961). However, the decrease in viscosity is not apparent when renneting occurs at a lower pH than the natural pH of milk, for example pH 6.2 (van Hooydonk et al., 1986). At a lower pH, there is a partial collapse of the hydrated κ-casein layer. It is then clear that the rennet reaction on κ-casein is complex, as the hydrolysis occurs on the surface of a much larger colloidal structure, and it is not a simple hydrolysis reaction on protein molecules in solution.
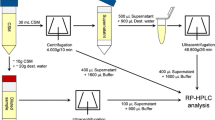
Rennet causes the release of the C-terminal portion of κ-casein, often referred to as the caseinomacropeptide (CMP), which diffuses away from the casein surface. A frequently used method to follow the extent of the reaction in milk is the determination of non-protein nitrogen (NPN) in the soluble phase; however, this method may not be sensitive enough to measure initial proteolysis rates. The release of CMP is better quantified using HPLC, after acid precipitation of the proteins, using trichloroacetic acid or perchloric acid (see for example, van Hooydonk and Olieman, 1982; López-Fandiño et al., 1993).
In unheated milk, all the κ-casein is accessed by the enzyme. The optimum pH for the enzymatic activity is 5.5–5.3, where the rate of cleavage is maximal (Visser et al., 1980; van Hooydonk et al., 1984). At lower pH it has been reported that general proteolysis on the other caseins (αs1-, αs2-, β-caseins) occurs (Lindqvist and Storgards, 1960), with important consequences for cheese ripening.
Several models have been proposed to describe the kinetics of gelation of the casein micelles; these aspects have been thoroughly examined in previous editions of this book (see for example, Hyslop, 2003). Most authors suggest that the reaction is a first order reaction (van Hooydonk et al., 1984). There are differences in the kinetics of the reaction between proteolysis of κ-casein in solution or in native casein micelles, as chymosin has strong affinity for para-κ-casein (Dalgleish, 1979; Dunnewind et al., 1996; de Roos et al., 2000).
The rate of the reaction is proportional to enzyme or protein concentration, and the relationship is linear for protein concentrations up to 8 % using skim milk concentrated by ultrafiltration (van Hooydonk et al., 1984). The initial velocity of the reaction reaches a plateau at higher milk protein concentrations (Garnot and Corre, 1980). It has been suggested that the reduction in the rate of the reaction with concentration may be due to reduction of the effective diffusion rate of the enzyme (van Hooydonk et al., 1984); however, it is important to note that processing history or serum composition may also have an effect. The reaction is strongly dependent on temperature, pH, calcium concentration, or micellar structure, and the effect of concentration on the rate of the enzymatic reaction, as well as the coagulation has yet to be fully elucidated.
There seems to be general agreement that the chymosin reaction is diffusion controlled, with a low energy of activation (van Hooydonk et al., 1984) and a constant reaction rate, which is affected by the volume fraction of the casein micelles. The enzyme is active on a very heterogeneous system; it is known that κ-casein can form disulphide-linked oligomeric aggregates (Groves et al., 1998) and it is probably present in this form in native casein micelles. The protein is not homogeneously present on the surface of the particles, as there is not enough κ-casein to cover the entire surface area (Dalgleish, 1998; Dalgleish and Corredig, 2012). The rate depends on the number of effective collisions, whereby the micelles diffuse very slowly compared to the rate of enzyme diffusion (van Hooydonk et al., 1984).
Chymosin activity is affected by ionic strength and type of ions present. The casein micelles are also strongly affected by ionic strength and in general, serum composition and it is challenging to distinguish between the effects of ionic strength on the reaction and that on the casein micelles structure. However, it has been demonstrated that the rate of hydrolysis is diminished by increasing the ionic strength to 0.1, for example using NaCl (Visser et al., 1980). In addition, a maximum rate of proteolysis has been reported for 8 mM calcium chloride (Bringe and Kinsella, 1986).
11.4 Colloidal Aspects of Rennet-Induced Aggregation of Casein Micelles
The clotting process triggered by chymosin has been studied for decades, and the aggregation is explained adopting models from colloidal theories. The mechanism of aggregation needs to take into account the relationship between the extent of κ-casein proteolysis and the probability of the casein micelles to aggregate when in close proximity with one another. As previously mentioned, κ-casein acts as a charged-polymer brush, hence its solvation and stability depends on solvent quality and charge density. The removal of the CMP from the surface results in a reduction of the overall charge of the particles, measured as a reduction in ζ-potential (Green, 1973; Anema and Klostermeyer, 1996); however, electrostatic repulsion is not the prime factor stabilizing casein micelles in skim milk. At large distances, the behaviour of casein micelles can be approximated to that of hard spheres. The presence of κ-casein on the surface causes steric repulsion forces to dominate over short-range van der Waal’s and electrostatic interactions, and the steric influence of this polyelectrolyte layer is strong enough to keep the casein micelles far apart (de Kruif and Zhulina, 1996; de Kruif, 1998), even at high concentrations (Bouchoux et al., 2010; Krishnankutty Nair et al., 2014).
In milk, at its natural pH, extensive (80–90 %) hydrolysis of κ-casein needs to occur before the casein micelles show visible aggregation (Dalgleish, 1979). Aggregation occurs only in the presence of calcium ions (Bringe and Kinsella, 1986; Martin et al., 2010). Temperature, pH and concentration of enzyme affect the kinetics of aggregation (Okigbo et al., 1985a, b; Kawaguchi et al., 1987; Crabbe, 2004). Aggregation of casein micelles is inhibited at refrigeration temperatures, even after a nearly complete release of the CMP. This behaviour has developed the general consensus that hydrophobic forces play a major role in casein aggregation; however, this may also be due to rearrangements in the structure of the casein micelles at low temperature.
The clotting reaction can be modeled after von Smoluchowski’s rate theory for bi-molecular reactions, and it is assumed to be diffusion-controlled and independent of the size of the reacting particles (Payens, 1979; Dalgleish, 1980). This model well explains the increase in the turbidity of casein micelle suspensions during renneting (Payens, 1979; Dalgleish, 1980). The collisions between the casein micelles will not be effective (will not lead to aggregates) until a sufficient portion of the surface can come into direct contact with another surface. The elimination of patches of depleted CMP is critical. A simple geometrical model of steric stabilization has been employed to describe this behaviour (Dalgleish and Holt, 1988). Once the protein particles are sufficiently modified, the collisions between the particles will lead to adhesion and the formation of clusters (Dalgleish, 1980). There is then a change from a stable colloidal system, with a high occurrence of ineffective particle encounters, to an unstable system, in a very short time scale, that is nearly instantaneous when a sufficient amount of κ-casein has been hydrolyzed. The apparent lag phase observed in milk from the addition of rennet to the visible milk gelation is caused by the time necessary for κ-casein to be hydrolyzed. This model is well supported by research that demonstrates that the ability of the casein particles to aggregate when partly treated with chymosin does not increase linearly with the extent of proteolysis (Dalgleish, 1979, 1980).
As the casein micelles come closer to each other, they are subjected to long range forces that rule how close they can approach. If closer approach can be achieved, then the interactions are short ranged, and may depend on the local composition and conformation of the κ-casein in the portion of the surfaces that could interact. This will not happen with intact layers. It has also been demonstrated that close approach by osmotic stressing cannot cause aggregation of the casein micelles (Krishnankutty Nair et al., 2013). The time necessary to observe visible milk gelation decreases with increasing enzyme concentration or protein concentration; however, literature results are contradictory in this regard, and may result from differences in the methods used to measure gelation and differences in the processing history of the sample (Dalgleish, 1980; Waungana et al., 1998; Karlsson et al., 2007; Salvatore et al., 2011; Sandra et al., 2011). The gelation point is more accurately identified using the extent of CMP release and a summary of the dependence of the gelation point on the amount of CMP released is shown in Fig. 11.2, for milk containing different concentrations of protein or treated at different pH values.
Several methods were developed over the years for monitoring the coagulation of milk, and a chronological list of them has been reported by Lucey (2002). Most of the available methods can accurately measure the milk coagulation time, and rheological measurements together with microstructural analysis can follow structure formation and the properties of the final gel; however, more analytical approaches are needed to follow structural rearrangements and structural heterogeneities once the gel has formed.
The early stages of gelation can be followed using light scattering. The changes in the diffusivity of the casein micelles allow measuring with accuracy the point of gelation, often expressed by a rapid increase in the radius of the casein micelles, or by a change in the turbidity of the milk. Before the point of gelation, there is a reduction of the casein micelle size (Walstra et al., 1981; Horne and Davidson, 1993), as shown in Fig. 11.3. After the addition of rennet, it is possible to follow a reduction in the viscosity of the milk, (which has been attributed to the release of CMP from the casein micelles), before the increase due to coagulation (de Kruif, 1999). The diffusivity of the casein micelles sharply decreases (as indicated by the sharp increase in the radius measured by light scattering) and the casein particles show arrested motion. This can be clearly measured by following the slope of the mean square displacement, measured using Diffusing Wave Spectroscopy (DWS), as shown in the insert of Fig. 11.4 (Alexander and Dalgleish, 2004; Gaygadzhiev et al., 2009). Figure 11.4 summarizes the development of light scattering and rheological parameters during a rennet gelation experiment. The changes in the turbidity parameter (1/l*) obtained by light scattering (transmission DWS) are related to the beginning of inter-particle interactions, and indicate that with the release of CMP the spatial correlation of the casein micelles begins to be affected at an earlier point than the complete release of CMP (Alexander and Dalgleish, 2004; Sandra et al., 2007).
Change in apparent diameter as a function of time from rennet addition, measured by dynamic light scattering. The casein micelles from untreated skim milk are extensively diluted in milk permeate, and rennet is added. The phase corresponding to the removal of the hairy layer of κ-casein is indicated by the arrow. For experimental details see Gaygadzhiev et al., (2009)
Representative data on a rennet induced aggregation experiment using skim milk, indicating the changes in radius (circles), 1/l* (turbidity, triangles) as measured by diffusing wave spectroscopy, compared to changes in the elastic modulus (G', squares) measured by rheology. Time indicates the time after addition of rennet. Insert shows mean square displacement as a function of time (not change in time scale); arrow indicates different time points during renneting
Dynamic oscillatory testing is often employed to follow the development of the gel structure in rennet-treated milk. The elastic modulus (G') shows a rapid increase at the point of gelation, in full agreement with the decrease in the diffusivity of the casein micelles, indicated by the increase in the apparent radius measured by light scattering. This rapid increase in the apparent size of the micelles or the elastic modulus is referred to as gelation point, and in milk at natural pH this point corresponds to about 90 % of CMP released from the micelles (Sandra et al., 2007). Using oscillatory rheology it is not only possible to measure the development of the gel structure, by following the changes in the elastic modulus (G'), but it is also possible to measure the transition point from a sol to a gel, often defined as a change of phase (tan δ = 1, where δ is the phase angle). Tan δ decreases rapidly through the gelation point, indicating that the increase in the stiffness is due to an increase in the number of linkages in the protein network, but not to the nature or strength of the bonds.
It is possible then to conclude that the kinetics of rennet-induced gelation strongly depend on the quality of the solvated κ-casein layer. At the natural pH of milk, gelation occurs only when nearly all the CMP has been removed from the surface of the casein micelles. Under these conditions, upon physical contact, the casein micelles no longer behave as rigid spheres, and can deform or even coalesce, leading to a loss of particle identity, showing “fusion” between the protein particles, similar to what has been described for model rubbery particles (Gauer et al., 2009).
At a lower pH, gelation occurs at a lower degree of hydrolysis (van Hooydonk et al., 1986) because of the decrease in steric and electrostatic repulsion. Similarly, the addition of ethanol can cause partial collapse of the κ-casein layer and a decrease in steric repulsion (Bringe and Kinsella, 1986; de Kruif, 1999; O’Connell et al., 2006). The internal organization of the proteins in the casein micelles and the amount of colloidal calcium phosphate present are not important to the initial stages of aggregation, and the behaviour of the casein micelles can be approximated to that of colloidal hard spheres (de Kruif, 1998). However, the internal characteristics and the extent of colloidal calcium phosphate present in the micelles become of importance after aggregation, as they affect the structural rearrangements of the network. It has been reported that a partial loss of colloidal calcium may affect charge interactions, and the formation of a weaker, more flexible protein gel (Choi et al., 2007). The composition of the serum phase and the presence of other molecules interacting with the casein micelles also can strongly affect the renneting properties of milk, at the early stages of aggregation as well as during structure formation. In particular, heat denatured whey proteins, soluble caseins and small molecular mass emulsifiers can affect the interactions of the casein micelles and the efficiency of the collisions (Gaygadzhiev et al., 2009; Ion Titapiccolo et al., 2010a; Kethiredipalli et al., 2010). For example, it has been shown that by addition of polysorbate, the casein micelles have a higher propensity to aggregate, and even at the natural pH of milk, the gelation time occurs at an earlier extent of CMP release than for control milk samples. On the other hand, recent work has demonstrated that, in the absence of added calcium, aggregation of casein micelles is inhibited by the presence of soluble caseins (Gaygadzhiev et al., 2009). The soluble proteins in milk at its natural pH may inhibit close range attachment of the micelles by shielding the hydrophobic or calcium-sensitive patches on the surface of the protein particles. This effect is demonstrated using dynamic light scattering under diluted conditions: while after addition of rennet there is still a decrease in the hydrodynamic diameter of the casein micelles, in the presence of sodium caseinate, there is no aggregation of the protein particles. The presence of soluble casein, and the re-arrangements of the casein micelles during cooling have also been considered as an important factor for the inhibition of rennet-induced aggregation at temperatures below 18 °C (Bansal et al., 2007).
11.5 Effect of Calcium Chloride on Rennet Induced Gelation of Casein Micelles
It is well recognized that the addition of calcium chloride to milk has positive effects on texture and cheese curd yield (Udabage et al., 2001). Calcium ions are essential for aggregation and gelation of casein micelles, and the reason behind this mechanism has yet to be fully established. Para-κ-casein binds to calcium ions more strongly than κ-casein (Bringe and Kinsella, 1986), and it has been recently hypothesized using molecular simulations that the charge asymmetry on the para-κ-casein may play a key role in affecting CMP conformation on the micelle surface and, as a consequence, its stabilizing properties (Ettelaie et al., 2014). Indeed, although free calcium ions shield negatively-charged amino acid residues, reducing the overall charge of the casein micelles, this cannot be the only reason behind its essential role in rennet-induced aggregation of casein micelles.
In milk, addition of 1 mM calcium chloride does not increase the enzymatic activity, as shown by kinetics of CMP release (Sandra et al., 2012), but it shortens coagulation time and forms stiffer gels (van Hooydonk et al., 1986; Sandra et al., 2012). The aggregation of the casein micelles occurs at a lower degree of κ-casein hydrolysis, by decreasing the extent of solvation of the polyelectrolyte brush around the micelles (van Hooydonk et al., 1986; de Kruif, 1999). The mode of addition of calcium (fast addition as CaCl2, or slowly by dialysis) to the milk does not seem to have an impact on the aggregation behaviour (Sandra et al., 2012). A recent study followed the increase in turbidity of milk after addition of rennet using diffusing wave spectroscopy and showed no difference in the turbidity behaviour as a function of CMP release with 1 mM calcium added, in spite of the earlier onset of coagulation of the micelles (Sandra et al., 2012). These results may suggest that calcium ions play a major role in strengthening short range interactions. Evidence of calcium binding to the micelles after rennet coagulation was also shown in a study on mixed acid and rennet coagulation, whereby soluble calcium was shown to be reduced during early acidification after complete CMP release (Salvatore et al., 2011). It is also important to note that although additional calcium increases stiffness of the gels, too much calcium added to milk can form weaker gels (Fox et al., 2000; Udabage et al., 2001).
While the amount of soluble calcium is of great significance in the early stages of rennet gelation, differences in the concentration of colloidal calcium affect the structural rearrangements of the gel network (Choi et al., 2007). Loss of colloidal calcium phosphate will weaken the internal structure of the micelles once the gel has formed, and will affect the forces stabilizing the gel, namely, hydrogen bonds, van der Waals interactions and calcium bridges. Hence, the presence of colloidal calcium is an important factor in determining the properties of the gel network. This can be clearly noted in the value of tan δ of rennet gels measured by rheology (Choi et al., 2007).
11.6 Effect of Acidification on Rennet Induced Gelation
Many cheese making processes involve an acidification step, usually occurring simultaneously to chymosin activity. A mild acidification can improve rennet coagulation properties, by shortening the coagulation time and strengthening the gel (Renault et al., 2000). As the pH decreases, the surface of the casein micelles changes, micellar calcium is released in the serum phase, but, at temperatures above 25 °C, this pH change does not cause a substantial release of protein in the soluble phase (Dalgleish and Law, 1988). At a more acidic pH there is also a faster release of CMP, as shown in Fig. 11.5.
With decreasing pH, the steric repulsion between casein micelles decreases and the attractive forces, as described quantitatively using an adhesive hard sphere model, increase (de Kruif, 1997). At about pH 5.6, there is a change in the apparent diameter of the micelles (Alexander and Dalgleish, 2005). Hence, the extent of CMP release necessary to cause the micelles to aggregate becomes smaller as the pH is decreased (van Hooydonk et al., 1986).
The mechanism of gel formation by acidification is quite different from that caused by renneting, as well as the properties of the final gel. In contrast with rennet-induced gelation, in the case of acid gelation, CMP is not released, but the micelles loose electrostatic repulsion because of the decrease in the overall charge on the casein micelles. This causes the collapse of the hairy layer and the decrease in steric stabilization, leading to aggregation near the isoelectric point of the proteins.
The rheological properties of milk gels obtained by a combination of acid and enzymatic gelation are less understood, as the two mechanisms occur simultaneously and the properties of the gel network formed are different than in the case of rennet alone, or acid alone (Lucey et al., 2000; Gastaldi et al., 2003). The order and the extent of the modification of the casein micelles will strongly affect the structure and properties of the final gel (Li and Dalgleish, 2006). In the case of low addition of rennet, whereby there is an insufficient release of CMP for aggregation to occur, by increasing the extent of proteolysis of κ-casein the pH of gelation increases (Gastaldi et al., 2003; Li and Dalgleish, 2006). On the other hand, in the case where a sufficient amount of CMP is released to cause rennet-induced aggregation, the release of calcium from the casein micelles during further acidification changes the properties of the gel (Li and Dalgleish, 2006; Salvatore et al., 2011).
In gels prepared with a combination of rennet and acid, the storage modulus is larger than that of either acid or rennet gelation alone. In addition, these gels show a very distinct development of the storage modulus, as exemplified in Fig. 11.6. When a rennet gel is formed at a pH where the calcium phosphate is still mostly present in the micelles, the gel modulus shows first a marked increase and a subsequent decrease caused by the release of colloidal calcium phosphate from the micelles. The development of the value of tan δ demonstrates the weakening of the gel network, explained by a weakening internal structure of casein micelles already part of a network, because of increased charge repulsion between the protein chains. A further decrease in pH then causes an increase in the storage modulus, indicating a further rearrangement of the structure (Fig. 11.6).
Rheological pattern of rennet gelation during fermentation with mesophilic bacteria. Storage modulus (G', solid line), and loss tangent (tan δ, broken line) as a function of time. The pH during fermentation at various points of the curve are indicated. Redrawn from Salvatore et al., (2011)
In summary, gels formed during partial enzymatic gelation and acidification will share both properties, but their characteristics will depend on the interactions between the particles, the presence of an inter-particle layer of CMP and the presence of colloidal calcium phosphate. This will lead to a different extent of breakdown of the internal micellar structure and formation of inter-particle interactions in the network, and may promote fusion of the casein micelles (Aichinger et al., 2003).
11.7 Effect of Temperature on Enzymatic Coagulation
It is common practice to cool the milk immediately after milking, to improve its microbiological quality. However, it has been demonstrated that cold refrigeration affects clotting time and reduces curd firmness (Walstra and van Vliet, 1986; Raynal and Remeuf, 2000). This effect has been attributed to the solubilization of β-casein from the micelles, resulting in a slight increase in the micellar stability. The cold-induced solubilisation of β-casein and calcium are partly reversed with re-warming of milk (Raynal and Remeuf, 2000).
When milk is incubated with rennet at refrigeration temperatures, the enzymatic reaction occurs slowly but no visible gelation is observed (Berridge, 1942). Recent work measured the rheological properties of milk during renneting at 10 °C and demonstrated that aggregation may occur at this temperature, but at a very slow rate. This effect has been recently attributed to the presence of β-casein, which may be playing a protective role being present in the serum phase and causing rearrangements to the casein micelle structure (Bansal et al., 2007).
11.8 Enzymatic Coagulation of Heated Milk
Heating of milk at temperatures above 60 °C causes structural changes to the whey proteins. These proteins unfold, expose their reactive sites and form aggregates, either in solution or on the surface of the casein micelles. The presence of heat-induced whey protein complexes in milk has important consequences for the technological properties of milk. The aggregates present in the non-sedimentable fraction of heated milk contain β-lactoglobulin, α-lactalbumin and κ-casein, and are <100 nm in diameter (Jean et al., 2006).
The presence of heat-induced aggregates can cause significant changes to the rate of rennet-induced aggregation and the properties of the curd. The effect has been attributed in the past to the reduction of the accessibility of chymosin to κ-casein, because of the linkage of denatured whey protein molecules to Cys11 and Cys88 of κ-casein, the loss of soluble calcium and the steric hindrance caused by the aggregates (van Hooydonk et al., 1987).
It has been recently demonstrated that the rate of CMP release is very similar in heated and unheated milk (Vasbinder et al., 2003; Kethireddipalli et al., 2010) and, regardless of where the κ-casein is located (i.e., in the serum complexes or on the casein micelles) the accessibility of the enzyme is similar (Mollé et al., 2006). The formation of heat-induced whey protein aggregates hinders interactions of casein micelles, decreasing the rate of effective collisions (Vasbinder et al., 2003). The aggregation is limited regardless of the distribution of the complexes on the micelles or in the serum phase. The whey protein aggregates present in the un-sedimentable fraction prior to renneting bind to the casein micelles as renneting proceeds, affecting the potentially interacting sites of the micelles (Kethireddipalli et al., 2010).
Rennet curds prepared from heated milk have poor texture, poor stretching and melting properties (Singh and Waungana, 2001). Strategies have been reported to restore, at least partially, the ability of skim milk to gel with rennet addition. The rennet coagulation properties of milk can be partially recovered after heating by acidifying milk to pH below 6.2, or by acidifying and then readjusting to pH 6.7, or by calcium addition (Banks et al., 1987; Singh and Waungana, 2001). However, the curds obtained with milk treated this way differ from traditional cheese curds. It is also important to note that by careful control of heating and modification to the processes, it is possible to transfer the whey proteins from milk to cheese curds, improving the yield. In this case also the properties of the cheese may be substantially different from the control cheese made from unheated milk (see for example, Chromik et al., 2010).
11.9 Renneting of High Pressure-Treated Milk
High pressure treatment induces changes in the milk constituents that affect the cheese making properties, in particular the rennet clotting time, the rate of gel formation, the stiffness and texture of the gel, as well as the water holding capacity of the curd (López-Fandiño et al., 1997; Needs et al., 2000). The distribution between soluble and colloidal calcium and phosphate is affected, and the increase in soluble calcium and phosphate reaches a plateau at about 300 MPa (López-Fandiño et al., 1997). This is caused by a change in the non-micellar casein fraction, with the extent of dissociation in the order β- > κ- > αs-caseins (López-Fandiño et al., 1996). The dissociation of casein micelles during high pressure processing is partly reversible, but depending on the pressure, can either induce aggregation or disruption of the casein micelles, with changes to the turbidity of the milk (Huppertz et al., 2006). Although the enzymatic hydrolysis (as measured by the release of CMP) seems to be little affected by high pressure, the changes in the size of the micelles reduce the clotting time (López-Fandiño et al., 1997). Indeed the presence of partially-disrupted casein micelles will cause an increase in the surface area for inter-micellar interactions, and will promote aggregation. At higher pressures (i.e., >300 MPa) whey protein denaturation occurs (López-Fandiño et al., 1996), and the formation of complexes with the casein micelles has a negative impact on casein aggregation. It has been reported that about 90 % of the β-lactoglobulin is no longer native at 400 MPa (López-Fandiño et al., 1996; Needs et al., 2000). Although the association of whey proteins with the casein micelles may cause hindrance to the secondary phase of gelation, curd yields increase, because of the incorporation of whey proteins and the increase in moisture content (Needs et al., 2000; Huppertz et al., 2006).
In addition to static high pressure, some work has also been reported on the effect of dynamic high pressure homogenization on rennet-induced aggregation of skim milk. The surface of the casein micelles is slightly altered by high pressure homogenization, causing a shorter coagulation time, as some κ-casein is removed (Sandra and Dalgleish, 2007).
11.10 Enzymatic Coagulation in Milk Concentrated by Membrane Filtration
In recent years, there has been an increased interest in understanding the effect of the volume fraction of casein micelles on the renneting properties of milk concentrates, as processes such as ultrafiltration and microfiltration are increasingly common unit operations in the dairy industry. The higher extent of linkages formed and the decreased mobility of the micelles may require changes in the cheese manufacturing processes, as they will affect yield, structure and texture of the final product.
Rennet aggregation is a diffusion controlled reaction (Dalgleish, 1980) and it may be affected by casein volume fraction. Earlier studies reported either a decreased or a constant reaction rate for milk concentrated by ultrafiltration (Garnot and Corre, 1980; van Hooydonk et al., 1984). The rate also depends on pH (with a lower increase at pH 6.2) (Garnot and Corre, 1980). Visible gelation of milk concentrated by ultrafiltration occurs earlier than in normal milk, with a higher firmness at higher protein concentration (Garnot et al., 1982).
Recent work reported the gelation of casein micelles as a function of concentration of milk by ultrafiltration, following the extent of CMP release (Salvatore et al., 2011; Sandra et al., 2011). These studies clearly demonstrated that in all cases, at the natural pH of milk, casein aggregation occurs at about 90 % CMP release (Sandra et al., 2011). The concentration also seems to affect the association of chymosin with the casein micelles. In a study with artificial micelles it was shown that in a more diluted system, chymosin dissociates faster than in concentrated systems (de Roos et al., 2000). This has important consequences to the recovery of chymosin in the cheese curd and cheese ripening (de Roos et al., 2000).
When treating concentrated milk, it is important to take into account the processing history of milk as well as the composition of the serum phase, as the amount of soluble calcium will affect the coagulation behaviour of milk (Ferrer et al., 2011). This is critical in the case of reconstitution of milk protein concentrates (Martin et al., 2010). The rennetability of these micellar concentrates depends on the ionic composition of the serum phase (Ferrer et al., 2008; Martin et al., 2010). Milk protein concentrates reconstituted in water do not gel upon rennet treatment, unless calcium is added to the serum phase. The amount of soluble casein present in the milk has to be carefully controlled as it may hinder the aggregation of the micellar fraction (Martin et al., 2010; Ferrer et al., 2011; Sandra et al., 2012).
11.11 Presence of Other Components in Milk
Although most mechanistic studies on the enzymatic gelation of casein micelles have been carried out in skim milk, in dairy products the aggregation occurs in the presence of other components, which may or may not affect the behaviour of the micelles.
In most milk products, fat globules play an important role in imparting structure and texture to the matrix, and contribute to creamy mouthfeel and flavour. The renneting behaviour of milk is affected by the size and the interfacial composition of the fat globules. In untreated whole milk, fat globules are polydisperse in size from less than 100 nm to about 10 μm in diameter (Michalski et al., 2002). Homogenization of milk modifies the size of the globules, resulting in an increase of their surface area. The original fat globule membrane material is no longer sufficient to stabilize the newly formed interfaces, and casein micelles are found preferentially adsorbed onto the interface, either intact or as fragments of the original micelle (Sharma and Dalgleish, 1993). The properties of rennet gels containing fat globules, prepared by homogenizing milk are quite different than those of unhomogenized milk, and have been widely studied, in particular for the effect on moisture retention, the whitening of cheese and increases in yield (Peters, 1956).
Recent data suggest that the release of CMP is faster in homogenized milk compared to skim milk, and all the κ-casein is available to chymosin hydrolysis (Ion Titapiccolo et al., 2010b). Homogenized milk coagulates faster than non-homogenized milk (Ion Titapiccolo et al., 2010b). The spreading of the caseins at the interface will increase the susceptibility of κ-casein to rennet, as previously hypothesized (Robson and Dalgleish, 1984). The micelles present at the surface of homogenized fat globules interact with the gel and aggregate with each other. The presence of these micelles does not modify their aggregation behaviour, but the system aggregates earlier than skim milk as a consequence of the earlier CMP release kinetics (Ion Titapiccolo et al., 2010b). However, milk homogenization may impair the quality of cheese obtained by rennet-induced gelation of milk, leading to soft curds containing high moisture levels.
Research has also been carried out using model systems consisting of milk gels filled with oil droplets. The rheological behaviour of these emulsion-filled milk gels depends on the structural properties of the filler particles, the viscoelasticity of the matrix as well as the physical properties of the droplets, the concentration of the components, and most importantly, the interactions between the interface of the droplet and the protein network (Sala et al., 2007).
In recombined systems, with a careful design of the interfacial composition of the fat globules and control of their particle size and colloidal state, it is possible to affect the structure of rennet gels, as the ability of the droplets to interact with the gel and participate in the formation of the casein network will result in very different structures. While interacting droplets will be part of the gel network, as in the case of homogenized fat globules containing casein micelles, oil droplets with interfacial material not subjected to destabilization with rennet will not become part of the network gel. Small non-interacting droplets will reside within the pores of the network, and will have little effect on the viscoelasticity of the system. Larger droplets or flocculated droplets will cause increases in the stiffness of the gel, even when not interacting. As the casein gel develops, the droplets are now entrapped inside the pores of the protein network, and the droplets, as well as the caseins have much less available space to interact, causing stiffening of the gel while the mobility of the droplets continue to decrease within the pores (Corredig et al., 2011). During cutting of the curd, these oil droplets may be lost in the serum phase, but have contributed to the structuring of the rennet gels.
Enzymatic cross-linking of caseins, which results in interesting properties for milk gels (increased gel strength and reduced syneresis), also affects the coagulation properties of milk. If rennet is added to casein micelles enzymatically cross-linked with transglutaminase (TGase; EC 2.3.2.13), the aggregation of para-κ-casein is reduced, due to a further level of steric stabilisation created by cross-linked micelles (Huppertz and de Kruif, 2007). Conversely, other authors (O’Sullivan et al., 2002; Özer et al., 2012) also demonstrated that the primary stage of milk coagulation (the release of CMP) is influenced by cross-linking. The coagulation times of milk with added TGase are strictly dependent on the amount of enzyme used, the coagulation temperature and the initial pH of the milk (Özer et al., 2012).
The drive to healthier products in the market place is also leading to the study of enzymatic coagulation in mixed systems containing polysaccharides, in quantities that could be either nutritionally significant, as dietary fiber, or as functional ingredients in low fat products. Polysaccharides are high molecular weight molecules that cause structuring in milk, and above a critical concentration can cause phase separation. The careful control of microphase separated domains during renneting is of increasing interest as it has the potential to create novel structures in dairy matrices. By controlling the kinetics and the extent of phase separation it is possible to form different gel microstructures (Acero-Lopez et al., 2010).
Recent work has been published on the effect of addition of polysaccharide molecules on the renneting properties and coagulation kinetics of skim milk (Fagan et al., 2006; Arango et al., 2013). The addition of fiber can change the time of rennet gelation as well as the stiffness and the microstructure of the gels. For example, in the case of skim milk containing κ-carrageenan and xanthan gum, increasing polysaccharide concentration causes inhibition of the aggregation phase, while adding high methoxyl pectin enhances aggregation. These changes have been attributed to differences in the structuring of these polysaccharide molecules (Tan et al., 2007).
The structuring behaviour of polysaccharide molecules depends on biopolymer concentration, milk volume fraction, environmental conditions, processing history and all these factors need to be carefully controlled to fine tune the final texture of the rennet gel. For example, above a certain concentration, phase separation may occur, and even before macroscopic phase separation, localized casein micelle clusters will form, limiting the formation of a self-supporting gel.
11.12 Conclusion and Outlook
The enzymatic gelation of milk is one of the most studied topics in food colloids, as the reaction is based on the gradual hydrolysis of a protein polyelectrolyte layer on the surface of the casein micelles. A better understanding of the renneting reaction has also great applied importance to dairy processors. By modulating environmental conditions, casein volume fraction and processing history of the milk, it is possible to control the preliminary stages of gelation, and form cheese curds with different properties, and as a consequence, influence the final cheese. Different conditions of enzymatic coagulation will also have important consequences to ripening, bacterial distribution and their lysis in the matrix.
The mode of action of the enzyme is well understood, and the details of the reaction well described, at least in the case of unprocessed milk at its natural pH. More knowledge is needed to link the changes occurring in the preliminary stages of gelation to the final gel, to be able to ultimately control the structure and form particular types of networks. Better experimental approaches are needed to be able to study the interactions occurring in the complex cheese curd matrix, to follow rearrangements of structure and how these are affected by the initial rennet coagulation and the destabilization of the casein micelles. Little is also known of the effects caused by partial enzymatic modifications to the casein micelles. Indeed, a multitude of processes are known where aggregation of para-κ-casein is one of the competing mechanisms occurring during milk gelation, and it is important to understand better how to fine tune the conditions to obtain a particular curd structure. Such processing conditions are often ruled by practical knowledge as it is still necessary to tease out the various mechanisms at play. This is obvious in rennet curds obtained in less traditional processes, whereby components, other than fat globules and native whey proteins may be present, or the processing history of the milk is complex, as for example, in reconstituted milk powders. This chapter highlights the current knowledge on the fundamental aspects of enzymatic induced coagulation of milk, but it clearly points out the need for more studies to bridge the gaps between model systems and practical application. In addition, it is clear that more needs to be understood on how to control the colloidal destabilization reaction to reach a particular structure and texture. Cheese is a nutritionally valued product widely consumed in various forms; a better knowledge of the complexity of the gelling process will lead to improved or novel products and processes.
References
Acero-Lopez A, Alexander M, Corredig M (2010) Diffusing wave spectroscopy and rheological studies of rennet-induced gelation of skim milk in the presence of pectin and κ-carrageenan. Int Dairy J 20:328–335
Aichinger PA, Michel M, Servais C, Dillmann M-L, Rouvet M, D’Amico N, Zink R, Klostermeyer H, Horne DS (2003) Fermentation of a skim milk concentrate with Streptococcus thermophilus and chymosin: structure, viscoelasticity and syneresis of gels. Colloids Surf B Biointerfaces 31:243–255
Alexander M, Dalgleish DG (2004) Application of transmission diffusing wave spectroscopy to the study of gelation of milk by acidification and rennet. Colloids Surf B Biointerfaces 38:83–90
Alexander M, Dalgleish DG (2005) Interactions between denatured serum proteins and casein micelles studied by diffusing wave spectroscopy. Langmuir 21:11380–11386
Anema SG, Klostermeyer H (1996) ζ-Potentials of casein micelles from reconstituted skim milk heated at 120°C. Int Dairy J 6:673–687
Arango O, Trujillo AJ, Castillo M (2013) Influence of fat replacement by inulin on rheological properties, kinetics of rennet milk coagulation, and syneresis of milk gels. J Dairy Sci 96:1984–1996
Banks JM, Stewart G, Muir DD, West IG (1987) Increasing the yield of cheddar cheese by the acidification of milk containing heat-denatured whey proteins. Milchwissenschaft 42:212–215
Bansal N, Fox PF, Mc Sweeney PLH (2007) Aggregation of rennet-altered casein micelles at low temperatures. J Agric Food Chem 55:3120–3126
Barbano DM, Rasmussen RR (1992) Cheese yield performance of fermentation-produced chymosin and other milk coagulants. J Dairy Sci 75:1–12
Berridge NJ (1942) The second phase of rennet coagulation. Nature 149:194–195
Bouchoux A, Gésan-Guiziou G, Pérez J, Cabane B (2010) How to squeeze a sponge: casein micelles under osmotic stress, a SAXS study. Biophys J 99:3754–3762
Bringe NA, Kinsella JE (1986) Influence of calcium chloride on the chymosin-initiated coagulation of casein micelles. J Dairy Res 53:371–379
Chitpinityol S, Crabbe MJC (1998) Chymosin and aspartic proteinases. Food Chem 61:395–418
Choi J, Horne DS, Lucey JA (2007) Effect of insoluble calcium concentration on rennet coagulation properties of milk. J Dairy Sci 90:2612–2623
Chromik C, Partschefeld C, Jaros D, Henle T, Rohm H (2010) Adjustment of vat milk treatment to optimize whey protein transfer into semi-hard cheese: a case study. J Food Eng 100:496–503
Corredig M, Ion Titapiccolo G, Gaygadzhiev Z, Alexander M (2011) Rennet-induced aggregation of milk containing homogenized fat globules. Effect of interacting and non-interacting fat globules observed using diffusing wave spectroscopy. Int Dairy J 21:679–684
Crabbe MJC (2004) Rennets: general and molecular aspects. In: Fox PF, McSweeney PLH, Cogan TM, Guinee TP (eds) Cheese: chemistry physics and microbiology, vol I, 3rd edn, General aspects. Elsevier Ltd, London, pp 19–45
Dalgleish DG (1979) Proteolysis and aggregation of casein micelles treated with immobilized or soluble chymosin. J Dairy Res 46:653–661
Dalgleish DG (1980) Effect of milk concentration on the rennet coagulation time. J Dairy Res 47:231–235
Dalgleish DG, Holt C (1988) A geometrical model to describe the initial aggregation of partly renneted casein micelles. J Colloid Interface Sci 123:80–84
Dalgleish DG, Law AJR (1988) pH-induced dissociation of bovine casein micelles. I. Analysis of liberated caseins. J Dairy Res 55:529–538
Dalgleish DG, Horne DS, Law AJR (1989) Size-related differences in bovine casein micelles. Biochim Biophys Acta 991:383–387
Dalgleish DG (1998) Casein micelles as colloids: surface structures and stabilities. J Dairy Sci 81:3013–3018
Dalgleish DG, Corredig M (2012) The structure of the casein micelle of milk and its changes during processing. Annu Rev Food Sci Technol 3:449–467
Danley DE, Geoghegan KF (1988) Structure and mechanism of formation of recombinant-derived chymosin C. J Biol Chem 263:9785–9789
de Kruif CG, Zhulina EB (1996) κ-Casein as a polyelectrolyte brush on the surface of casein micelles. Colloids Surf A Physicochem Eng Asp 117:151–159
de Kruif CG (1997) Skim milk acidification. J Colloid Interface Sci 185:19–25
de Kruif CG (1998) Supra-aggregates of casein micelles as a prelude to coagulation. J Dairy Sci 81:3019–3028
de Kruif CG (1999) Casein micelle interactions. Int Dairy J 9:183–188
de Kruif CG, Huppertz T, Urban VS, Petukhov AV (2012) Casein micelles and their internal structure. Adv Colloid Interface Sci 171–172:36–52
de Roos AL, Geurts TJ, Walstra P (2000) The association of chymosin with artificial casein micelles. Int Dairy J 10:225–232
Dunnewind B, de Roos AL, Geurtz TJ (1996) Association of chymosin with caseins in solution. Neth Milk Dairy J 50:121–133
Ettelaie R, Khandelwal N, Wilkinson R (2014) Interactions between casein layers adsorbed on hydrophobic surfaces from self consistent field theory: κ-casein versus para-κ-casein. Food Hydrocolloids 34:236–246
Fagan CC, O’Donnell CP, Cullen PJ, Brennan CS (2006) The effect of dietary fiber inclusion on milk coagulation kinetics. J Food Eng 77:261–268
Farrell HM Jr, Wickham ED, Dower HJ, Piotrowski EG, Hoagland PD, Cooke PH, Groves ML (1999) Characterization of the particles of purified kappa-casein: trypsin as a probe of surface-accessible residues. J Protein Chem 18:637–652
Ferrer MA, Hill AR, Corredig M (2008) Rheological properties of rennet gels containing milk protein concentrates. J Dairy Sci 91:959–969
Ferrer MA, Alexander M, Corredig M (2011) Does ultrafiltration have a lasting effect on the physico-chemical properties of the casein micelles? Dairy Sci Technol 91:151–170
Fox PF, Guinee TP, Cogan TM, McSweeney PLH (2000) Fundamentals of cheese science. Aspen, Gaithersburg
Fox PF, McSweeney PLH, Cogan TM, Guinee TP (2004) Cheese: chemistry, physics and microbiology, vol 2, 3rd edn, Major cheese groups. Elsevier, London, pp 1–434
Galán E, Prados F, Pino A, Tejada L, Fernández-Salguero J (2008) Influence of different amounts of vegetable coagulant from cardoon Cynara cardunculus and calf rennet on the proteolysis and sensory characteristics of cheeses made with sheep milk. Int Dairy J 18:93–98
Galán E, Cabezas L, Fernández-Salguero J (2012) Proteolysis, microbiology and sensory properties of ewes’ milk cheese produced with plant coagulant from cardoon Cynara cardunculus, calf rennet or a mixture thereof. Int Dairy J 25:92–96
Garnot P, Corre C (1980) Influence of milk protein concentration on the gelling activity of chymosin and bovine pepsin. J Dairy Res 47:103–111
Garnot P, Rank TC, Olson NF (1982) Influence of protein and fat contents of ultrafiltered milk on rheological properties of gels formed by chymosin. J Dairy Sci 65:2267–2273
Gastaldi E, Trial N, Guillaume C, Bourret E, Gontard N, Cuq L (2003) Effect of controlled kappa-casein hydrolysis on rheological properties of acid milk gels. J Dairy Sci 86:704–711
Gauer C, Jia Z, Wu H, Morbidelli M (2009) Aggregation kinetics of coalescing polymer colloids. Langmuir 25:9703–9713
Gaygadzhiev Z, Alexander M, Corredig M (2009) Sodium caseinate-stabilized fat globules inhibition of the rennet-induced gelation of casein micelles studied by diffusing wave spectroscopy. Food Hydrocoll 23:1134–1138
Gilliland GL, Winborne EL, Nachman J, Wlodawer A (1990) The three-dimensional structure of recombinant bovine chymosin at 2.3 Å resolution. Proteins 8:82–101
Green ML (1973) Studies on the mechanism of clotting of milk. Neth Milk Dairy J 27:278–285
Groves ML, Wickham ED, Farrell HM (1998) Environmental effects on disulfide bonding patterns of bovine κ-casein. J Protein Chem 17:73–84
Gutchina E, Rumsh L, Ginodman L, Majer P, Andreeva N (1996) Post X-ray crystallographic studies of chymosin: the existence of two structural forms and the regulation of activity by the interaction with the histidine-proline cluster of κ-casein. FEBS Lett 379:60–62
Horne DS, Davidson CM (1993) Direct observation of decrease in size of casein micelles during the initial stages of renneting of milk. Int Dairy J 3:61–71
Horne DS, Banks JM (2004) Rennet-induced coagulation of milk. In: Fox PF, McSweeney PLH, Cogan TM, Guinee TP (eds) Cheese: chemistry physics and microbiology, vol I, 3rd edn, General aspects. Elsevier, London, pp 47–70
Huppertz T, Smiddy MA, Upadhyay VK, Kelly AL (2006) High-pressure induced changes in bovine milk: a review. Int J Dairy Technol 59:58–66
Huppertz T, de Kruif CG (2007) Rennet-induced coagulation of enzymatically cross-linked casein micelles. Int Dairy J 17:442–447
Hyslop DB (2003) Enzymatic coagulation of milk. In: Fox PF, McSweeney PLH (eds) Advanced dairy chemistry, vol 1, 3rd edn, Proteins, Part B. Kluwer Academic, New York, pp 839–878
Ion Titapiccolo G, Corredig M, Alexander M (2010a) Modifications to casein micelles renneting functionality caused by non-ionic surfactants. J Dairy Sci 93:506–514
Ion Titapiccolo G, Alexander M, Corredig M (2010b) Rennet induced aggregation of homogenized milk: impact of the presence of fat globules on the structure of casein gels. Dairy Sci Technol 90:623–639
Jaros D, Seitler K, Rohm H (2008) Enzymatic coagulation of milk: animal rennets and microbial coagulants differ in their gelation behaviour as affected by pH and temperature. Int J Food Sci Technol 43:1721–1727
Jean K, Renan M, Famelart MH, Guyomarc’h F (2006) Structure and surface properties of the serum heat-induced protein aggregates isolated from heated skim milk. Int Dairy J 16:303–315
Jensen JL, Mølgaard A, Navarro Poulsen J-C, Harboe MK, Simonsen JB, Lorentzen AM, Hjernø K, van den Brink JM, Qvist KB, Larsen S (2013) Camel and bovine chymosin: the relationship between their structures and cheese-making properties. Acta Crystallogr D69:901–913
Kappeler SR, van den Brink HJM, Rahbek-Nielsen H, Farah Z, Puhan Z, Hansen EB, Johansen E (2006) Characterization of recombinant camel chymosin reveals superior properties for the coagulation of bovine and camel milk. Biochem Biophys Res Commun 342:647–654
Karlsson AO, Ipsen R, Ardö Y (2007) Rheological properties and microstructure during rennet induced coagulation of UF concentrated skim milk. Int Dairy J 17:674–682
Kawaguchi Y, Kosugi S, Sasaki K, Uozumi T, Beppu T (1987) Production of chymosin in Escherichia coli cells and its enzymatic properties. Agric Biol Chem 51:1871–1877
Kethireddipalli P, Hill AR, Dalgleish DG (2010) Protein interactions in heat-treated milk and effect on rennet coagulation. Int Dairy J 20:838–843
Krishnankutty Nair P, Dalgleish DG, Corredig M (2013) Colloidal properties of concentrated heated milk. Soft Matter 9:3815–3824
Krishnankutty Nair P, Alexander M, Dalgleish DG, Corredig M (2014) Physico-chemical properties of casein micelles in unheated skim milk concentrated by osmotic stressing: interactions and changes in the composition of the serum phase. Food Hydrocoll 34:46–53
Li J, Dalgleish DG (2006) Controlled proteolysis and the properties of milk gels. J Agric Food Chem 54:4687–4695
Lindqvist B, Storgards T (1960) An electrophoretic investigation of the degradation of β-casein by crystalline rennin. Acta Chem Scan 14:757–764
López-Fandiño R, Olano A, San José C, Ramos M (1993) Application of reversed-phase HPLC to the study of proteolysis in UHT milk. J Dairy Res 60:111–116
López-Fandiño R, Carrascosa AV, Olano A (1996) The effects of high pressure on whey protein denaturation and cheese making properties of raw milk. J Dairy Sci 79:929–936
López-Fandiño R, Ramos M, Olano A (1997) Rennet coagulation of milk subjected to high pressures. J Agric Food Chem 45:3233–3237
Lucey JA, Tamehana M, Singh H, Munro PA (2000) Rheological properties of milk gels formed by a combination of rennet and glucono-delta-lactone. J Dairy Res 67:415–427
Lucey JA (2002) ADSA Foundation Scholar Award. Formation and physical properties of milk protein gels. J Dairy Sci 85:281–294
Marchin S, Puteaux JL, Pignon F, Leonil J (2007) Effects of the environmental factors on the casein micelle structure studied by cryo transmission electron microscopy and small angle X-ray scattering/ultrasmall angle X-ray scattering. J Chem Phys 126:045101–045110
Martin GJO, Williams RPW, Dunstan DE (2010) Effect of manufacture and reconstitution of milk protein concentrate powder on the size and rennet gelation behavior of casein micelles. Int Dairy J 20:128–131
Michalski MC, Cariou R, Michel F, Garnier C (2002) Native vs. damaged milk fat globules: membrane properties affect the viscoelasticity of milk gels. J Dairy Sci 85:2451–2461
Mollé D, Jean K, Guyomarc’h F (2006) Chymosin sensitivity of the heat-induced serum protein aggregates isolated from skim milk. Int Dairy J 16:1435–1441
Needs EC, Stenning RA, Gill AL, Ferragut V, Rich GT (2000) High pressure treatment of milk: effects on casein micelle structure and on enzymic coagulation. J Dairy Res 67:31–42
Newman M, Safro M, Frazao C, Khan G, Zdanov A, Tickle IJ, Blundell TL, Andreeva N (1991) X-ray analyses of aspartic proteinase IV. Structure and refinement at 2.2 Å resolution of bovine chymosin. J Mol Biol 221:1295–1309
Newman M, Watson F, Roychowdhury P, Jones H, Badasso M, Cleasby A, Wood SP, Tickle IJ, Blundell TL (1993) X-ray analyses of aspartic proteinase V. Structure and refinement at 2.0 Å resolution of the aspartic proteinase from Mucor pusillus. J Mol Biol 230:260–283
O’Connell JE, Saracino P, Huppertz T, Uniake T, de Kruif CG, Kelly AL, Fox PF (2006) Influence of ethanol on the rennet-induced coagulation of milk. J Dairy Res 73:312–317
O’Sullivan MM, Kelly AL, Fox PF (2002) Influence of transglutaminase on some physico-chemical properties of milk. J Dairy Res 69:433–442
Okigbo LM, Richardson GH, Brown RJ, Ernstrom CA (1985a) Effects of pH, calcium chloride, and chymosin concentration on coagulation properties of abnormal and normal milk. J Dairy Sci 68:2527–2533
Okigbo LM, Richardson GH, Brown RJ, Ernstrom CA (1985b) Interactions of calcium, pH, temperature, and chymosin during milk coagulation. J Dairy Sci 68:3135–3142
Özer B, Guyot C, Kulozik U (2012) Simultaneous use of transglutaminase and rennet in milk coagulation: Effect of initial milk pH and renneting temperature. Int Dairy J 24:1–7
Palmer DS, Christensen AU, Sørensen J, Celik L, Qvist KB, Schiøtt B (2010) Bovine chymosin: A computational study of recognition and binding of bovine κ-casein. Biochemistry 49:2563–2573
Payens TAJ (1979) Casein micelles: the colloid-chemical approach. J Dairy Res 46:291–306
Peters II (1956) Cheddar cheese made from pasteurized milk homogenized at various pressure. J Dairy Sci 39:1083–1088
Pino A, Prados F, Galán E, McSweeney PLH, Fernández-Salguero J (2009) Proteolysis during the ripening of goats’ milk cheese made with plant coagulant or calf rennet. Food Res Int 42:324–330
Raynal K, Remeuf F (2000) Effect of storage at 4°C on the physicochemical and renneting properties of milk: a comparison of caprine, ovine and bovine milks. J Dairy Res 67:199–207
Renault C, Gastaldi E, Cuq JL, Tarodo De la Fuente B (2000) Effect of temperature of milk acidification on rennet gel properties. J Food Sci 65:630–634
Robson EW, Dalgleish DG (1984) Coagulation of homogenized milk particles by rennet. J Dairy Res 51:417–424
Sala G, Van Aken GA, Cohen Stuart MA, Van de Velde F (2007) Effect of droplet-matrix interactions on large deformation properties of emulsion-filled gels. J Texture Stud 38:511–535
Salvatore E, Pirisi A, Corredig M (2011) Gelation properties of casein micelles during combined renneting and bacterial fermentation: effect of concentration by ultrafiltration. Int Dairy J 21:848–856
Sandra S, Alexander M, Dalgleish DG (2007) The rennet coagulation mechanism of skim milk as observed by transmission diffusing wave spectroscopy. J Colloid Interface Sci 308:364–373
Sandra S, Dalgleish DG (2007) The effect of ultra high-pressure homogenization (UHPH) on rennet coagulation properties of unheated and heated fresh skimmed milk. Int Dairy J 17:1043–1052
Sandra S, Cooper C, Alexander M, Corredig M (2011) Coagulation properties of ultrafiltered milk retentates measured using rheology and diffusing wave spectroscopy. Food Res Int 44:951–956
Sandra S, Ho M, Alexander M, Corredig M (2012) Effect of soluble calcium on the renneting properties of the casein micelles as measured by rheology and diffusing wave spectroscopy. J Dairy Sci 95:75–82
Scott Blair GW, Oosthuizen JC (1961) A viscometric study of the breakdown of casein in milk by rennin and rennet. J Dairy Res 28:165–173
Sharma R, Dalgleish DG (1993) Interactions between milk serum proteins and synthetic fat globule membrane during heating of homogenized whole milk. J Agric Food Chem 41:1407–1412
Sielecki AR, Fedorov AA, Boodhoo A, Andreeva NS, James MNG (1990) Molecular and crystal structures of monoclinic porcine pepsin refined at 1.8 Å resolution. J Mol Biol 214:143–170
Singh H, Waungana A (2001) Influence of heat treatment of milk on cheesemaking properties. Int Dairy J 11:543–551
Silva SV, Malcata FX (2005) Studies pertaining to coagulant and proteolytic activities of plant proteases from Cynara cardunculus. Food Chem 89:19–26
Sørensen J, Palmer DS, Qvist KB, Schiøtt B (2011) Initial stage of cheese production: a molecular modeling study of bovine and camel chymosin complexed with peptides from the chymosin-sensitive region of κ-casein. J Agric Food Chem 59:5636–5647
Tan YL, Ye A, Singh H, Hemar Y (2007) Effects of biopolymer addition on the dynamic rheology and microstructure of renneted skim milk systems. J Texture Stud 38:404–422
Udabage P, McKinnon IR, Augustin MA (2001) Effects of mineral salts and calcium-chelating agents on the gelation of renneted skim milk. J Dairy Sci 84:1569–1575
Vallejo JA, Ageitos JM, Poza M, Villa TG (2012) Short communication: a comparative analysis of recombinant chymosins. J Dairy Sci 95:609–613
van Hooydonk ACM, Olieman C (1982) A rapid and sensitive high-performance liquid chromatography method of following the action of chymosin in milk. Neth Milk Dairy J 36:153–158
van Hooydonk ACM, Olieman C, Hagedoorn HG (1984) Kinetics of the chymosin-catalysed proteolysis of κ-casein in milk. Neth Milk Dairy J 38:207–222
van Hooydonk ACM, Boerrigter IJ, Hagedoorn HG (1986) pH-induced physico-chemical changes of casein micelles in milk and their effect on renneting. 2. Effect of pH on renneting of milk. Neth Milk Dairy J 40:297–313
van Hooydonk ACM, de Koster PG, Boerrigter IJ (1987) The renneting properties of heated milk. Neth Milk Dairy J 41:3–18
Vasbinder AJ, Rollema HS, de Kruif CG (2003) Impaired rennetability of heated milk; study of enzymatic hydrolysis and gelation kinetics. J Dairy Sci 86:1548–1555
Visser S, van Rooijen PJ, Slangen CJ (1980) Peptide substrates for chymosin (rennin). Isolation and substrate behaviour of two tryptic fragments of bovine κ-casein. Eur J Biochem 108:415–421
Visser S, Slangen CJ, van Rooijen PJ (1987) Peptide substrates for chymosin (rennin): interaction sites in κ-casein-related sequences located outside the (103–108) hexapeptide region that fits into the enzyme’s active-site cleft. Biochem J 244:553–558
Walstra P, Bloomfield VA, Wei GJ, Jenness R (1981) Effect of chymosin action on the hydrodynamic diameter of casein micelles. Biochim Biophys Acta 669:258–259
Walstra P, van Vliet T (1986) The physical chemistry of curd making. Neth Milk Dairy J 40:241–259
Waungana A, Singh H, Bennett RJ (1998) Rennet coagulation properties of skim milk concentrated by ultrafiltration: effects of heat treatment and pH adjustment. Food Res Int 31:645–651
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer Science+Business Media New York
About this chapter
Cite this chapter
Corredig, M., Salvatore, E. (2016). Enzymatic Coagulation of Milk. In: McSweeney, P., O'Mahony, J. (eds) Advanced Dairy Chemistry. Springer, New York, NY. https://doi.org/10.1007/978-1-4939-2800-2_11
Download citation
DOI: https://doi.org/10.1007/978-1-4939-2800-2_11
Publisher Name: Springer, New York, NY
Print ISBN: 978-1-4939-2799-9
Online ISBN: 978-1-4939-2800-2
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)