Abstract
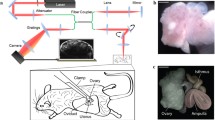
The oviduct (or fallopian tube) serves as the site where a number of major reproductive events occur for the start of a new life in mammals. Understanding the oviduct physiology is essential to uncover hidden mechanisms of the human reproduction and its disorders, yet the current analysis of the oviduct that is largely limited to in vitro imaging is a significant technical hurdle. To overcome this barrier, we have recently developed in vivo approaches based on optical coherence tomography for structural and functional imaging of the mouse oviduct. In this chapter, we describe the details of such live imaging methods that allow for three-dimensional visualization of the oviduct wall morphology, microscale mapping of the oviduct cilia beat frequency, and high-resolution observation of the cumulus–oocyte complex at the cellular level. We expect this set of imaging tools will enable novel studies toward a comprehensive knowledge of the mammalian reproduction.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Coy P, García-Vázquez FA, Visconti PE, Avilés M (2012) Roles of the oviduct in mammalian fertilization. Reproduction 144(6):649–660. https://doi.org/10.1530/rep-12-0279
Li S, Winuthayanon W (2017) Oviduct: roles in fertilization and early embryo development. J Endocrinol 232(1):R1–R26. https://doi.org/10.1530/joe-16-0302
Besenfelder U, Havlicek V, Brem G (2012) Role of the oviduct in early embryo development. Reprod Domest Anim 47(Suppl 4):156–163. https://doi.org/10.1111/j.1439-0531.2012.02070.x
Lyons RA, Saridogan E, Djahanbakhch O (2006) The reproductive significance of human fallopian tube cilia. Hum Reprod Update 12(4):363–372. https://doi.org/10.1093/humupd/dml012
Abe H, Oikawa T (1993) Observations by scanning electron microscopy of oviductal epithelial cells from cows at follicular and luteal phases. Anat Rec 235(3):399–410. https://doi.org/10.1002/ar.1092350309
Teilmann SC, Byskov AG, Pedersen PA, Wheatley DN, Pazour GJ, Christensen ST (2005) Localization of transient receptor potential ion channels in primary and motile cilia of the female murine reproductive organs. Mol Reprod Dev 71(4):444–452. https://doi.org/10.1002/mrd.20312
Bylander A, Nutu M, Wellander R, Goksör M, Billig H, Larsson DJ (2010) Rapid effects of progesterone on ciliary beat frequency in the mouse fallopian tube. Reprod Biol Endocrinol 8(1):1–8. https://doi.org/10.1186/1477-7827-8-48
Burton JC, Wang S, Stewart CA, Behringer RR, Larina IV (2015) High-resolution three-dimensional in vivo imaging of mouse oviduct using optical coherence tomography. Biomed Opt Express 6(7):2713–2723. https://doi.org/10.1364/BOE.6.002713
Wang S, Burton JC, Behringer RR, Larina IV (2015) In vivo micro-scale tomography of ciliary behavior in the mammalian oviduct. Sci Rep 5:13216. https://doi.org/10.1038/srep13216. http://www.nature.com/articles/srep13216#supplementary-information
Fercher AF, Drexler W, Hitzenberger CK, Lasser T (2003) Optical coherence tomography—principles and applications. Rep Prog Phys 66(2):239
Schmitt JM, Xiang SH, Yung KM (1999) Speckle in optical coherence tomography. J Biomed Opt 4(1):95–105. https://doi.org/10.1117/1.429925
Cho A, Haruyama N, Kulkarni AB (2009) Generation of transgenic mice. In: Juan S. Bonifacino et al (eds) Current protocols in cell biology. Chapter19:Unit 19.11. John Willey & Sons, Inc., New Jersey. doi:https://doi.org/10.1002/0471143030.cb1911s42
Ploutarchou P, Melo P, Day AJ, Milner CM, Williams SA (2015) Molecular analysis of the cumulus matrix: insights from mice with O-glycan-deficient oocytes. Reproduction 149(5):533–543. https://doi.org/10.1530/REP-14-0503
Oldenburg AL, Chhetri RK, Hill DB, Button B (2012) Monitoring airway mucus flow and ciliary activity with optical coherence tomography. Biomed Opt Express 3(9):1978–1992. https://doi.org/10.1364/boe.3.001978
Wang S, Burton JC, Behringer RR, Larina IV (2016) Functional optical coherence tomography for high-resolution mapping of cilia beat frequency in the mouse oviduct in vivo. Proc SPIE 9689:96893R-96895
Caligioni C (2009) Assessing reproductive status/stages in mice. In: Jacqueline N. Crawley et al (eds) Current protocols in neuroscience. Appendix:Appendix-4I. John Willey & Sons, Inc., New Jersey. doi:https://doi.org/10.1002/0471142301.nsa04is48
Acknowledgments
This work was supported by the National Institute of Health grant R01HL120140 (I.V.L.) and the American Heart Association grant 16POST30990070 (S.W.).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Wang, S., Larina, I.V. (2018). In Vivo Imaging of the Mouse Reproductive Organs, Embryo Transfer, and Oviduct Cilia Dynamics Using Optical Coherence Tomography. In: Delgado-Olguin, P. (eds) Mouse Embryogenesis. Methods in Molecular Biology, vol 1752. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-7714-7_5
Download citation
DOI: https://doi.org/10.1007/978-1-4939-7714-7_5
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-7713-0
Online ISBN: 978-1-4939-7714-7
eBook Packages: Springer Protocols