Abstract
Background
The co-occurrence of diabetes and osteoporosis is common in postmenopausal women. For the treatment of postmenopausal osteoporosis, current guidelines recommend initial treatment with bisphosphonates, but it is unclear whether bisphosphonates provide a similar degree of therapeutic efficacy in patients with diabetes. This study sought to compare the efficacy of monthly oral ibandronate for retaining bone mineral density (BMD) in diabetic and non-diabetic postmenopausal women with osteoporosis.
Methods
Postmenopausal osteoporotic women with or without diabetes were enrolled in this study from three hospitals in an open-label approach from 2018 to 2020. Each group of patients received oral ibandronate 150 mg once monthly for 1 year. BMD, trabecular bone score (TBS), serum C-terminal telopeptide of type I collagen (CTx) and procollagen type 1 N-terminal propeptide (P1NP) were evaluated prospectively. Treatment-emergent adverse events and changes in glucose metabolism during drug use were also monitored.
Results
Among the 120 study participants, 104 (86.7%) completed the study. Following 1 year of treatment, BMD increased by 3.41% vs. 3.71% in the lumbar spine, 1.30% vs. 1.18% in the femur neck, and 1.51% vs. 1.58% in the total hip in the non-diabetes and diabetes groups, respectively. There were no significant differences in BMD changes between the groups, and the differences in CTx or P1NP changes between groups were not significant. We did not observe any significant differences in baseline TBS values or the degree of change between before and after 1 year of ibandronate treatment in either group in this study. A total of 11 adverse events (9.2%) that recovered without sequelae occurred among the 120 included patients, and there was no significant difference in the frequency of adverse events between the groups (p = 0.862). The changes in fasting glucose and glycated hemoglobin levels between before and after treatment were not significant in the diabetic group.
Conclusions
Bisphosphonate therapy showed similar increases in BMD and decreases in CTx and P1NP of postmenopausal women with and without diabetes. Monthly oral ibandronate can be a safe and effective therapeutic option in postmenopausal osteoporosis patients with type 2 diabetes.
Trial registration
NCT number: NCT05266261, Date of registration: 04 March 2022.
Similar content being viewed by others
Background
Type 2 diabetes mellitus (T2DM) and osteoporosis are common in postmenopausal women [1]. Previous studies have reported that diabetes can cause deterioration of bone quality due to accumulation of advanced glycosylation end products in bone architecture or progressive microangiopathy surrounding the bone environment [2,3,4,5]. In addition, the risk of falls increases due to the occurrence of hypoglycemic events [6] and the diabetic complications such as diabetic retinopathy [7], diabetic peripheral neuropathy [8], and diabetic autonomic neuropathy. Therefore, more attention is needed to treat osteoporosis and prevent fractures in diabetic patients [9].
In postmenopausal women at high risk of fracture, current guidelines recommend initial treatment with bisphosphonates including alendronate, risedronate, zoledronic acid, and ibandronate to reduce fracture risk [10, 11]. However, it is unclear whether bisphosphonates provide a similar degree of therapeutic efficacy in patients with diabetes, who can present with complex situations; such as poor bone quality, multiple comorbidities, or use of various medications. Bisphosphonates reduce osteocalcin level by inhibiting osteoclast activity [12], and osteocalcin is associated with insulin secretion and sensitivity [13]. Not surprisingly, concerns have been raised that bisphosphonate therapy can adversely affect glucose metabolism [14, 15].
However, few studies have investigated the efficacy and safety of ibandronate in patients with T2DM. Ibandronate is a therapeutic agent that can be applied with intermittent dosing because it is sequestered in the bone with high binding affinity [16]. Once-monthly oral ibandronate, in a 150 mg tablet, has been deemed effective for increasing bone mineral density (BMD) and preventing osteoporotic fractures [17, 18]. Although the effectiveness of ibandronate for preventing non-vertebral fractures is controversial, it is used widely due to the convenience of once-monthly dosing. Based on previous studies [19, 20], we hypothesized that the effect of ibandronate in T2DM patients is not drastically different than in non-diabetic patients.
This study was conducted to provide evidence of the efficacy and safety of once-monthly ibandronate, at a 150 mg dose, in women with post-menopausal osteoporosis and T2DM. In addition, the influence of ibandronate on glucose metabolism was investigated in diabetic patients.
Methods
Study design
This study was a multicenter, prospective clinical trial with an open-label design. Patients were enrolled from Samsung Medical Center, Yeouido St. Mary’s Hospital, or Seoul National University Bundang Hospital, between 2018 and 2020. The study protocol was reviewed and approved by the institutional review board of Samsung Medical Center (SMC 2018–02-054), Yeouido St.Mary’s Hospital (SC18MEDV0024), and Seoul National University Bundang Hospital (B-1712/439–003). The study was performed in accordance with Declaration of Helsinki, and informed consent was obtained from all subjects. The current study was a prospective open-label clinical trial, and the protocol was submitted World Health Organization International Clinical Trial Registry Platform (NCT number: NCT05266261, Date of registration: 4 March 2022).
Participants
The patients recruited were as those who had previously visited the hospital for other diseases, and who consented to the clinical trial explanation from the attending physician. Patients were enrolled based on the following inclusion criteria: a) age of at least 55 years at the time of screening; b) postmenopausal woman, defined as the absence of menstruation for at least 12 consecutive months; and c) a diagnosis of osteoporosis (indicated by a BMD T-score ≤ − 2.5 points in the lumbar spine, total hip, or femoral neck). Exclusion criteria were as follows: a) a history of osteoporosis treatment within 3 years of the study, b) underlying disease (e.g., heart failure, liver disease, renal disease, or malignancy) and/or the use of drugs that affect bone metabolism (e.g., steroids, immunosuppressants, gonadotropin-releasing hormone agonists, aromatase inhibitors, thiazolidinedione drugs, anticonvulsants, and antidepressants); and c) a history of adverse effects of bisphosphonate or difficulty taking the drug due to an inability to sit or the presence of upper gastrointestinal disease. Criteria for stopping the clinical trial were as follows: a) withdrawal of consent from study subjects, b) serious adverse reaction confirmed by the investigators c) violation of the protocol; or d) non-cooperation of subjects.
Interventions
One tablet (150 mg of ibandronate + 24,000 IU of cholecalciferol) was provided on the same date each month with a sufficient amount of water at least 1 hour before the ingestion of food and other drugs. Calcium and vitamin D replacements were administered for all subjects during the study period (daily oral formulation: 1250 mg of calcium carbonate + 1000 IU of cholecalciferol). Study activities were discontinued if the subject withdrew consent or reported an adverse reaction to the study drug.
Measurements
Subjects visited the clinic every 6 months and were instructed to fast for more than 8 hours in the morning of the visit day; blood pressure, pulse rate, respiration rate, and body temperature were measured at rest, and blood sampling was performed. BMD was measured at different centers using two types of dual-energy X-ray absorptiometry devices, either the Lunar Prodigy Advance (GE Healthcare., Chicago, IL, USA) or Hologic Delphi W (Hologic Inc., Marlborough, MA, USA), and was evaluated at baseline and at the end of the study. Harmonizing equations [21] were used to compare the baseline BMD values of the study groups. The trabecular bone score (TBS) of the lumbar spine was evaluated in the patient group that used the Lunar scanner. Serum C-terminal telopeptide of type I collagen (CTx) and procollagen type 1 N-terminal propeptide (P1NP) levels were measured by immunoassay methods using Elecsys kits – 07296355001 V4 and 07296509001 V4 (Roche Diagnostic Corp., Basel, Switzerland).
Outcomes
The primary efficacy endpoint was the percentage change of BMD at the lumbar spine, femur neck, or total hip after 1 year, and secondary efficacy endpoints were percentage changes in the serum bone turnover markers CTx and P1NP. The primary safety endpoint was an adverse event requiring drug discontinuation; secondary safety endpoints were changes in fasting glucose or glycosylated hemoglobin (HbA1c) level at the start or end of the study in diabetic patients.
Sample size
When the percentage change in lumbar spine BMD was compared between diabetic and non-diabetic patients, a difference of at least 2.0 percentage points was considered clinically meaningful, with consideration of error. The standard deviation was assumed to be 4.5 based on a previous study, and initial study planning calculated that about 45.5 study subjects were required for each group to achieve results with 80% power and at a 5% significance level (two-tailed test). Considering the anticipated dropout rate of 20%, a total of 120 patient enrollments were planned.
Statistical analysis
Data normality was assessed through the Shapiro-Wilk Test. For continuous variables that formed normal distributions, the mean and standard deviation values were expressed and compared using a t-test. For continuous variables that did not form normal distributions, the median and interquartile ranges (IQR) were expressed and compared with the Wilcoxon rank-sum test. To analyze the percentage difference in BMD change, an analysis of covariance was performed with age as a covariate, and the result was expressed as the least square mean and its 95% confidence interval. Changes in bone turnover markers were analyzed using the generalized estimating equation for repeated-measures analysis and compared using Tukey’s method for multiple comparisons. Two sided P values less than 0.05 were considered statistically significant. Statistical analysis was performed using R version 4.1.0 (R Foundation for Statistical Computing, Vienna, Austria).
Results
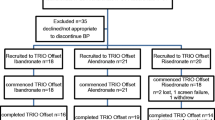
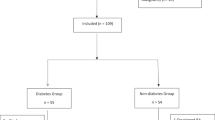
Baseline characteristics
Among the 120 study participants, 104 (86.7%) completed the study (Fig. 1), and group analysis indicated that the diabetic group was older and had higher body mass index (BMI) values. However, BMD, TBS and T-scores were not significantly different between the two groups, and there were no significant differences in baseline bone turnover markers or serum vitamin D level between the two groups (Table 1). The duration of T2DM in the diabetic group was a median of 12 years (IQR, 2–17.25 years). Treatment methods for T2DM varied from mono-drug therapy to insulin use. The baseline HbA1c level for T2DM patients ranged from 5.5 to 9.1% (Additional File 1: Fig. S1).
Efficacy
Following 1 year of treatment, the BMD increased by 3.5% in the lumbar spine, 1.2% in the femur neck, and 1.5% in the total hip area for all treated patients. When the increments in BMD were investigated for each group, the results were 3.41% vs. 3.71% for the lumbar spine, 1.30% vs. 1.18% for the femur neck, and 1.51% vs. 1.58% in the total hip in the non-diabetes and diabetes group, respectively. There was no significant difference in these BMD changes between groups (Fig. 2). Changes in TBS values of the lumbar spine were analyzed for patients who had undergone imaging with the Lunar scanner. The mean (standard deviation) TBS values were 1.310 (0.058) at baseline and 1.333 (0.071) at the end of the study in the non-diabetic group and 1.289 (0.076) and 1.288 (0.088) in the diabetic group, respectively. Unlike BMD, no significant change in the TBS between before and after treatment was observed (p = 0.20 and p = 0.80).
There was no difference in baseline level for changes in bone turnover markers between groups (Fig. 3). The levels of bone turnover markers decreased significantly following treatment (p < 0.001). The degrees of reduction at 6 months in the non-diabetes and diabetes groups were 58.1% vs. 48.6% (p = 0.09) for CTx and 54.9% vs. 51.1% (p = 0.19) for P1NP, respectively. In the diabetic group, the efficacy of ibandronate on BMD and bone turnover markers were evaluated according to the duration of diabetes, treatment method, and baseline HbA1c of the patient, but no significant difference was observed (Additional File 2: Table S1).
The number of patients with previously symptomatic compression fractures reported at the baseline screening visit was six in the non-diabetic group and 13 in the diabetic group, respectively. More patients in the diabetic group tended to report osteoporotic fractures, but this difference was not statistically significant. Radiologically confirmed lumbar compression fractures were investigated through baseline X-ray imaging, and nine and 15 patients in the non-diabetic group and the diabetic group, respectively, were identified as having such fractures; this difference was not statistically significant. Newly developed fractures were not analyzed as a study endpoint because the follow-up treatment period was limited to 1 year. However, no patients reported symptomatic fractures, including atypical fractures, during the study period. During follow-up, one radiologic compression fracture of the lumbar spine (L2) was confirmed in a patient in the diabetic group.
Safety
A total of 11 adverse events (9.2%) occurred among the 120 included patients (Table 2). One patient in the non-diabetic group developed a persistent fever and was withdrawn from the study based on the investigator’s decision. Two patients in the non-diabetic group and three patients in the diabetic group stopped treatment due to myalgia. One patient in each group requested to discontinue the study due to dyspepsia, and one patient in the non-diabetic group and two patients in the diabetic group requested to discontinue the study due to weight loss. All adverse reactions recovered without sequelae after discontinuation of the drug. There was no significant difference in the frequency of adverse events between the two groups (p = 0.862). To evaluate the effects of ibandronate on glucose metabolism, levels of fasting glucose and HbA1c were evaluated in the diabetic group, and the differences between before and after treatment were not statistically significant (Table 3).
Discussion
The purpose of this study was to determine whether monthly oral ibandronate therapy is as effective for retaining BMD in diabetic patients compared to non-diabetic patients. No significant difference was observed in the changes in BMD and suppression of bone turnover markers between the diabetic and non-diabetic groups. In addition, measurements of fasting glucose and HbA1c for evaluating glucose metabolism in diabetic patients did not significantly differ between before and after treatment.
In this study population, although the T2DM patient group was older, the baseline BMD and T-score values were not statistically different between the two groups. Therefore, patient age was corrected for analysis of covariance to compare differences in BMD changes. The average increase in lumbar spine BMD was 3.5% in all patients in this study, and there were increases of 3.4 and 3.7% in BMD in the lumbar spine in the non-diabetic and diabetic groups, respectively (Fig. 2). The observed 3.5% increase in BMD was lower than that reported in Western studies, but this result is similar to findings in a meta-analysis of randomized controlled trials [22]. There was also no significant difference in BMD change between the two groups in the femoral neck or the total hip area, which showed an increase less than 2%. However, the effect of ibandronate on lumbar spine BMD was confirmed, which is consistent with the results of a previous large-scale study that documented significant prevention of vertebral fracture with ibandronate therapy [23].
Discordance has been reported in that patients with T2DM have a high BMD but are at an increased fracture risk [24, 25]. Previous investigators have reported significantly lower TBS values in T2DM patients, and thus attempted to explain the increased fracture risk in diabetic patients through the mechanism of low TBS values [26, 27]. However, we did not observe any significant differences in baseline TBS values or the degree of change between before and after one year of ibandronate treatment in either group in this study. This is consistent with previous observations that changes in TBS were less responsive to bisphosphonate treatment compared to BMD [28, 29]. The coefficient of variation indicating reproducibility or precision of TBS is more than twice that of BMD [30], and it is thought that the number of patients confirming statistical significance is greater than the sample size of this study. Therefore, the interpretation of TBS in T2DM patients may be controversial, and further studies are needed.
Similar to the results of previous reports [31, 32], the degree of biomarker reduction was about 50% for both CTx and P1NP in this study. There was no significant difference in baseline biomarker levels in the two groups, and the effect could be verified by the decrease in bone turnover markers without a significant difference between the diabetic and non-diabetic groups (Fig. 3). Diabetic patients generally tended to exhibit low bone turnover [33, 34], but bone turnover markers were not significantly different in this study. This may be due to the fact that most patients had good glycemic control with a HbA1c level under 7%. However, considering that the diabetic group was older than the non-diabetic group, it is a meaningful that the changes in bone turnover markers were not significantly different in this study. Low bone turnover in diabetic patients can be related to side effects associated with the use of bisphosphonates. However, the inhibition of bone turnover in the diabetic group was similar to that in the non-diabetic group according to the results of this study. In addition, similar changes in bone turnover markers in both groups prove that it is justified to use bone turnover markers to monitor drug efficacy even in diabetic patients.
In terms of safety, the proportion of patients who discontinued study participation was almost the same in both groups, and there was no significant difference in the incidence of adverse effects between the groups (Table 2). Although the diabetic group was older and more side effects can be expected in this population due to polypharmacy [35], the absence of a difference in the incidence of side effects in the groups demonstrates the safety of the drug. Therefore, we suggest that monthly ibandronate (150 mg ibandronate + 24,000 IU vitamin D tablet) is tolerable in postmenopausal women with diabetes.
Bisphosphonate therapy reduced bone turn-over and resulted in lowering osteocalcin secretion, which may influence glucose metabolism. However, it is not clear how bisphosphonate treatment affects glucose metabolism in diabetic patients. Researchers previously reported that the effect of altered osteocalcin levels on glucose metabolism with bisphosphonate therapy was inconsequential [36, 37]. Some studies insist a rather favorable effect of bisphosphonate therapy in diabetic patients [38, 39]. The results of this study also show no differences in fasting glucose and HbA1c levels between before and after treatment (Table 3). Based on the results of our study, the effect of ibandronate on diabetes is not significant, so the use of ibandronate in T2DM patients appears to be safe in terms of glucose metabolism. Subgroup analysis was performed to further assess the diabetic group of this study based on patient distribution (Additional File 1: Fig. S1) and treatment effects (Additional File 2: Table S1). However, no significant difference was noted in the efficacy of ibandronate according to diabetic status.
This study had several limitations. First, researcher intervention cannot be excluded since the study was conducted with an open-label design. In particular, the recruitment process of patients in this study was conducted with those who received research proposals from the attending physician, rather than open recruitment. Second, the number of subjects in this study was smaller than in the previous study [40], and the research approach might have overlooked an effect on femur neck and total hip BMD, where the effect of ibandronate was relatively small. In addition, a larger number of patients is needed to investigate TBS change due to precision error. Third, patients in the diabetes group were composed of patients with good glycemic control and different diabetic treatment methods, it is difficult to confirm definite influence of ibandronate on glucose metabolism from this study alone. Therefore, additional studies are necessary for uncontrolled diabetes mellitus or type 1 diabetic patients. Finally, it is difficult to evaluate the effect of ibandronate treatment on long-term fracture prevention based only on the results of this study because of the one-year study duration. Nevertheless, this study is a rare study designed to investigate the difference in treatment response to osteoporosis medications between patients with and without diabetes, and the results provide data that support the use of ibandronate therapy in patients with diabetes. To the best of our knowledge, this study is the first study on the use of ibandronate in diabetic patients.
Conclusions
In conclusion, bisphosphonate therapy showed similar increases in BMD and decreases in bone turnover markers in postmenopausal women with and without diabetes. Monthly oral ibandronate can be a safe and effective therapeutic option in postmenopausal osteoporosis patients with T2DM.
Availability of data and materials
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Abbreviations
- T2DM :
-
Type 2 diabetes mellitus
- BMD :
-
Bone mineral density
- DEXA :
-
Dual-energy X-ray absorptiometry
- TBS :
-
Trabecular bone score
- CTx :
-
C-terminal telopeptide of type I collagen
- P1NP :
-
Procollagen type 1 N-terminal propeptide
- HbA1c :
-
Glycosylated hemoglobin
- IQR :
-
Interquartile ranges
- BMI :
-
Body mass index
References
Paschou SA, Dede AD, Anagnostis PG, Vryonidou A, Morganstein D, Goulis DG. Type 2 diabetes and osteoporosis: A guide to optimal management. J Clin Endocrinol Metab. 2017;102(10):3621–34.
Brownlee M, Cerami A, Vlassara H. Advanced glycosylation end products in tissue and the biochemical basis of diabetic complications. N Engl J Med. 1988;318(20):1315–21.
Kusumbe AP, Ramasamy SK, Adams RH. Coupling of angiogenesis and osteogenesis by a specific vessel subtype in bone. Nature. 2014;507(7492):323–8.
Schwartz AV. Diabetes mellitus: does it affect bone? Calcif Tissue Int. 2003;73(6):515–9.
Hamann C, Kirschner S, Gunther KP, Hofbauer LC. Bone, sweet bone--osteoporotic fractures in diabetes mellitus. Nat Rev Endocrinol 2012;8(5):297–305.
Chiang JI, Li TC, Li CI, Liu CS, Meng NH, Lin WY, et al. Visit-to-visit variation of fasting plasma glucose is a predictor of hip fracture in older persons with type 2 diabetes: the Taiwan diabetes study. Osteoporos Int. 2016;27(12):3587–97.
Ivers RQ, Cumming RG, Mitchell P, Peduto AJ. Blue Mountains eye S. diabetes and risk of fracture: the Blue Mountains eye study. Diabetes Care. 2001;24(7):1198–203.
Kim JH, Jung MH, Lee JM, Son HS, Cha BY, Chang SA. Diabetic peripheral neuropathy is highly associated with nontraumatic fractures in Korean patients with type 2 diabetes mellitus. Clin Endocrinol. 2012;77(1):51–5.
Ferrari SL, Abrahamsen B, Napoli N, Akesson K, Chandran M, Eastell R, et al. Diagnosis and management of bone fragility in diabetes: an emerging challenge. Osteoporos Int. 2018;29(12):2585–96.
Camacho PM, Petak SM, Binkley N, Diab DL, Eldeiry LS, Farooki A, et al. American Association of Clinical Endocrinologists/American College of Endocrinology Clinical Practice Guidelines for the diagnosis and treatment of postmenopausal Osteoporosis-2020 update. Endocr Pract. 2020;26(Suppl 1):1–46.
Shoback D, Rosen CJ, Black DM, Cheung AM, Murad MH, Eastell R. Pharmacological Management of Osteoporosis in postmenopausal women: an Endocrine Society guideline update. J Clin Endocrinol Metab. 2020;105(3).
Cooper C, Emkey RD, McDonald RH, Hawker G, Bianchi G, Wilson K, et al. Efficacy and safety of oral weekly ibandronate in the treatment of postmenopausal osteoporosis. J Clin Endocrinol Metab. 2003;88(10):4609–15.
Fernández-Real JM, Izquierdo M, Ortega F, Gorostiaga E, Gómez-Ambrosi J, Moreno-Navarrete JM, et al. The relationship of serum osteocalcin concentration to insulin secretion, sensitivity, and disposal with hypocaloric diet and resistance training. J Clin Endocrinol Metab. 2009;94(1):237–45.
Yeap BB, Chubb SA, Flicker L, McCaul KA, Ebeling PR, Beilby JP, et al. Reduced serum total osteocalcin is associated with metabolic syndrome in older men via waist circumference, hyperglycemia, and triglyceride levels. Eur J Endocrinol. 2010;163(2):265–72.
Kindblom JM, Ohlsson C, Ljunggren O, Karlsson MK, Tivesten A, Smith U, et al. Plasma osteocalcin is inversely related to fat mass and plasma glucose in elderly Swedish men. J Bone Miner Res. 2009;24(5):785–91.
Leu CT, Luegmayr E, Freedman LP, Rodan GA, Reszka AA. Relative binding affinities of bisphosphonates for human bone and relationship to antiresorptive efficacy. Bone. 2006;38(5):628–36.
Chesnut CH 3rd, Skag A, Christiansen C, Recker R, Stakkestad JA, Hoiseth A, et al. Effects of oral ibandronate administered daily or intermittently on fracture risk in postmenopausal osteoporosis. J Bone Miner Res. 2004;19(8):1241–9.
Miller PD, Recker RR, Reginster JY, Riis BJ, Czerwinski E, Masanauskaite D, et al. Efficacy of monthly oral ibandronate is sustained over 5 years: the MOBILE long-term extension study. Osteoporos Int. 2012;23(6):1747–56.
Schwartz AV. Efficacy of osteoporosis therapies in diabetic patients. Calcif Tissue Int. 2017;100(2):165–73.
Anagnostis P, Paschou SA, Gkekas NN, Artzouchaltzi AM, Christou K, Stogiannou D, et al. Efficacy of anti-osteoporotic medications in patients with type 1 and 2 diabetes mellitus: a systematic review. Endocrine. 2018;60(3):373–83.
Fan B, Lu Y, Genant H, Fuerst T, Shepherd J. Does standardized BMD still remove differences between Hologic and GE-lunar state-of-the-art DXA systems? Osteoporos Int. 2010;21(7):1227–36.
Lee YH, Song GG. Efficacy and safety of monthly 150 mg oral ibandronate in women with postmenopausal osteoporosis: a systematic review and meta-analysis of randomized controlled trials. Korean J Intern Med. 2011;26(3):340–7.
Harris ST, Reginster JY, Harley C, Blumentals WA, Poston SA, Barr CE, et al. Risk of fracture in women treated with monthly oral ibandronate or weekly bisphosphonates: the eValuation of IBandronate efficacy (VIBE) database fracture study. Bone. 2009;44(5):758–65.
Vestergaard P. Discrepancies in bone mineral density and fracture risk in patients with type 1 and type 2 diabetes--a meta-analysis. Osteoporos Int. 2007;18(4):427–44.
de L II, van der Klift M, de Laet CE, van Daele PL, Hofman A, Pols HA. Bone mineral density and fracture risk in type-2 diabetes mellitus: the Rotterdam study. Osteoporos Int. 2005;16(12):1713–20.
Leslie WD, Aubry-Rozier B, Lamy O, Hans D. Manitoba bone density P. TBS (trabecular bone score) and diabetes-related fracture risk. J Clin Endocrinol Metab. 2013;98(2):602–9.
Kim JH, Choi HJ, Ku EJ, Kim KM, Kim SW, Cho NH, et al. Trabecular bone score as an indicator for skeletal deterioration in diabetes. J Clin Endocrinol Metab. 2015;100(2):475–82.
Senn C, Gunther B, Popp AW, Perrelet R, Hans D, Lippuner K. Comparative effects of teriparatide and ibandronate on spine bone mineral density (BMD) and microarchitecture (TBS) in postmenopausal women with osteoporosis: a 2-year open-label study. Osteoporos Int. 2014;25(7):1945–51.
Di Gregorio S, Del Rio L, Rodriguez-Tolra J, Bonel E, García M, Winzenrieth R. Comparison between different bone treatments on areal bone mineral density (aBMD) and bone microarchitectural texture as assessed by the trabecular bone score (TBS). Bone. 2015;75:138–43.
Bandirali M, Di Leo G, Messina C, Pastor Lopez MJ, Mai A, Ulivieri FM, et al. Reproducibility of trabecular bone score with different scan modes using dual-energy X-ray absorptiometry: a phantom study. Skelet Radiol. 2015;44(4):573–6.
Lewiecki EM, Keaveny TM, Kopperdahl DL, Genant HK, Engelke K, Fuerst T, et al. Once-monthly oral ibandronate improves biomechanical determinants of bone strength in women with postmenopausal osteoporosis. J Clin Endocrinol Metab. 2009;94(1):171–80.
Eekman DA, Bultink IE, Heijboer AC, Dijkmans BA, Lems WF. Bone turnover is adequately suppressed in osteoporotic patients treated with bisphosphonates in daily practice. BMC Musculoskelet Disord. 2011;12:167.
Hygum K, Starup-Linde J, Harslof T, Vestergaard P, Langdahl BL. MECHANISMS IN ENDOCRINOLOGY: diabetes mellitus, a state of low bone turnover - a systematic review and meta-analysis. Eur J Endocrinol. 2017;176(3):R137–R57.
Purnamasari D, Puspitasari MD, Setiyohadi B, Nugroho P, Isbagio H. Low bone turnover in premenopausal women with type 2 diabetes mellitus as an early process of diabetes-associated bone alterations: a cross-sectional study. BMC Endocr Disord. 2017;17(1):72.
Peron EP, Ogbonna KC, Donohoe KL. Antidiabetic medications and polypharmacy. Clin Geriatr Med 2015;31(1):17–27, vii.
Schwartz AV, Schafer AL, Grey A, Vittinghoff E, Palermo L, Lui LY, et al. Effects of antiresorptive therapies on glucose metabolism: results from the FIT, HORIZON-PFT, and FREEDOM trials. J Bone Miner Res. 2013;28(6):1348–54.
Hong SH, Koo JW, Hwang JK, Hwang YC, Jeong IK, Ahn KJ, et al. Changes in serum osteocalcin are not associated with changes in glucose or insulin for osteoporotic patients treated with bisphosphonate. J Bone Metab. 2013;20(1):37–41.
Karimi Fard M, Aminorroaya A, Kachuei A, Salamat MR, Hadi Alijanvand M, Aminorroaya Yamini S, et al. Alendronate improves fasting plasma glucose and insulin sensitivity, and decreases insulin resistance in prediabetic osteopenic postmenopausal women: A randomized triple-blind clinical trial. J Diabetes Investig. 2019;10(3):731–7.
Viggers R, Al-Mashhadi Z, Starup-Linde J, Vestergaard P. Alendronate use and risk of type 2 diabetes: A Nationwide Danish nested case-control study. Front Endocrinol (Lausanne). 2021;12:771426.
Miller PD, McClung MR, Macovei L, Stakkestad JA, Luckey M, Bonvoisin B, et al. Monthly oral ibandronate therapy in postmenopausal osteoporosis: 1-year results from the MOBILE study. J Bone Miner Res. 2005;20(8):1315–22.
Acknowledgements
We would like to thank all the participants of the studies, and Handok Pharmaceutical that funded the research.
Funding
This work was supported by the research fund of Handok Pharmaceutical.
Author information
Authors and Affiliations
Contributions
Jinyoung Kim and Kyoung Min Kim contributed equally to this work. Kyoung Min Kim, Ki-Hyun Baek and Yong-Ki Min designed the study, enrolled the patients, quality assessment. Soo Lim and Moo-Il Kang performed literature screening and data extraction. Jinyoung Kim conducted data extraction and data analysis, and wrote the original draft. Kyoung Min Kim and Ki-Hyun Baek contributed to reviewing and editing of the manuscript. All authors have read the final manuscript and agreed to publication. Correspondence to Ki-Hyun Baek or Yong-Ki Min.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
The study protocol was reviewed and approved by the institutional review board of Samsung Medical Center (SMC 2018–02-054), Yeouido St.Mary’s Hospital (SC18MEDV0024), and Seoul National University Bundang Hospital (B-1712/439–003). The study was performed in accordance with Declaration of Helsinki, and informed consent was obtained from all subjects.
Consent for publication
Not applicable for this study.
Competing interests
The funder had no role in data collection, analysis, or preparation of the manuscript. The authors have no other conflicts of interest to declare.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Kim, J., Kim, K.M., Lim, S. et al. Efficacy of bisphosphonate therapy on postmenopausal osteoporotic women with and without diabetes: a prospective trial. BMC Endocr Disord 22, 99 (2022). https://doi.org/10.1186/s12902-022-01010-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12902-022-01010-w