Abstract
Injection of intraovarian platelet-rich plasma (PRP) was recently presented in terms of improvement ovarian function in women with a poor ovarian response (POR) or primary ovarian insufficiency (POI). In a before and after study, 17 poor responder women and 9 women with the diagnosis of POI were recruited. The multifocal intramedullary infusion of 1.5 ml activated PRP was performed into each ovary. The majority of women in both groups received the second PRP injection with the twofold increase in the dosage to 3ml, 3 months after the first injection. Evaluation of serum anti-mullerian hormone (AMH), follicle-stimulating hormone (FSH), luteinizing hormone (LH), and estradiol (E2) was performed. In addition, all women were followed with regard to pregnancy outcome up to delivery. In the POI group, menstrual restoration was monitored. The significant difference was not detected regarding the hormonal profile between the three time points in both groups. With regard to pregnancy outcome, 8/17 (47%) of PORs had spontaneous pregnancy in response to PRP injection. Of those, three women (37.55%) had abortions, whereas 4 pregnancies (50%) led to healthy live births, and one woman (12.5%) was in the 24th week of her pregnancy. Menstruation recovery occurred among 22.2% of women with POI after the second PRP injection, but no one became pregnant. Intraovarian injection of autologous PRP might be considered an alternative treatment in poor responders. As for women with POI, it is questionable whether PRP could induce menstrual recovery.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Both women with primary ovarian insufficiency (POI) and poor ovarian responders (PORs) with poor fertility outcomes are considered the main challenges of reproductive science. POI, which is described by significantly reduced ovarian reserve, menstrual irregularity, or amenorrhea earlier than 40 years of age, affects 1% of women at reproductive age [1]. At present, egg donation is the only treatment option for women with POI, and that is not usually welcomed by infertile couples who desire to have their own genetic offspring. On the other hand, PORs are presented by the low response to ovarian stimulation protocols with a frequency of 9 to 24% [2, 3]. Despite several approaches that have been investigated to improve the assisted reproductive technology (ART) outcome in PORs, the pregnancy rate remains low in these patients. Recently, platelet-rich plasma (PRP) has been widely used in regenerative treatment in different fields such as orthopedics, dermatology, dentistry, and aesthetic surgery [4,5,6,7]. PRP is platelet-rich blood plasma supplemented with cytokines and growth factors. Accumulation of platelet in a tissue stimulates cell proliferation and tissue regeneration through protein secretion in response to cytokines, and growth factors [8] lead to revert the cellular damage and tissue rejuvenation [9]. In the field of infertility, PRP was primarily used for the treatment of thin endometrium [10] and recurrent implantation failure [11]. Recently, it has been investigated in the cases of ovarian insufficiency [12, 13]. Since the studies which assessed the PRP beneficial effects on ovarian parameters are limited, in this study, we evaluated whether the intraovarian injection of autologous PRP would improve ovarian reserve through ovarian rejuvenation thus causing spontaneous pregnancy or menstrual recovery in women with POI and poor responders.
Materials and Methods
The study was conducted at Yazd Research and Clinical Center for Infertility, Yazd Reproductive Sciences Institute, between September 2018 and February 2020. The research proposal was approved by the Ethics Committee of Yazd Reproductive Sciences Institute, Shahid Sadoughi University of Medical Sciences, Yazd, Iran (IR.SSU.RSI.REC.1397.004). A written informed consent for participating in this trial was obtained from all poor responders and women with POI who rejected the oocyte donation program. This research project was registered in the Iranian Registry of Clinical Trials (IRCT) with the code of IRCT20180818040828N2.
Patient Selection
Twenty-six cases: 17 poor responder women and 9 women with the diagnosis of POI were recruited. POI patients were diagnosed according to ESHRE guideline: onset prior to 40 years of age, oligo-/amenorrhea for at least 4 months, and an elevated FSH level > 25 IU/l on two occasions > 4 weeks apart [14]. PORs were selected based on Bologna criteria: age of >40 years or any other risk factor for POR, history of POR detected by 3 or fewer oocytes in previous conventional stimulation protocols, and low ovarian reserve tests including anti-mullerian hormone (AMH <1.1 ng/ml) or antral follicular count (AFC <5 follicles). The PORs must fulfill at least two of the three mentioned criteria [15].
Exclusion criteria included body mass index (BMI) above 30 or less than 18, autoimmune diseases, thrombophilic disorders, ovarian insufficiency secondary to sex chromosome etiology, sexually transmitted diseases, tubal factor infertility, endocrine disorders such as thyroid dysfunction, endometriosis, previous major lower abdominal surgery and pelvic adhesions, renal failure, malignancy, and couples with abnormal semen parameters. All iatrogenic POI cases were also excluded. Participants did not receive any hormonal therapy 1 month prior to enrollment, during PRP treatment and for a 1-month period post-PRP injection. Screening tests for HIV (human immunodeficiency virus) antibody and HBS (hepatitis B surface) antigen were performed for all women.
Sample Preparation
PRP was prepared from autologous blood using the manufacturer’s instruction (Rooyagen, Tehran, Iran). Briefly, for each PRP infusion, approximately 20 ml of peripheral venous blood was drawn in the syringe that contained 3 ml of acid citrate A anticoagulant solution (ACD-A) (Arya Mabna Tashkhis, Iran) and centrifuged immediately at 1600g for 10 min. The blood was divided into three layers including red blood cells at the bottom, a buffy coat layer, and cellular plasma as the supernatant. The plasma layer and buffy coat were transferred to another tube and centrifuged at 3500g for 5 min to achieve 3 ml PRP of 3 to 5 times higher than basal blood samples [16]. Prepared PRP was stored for 1 h at a temperature of 4°C before injection, and PRP activation was performed using calcium gluconate (CG) in a 1:9 ratio [17, 18].
Intraovarian Injection
The injection was randomly scheduled in POI women with amenorrhea, whereas in POI women who were oligomenorrheic and PORs, PRP injection was done 10 days after the beginning of menstrual bleeding. PRP injection was done according to the previously defined method [13]. The multifocal intramedullary infusion of 1.5 ml activated PRP was performed using a 17-gauge single lumen needle into each ovary under minimal sedation via transvaginal ultrasound monitoring.
In the case of POI, a quantity of the injected PRP leaked into the peritoneal cavity due to atrophic ovaries with decreased volume. By the end of the procedure, accurate ultrasonography was performed to evaluate the pelvic area concerning the amount of leakage and vascular integrity. The majority of women in both groups received the second PRP injection with a twofold increase in the dosage to 3ml, 3 months after the first injection. Only five patients in the POR group did not receive the second PRP injection due to the spontaneous conception after the first PRP injection, which will be discussed in the “Result” section.
Hormonal Assessment and Follow-Up
Ovarian function was assessed through hormonal measurement in all women. Evaluation of serum AMH (ELISA immunoassay kit, Ansh Lab, Webster, USA), follicle-stimulating hormone (FSH) (AccuBind ELISA Kits, CA, USA), luteinizing hormone (LH) (AccuBind ELISA Kits, CA, USA), and estradiol (E2) (AccuBind ELISA Kits, CA, USA) was performed three times: prior to PRP injection and two consecutive months after the first PRP injection. In PORs, hormonal assessment was done on day 3 of the menstrual cycle. Intra-assay coefficient of variation for AMH was <10%, inter-assay coefficient of variation was <12%, detection range was 0.06–14.2 ng/mL, and minimum detectable concentration was 0.023 ng/mL.
In addition, all women were followed up for 1 year after the first PRP injection in terms of pregnancy outcome, and pregnant cases were observed up to delivery. In the POI group, menstrual restoration was monitored.
Outcome Parameters
Chemical pregnancy was considered a positive serum beta human gonadotropin hormone (hCG) that might progress into a clinical pregnancy or result in an early miscarriage. Clinical pregnancy was defined by detecting fetal heart activity in transvaginal ultrasonography 5 weeks after positive beta hCG. Abortion was pregnancy loss before the 20th week of gestation, and ongoing pregnancy was defined as pregnancies which continued after 12 weeks.
Statistical Analysis
The Statistical Package for Social Sciences 20(SPSS, SPSS Inc, Chicago) was used for data analysis. Values were expressed as the mean ± standard deviation (SD). Comparison of parametric and non-parametric variables before and after the intervention was performed using the paired t-test and Wilcoxon signed-rank test respectively. P < 0.05 was considered statistically significant. A confidence level was set at 95%.
Result
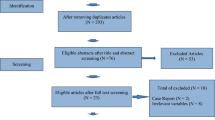
A total of 30 women, 19 poor responder patients and 11 women with the diagnosis of POI, were initially recruited. Of those, 2 women in each group did not meet the inclusion criteria. Finally, 17 PORs and 9 women with POI received PRP injection (Fig. 1). The mean age of women was 35.47 ± 4.34 years and 33.66 ± 4.84 years in PORs and POI groups, respectively. The mean duration post the menstrual cessation was 8.11±3.29 years in women with POI.
Demographic and clinical features of the poor responders and women with POI were listed in Tables 1 and 2, respectively.
Hormonal Profile and Pregnancy Outcome in PORs
The significant differences were not detected in FSH and LH levels 1 month and 2 months following PRP injection (Table 3, Fig. 2a and b). A non-significant elevation was detected in the E2 level 1 month post-PRP injection followed by an insignificant decline after the 2 months of PRP administration (Table 3, Fig. 2d). The serum AMH level was increased slightly 1 month after PRP treatment, even though the difference was not statistically significant. The serum AMH level showed an insignificant reduction 2 months post-PRP injection (Fig. 2c).
The mean serum levels of FSH (a), LH (b), AMH (d), and E2 (d) at three time points: prior to PRP injection, 1 month post-PRP injection, and 2 months post-PRP injection in the POR group; confidence level was set at 95%; data are presented as mean ± SD; LH, luteinizing hormone; FSH, follicular stimulating hormone; AMH, anti-mullerian hormone; E2, estradiol; POR, poor ovarian responder
With regard to pregnancy outcome, 8/17 (47%) of PORs had spontaneous pregnancy in response to PRP injection. Of those, three women (37.5%) had abortions after chemical pregnancy, whereas 4 pregnancies (50%) led to healthy live births, and one woman (12.5%) reached ongoing pregnancy. In detail, five patients received only the first PRP injection. Three of them refused the second injection due to spontaneous chemical pregnancy leading to abortion (cases 1, 2, 17), which means clinical pregnancy did not occur; one of them withdrew from the trial because of spontaneous conception just before the second injection (case 15); and the last one simply refused to take the second injection for personal reasons, although interestingly she became pregnant 6 months after the first injection (case 3). It is worth noting that the conception in the last two cases led to healthy live births (Table 1). Two women experienced spontaneous pregnancy 3 months after the second PRP injection. One pregnancy led to a healthy live birth (case 6), and the other pregnant women had the gestational age of 24 weeks at the time of manuscript submission (case 16). All women were allowed to conceive spontaneously. Only one woman, who had a remarkable increase in the serum AMH level after the second PRP injection, underwent in vitro fertilization (IVF) cycle 2 months later at her own insistence. But then, the cycle was canceled due to poor ovarian response to the stimulation protocol; interestingly, a spontaneous conception occurred 6 months post-PRP injection resulting in a live birth with a healthy baby boy (case 8).
Hormonal Profile and Pregnancy Outcome in POI Women
Serum FSH and LH were not decreased significantly for two consecutive months after PRP injection (Table 3, Fig. 3a and b). E2 showed a non-significant increase 1 month post-PRP injection with a subsequent decrease 2 months after PRP administration. Even though the E2 levels showed an elevation at two time points after PRP treatment in comparison with the level prior to PRP injection, the differences were not statistically significant (Table 3, Fig. 3d). Serum AMH level was increased 1 month after PRP injection; however, the AMH concentration was dropped below the level prior to PRP administration 2 months post-PRP injection. The differences were not statistically significant (Table 3, Fig. 3c).
The mean serum levels of FSH (a), LH (b), AMH (c), and E2 (d) at three time points: prior to PRP injection, 1 month post-PRP injection, and 2 months post-PRP injection in the POI group; confidence level was set at 95%; data are presented as mean ± SD; LH, luteinizing hormone; FSH, follicular stimulating hormone; AMH, anti-mullerian hormone; E2, estradiol; POI, primary ovarian insufficiency
Menstruation recovery occurred among 22.2% of women with POI after the second PRP injection, but no one became pregnant.
Discussion
In the current study, we examined the influence of intraovarian injection of autologous PRP on ovarian reserve factors and pregnancy outcome in women diagnosed with POI and PORs. Intraovarian PRP was injected to a total of 17 PORs and 7 women with POI. Among PORs, 8 women (47%) conceived spontaneously with a live birth rate of 50%, while POI women achieved no pregnancy, but menstruation recovery occurred in 22% of them. AMH, FSH, LH, and E2 levels did not change significantly before and after PRP injection in both groups.
Previously, the application of PRP for patients with thin endometrium and poor receptivity revealed successful results in terms of thickening endometrium and good pregnancy outcome following assisted reproduction [10, 19]. For the first time, Sills and colleagues reported intraovarian PRP injection in four women with poor response to ovarian stimulation and amenorrhea. They observed improvement in hormonal profile, oocyte retrieval, and blastocyst formation in all women after PRP administration [18]. Afterwards, Sfakianoudis and colleagues presented the first case of in vitro fertilized biochemical pregnancy after the intraovarian injection of autologous PRP. However, this pregnancy led to spontaneous abortion in the fifth week of gestational age [20].
All the aforementioned studies were performed based on the theory that the degenerative processes lead to the ovarian insufficiency through molecular pathways dysfunction which regulates the ovarian vascularization. On the other hand, some researchers believe that poor ovarian response could be treated by resourcing nutrients and hormonal supply [11, 13, 20,21,22]. By the introduction of PRP, it is assumed that platelet-derived factors may promote ovarian angiogenesis and stimulate follicular development by recovering ovarian microenvironment [13, 23]. PRP contains a high concentration of cytokines and growth factors such as platelet-derived growth factor, insulin-like growth factor, vascular endothelial growth factor, a platelet-derived angiogenic factor, and interleukin-8. All mentioned factors have a key role in tissue regenerative and healing [24], which is supposed to restore folliculogenesis and ovarian hormonal profile after intraovarian injection.
Unexpectedly, in our study, AMH and E2 levels along with FSH and LH concentrations did not change significantly within two consecutive months post-PRP injection in both POI and POR groups. Nevertheless, E2 and AMH levels showed a non-significant increase 1 month post-PRP injection. The levels dropped again 2 months after PRP administration. The small sample size of the study did not allow us to assess ovarian response predictors. As the other studies with larger sample size evaluated the possible relationship between the ovarian response to PRP injection and patients’ characteristics such as age, body mass index, baseline platelet concentration, pretreatment antral follicle count, or hormone levels [12, 18]. In contrast, the other studies reported a significant reduction in the patients’ FSH and LH levels plus increased AMH and E2 levels after PRP injection [20, 25, 26]. However, in the Cakiroglu study, similar to our results, the FSH level remained unchanged among POI women after PRP treatment [12].
With regard to pregnancy outcome, our result showed a reasonable pregnancy rate of 47% with a live birth rate of 50% of the pregnant women in poor responders. All pregnancies occurred spontaneously after the PRP administration. Only one woman was stimulated during an IVF cycle that was canceled due to the poor ovarian response. However, the patient became pregnant spontaneously. In a clinical trial, Farimani and colleagues compared the number of oocytes retrieved before and after PRP injection and reported an increase in the oocyte and embryos obtained after PRP administration. Three of the 19 women became pregnant including two spontaneous chemical pregnancies and one in vitro-fertilized clinical pregnancy that led to a healthy live birth [27]. Another case series presented 3 poor responder women who were treated with autologous PRP ovarian infusion. They reported a natural ongoing pregnancy (24 weeks of gestation), an uncomplicated ongoing pregnancy (17 weeks of gestation), and a successful live birth. The last two cases became pregnant after IVF cycles [26]. Moreover, Stojkovska and colleagues compared 40 poor responder women undergoing IVF with or without intraovarian injection of PRP. The result showed non-significant higher implantation and live birth rates in patients with PRP treatment [28].
In total, spontaneous conception is infrequent among women with POI, ranging from 2.2 to 14.2% using hormonal therapy, IVF, or stem cell therapy [29]. In this study, no cases of pregnancy occurred among women with POI after PRP administration, while 22% of women experienced menstrual recovery. On the contrary, other studies reported the beneficial effects of PRP injection on pregnancy outcome in these women. Pantos and colleagues performed intraovarian PRP injection for two women with POI aged 40 and 27 years and one 46-year-old menopausal woman. Menstrual restoration and improvement of the hormonal profile occurred in all three women. Furthermore, all women became pregnant naturally within 2–6 months after PRP treatment with uncomplicated ongoing pregnancies at the time of the case series publication [13]. A recent study with a large sample size performed intraovarian PRP injection for 311 women with ovarian insufficiency. Of those, 23 women (7.4%) had spontaneous pregnancy, 201 (64.8%) women who developed antral follicle(s) underwent IVF cycles, and 87 (27.8%) with no antral follicles did not receive further treatment. Out of 201 women undergoing IVF, 57 women had embryo transfer resulting in 13 pregnancies. Finally, of the 311 women who received PRP treatment, 25 (8.0%) achieved ongoing pregnancy or live birth either spontaneously or after IVF. The authors concluded that the intraovarian injection of autologous PRP could be considered an alternative treatment option among women with POI [12]. More recently, Sfakianoudis and his group conducted four pilot studies on PORs, POIs, perimenopause, and menopause women with 30 participants in each arm. PRP treatment in PORs resulted in a significant increase in the number of retrieved and metaphase II oocytes as well as the number of two pronuclei embryos and embryos reaching cleavage stage. In addition, the cancelation rate was significantly decreased. No information regarding pregnancy in this group of patients was reported. In the POI group, 60% of women achieved menstrual cycle recovery, along with reduced FSH levels. Three cases of spontaneous pregnancy leading to live birth were reported. Twenty four (80%) of perimenopausal women presented menstrual regularity and decrease FSH levels after PRP injection. Among them, 4 women conceived naturally including one spontaneous abortion and three healthy live births. 43.3% of menopause participants showed menstrual restoration and a significant decline in FSH levels. One woman became pregnant spontaneously leading to a healthy live birth [25].
Although, our study presented a reasonable pregnancy outcome and live birth for PORs using PRP treatment, it failed to show the beneficial effects of the intraovarian PRP injection in the management of women with POI. Despite the fact that this new technique brings hope to the field of reproductive medicine, this therapeutic choice should be individualized among the various subgroups of infertile women with different ages and hormonal profiles. Furthermore, some important questions have to be answered about the administration methods, number, and time intervals between the injections, as well as the adverse effects of resources containing factors with a much higher concentration than the natural environment on the embryos and potential child.
The main limitation of this study was the small sample size and lack of control group as well as randomization, mostly related to the “before and after” nature of the study.
In conclusion, intraovarian injection of autologous PRP might be considered an alternative treatment option in poor responders. However, the safety and efficacy of this novel therapeutic method, along with its short-term and long-term side effects, need to be investigated in more high-quality studies prior to clinical application.
Data Availability
All data and materials are available and transparent.
References
Nelson LM. Clinical practice. Primary ovarian insufficiency. N Engl J Med. 2009;360(6):606–14. https://doi.org/10.1056/NEJMcp0808697.
Papathanasiou A, Searle BJ, King NM, Bhattacharya S. Trends in 'poor responder' research: lessons learned from RCTs in assisted conception. Hum Reprod Update. 2016;22(3):306–19. https://doi.org/10.1093/humupd/dmw001.
Sunkara SK, Tuthill J, Khairy M, El-Toukhy T, Coomarasamy A, Khalaf Y, et al. Pituitary suppression regimens in poor responders undergoing IVF treatment: a systematic review and meta-analysis. Reprod Biomed Online. 2007;15(5):539–46. https://doi.org/10.1016/s1472-6483(10)60386-0.
Abu-Ghname A, Perdanasari AT, Davis MJ, Reece EM. Platelet-rich plasma: principles and applications in plastic surgery. Semin Plast Surg. 2019;33(3):155–61. https://doi.org/10.1055/s-0039-1693400.
Stahli A, Strauss FJ, Gruber R. The use of platelet-rich plasma to enhance the outcomes of implant therapy: a systematic review. Clin Oral Implants Res. 2018;29(Suppl 18):20–36. https://doi.org/10.1111/clr.13296.
Whitney KE, Liebowitz A, Bolia IK, Chahla J, Ravuri S, Evans TA, et al. Current perspectives on biological approaches for osteoarthritis. Ann N Y Acad Sci. 2017;1410(1):26–43. https://doi.org/10.1111/nyas.13554.
Zhang M, Park G, Zhou B, Luo D. Applications and efficacy of platelet-rich plasma in dermatology: A clinical review. J Cosmet Dermatol. 2018;17(5):660–5. https://doi.org/10.1111/jocd.12673.
Pavlovic V, Ciric M, Jovanovic V, Stojanovic P. Platelet rich plasma: a short overview of certain bioactive components. Open Med (Wars). 2016;11(1):242–7. https://doi.org/10.1515/med-2016-0048.
Ramaswamy Reddy SH, Reddy R, Babu NC, Ashok GN. Stem-cell therapy and platelet-rich plasma in regenerative medicines: a review on pros and cons of the technologies. J Oral Maxillofac Pathol. 2018;22(3):367–74. https://doi.org/10.4103/jomfp.JOMFP_93_18.
Zadehmodarres S, Salehpour S, Saharkhiz N, Nazari L. Treatment of thin endometrium with autologous platelet-rich plasma: a pilot study. JBRA Assist Reprod. 2017;21(1):54–6. https://doi.org/10.5935/1518-0557.20170013.
Farimani M, Poorolajal J, Rabiee S, Bahmanzadeh M. Successful pregnancy and live birth after intrauterine administration of autologous platelet-rich plasma in a woman with recurrent implantation failure: a case report. Int J Reprod Biomed (Yazd). 2017;15(12):803–6.
Cakiroglu Y, Saltik A, Yuceturk A, Karaosmanoglu O, Kopuk SY, Scott RT, et al. Effects of intraovarian injection of autologous platelet rich plasma on ovarian reserve and IVF outcome parameters in women with primary ovarian insufficiency. Aging (Albany NY). 2020;12(11):10211–22. https://doi.org/10.18632/aging.103403.
Pantos K, Simopoulou M, Pantou A, Rapani A, Tsioulou P, Nitsos N, et al. A Case Series on Natural conceptions resulting in ongoing pregnancies in menopausal and prematurely menopausal women following platelet-rich plasma treatment. Cell Transplant. 2019;28(9-10):1333–40. https://doi.org/10.1177/0963689719859539.
European Society for Human R, Embryology Guideline Group on POI, Webber L, Davies M, Anderson R, Bartlett J, et al. ESHRE Guideline: management of women with premature ovarian insufficiency. Hum Reprod. 2016;31(5):926–37. https://doi.org/10.1093/humrep/dew027.
Ferraretti AP, La Marca A, Fauser BC, Tarlatzis B, Nargund G, Gianaroli L, et al. ESHRE consensus on the definition of ‘poor response’ to ovarian stimulation for in vitro fertilization: the Bologna criteria. Hum Reprod. 2011;26(7):1616–24. https://doi.org/10.1093/humrep/der092.
Eftekhar M, Neghab N, Naghshineh E, Khani P. Can autologous platelet rich plasma expand endometrial thickness and improve pregnancy rate during frozen-thawed embryo transfer cycle? A randomized clinical trial. Taiwan J Obstet Gynecol. 2018;57(6):810–3. https://doi.org/10.1016/j.tjog.2018.10.007.
Gkini MA, Kouskoukis AE, Tripsianis G, Rigopoulos D, Kouskoukis K. Study of platelet-rich plasma injections in the treatment of androgenetic alopecia through an one-year period. J Cutan Aesthet Surg. 2014;7(4):213–9. https://doi.org/10.4103/0974-2077.150743.
Sills ES, Rickers NS, Li X, Palermo GD. First data on in vitro fertilization and blastocyst formation after intraovarian injection of calcium gluconate-activated autologous platelet rich plasma. Gynecol Endocrinol. 2018;34(9):756–60. https://doi.org/10.1080/09513590.2018.1445219.
Frantz N, Ferreira M, Kulmann MI, Frantz G, Bos-Mikich A, Oliveira R. Platelet-rich plasma as an effective alternative approach for improving endometrial receptivity - a clinical retrospective study. JBRA Assist Reprod. 2020;24(4):442–6. https://doi.org/10.5935/1518-0557.20200026.
Sfakianoudis K, Simopoulou M, Nitsos N, Rapani A, Pappas A, Pantou A, et al. Autologous platelet-rich plasma treatment enables pregnancy for a woman in premature menopause. J Clin Med. 2018;8(1). https://doi.org/10.3390/jcm8010001.
Herraiz S, Romeu M, Buigues A, Martínez S, Díaz-García C, Gómez-Seguí I, et al. Autologous stem cell ovarian transplantation to increase reproductive potential in patients who are poor responders. Fertil Steril. 2018;110(3):496-505.e1. https://doi.org/10.1016/j.fertnstert.2018.04.025.
Ozcan P, Takmaz T, Tok OE, Islek S, Yigit EN, Ficicioglu C. The protective effect of platelet-rich plasma administrated on ovarian function in female rats with Cy-induced ovarian damage. J Assist Reprod Genet. 2020;37(4):865–73. https://doi.org/10.1007/s10815-020-01689-7.
Peterson JE, Zurakowski D, Italiano JE Jr, Michel LV, Fox L, Klement GL, et al. Normal ranges of angiogenesis regulatory proteins in human platelets. Am J Hematol. 2010;85(7):487–93. https://doi.org/10.1002/ajh.21732.
Dawood AS, Salem HA. Current clinical applications of platelet-rich plasma in various gynecological disorders: an appraisal of theory and practice. Clin Exp Reprod Med. 2018;45(2):67–74. https://doi.org/10.5653/cerm.2018.45.2.67.
Sfakianoudis K, Simopoulou M, Grigoriadis S, Pantou A, Tsioulou P, Maziotis E, et al. Reactivating ovarian function through autologous platelet-rich plasma intraovarian infusion: pilot data on premature ovarian insufficiency, perimenopausal, menopausal, and poor responder women. J Clin Med. 2020;9(6). https://doi.org/10.3390/jcm9061809.
Sfakianoudis K, Simopoulou M, Nitsos N, Rapani A, Pantou A, Vaxevanoglou T, et al. A case series on platelet-rich plasma revolutionary management of poor responder patients. Gynecol Obstet Invest. 2019;84(1):99–106. https://doi.org/10.1159/000491697.
Farimani M, Heshmati S, Poorolajal J, Bahmanzadeh M. A report on three live births in women with poor ovarian response following intra-ovarian injection of platelet-rich plasma (PRP). Molecular biology reports. 2019;46(2):1611–6. https://doi.org/10.1007/s11033-019-04609-w.
Stojkovska S, Dimitrov G, Stamenkovska N, Hadzi-Lega M, Petanovski Z. Live birth rates in poor responders’ group after previous treatment with autologous platelet-rich plasma and low dose ovarian stimulation compared with poor responders used only low dose ovarian stimulation before in vitro fertilization. Open Access Maced J Med Sci. 2019;7(19):3184–8. https://doi.org/10.3889/oamjms.2019.825.
Fraison E, Crawford G, Casper G, Harris V, Ledger W. Pregnancy following diagnosis of premature ovarian insufficiency: a systematic review. Reprod Biomed Online. 2019;39(3):467–76. https://doi.org/10.1016/j.rbmo.2019.04.019.
Acknowledgements
The authors thank Dr. Hossein Falahzadeh, Professor of Biostatistics, for the statistical assistance. We acknowledge the Hormone laboratory group and operating room personnel at the Yazd Reproductive Sciences Institute for their great contribution to the entire laboratory work and data gathering.
Code Availability
The Ethics Code and IRCT Code are available.
Funding
The research leading to these results received funding from Yazd Reproductive Sciences Institute, Shahid Sadoughi University of Medical Sciences, Yazd, Iran, under Grant Agreement No. 68876.
Author information
Authors and Affiliations
Contributions
Abbas Aflatoonian and Marzieh Lotfi conceived and designed the study. Abbas Aflatoonian and Lida Saeed performed the experiments. Marzieh Lotfi and Nasim Tabibnejad gathered and analyzed the data, wrote the draft, and revised the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. This study was approved by the Ethics Committee of Yazd Reproductive Sciences Institute, Shahid Sadoughi University of Medical Sciences, Yazd, Iran (IR.SSU.RSI.REC.1397.004).
Consent to Participate
A written informed consent for participating in this trial was obtained from all patients.
Consent for Publication
Patients signed informed consent regarding publishing their data.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Aflatoonian, A., Lotfi, M., Saeed, L. et al. Effects of Intraovarian Injection of Autologous Platelet-Rich Plasma on Ovarian Rejuvenation in Poor Responders and Women with Primary Ovarian Insufficiency. Reprod. Sci. 28, 2050–2059 (2021). https://doi.org/10.1007/s43032-021-00483-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43032-021-00483-9