Abstract
A wide range of techniques and methods are actively invented by clinicians and scientists who are dedicated to the field of musculoskeletal tissue regeneration. Biological, chemical, and physiological factors, which play key roles in musculoskeletal tissue development, have been extensively explored. However, physical stimulation is increasingly showing extreme importance in the processes of osteogenic and chondrogenic differentiation, proliferation and maturation through defined dose parameters including mode, frequency, magnitude, and duration of stimuli. Studies have shown manipulation of physical microenvironment is an indispensable strategy for the repair and regeneration of bone and cartilage, and biophysical cues could profoundly promote their regeneration. In this article, we review recent literature on utilization of physical stimulation, such as mechanical forces (cyclic strain, fluid shear stress, etc.), electrical and magnetic fields, ultrasound, shock waves, and substrate stimuli, to promote the repair and regeneration of bone and cartilage tissue. Emphasis is placed on the mechanism of cellular response and the potential clinical usage of these stimulations for bone and cartilage regeneration.
Lay Summary
Bone and cartilage regenerative engineering aims to create stable, bioactive, and native tissue-like scaffolds which can repair bone and cartilage damages. These scaffolds are often combined with chondrogenic/osteogenic cells or stem cells to create replacement tissue grafts with enhanced regenerative capability. In this approach, physical stimulations such as ultrasound, mechanical force, electrical charge, and magnetic field have significant impacts on cell fate and behavior through regulating various intracellular signaling pathways. The review provides a comprehensive understanding and broad overview of literature on effects of different physical stimulations on cellular behaviors and signaling pathways, which have been reported to induce growth of bone and cartilage. The knowledge lay a strong foundation for the development of future “smart” tissue grafts that can effectively repair bone and cartilage under physical stimulations. Other future works will focus on combining different physical stimulations and fine-tuning parameters of such stimulations to obtain optimal cartilage and bone regeneration.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Classical tissue regenerative engineering is an interdisciplinary field of advanced material science, cell biology, and developmental biology, with the aim of promoting the regeneration of complex tissues and organs [1]. In this process, natural or synthetic scaffolds, cells, and growth factors combine to form a construct, structurally, functionally, and mechanically similar to the native tissue that requires repair [2]. It is well known that bone disorders such as osteoporosis, bone fractures, and cartilage disease, like osteoarthritis, commonly occur due to abnormal physiology or physical injury. Several techniques and strategies have emerged to promote their regeneration. For example, guided bone regeneration (GBR) has been widely utilized as a simple therapeutic technique for effective bone reconstruction [3,4,5]. Autologous chondrocyte implantation (ACI) and human mesenchymal stem cell (hMSC)-based treatments are promising strategies for cartilage regeneration. However, for bony defects and cartilage degeneration, reconstructing tissues with sufficient mechanical strength and native tissue-like function is one of the major challenges. In bone regeneration, a common obstacle is fibrous connective tissue rapidly occupying the bony defect rather than normal bone formation (osteogenesis) occurring. The resulting fibrous connective tissue buildup, with its low mechanical strength and cartilage-like structure, creates defective bone. In cell-based cartilage regenerative therapies, both ACI and hMSC-based treatments have shown critical drawbacks for clinical use. Dedifferentiated chondrocytes form fibrocartilage instead of hyaline cartilage after transplantation in ACI and endochondral ossification following hypertrophic differentiation of hMSCs frequently occurs in hMSC-based osteoarthritis treatments [6,7,8,9,10,11,12].
To address these issues, people have extensively researched biochemical stimuli including platelet-rich plasma, novel biomaterial scaffolds, and various growth factors; however, most attention was put into the chemical and biological behaviors [13,14,15,16,17]. Since bone and cartilage are exposed to multiple internal and external physical forces, biomechanical environment plays an important role in maintaining, repairing, and remodeling their respective tissues to meet functional demands and maintain the tissue homeostasis. In fact, the physical properties of the cell micro-environment are equally important as the biochemical properties. For example, it was shown that altering the stiffness of the extracellular matrix (ECM) could direct stem cell differentiation, with increasing stiffness directing differentiation toward more mechanically competent tissues, such as cartilage and bone, and away from the more delicate adipose and neuronal tissues [18].
Physical stimuli (cyclic strain, electricity, electromagnetism, ultrasound, shock wave, and laser) have already shown active roles in bone and cartilage regeneration in vitro and in vivo [19,20,21,22,23]. In cell-based musculoskeletal tissue engineering, these physical stimuli (Fig. 1) have been found to induce hMSC proliferation, modulate their behaviors, and support their differentiation by modulating their intracellular signaling pathways. This suggests that the use of such stimuli can be a promising strategy to improve bone fracture healing and cartilage regeneration. To date, some physical manipulations have already been introduced into clinical applications for bone and cartilage regeneration. The objective of this review is to identify the main physical stimulation methods that have been utilized in bone and cartilage repair and elucidate possible mechanisms of cellular response.
Physical Stimulation for Bone Regeneration and Fracture Healing
Mechanical Forces
It is well known that both extrinsic and intrinsic mechanical forces can induce tissue resistance and adaptation. The induced tissue forces are transmitted to the micromechanical environment of resident cells and thus influence the intracellular forces. Cells can subsequently modify their micromechanical environments via cytoskeletal rearrangement or molecular cascade transduction activation. This ultimately alters synthesis or degradation of the extracellular matrix and feeds back to alter cellular sensitivity to incoming mechanical forces [24]. Studies have demonstrated that appropriate mechanical forces are important for bone cell localization, orientation, metabolism, and homeostasis [25]. The most common mechanical forces that benefit this are cyclic strain and fluid shear stress.
Cyclic Strain
Cycles of loading and unloading cause the compression and relaxation of the ECM, which induce strain on the cells of bone or cartilage. Cyclic strain includes repeated tensile strain as well as cyclic compressive strain. Cartilage and bone are constantly exposed to cyclic strain when an individual body is moving in daily life. Tensile strain is clinically used for bone engineering in distraction osteogenesis (a surgical procedure used to repair bone by creating a fracture between two bone segments, then moving the segments slowly apart from each other). The magnitude of tensile strain is important in bone development and in the fate determination of MSCs. For example, the magnitude of tensile strain was reportedly related to inhibition of adipogenesis (a process balancing osteogenesis and chondrogenesis) and the introduction of interruptions (rests) in strain application showed no significant effect [26]. The equi-biaxial cyclic tensile strain significantly reduced adipogenesis in mouse adipose-derived mesenchymal stem cells (ASCs) [27]. Studies have shown cyclic strain could increase bone-to-adipose ratio via Wnt pathways and upregulate the expression of palladin (an actin-associated protein), to promote the osteogenesis. Stretch-activated cation channels may also contribute to osteogenesis [28]. Forces acting on cells may change protein conformation and thus expose the binding sites in a functionally relevant way [29]. Elements of the cytoskeleton bridging actin fibers (including lamin proteins) to the nuclear membrane also show an important role in osteogenesis during this mechanical stimulation [29].
In addition, the piezoelectric properties of bone make it generate electricity in response to mechanical stress. The amplitude of the electrical potential generated in stressed bone is determined by the rate and magnitude of the applied load and the resulting bone deformation. The electrical polarity is dependent upon the directions of loading and bending. Normally, when bone is bent, the concave sides (under compression) become negatively charged and the convex sides (under tensile) become positively charged, which make the bone grow more on the compressive side and degrade more on the stretched side [30]. In this case, the mechanical stress is also capable of stimulating bone regeneration through electrical-induced pathways, the mechanism of which is detailed in the following electrical stimulation section.
Fluid Shear Stress
The circulatory system (e.g., flow of blood) also produces pulsatile or oscillating shear stress on musculoskeletal cells. The shear stress induced by fluid flow plays a significant role in bone development, especially in the osteogenic differentiation process of stem cells. Studies have shown the application of both continuous flow and pulsating fluid flow (PFF) to increase osteogenic differentiation of ASCs as compared to static cultures. The greatest osteogenic induction was seen with PFF. Tjabringa et al. state that 3 h after PFF application, gene expression of Runx2 was increased, while that of osteopontin (OPN) did not change, suggesting PFF may affect the early stages, but not the late stages of osteogenic differentiation. This is because Runx2 expression is an indicator of early osteogenesis and OPN expression is an indicator of late osteogenesis. The enhancement of osteogenesis from fluid flow may relate to the distribution of nutrient and growth factors in the cell. Fröhlich et al. found enhanced expression of bone-specific markers in perfusion cultures with uniform distribution, as compared with static culture only present at the outer regions in static culture [31]. Thus, the improved osteogenesis from fluid flow may be attributed to the better distribution of nutrients and growth factors.
The flow stimulation of ASC osteogenesis may be explained by an indirect mechanism via polyamines, the enzyme Cox-2, and nitric oxide (NO). Studies have shown PFF to increase the gene expression of spermidine/spermine-N(1)-acetyltransferase (SSAT), an enzyme associated with polyamine activity [32]. Higher intracellular calcium activity may also be involved in shear-induced osteogenesis through PKC and ERK 1/2 pathways, downstream of NO production, in PFF-activated ASC osteogenesis [33]. Fluid shear stress can also upregulate the expression of integrin α5β1, which has been identified as an important factor in promoting osteogenesis through ERK 1/2 activation [33]. ERK activation is proven to be important in determining osteoblast survival, proliferation, and differentiation [34].
The Molecular Mechanism of Mechanical Force Transduction
Once the cell has detected a local mechanical stimulus, the signal needs to be converted into a biochemical response. For the general pattern of mechanical force sensing machinery in the musculoskeletal system, the ECM-integrin-cytoskeletal signaling axis has gained the most attention (Fig. 2). Transmembrane receptors called integrins connect the ECM to intracellular cytoskeleton elements consisting of actin filaments, non-muscle myosin, and associated proteins [35]. The cytoskeleton achieves structural cohesion by creating a dynamic balance between the counteracting forces of compression and tension [36]. The force-induced conformational changes of the cytoskeleton directly alter chromatin structure and thus modulate gene transcriptional activity. This occurs via direct connections of cytoskeletal elements to DNA [37] or by activating integrin-mediated intracellular pathways that involve focal adhesion kinases (FAKs) or Src tyrosine kinases [38]. Neighboring cells that are attached to the affected cell via cadherin-containing adhesion complexes could be mechanically transferred, accordingly inducing molecular changes [39].
Possible pathways involved in the biological response to mechanical stress, ES, US, and shock wave stimulations on bone cells. Several pathways of MAPK/ERK, Wnt/β-catenin, PI3K/Akt, TGF-β/BMP, NF-κB, PKA, PKC, and Ca2+ signaling could be regulated in response to biophysical stimulations, to enhance the cell proliferation and differentiation and to modulate the inflammatory response by modulating the expressing of bone markers Rux2, BMP2/4, OCM, Osx, etc. or other related regulators. GSK-3β glycogen synthase kinase-3 beta; TRK tyrosine kinase receptor; TCF/LEF T cell factor/lymphoid enhancer factor; PI3K phosphatidylinositide 3-kinases; TGF-β transforming growth factor beta; BMP bone morphogenetic proteins; AKT protein kinase B; mTOR mechanistic target of rapamycin; NF-κB nuclear factor kappa-light-chain-enhancer of activated B cells; PGE2 prostaglandin E2; AC adenylyl cyclase; cAMP cyclic adenosine monophosphate; PKA protein kinase A; CREB cAMP response element-binding protein; PKC protein kinase C; MAPK mitogen-activated protein kinase; ERK extracellular signal-regulated kinases; FAK focal adhesion kinase; GPCR G protein-coupled receptor; OCN osteocalcin; Osx osterix; ES electrical stimulation; US ultrasound; TRK tyrosine kinase
On the other hand, integrin-mediated transmission of membrane strain induces activation of Akt, resulting in downstream activation of both β-catenin and Ras homolog gene family member A (RhoA). This increases cell stiffness, which results in repression of adipogenic genes [38]. As an effect of force, calcium influx is frequently regulated by voltage-sensitive calcium channels (VSCC). These channels are partially anchored in the cell membrane and, thus, are capable of attaching to the ECM and responding to mechanical stimulation in osteoblasts [40]. In vitro inhibition of T type VSCC significantly reduces the expression of both early and late mechanoresponsive genes in osteoblasts [41]. However, the mechanism of mechanical force transduction is complex and not clear, warranting further investigation.
Electrical and Electromagnetic Stimulation
Electrical Stimulation
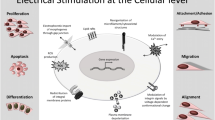
Physiological electric fields (EFs) serve as an efficient tool to control and adjust the cellular and tissue homeostasis. The human body generates a biological EF ranging between 10 and 60 mV at various locations [42]. Bioelectricity is very important in the wound healing process. When a wound is created, a steady direct current (DC) EF is initiated. This endogenous EF guides cell migration toward the wound edge. On the contrary, wound healing is compromised when the EF is inhibited [43]. In 1953, Yasuda et al. applied continuous electrical current to a rabbit femur for 3 weeks and demonstrated new bone formation around the cathode [44]. Since then, use of EFs for bone healing applications has been widely researched [45]. Capacitive coupling electric field (CCEF) and inductive coupling electromagnetic field (EMF) are also being used more frequently in recent years. Both DC and alternating current (AC) have been observed to enhance osteogenesis when cells at the cathode are stimulated with a current of 5–100 μA [46].
Electrical potentials have been proven to play an important role in bone cell proliferation, migration, and remodeling both in vitro and in vivo [47, 48]. Some implant materials, such as electrically active ceramics, including polarized hydroxyapatite (HA), and piezoelectric ceramics, which produce an electrical potential under mechanical loading, have been found to induce bone ingrowth and improve bone formation around implants respectively. The mechanism by which electrical activity influences biological responses is likely to result from preferential adsorption of proteins and ions onto the charged surface. Numerous studies have emphasized the importance of surface charge species on cell behavior at the biomaterial interface [47, 49, 50]. In calvarial bones of rats, after implanting electrically polarized HA plates, improved bone ingrowth and enhanced osteoblast activity were observed, with complete bone penetration into polarized implants occurring as early as 3 weeks [47]. In this study, the bone formation increase that occurred on the negatively charged surfaces (N-surfaces) of the polarized implants was likely due to accumulating Ca2+ ions on the surfaces. Molecules such as fibronectin, osteocalcin, and bone morphogenetic proteins (BMPs), on the other hand, adhere to the positively charged surfaces (P-surfaces) to improve osteoblast migration [47]. Nakamura et al. also observed the surface charge of polarized HA influencing protein adsorption onto the HA surface and thus enhancing the osteoconductivity of HA. Fibrin was presented as a key protein in the early stages of osteoconduction. Its adsorption was accelerated on both N-surfaces and P-surfaces through ionic and pH changes via attracting calcium ions and –COOH groups of fibrin respectively. Specifically, –COOH groups of fibrin were attracted to the P-surfaces, while calcium ions were attracted to the N-surfaces. The resulting positively charged ion layer further encouraged fibrin adsorption (Fig. 3). Subsequently, a network scaffold is formed by absorbed fibrin, platelets, and osseous cells. After adhesion to the fibrin on the P-surface occurs through integrin α2bβ3, the activated platelets further release a variety of growth factors that stimulate the osseous cells. The coagulation components played an important role in the early stages of osteoconduction [49]. In addition, hyaluronan, an extracellular matrix component, also plays a key role in the cellular interactions with charged surfaces. The negatively charged surface of osteoblasts that hyaluronan induced has been shown to mediate initial contact between cell and metal surfaces [51].
The exact mechanism underlying the intracellular signal transduction of ES in bone repair is still unclear. Several hypotheses are shown in Fig. 2. (1) External EFs could alter the ion flux via cell membrane proteins (such as ion channels, transporters, pumps, and enzymes) and subsequently lead to an ion concentration (such as Ca2+, Na+, Cl−, and K+) change, which may cause a depolarization of excitable cells and trigger the related cellular signaling [52]. For example, electrical stimulation (ES) could activate the phosphatidylinositol-3-kinase (PI3K) and mammalian target of rapamycin (mTOR) pathways, which lead to the transcription of transforming growth factor-β (TGF-β) family factors such as BMP-4. (2) Applied current could change the cell gap junctions, which affect the exchange of certain signaling molecules such as calcium, cyclic nucleotides, and inositol phosphates [53]. Much evidence indicates that gap junction communication is necessary for the development and maintenance of a differentiated osteoblast phenotype, including the production of alkaline phosphatase, osteocalcin (OCN), bone sialoprotein, and collagen [54]. (3) EFs may also affect ligand-receptor binding by changing the conformation or expression of receptors. For example, EFs could increase the expression of adenosine A2A receptors (A2ARs) or integrin-β molecules, both of which influence their related intracellular pathways with roles in anti-inflammatory and differentiation processes [55]. (4) EFs may also stimulate higher metabolic activity, which could induce intracellular ATP depletion and thus alter the membrane characteristics such as endo- and exocytosis, adhesion, and motility [56]. (5) EFs could change ECM compositions by affecting the ECM components including soluble ions and charged groups in glycosaminoglycans (GAGs) and proteins [57].
Electromagnetic Stimulation
Pulsed electromagnetic fields (PEMFs), which are generated from an unsteady current being passed through a coil, have been approved by the FDA to treat nonunions of bone fractures and related problems since 1979 [58]. Under PEMF stimulation, osteoblasts were found to exhibit increased osteogenesis caused by elevated expression of TGF-β1 [59] and BMP-2/4 [60] and reinforced intracellular calcium transients [61]. In an ovariectomized rat model, PEMFs were found to prevent ovariectomy-induced bone loss through activation of the Wnt/ β-catenin signaling pathway [62]. In an identical model, long-term PEMF stimulation treatment alleviated lumbar vertebral osteoporosis by increasing bone formation and suppressing bone resorption through regulation of the Wnt3a/LRP5/β-catenin and OPG/RANKL/RANK signaling pathways [63]. Ehnert et al. identified a specific extremely low-frequency pulsed electromagnetic field (ELF-PEMF) (10 to 90.6 Hz) that supports human osteoblast function in an ERK1/2-dependent manner. The ELF-PEMF by producing non-toxic amounts of reactive oxygen species (ROS) induced anti-oxidative defense mechanisms in these cells [64, 65].
In bone tissue engineering, PEMFs were found to modulate the cell cycle of MSCs of different origins and enhance their differentiation and proliferation. This could be seen by their enhanced production of ECM and growth/differentiation factors including TGF-β and BMPs [23, 66]. A wide range of electromagnetic stimulation frequencies (between 2 and 123 Hz) have been shown to be effective in improving osteogenic stimulation of ASCs [67], characterized by increased intracellular calcium and Alizarin Red S staining after 14 days induction [68]. The stimulation increased alkaline phosphatase activity and cytoskeleton tension. It also induced higher expression of ALP, OPN, collagen type I (Col I), and Runx2 after 21 days induction [67]. PEMFs were also used as an adjuvant element in many studies, along with osteoinductive medium. However, which PEMF parameters (dose, frequency, and intensity) enable the most optimal repair in a clinical setting is still an unanswered question.
Similar to the mechanism of mechanical stress, that of PEMFs on bone regeneration is more complicated than initially expected [69]. PEMFs may play roles through (1) changing the physical and chemical properties of a cell membrane by altering the ion flux and membrane potentials [70, 71]; (2) affecting the assembly and arrangement of the actin cytoskeleton; (3) modulating the intracellular Wnt/β-catenin and TGF-β/BMP signaling pathways, leading to upregulated expression of key cytokines such as TGF-β1 and BMP 2/4 [59, 60, 62]; and (4) regulating the oxidative state of cell [72, 73]. However, the precise cellular mechanism is still unclear. More mechanistic investigations are needed.
Ultrasound
Ultrasound (US) usually refers to a longitudinal wave propagation, a special type of sonic wave with a frequency greater than 20 kHz (this is the upper limit of human audibility), that causes local oscillation of particles. Ultrasound with a frequency around 3~10 MHz is widely used in clinical settings for diagnostic and therapeutic purposes. It is also one of the well-established therapeutic physical stimuli for bone healing. Since ultrasound was first reported to stimulate bone healing in 1950 [74], numerous efforts have been spent over the past several decades to prove its therapeutic effects in animal models [75, 76]. In particular, low-intensity pulsed ultrasound stimulation (LIPUS), using intensities less than 50 mW/cm2, was reported to improve ECM synthesis, accelerate bone healing, and reactivate failed healing processes [75, 77]. The use of ultrasound in improving bone regeneration was recently approved by the FDA for human application. Ultrasound-based non-viral gene delivery was also recently found to induce bone formation in vivo [78]. In in vitro cell studies, ultrasound was found to enhance the expression of osteoblast maturation markers, such as OCN, bone sialoprotein (BSP), and Ca2+ [79,80,81,82,83,84]. With treatment of LIPUS, the expression of chemokines such as monocyte-chemoattractant protein (MCP)-1, macrophage-inflammatory protein (MIP)-1, and receptor-activator of nuclear factor kappa-Β ligands (RANKL) is enhanced, and mechanoreceptor angiotensin II type I receptor (ATI) is activated on the surfaces of osteoblasts [85]. Under LIPUS exposure, the production of NO and prostaglandin E2 (PGE2) was upregulated. The former is a free radical gas involved in the regulation of vascular endothelial growth factor (VEGF) expression and is important to bone formation [86]; the latter is an arachidonic acid-derived metabolite, associated with bone formation and resorption. In differentiating murine osteoblasts, LIPUS has been found to enhance RANKL gene expression 10-fold compared to unstimulated controls after 3 weeks of LIPUS treatment [85]. It was also reported that LIPUS combined with growth factors such as calcium-regulating hormones, 1,25-dihydroxyvitamin D3 (1,25-(OH)2D3) [87], BMP2 [88], or BMP7 [89] can stimulate bone repair.
LIPUS-induced bone healing can be influenced by the processes of (1) inflammation, (2) soft callus formation, (3) angiogenesis, (4) early osteogenesis, (5) bone formation, and (6) bone remodeling [90]. There are several theories to illustrate these mechanisms (Fig. 2). In the first theory, oscillatory displacement of the cell membrane caused by the ultrasound wave triggers oscillatory displacement between intracellular elements of different densities [91]. The very low strains induced by the ultrasound on cells in vitro have been reported to induce a prompt fluidization of the cytoskeleton together with an acceleration of cytoskeletal remodeling events [92]. The second theory is the bilayer sonophore model. In this model, ultrasound application periodically pulls the two lipid layers apart and back, leading to intramembranous hydrophobic spaces expanding and contracting accordingly [93]. In the third theory, integrins play a key role in converting LIPUS signals into biochemical responses [94]. The type of integrins including α2, α5, β1, and β3 integrins varies in response to different cell origins [81, 95, 96]. In the fourth theory, ultrasound induces intracellular stress and strain which are maximized within the cell at two distinct resonant frequencies. Stimulated load-inducible gene expression, therefore, is maximized when the excitation frequency matches the cell’s resonant frequency [97]. The fifth theory is related to P2Y receptor activation. P2Y receptors are G protein-coupled receptors (GPCRs) that are activated by adenine and uridine nucleotides and nucleotide sugars. Studies have shown LIPUS treatment to induce osteoblastogenesis by releasing purines, such as ATP, and activating P2Y receptors [98]. The sixth theory involves calcium signaling regulation. Ca2+ signals are oscillatory and these signals (also generated via the RhoA GTPase pathway) are crucial for bone marrow-derived mesenchymal stem cell (BMSC) differentiation [99]. Finally, the last theory is about the connexin-mediated gap junction. Studies have shown that gap junctions are essential for LIPUS’s effect on osteogenic differentiation of MSCs [84, 100].
In addition, ultrasound can modulate the micro-environment by heating, cavitation, acoustic streaming, or triggering delivery of growth factors to engineered cells [101]. The physical effects of LIPUS that induce biological responses can be divided into thermal and non-thermal categories. Temperature increases can regulate thermo-sensitive enzymes like metalloproteinase, which are important for bone matrix remodeling. Non-thermal effects include oscillatory strains induced by ultrasonic waves (which can directly affect the mechanosensitive elements at very high frequencies), acoustic radiation forces (resulting in a low-frequency cyclic mechanical stimulus), strain gradients, and fluid flow (such as acoustic streaming and micro-streaming). Radiation force, fluid flow, and strain gradients can create shear stresses on cell membranes. Acoustic streaming and micro-streaming can play important roles in vitro. The former results in nutrient redistribution via improved circulation of molecules in the culture medium or via increased fluid flow in vivo [102]; the latter is generated in response to oscillating gas bubbles or other small acoustic inhomogeneities and causes circulatory movement of fluid [101].
The involved pathways in US treatment are complex. In murine MC3T3-E1 pre-osteoblasts, COX-2 expression, which is important for PGE2 production, was regulated via FAK and mitogen-activated protein kinases (MAPKs), Erk1/2, PI3K, and Akt kinase signaling in response to ultrasound treatment [95]. The expression of iNOS (in charge of the NO production) was found to be induced by ultrasound through the canonical NF-κB pathway which is preceded by activation of Ras, Raf-1, MEK, Erk, and IKKα/β kinases [103]. LIPUS induced p38 MAPKs and Erk1/2 MAPKs which were found to be crucial in the process of osteogenic differentiation in human periodontal ligament cells (HPDLCs) and the murine pluripotent mesenchymal cell line C2C12 [84, 104]. However, more investigations are needed to illustrate the involved signaling transduction cascade under ultrasound exposure.
Shock Wave
A shock wave is a kind of short-duration, acoustic pressure wave consisting of two phases, the positive phase evoking compressive stress (peak pressure: 30–100 MPa) and the negative phase arousing tensile and shear stress (negative pressure). These waves can be produced by various generators such as electro-hydraulic, electromagnetic, piezoelectric, or pneumatic generators [105]. After propagating into tissue, shock waves may lead to micro-bubbles of liquid molecules and cavitation effects on the focal area. Shock waves were introduced to increase cell membrane permeability and facilitate the delivery of macromolecules into cells [106]. Use of shock waves, referred to as extracorporeal shock wave therapy (ESWT), is normally looked as “extracorporeal” and “non-invasive” stimulation mainly focused on the treated area. It is known that ESWT is able to relieve pain, reduce inflammation, induce neo-angiogenesis, and stimulate stem cell activities, thus improving tissue regeneration and healing. ESWT has been applied in the musculoskeletal field as orthotripsy and regenerative medicine to promote bone remodeling [107,108,109], restore the healing process in cases of non-unions [110], loosen the bone cement during revision arthroplasty [111], and enhance bone callus formation during bone lengthening [112]. Shock waves also can promote osteoblast growth and differentiation as well as their expression of TGF-β1 in a dose-dependent manner [113]. In osteoarthritis (OA) treatment, ESWT was used to regulate subchondral bone remodeling and improve trabecular microarchitecture. Compared to the non-treatment OA group, the ESWT-treated group showed an increased osteocyte count and a higher percentage of subchondral trabecular bone [114]. Increased proliferation and migratory capacity were also shown in human BMSCs when exposed to shock waves [115]. ASCs exposed to ESWT have shown enhanced production of osteogenic markers such as RUNX2, ALP, and mineralized matrix. However, the production of reactive oxygen species (ROS) was also increased [116]. ESWT could also affect the growth ratio of bone marrow osteoprogenitor cells to bone nodules, which is related to the induction of TGF-β1 molecules [117].
The acoustic shock wave induces tissue to absorb, reflect, refract, and propagate the mechanical pulsed energy. The mechanisms of shock waves’ effects on bone healing are possibly related to the micro-fractures and cavitation they induce [118]. The micro-fractures and cavitation may trigger the initiation of remodeling cycles and neovascularization [108, 119]. Thus, they regulate the growth and maturation of osteoprogenitor cells, membrane polarization, expression of BMPs, and activation of the so-called mechanotransduction pathways that are related to acoustic stimulations [107, 120,121,122,123]. During the mechanotransduction process, mechanosensory components in cell membranes such as integrins, ion channels, and various sensors and growth factor receptors may be activated by shock wave-induced forces. Several signaling pathways (e.g., MAPK-ERK pathway, P13K-Akt-iNOS pathway) may be involved in the corresponding biological events of cytoskeletal rearrangement and nuclear expression modulation [124]. ESWT can also regulate the sub-membrane reduction-oxidation (redox) reactions elicited by early O2 production for tyrosine kinase-mediated ERK activation, resulting in phosphorylation of CBFA1 (core-binding factor alpha1), the transcription factor for osteoblastic differentiation [125]. However, the overlap of several pathways and interactions between them make it more complicated to illustrate the exact signal transduction. More investigation needs to be done to clarify the precise molecular mechanism before translating it to clinic use.
Substrate Stimulus
The nature of growth surfaces, scaffold or substrate, always plays a significant role in influencing the cell behavior. Good osteoconductivity and osteogenic ability are prerequisites for scaffolds used in bone engineering to promote the new bone formation. The idea is that scaffolds should provide a good environment to guarantee secure attachment, survival, and distribution of osteogenic cells grown into or surrounding them. The substrate stimulus could directly affect the structural changes of bone or cartilage cells through integrins, focal adhesions, or the actin cytoskeleton. Indirect mechanisms via G proteins or ion channels are also possible. In scaffold-induced osteogenesis, increased phosphorylation by FAKs at tyrosine 397 was observed [126]. Stimulation by scaffold ions may drive osteogenesis of ASCs through an indirect mechanism in which signals are transduced through receptors, ion channels, or G proteins to the nucleus where the expression of related genes was regulated. For example, the calcium ions could enter the cell via calcium receptors which interact with G proteins. Calcium ions have been shown to stimulate proliferation of osteoblasts and magnesium ions have been associated with increased mineralization [127]. McCullen et al. also showed that ionic calcium enhanced mineralization in human ASCs [128]. Additionally, nanoscale topographical features in growth substrates influence stem cell behavior [29]. Elasticity also has a plausible effect on the osteogenesis of ASCs [18, 26, 68, 129].
The area of cell adhesion onto the matrix substrate has been also shown to regulate cell behavior. For example, stem cells forced to attach on large fibronectin islands show an elongated morphology, different from the more rounded shape that occurs when attached on smaller islands. The enhanced osteogenic commitment was due to increased RhoA and Rho-associated protein kinase (ROCK) activity [130].
Other Physical Factors
Besides the aforementioned physical stimulations on bone regeneration, some other physical methods have also been introduced into bone regeneration and healing. Laser periodontal therapy (LPT) is a laser-based procedure developed as an effective debridement technique to treat periodontitis [131]. Low-level laser therapy (LLLT) with proper doses and output powers was also reported to stimulate cellular metabolism, increase protein synthesis, and subsequently enhance bone regeneration [132]. Temperature could rise in a surgical operation like osteotomies and the elevated temperature could disrupt the bone healing [133]. Heat generation during osteotomy is one of the important factors influencing the development of osseointegration [134]. One recent study showed ultraviolet (UV)/O3 irradiation for ≥ 5 min significantly decontaminating H3PO4-modified hydroxyapatite surfaces and improving their wettability, thus facilitated osteoblast growth and function [135].
Physical Stimulation for Cartilage Regeneration
Mechanical Forces
Mechanical load is an important regulator of chondrocyte metabolism and is required for maintaining normal cartilage matrix properties. The natural environment of articular chondrocytes in the body is a dynamic mechanical one involving various biomechanical forces including compression, tensile stretching, shear stress, hydrostatic pressure, and osmotic stress [136]. These forces have varying origins. The direct contact between joint surfaces can produce both static and dynamic compression. Tensile loading can result from physical activities such as gymnastics. The synovial fluid in articular cavities generates shear stress. Charged proteoglycan (PG) in the cartilage matrix makes hydrostatic pressure in chondrocytes. Lastly, osmotic stress is the result of the influx and efflux of fluid within the cartilage matrix during joint loading [136, 137]. Overall, both catabolic and anabolic factors contribute to ECM synthesis and remodeling in response to mechanical stimulation [138, 139].
Extra mechanical stimuli can also influence the metabolism and gene expression patterns of normal and osteoarthritic chondrocytes. For example, chondrocytes from OA cartilage can benefit from optimized compressive stimulation by enhancing the biosynthetic activity reflected by greater ECM production. One in vitro study has shown short-term compressive stimulation can significantly induce aggrecan (ACAN), COL2A1, COL1A1, proteoglycan 4 (PRG4), and COL10A1 gene expression in a zone-dependent manner, while long-term compression can increase collagen type II, ACAN immunostaining, and total GAG content [140]. Dynamic compression upregulates the gene expression of ACAN and type II collagen, while static compression downregulates it in chondrocytes [141]. It is known that mechanical stimulation not only changes the biosynthesis of load-bearing ECM molecules (e.g., aggrecan and collagen type II) but also regulates articular cartilage lubrication molecules (e.g., PRG4, lubricin, superficial zone protein (SZP)) [142, 143]. A high shear strain can induce extensive rearrangements of the focal adhesions and the actin cytoskeleton of the chondrocytes [144]. Cyclic hydrostatic pressure was reported to stimulate the chondrogenic differentiation of BMSCs in pellet cultures [145]. Under intermittent hydrostatic pressure, inhibited matrix metalloproteinase and pro-inflammatory mediator release was observed in human osteoarthritic chondrocytes in vitro [146].
Mechanical stimulation can also be exerted on MSC-seeded constructs for cartilage tissue engineering applications. For example, dynamic compression combined with exogenous SOX-9 promotes chondrogenesis of ACSs in a poly(lactic-co-glycolic acid) (PLGA) scaffold [147]. Compressive loading upregulated the expression levels of chondrogenic genes in MSC-seeded porous hyaluronan-gelatin constructs [148]. Cyclic tensile loading enhanced PG synthesis rates in MSC-seeded collagen-PG scaffolds [149]. A combination of shear and dynamic compression leads to chondrogenesis of human MSCs [150]. Some research has shown chondrogenesis of human BMSCs in fibrin-polyurethane composites could be modulated by frequency and amplitude of dynamic compression and shear stress [151]. However, the responses of MSCs to mechanical stimulation sometime are different from those of chondrocytes. Huang et al. demonstrated that 21 days of compressive loading significantly reduces the compressive mechanical properties and biochemical contents of bovine MSC-seeded agarose constructs [152]. Thorpe et al. recently showed that both the compressive modulus and PG content of porcine MSC-seeded agarose constructs were significantly reduced in a long-term, dynamic mechanical compression application, compared to free-swelling controls [153]. It is important to highlight that mechanical loading could synergistically improve the composition and mechanical properties of neocartilage when integrated with growth factors, such as TGF-β and insulin-like growth factor-1 (IGF-1) [154].
The molecular mechanisms of mechanical signal transduction in chondrocytes are complex and not fully understood. The physical properties of chondrocytes are related to this mechanical force transduction. For example, the pericellular matrix (PCM) of chondrocytes was proven to transfer forces between the cell cytoskeleton and its ECM. The viscoelasticity of chondrocytes, which is determined by the integrity and organization of their actin filaments and intermediate filaments, is also very important in cartilage’s response to the mechanical force. Mismatches in viscoelastic properties could result in further cartilage degradation due to the disparities between neocartilage and adjacent tissues. The actin and intermediate filaments bear the cytoskeletal tension and the microtubules serve as struts to resist compression. Chondrocytes could respond to mechanical stimuli by remodeling their actin cytoskeleton, which is linked to the ECM through focal adhesions. These focal adhesions transfer the signal of external physical forces into the appropriate biochemical events in a Rho kinase-dependent manner [155]. Specifically, the Rho GTPases activate ROCKs, which phosphorylate and activate Lim kinases, which in turn phosphorylate and inhibit the actin-depolymerizing protein, cofilin. Studies have shown the actin cytoskeleton was changed by dynamic compression acting on agarose-embedded chondrocytes. Rho kinase activity is required for this actin reorganization and the change in gene expression that occurs [155]. Chondrocyte nuclei and their nucleoskeletons also play a vital role in mechanotransduction. A 15% compressive strain greatly decreases the height and volume of the chondrocyte and its nucleus. The actin cytoskeleton plays an important role in this deformation behavior [156]. The signal transduction mechanisms of these force-induced biochemical responses are still obscure. There are several pathways involved as shown in Fig. 4.
Possible pathways are regulated in response to extra biophysical stimulations on chondrocytes. Biophysical stimulations could act through one or a combination of MAPK/ERK, PKA/CREB, Wnt/β-catenin, PI3K/Akt, TGF-β/BMP, NF-κB, PKA, PKC, and Ca2+ signaling pathways to enhance cell proliferation, survival, and differentiation and to modulate the inflammatory response by regulating the expressing of cartilage markers Sox9, TGF-β1, collagen type II, ACAN, etc
Integrin Signaling
Fibronectin and integrin receptors provide the vital connection between the ECM and the cytoskeleton [157] involved in cell adhesion, ECM remodeling, and chondrocyte metabolism [158]. Studies have shown that human articular chondrocytes use α5β1 integrins as mechanoreceptors. Stimulation of this integrin modulates ion channels, the actin cytoskeleton, focal adhesion, paxillin, and β-catenins. As a result, IL-4 is secreted in an autocrine manner via type II receptors, to induce membrane hyperpolarization, increase levels of aggrecan, and decrease matrix metalloproteinase 3 expression. However, healthy chondrocytes show a different response than osteoarthritic cartilage [159].
Purinergic Signaling
Extracellular purines, such as ATP, adenosine, and pyrimidine, which act as extracellular signaling molecules, activate related purinergic receptors [160]. ATP is involved in the mechanotransduction signaling cascade in chondrocytes by paracrine, upregulate matrix production, and decrease MMP 3 activity after mechanical stimulation. The purinergic pathway that ATP induces activates intracellular Ca2+ signaling [161, 162].
Calcium Signaling
The calcium signaling includes the intracellular PLC-inositol 1,4,5-trisphosphate pathway, stretch-activated ion channels, and the transient receptor potential vanilloid 4 (TRPV4) pathway. Under mechanical press, an influx of Ca2+ could induce a wave of Ca2+ uptake through mechanosensitive ion channels from the extracellular medium. The Ca2+ concentration transiently increasing in the intracellular site may be one of the earliest events in the response of chondrocytes to mechanical stimulation [163]. TRPV4 not only regulates SOX9 expression, but it also mediates the response to osmotic stress, especially for hypoosmotic stress [164, 165]. Through Src kinases, Ca2+ also regulates the integrin-mediated signaling pathway, converged on ERK-MAPK by a single application of cyclic compression (1 kPa, 1 Hz, 30 min). Mechanical stimuli including compression, fluid flow, hydrostatic pressure, and osmotic stress can influence Ca2+ signaling in chondrocytes [162, 166,167,168,169,170,171].
MAPK/ERK Pathway
Several studies have shown downstream activation of MAPK pathways when chondrocytes were subjected to mechanical stimulation. MAPK signaling was found to be force-dependent in intact cartilage. Mechanical compression activated ERK1/2, JNK, and p38 pathways by stimulating phosphorylation in distinct temporal patterns [172]. Shear- and compression-induced chondrocyte transcription requires MAPK activation in cartilage explants [173]. One microarray study has shown that hyperosmotic stress leads to regulation of a wide variety of genes, which involves transduction through p38 MAPK and ERK1/2 pathways [174].
TGF-β Signaling
TGF-β signaling has been shown to be involved in the responses of chondrocytes and MSCs to mechanical stimuli. For example, dynamic compression transiently activates Smad2/3 in chondrocyte-agarose constructs [175] and bovine bone marrow stromal constructs during chondrogenesis [176]. Mechanical load promotes chondrogenesis of hMSCs through the TGF-β pathway by upregulating TGF-β gene expression and protein synthesis [177].
Electrical Stimulation
The electrochemical properties of articular cartilage occur from the electrically charged nature of the tissue. Electric potentials develop in cartilage by the flow of charged particles across negatively charged PG in and out of the ECM [178, 179]. Applying an external electric potential or current to cartilage can produce stress and deformation in the tissue [179]. It has been hypothesized that the electric fields associated with the dynamic loading of cartilage may affect its growth, remodeling, and biosynthesis [180]. Two modes of electrical stimulation are commonly utilized: (1) direct current (DC) and (2) capacitive coupling (CC). DC (5 mA) was reported to stimulate the differentiation of MSCs into chondrocytes and enhance the proliferation of differentiated chondrocytes [181]. Chondrocytes exhibiting cathodal migration when subjected to applied DC electric fields have also been reported [182, 183]. As early as 1978, the effect of a capacitively coupled electric field (CCEF) on chondrocyte DNA synthesis via Na+ and Ca2+ fluxes was reported [184]. Selective capacitively coupled electrical signals could upregulate the gene expression of cartilage matrix proteins (e.g., ACAN and type II collagen), in which the duration, response time, amplitude, duty cycle, and frequency could affect matrix production [185,186,187]. Even in the presence of interleukin (IL)-1β, a defined capacitively induced electrical signal could result in significant upregulation of cartilage matrix proteins while significantly attenuating the upregulation of MMPs in full-thickness osteoarthritic adult human articular cartilage explants [187].
However, how the electrical signals transform and influence cell behavior is still unclear. There are several pathways that are potentially involved: (1) adenosine receptors—evidence has shown that adenosine receptors have been implicated in the electrotransduction process for cartilage [188]. Stimulation of both the high-affinity A2a and low-affinity A2b adenosine receptors resulted in elevated cyclic AMP and subsequent activation of anti-inflammatory pathways via protein kinase A (PKA) and exchange protein-activated directly by cyclic AMP (EPAC). This, in turn, leads to the suppression of NO and PGE2 and downstream feedback inhibition of tumor necrosis factor (TNF)-α and IL-1β [189]. (2) Calcium signaling—studies have shown CCEF (60 kHz, 20 mV/cm) to increase the expression of cartilage matrix protein genes and suppress the expression of the MMP gene in bovine articular chondrocytes. This occurs by an influx of Ca2+ through voltage-gated calcium channels rather than from intracellular Ca2+ repositories and has downstream consequences on calmodulin, calcineurin, and nuclear factor of activated T cells (NF-AT) rather than on phospholipase C and IP(3) [190]. The transduction pathway of pulsed CCEF stimulates human chondrocyte proliferation, which involves calcium, calmodulin, cGMP, and nitric oxide synthase [191].
Magnetic Stimulation
There are two modes of magnetic stimulation: (1) static magnetic fields (SMFs), which are generated from the material properties of a permanent magnet, and (2) PEMF. It is well established that cellular responses to magnetic stimulation depend on the intensity, frequency, type of field (static or oscillatory), waveform (sinusoidal, square, etc.), cell status (precursor or differentiated), and type of cell exposed [192, 193]. In in vitro human chondrocyte cultures, 0.6 T SMFs significantly increase chondrocyte proliferation and viability [194]. Additionally, 0.4 T SMFs were also reported to promote PG synthesis of primary bovine and human articular chondrocytes and potentiate the chondrogenic differentiation of human bone marrow stromal cells. The differentiation was synergistically augmented in the presence of TGF-β3 [195]. However, there have been relatively few studies on the effect of SMFs on chondrocytes, compared to using PEMFs. PEMFs have been clinically investigated in OA patients with encouraging results [196, 197]. In in vitro studies, PEMFs have been found to cause a number of physiological effects in both monolayer chondrocyte cultures and tissue explant models. These benefits include enhanced proliferation [198,199,200], anabolic activities, PG synthesis [201,202,203,204], and anti-inflammatory responses [188, 205]. After surgical implantation of neocartilage in vivo, PEMFs prevent the catabolic effects of inflammation by upregulation of A2A receptors. The combination of EMFs and IGFs has been reported in vivo to provide a more chondroprotective effect than any single treatment alone [202]. In stem cell-based tissue engineering for chondrogenesis, studies both in vitro and in vivo have shown that EMF exposure may promote chondrogenic differentiation [206, 207], increase ECM component synthesis, and control the inflammatory events, all while downregulating the expression of inflammatory mediators such as IL-1 and MMPs [22, 208,209,210].
As for the mechanisms of magnetic stimulation, studies have shown that MFs (e.g., PEMFs) alter ion channels, ligand-binding sites, and receptor density and distribution in the cell membrane, consequently affecting transmembrane signaling [211]. To date, there is no identified magnetoreceptor or even a magnetotransduction theory to clarify the cellular responses to MF stimulation. Lorentz force that is produced by the movement of charged ions in an MF may play a key role in the biological behaviors that MFs induce. Several intracellular molecular signaling pathways are involved in the stimulation: (1) calcium signaling—two studies have shown the involvement of the Ca2+ signaling pathway in chondrogenesis under the stimulation of MFs. One study has shown strong MFs (3 T) significantly increase the intracellular Ca2+ concentration after 6 h of exposure and cause deleterious effects on human chondrocytes [212]. Another study has shown moderate strength (0.4 T) SMFs induce chondrogenic differentiation of human BMSCs and increases intracellular Ca2+ within 20 s of exposure [195]. (2) TGF-β signaling—the enhancement of chondrogenesis by a PEMF is associated with an increase in TGF-β1 synthesis, mediated by binding of AP-1, which may be modulated by phosphorylation of JNK. Moderate-strength magnetic fields (0.4 T) can induce chondrogenesis of BMSCs by upregulating the expression of the cartilage maker gene SOX9, type II collagen, and aggrecan through a TGF-β-dependent pathway. SMFs alone cause TGF-β secretion in BMSC chondrogenic culture and these effects of SMFs could be abrogated by the TGF-β receptor blocker SB-431542 [195]. (3) adenosine A2AR-mediated pathway—PEMFs have been shown to reduce the concentration of TNFα and IL-1β via the upregulation of A2AR and regulation of the NF-κB pathway [189, 213]. (4) MAPK signaling—Hsieh et al. found that strong (3 T) MFs upregulate the phosphorylation of ERK1 and ERK2 after 8- to 96-h exposure times [212].
Ultrasound
LIPUS is a non-invasive, efficient, and cost-effective method of improving the healing of osteochondral defects [214] and is a good supporter of cartilage regeneration [215]. These effects of US are showed by enhancing the viability of cells, matrix protein synthesis, and matrix integrity with no need for exogenous TGF-β. Both in vitro and in vivo studies have reported that LIPUS induces the expression of type II collagen and PG in chondrocyte cells and cartilage tissue [216,217,218,219,220,221]. Choi et al. have shown that LIPUS of 200~300 mW/cm2 increases the expression of type II collagen by 50% and PG by 30% in a 3D alginate culture of human articular chondrocyte [222]. In the same culture, LIPUS treatment could reduce the matrix degradation by inhibiting the catabolic gene expression (e.g., MMP1) [215]. In the early stages of papain-induced arthritis in rats and full-thickness osteochondral defects in rabbits, LIPUS was shown to promote the repair process of arthritic cartilage [221]. In a rabbit OA model, LIPUS was reported to significantly reduce the severity of OA-induced structural damage in the cartilage and synovium when combined with hyaluronate treatment [223].
Furthermore, LIPUS has been tested to efficiently induce the chondrogenic differentiation of MSCs both in vitro and in vivo. For example, LIPUS treatment of 200 mW/cm2 has been found to promote the chondrogenic marker expressions of COL2A1, ACAN, and Sox-9 in the early stages of rabbit MSC chondrogenesis [224]. The chondrogenesis of human MSCs seeded in a 3D scaffold was also enhanced when exposed to LIPUS [225]. In in vivo studies, LIPUS treatments greatly enhanced the chondrogenic differentiation of MSCs, reflected by increased total collagen and GAG content, compared to the control [226, 227]. LIPUS has also been reported to enhance the chondrogenesis process even without exogenous TGF-β, which is a well-known inducer of chondrogenesis [215]. The combined use of LIPUS and growth factor, TGF-β3, has been reported to improve the chondrogenic differentiation of a pellet culture of hMSCs [228]. In monolayer cultures, the synergistic use of ultrasound and TGF-β1 was observed to greatly enhance the expression of Sox9, aggrecan, and COL2A1 expression [229]. In addition, LIPUS was found to accelerate the proliferation of chondrocytes (harvested from rats and pigs) in monolayer cultures [230, 231].
Understanding of the effect of ultrasound on the biological behaviors of chondrocytes is still elusive. Many theories have been postulated: (1) the calcium pathway—even at low intensities, ultrasounds could increase the intracellular concentration of Ca2+ and that blocking this rise with intracellular calcium chelating agents, or inhibiting Ca2+/ATPase, abolishes the stimulatory effect of ultrasound on PG synthesis [21]. (2) The MAPK/ERK pathway [97, 232]—the phosphorylation of FAK, Src, p130Cas, CrkII, and Erk1/2 may be increased in primary human chondrocytes under continuous LIPUS stimulation [232]. (3) The integrin/PI3K/Akt pathway—this pathway was found to be linked with the increased proliferation rate of primary pig articular chondrocytes in response to LIPUS [230].
Shock Wave
An increasing number of studies have demonstrated the therapeutic effects of ESWT on the treatment or management of OA. ESWT has been shown to possess chondroprotective effects and cartilage repair roles [114, 233,234,235], and to reduce the concentration of interleukin-10 and TNF-α in OA chondrocytes [236]. ESWT has also been shown to inhibit the expression of MMP1 and MMP3 in a rabbit OA model [237]. Furthermore, in this model, the chondroprotective effects of ESWT show time-dependence [235]. The application of 600 impulse shockwaves at 1.5 × 105 Pa each on the knee joint of OA rabbit models decreased the production of NO and rate of chondrocyte apoptosis [238]. However, excessive bouts of ESWT treatments or overdose of ESWT treatments may cause negative effects on OA cartilage repair, instead of inducing the favorable promotion [106, 239]. Studies have shown smaller denudations greater enhance chondrocyte formation and density for osteochondritis dissecans of rabbit knees when they are exposed to ESWT [240]. In addition, ESWT has also been reported to attenuate pain in human OA, enhancing the functional ability and decreasing the Western Ontario and McMaster University Osteoarthritis Index (WOMAC) pain score in the ESWT treatment group [241, 242]. ESWT has also been shown to enhance the expression of crucial factors for chondrogenesis such as TGF-β, IGF, and fibroblast growth factors (FGFs) [243].
Although ESWT has shown positive effects of chondroprotection, subchondral bone remodeling, pain reduction, and motor function improvements, the exact mechanism remains unclear. The reduced expression of catabolic genes and inflammatory factors such as IL-1, IL-10, and TNF-α may partly contribute to the benefits of ESWT on OA patient. Greater investigation is required.
Other Physical Factors
Han et al. [171] found that Ca2+ signaling in chondrocytes occurs more quickly and with greater magnitude when the temperature is increased. This fact suggests that Ca2+ signaling of chondrocytes is also regulated by other, cellular, physical environments, such as ECM topography and temperature. Periodic heat shock at 41 °C for 1 h was found to significantly accelerate the chondrogenic differentiation of human mesenchymal stem cells in a pellet culture [244]. However, thermal stimulations of chondrogenesis have not been well explored. Studies also indicate that low-energy light could directly stimulate chondrogenesis. LLLT using 660 and 780 nm wavelengths benefits the regeneration of OA by accelerating the initial breakdown of cartilage, destroyed by collagenase, and stimulating fibroblasts to synthesize the repairing collagen III [245]. UV irradiation is also normally used on engineered cartilage. A number of studies have shown the substrate’s physical properties and its topological structures to be critical for the chondrocyte’s differentiation, redifferentiation, and maturation [246]. A suitable substrate for seeding cells may help cartilage regeneration through mechanical, biological, and chemical effects in cartilage regeneration [247].
Conclusion
This review has elaborated on the application of physical stimulation, including electrical and electromagnetic field, mechanical force (tensile strain and fluid flow), ultrasound, shock wave, and others on bone and cartilage regeneration. A better understanding of how these factors act on the cells of bone and cartilage will allow better prediction and control in bone and cartilage tissue engineering approaches. In the review, the efficacy and mechanism of these biophysical stimulations were displayed. Pathway analyses among diverse classes of stimuli were described. The combination of different stimuli can result in synergistic therapeutic effects for the treatment of bone and cartilage diseases. Many pathways were postulated to explain the cell response to physical stimulation. Some pathways were complementary and overlapped, making it challenging to decipher the precise contribution and timing of activation of individual pathways. Yet, it is clear that the physical factors play very critical roles in the biological processes. The applications of tensile strain, shear stress, electromagnetic fields, and ultrasound are among many options to enhance osteogenesis and chondrogenesis of human stem cell. Therefore, direct physical intervention is an appealing approach and should be profoundly exploited to improve clinical outcomes in bone and cartilage regeneration.
However, when treating bone and cartilage disorders by physical intervention, both the positive and negative effects should be taken into consideration. There still exist the following major challenges in clinical use.
-
(1)
The specific forms of physical stimulation and their dose effects and application timings must be carefully determined and validated.
-
(2)
Advanced techniques and devices must be addressed in achieving focalized stimulus delivery with adjustable signal type and intensity. Emphasis should especially be placed on how to monitor the healing process. The development of effective and reliable treatment protocols is a prerequisite.
-
(3)
Efficient in vivo assessing technologies must also be obtained. Although the positive effects of some physical factors have been identified in vitro, the exact value of these factors in native bone and cartilage tissues has not been determined. Well-designed animal experiments need to be conducted as physical stimulation signal and dose effects on regeneration and repair of tissue should be firmly established and quantified.
-
(4)
An optimal strategy for bone and cartilage engineering may be to incorporate different physical stimuli with growth factors and biomaterials. How do the physical factors influence each other and what are the underlying mechanisms should be further investigated.
-
(5)
For cartilage repair, the low friction properties, collagen organization and crosslinking, and the related test standards should gain more emphasis.
Compared to bone, with its multiple cell types, vascularity, and high capability for innate repair, regeneration of cartilage faces more challenges [11]. Although the biochemical components of neocartilage (e.g., PG and collagen) can be controlled and modified, to date, no tissue-engineered neocartilage has been able to simultaneously match native cartilage’s compressive, friction, and tensile properties under large deformations and motions [11, 193]. Joint forces over a large range of motion can take a devastating toll on neocartilage [11]. Therefore, the synthetic cartilage should have sufficient mechanical strength and compliance to sustain various forces and act as a cushion, respectively. In addition, the biomechanical response of native cartilage to loading varies widely and is time-dependent. Thus, a big challenge is defining, evaluating, and determining the specific mechanical properties, required for the replacement cartilages at specific implantation sites. Even if proper compressive properties match those of native tissues, the properties may not maintain when transplanted in vivo. Another challenge is how to properly integrate replacement tissues with the adjacent cartilage to provide stable, biologic fixation, load distribution, and proper mechanotransduction [248].
To some extent, within bone and cartilage cells, the signals from diverse stimuli overlap and the cells show a coordinated response. The combination of multiple stimuli may be better than any single stimulation to facilitate bone and cartilage regeneration. For example, enhanced differentiation of pre-osteoblasts was observed when simultaneously stimulating cells with cyclic strain and ultrasound [249]. To date, a combinatory and continuous application of strain, fluid shear, or electromagnetic fields with soluble small molecules is increasingly popular to supplement standard in vitro methods of inducing osteogenesis and chondrogenesis. Altogether, the success in repairing and regenerating musculoskeletal tissues depends on creating a musculoskeletal tissue with optimized mechanical, biological, and chemical characteristics. This requires interdisciplinary approaches and collaborative work between biologists, physicists, medical doctors, and engineers to develop novel, effective regenerative therapies.
Abbreviations
- GBR:
-
guided bone regeneration
- ACI:
-
autologous chondrocyte implantation
- hMSCs:
-
human mesenchymal stem cells
- ECM:
-
extracellular matrix
- ASCs:
-
mouse adipose-derived mesenchymal stem cells
- PFF:
-
pulsating fluid flow
- OPN:
-
osteopontin
- SSAT:
-
spermidine/spermine-N(1)-acetyltransferase
- FAK:
-
focal adhesion kinase
- RhoA:
-
Ras homolog gene family member A
- VSCC:
-
voltage-sensitive calcium channels
- ES:
-
electrical stimulation
- EF:
-
electric field
- DC:
-
direct current
- CCEF:
-
capacitive coupling electric field
- EMF:
-
electromagnetic field
- AC:
-
alternating current
- PI3K:
-
phosphatidylinositol-3-kinase
- mTOR:
-
mammalian target of rapamycin
- TGF-β:
-
transforming growth factor-β
- A2AR:
-
adenosine A2A receptors
- GAGs:
-
glycosaminoglycans
- PEMFs:
-
pulsed electromagnetic fields
- ELF-PEMF:
-
extremely low-frequency pulsed electromagnetic field
- ROS:
-
reactive oxygen species
- Col I:
-
collagen type I
- GSK-3β:
-
glycogen synthase kinase-3 beta
- TRK:
-
tyrosine kinase receptor
- TCF/LEF:
-
T cell factor/lymphoid enhancer factor
- PI3K:
-
phosphatidylinositide 3-kinases
- TGF-β:
-
transforming growth factor beta
- BMP:
-
bone morphogenetic proteins
- AKT:
-
protein kinase B
- mTOR:
-
mechanistic target of rapamycin
- NF-κB:
-
nuclear factor kappa-light-chain-enhancer of activated B cells
- PGE2:
-
prostaglandin E2
- AC:
-
adenylyl cyclase
- cAMP:
-
cyclic adenosine monophosphate
- PKA:
-
protein kinase A
- CREB:
-
cAMP response element-binding protein
- PKC:
-
protein kinase C
- MAPK:
-
mitogen-activated protein kinase
- ERK:
-
extracellular signal-regulated kinases
- FAK:
-
focal adhesion kinase
- GPCR:
-
G protein-coupled receptor
- OCN:
-
osteocalcin
- Osx:
-
osterix
- US:
-
ultrasound
- LIPUS:
-
low-intensity pulsed ultrasound
- BSP:
-
bone sialoprotein
- MCP:
-
monocyte-chemoattractant protein
- MIP:
-
macrophage-inflammatory protein
- RANKL:
-
receptor activator of nuclear factor kappa-Β ligand
- ATI:
-
angiotensin II type I receptor
- NO:
-
nitric oxide
- PGE2:
-
prostaglandin E2
- VEGF:
-
vascular endothelial growth factor
- GPCRs:
-
G protein-coupled receptors
- BMSCs:
-
bone marrow-derived mesenchymal stem cells
- ESWT:
-
extracorporeal shock wave therapy
- CBFA1:
-
core-binding factor alpha1
- ROCK:
-
RhoA and Rho-associated protein kinase
- PG:
-
proteoglycan
- ACAN:
-
aggrecan
- PRG4:
-
proteoglycan 4
- SZP:
-
superficial zone protein
- PCM:
-
pericellular matrix
- TRPV4:
-
transient receptor potential vanilloid 4
- CC:
-
capacitive coupling
- EPAC:
-
exchange proteins activated directly by cyclic AMP
- TNF-α:
-
tumor necrosis factor-α
- NF-AT:
-
nuclear factor of activated T cells
- SMFs:
-
static magnetic fields
- WOMAC:
-
Western Ontario and McMaster University Osteoarthritis Index
- LLLT:
-
low-level laser therapy
References
Laurencin CT, Khan Y. Regenerative engineering. Sci Transl Med. 2012;4(160):160ed9. https://doi.org/10.1126/scitranslmed.3004467.
Lo KW, Jiang T, Gagnon KA, Nelson C, Laurencin CT. Small-molecule based musculoskeletal regenerative engineering. Trends Biotechnol. 2014;32(2):74–81. https://doi.org/10.1016/j.tibtech.2013.12.002.
Ueyama Y, Ishikawa K, Mano T, Koyama T, Nagatsuka H, Suzuki K, et al. Usefulness as guided bone regeneration membrane of the alginate membrane. Biomaterials. 2002;23(9):2027–33.
Davey AK, Maher PJ. Surgical adhesions: a timely update, a great challenge for the future. J Minim Invasive Gynecol. 2007;14(1):15–22. https://doi.org/10.1016/j.jmig.2006.07.013.
Kikuchi M, Koyama Y, Yamada T, Imamura Y, Okada T, Shirahama N, et al. Development of guided bone regeneration membrane composed of beta-tricalcium phosphate and poly (L-lactide-co-glycolide-co-epsilon-caprolactone) composites. Biomaterials. 2004;25(28):5979–86. https://doi.org/10.1016/j.biomaterials.2004.02.001.
Cournil-Henrionnet C, Huselstein C, Wang Y, Galois L, Mainard D, Decot V, et al. Phenotypic analysis of cell surface markers and gene expression of human mesenchymal stem cells and chondrocytes during monolayer expansion. Biorheology. 2008;45(3–4):513–26.
Hubka KM, Dahlin RL, Meretoja VV, Kasper FK, Mikos AG. Enhancing chondrogenic phenotype for cartilage tissue engineering: monoculture and coculture of articular chondrocytes and mesenchymal stem cells. Tissue Eng B Rev. 2014;20(6):641–54. https://doi.org/10.1089/ten.TEB.2014.0034.
Peterson L, Minas T, Brittberg M, Nilsson A, Sjogren-Jansson E, Lindahl A. Two- to 9-year outcome after autologous chondrocyte transplantation of the knee. Clin Orthop Relat Res. 2000;374:212–34.
Roberts S, McCall IW, Darby AJ, Menage J, Evans H, Harrison PE, et al. Autologous chondrocyte implantation for cartilage repair: monitoring its success by magnetic resonance imaging and histology. Arthritis Res Ther. 2003;5(1):R60–73.
Freyria AM, Mallein-Gerin F. Chondrocytes or adult stem cells for cartilage repair: the indisputable role of growth factors. Injury. 2012;43(3):259–65. https://doi.org/10.1016/j.injury.2011.05.035.
Huey DJ, Hu JC, Athanasiou KA. Unlike bone, cartilage regeneration remains elusive. Science. 2012;338(6109):917–21. https://doi.org/10.1126/science.1222454.
Puetzer JL, Petitte JN, Loboa EG. Comparative review of growth factors for induction of three-dimensional in vitro chondrogenesis in human mesenchymal stem cells isolated from bone marrow and adipose tissue. Tissue Eng B Rev. 2010;16(4):435–44. https://doi.org/10.1089/ten.TEB.2009.0705.
Park YJ, Kim KH, Lee JY, Ku Y, Lee SJ, Min BM, et al. Immobilization of bone morphogenetic protein-2 on a nanofibrous chitosan membrane for enhanced guided bone regeneration. Biotechnol Appl Biochem. 2006;43(Pt 1):17–24. https://doi.org/10.1042/BA20050075.
Camargo PM, Lekovic V, Weinlaender M, Vasilic N, Madzarevic M, Kenney EB. Platelet-rich plasma and bovine porous bone mineral combined with guided tissue regeneration in the treatment of intrabony defects in humans. J Periodontal Res. 2002;37(4):300–6.
Lee SJ, Park YJ, Park SN, Lee YM, Seol YJ, Ku Y, et al. Molded porous poly (L-lactide) membranes for guided bone regeneration with enhanced effects by controlled growth factor release. J Biomed Mater Res. 2001;55(3):295–303.
Cacciafesta V, Dalstra M, Bosch C, Melsen B, Andreassen TT. Growth hormone treatment promotes guided bone regeneration in rat calvarial defects. Eur J Orthod. 2001;23(6):733–40.
Damien E, Hing K, Saeed S, Revell PA. A preliminary study on the enhancement of the osteointegration of a novel synthetic hydroxyapatite scaffold in vivo. J Biomed Mater Res A. 2003;66((2):241–6. https://doi.org/10.1002/jbm.a.10564.
Engler AJ, Sen S, Sweeney HL, Discher DE. Matrix elasticity directs stem cell lineage specification. Cell. 2006;126(4):677–89. https://doi.org/10.1016/j.cell.2006.06.044.
Kohavi D, Pollack SR, Brighton C. Short-term effect of guided bone regeneration and electrical stimulation on bone growth in a surgically modelled resorbed dog mandibular ridge. Biomater Artif Cells Immobilization Biotechnol. 1992;20(1):131–8.
Bonassar LJ, Grodzinsky AJ, Frank EH, Davila SG, Bhaktav NR, Trippel SB. The effect of dynamic compression on the response of articular cartilage to insulin-like growth factor-I. J Orthop Res. 2001;19(1):11–7. https://doi.org/10.1016/S0736-0266(00)00004-8.
Parvizi J, Parpura V, Greenleaf JF, Bolander ME. Calcium signaling is required for ultrasound-stimulated aggrecan synthesis by rat chondrocytes. J Orthop Res. 2002;20(1):51–7. https://doi.org/10.1016/S0736-0266(01)00069-9.
Ciombor DM, Lester G, Aaron RK, Neame P, Caterson B. Low frequency EMF regulates chondrocyte differentiation and expression of matrix proteins. J Orthop Res. 2002;20(1):40–50. https://doi.org/10.1016/S0736-0266(01)00071-7.
Chao EY, Inoue N. Biophysical stimulation of bone fracture repair, regeneration and remodelling. Eur Cell Mater. 2003;6:72–84. discussion −5
Thompson WR, Scott A, Loghmani MT, Ward SR, Warden SJ. Understanding mechanobiology: physical therapists as a force in mechanotherapy and musculoskeletal regenerative rehabilitation. Phys Ther. 2016;96(4):560–9. https://doi.org/10.2522/ptj.20150224.
Bodle JC, Hanson AD, Loboa EG. Adipose-derived stem cells in functional bone tissue engineering: lessons from bone mechanobiology. Tissue Eng Part B Rev. 2011;17(3):195–211. https://doi.org/10.1089/ten.TEB.2010.0738.
Hanson AD, Marvel SW, Bernacki SH, Banes AJ, van Aalst J, Loboa EG. Osteogenic effects of rest inserted and continuous cyclic tensile strain on hASC lines with disparate osteodifferentiation capabilities. Ann Biomed Eng. 2009;37(5):955–65. https://doi.org/10.1007/s10439-009-9648-7.
Huang SC, Wu TC, Yu HC, Chen MR, Liu CM, Chiang WS, et al. Mechanical strain modulates age-related changes in the proliferation and differentiation of mouse adipose-derived stromal cells. BMC Cell Biol. 2010;11:18. https://doi.org/10.1186/1471-2121-11-18.
Kearney EM, Farrell E, Prendergast PJ, Campbell VA. Tensile strain as a regulator of mesenchymal stem cell osteogenesis. Ann Biomed Eng. 2010;38(5):1767–79. https://doi.org/10.1007/s10439-010-9979-4.
Teo BK, Ankam S, Chan LY, Yim EK. Nanotopography/mechanical induction of stem-cell differentiation. Methods Cell Biol. 2010;98:241–94. https://doi.org/10.1016/S0091-679X(10)98011-4.
Isaacson BM, Bloebaum RD. Bone bioelectricity: what have we learned in the past 160 years? J Biomed Mater Res A. 2010;95((4):1270–9. https://doi.org/10.1002/jbm.a.32905.
Frohlich M, Grayson WL, Marolt D, Gimble JM, Kregar-Velikonja N, Vunjak-Novakovic G. Bone grafts engineered from human adipose-derived stem cells in perfusion bioreactor culture. Tissue Eng Part A. 2010;16(1):179–89. https://doi.org/10.1089/ten.TEA.2009.0164.
Tjabringa GS, Vezeridis PS, Zandieh-Doulabi B, Helder MN, Wuisman PI, Klein-Nulend J. Polyamines modulate nitric oxide production and COX-2 gene expression in response to mechanical loading in human adipose tissue-derived mesenchymal stem cells. Stem Cells. 2006;24(10):2262–9. https://doi.org/10.1634/stemcells.2005-0625.
Liu L, Yuan W, Wang J. Mechanisms for osteogenic differentiation of human mesenchymal stem cells induced by fluid shear stress. Biomech Model Mechanobiol. 2010;9(6):659–70. https://doi.org/10.1007/s10237-010-0206-x.
Jessop HL, Rawlinson SC, Pitsillides AA, Lanyon LE. Mechanical strain and fluid movement both activate extracellular regulated kinase (ERK) in osteoblast-like cells but via different signaling pathways. Bone. 2002;31(1):186–94.
Hynes RO. Integrins: a family of cell surface receptors. Cell. 1987;48(4):549–54.
Ingber DE. Tensegrity: the architectural basis of cellular mechanotransduction. Annu Rev Physiol. 1997;59:575–99. https://doi.org/10.1146/annurev.physiol.59.1.575.
Wang N, Tytell JD, Ingber DE. Mechanotransduction at a distance: mechanically coupling the extracellular matrix with the nucleus. Nat Rev Mol Cell Biol. 2009;10(1):75–82. https://doi.org/10.1038/nrm2594.
Thompson WR, Guilluy C, Xie Z, Sen B, Brobst KE, Yen SS, et al. Mechanically activated Fyn utilizes mTORC2 to regulate RhoA and adipogenesis in mesenchymal stem cells. Stem Cells. 2013;31(11):2528–37. https://doi.org/10.1002/stem.1476.
Leckband DE, le Duc Q, Wang N, de Rooij J. Mechanotransduction at cadherin-mediated adhesions. Curr Opin Cell Biol. 2011;23(5):523–30. https://doi.org/10.1016/j.ceb.2011.08.003.
Thompson WR, Majid AS, Czymmek KJ, Ruff AL, Garcia J, Duncan RL, et al. Association of the alpha(2)delta(1) subunit with Ca(v)3.2 enhances membrane expression and regulates mechanically induced ATP release in MLO-Y4 osteocytes. J Bone Miner Res. 2011;26(9):2125–39. https://doi.org/10.1002/jbmr.437.
Srinivasan PP, Parajuli A, Price C, Wang L, Duncan RL, Kirn-Safran CB. Inhibition of T-type voltage sensitive calcium channel reduces load-induced OA in mice and suppresses the catabolic effect of bone mechanical stress on chondrocytes. PLoS One. 2015;10(5):e0127290. https://doi.org/10.1371/journal.pone.0127290.
Foulds IS, Barker AT. Human skin battery potentials and their possible role in wound healing. Br J Dermatol. 1983;109(5):515–22.
Song B, Zhao M, Forrester JV, McCaig CD. Electrical cues regulate the orientation and frequency of cell division and the rate of wound healing in vivo. Proc Natl Acad Sci U S A. 2002;99(21):13577–82. https://doi.org/10.1073/pnas.202235299.
Ryaby JT. Clinical effects of electromagnetic and electric fields on fracture healing. Clin Orthop Relat Res 1998(355 Suppl):S205–15.
Manjhi J, Mathur R, Behari J. Effect of low level capacitive-coupled pulsed electric field stimulation on mineral profile of weight-bearing bones in ovariectomized rats. J Biomed Mater Res B Appl Biomater. 2010;92(1):189–95. https://doi.org/10.1002/jbm.b.31505.
Aaron RK, Ciombor DM, Simon BJ. Treatment of nonunions with electric and electromagnetic fields. Clin Orthop Relat Res. 2004;419:21–9.
Itoh S, Nakamura S, Nakamura M, Shinomiya K, Yamashita K. Enhanced bone ingrowth into hydroxyapatite with interconnected pores by electrical polarization. Biomaterials. 2006;27(32):5572–9. https://doi.org/10.1016/j.biomaterials.2006.07.007.
Nakamura M, Nagai A, Tanaka Y, Sekijima Y, Yamashita K. Polarized hydroxyapatite promotes spread and motility of osteoblastic cells. J Biomed Mater Res A. 2010;92(2):783–90. https://doi.org/10.1002/jbm.a.32404.
Nakamura M, Sekijima Y, Nakamura S, Kobayashi T, Niwa K, Yamashita K. Role of blood coagulation components as intermediators of high osteoconductivity of electrically polarized hydroxyapatite. J Biomed Mater Res A. 2006;79(3):627–34. https://doi.org/10.1002/jbm.a.30827.
Nakamura S, Kobayashi T, Nakamura M, Itoh S, Yamashita K. Electrostatic surface charge acceleration of bone ingrowth of porous hydroxyapatite/beta-tricalcium phosphate ceramics. J Biomed Mater Res A. 2010;92(1):267–75. https://doi.org/10.1002/jbm.a.32354.
Finke B, Luethen F, Schroeder K, Mueller PD, Bergemann C, Frant M, et al. The effect of positively charged plasma polymerization on initial osteoblastic focal adhesion on titanium surfaces. Biomaterials. 2007;28(30):4521–34. https://doi.org/10.1016/j.biomaterials.2007.06.028.
Aaron RK, Boyan BD, Ciombor DM, Schwartz Z, Simon BJ. Stimulation of growth factor synthesis by electric and electromagnetic fields. Clin Orthop Relat Res. 2004;419:30–7.
Stains JP, Civitelli R. Gap junctions in skeletal development and function. Biochim Biophys Acta. 2005;1719(1–2):69–81. https://doi.org/10.1016/j.bbamem.2005.10.012.
Stains JP, Civitelli R. Cell-to-cell interactions in bone. Biochem Biophys Res Commun. 2005;328(3):721–7. https://doi.org/10.1016/j.bbrc.2004.11.078.
Ciombor DM, Aaron RK. The role of electrical stimulation in bone repair. Foot Ankle Clin. 2005;10(4):579–93, vii. https://doi.org/10.1016/j.fcl.2005.06.006.
Orr AW, Helmke BP, Blackman BR, Schwartz MA. Mechanisms of mechanotransduction. Dev Cell. 2006;10(1):11–20. https://doi.org/10.1016/j.devcel.2005.12.006.
Kotwal A, Schmidt CE. Electrical stimulation alters protein adsorption and nerve cell interactions with electrically conducting biomaterials. Biomaterials. 2001;22(10):1055–64.
Fioravanti A, Nerucci F, Collodel G, Markoll R, Marcolongo R. Biochemical and morphological study of human articular chondrocytes cultivated in the presence of pulsed signal therapy. Ann Rheum Dis. 2002;61(11):1032–3.
Zhuang H, Wang W, Seldes RM, Tahernia AD, Fan H, Brighton CT. Electrical stimulation induces the level of TGF-beta1 mRNA in osteoblastic cells by a mechanism involving calcium/calmodulin pathway. Biochem Biophys Res Commun. 1997;237(2):225–9. https://doi.org/10.1006/bbrc.1997.7118.
Bodamyali T, Bhatt B, Hughes FJ, Winrow VR, Kanczler JM, Simon B, et al. Pulsed electromagnetic fields simultaneously induce osteogenesis and upregulate transcription of bone morphogenetic proteins 2 and 4 in rat osteoblasts in vitro. Biochem Biophys Res Commun. 1998;250(2):458–61. https://doi.org/10.1006/bbrc.1998.9243.
Tong J, Sun L, Zhu B, Fan Y, Ma X, Yu L, et al. Pulsed electromagnetic fields promote the proliferation and differentiation of osteoblasts by reinforcing intracellular calcium transients. Bioelectromagnetics. 2017;38(7):541–9. https://doi.org/10.1002/bem.22076.
Zhou J, He H, Yang L, Chen S, Guo H, Xia L, et al. Effects of pulsed electromagnetic fields on bone mass and Wnt/beta-catenin signaling pathway in ovariectomized rats. Arch Med Res. 2012;43(4):274–82. https://doi.org/10.1016/j.arcmed.2012.06.002.
Lei T, Liang Z, Li F, Tang C, Xie K, Wang P, et al. Pulsed electromagnetic fields (PEMF) attenuate changes in vertebral bone mass, architecture and strength in ovariectomized mice. Bone. 2018;108:10–9. https://doi.org/10.1016/j.bone.2017.12.008.
Ehnert S, Fentz AK, Schreiner A, Birk J, Wilbrand B, Ziegler P, et al. Extremely low frequency pulsed electromagnetic fields cause antioxidative defense mechanisms in human osteoblasts via induction of *O2(-) and H2O2. Sci Rep. 2017;7(1):14544. https://doi.org/10.1038/s41598-017-14983-9.
Ehnert S, Falldorf K, Fentz AK, Ziegler P, Schroter S, Freude T, et al. Primary human osteoblasts with reduced alkaline phosphatase and matrix mineralization baseline capacity are responsive to extremely low frequency pulsed electromagnetic field exposure—clinical implication possible. Bone Rep. 2015;3:48–56. https://doi.org/10.1016/j.bonr.2015.08.002.
Dimitriou R, Babis GC. Biomaterial osseointegration enhancement with biophysical stimulation. J Musculoskelet Neuronal Interact. 2007;7(3):253–65.
Hammerick KE, James AW, Huang Z, Prinz FB, Longaker MT. Pulsed direct current electric fields enhance osteogenesis in adipose-derived stromal cells. Tissue Eng Part A. 2010;16(3):917–31. https://doi.org/10.1089/ten.TEA.2009.0267.
McCullen SD, McQuilling JP, Grossfeld RM, Lubischer JL, Clarke LI, Loboa EG. Application of low-frequency alternating current electric fields via interdigitated electrodes: effects on cellular viability, cytoplasmic calcium, and osteogenic differentiation of human adipose-derived stem cells. Tissue Eng Part C Methods. 2010;16(6):1377–86. https://doi.org/10.1089/ten.TEC.2009.0751.
Victoria G, Petrisor B, Drew B, Dick D. Bone stimulation for fracture healing: what’s all the fuss? Indian J Orthop. 2009;43(2):117–20. https://doi.org/10.4103/0019-5413.50844.
Panagopoulos DJ, Karabarbounis A, Margaritis LH. Mechanism for action of electromagnetic fields on cells. Biochem Biophys Res Commun. 2002;298(1):95–102.
Ross CL, Siriwardane M, Almeida-Porada G, Porada CD, Brink P, Christ GJ, et al. The effect of low-frequency electromagnetic field on human bone marrow stem/progenitor cell differentiation. Stem Cell Res. 2015;15(1):96–108. https://doi.org/10.1016/j.scr.2015.04.009.
Funk RH, Monsees T, Ozkucur N. Electromagnetic effects—from cell biology to medicine. Prog Histochem Cytochem. 2009;43(4):177–264. https://doi.org/10.1016/j.proghi.2008.07.001.
Cifra M, Fields JZ, Farhadi A. Electromagnetic cellular interactions. Prog Biophys Mol Biol. 2011;105(3):223–46. https://doi.org/10.1016/j.pbiomolbio.2010.07.003.
Buchtala V. Present state of ultrasound therapy. Dia Med. 1950;22(70):2944–50.
Claes L, Willie B. The enhancement of bone regeneration by ultrasound. Prog Biophys Mol Biol. 2007;93(1–3):384–98. https://doi.org/10.1016/j.pbiomolbio.2006.07.021.
Jung YJ, Kim R, Ham HJ, Park SI, Lee MY, Kim J, et al. Focused low-intensity pulsed ultrasound enhances bone regeneration in rat calvarial bone defect through enhancement of cell proliferation. Ultrasound Med Biol. 2015;41(4):999–1007. https://doi.org/10.1016/j.ultrasmedbio.2014.11.008.
Saito M, Fujii K, Tanaka T, Soshi S. Effect of low- and high-intensity pulsed ultrasound on collagen post-translational modifications in MC3T3-E1 osteoblasts. Calcif Tissue Int. 2004;75(5):384–95. https://doi.org/10.1007/s00223-004-0292-9.
Sheyn D, Kimelman-Bleich N, Pelled G, Zilberman Y, Gazit D, Gazit Z. Ultrasound-based nonviral gene delivery induces bone formation in vivo. Gene Ther. 2008;15(4):257–66. https://doi.org/10.1038/sj.gt.3303070.
Angle SR, Sena K, Sumner DR, Virdi AS. Osteogenic differentiation of rat bone marrow stromal cells by various intensities of low-intensity pulsed ultrasound. Ultrasonics. 2011;51(3):281–8. https://doi.org/10.1016/j.ultras.2010.09.004.
Suzuki A, Takayama T, Suzuki N, Sato M, Fukuda T, Ito K. Daily low-intensity pulsed ultrasound-mediated osteogenic differentiation in rat osteoblasts. Acta Biochim Biophys Sin Shanghai. 2009;41(2):108–15.
Yang RS, Lin WL, Chen YZ, Tang CH, Huang TH, Lu BY, et al. Regulation by ultrasound treatment on the integrin expression and differentiation of osteoblasts. Bone. 2005;36(2):276–83. https://doi.org/10.1016/j.bone.2004.10.009.
Unsworth J, Kaneez S, Harris S, Ridgway J, Fenwick S, Chenery D, et al. Pulsed low intensity ultrasound enhances mineralisation in preosteoblast cells. Ultrasound Med Biol. 2007;33(9):1468–74. https://doi.org/10.1016/j.ultrasmedbio.2006.12.003.
Dalla-Bona DA, Tanaka E, Oka H, Yamano E, Kawai N, Miyauchi M, et al. Effects of ultrasound on cementoblast metabolism in vitro. Ultrasound Med Biol. 2006;32(6):943–8. https://doi.org/10.1016/j.ultrasmedbio.2006.01.015.
Ren L, Yang Z, Song J, Wang Z, Deng F, Li W. Involvement of p38 MAPK pathway in low intensity pulsed ultrasound induced osteogenic differentiation of human periodontal ligament cells. Ultrasonics. 2013;53(3):686–90. https://doi.org/10.1016/j.ultras.2012.10.008.
Bandow K, Nishikawa Y, Ohnishi T, Kakimoto K, Soejima K, Iwabuchi S, et al. Low-intensity pulsed ultrasound (LIPUS) induces RANKL, MCP-1, and MIP-1beta expression in osteoblasts through the angiotensin II type 1 receptor. J Cell Physiol. 2007;211(2):392–8. https://doi.org/10.1002/jcp.20944.
Wang FS, Kuo YR, Wang CJ, Yang KD, Chang PR, Huang YT, et al. Nitric oxide mediates ultrasound-induced hypoxia-inducible factor-1alpha activation and vascular endothelial growth factor—a expression in human osteoblasts. Bone. 2004;35(1):114–23. https://doi.org/10.1016/j.bone.2004.02.012.
Ito M, Azuma Y, Ohta T, Komoriya K. Effects of ultrasound and 1,25-dihydroxyvitamin D3 on growth factor secretion in co-cultures of osteoblasts and endothelial cells. Ultrasound Med Biol. 2000;26(1):161–6.
Kim TH, Oh SH, Na SY, Chun SY, Lee JH. Effect of biological/physical stimulation on guided bone regeneration through asymmetrically porous membrane. J Biomed Mater Res A. 2012;100((6):1512–20. https://doi.org/10.1002/jbm.a.34086.
Lee SY, Koh A, Niikura T, Oe K, Koga T, Dogaki Y, et al. Low-intensity pulsed ultrasound enhances BMP-7-induced osteogenic differentiation of human fracture hematoma-derived progenitor cells in vitro. J Orthop Trauma. 2013;27(1):29–33. https://doi.org/10.1097/BOT.0b013e3182519492.
Azuma Y, Ito M, Harada Y, Takagi H, Ohta T, Jingushi S. Low-intensity pulsed ultrasound accelerates rat femoral fracture healing by acting on the various cellular reactions in the fracture callus. J Bone Miner Res. 2001;16(4):671–80. https://doi.org/10.1359/jbmr.2001.16.4.671.
Or M, Kimmel E. Modeling linear vibration of cell nucleus in low intensity ultrasound field. Ultrasound Med Biol. 2009;35(6):1015–25. https://doi.org/10.1016/j.ultrasmedbio.2008.11.011.
Mizrahi N, Zhou EH, Lenormand G, Krishnan R, Weihs D, Butler JP, et al. Low intensity ultrasound perturbs cytoskeleton dynamics. Soft Matter. 2012;8(8):2438–43. https://doi.org/10.1039/C2SM07246G.
Krasovitski B, Frenkel V, Shoham S, Kimmel E. Intramembrane cavitation as a unifying mechanism for ultrasound-induced bioeffects. Proc Natl Acad Sci U S A. 2011;108(8):3258–63. https://doi.org/10.1073/pnas.1015771108.
Pounder NM, Harrison AJ. Low intensity pulsed ultrasound for fracture healing: a review of the clinical evidence and the associated biological mechanism of action. Ultrasonics. 2008;48(4):330–8. https://doi.org/10.1016/j.ultras.2008.02.005.
Tang CH, Yang RS, Huang TH, Lu DY, Chuang WJ, Huang TF, et al. Ultrasound stimulates cyclooxygenase-2 expression and increases bone formation through integrin, focal adhesion kinase, phosphatidylinositol 3-kinase, and Akt pathway in osteoblasts. Mol Pharmacol. 2006;69(6):2047–57. https://doi.org/10.1124/mol.105.022160.
Watabe H, Furuhama T, Tani-Ishii N, Mikuni-Takagaki Y. Mechanotransduction activates alpha(5)beta(1) integrin and PI3K/Akt signaling pathways in mandibular osteoblasts. Exp Cell Res. 2011;317(18):2642–9. https://doi.org/10.1016/j.yexcr.2011.07.015.
Louw TM, Budhiraja G, Viljoen HJ, Subramanian A. Mechanotransduction of ultrasound is frequency dependent below the cavitation threshold. Ultrasound Med Biol. 2013;39(7):1303–19. https://doi.org/10.1016/j.ultrasmedbio.2013.01.015.
Alvarenga EC, Rodrigues R, Caricati-Neto A, Silva-Filho FC, Paredes-Gamero EJ, Ferreira AT. Low-intensity pulsed ultrasound-dependent osteoblast proliferation occurs by via activation of the P2Y receptor: role of the P2Y1 receptor. Bone. 2010;46(2):355–62. https://doi.org/10.1016/j.bone.2009.09.017.
Sun S, Liu Y, Lipsky S, Cho M. Physical manipulation of calcium oscillations facilitates osteodifferentiation of human mesenchymal stem cells. FASEB J. 2007;21(7):1472–80. https://doi.org/10.1096/fj.06-7153com.
Sena K, Angle SR, Kanaji A, Aher C, Karwo DG, Sumner DR, et al. Low-intensity pulsed ultrasound (LIPUS) and cell-to-cell communication in bone marrow stromal cells. Ultrasonics. 2011;51(5):639–44. https://doi.org/10.1016/j.ultras.2011.01.007.
Padilla F, Puts R, Vico L, Raum K. Stimulation of bone repair with ultrasound: a review of the possible mechanic effects. Ultrasonics. 2014;54(5):1125–45. https://doi.org/10.1016/j.ultras.2014.01.004.
Argintar E, Edwards S, Delahay J. Bone morphogenetic proteins in orthopaedic trauma surgery. Injury. 2011;42(8):730–4. https://doi.org/10.1016/j.injury.2010.11.016.
Hou CH, Lin J, Huang SC, Hou SM, Tang CH. Ultrasound stimulates NF-kappaB activation and iNOS expression via the Ras/Raf/MEK/ERK signaling pathway in cultured preosteoblasts. J Cell Physiol. 2009;220(1):196–203. https://doi.org/10.1002/jcp.21751.
Ikeda K, Takayama T, Suzuki N, Shimada K, Otsuka K, Ito K. Effects of low-intensity pulsed ultrasound on the differentiation of C2C12 cells. Life Sci. 2006;79(20):1936–43. https://doi.org/10.1016/j.lfs.2006.06.029.
Mittermayr R, Antonic V, Hartinger J, Kaufmann H, Redl H, Teot L, et al. Extracorporeal shock wave therapy (ESWT) for wound healing: technology, mechanisms. and clinical efficacy Wound Repair Regen. 2012;20(4):456–65. https://doi.org/10.1111/j.1524-475X.2012.00796.x.
Byron CR, Benson BM, Stewart AA, Stewart MC. Effects of radial shock waves on membrane permeability and viability of chondrocytes and structure of articular cartilage in equine cartilage explants. Am J Vet Res. 2005;66(10):1757–63.
Wang CJ, Wang FS, Ko JY, Huang HY, Chen CJ, Sun YC, et al. Extracorporeal shockwave therapy shows regeneration in hip necrosis. Rheumatology (Oxford, England). 2008;47(4):542–6. https://doi.org/10.1093/rheumatology/ken020.
Ma HZ, Zeng BF, Li XL. Upregulation of VEGF in subchondral bone of necrotic femoral heads in rabbits with use of extracorporeal shock waves. Calcif Tissue Int. 2007;81(2):124–31. https://doi.org/10.1007/s00223-007-9046-9.
Mont MA, Jones LC, Seyler TM, Marulanda GA, Saleh KJ, Delanois RE. New treatment approaches for osteonecrosis of the femoral head: an overview. Instr Course Lect. 2007;56:197–212.
Wang CJ, Yang KD, Ko JY, Huang CC, Huang HY, Wang FS. The effects of shockwave on bone healing and systemic concentrations of nitric oxide (NO), TGF-beta1, VEGF and BMP-2 in long bone non-unions. Nitric Oxide. 2009;20(4):298–303. https://doi.org/10.1016/j.niox.2009.02.006.
Weinstein JN, Oster DM, Park JB, Park SH, Loening S. The effect of the extracorporeal shock-wave lithotriptor on the bone-cement interface in dogs. Clin Orthop Relat R. 1988;235:261–7.
Narasaki K, Shimizu H, Beppu M, Aoki H, Takagi M, Takashi M. Effect of extracorporeal shock waves on callus formation during bone lengthening. J Orthop Sci. 2003;8(4):474–81. https://doi.org/10.1007/s00776-003-0664-4.
Hofmann A, Ritz U, Hessmann MH, Alini M, Rommens PM, Rompe JD. Extracorporeal shock wave-mediated changes in proliferation, differentiation, and gene expression of human osteoblasts. J Trauma. 2008;65(6):1402–10. https://doi.org/10.1097/TA.0b013e318173e7c2.
Wang CJ, Weng LH, Ko JY, Wang JW, Chen JM, Sun YC, et al. Extracorporeal shockwave shows regression of osteoarthritis of the knee in rats. J Surg Res. 2011;171(2):601–8. https://doi.org/10.1016/j.jss.2010.06.042.
Suhr F, Delhasse Y, Bungartz G, Schmidt A, Pfannkuche K, Bloch W. Cell biological effects of mechanical stimulations generated by focused extracorporeal shock wave applications on cultured human bone marrow stromal cells. Stem Cell Res. 2013;11(2):951–64. https://doi.org/10.1016/j.scr.2013.05.010.
Catalano MG, Marano F, Rinella L, de Girolamo L, Bosco O, Fortunati N, et al. Extracorporeal shockwaves (ESWs) enhance the osteogenic medium-induced differentiation of adipose-derived stem cells into osteoblast-like cells. J Tissue Eng Regen Med. 2017;11(2):390–9. https://doi.org/10.1002/term.1922.
Wang FS, Yang KD, Chen RF, Wang CJ, Sheen-Chen SM. Extracorporeal shock wave promotes growth and differentiation of bone-marrow stromal cells towards osteoprogenitors associated with induction of TGF-beta1. J Bone Joint Surg. 2002;84(3):457–61.
Da Costa Gomez TM, Radtke CL, Kalscheur VL, Swain CA, Scollay MC, Edwards RB, et al. Effect of focused and radial extracorporeal shock wave therapy on equine bone microdamage. Vet Surg. 2004;33(1):49–55.
Wang CJ, Wang FS, Yang KD, Weng LH, Hsu CC, Huang CS, et al. Shock wave therapy induces neovascularization at the tendon-bone junction. A study in rabbits. J Orthop Res. 2003;21(6):984–9. https://doi.org/10.1016/S0736-0266(03)00104-9.
Chen YJ, Kuo YR, Yang KD, Wang CJ, Sheen Chen SM, Huang HC, et al. Activation of extracellular signal-regulated kinase (ERK) and p38 kinase in shock wave-promoted bone formation of segmental defect in rats. Bone. 2004;34(3):466–77. https://doi.org/10.1016/j.bone.2003.11.013.
Wang FS, Yang KD, Kuo YR, Wang CJ, Sheen-Chen SM, Huang HC, et al. Temporal and spatial expression of bone morphogenetic proteins in extracorporeal shock wave-promoted healing of segmental defect. Bone. 2003;32(4):387–96.
Yip HK, Chang LT, Sun CK, Youssef AA, Sheu JJ, Wang CJ. Shock wave therapy applied to rat bone marrow-derived mononuclear cells enhances formation of cells stained positive for CD31 and vascular endothelial growth factor. Circ J. 2008;72(1):150–6.
Wang FS, Wang CJ, Huang HJ, Chung H, Chen RF, Yang KD. Physical shock wave mediates membrane hyperpolarization and Ras activation for osteogenesis in human bone marrow stromal cells. Biochem Biophys Res Commun. 2001;287(3):648–55. https://doi.org/10.1006/bbrc.2001.5654.
McKinlay A, Vecchia P, Ziegelberger G, Greinert R. Progress in biophysics and molecular biology. Editorial Prog Biophys Mol Biol. 2011;107(3):311. https://doi.org/10.1016/j.pbiomolbio.2011.09.021.
Wang FS, Wang CJ, Sheen-Chen SM, Kuo YR, Chen RF, Yang KD. Superoxide mediates shock wave induction of ERK-dependent osteogenic transcription factor (CBFA1) and mesenchymal cell differentiation toward osteoprogenitors. J Biol Chem. 2002;277(13):10931–7. https://doi.org/10.1074/jbc.M104587200.
Marino G, Rosso F, Cafiero G, Tortora C, Moraci M, Barbarisi M, et al. Beta-tricalcium phosphate 3D scaffold promote alone osteogenic differentiation of human adipose stem cells: in vitro study. J Mater Sci Mater Med. 2010;21(1):353–63. https://doi.org/10.1007/s10856-009-3840-z.
Liu Q, Cen L, Yin S, Chen L, Liu G, Chang J, et al. A comparative study of proliferation and osteogenic differentiation of adipose-derived stem cells on akermanite and beta-TCP ceramics. Biomaterials. 2008;29(36):4792–9. https://doi.org/10.1016/j.biomaterials.2008.08.039.
McCullen SD, Haslauer CM, Loboa EG. Musculoskeletal mechanobiology: interpretation by external force and engineered substratum. J Biomech. 2010;43(1):119–27. https://doi.org/10.1016/j.jbiomech.2009.09.017.
Joshi SD, Webb K. Variation of cyclic strain parameters regulates development of elastic modulus in fibroblast/substrate constructs. J Orthop Res. 2008;26(8):1105–13. https://doi.org/10.1002/jor.20626.
McBeath R, Pirone DM, Nelson CM, Bhadriraju K, Chen CS. Cell shape, cytoskeletal tension, and RhoA regulate stem cell lineage commitment. Dev Cell. 2004;6(4):483–95.
Gregg RH 2nd, McCarthy D. Laser periodontal therapy for bone regeneration. Dentistry Today. 2002;21(5):54–9.
Zein R, Selting W, Benedicenti S. Effect of low-level laser therapy on bone regeneration during osseointegration and bone graft. Photomed Laser Surg. 2017;35(12):649–58. https://doi.org/10.1089/pho.2017.4275.
Isler SC, Cansiz E, Tanyel C, Soluk M, Selvi F, Cebi Z. The effect of irrigation temperature on bone healing. Int J Med Sci. 2011;8(8):704–8.
Mishra SK, Chowdhary R. Heat generated by dental implant drills during osteotomy—a review: heat generated by dental implant drills. J Indian Prosthodont Soc. 2014;14(2):131–43. https://doi.org/10.1007/s13191-014-0350-6.
Yasuda K, Okazaki Y, Abe Y, Tsuga K. Effective UV/ozone irradiation method for decontamination of hydroxyapatite surfaces. Heliyon. 2017;3(8):e00372. https://doi.org/10.1016/j.heliyon.2017.e00372.
Chen C, Tambe DT, Deng L, Yang L. Biomechanical properties and mechanobiology of the articular chondrocyte. Am J Physiol Cell Physiol. 2013;305(12):C1202–8. https://doi.org/10.1152/ajpcell.00242.2013.
Guilak F, Erickson GR, Ting-Beall HP. The effects of osmotic stress on the viscoelastic and physical properties of articular chondrocytes. Biophys J. 2002;82(2):720–7. https://doi.org/10.1016/S0006-3495(02)75434-9.
Kisiday JD, Lee JH, Siparsky PN, Frisbie DD, Flannery CR, Sandy JD, et al. Catabolic responses of chondrocyte-seeded peptide hydrogel to dynamic compression. Ann Biomed Eng. 2009;37(7):1368–75. https://doi.org/10.1007/s10439-009-9699-9.
Nicodemus GD, Bryant SJ. Mechanical loading regimes affect the anabolic and catabolic activities by chondrocytes encapsulated in PEG hydrogels. Osteoarthr Cartil. 2010;18(1):126–37. https://doi.org/10.1016/j.joca.2009.08.005.
Jeon JE, Schrobback K, Hutmacher DW, Klein TJ. Dynamic compression improves biosynthesis of human zonal chondrocytes from osteoarthritis patients. Osteoarthr Cartil. 2012;20(8):906–15. https://doi.org/10.1016/j.joca.2012.04.019.
Shieh AC, Athanasiou KA. Dynamic compression of single cells. Osteoarthr Cartil. 2007;15(3):328–34. https://doi.org/10.1016/j.joca.2006.08.013.
Nugent GE, Aneloski NM, Schmidt TA, Schumacher BL, Voegtline MS, Sah RL. Dynamic shear stimulation of bovine cartilage biosynthesis of proteoglycan 4. Arthritis Rheum. 2006;54(6):1888–96. https://doi.org/10.1002/art.21831.
Grad S, Lee CR, Gorna K, Gogolewski S, Wimmer MA, Alini M. Surface motion upregulates superficial zone protein and hyaluronan production in chondrocyte-seeded three-dimensional scaffolds. Tissue Eng. 2005;11(1–2):249–56. https://doi.org/10.1089/ten.2005.11.249.
Ofek G, Dowling EP, Raphael RM, McGarry JP, Athanasiou KA. Biomechanics of single chondrocytes under direct shear. Biomech Model Mechanobiol. 2010;9(2):153–62. https://doi.org/10.1007/s10237-009-0166-1.
Angele P, Yoo JU, Smith C, Mansour J, Jepsen KJ, Nerlich M, et al. Cyclic hydrostatic pressure enhances the chondrogenic phenotype of human mesenchymal progenitor cells differentiated in vitro. J Orthop Res. 2003;21(3):451–7. https://doi.org/10.1016/S0736-0266(02)00230-9.
Trindade MC, Shida J, Ikenoue T, Lee MS, Lin EY, Yaszay B, et al. Intermittent hydrostatic pressure inhibits matrix metalloproteinase and pro-inflammatory mediator release from human osteoarthritic chondrocytes in vitro. Osteoarthr Cartil. 2004;12(9):729–35. https://doi.org/10.1016/j.joca.2004.05.008.
Zhang Y, Tang CL, Chen WJ, Zhang Q, Wang SL. Dynamic compression combined with exogenous SOX-9 promotes chondrogenesis of adipose-derived mesenchymal stem cells in PLGA scaffold. Eur Rev Med Pharmacol Sci. 2015;19(14):2671–8.
Shahin K, Doran PM. Tissue engineering of cartilage using a mechanobioreactor exerting simultaneous mechanical shear and compression to simulate the rolling action of articular joints. Biotechnol Bioeng. 2012;109(4):1060–73. https://doi.org/10.1002/bit.24372.
McMahon LA, Reid AJ, Campbell VA, Prendergast PJ. Regulatory effects of mechanical strain on the chondrogenic differentiation of MSCs in a collagen-GAG scaffold: experimental and computational analysis. Ann Biomed Eng. 2008;36(2):185–94. https://doi.org/10.1007/s10439-007-9416-5.
Schatti O, Grad S, Goldhahn J, Salzmann G, Li Z, Alini M, et al. A combination of shear and dynamic compression leads to mechanically induced chondrogenesis of human mesenchymal stem cells. Eur Cell Mater. 2011;22:214–25.
Li Z, Yao SJ, Alini M, Stoddart MJ. Chondrogenesis of human bone marrow mesenchymal stem cells in fibrin-polyurethane composites is modulated by frequency and amplitude of dynamic compression and shear stress. Tissue Eng Part A. 2010;16(2):575–84. https://doi.org/10.1089/ten.TEA.2009.0262.
Huang AH, Farrell MJ, Kim M, Mauck RL. Long-term dynamic loading improves the mechanical properties of chondrogenic mesenchymal stem cell-laden hydrogel. Eur Cell Mater. 2010;19:72–85.
Thorpe SD, Buckley CT, Vinardell T, O'Brien FJ, Campbell VA, Kelly DJ. Dynamic compression can inhibit chondrogenesis of mesenchymal stem cells. Biochem Biophys Res Commun. 2008;377(2):458–62. https://doi.org/10.1016/j.bbrc.2008.09.154.
Kock L, van Donkelaar CC, Ito K. Tissue engineering of functional articular cartilage: the current status. Cell Tissue Res. 2012;347(3):613–27. https://doi.org/10.1007/s00441-011-1243-1.
Haudenschild DR, D'Lima DD, Lotz MK. Dynamic compression of chondrocytes induces a Rho kinase-dependent reorganization of the actin cytoskeleton. Biorheology. 2008;45(3–4):219–28.
Guilak F. Compression-induced changes in the shape and volume of the chondrocyte nucleus. J Biomech. 1995;28(12):1529–41.
Ingber D. Integrins as mechanochemical transducers. Curr Opin Cell Biol. 1991;3(5):841–8.
Shakibaei M. Inhibition of chondrogenesis by integrin antibody in vitro. Exp Cell Res. 1998;240(1):95–106. https://doi.org/10.1006/excr.1998.3933.
Salter DM, Millward-Sadler SJ, Nuki G, Wright MO. Integrin-interleukin-4 mechanotransduction pathways in human chondrocytes. Clin Orthop Relat Res 2001(391 Suppl):S49–60.
Baroja-Mazo A, Barbera-Cremades M, Pelegrin P. The participation of plasma membrane hemichannels to purinergic signaling. Biochim Biophys Acta. 2013;1828(1):79–93. https://doi.org/10.1016/j.bbamem.2012.01.002.
Millward-Sadler SJ, Wright MO, Flatman PW, Salter DM. ATP in the mechanotransduction pathway of normal human chondrocytes. Biorheology. 2004;41(3–4):567–75.
Pingguan-Murphy B, El-Azzeh M, Bader DL, Knight MM. Cyclic compression of chondrocytes modulates a purinergic calcium signalling pathway in a strain rate- and frequency-dependent manner. J Cell Physiol. 2006;209(2):389–97. https://doi.org/10.1002/jcp.20747.
Guilak F, Zell RA, Erickson GR, Grande DA, Rubin CT, McLeod KJ, et al. Mechanically induced calcium waves in articular chondrocytes are inhibited by gadolinium and amiloride. J Orthop Res. 1999;17(3):421–9. https://doi.org/10.1002/jor.1100170319.
Guilak F, Leddy HA, Liedtke W. Transient receptor potential vanilloid 4: the sixth sense of the musculoskeletal system? Ann N Y Acad Sci. 2010;1192:404–9. https://doi.org/10.1111/j.1749-6632.2010.05389.x.
Muramatsu S, Wakabayashi M, Ohno T, Amano K, Ooishi R, Sugahara T, et al. Functional gene screening system identified TRPV4 as a regulator of chondrogenic differentiation. J Biol Chem. 2007;282(44):32158–67. https://doi.org/10.1074/jbc.M706158200.
Roberts SR, Knight MM, Lee DA, Bader DL. Mechanical compression influences intracellular Ca2+ signaling in chondrocytes seeded in agarose constructs. J Appl Physiol (1985). 2001;90(4):1385–91. https://doi.org/10.1152/jappl.2001.90.4.1385.
Kono T, Nishikori T, Kataoka H, Uchio Y, Ochi M, Enomoto K. Spontaneous oscillation and mechanically induced calcium waves in chondrocytes. Cell Biochem Funct. 2006;24(2):103–11. https://doi.org/10.1002/cbf.1304.
Edlich M, Yellowley CE, Jacobs CR, Donahue HJ. Oscillating fluid flow regulates cytosolic calcium concentration in bovine articular chondrocytes. J Biomech. 2001;34(1):59–65.
Browning JA, Saunders K, Urban JP, Wilkins RJ. The influence and interactions of hydrostatic and osmotic pressures on the intracellular milieu of chondrocytes. Biorheology. 2004;41(3–4):299–308.
Chao PH, West AC, Hung CT. Chondrocyte intracellular calcium, cytoskeletal organization, and gene expression responses to dynamic osmotic loading. Am J Physiol Cell Physiol. 2006;291(4):C718–25. https://doi.org/10.1152/ajpcell.00127.2005.
Han SK, Wouters W, Clark A, Herzog W. Mechanically induced calcium signaling in chondrocytes in situ. J Orthop Res. 2012;30(3):475–81. https://doi.org/10.1002/jor.21536.
Fanning PJ, Emkey G, Smith RJ, Grodzinsky AJ, Szasz N, Trippel SB. Mechanical regulation of mitogen-activated protein kinase signaling in articular cartilage. J Biol Chem. 2003;278(51):50940–8. https://doi.org/10.1074/jbc.M305107200.
Fitzgerald JB, Jin M, Chai DH, Siparsky P, Fanning P, Grodzinsky AJ. Shear- and compression-induced chondrocyte transcription requires MAPK activation in cartilage explants. J Biol Chem. 2008;283(11):6735–43. https://doi.org/10.1074/jbc.M708670200.
Tew SR, Vasieva O, Peffers MJ, Clegg PD. Post-transcriptional gene regulation following exposure of osteoarthritic human articular chondrocytes to hyperosmotic conditions. Osteoarthr Cartil. 2011;19(8):1036–46. https://doi.org/10.1016/j.joca.2011.04.015.
Bougault C, Aubert-Foucher E, Paumier A, Perrier-Groult E, Huot L, Hot D, et al. Dynamic compression of chondrocyte-agarose constructs reveals new candidate mechanosensitive genes. PLoS One. 2012;7(5):e36964. https://doi.org/10.1371/journal.pone.0036964.
Mouw JK, Connelly JT, Wilson CG, Michael KE, Levenston ME. Dynamic compression regulates the expression and synthesis of chondrocyte-specific matrix molecules in bone marrow stromal cells. Stem Cells. 2007;25(3):655–63. https://doi.org/10.1634/stemcells.2006-0435.
Li Z, Kupcsik L, Yao SJ, Alini M, Stoddart MJ. Mechanical load modulates chondrogenesis of human mesenchymal stem cells through the TGF-beta pathway. J Cell Mol Med. 2010;14(6A):1338–46. https://doi.org/10.1111/j.1582-4934.2009.00780.x.
Frank EH, Grodzinsky AJ. Cartilage electromechanics—I. Electrokinetic transduction and the effects of electrolyte pH and ionic strength. J Biomech. 1987;20(6):615–27.
Frank EH, Grodzinsky AJ. Cartilage electromechanics—II. A continuum model of cartilage electrokinetics and correlation with experiments. J Biomech. 1987;20(6):629–39.
MacGinitie LA, Gluzband YA, Grodzinsky AJ. Electric field stimulation can increase protein synthesis in articular cartilage explants. J Orthop Res. 1994;12(2):151–60. https://doi.org/10.1002/jor.1100120202.
Nogami H, Aoki H, Okagawa T, Mimatsu K. Effects of electric current on chondrogenesis in vitro. Clin Orthop Relat Res. 1982;163:243–7.
Chao PH, Roy R, Mauck RL, Liu W, Valhmu WB, Hung CT. Chondrocyte translocation response to direct current electric fields. J Biomech Eng. 2000;122(3):261–7.
Gunja NJ, Dujari D, Chen A, Luengo A, Fong JV, Hung CT. Migration responses of outer and inner meniscus cells to applied direct current electric fields. J Orthop Res. 2012;30(1):103–11. https://doi.org/10.1002/jor.21489.
Rodan GA, Bourret LA, Norton LA. DNA synthesis in cartilage cells is stimulated by oscillating electric fields. Science. 1978;199(4329):690–2.
Wang W, Wang Z, Zhang G, Clark CC, Brighton CT. Up-regulation of chondrocyte matrix genes and products by electric fields. Clin Orthop Relat Res 2004(427 Suppl):S163–73.
Brighton CT, Wang W, Clark CC. Up-regulation of matrix in bovine articular cartilage explants by electric fields. Biochem Biophys Res Commun. 2006;342(2):556–61. https://doi.org/10.1016/j.bbrc.2006.01.171.
Brighton CT, Wang W, Clark CC. The effect of electrical fields on gene and protein expression in human osteoarthritic cartilage explants. J Bone Joint Surg Am. 2008;90(4):833–48. https://doi.org/10.2106/JBJS.F.01437.
Varani K, De Mattei M, Vincenzi F, Gessi S, Merighi S, Pellati A, et al. Characterization of adenosine receptors in bovine chondrocytes and fibroblast-like synoviocytes exposed to low frequency low energy pulsed electromagnetic fields. Osteoarthr Cartil. 2008;16(3):292–304. https://doi.org/10.1016/j.joca.2007.07.004.
Tesch AM, MacDonald MH, Kollias-Baker C, Benton HP. Chondrocytes respond to adenosine via A(2)receptors and activity is potentiated by an adenosine deaminase inhibitor and a phosphodiesterase inhibitor. Osteoarthr Cartil. 2002;10(1):34–43. https://doi.org/10.1053/joca.2001.0479.
Xu J, Wang W, Clark CC, Brighton CT. Signal transduction in electrically stimulated articular chondrocytes involves translocation of extracellular calcium through voltage-gated channels. Osteoarthr Cartil. 2009;17(3):397–405. https://doi.org/10.1016/j.joca.2008.07.001.
Fitzsimmons RJ, Gordon SL, Kronberg J, Ganey T, Pilla AA. A pulsing electric field (PEF) increases human chondrocyte proliferation through a transduction pathway involving nitric oxide signaling. J Orthop Res. 2008;26(6):854–9. https://doi.org/10.1002/jor.20590.
Dini L, Abbro L. Bioeffects of moderate-intensity static magnetic fields on cell cultures. Micron. 2005;36(3):195–217. https://doi.org/10.1016/j.micron.2004.12.009.
Brady MA, Waldman SD, Ethier CR. The application of multiple biophysical cues to engineer functional neocartilage for treatment of osteoarthritis. Part I: cellular response. Tissue Eng Part B Rev. 2015;21(1):1–19. https://doi.org/10.1089/ten.TEB.2013.0757.
Stolfa S, Skorvanek M, Stolfa P, Rosocha J, Vasko G, Sabo J. Effects of static magnetic field and pulsed electromagnetic field on viability of human chondrocytes in vitro. Physiol Res. 2007;56(Suppl 1):S45–9.
Amin HD, Brady MA, St-Pierre JP, Stevens MM, Overby DR, Ethier CR. Stimulation of chondrogenic differentiation of adult human bone marrow-derived stromal cells by a moderate-strength static magnetic field. Tissue Eng Part A. 2014;20(11–12):1612–20. https://doi.org/10.1089/ten.tea.2013.0307.
Trock DH, Bollet AJ, Dyer RH Jr, Fielding LP, Miner WK, Markoll R. A double-blind trial of the clinical effects of pulsed electromagnetic fields in osteoarthritis. J Rheumatol. 1993;20(3):456–60.
Vavken P, Arrich F, Schuhfried O, Dorotka R. Effectiveness of pulsed electromagnetic field therapy in the management of osteoarthritis of the knee: a meta-analysis of randomized controlled trials. J Rehabil Med. 2009;41(6):406–11. https://doi.org/10.2340/16501977-0374.
Sakai A, Suzuki K, Nakamura T, Norimura T, Tsuchiya T. Effects of pulsing electromagnetic fields on cultured cartilage cells. Int Orthop. 1991;15(4):341–6.
Pezzetti F, De Mattei M, Caruso A, Cadossi R, Zucchini P, Carinci F, et al. Effects of pulsed electromagnetic fields on human chondrocytes: an in vitro study. Calcif Tissue Int. 1999;65(5):396–401.
De Mattei M, Caruso A, Pezzetti F, Pellati A, Stabellini G, Sollazzo V, et al. Effects of pulsed electromagnetic fields on human articular chondrocyte proliferation. Connect Tissue Res. 2001;42(4):269–79.
De Mattei M, Pasello M, Pellati A, Stabellini G, Massari L, Gemmati D, et al. Effects of electromagnetic fields on proteoglycan metabolism of bovine articular cartilage explants. Connect Tissue Res. 2003;44(3–4):154–9.
De Mattei M, Pellati A, Pasello M, Ongaro A, Setti S, Massari L, et al. Effects of physical stimulation with electromagnetic field and insulin growth factor-I treatment on proteoglycan synthesis of bovine articular cartilage. Osteoarthr Cartil. 2004;12(10):793–800. https://doi.org/10.1016/j.joca.2004.06.012.
Bobacz K, Graninger WB, Amoyo L, Smolen JS. Effect of pulsed electromagnetic fields on proteoglycan biosynthesis of articular cartilage is age dependent. Ann Rheum Dis. 2006;65(7):949–51. https://doi.org/10.1136/ard.2005.037622.
Chang SH, Hsiao YW, Lin HY. Low-frequency electromagnetic field exposure accelerates chondrocytic phenotype expression on chitosan substrate. Orthopedics. 2011;34(1):20. https://doi.org/10.3928/01477447-20101123-10.
Ongaro A, Pellati A, Masieri FF, Caruso A, Setti S, Cadossi R, et al. Chondroprotective effects of pulsed electromagnetic fields on human cartilage explants. Bioelectromagnetics. 2011;32(7):543–51. https://doi.org/10.1002/bem.20663.
Mayer-Wagner S, Passberger A, Sievers B, Aigner J, Summer B, Schiergens TS, et al. Effects of low frequency electromagnetic fields on the chondrogenic differentiation of human mesenchymal stem cells. Bioelectromagnetics. 2011;32(4):283–90. https://doi.org/10.1002/bem.20633.
Chen CH, Lin YS, Fu YC, Wang CK, Wu SC, Wang GJ, et al. Electromagnetic fields enhance chondrogenesis of human adipose-derived stem cells in a chondrogenic microenvironment in vitro. J Appl Physiol (1985). 2013;114(5):647–55. https://doi.org/10.1152/japplphysiol.01216.2012.
Levin M. Bioelectromagnetics in morphogenesis. Bioelectromagnetics. 2003;24(5):295–315. https://doi.org/10.1002/bem.10104.
Robinson KR, Messerli MA. Left/right, up/down: the role of endogenous electrical fields as directional signals in development, repair and invasion. BioEssays. 2003;25(8):759–66. https://doi.org/10.1002/bies.10307.
Lippiello L, Chakkalakal D, Connolly JF. Pulsing direct current-induced repair of articular cartilage in rabbit osteochondral defects. J Orthop Res. 1990;8(2):266–75. https://doi.org/10.1002/jor.1100080216.
Brady MA, Waldman SD, Ethier CR. The application of multiple biophysical cues to engineer functional neocartilage for treatment of osteoarthritis. Part II: signal transduction. Tissue Eng Part B Rev. 2015;21(1):20–33. https://doi.org/10.1089/ten.TEB.2013.0760.
Hsieh CH, Lee MC, Tsai-Wu JJ, Chen MH, Lee HS, Chiang H, et al. Deleterious effects of MRI on chondrocytes. Osteoarthr Cartil. 2008;16(3):343–51. https://doi.org/10.1016/j.joca.2007.07.001.
Varani K, Vincenzi F, Tosi A, Targa M, Masieri FF, Ongaro A, et al. Expression and functional role of adenosine receptors in regulating inflammatory responses in human synoviocytes. Br J Pharmacol. 2010;160(1):101–15. https://doi.org/10.1111/j.1476-5381.2010.00667.x.
Cook SD, Salkeld SL, Popich-Patron LS, Ryaby JP, Jones DG, Barrack RL. Improved cartilage repair after treatment with low-intensity pulsed ultrasound. Clin Orthop Relat Res 2001(391 Suppl):S231–43.
Min BH, Choi BH, Park SR. Low intensity ultrasound as a supporter of cartilage regeneration and its engineering. Biotechnol Bioproc E. 2007;12(1):22–31. https://doi.org/10.1007/Bf02931799.
Parvizi J, Wu CC, Lewallen DG, Greenleaf JF, Bolander ME. Low-intensity ultrasound stimulates proteoglycan synthesis in rat chondrocytes by increasing aggrecan gene expression. J Orthop Res. 1999;17(4):488–94. https://doi.org/10.1002/jor.1100170405.
Zhang ZJ, Huckle J, Francomano CA, Spencer RG. The effects of pulsed low-intensity ultrasound on chondrocyte viability, proliferation, gene expression and matrix production. Ultrasound Med Biol. 2003;29(11):1645–51.
Nishikori T, Ochi M, Uchio Y, Maniwa S, Kataoka H, Kawasaki K, et al. Effects of low-intensity pulsed ultrasound on proliferation and chondroitin sulfate synthesis of cultured chondrocytes embedded in Atelocollagen gel. J Biomed Mater Res. 2002;59(2):201–6.
Yang KH, Parvizi J, Wang SJ, Lewallen DG, Kinnick RR, Greenleaf JF, et al. Exposure to low-intensity ultrasound increases aggrecan gene expression in a rat femur fracture model. J Orthop Res. 1996;14(5):802–9. https://doi.org/10.1002/jor.1100140518.
Zhang Z, Huckle J, Francomano CA, Spencer RG. The influence of pulsed low-intensity ultrasound on matrix production of chondrocytes at different stages of differentiation: an explant study. Ultrasound Med Biol. 2002;28(11–12):1547–53.
Huang MH, Ding HJ, Chai CY, Huang YF, Yang RC. Effects of sonication on articular cartilage in experimental osteoarthritis. J Rheumatol. 1997;24(10):1978–84.
Choi BH, Woo JI, Min BH, Park SR. Low-intensity ultrasound stimulates the viability and matrix gene expression of human articular chondrocytes in alginate bead culture. J Biomed Mater Res A. 2006;79((4):858–64. https://doi.org/10.1002/jbm.a.30816.
Park SR, Park SH, Jang KW, Cho HS, Cui JH, An HJ, et al. The effect of sonication on simulated osteoarthritis. Part II: alleviation of osteoarthritis pathogenesis by 1 MHz ultrasound with simultaneous hyaluronate injection. Ultrasound Med Biol. 2005;31(11):1559–66. https://doi.org/10.1016/j.ultrasmedbio.2005.07.001.
Lee HJ, Choi BH, Min BH, Son YS, Park SR. Low-intensity ultrasound stimulation enhances chondrogenic differentiation in alginate culture of mesenchymal stem cells. Artif Organs. 2006;30(9):707–15. https://doi.org/10.1111/j.1525-1594.2006.00288.x.
Schumann D, Kujat R, Zellner J, Angele MK, Nerlich M, Mayr E, et al. Treatment of human mesenchymal stem cells with pulsed low intensity ultrasound enhances the chondrogenic phenotype in vitro. Biorheology. 2006;43(3,4):431–43.
Cui JH, Park SR, Park K, Choi BH, Min BH. Preconditioning of mesenchymal stem cells with low-intensity ultrasound for cartilage formation in vivo. Tissue Eng. 2007;13(2):351–60. https://doi.org/10.1089/ten.2006.0080.
Cui JH, Park K, Park SR, Min BH. Effects of low-intensity ultrasound on chondrogenic differentiation of mesenchymal stem cells embedded in polyglycolic acid: an in vivo study. Tissue Eng. 2006;12(1):75–82. https://doi.org/10.1089/ten.2006.12.75.
Ebisawa K, Hata K, Okada K, Kimata K, Ueda M, Torii S, et al. Ultrasound enhances transforming growth factor beta-mediated chondrocyte differentiation of human mesenchymal stem cells. Tissue Eng. 2004;10(5–6):921–9. https://doi.org/10.1089/1076327041348437.
Lai CH, Chen SC, Chiu LH, Yang CB, Tsai YH, Zuo CS, et al. Effects of low-intensity pulsed ultrasound, dexamethasone/TGF-beta1 and/or BMP-2 on the transcriptional expression of genes in human mesenchymal stem cells: chondrogenic vs. osteogenic differentiation. Ultrasound Med Biol. 2010;36(6):1022–33. https://doi.org/10.1016/j.ultrasmedbio.2010.03.014.
Takeuchi R, Ryo A, Komitsu N, Mikuni-Takagaki Y, Fukui A, Takagi Y, et al. Low-intensity pulsed ultrasound activates the phosphatidylinositol 3 kinase/Akt pathway and stimulates the growth of chondrocytes in three-dimensional cultures: a basic science study. Arthritis Res Ther. 2008;10(4):R77. https://doi.org/10.1186/ar2451.
Mukai S, Ito H, Nakagawa Y, Akiyama H, Miyamoto M, Nakamura T. Transforming growth factor-beta1 mediates the effects of low-intensity pulsed ultrasound in chondrocytes. Ultrasound Med Biol. 2005;31(12):1713–21. https://doi.org/10.1016/j.ultrasmedbio.2005.07.012.
Whitney NP, Lamb AC, Louw TM, Subramanian A. Integrin-mediated mechanotransduction pathway of low-intensity continuous ultrasound in human chondrocytes. Ultrasound Med Biol. 2012;38(10):1734–43. https://doi.org/10.1016/j.ultrasmedbio.2012.06.002.
Wang CJ, Weng LH, Ko JY, Sun YC, Yang YJ, Wang FS. Extracorporeal shockwave therapy shows chondroprotective effects in osteoarthritic rat knee. Arch Orthop Traum Su. 2011;131(8):1153–8. https://doi.org/10.1007/s00402-011-1289-2.
Wang CJ, Hsu SL, Weng LH, Sun YC, Wang FS. Extracorporeal shockwave therapy shows a number of treatment related chondroprotective effect in osteoarthritis of the knee in rats. BMC Musculoskel Dis. 2013;14. doi:Artn 44. https://doi.org/10.1186/1471-2474-14-44.
Wang CJ, Sun YC, Wong T, Hsu SL, Chou WY, Chang HW. Extracorporeal shockwave therapy shows time-dependent chondroprotective effects in osteoarthritis of the knee in rats. J Surg Res. 2012;178(1):196–205. https://doi.org/10.1016/j.jss.2012.01.010.
Moretti B, Iannone F, Notarnicola A, Lapadula G, Moretti L, Patella V et al. Extracorporeal shock waves down-regulate the expression of interleukin-10 and tumor necrosis factor-alpha in osteoarthritic chondrocytes. BMC Musculoskel Dis. 2008;9. doi:Artn 16 https://doi.org/10.1186/1471-2474-9-16.
Wang P, Liu C, Yang XT, Wei XF, Zhou YJ, Yang L, et al. Effect of extracorporeal shock wave therapy on cartilage and subchondral bone remodeling in rabbits with ACLT-induced osteoarthritis. Sichuan Da Xue Xue Bao Yi Xue Ban. 2014;45(1):120–5.
Zhao Z, Ji H, Jing R, Liu C, Wang M, Zhai L, et al. Extracorporeal shock-wave therapy reduces progression of knee osteoarthritis in rabbits by reducing nitric oxide level and chondrocyte apoptosis. Arch Orthop Trauma Surg. 2012;132(11):1547–53. https://doi.org/10.1007/s00402-012-1586-4.
Wang CJ, Hsu SL, Weng LH, Sun YC, Wang FS. Extracorporeal shockwave therapy shows a number of treatment related chondroprotective effect in osteoarthritis of the knee in rats. BMC Musculoskelet Disord. 2013;14:44. https://doi.org/10.1186/1471-2474-14-44.
Lyon R, Liu XC, Kubin M, Schwab J. Does extracorporeal shock wave therapy enhance healing of osteochondritis dissecans of the rabbit knee?: a pilot study. Clin Orthop Relat Res. 2013;471(4):1159–65. https://doi.org/10.1007/s11999-012-2410-8.
Chen TW, Lin CW, Lee CL, Chen CH, Chen YJ, Lin TY, et al. The efficacy of shock wave therapy in patients with knee osteoarthritis and popliteal cyamella. Kaohsiung J Med Sci. 2014;30(7):362–70. https://doi.org/10.1016/j.kjms.2014.03.006.
Zhao Z, Jing R, Shi Z, Zhao B, Ai Q, Xing G. Efficacy of extracorporeal shockwave therapy for knee osteoarthritis: a randomized controlled trial. J Surg Res. 2013;185(2):661–6. https://doi.org/10.1016/j.jss.2013.07.004.
Frairia R, Berta L. Biological effects of extracorporeal shock waves on fibroblasts. A review. Muscles Ligaments Tendons J. 2011;1(4):138–47.
Chen J, Li CH, Wang SH. Periodic heat shock accelerated the chondrogenic differentiation of human mesenchymal stem cells in pellet culture. PLoS One. 2014;9(3):e91561. https://doi.org/10.1371/journal.pone.0091561.
Mangueira NM, Xavier M, de Souza RA, Salgado MA, Silveira L Jr, Villaverde AB. Effect of low-level laser therapy in an experimental model of osteoarthritis in rats evaluated through Raman spectroscopy. Photomed Laser Surg. 2015;33(3):145–53. https://doi.org/10.1089/pho.2014.3744.
Goldshmid R, Cohen S, Shachaf Y, Kupershmit I, Sarig-Nadir O, Seliktar D, et al. Steric interference of adhesion supports in-vitro chondrogenesis of mesenchymal stem cells on hydrogels for cartilage repair. Sci Rep. 2015;5:12607. https://doi.org/10.1038/srep12607.
Rai V, Dilisio MF, Dietz NE, Agrawal DK. Recent strategies in cartilage repair: a systemic review of the scaffold development and tissue engineering. J Biomed Mater Res A. 2017;105(8):2343–54. https://doi.org/10.1002/jbm.a.36087.
Khan IM, Gilbert SJ, Singhrao SK, Duance VC, Archer CW. Cartilage integration: evaluation of the reasons for failure of integration during cartilage repair. A review. Eur Cell Mater. 2008;16:26–39.
Kang KS, Lee SJ, Lee HS, Moon W, Cho DW. Effects of combined mechanical stimulation on the proliferation and differentiation of pre-osteoblasts. Exp Mol Med. 2011;43(6):367–73. https://doi.org/10.3858/emm.2011.43.6.040.
Funding
The authors thank NIH for the research support (1R21EB024787).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Huang, X., Das, R., Patel, A. et al. Physical Stimulations for Bone and Cartilage Regeneration. Regen. Eng. Transl. Med. 4, 216–237 (2018). https://doi.org/10.1007/s40883-018-0064-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40883-018-0064-0