Abstract
Purpose
Economic evaluation of health services commonly requires information regarding health-state utilities. Sometimes this information is not available but non-utility measures of quality of life may have been collected from which the required utilities can be estimated. This paper examines the possibility of mapping a non-utility-based outcome, the Sydney Asthma Quality of Life Questionnaire (AQLQ-S), onto five multi-attribute utility instruments: Assessment of Quality of Life 8 Dimensions (AQoL-8D), EuroQoL 5 Dimensions 5-Level (EQ-5D-5L), Health Utilities Index Mark 3 (HUI3), 15 Dimensions (15D), and the Short-Form 6 Dimensions (SF-6D).
Methods
Data for 856 individuals with asthma were obtained from a large Multi-Instrument Comparison (MIC) survey. Four statistical techniques were employed to estimate utilities from the AQLQ-S. The predictive accuracy of 180 regression models was assessed using six criteria: mean absolute error (MAE), root mean squared error (RMSE), correlation, distribution of predicted utilities, distribution of residuals, and proportion of predictions with absolute errors <0.0.5. Validation of initial ‘primary’ models was carried out on a random sample of the MIC data.
Results
Best results were obtained with non-linear models that included a quadratic term for the AQLQ-S score along with demographic variables. The four statistical techniques predicted models that performed differently when assessed by the six criteria; however, the best results, for both the estimation and validation samples, were obtained using a generalised linear model (GLM estimator).
Conclusions
It is possible to predict valid utilities from the AQLQ-S using regression methods. We recommend GLM models for this exercise.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
The Sydney Asthma Quality of Life Questionnaire (AQLQ-S) was designed to measure functional problems in adults who have asthma. However, it is currently not possible to estimate health utilities based on the AQLQ-S because of methodological constraints. |
Using regression approaches, our study showed that it is possible to predict health-state utilities for five commonly used multi-attribute utility instruments (MAUIs) from AQLQ-S responses, i.e. the Assessment of Quality of Life 8 Dimensions (AQoL-8D), EuroQoL 5 Dimensions 5-Level (EQ-5D-5L), Health Utilities Index Mark 3 (HUI3), 15 Dimensions (15D), and the Short-Form 6 Dimensions (SF-6D). |
The results of this study can be used to inform utility estimation within future economic evaluations of interventions targeted at populations of people with asthma. |
1 Introduction
The global prevalence, morbidity, mortality and economic burden associated with asthma have been increasing over the years [1]. Asthma affects between 1 and 18 % of the population in different countries, with an estimated 300 million individuals affected worldwide [2, 3]. Its effect on health-related quality of life (HRQoL) is increasingly being measured to inform patient management and policy decisions, including decisions relating to the share of the health budget that should be allocated to the treatment of asthma [4–9]. Many decision bodies, including the UK National Institute for Health and Care Excellence (NICE) and the Australian Pharmaceutical Benefits Advisory Committee (PBAC) and Medical Services Advisory Committee (MSAC) recommend the use of cost-utility analysis (CUA) [10–12], which estimates and compares the cost per additional quality-adjusted life-year (QALY) obtained from each service where QALYs are calculated as life-years times an index of the utility of the relevant health state measured on a 0–1 (death to full health) scale [13, 14]. Increasingly, utilities have been derived from a limited number of multi-attribute utility instruments (MAUIs) [15, 16]; however, MAUIs are often perceived as being less sensitive to particular conditions than non-utility, condition-specific quality-of-life (QoL) measures [17].
The Sydney Asthma Quality of Life Questionnaire (AQLQ-S) is a non-utility-based asthma-specific QoL instrument that was developed to measure functional problems in adults who have asthma [18, 19]. A recent review identified it as one of the most commonly used asthma-specific QoL measures [6]. Compared with one of its variants (the McMaster Asthma Quality of Life Questionnaire [AQLQ-McMaster]), the AQLQ-S has been shown to have lower respondent burden and is therefore preferred by researchers and respondents for inclusion as an asthma-specific QoL measure in broader population health surveys [9]. A limitation of the AQLQ-S for economic evaluation is that it does not have utility weights and cannot therefore be used to estimate QALYs, as needed for a CUA.
This limitation may be overcome by creating an algorithm that predicts utility scores from the AQLQ-S. To date, no such mapping algorithm has been created. Tsuchiya et al. [20] employed ordinary least squares (OLS) and multinomial logistic regression to map the AQLQ-McMaster onto an MAUI, the EuroQoL 5 Dimension 3-Level (EQ-5D-3L), using a sample of 3000 individuals. While the authors concluded that it was possible to estimate a robust relationship between EQ-5D-3L utilities and the AQLQ-McMaster, the study was limited by the exclusion of sociodemographic variables and by only mapping to the EQ-5D-3L, which performs less well on tests of sensitivity and content validity than other MAUIs [21].
Using data from a large Multi-Instrument Comparison (MIC) study [22], the present paper develops mapping algorithms that use the AQLQ-S and patient socio-demographic characteristics to predict utilities for the five most commonly used MAUIs, which are listed in Fig. 1 along with their common abbreviation and major reference in the literature.
2 Methods
We followed the newly developed ‘Mapping onto Preference-Based Measures Reporting Standards’ (MAPS) checklist in conducting this study [23]. The target instruments for mapping were the AQoL-8D, EQ-5D-5L, HUI 3, 15D and SF-6D, while the source instrument was the AQLQ-S.
2.1 Instruments
2.1.1 Sydney Quality of Life Questionnaire (AQLQ-S)
This 20-item, condition-specific instrument was developed to measure the functional impairments that are most troublesome to adults (17–70 years) living with asthma [18, 24], and consists of four domains, some with overlapping items: breathlessness (five items), mood disturbance (five items), social disruption (seven items), and concerns for health (seven items). It has shown good validity when used within asthma populations [19, 25–27]. Results may be reported as average scores for each of the four domains or as a simple score that may be reduced to a 0–1 (worst–best) scale.
2.1.2 Assessment of Quality of Life 8 Dimensions (AQoL-8D)
This is an eight-dimension MAUI designed to assess HRQoL across health conditions, and is applicable to individuals aged ≥16 years [28, 29]. The AQoL-8D measures the following dimensions: independent living, relationships, mental health, coping, pain, senses, happiness and self-worth [30]. Using the time trade-off (TTO) approach [31], population preference weights were obtained from the Australian population, resulting in utilities ranging from −0.094 to 1 [32]. The validity of the AQoL-8D has been proven in multiple patient populations [21, 33–35].
2.1.3 EuroQoL Dimensions 5-Level (EQ-5D-5L)
This is a measure of HRQoL suitable for use on individuals aged ≥18 years, and comprised of five single-item dimensions of health: mobility, self-care, usual activities, pain/discomfort, and anxiety/depression [36]. It is a modification of the original EQ-5D-3L and includes five, rather than three, levels of impairment in each domain: no, slight, moderate, severe, and extreme problems in the relevant dimension of health [37]. Using these responses, the EQ-5D-5L is able to distinguish between 3125 states of health. A UK-specific algorithm developed using TTO techniques was used to convert the EQ-5D-5L health description into a valuation ranging from −0.281 to 1 [38]. Scores less than 0 represent health states that are worse than death [39]. The EQ-5D-5L has been validated in differentiated clinical populations [40–42].
2.1.4 Health Utilities Index Mark 3 (HUI3)
The HUI3 is an HRQoL outcome that measures eight domains, namely vision, hearing, speech, ambulation/mobility, pain, dexterity, emotion, and cognition [43, 44]. Each of these domains has five to six rank-ordered response options. Utilities were developed using a visual analogue scale (VAS) and the Standard Gamble (SG) technique, and ranged from −0.36 to 1 [44]. The HUI3 has been validated in diverse clinical conditions and is suitable for individuals aged 5 years and older [43, 45, 46].
2.1.5 Short-Form 6 Dimensions (SF-6D)
This MAUI was derived from the Short-Form 36 dimensions (SF-36), a 36-item generic HRQoL instrument designed to measure general health concepts across different ages, diseases and treatment groups [47]. The SF-6D consists of six dimensions: vitality, physical functioning, pain, role functioning, social functioning and mental health [48]. The number of levels per dimension varies from four to six. Utilities, developed using the SG approach, can be derived from 11 of the 36 items in the SF-36, and range from 0.291 to 1 [49]. The validity of the SF-6D has been demonstrated in differentiated populations with variable clinical conditions [46, 50–52].
2.1.6 15 Dimensions (15D)
This MAUI is suitable for individuals aged ≥16 years and has 15 HRQoL dimensions, namely mobility, vision, hearing, breathing, sleeping, eating, speech, excretion, usual activities, mental function, discomfort and symptoms, depression, distress, vitality and sexual activity [53]. Each of these dimensions has five ordinal levels of severity [53]. It is well-validated in various clinical populations [54–56], and health states defined by the MAUI can be converted into utilities (ranging from 0 to 1) that were derived as a weighted average of VAS scores for the 15 dimensions [48, 53].
A comparison between the dimensions of the AQLQ-S and those of the five MAUIs shown in Fig. 2 depicts the conceptual overlap between these instruments.
Comparisons between the dimensions of the AQLQ-S and the MAUIs. AQLQ-S Sydney Asthma Quality of Life Questionnaire, AQoL-8D Assessment of Quality of Life 8 Dimensions, EQ-5D-5L EuroQoL 5 Dimensions 5-Level, HUI3 Health Utilities Index Mark 3, SF-6D Short-Form 6 Dimensions, 15D 15 Dimensions, MAUIs multi-attribute utility instruments
2.2 Data
A large MIC survey was carried out in six countries: Australia, Canada, Germany, Norway, the UK and the US, details of which are provided elsewhere [34]. The online survey was administered by a global company (CINT Pty Ltd), to a demographically representative group of the healthy population in each country and to patients in seven major disease areas. Quotas were applied to obtain a target number of respondents in each of the chronic disease areas. Only patients with asthma were included in the present study. Data collected included age, gender, educational level, country of residence, ethnicity, marital status, occupational status, income level, body mass index (BMI), smoking status and responses to the six instruments described above. Data were collected between October 2011 and January 2014, and all participants gave their informed consent prior to inclusion in the study. Ethical approval was granted by the Monash University Human Research Ethics Committee (MUHREC) [CF11/3192–2011001748].
2.3 Statistical Analysis
All analyses were conducted in STATA version 14.1 [57], and the analysis was conducted in two stages. In the first stage, the correlation between the AQLQ-S scores and the five MAUIs was assessed using scatter diagrams and Spearman’s rank correlation coefficients.
In the second stage, independent variables were chosen for inclusion in the regression models. Highly correlated independent variables (r > |0.7|) [58] were identified using Spearman’s rank correlation, and a decision was made with respect to which variables to include in the analysis. The independent variables, including the AQLQ-S, were mapped onto each of the five MAUIs using the nine models described in Table 1. Specific patient characteristics were included when they improved the predictive ability of the models (see Sect. 2.5 for measures of predictive ability). The effect of including dummy variables (representing each of the six countries in the MIC study) in all of the nine models was also tested within a sensitivity analysis. The following regression model families were used in the mapping:
-
OLS regression models These have been the most widely used models in mappings [17]; however they have a potential limitation, i.e. the presence of a data ceiling can lead to inconsistent coefficient estimates [59, 60].
-
Censored least absolute deviations (CLAD) This technique takes the ceiling effect into account and is also robust to heteroscedastic and skewed data [61]. It is consistent and asymptotically normal for a wide class of error distributions [62].
-
The generalised linear model (GLM) This family of models is also robust to heteroscedasticity and skewness [63]. The choice of the GLM distribution and link was guided by the modified park test suggested by Manning [64].
-
The Beta Binomial (BB) regression model The BB model can estimate unimodal or bimodal utilities while being robust to skewness [65, 66]. A limitation of the model is that it restricts utilities to a 0 to 1 range [66]. However, utilities in our data set were positive, except for a small number of observations for the EQ-5D-5L (0.7 %) and HUI3 (1.05 %). As done elsewhere, these data were set equal to 0 [67].
2.4 Estimation and Validation of Primary Models
We used an approach similar to previous studies to estimate ‘primary’ or ‘estimation’ models from a subset of the data, and validated these with the remaining data [68–71]. In this ‘hold-out’ approach, data were split into two parts: an ‘estimation sample’ consisting of two-thirds of the data (793 observations) that were used to construct the primary models, and a ‘validation sample’ consisting of the remaining third (396 observations) that were used for validation. A total of 180 primary models were estimated (four model families × nine model specifications × five MAUI-dependent variables). These primary models were then tested on the validation sample to assess their predictive ability.
2.5 Assessment of Predictive Ability
Predicted utilities from each of the 180 models were estimated using STATA’s inbuilt post-estimation commands. The predictive ability of the models was primarily assessed using two measures of predictive error [72]: the root mean squared error (RMSE) and the mean absolute error (MAE), with lower values of the measures implying a better performing predictive model. To calculate the RMSE, the difference between the observed and predicted values of the MAUIs was squared and then summed over all observations. The RMSE was then estimated as the square root of the mean of these summed values. The MAE was calculated by summing the absolute difference between the observed and predicted values of the MAUIs and the estimated mean of these summed values. Where the RMSE and MAE indicated different results in the validation sample, and as recommended in the literature [73], more weight was placed on the RMSE, particularly when the distribution of the error from the model was Gaussian. Performance of the models was further assessed using four additional criteria estimated using the validation sample, namely (1) the ranges of, and Spearman’s rank correlations between, the predicted and observed utilities; (2) an examination of the distributions of the predicted and observed utilities to determine how closely predicted values matched observed scores [74]; (3) assessment of the distribution of the residuals (observed minus predicted utilities) to determine bias in the predicted utilities [75]; and (iv) assessment of the proportion of predicted utilities deviating from observed values by <0.03 or 0.05 [76]. A breakdown of which regression model family, model specification (among the nine models) and MAUI prediction (AQoL-8D, EQ-5D-5L, HUI3, SF-6D or 15D) performed best according to the six selection criteria (RMSE, MAE and the four additional criteria) is also presented. The best-fitting models overall, based primarily on the performance of their measures of predictive error, were re-estimated using data from the entire sample.
Complete data sets were available for all of the instruments and demographic data analysed.
3 Results
3.1 Demographic and Other Characteristics
Table 2 presents summary statistics for 856 study participants. No significant differences in the instrument scores were observed between the estimation and validation samples. Mean utilities were highest for the 15D (mean 0.85) and lowest for the AQoL-8D (mean 0.69). The majority of individuals in the sample were <45 years of age (58 %), female (62 %), married or living with a partner (59 %), non-smokers (77 %), educated beyond high school (71 %), and had a good or very good standard of living (88 %). There were no statistically significant differences between the estimation and validation sample in terms of patient characteristics. All six countries were fairly represented in the dataset.
3.2 Bivariate Relationship between AQLQ-S and Multi-Attribute Utility Instruments (MAUIs)
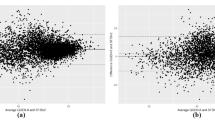
The dimensions of the AQLQ-S and MAUIs are compared in Fig. 2. When contrasted against the dimensions of the AQLQ-S, the 15D had the greatest number of overlapping dimensions (12/15), followed by the SF-6D (5/6), AQoL-8D (6/8), EQ-5D-5L (4/5) and HUI3 (2/8). Figure 3 depicts the relationship between the AQLQ-S total scores and utilities for each of the five MAUIs. The plots show moderate to strong correlation for all comparisons, with the lowest being between the AQLQ-S and the HUI3 (0.458), and the highest being between the AQLQ-S and the 15D (0.544).
Scatter plots between the AQLQ-S total scores and utilities of each of the MAUIs, as well as corresponding correlation coefficients. AQLQ-S Sydney Asthma Quality of Life Questionnaire, MAUIs multi-attribute utility instruments, AQoL-8D Assessment of Quality of Life 8 Dimensions, EQ-5D-5L EuroQoL 5 Dimensions 5-Level, HUI3 Health Utilities Index Mark 3, SF-6D Short-Form 6 Dimensions, 15D 15 Dimensions
3.3 Assessment of Model Predictive Ability
Selection criteria statistics for assessing the predictive ability of the 180 models are presented in electronic supplementary Table 1 for both the estimation and validation samples. These were used to rank each model, resulting in rankings that were sufficiently consistent to permit the selection of a ‘shortlist’ of 10 best-fitting models. The short list for each MAUI, as well as for all MAUIs combined, is shown in electronic supplementary Table 2. A total of nine regression algorithms were candidates for best predicting models as they were the best-fitting models based on the selection criteria statistics: AQoL-8D – OLS (9), AQoL-8D – GLM (9), 15D – OLS (9), 15D – GLM (9), 15D – OLS (9) and 15D – CLAD (8) in the estimation sample, and AQoL-8D – GLM (8), 15D – GLM (8), and 15D – CLAD (5) in the validation sample. Selection criteria statistics estimated using these models ranged from (figures given for the estimation and validation samples) 0.0950 to 0.0973 and 0.0834 to 0.0866 (RMSE), 0.0730 to 0.0740 and 0.0645 to 0.0665 (MAE), 0.6120 to 0.6420 and 0.6370 to 0.6610 (correlation), and 43–49 % and 43–46 % (proportion of predictions with absolute errors <0.0.5). In addition, the ‘minimum to maximum’ range of the predicted probabilities for all nine models was narrower than that for the observed utilities, while the distribution of the residuals for both samples all appear close to being normally distributed (supporting our decision to put more weight on the RMSE for selecting the best-fitting model [67]). Below, these nine regression models are now analysed in order to give a breakdown of which regression model family, model specification and MAUI prediction performed best according to the selection criteria.
With respect to the performance of the regression model families, there were some mixed results. Overall, however, the OLS and GLM performed best on correlation, MAE and RMSE and the CLAD on the proportion of predicted utilities deviating from mean observed utilities by <0.05 (electronic supplementary Table 2). The OLS and GLM predicted mean utilities whose values were closest to those of the observed utilities; however, the CLAD predicted more utilities whose distribution ‘mimicked’ that of the observed values. Fewer CLAD-predicted utilities also deviated from the mean utilities by >0.05. Based on best performance on the most criteria, the OLS and GLM were deemed to have been better models.
In terms of model specification, electronic supplementary Table 2 shows that, regardless of the MAUI predicted, model specifications (8) and (9) performed the best. Model (8) was a non-linear model that included a quadratic term of the AQLQ-S, as well as demographic characteristics as independent variables. Interaction terms were added to these independent variables in model (9).
With respect to prediction of specific MAUIs (electronic supplementary Table 2), the prediction of 15D was the strongest when assessed using the RMSE, MAE and proportion of predicted utilities deviating from mean observed utilities by <0.05, while that for the AQoL-8D was strongest when correlation was assessed. There was mixed performance from the ‘AQLQ-S to AQoL-8D’ and ‘AQLQ-S to EQ-5D-5L’ predictions. Therefore, based on best performance on the most criteria the ‘AQLQ-S to 15D’ prediction was, on average, the strongest, followed by the ‘AQLQ-S to SF-6D’, while the ‘AQLQ-S to HUI3’ prediction was the weakest.
3.4 Best-Performing Models Overall for All MAUIs
When the regression model families and model specifications are considered together, GLM (8) performed best on the RMSE and MAE (except for the HUI3 and SF-6D predictions where CLAD (8) performed best on the MAE). In the estimation sample, GLM (8) was ranked within the top four best-performing models for all MAUI predictions in terms of the RMSE and MAE (except for the EQ-5D-5L prediction, where it was ranked outside the top 10 on the MAE, and for the HUI3 prediction, where it was ranked fifth on both the RMSE and MAE). In the validation sample, GLM (8) overpredicted mean utilities whose range (minimum to maximum) was also narrower than that of the observed utilities (Table 3). Although the range of utilities predicted by GLM (8) was again narrower than that of the observed utilities in the estimation sample, the mean predicted and observed utilities were the same (Table 3). However, an examination of the measures of spread (particularly the 25th percentile, median and 75th percentile) shows that the distributions of predicted utilities in both samples were similar to those for the observed utilities. Spearman’s rank correlations between GLM (8) predicted and observed utilities in both samples all showed moderate correlation (range 0.51–0.66). Finally, the plots of residuals for GLM (8) (Fig. 4) for comparable predictions in the estimation and validation samples look significantly different but appear close to being normally distributed. Including country dummies in all model specifications within the sensitivity analysis did not result in better-performing models (predictive accuracy results of the 10 best-fitting models across all MAUIs are shown in electronic supplementary Table 3). On this basis, and in order to have parsimonious prediction models, preference was given to models without country dummies. In particular, GLM (8) was chosen as relatively best-fitting in both samples, and then re-estimated using data from the entire sample. The regression model coefficients for predicting the five MAUIs using GLM (8) are shown in Table 4. To predict 15D utilities from the AQLQ-S, for instance, the following equation would have to be used:
4 Discussion
The AQLQ-S is a non-preference-based measure of QoL for people with asthma, frequently used in clinical and epidemiological studies in Australia and internationally [6]. As it is not a utility instrument, it cannot be used for comparisons between interventions for disparate services. This limitation is overcome by mapping the AQLQ-S onto an MAUI. The estimated utilities from the mappings may then be used to calculate QALYs, and for the conduct of CUA. The present study has provided such mapping functions for each of the major MAUIs. As there were slight differences in the estimated utilities, the choice of which MAUIs to map onto must be guided by whether the health-state classification system of each MAUI reflects the domains deemed most important for the condition under consideration.
The AQLQ-S demonstrated strong positive association with all the MAUIs, implying good convergent validity between them. In the preliminary analysis, correlation was highest between the AQLQ-S and the 15D, and lowest between the AQLQ-S and the HUI3. The strong correlation with the 15D is a reflection of the close correspondence of the conceptualisation of the dimensions of health in the two instruments [77].
There were some mixed results among the regression algorithms for the best-predictive models (assessed according to correlation, RMSE, MAE and percentage of absolute differences between predicted and observed utilities of <0.05). The range of these statistics (0.0834–0.0973, RMSE; 0.0645–0.0740, MAE; 0.6420–0.6610, correlation; and 46–49 % of absolute differences between predicted and observed utilities of <0.05) for these models were all within acceptable ranges of published estimates [17], making the selection of the optimal algorithms for each MAUI difficult; however, differences between these models were small. It was not possible to compare our results with those of comparable analyses as our study was the first to map the AQLQ-S onto MAUIs, and the first to provide mappings for all of the major MAUIs. However, the RMSE estimates obtained in this study were substantially lower than those reported by Tsuchiya et al. [20] for the mapping of the AQLQ-McMaster onto the EQ-5D-3L (range 0.2024–0.2775).
For economic evaluation, mean estimates are of great importance [75, 78, 79]. Using this criterion, our results show that the OLS and GLM performed best as they predicted mean scores that were closest to the mean observed utilities. However, if an analyst is also interested in accurate prediction across the whole distribution of utilities, then the performance of the CLAD was the best because, compared with the OLS and GLM, CLAD models predicted mean utilities that had a wider range (minimum to maximum) that more closely described the variation of observed utilities, implying that CLAD-predicted utilities had a better spread of predicted values than the OLS and GLM models. This result has also been seen elsewhere [74, 75]. Generally though, all three model families predicted utilities with narrower ranges than those of observed utilities, a result seen in other research [68, 74, 79], and may have been due to few patients having scores or utilities at the lower or upper scales of the instruments used in this study.
Some limitations in our data and analysis need to be noted. First, no suitable out-of-sample dataset was available and therefore in-sample validation of mapping algorithms, successfully used in a number of other mapping studies [68–71], was applied; However, it is desirable that the algorithms should be validated on an external dataset. Second, asthma status was self-reported and therefore subject to reporting biases. Third, our sample may not be fully representative of the asthma population as we used a self-selected sample of respondents, namely people who used the internet and were part of the online database of CINT Pty Ltd; however, there are no strong prior reasons for believing that this should skew the functional relationships reported here. The sample included a wide representation across six countries, and the study participants were reflective of a broad range of sociodemographic characteristics. Finally, the same preference weights were used regardless of the nationality of the respondents as national weights do not exist for all of the MAUIs. However, it has been shown that the content of MAUIs has a greater impact on utilities than the difference in intercountry preference weights [34].
5 Conclusions
Directly collecting data on utilities will always be the best way of measuring QoL for the purpose of conducting a CUA. When this has not been done, our results demonstrate the possibility of predicting utilities if data on the AQLQ-S have been collected. We recommend using a GLM (8) mapping function for this exercise.
References
Braman SS. The global burden of asthma. Chest. 2006;130(1 Suppl):4s–12s.
Global Initiative for Asthma. Global strategy for asthma management and prevention. Global Initiative for Asthma; 2015.
Global Initiative for Asthma. Pocket guide for asthma management and prevention (for adults and children older than 5 years). Global Initiative for Asthma; 2015.
Eberhart NK, Sherbourne CD, Edelen MO, Stucky BD, Sin NL, Lara M. Development of a measure of asthma-specific quality of life among adults. Qual Life Res. 2014;23(3):837–48.
Apfelbacher C, Paudyal P, Bulbul A, Smith H. Measurement properties of asthma-specific quality-of-life measures: protocol for a systematic review. Syst Rev. 2014;3:83.
Apfelbacher CJ, Hankins M, Stenner P, Frew AJ, Smith HE. Measuring asthma-specific quality of life: structured review. Allergy. 2011;66(4):439–57.
Norman G, Faria R, Paton F, Llewellyn A, Fox D, Palmer S, et al. Omalizumab for the treatment of severe persistent allergic asthma: a systematic review and economic evaluation. Health Technol Assess. 2013;17(52):1–342.
Frew E, Hankins M, Smith HE. Patient involvement in the development of asthma-specific patient-reported outcome measures: a systematic review. J Allergy Clin Immunol. 2013;132(6):1434–6.
Australian Centre for Asthma Monitoring. Measuring the impact of asthma on quality of life in the Australian population. Canberra: Australian Institute of Health and Welfare; 2004.
Buxton MJ. Economic evaluation and decision making in the UK. Pharmacoeconomics. 2006;24(11):1133–42.
Harris A, Bulfone L. Getting value for money: the Australian experience. In: Jost TS, editor. Health care coverage determinations: an international comparative study. Maidenhead: Open University Press; 2004.
National Institute for Health and Care Excellence. Guide to the methods of technology appraisal 2013. London: National Institute for Health and Care Excellence; 2013.
Morris S, Devlin N, Parkin D. Economic analysis in health care. Chichester: Wiley; 2007.
Drummond MF, Sculpher M, O’Brien B, Stoddart GL, Torrance GW. Methods for the economic evaluation of health care programmes. Oxford: Oxford University Press; 2005.
Brazier J, Ratcliffe J, Salomon J, Tsuchiya A. Measuring and valuing health benefits for economic evaluation. Oxford: Oxford University Press; 2007.
Richardson J, McKie J, Bariola E. Multi attribute utility instruments and their use. In: Culyer AJ, editor. Encyclopedia of health economics. San Diego: Elsevier Science; 2014. p. 341–57.
Brazier JE, Yang Y, Tsuchiya A, Rowen DL. A review of studies mapping (or cross walking) non-preference based measures of health to generic preference-based measures. Eur J Health Econ. 2010;11(2):215–25.
Marks GB, Dunn SM, Woolcock AJ. A scale for the measurement of quality of life in adults with asthma. J Clin Epidemiol. 1992;45(5):461–72.
Marks GB, Dunn SM, Woolcock AJ. An evaluation of an asthma quality of life questionnaire as a measure of change in adults with asthma. J Clin Epidemiol. 1993;46(10):1103–11.
Tsuchiya A, Brazier J, McColl E, Parkin D. Deriving preference-based single indices from non-preference based condition-specific instruments: converting AQLQ into EQ5D indices. Ref. 02/1. Sheffield Health Economics Group Discussion Paper Series. 2002.
Richardson J, Khan MA, Iezzi A, Maxwell A. Measuring the sensitivity and construct validity of six utility instruments in seven disease states. Med Decis Making. 2016;36(2):147–59.
Richardson J, Iezzi A, Khan MA, Maxwell A. Cross-national comparison of twelve quality of life instruments. MIC paper 1: background, questions, instruments. Research paper 76. Melbourne, VIC: Centre for Health Economics, Monash University; 2012.
Petrou S, Rivero-Arias O, Dakin H, Longworth L, Oppe M, Froud R, et al. The MAPS reporting statement for studies mapping onto generic preference-based outcome measures: explanation and elaboration. Pharmacoeconomics. 2015;33(10):993–1011.
Spilker B. Quality of life and pharmacoeconomics in clinical trials. Philadelphia: Lippincott-Raven; 1996.
Katz PP, Eisner MD, Henke J, Shiboski S, Yelin EH, Blanc PD. The Marks Asthma Quality of Life Questionnaire: further validation and examination of responsiveness to change. J Clin Epidemiol. 1999;52(7):667–75.
Ware JE Jr, Kemp JP, Buchner DA, Singer AE, Nolop KB, Goss TF. The responsiveness of disease-specific and generic health measures to changes in the severity of asthma among adults. Qual Life Res. 1998;7(3):235–44.
Bayliss MS, Espindle DM, Buchner D, Blaiss MS, Ware JE. A new tool for monitoring asthma outcomes: the ITG Asthma Short Form. Qual Life Res. 2000;9(4):451–66.
Hawthorne G, Richardson J, Osborne R. The Assessment of Quality of Life (AQoL) instrument: a psychometric measure of health-related quality of life. Qual Life Res. 1999;8(3):209–24.
Richardson J, Hawthorne G. The Australian quality of life (AQoL) instrument: psychometric properties of the descriptive system and inital validation. Aust Stud Health Service Adm. 1998;85:315–42.
Richardson J, Khan MA, Chen G, Iezzi A, Maxwell A. Population norms and Australian profile using the Assessment of Quality of Life (AQoL) 8D Utility Instrument. Melbourne: Centre for Health Economics, Monash University; 2012.
Hawthorne G, Korn S, Richardson J. Population norms for the AQoL derived from the 2007 Australian National Survey of Mental Health and Wellbeing. Aust NZ J Public Health. 2013;37(1):7–16.
Richardson J, Sinha K, Iezzi A, Khan MA. Modelling utility weights for the Assessment of Quality of Life (AQoL)-8D. Qual Life Res. 2014;23(8):2395–404.
Richardson J, Iezzi A, Khan MA, Maxwell A. Validity and reliability of the Assessment of Quality of Life (AQoL)-8D multi-attribute utility instrument. Patient. 2014;7(1):85–96.
Richardson J, Khan MA, Iezzi A, Maxwell A. Comparing and explaining differences in the magnitude, content, and sensitivity of utilities predicted by the EQ-5D, SF-6D, HUI 3, 15D, QWB, and AQoL-8D multiattribute utility instruments. Med Decis Making. 2015;35(3):276–91.
Richardson J, Chen G, Khan MA, Iezzi A. Can multi attribute utility instruments adequately account for subjective wellbeing? Med Decis Making. 2015;35(3):292–304.
Cheung K, Oemar M, Oppe M, Rabin R. EQ-5D user guide: basic information on how to use EQ-5D—Version 2.0. Rotterdam: EuroQoL Group; 2009.
Herdman M, Gudex C, Lloyd A, Janssen M, Kind P, Parkin D, et al. Development and preliminary testing of the new five-level version of EQ-5D (EQ-5D-5L). Qual Life Res. 2011;20(10):1727–36.
Devlin N, Shah K, Feng Y, Mulhern B, Hout B. Valuing health-related quality of life: an EQ-5D-5L value set for England. Research paper 16/01. Office of Health Economics; 2016.
Kind P, Hardman G, Macran S. UK population norms for EQ-5D: discussion paper 172. York: University of York, Centre for Health Economics; 1999.
Janssen M, Pickard A, Golicki D, Gudex C, Niewada M, Scalone L, et al. Measurement properties of the EQ-5D-5L compared to the EQ-5D-3L across eight patient groups: a multi-country study. Qual Life Res. 2013;22(7):1717–27.
Conner-Spady BL, Marshall DA, Bohm E, Dunbar MJ, Loucks L, Khudairy AA, et al. Reliability and validity of the EQ-5D-5L compared to the EQ-5D-3L in patients with osteoarthritis referred for hip and knee replacement. Qual Life Res. 2015;24(7):1775–84.
Golicki D, Niewada M, Buczek J, Karlinska A, Kobayashi A, Janssen MF, et al. Validity of EQ-5D-5L in stroke. Qual Life Res. 2015;24(4):845–50.
Horsman J, Furlong W, Feeny D, Torrance G. The Health Utilities Index (HUI): concepts, measurement properties and applications. Health Qual Life Outcomes. 2003;1:54.
Feeny D, Furlong W, Torrance GW, Goldsmith CH, Zhu Z, DePauw S, et al. Multiattribute and single-attribute utility functions for the health utilities index mark 3 system. Med Care. 2002;40(2):113–28.
Moy ML, Fuhlbrigge AL, Blumenschein K, Chapman RH, Zillich AJ, Kuntz KM, et al. Association between preference-based health-related quality of life and asthma severity. Ann Allergy Asthma Immunol. 2004;92(3):329–34.
McTaggart-Cowan HM, Marra CA, Yang Y, Brazier JE, Kopec JA, FitzGerald JM, et al. The validity of generic and condition-specific preference-based instruments: the ability to discriminate asthma control status. Qual Life Res. 2008;17(3):453–62.
Ware JE Jr, Sherbourne CD. The MOS 36-item short-form health survey (SF-36). I: conceptual framework and item selection. Med Care. 1992;30(6):473–83.
Brazier JE, Ratcliffe J, Salomon J, Tsuchiya A. Measuring and valuing health benefits for economic evaluation. Oxford: Oxford University Press; 2007.
Brazier J, Roberts J, Deverill M. The estimation of a preference-based measure of health from the SF-36. J Health Econ. 2002;21(2):271–92.
Harrison M, Davies L, Bansback N, McCoy M, Verstappen S, Watson K, et al. The comparative responsiveness of the EQ-5D and SF-6D to change in patients with inflammatory arthritis. Qual Life Res. 2009;18(9):1195–205.
Kontodimopoulos N, Pappa E, Papadopoulos A, Tountas Y, Niakas D. Comparing SF-6D and EQ-5D utilities across groups differing in health status. Qual Life Res. 2009;18(1):87–97.
Goncalves Campolina A, Bruscato Bortoluzzo A, BosiFerraz M, Mesquita Ciconelli R. Validity of the SF-6D index in Brazilian patients with rheumatoid arthritis. Clin Exp Rheumatol. 2009;27(2):237–45.
Sintonen H. The 15D instrument of health-related quality of life: properties and applications. Ann Med. 2001;33(5):328–36.
Sintonen H. The 15D-measure of health-related quality of life. I: reliability, validity and sensitivity of its health state descriptive system. Melbourne: National Centre for Health Program Evaluation; 1994.
Haapaniemi TH, Sotaniemi KA, Sintonen H, Taimela E. The generic 15D instrument is valid and feasible for measuring health related quality of life in Parkinson’s disease. J Neurol Neurosurg Psychiatry. 2004;75(7):976–83.
Linde L, Sorensen J, Ostergaard M, Horslev-Petersen K, Hetland ML. Health-related quality of life: validity, reliability, and responsiveness of SF-36, 15D, EQ-5D [corrected] RAQoL, and HAQ in patients with rheumatoid arthritis. J Rheumatol. 2008;35(8):1528–37.
StataCorp LP. Intercooled Stata 131 for windows. College Station: StataCorp LP; 2014.
Rumsey DJ. Statistics II for dummies. Hoboken: Wiley Publishing, Inc; 2009.
Gray AM, Rivero-Arias O, Clarke PM. Estimating the association between SF-12 responses and EQ-5D utility values by response mapping. Med Decis Making. 2006;26(1):18–29.
Long JS. Regression models for categorical and limited dependent. A volume in the Sage Series for Advanced Quantitative Techniques. Thousand Oaks: Sage Publications; 1997.
Chay KY, Powell JL. Semiparametric censored regression models. J Econ Perspect. 2001;15(4):29–42.
Johnston J, DiNardo J. Econometric methods. London: The McGraw-Hill Companies, Inc; 1997.
McCullagh P, Nelder JA. Generalized linear models. 2nd ed. London: Chapman & Hall; 1989.
Manning WG. The logged dependent variable, heteroscedasticity, and the retransformation problem. J Health Econ. 1998;17(3):283–95.
Briggs A, Claxton K, Sculpher M. Decision modelling for health economic evaluation. Oxford: Oxford University Press; 2006.
Ospina R, Ferrari SLP. A general class of zero-or-one inflated beta regression models. Comput Stat Data Anal. 2012;56(6):1609–23.
Khan I, Morris S. A non-linear beta-binomial regression model for mapping EORTC QLQ-C30 to the EQ-5D-3L in lung cancer patients: a comparison with existing approaches. Health Qual Life Outcomes. 2014;12(1):1–16.
Longworth L, Yang Y, Young T, Mulhern B, Hernández Alava M, Mukuria C, et al. Use of generic and condition-specific measures of health-related quality of life in NICE decision-making: a systematic review, statistical modelling and survey. Health Technol Assess. 2014;18(9):1–224.
Brennan D, Spencer AJ. Mapping oral health related quality of life to generic health state values. BMC Health Serv Res. 2006;6(1):96.
Sauerland S, Weiner S, Dolezalova K, Angrisani L, Noguera CM, Garcia-Caballero M, et al. Mapping utility scores from a disease-specific quality-of-life measure in bariatric surgery patients. Value Health. 2009;12(2):364–70.
Bansback N, Marra C, Tsuchiya A, Anis A, Guh D, Hammond T, et al. Using the health assessment questionnaire to estimate preference-based single indices in patients with rheumatoid arthritis. Arthritis Rheum. 2007;57(6):963–71.
Daniel WW, Terrell JC. Business statistics. Boston: Houghton Mifflin Company; 1995.
Chai T, Draxler RR. Root mean square error (RMSE) or mean absolute error (MAE)? Arguments against avoiding RMSE in the literature. Geosci Model Dev. 2014;7(3):1247–50.
Cheung YB, Thumboo J, Gao F, Ng GY, Pang G, Koo WH, et al. Mapping the English and Chinese versions of the Functional Assessment of Cancer Therapy-General to the EQ-5D utility index. Value Health. 2009;12(2):371–6.
Kaambwa B, Billingham L, Bryan S. Mapping utility scores from the Barthel index. Eur J Health Econ. 2013;14(2):231–41.
Dakin H, Petrou S, Haggard M, Benge S, Williamson I. Mapping analyses to estimate health utilities based on responses to the OM8-30 Otitis Media Questionnaire. Qual Life Res. 2010;19(1):65–80.
Brazier J, Rowen D. Alternatives to EQ-5D for generating health state utility values. Contract no. 11. Sheffield: Decision Support Unit, School of Health and Related Research, University of Sheffield; 2011.
Khan KA, Petrou S, Rivero-Arias O, Walters SJ, Boyle SE. Mapping EQ-5D utility scores from the PedsQL generic core scales. Pharmacoeconomics. 2014;32(7):693–706.
Pinedo-Villanueva RA, Turner D, Judge A, Raftery JP, Arden NK. Mapping the Oxford hip score onto the EQ-5D utility index. Qual Life Res. 2013;22(3):665–75.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This work was supported through an Australian National Health and Medical Research Council (NHMRC) project Grant (Grant Number 1006334).
Contribution of authors
Jeff Richardson contributed to the study inception and writing of the NHMRC grant application. Billingsley Kaambwa analysed the data, interpreted the results, and wrote the first draft of the manuscript. Julie Ratcliffe, Gang Chen, Angelo Iezzi, Aimee Maxwell, and Jeff Richardson contributed to the interpretation of results and revision of the manuscript. All authors have read and approved the final manuscript. Billingsley Kaambwa is the guarantor of the manuscript.
Conflict of interest
Billingsley Kaambwa, Gang Chen, Julie Ratcliffe, Angelo Iezzi, Aimee Maxwell, and Jeff Richardson declare that they have no conflicts of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Ethical approval was granted by the MUHREC (CF11/3192–2011001748).
Informed consent
Informed consent was obtained from all individual participants included in the study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kaambwa, B., Chen, G., Ratcliffe, J. et al. Mapping Between the Sydney Asthma Quality of Life Questionnaire (AQLQ-S) and Five Multi-Attribute Utility Instruments (MAUIs). PharmacoEconomics 35, 111–124 (2017). https://doi.org/10.1007/s40273-016-0446-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40273-016-0446-4