Abstract
Background
Accurate assessment of preference-based health-related quality of life is important in determining the value of asthma interventions.
Objective
To examine the sensitivity and responsiveness of the EQ-5D and the AQL-5D to differences in asthma control measured by the Asthma Control Questionnaire (ACQ-5).
Methods
The Observational Study of Asthma Control and Outcomes was a prospective survey of persistent asthma patients ≥12 years old in Kaiser Colorado. Patients received a survey three times in 1 year, including the ACQ-5, AQL-5D and EQ-5D-3L (including VAS). Censored Least Absolute Deviations (CLAD) and logistic regression were used, controlling for sociodemographics and smoking.
Results
There were 6666 completed surveys (1799 individuals completed all three survey waves). After controlling for covariates, each one-point increase in ACQ-5 was associated with a decrease of 0.066, 0.058, 0.074 and 6.12 in EQ-5D(US), EQ-5D(UK), AQL-5D and VAS scores. Uncontrolled asthma (ACQ-5 > 1.5) was associated with a decrease of 0.15, 0.17, 0.11 and 10, respectively (vs. ACQ ≤ 1.5). AQL-5D scores were statistically significantly different across categories of ACQ-5 scores of 0.5 (the minimum clinically important difference [MCID]), while EQ-5D scores were not significant across most categories. The AQL-5D appeared more robust to changes in control over time (responsiveness) compared to EQ-5D-3L.
Conclusion
The AQL-5D appears more responsive to changes in asthma control over time and more sensitive to detecting differences corresponding to the ACQ-5 MCID than the EQ-5D-3L. Using the EQ-5D-3L without an asthma-specific measure such as the AQL-5D may miss clinically important changes in asthma control.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
Many studies document the negative effect of poor asthma control on health-related quality of life (HRQoL) [1–4]. The National Asthma Education and Prevention Program guidelines underscore the importance of asthma control on HRQL and as a goal of treatment [5–7]. Instruments such as the Asthma Control Questionnaire (ACQ) have been developed to measure asthma control [8].
In order to implement asthma treatments or disease management interventions, payers (insurance companies, governments, etc.) must find adequate value associated with the intervention. Payers use preference-based HRQoL (pbHRQoL) with survival to evaluate the relative value of asthma interventions. In the United Kingdom, for example, the National Institute for Health and Clinical Effectiveness (NICE) requires a comprehensive cost-utility analysis before the National Health Service (NHS) will pay for the use of treatments or procedures. Similar requirements exist in Australia, Canada and many other countries, particularly in Europe. Many payers in the USA also incorporate cost-effectiveness analyses in their reimbursement decision-making process.
Measuring treatment-related changes in HRQoL over time requires an instrument that is responsive and interpretable (able to detect an important and clinically meaningful change over time) [9]. Previous research comparing asthma-specific and generic measures of pbHRQoL has been based on cross-sectional associations, not longitudinal changes over time [10–12]. While these analyses are informative in assessing instrument validity, they are not directly applicable to assessing an instrument’s ability to measure meaningful changes in asthma control over time. Most cost-effectiveness studies of asthma interventions are limited in that they are forced to assume that cross-sectional relationships translate to longitudinal changes (i.e., that relationships across individuals would be similar to relationships observed for within person changes over time). Given that asthma interventions are aimed to improve control within a person over time, assessing responsiveness of pbHRQoL instruments is integral.
The purpose of this analysis is to: (1) compare the sensitivity and responsiveness of the EuroQol five-dimensional questionnaire (EQ-5D) and the five-dimensional Asthma Quality of Life Questionnaire (AQL-5D) to distinguish between asthma control states; (2) examine the impact of asthma control on specific domains and (3) quantify the magnitude of utility estimates associated with asthma control states.
Methods
Data source
The Observational Study of Asthma Control and Outcomes (OSACO) was a prospective survey of patients with persistent asthma in Kaiser Permanente of Colorado (KPCO) [13]. KPCO is a group model, closed panel, nonprofit HMO providing health care services to more than 625,000 members. Eligible individuals ≥12 years of age were sent surveys during three waves over 1 year: April–August 2011, September–December 2011 and March–June 2012. The study design included three waves in 1 year to capture potential seasonal variation in asthma symptoms. Subsequent surveys were only sent to patients who completed the previous wave. For each wave, patients who did not respond after two attempted mailings were contacted by phone to complete the survey. The OSACO study was approved by the Kaiser Colorado and Regis University Institutional Review Boards. Adults and parents of adolescents gave consent to participate in the study.
In addition to the EQ-5D, AQL-5D and ACQ-5, the survey included questions on asthma control, exacerbations, smoking status, sex, family income level, race, educational attainment and ethnicity. Exacerbations were ascertained by asking: “In the last 6 weeks, have you experienced serious asthma symptoms requiring you to take oral steroid medications?”
Instruments
Asthma control
Asthma control was assessed by the Asthma Control Questionnaire (ACQ-5) [8]. Patients were asked to recall how their asthma has been during the previous week and to respond to the questions on a 7-point scale (0 = no impairment, 6 = maximum impairment). The ACQ-5 asks patients about asthma symptoms: nighttime awakening, morning symptoms, activity limitations, shortness of breath and wheeze. Questions are equally weighted—the final score is the mean of all questions between 0 (totally controlled) and 6 (severely uncontrolled). The ACQ has been shown to have strong discriminative properties in both detecting differences between patients with different levels of asthma control and within-patient change in asthma control over time [14, 15.] The self-administered adult version of the ACQ has been validated in children 11 years and older [16].
A change or difference of 0.5 is considered by the developer to be the MCID [17]. In the current analysis, we explored a variety of ACQ-5 cut points to assess the sensitivity of the pbHRQoL instruments to distinguish between levels of control. Previous research has suggested a few potential cut points for the ACQ. The developers of the ACQ argue that to be confident that a patient has well-controlled asthma, the optimal cut point is 0.75 [18]. Alternatively, the authors recommend using a cut point of 1.5 to be confident that the patient has inadequately controlled asthma. Sastre et al. estimated the cut point between controlled and uncontrolled asthma was approximately 0.83 for the ACQ without lung function data. The authors also note that an ACQ score between 0.83 and 1.5 indicates some level of control and suggest that a hard cut point for uncontrolled asthma is closer to 1.5. Based on this literature, the current research examined the sensitivity of pbHRQoL instruments to distinguish between controlled and uncontrolled asthma using three potential cut points: 0.75, 0.83 and 1.5.
EQ-5D
The EQ-5D-3L is a five-item descriptive system measuring 5 dimensions of health status (mobility, self-care, usual activities, pain/discomfort and anxiety/depression) with 3 levels per dimension (no problem, some problems and extreme problems) based on the patient’s current health state [19]. Preferences for the different health states described by the EQ-5D are elicited from a general population sample from which a scoring algorithm is created to define a preference-based score (“utility”) for each possible health state [20.] The EQ-5D has been extensively validated as a generic pbHRQoL instrument in a variety of conditions and populations in over 6000 publications [21.] In addition, Pickard et al. [22] have reviewed the evidence of the construct validity, test–retest reliability and responsiveness of the EQ-5D in asthma. The EQ-5D-3L has been used in at least 29 studies including children [23]. Initial studies by Hennessy and Kind reported adequate performance of the EQ-5D in adolescents aged 12–18 years [24]. Subsequent development of the child-specific version of the EQ-5D, the EuroQol child-friendly version EQ-5D-Y (youth), was focused on children aged 7–12 years [25]. The current research used the adult version of the EQ-5D-3L.
This research included the EQ-5D tariffs for the USA [20] and the U.K. [26]. Because the AQL-5D is based on U.K. preferences, the EQ-5D scores based on the UK preferences would be more comparable theoretically than the EQ-5D U.S. tariffs. In addition, the EQ-5D VAS scores provided are based on a different preference elicitation method and may not be directly comparable.
AQL-5D
The Asthma Quality of Life Utility Index (AQL-5D) is a five-item asthma-specific “utility” instrument derived from the mini Asthma Quality of Life Questionnaire (mAQLQ). The mAQLQ is a disease-specific HRQL instrument that measures 15 items derived from the original 32-item AQLQ developed by Juniper [27]. The mAQLQ has 4 domains including symptoms, activity limitation, emotional function and environmental stimuli. The patient recall time horizon for the mAQLQ is the previous 2 weeks. The AQL-5D consolidates items of the mAQLQ to derive an asthma-specific health status classification system resulting in five domains: concern about asthma, shortness of breath, pollution, sleep and activity [28]. There are seven possible levels per domain ranging from “none of the time” to “all of the time”. The “utility” scoring function for the AQL-5D was derived by asking a general population in the U.K. to rate their preferences for asthma-specific health states derived from the AQL-5D questionnaire [29, 30]. Previous research has documented the validity of the AQL-5D in asthma [10, 29, 30]. The AQLQ was modified for adolescents and adults in the revised AQLQ12+ [31]. Although only one word was changed to derive the AQLQ12+, the mAQLQ has not been validated in adolescent populations.
Statistical analysis
Study variables for this analysis included asthma control, pbHRQoL, exacerbations and sociodemographic characteristics. Unadjusted mean and standard deviations for all instruments were calculated across ACQ-5 levels.
Censored least absolute deviations estimator (CLAD) was used for all pbHRQoL scores (except the VAS) because of their unique statistical properties. The reasoning and justification for using CLAD have been extensively discussed elsewhere [32]. Briefly, because of clustering of a high number of individuals around 1.0 (called “censoring”), use of OLS results in biased estimates. Alternatives like Tobit are also problematic due to heteroskedasticity of errors. VAS scores in these data did not share the same complexities (no clustering at 100), and hence, OLS regression was used. For analyses using CLAD, standard errors were computed by bootstrapping with 130 iterations; this method allows for a robust calculation of standard errors [33].
Analyses were also conducted to examine the relative importance of asthma control on the different domains of the EQ-5D and AQL-5D. Dichotomous dependent variables were created for each EQ-5D and AQL-5D domain by collapsing all levels within a given domain into two (no reported problems or any reported problems). For example, level 1 (no problems) on the EQ-5D was categorized as 0 and levels 2 and 3 (some problems or extreme problems) were categorized as 1. Logistic regression was then conducted for each domain. The main independent variable was poorly controlled asthma (ACQ-5 > 1.5 vs. ACQ ≤ 1.5).
All of the cross-sectional regression analyses controlled for the following covariates: wave and group fixed effects (“group” differentiates between individuals who self-completed the survey on-site or by mail), age, sex, family income level, race, educational attainment, ethnicity and smoking status. The following series of cross-sectional regression analyses were conducted:
-
(1)
Dependent variable: pbHRQoL scores (continuous). Independent variable: ACQ-5 score (continuous). Results presented: Table 3.
-
(2)
Dependent variable: pbHRQoL scores (continuous). Independent variable: dichotomous—exacerbation—yes/no. Results presented: Table 3.
-
(3)
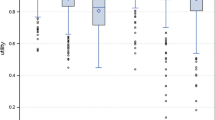
Dependent variable: pbHRQoL scores (continuous). Independent variable: ACQ-5 > cut point (e.g., >0.75 vs. not). Results presented: Fig. 1.
-
(4)
Dependent variable: pbHRQoL scores (continuous). Independent variable: ACQ-5 categories cut by 0.5 (ACQ-5 0–0.5 reference). Results presented: Fig. 2.
Fig. 2 HRQoL scores by ACQ-5 categories (cross-sectional; waves 1–3). Asterisk statistically significantly different than previous ACQ-5 score category (p < 0.05). Regression of HRQoL instrument on ACQ-5 categories (ACQ-5 0–0.5 reference) controlling for wave, group, age, gender, income, race, ethnicity, education and smoking
-
(5)
Logistic regression. Dependent variable: pbHRQoL questionnaire (binary—any reported problems vs no reported problems). Independent variable: (binary—ACQ-5 > 1.5 vs ≤1.5). Results presented: Fig. 3.
Fig. 3 Odds of Reporting Problems on HRQoL Questionnaires for ACQ-5 > 1.5 (compared to ACQ ≤ 1.5) (cross-sectional; waves 1–3). Odds of reporting any problems versus no problems for each domain of each questionnaire, comparing ACQ-5 > 1.5 versus ACQ ≤ 1.5. All odds ratios were statistically significant (p < 0.01). Logistic regression of reported problems for domain (any reported problems = 1) on uncontrolled asthma (ACQ-5 > 1.5) controlling for wave, group, age, gender, income, race, ethnicity, education and smoking
In order to examine the responsiveness of the respective instruments, analyses were conducted of the change in scores from one wave to the next. For the independent variables in the regressions, five categorical variables were created based on the ACQ-5 MCID: ACQ-5 Increase (0 < ACQ5 < 0.5); ACQ-5 Increase (ACQ5 ≥ 0.5); ACQ-5 Decrease (−0.5 < ACQ5 < 0); ACQ-5 Decrease (ACQ5 ≤ −0.5) and no change. The dependent variable was the change in pbHRQoL score (continuous), regressed on the aforementioned categorical independent variables, controlling for the covariates listed above (no change was the reference group).
Results
A total of 5951 individuals were eligible for the study. Of this eligible population, 2681 completed the first survey (Wave 1); 2186 also completed the second survey (Wave 2); and 1799 individuals completed all three surveys, for a total of 6666 completed surveys (Table 1). (There were 387 individuals who completed only two surveys and 495 who completed only one). Of the 6666 completed surveys, 1273 (19 %) were completed on-site. The mean ACQ-5 score was 0.99, with most scores falling between 0 and 1 (55 %) or 1 and 2 (30 %) and very few responses with ACQ-5 > 4.0. Mean EQ-5D(UK) scores are lower than the AQL-5D and EQ-5D US. The VAS (/100) has the lowest mean score of the four instruments.
Cross-sectional analyses
Unadjusted results from Table 2 show that pbHRQoL scores are associated with ACQ-5 scores, with poor control associated with lower scores on all measures. Adjusted results also demonstrate a strong association between poor asthma control and lower pbHRQoL. Table 3 shows the marginal decrement for each one-point increase in the ACQ-5, after controlling for covariates. AQL-5D scores appear to have the greatest magnitude association.
In adjusted analyses, all instruments appear sensitive to differentiating common cut points. Figure 1 shows that the dichotomous cut points indicating uncontrolled asthma (ACQ-5 > 0.75; >0.83 and >1.5) are associated with a greater magnitude decrement in EQ-5D than AQL-5D scores. The AQL-5D appears to be more sensitive to differences across the cut points compared to the EQ-5D(US) and EQ-5D(UK) and the direction is more consistent, with higher cut points resulting in lower scores (−0.086, −0.091, −0.110). The magnitude of decrement associated with exacerbation appears to be greatest for the EQ-5D(UK) (variability is also greatest with wide confidence intervals), while EQ-5D(US) and AQL-5D appear to be similar (Table 3).
Figure 2 shows the association between HRQL scores for categories of ACQ-5 scores separated by 0.5. The AQL-5D shows a statistically significant difference between even the mildest asthma control states such as 0–0.5 versus 0.5–1.0 and is able to differentiate between all ACQ-5 categories: The 95 % confidence bands show that there is a statistically significant difference between all categories except above ACQ-5 > 4.0 (where the sample size is very limited). It appears to have a robust and monotonically decreasing relationship with increasing categories of ACQ-5 scores. The EQ-5D(UK) and EQ-5D(US) fail to distinguish between very mild states such as 0–0.5 versus 0.5–1.0 and fail to differentiate between most ACQ-5 categories. They appear to have a more pronounced magnitude decrease associated with lower ACQ-5 scores; however, ACQ-5 1–1.5 scores are −0.12 and −0.14 for the EQ-5D(UK) and EQ-5D(US), respectively. Both show a significant drop at ACQ > 1.0 followed by a flatter decreasing relationship with subsequent categories of ACQ-5 scores compared to the other instruments. VAS scores are statistically significantly different for the mildest states, and appear to have a monotonically decreasing association with ACQ-5 scores, but scores are not statistically significantly different for ACQ-5 above 2.5.
The odds of reporting any problems (vs. no problems) on each questionnaire are shown in Fig. 3. The comparison is controlled vs. uncontrolled asthma (ACQ-5 > 1.5 vs. ACQ ≤ 1.5). For example, individuals with ACQ > 1.5 have 6 times higher odds of reporting some level of problems on the usual activities domain of the EQ-5D questionnaire than those with ACQ ≤ 1.5. The likelihood of reporting some problems appeared to be statistically significantly associated with uncontrolled asthma for all EQ-5D domains. Individuals with uncontrolled asthma were most likely to report problems on the usual activities and self-care domains of the EQ-5D. In contrast, individuals with uncontrolled asthma have much greater odds of reporting some level of problems on all five domains of the AQL-5D (odds ratios 5.7–48.5). Shortness of breath appeared to be the most sensitive domain to uncontrolled asthma, but domains of the AQL-5D appeared to be more sensitive to problems than EQ-5D domains in general.
Longitudinal analyses
Table 4 displays the results of the analysis of responsiveness, and Table 5 (appendix) presents the corresponding numbers of individuals for each change category. The AQL-5D appears to be the most robust, detecting statistically significant differences in ACQ-5 scores across all categories (compared to no change) except one. In addition, the confidence intervals of each category do not overlap, showing the AQL-5D capable of distinguishing statistically significant differences between all groups as well. The EQ-5D UK was able to differentiate between no change and changes greater than the MCID. However, improvements in asthma control meeting or exceeding the MCID (ACQ-5 change ≤−0.5) were not statistically significantly different than “no change” for W3–W2. The EQ-5D US appeared to be the least robust: Only decreases in the ACQ-5 from Wave 2 to Wave 1 and increases from Wave 2 to Wave 3 meeting/exceeding the MCID were statistically significant. Both the EQ-5D UK and EQ-5D US had overlapping confidence intervals, suggesting that these instruments were not capable of differentiating between the categories.
Discussion
Previous cross-sectional analyses have shown that generic measures such as the EQ-5D are able to distinguish between the highest and lowest levels of control but lack sensitivity for the middle [10]. The current research provides new information by examining the responsiveness over time and item-level functioning of these instruments, as well as their ability to discriminate across the MCID of the ACQ-5. Our results suggest that the AQL-5D is more responsive to changes in asthma control over time and is better able to differentiate between granular levels of control than the EQ-5D(UK) and EQ-5D(US).
Overall, all pbHRQoL instrument scores appeared to be associated with poorly controlled asthma and exacerbations. However, the AQL-5D appears to have greater construct validity in measuring pbHRQoL associated with asthma control than either EQ-5D index: The AQL-5D appears to be more sensitive to distinguishing statistically significant differences across the spectrum of ACQ-5 scores categorized by 0.5. In contrast, the EQ-5D(UK) and EQ-5D(US) scores were not statistically significantly different for comparisons of most categories of ACQ-5 scores.
There is some disagreement about whether asthma-specific pbHRQoL measures are more appropriate than generic measures to assess asthma interventions [10]. For example, NICE has explicitly published guidelines requiring the use of the EQ-5D for all cost-utility analyses. Others, however, have argued that disease-specific measures are more sensitive to improvements in health status in asthma. NICE may consider the use of an asthma-specific pbHRQoL instrument in place of the EQ-5D only if sufficient evidence demonstrates deficiency in the EQ-5D. The lack of responsiveness of the EQ-5D to changes in asthma control as well as its insensitivity to distinguish across the MCID of the ACQ-5 may limit its usefulness in assessing the cost-utility of asthma treatments. A treatment that improves asthma control over time may not show significant improvement in EQ-5D scores, which could result in allocation of resources away from asthma interventions and denial of reimbursement for treatments that improve control.
Unlike disease-specific instruments, however, generic instruments such as the EQ-5D have the benefit of consistency across diseases for informing decisions from the health system perspective. Use of varied disease-specific instruments may improve sensitivity within conditions but would lead to incomparability across diseases for a health system. Ideally, future generation of generic pbHRQoL instruments would include a respiratory-related domain or bolt-on. Currently, however, if the EQ-5D is used in cost-utility analyses, it seems prudent that an asthma-specific instrument such as the AQL-5D can be considered in addition.
The EQ-5D is a generic instrument and does not contain any respiratory-specific items or domains. The EQ-5D appears sensitive to crude differences between controlled and uncontrolled asthma. However, it is not well understood how the symptoms of poor asthma control impact EQ-5D domains. Results of this analysis show that all EQ-5D domains are sensitive to poorly controlled asthma, but the self-care and usual activities domains are most sensitive. In additional sensitivity analyses comparing ACQ-5 scores >3.5 (vs ≤3.5) on the odds of reporting some/any problems per EQ-5D domain, the self-care domain was by far the most likely to show problems (odds > 50) followed by usual activities (odds > 25). Future research should examine the nature of this relationship more specifically.
In contrast, the AQL-5D is an asthma-specific instrument. Our results show that poorly controlled individuals have a high likelihood of reporting problems on all domains of the AQL-5D. Shortness of breath appears to have the strongest association, followed by sleep and activity. In sensitivity analyses comparing ACQ-5 scores >3.5 (vs ≤3.5) on the odds of reporting some problems per AQL-5D domain, there was perfect association (meaning that all individuals who had ACQ > 3.5 reported at least some problems) for these three domains. This may also partially explain the stronger correlation between the AQL-5D and the ACQ-5. Both contain domains specific to shortness of breath, nighttime awakening and activity limitations.
The recall window differs across the three instruments. The EQ-5D asks patients to rate their current health state while the AQL-5D is based on the previous 2 weeks and the ACQ-5 is based on the previous week. It is possible that the different recall windows of the instruments could influence the correlation between scores and may even compromise the ability to compare these instruments. Asthma symptoms can be variable from 1 week to the next, and the potential direction of the bias due to different recall horizons is unclear.
In addition, the items within the AQL-5D may be correlated because they are all related to asthma HRQoL. If the items within the AQL-5D are correlated, the single summary score of the AQL-5D may be more sensitive to small changes in asthma control. This may contribute to greater sensitivity of the AQL-5D to changes in asthma control. The AQL-5D contains five levels per item, while the EQ-5D-3L contains only three levels per item. It is unclear whether the more recent five-level EQ-5D-5L would be more sensitive to changes in asthma control than the EQ-5D-3L used in this research.
The inclusion of adolescents aged ≥12 years may be a limitation in this study. Validation studies have shown the self-administered adult version of the ACQ to be valid in populations as young as 11 years old [16]. While the EQ-5D-3L has been shown to be valid in adolescents ≥12 years of age [24], there may be some limitation to the use of the adult version in our study among adolescents subjects. The m-AQLQ (and hence the AQL-5D), however, has not been well validated in adolescent populations.
The results of this study may not be generalizable to other populations. The sample in this study was less likely to be African American, Hispanic and poor compared to the U.S. general population. The inclusion of older individuals with potential comorbidities may also limit the results of this study. Regression analyses did not control for comorbidities. The existence of comorbidities may differentially impact pbHRQoL, and results should be interpreted with caution.
There is the potential for selection bias in this study. Individuals who were willing to participate in multiple surveys may be different than those not. The statistical analysis controlled for survey administration timing and group as well as explicitly controlling for sociodemographic characteristics. Patients ≥50 years of age prescribed ipratropium or tiotropium were excluded from this analysis in order to avoid misclassifying COPD patients as persistent asthma patients [13]. This, however, may have resulted in unintentionally excluding some asthma patients.
This research provides important insight into the specific relationship between asthma control and pbHRQoL. The EQ-5D and AQL-5D appear sensitive to differentiating between controlled and uncontrolled asthma and exacerbations. The AQL-5D appears more responsive to changes in asthma control over time and more sensitive to distinguishing across the MCID in ACQ-5 scores. Of the five EQ-5D domains, self-care and usual activities appear most sensitive to uncontrolled asthma. To fully capture the benefits of asthma treatments and interventions, it seems prudent to include both a generic measure like the EQ-5D and an asthma-specific measure like the AQL-5D.
References
Sullivan, P. W., Smith, K. L., Ghushchyan, V. H., Globe, D. R., Lin, S. L., & Globe, G. (2013). Asthma in USA: Its impact on health-related quality of life. Journal of Asthma, 50(8), 891–899.
Vollmer, W. M., Markson, L. E., O’Connor, E., Sanocki, L. L., Fitterman, L., Berger, M., et al. (1999). Association of asthma control with health care utilization and quality of life. American Journal of Respiratory and Critical Care Medicine, 160(5 Pt 1), 1647–1652.
Chen, H., Gould, M. K., Blanc, P. D., Miller, D. P., Kamath, T. V., Lee, J. H., et al. (2007). Asthma control, severity, and quality of life: Quantifying the effect of uncontrolled disease. Journal of Allergy and Clinical Immunology, 120(2), 396–402.
Siroux, V., Boudier, A., Anto, J. M., Cazzoletti, L., Accordini, S., Alonso, J., et al. (2008). Quality-of-life and asthma-severity in general population asthmatics: Results of the ECRHS II Study. Allergy, 63(5), 547–554.
Cockcroft, D. W., & Swystun, V. A. (1996). Asthma control versus asthma severity. Journal of Allergy and Clinical Immunology, 98(6 Pt 1), 1016–1018.
Pakhale, S., Mulpuru, S., & Boyd, M. (2011). Optimal management of severe/refractory asthma. Clinical Medicine Insights Circulatory, Respiratory and Pulmonary Medicine, 5, 37–47.
Rosenzweig, J. R. C., Edwards, L., Lincourt, W., Dorinsky, P., & ZuWallack, R. L. (2004). The relationship between health-related quality of life, lung function and daily symptoms in patients with persistent asthma. Respiratory Medicine, 98(12), 1157–1165.
Juniper, E. F., O’Byrne, P. M., Guyatt, G. H., Ferrie, P. J., & King, D. R. (1999). Development and validation of a questionnaire to measure asthma control. European Respiratory Journal, 14(4), 902–907.
Guyatt, G. H., Osoba, D., Wu, A. W., Wyrwich, K. W., & Norman, G. R. (2002). Methods to explain the clinical significance of health status measures. Mayo Clinic Proceedings, 77(4), 371–383.
McTaggart-Cowan, H. M., Marra, C. A., Yang, Y., Brazier, J. E., Kopec, J. A., FitzGerald, J. M., et al. (2008). The validity of generic and condition-specific preference-based instruments: the ability to discriminate asthma control status. Quality of Life Research, 17(3), 453–462.
Moy, M. L., Fuhlbrigge, A. L., Blumenschein, K., Chapman, R. H., Zillich, A. J., Kuntz, K. M., et al. (2004). Association between preference-based health-related quality of life and asthma severity. Annals of Allergy, Asthma & Immunology, 92(3), 329–334.
Nishimura, K., Oga, T., Ikeda, A., Hajiro, T., Tsukino, M., & Koyama, H. (2008). Comparison of health-related quality of life measurements using a single value in patients with asthma and chronic obstructive pulmonary disease. Journal of Asthma, 45(7), 615–620.
Sullivan, P. W., Campbell, J. D., Globe, G., Ghushchyan, V. H., Bender, B., Schatz, M., et al. (2015). Measuring the effect of asthma control on exacerbations and health resource use. Journal of Allergy and Clinical Immunology, 136(5), 1409.e6–1411.e6
Juniper, E. F., Buist, A. S., Cox, F. M., Ferrie, P. J., & King, D. R. (1999). Validation of a standardized version of the Asthma Quality of Life Questionnaire. Chest, 115(5), 1265–1270.
QOLTECH. Asthma Control Questionnaire 2010 [cited 2010 March 5 2010]. http://www.qoltech.co.uk/acq.html.
Juniper, E. F., Gruffydd-Jones, K., Ward, S., & Svensson, K. (2010). Asthma Control Questionnaire in children: validation, measurement properties, interpretation. European Respiratory Journal, 36(6), 1410–1416.
Juniper, E. F., Svensson, K., Mork, A. C., & Stahl, E. (2005). Measurement properties and interpretation of three shortened versions of the asthma control questionnaire. Respiratory Medicine, 99(5), 553–558.
Juniper, E. F., Bousquet, J., Abetz, L., & Bateman, E. D. (2006). Identifying ‘well-controlled’ and ‘not well-controlled’ asthma using the Asthma Control Questionnaire. Respiratory Medicine, 100(4), 616–621.
Brooks, R. G., Jendteg, S., Lindgren, B., Persson, U., & Bjork, S. (1991). EuroQol: health-related quality of life measurement. Results of the Swedish questionnaire exercise. Health Policy, 18(1), 37–48.
Shaw, J. W., Johnson, J. A., Coons, S. J. (2005). U.S. valuation of the EQ-5D health states: development and testing of the D1 valuation model. Medical Care, 43(3), 203–220
The EuroQol Group. EQ-5D Reference Search 2016 [cited 2013 May 10]. http://www.euroqol.org/eq-5d-publications/search.html.
Pickard, A. S., Wilke, C., Jung, E., Patel, S., Stavem, K., & Lee, T. A. (2008). Use of a preference-based measure of health (EQ-5D) in COPD and asthma. Respiratory Medicine, 102(4), 519–536.
Noyes, J., & Edwards, R. T. (2011). EQ-5D for the assessment of health-related quality of life and resource allocation in children: a systematic methodological review. Value Health, 14(8), 1117–1129.
Hennessy, S., Kind, P. (2002). Measuring health status in children: Developing and testing a child-friendly version of EQ-5D. In 19th plenary meeting of the EuroQol Group; York, United Kingdom.
Wille, N., Badia, X., Bonsel, G., Burstrom, K., Cavrini, G., Devlin, N., et al. (2010). Development of the EQ-5D-Y: A child-friendly version of the EQ-5D. Quality of Life Research, 19(6), 875–886.
Dolan, P. (1997). Modeling valuations for EuroQol health states. Medical Care, 35(11), 1095–1108.
Juniper, E. F., Guyatt, G. H., Cox, F. M., Ferrie, P. J., & King, D. R. (1999). Development and validation of the Mini Asthma Quality of Life Questionnaire. European Respiratory Journal, 14(1), 32–38.
Young, T. A., Yang, Y., Brazier, J. E., & Tsuchiya, A. (2011). The use of rasch analysis in reducing a large condition-specific instrument for preference valuation: the case of moving from AQLQ to AQL-5D. Medical Decision Making, 31(1), 195–210.
Yang, Y., Brazier, J. E., Tsuchiya, A., & Young, T. A. (2011). Estimating a preference-based index for a 5- dimensional health state classification for asthma derived from the asthma quality of life questionnaire. Medical Decision Making, 31(2), 281–291
Yang, Y., Brazier, J. E., Tsuchiya, A., Young, T. A. (2002). Estimating a preference-based single index from the Asthma Quality of Life Questionnaire (AQLQ). Sheffield: Health Economics and Decision Science (HEDS) Section at the School of Health and Related Research (ScHARR), University of Sheffield, 07/02. Report No.
Juniper, E. F., Svensson, K., Mork, A. C., & Stahl, E. (2005). Modification of the asthma quality of life questionnaire (standardised) for patients 12 years and older. Health Qual Life Outcomes, 3, 58.
Sullivan, P. W., Lawrence, W. F., & Ghushchyan, V. (2005). A national catalog of preference-based scores for chronic conditions in the United States. Medical Care, 43(7), 736–749.
Efron, B., & Tibshirani, R. J. (1986). Bootstrap methods for standard errors, confidence intervals, and other measures of statistical accuracy. Statistical Science, 1, 54–77.
Acknowledgments
The authors thank Denise Globe, PhD for her valuable contribution to the conception and design of the study. Funding source: This study was funded by Amgen Inc.
Author information
Authors and Affiliations
Corresponding author
Appendix
Rights and permissions
About this article
Cite this article
Sullivan, P.W., Ghushchyan, V.H., Campbell, J.D. et al. Measurement of utility in asthma: evidence indicating that generic instruments may miss clinically important changes. Qual Life Res 25, 3017–3026 (2016). https://doi.org/10.1007/s11136-016-1357-8
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11136-016-1357-8