Abstract
The influence of the interaction between Phoma sp. GS8-2 (GS8-2) and the arbuscular mycorrhizal fungus Glomus mosseae (Gm) on the development of soil-borne diseases, microbial population, and plant growth in cucumber, bentgrass and tomato plants was investigated under controlled conditions. The composite inoculation of GS8-2 and Gm yielded varied effects on suppression of symptoms of soil-borne pathogens depending on the host-pathogen combination involved. The GS8-2 + Gm had neutral effect in reducing damping-off disease of cucumber caused by Rhizoctonia solani AG-4 and the crown and root rot disease of tomato caused by Fusarium oxysporum f. sp. radicis-lycopersici (Forl), but exacerbated the brown patch disease caused by R. solani AG2-2 in bentgrass. In tomato plants, Forl populations in the roots and rhizosphere were significantly reduced by GS8-2 + Gm. Results also show that the composite inoculation of GS8-2 and Gm resulted to a synergistic effect on the reduction of fungal and bacterial populations in roots and rhizosphere of the plants. Plant growth enhancement was due to the individual effect of GS8-2 or Gm but not their interaction. GS8-2 root colonization of cucumber and tomato plants decreased significantly in dual inoculated plants compared to plants inoculated with GS8-2 alone; while no significant differences were found in the Gm root colonization of the three plant species indicating that GS8-2 had no effect on Gm.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Most soil-borne pathogens are difficult to manage even with the conventional use of soil fumigants (Edwards et al. 1998; Défago and Keel 1995), and one chief alternative to fungicides is biocontrol. The need for biocontrol agents in modern crop production systems fuels the directed effort towards the search for more efficient biocontrol agents which can potentially replace these deleterious chemical pesticides. The use of mixes of two or more beneficial organisms, have long been studied as it offers an array of advantages over single beneficial organism, including efficacy and efficiency (Raupach and Kloepper 1998; Guetsky et al. 2001; de Boer et al. 2003; Roberts et al. 2005; Haggag and Nofal 2006). Among the beneficial microorganisms, arbuscular mycorrhizal fungi (AMF) are unique because they present a wide array of benefits to the plants. AM symbiosis is known to control soil-borne diseases (Azcón-Aguilar et al. 2002; Whipps 2004; Pozo et al. 2010) and has a well-established role in soil fertility and improvement of plant nutritional status (Jeffries and Barea 2001; Jeffries et al. 2003) through several mechanisms, including increased availability of plant nutrients and the increased uptake of nutrients that are usually immobile (Smith and Read 2008).
Akin to the plant growth-promoting rhizobacteria, the plant growth-promoting fungi (PGPF) are generally non-pathogenic soil inhabiting saprotrophs (Hyakumachi 1994) reported to promote growth and suppress several plant diseases upon root colonization in some plants (Shivanna et al. 1996, 2005; Koike et al. 2001; Hyakumachi and Kubota 2004; Chandanie et al. 2005, 2006; Horinouchi et al. 2007; Saldajeno and Hyakumachi 2011). Biocontrol of plant pathogens attained by the PGPF strains is believed to be mediated through several mechanisms including hyperparatism, antibiosis, competition and induction of systemic resistance (Hyakumachi and Kubota 2004). The PGPF Phoma sp. Saccardo are generally poor spore-formers (Chandanie et al. 2005) and the isolate GS8-2 is a poor root colonizer (Meera et al. 1995a) but has biocontrol activities against several root and foliar diseases (Koike et al. 2001; Meera et al. 1994; 1995a, b; Chandanie et al. 2006). Published studies on the interaction between beneficial rhizosphere microorganisms like the AMF and the PGPF is inadequate (Vázquez et al. 2000; Chandanie et al. 2006). A better understanding of the interaction effects between two or more beneficial microorganisms could lead to improved practical usage of beneficial microorganisms as biocontrol agents or biofertilizers. In this study, we hypothesize a synergistic effect of interaction between the PGPF Phoma sp. isolate GS8-2 and the AMF Glomus mosseae (Nicol. and Gerd.) Gerd. and Trappe (Gm) in their suppression of the symptom development of soil-borne diseases. To achieve this goal, we compositely inoculated 3 plant species (cucumber, bentgrass, and tomato) with a barley grain inoculum (BGI) of GS8-2 and a commercial formulation of Gm and examined its impact on disease development of two soil-borne pathogens. Moreover, the quantitative changes in the population of the other microorganisms and the co-inoculants in the roots and the rhizosphere of the host plants were determined and the shoot dry weights (SDW) of the host plants were measured.
Materials and methods
Host plants and fungal isolates
The PGPF isolate was GS8-2 maintained in potato dextrose agar (PDA) slants at the PGPF collection of the Laboratory of Plant Pathology, Gifu University, Japan. Inoculum production was prepared following the procedure described by Chandanie et al. (2005, 2006) and the ground BGI was stored at 4°C until use. The AMF utilized was Gm obtained from a commercial inoculum (Idemitsu Kosan Co. Ltd., Tokyo, Japan) containing spores, colonized root fragments and soil. Rhizoctonia solani Kühn AG4 HG-II isolate Mat 7 and R. solani AG2-2 IIIB isolate K2-5 were used as the damping-off and brown patch pathogens, respectively. Damping-off pathogen inoculum was prepared using barley grains following the same procedure previously described in preparing the GS8-2 inoculum with minor modifications. R. solani AG4 HG-II Mat 7 was cultured in PDA for 2 days prior to inoculation to sterile barley grains. The brown patch pathogen inoculum was fungal agar discs (6 mm diameter) prepared from the actively growing margin of a 7-day old PDA culture of R. solani AG2-2 IIIB K2-5. Fusarium oxysporum f. sp. radicis-lycopersici Jarvis and Shoemaker isolate KU11 (Forl) was used as the crown and root rot pathogen. Spore suspension was collected from a 7-day old PDA culture using autoclaved distilled water (sdH2O) and filtered through 3 layers of cheesecloth. The spore concentration was adjusted to 1 × 106 spores ml-1 using a haemocytometer. The host plants were cucumber (Cucumis sativus cv. Tokiwa Jibai), bentgrass (Agrostis palustris Hugus), and tomato (Lycopersicon esculentum cv. House Momotaro).
Experimental design, biological treatments, and growth conditions
The experiments were set up as a 2 by 2 factorial experiment with three replicates per treatment. All experiments were repeated at least thrice. The potting medium was blended with GS8-2 and/or Gm inocula (2% w/w) giving a total of four treatments for all experiments: (A) GS8-2 and Gm (GS8-2 + Gm); (B) GS8-2 alone (GS8-2); (C) Gm alone (Gm); and (X) Uninoculated control (Control). The non-mycorrhizal treatments (B and X) were amended with the same amount of autoclaved Gm inoculum, while non-Phoma treatments (C and X) got the same amount of autoclaved and milled barley kernels. The potting medium was a clay loam soil obtained from the experimental field of Gifu University, Yanagido, Gifu City, containing 0.10% total N and 18.6 mg/100 g available P, with a pH of 6.5 (soil analysis done by Tokachi Nokyoren Agricultural Research Institute, Sapporo, Japan). The soil was sieved with 2 mm mesh, autoclaved, and mixed (1:1 w/w) with sterile commercial river sand (Aichi Toyota Ishimura, Aichi Prefecture, Japan). Mineral nutrients were added to the soil-sand mixture as in Green et al. (1999) just before sowing. Similar initial microflora communities were established in non-mycorrhizal treatments with that of the mycorrhizal treatments (Calvet et al. 1993; Green et al. 1999), by adding AMF inoculum filtrate to each non-mycorrhizal pot. The filtrate was prepared according to Calvet et al. (1993), and the suspension was filtered through a 38 μm sieve to exclude AMF propagules. Plants were maintained in a growth chamber at 25°C, 75% relative humidity, and 12 h light (300 μE m-2 s-1) or at the greenhouse at 25°C, and 12 h light period until sampling.
Disease Suppression Experiments
-
1.
Inoculation of the GS8-2 and Gm and the challenge inoculation of the pathogens
Pre-germinated cucumber seeds were sown in paper pots (Nippon Beet Sugar Co. Ltd., Japan) (6.5 cm depth x 1.5 cm diameter) filled with potting medium inoculated with GS8-2 and/or Gm. The non-mycorrhizal and mycorrhizal pots were added with Gm inoculum filtrate and sdH2O at 1 ml/pot, respectively. The seedlings were grown for 10 days in the growth chamber under the conditions described above after which the seedlings with the potting medium were transplanted into 200 ml pots filled with the potting medium inoculated with the pathogen R. solani AG-4 HG II Mat 7 at 0.01% (w/w) inoculum density. The plants were then kept in the growth chamber for additional 7 days for symptom development.
For bentgrass, plastic pots (6 cm diameter x 7.8 cm depth) were filled with the thoroughly mixed potting medium and BGI of GS8-2 and/or Gm inoculum. The Gm inoculum filtrate and sdH2O were added as previously described at the rate of 5 ml pot-1 to the non-mycorrhizal and mycorrhizal pots, respectively. Thirty milligrams of bentgrass seeds were sown per pot and were grown for 6 weeks in a growth chamber with the same conditions previously described. Six-week old plants were trimmed to 5 mm height and an agar disc of R. solani AG2-2 IIIB K2-5 (6 mm diameter) was inserted in a hole (6 mm diameter x 5 mm depth) at the center of the pot. The plants were then kept in the growth chamber for another week for symptom development. In the case of tomato, pre-germinated tomato seeds were sown in plastic pots (6 cm diameter x 7.8 cm depth) filled with the potting medium and inocula mixtures. The AMF inoculum filtrate and sdH2O were added to the pots as described above. The seedlings were grown for 6 weeks in the greenhouse at 25°C and 12 h light period. After 6 weeks, a 10-ml spore suspension of Forl was added per pot and plants were maintained for another 4 weeks for symptom development.
-
2.
Disease Assessment
Disease severity (DS) assessments for cucumber, bentgrass and tomato were calculated based on the disease index (DI) as: DS = Sum of the DI/total number of plants, where DI was specific for each plant. In cucumber, DS assessment was done 7 days after transplanting in the pathogen-inoculated soil medium and the DI was measured on a 0–4 scale in which 0 = no lesion on hypocotyl (healthy seedling); 1 = lesions cover ≤10% of hypocotyl; 2 = lesions cover 11-50% of hypocotyl; 3 = lesions cover 51-90% of hypocotyl; 4 = dead seedling. For bentgrass, DS assessment was done 1 week after inoculation (7 weeks after planting, WAP) and the DI was measured on a 0–4 scale in which 0 = Intact (less than 5% browning or healthy seedling); 1= 5–25% browning; 2 = 26–50% browning; 3 = 51–75% browning; 4 = ≥75% browning. In tomato, 4 weeks after inoculation (10 WAP), DS was calculated based on the DI which was measured on a 0–4 scale in which 0 = no crown and root rot symptoms (healthy plants); 1 = ≤10% rotting of crown and roots; 2 = 11–30% rotting of crown and roots; 3 = 31–50% rotting of crown and roots; 4 = ≥ 50% rotting of crown and roots or dead plant.
The degree of disease suppression attained by inoculating the three different plant species with GS8-2 and/or Gm was calculated based on the DS as follows: Protection = [(A-B)/A] x 100% where A = DS in control plants and B = DS in treated plants.
Measuring growth enhancement, root colonization of co-inoculants, and microbial population
Pre-germinated cucumber or tomato seeds were sown in autoclaved 200 ml (6 cm diameter x 7.8 cm depth) plastic pots filled with 180 g potting medium inoculated with GS8-2 and/or Gm. For bentgrass, about 30 mg of bentgrass seeds were sown per pot. After growing cucumber plants for 4 weeks and bentgrass and tomato plants for 6 weeks, the plants were uprooted and the shoots separated from the roots. The shoots were oven-dried at 60°C for 48 h and the dry weights were measured. Root colonization by GS8-2 was evaluated utilizing the whole root samples. One cm long root segments previously surface disinfected with 0.5% NaOCl and rinsed with sdH2O were blotted dry on sterile filter paper just before plating. Dried roots were plated on PDA amended with chloramphenicol (250 mg l-1), and incubated for 2–3 days at 25°C. After incubation, GS8-2 colonies growing from root segments were identified and the isolation frequency was determined by counting the number of colonized segments.
The Gm root colonization was evaluated by staining 1 cm root samples with 0.05% chlorazol black E (CBE) (Brundrett et al. 1984). The percent root length colonized by hyphae, arbuscles and vesicles of Gm was determined using the magnified intersection method of McGonigle et al. (1990).
The microbial population in the roots and rhizosphere were determined by the dilution plate technique. Upon uprooting the plants, the roots were shaken gently to separate the bulk from the rhizosphere soil. The soil adhering to the roots was collected as rhizosphere soil and tenfold soil dilutions were prepared and plated on fungal selective Martin’s Rose Bengal medium (Martin 1950) and bacterial selective Tryptic Soy Agar (TSA) medium and incubated for 2–3 days at 25°C. After incubation, colonies were counted and population was quantified as fungal or bacterial colony-forming units (CFU) g-1 of dry soil. For quantifying microbial population in the roots, root segments were homogenized for 20 min at 8000 rpm (Type B-A; Teraoka Toyo Keisokuki, Osaka, Japan) and tenfold dilutions were plated on Martin’s Rose Bengal medium or TSA, and fungal and bacterial colonies were counted and population was quantified as CFU g-1 fresh roots after 2–3 days of incubation.
Measuring population of the soil-borne pathogens in the roots and rhizosphere of plants
The Forl population in the roots and the rhizosphere of tomato plants were determined following the same procedure previously described in determining the microbial population in the roots and the rhizosphere. The tenfold root and soil dilutions were plated on Komada medium (Komada 1975) and incubated for 2–3 days at 25°C after which Forl colonies were counted. In the case of R. solani in cucumber and bentgrass, we did not measure the pathogen population neither in the roots nor in the rhizosphere because of two limitations: the existence of several inoculum units in the soil or in the roots which makes quantification difficult and the lack of an effective quantitative medium for R. solani.
DNA extraction and denaturing gradient gel electrophoresis
To verify the microbial population from the roots and rhizosphere counted through the selective media, the fungal and bacterial community from rhizosphere soil of 4-week old cucumber plants was determined through the denaturing gradient gel electrophoresis (DGGE). Whole fungal and bacterial community DNA was extracted from 0.2 g rhizosphere soil samples utilizing the bead-beating technique using the Fast DNA spin kit for soil DNA extraction (Bio 101 Inc., Carlsbad, USA) according to the manufacturer’s protocol. Further DNA purification was carried out using the Geneclean II Kit (Bio 101, Inc.) and DNA were stored in TE buffer at −80°C until use. Amplification of the fungal and bacterial community DNA was done using primers that anneal to the conserved regions of the 18 s rDNA and 16 s rDNA of fungi and eubacteria, according to Vainio and Hantula (2000) and Muyzer et al. (1993), respectively. Profiles of the amplified 18 s rDNA and 16 s rDNA sequences were produced by DGGE as in Vainio and Hantula (2000) and Müller et al. (2001), respectively.
Statistical analyses
The data were subjected to Levene’s test to determine homogeneity, after which to two-way ANOVA analysis except for GS8-2 and Gm colonization which were compared using Student’s T-test at P ≤ 0.05. When ANOVA indicated a significant F value, treatment means were separated by the Fisher’s least significant difference (LSD) test at P ≤ 0.05. Data on microbial population were analyzed after logarithmic transformation. All analyses were performed using XLSTAT Pro statistical analysis software (Addinsoft, New York, NY, USA).
Results
Impact on disease severity
Cucumber seedlings pre-treated with GS8-2 and/or Gm 10 days prior to pathogen inoculation inhibited damping-off symptom development compared to the control (Table 1) but there is no interaction between the two beneficial microorganisms in inhibiting disease symptoms. In bentgrass, a negative synergistic effect of interaction between the GS8-2 and Gm was observed. The application of either GS8-2 or Gm was at par with each other in reducing brown patch symptoms but the co-inoculation of GS8-2 and Gm intensified brown patch symptoms development such that it was not significantly different with the control plants. In tomato plants, a significant reduction in the disease symptoms was observed in GS8-2 + Gm treatment compared to the control but the ANOVA analysis shows that only the GS8-2 significantly contributed to the suppression of symptom development of the crown and root rot disease.
Impact on the population of the soil-borne pathogens in the roots and rhizosphere of plants
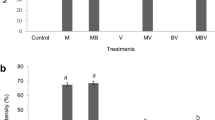
The GS8-2 + Gm highly reduced Forl population in the roots and rhizosphere of tomato plants compared to other treatments (Fig. 1). Either GS8-2 or Gm inoculated alone similarly reduced Forl in the roots and rhizosphere compared to the control. The interaction between GS8-2 and Gm significantly reduced the crown and root rot pathogen population in the roots but not in the rhizosphere (Table 2). The R. solani populations in the roots and rhizosphere of cucumber and bentgrass were not quantified because of limitations previously mentioned.
Number of colony forming units (CFU) of F. oxysporum f. sp. radicis-lycopersici per gram fresh roots and rhizosphere soil of tomato plants inoculated or not with Phoma sp. GS8-2 and/or G. mosseae (Gm) 6 weeks after planting. Bar values are means of six plants. Values with different letters indicate a significant difference (P ≤ 0.05) between treatments according to Fisher’s LSD test
Impact on microbial population in the roots and rhizosphere of plants
The fungal and bacterial population in the roots and rhizosphere of the cucumber, bentgrass and tomato plants were significantly reduced by the composite amendment of GS8-2 and Gm compared to the control except for the bacterial population in the bentgrass rhizosphere at 4WAP (Table 3). In most instances, either GS8-2 or Gm alone treatments similarly reduced significantly the microbial population in the roots and rhizosphere compared to the control except for those in the bentgrass rhizosphere. Interestingly, the ANOVA analysis showed that Gm was affecting both the fungal and bacterial populations; GS8-2 was affecting mainly the fungal population; while the interaction between the two was found to be generally affecting the bacterial population in the roots and/or rhizosphere of the three plants. In the DGGE analysis, it was found that there were shifts in the fungal or bacterial population in the rhizosphere as some bands were found to be absent or present in the different treatments (Fig. 2).
Impact on plant growth
The inoculation of GS8-2 and/or Gm significantly enhanced the SDW of the three plants compared to the control. The co-inoculation of GS8-2 with Gm gave the best enhancement of the SDW of cucumber and tomato, while Gm was at par with GS8-2 in enhancing the growth of bentgrass. The growth enhancement was due to the effects of either GS8-2 or Gm but not their interaction. In bentgrass, growth enhancement was due to GS8-2 only (Table 4).
Impact on root colonization of plants by Phoma sp. GS8-2 and G. mosseae
The re-isolation frequency of GS8-2 from whole root segments of cucumber and tomato plants decreased significantly in dual inoculated plants compared to plants inoculated only with GS8-2 but this was not observed in bentgrass. Since GS8-2 hardly produces spores, its rhizosphere colonization was not determined. No significant differences were found in the Gm root colonization of the three plants indicating that GS8-2 had no effect on Gm (Table 5).
Discussion
Our results demonstrated that either GS8-2 or Gm when applied individually could significantly suppress the disease symptoms development of damping-off and brown patch in cucumber and bentgrass, respectively. In tomato plants, the DS values show that either GS8-2 or Gm did not effectively contained symptom development of crown and root rot caused by Forl compared to the control but the ANOVA analysis shows that GS8-2 significantly contributed to the suppression of disease symptom development while Gm did not. These results indicate that Gm protected plants against soil-borne pathogens and reconfirm what previous studies have shown that Gm could efficiently reduce incidence of root diseases (Pozo et al. 2002; Ozgonen and Erkilic 2007; Martínez-Medina et al. 2009; 2011) but protection is not broad-spectrum (Azcón-Aguilar and Barea 1996; Pozo and Azcón-Aguilar 2007). Similarly, these results indicate that the PGPF Phoma sp. GS8-2 could protect plants against soil-borne diseases and substantiates reported effectiveness against Fusarium wilt in cucumber (Koike et al. 2001). We considered that the co-inoculation of GS8-2 and Gm could give a much better protection values than what these beneficial organisms can achieve individually, however, our results show that the interaction between GS8-2 and Gm yielded negative to neutral effect on the suppression of soil-borne pathogens: it exacerbated brown patch of bentgrass while it had no effect on cucumber damping-off and tomato crown and root rot. These results indicate that the protection conferred by Gm in combination with GS8-2 against soil-borne diseases seems to be dependent on the host plant-pathogen combination involved. Contrary to our results, several studies concerning co-inoculations of AMF with saprotrophs proved effective against soil-borne pathogens. For instance, crown and root rot of tomato caused by Forl was reduced by the co-inoculation of Trichoderma harzianum and Glomus intraradices (Datnoff et al. 1995); root rot of geranium plants caused by Fusarium solani and Macrophomina phaseolina were inhibited by T. harzianum or Penicillium oxalicum and G. mosseae (Haggag and Abd-El latif 2001); damping-off of cucumber caused by R. solani was reduced by Penicillium simplicissimum or T. harzianum and G. mosseae (Chandanie et al. 2009); and Fusarium wilt of melons caused by Fusarium oxysporum f. sp. melonis was controlled by T. harzianum and several AMF species (Martínez-Medina et al. 2009; 2011).
Our results further show that in tomato plants, although significant differences were not observed in the DS, there was a significant reduction of Forl population in the roots and in the rhizosphere of plants treated with either GS8-2 or Gm alone compared to the control. The reduction in the Forl population could be due to several possibilities: (i) quality and/or quantity of exudates from treated roots or from AMF itself inhibited Forl; (ii) competition of colonization sites between Forl and GS8-2 and/or Gm; and, (iii) prior-colonization-possession phenomenon (Toyota et al. 1995), which could be attributed to the exhaustion of nutrients by the established GS8-2 and Gm. Mycorrhization of the host plants could have altered the quality and quantity of root exudates (Norman and Hooker 2000; Filion et al. 2003; Scheffknecht et al. 2006) while the extra-matrical hyphae of Gm itself could produce fungal exudates (Linderman 1988; Marschner and Timonen 2005) which affected Forl population. Additionally, one mechanism by which the PGPF Phoma sp. suppresses soil-borne diseases is through competition (Hyakumachi and Kubota 2004), and lastly, the Forl inoculum was added 6 WAP, by which time the GS8-2 and Gm were already fully established in the roots and in the soil. Unfortunately, we were not able to validate this reduction of pathogen population in cucumber and bentgrass mainly because of 2 limitations: (i) R. solani inoculum units can either exist as sclerotia or hyphal fragments in the soil or in the roots making it tricky to determine the exact CFU; and, (ii) the unavailability of a good selective medium for R. solani to date.
The reduction in the Forl population surprisingly did not relate to the suppression of crown and root rot disease symptoms. As Forl is known to secrete cell wall degrading enzymes (Fernández et al. 1993), it is plausible that minute amounts of these enzymes were potent enough to cause considerable crown and root rotting.
We observed significant quantitative changes in the microbial community in the roots and in the rhizosphere of the inoculated compared to the uninoculated plants. The AMF could affect microbial community composition around the mycorrhizal roots in 2 ways: (i) directly by producing fungal exudates (Marschner and Timonen 2005) and/or (ii) indirectly by modifying root exudates (Linderman 1988) amount and/or composition (Marschner et al. 1997; Vierheilig 2004; Marschner and Timonen 2005; Scheffknecht et al. 2006). Similarly, saprotrophic fungi like the PGPF could affect root exudation amount, pattern and composition (Hale and Moore 1979). We assume that the PGPF could affect rhizosphere microbial communities, comparable with AMF or PGPR (Castro-Sowinski et al. 2007). These changes in root exudation brought about by the colonization of AMF and PGPF, and by the fungal exudates directly secreted by these fungi themselves that resulted in the selective effect on microbial quantities, could also promote biocontrol of pathogens (Whipps 2004; Pozo et al. 2010). However, this effect is apparently not demonstrated in our results for still unknown reasons.
Varied effects of Gm were observed on GS8-2 colonization in the roots of dual inoculated plants depending on the host plant involved. An antagonistic effect of Gm towards GS8-2 colonization in cucumber and tomato plants was observed while no effect was observed in bentgrass. These results affirm previous reported effects of combined inoculation of AMF with saprotrophic fungi which either showed antagonistic, synergistic or neutral effect on the population of the saprotrophic co-inoculant (McAllister et al. 1997; Garcia-Romera et al. 1998; Ravnskov et al. 2006; Martínez-Medina et al. 2009). The effect of combined AMF-saprotrophic fungi inoculations on the population of each co-inoculant is highly variable; affected by different factors such as the species of AMF or saprotrophic fungus involved. Results could vary even within the isolates of the same species of the AMF or saprotrophic fungus (Datnoff et al. 1995; Rousseau et al. 1996; Green et al. 1999; Martinez et al. 2004). Interestingly, although a negative effect of the Gm was observed in the root colonization of GS8-2, no negative effect was observed with regards to its biocontrol capacity against soil-borne pathogens. Furthermore, our results verify reports that plant species vary in the degree to which they respond to AMF (Jeffries and Barea 2001; Smith and Read 2008) and show that Phoma sp. GS8-2 did not affect the Gm root colonization in cucumber, bentgrass or tomato plants. In bentgrass, the negligible Gm root colonization did not relate to the ability of Gm to suppress the brown patch disease symptoms. This result is an exception to the general observation that only a well-established AMF symbiosis can provide good mycorrhizal protection (Slezack et al. 2000; Khaosaad et al. 2007; Pozo et al. 2010), but Hart and Reader (2002) reported that percent root length colonization does not always reflect the extent of colonization of AMF.
Plant growth enhancement observed in this study is primarily due to the individual effect of GS8-2 or Gm. This result supports previous studies with other PGPF in which no additive effect were observed between Gm and Phoma sp. or Gm and F. equiseti isolates in growth enhancement of cucumber plants (Chandanie et al. 2005; Saldajeno and Hyakumachi 2011). In contrast to our results, additive effects on plant growth by the interaction between the AMF and other beneficial soil microorganisms have been reported elsewhere (Calvet et al. 1993; Fracchia et al. 2000; Haggag and Abd-El latif 2001; Ravnskov et al. 2006; Martínez-Medina et al. 2009).
In summary, our study which demonstrates the effectiveness of either GS8-2 or Gm applied alone against several soil-borne diseases equally suggests that the effectiveness of their co-inoculation against soil-borne pathogens depends on the host-pathogen combination involved. In this instance, the GS8-2 + Gm has neutral effect in reducing damping-off disease of cucumber caused by R. solani AG-4 and the crown and root rot disease of tomato caused by Forl, and an antagonistic effect on the brown patch disease caused by R. solani AG2-2 in bentgrass. This study underscores the interaction between two beneficial microorganisms which could be very effective individually but not when applied together.
References
Azcón-Aguilar C, Barea JM (1996) Arbuscular mycorrhizas and biological control of soil-borne plant pathogens—an overview of the mechanisms involved. Mycorrhiza 6:457–464
Azcón-Aguilar C, Jaizme-Vega M, Calvet C (2002) The contribution of arbuscular mycorrhizal fungi to control of soil-borne plant pathogens. In: Gianinazzi S, Schüepp H, Barea JM, Haselwandter K (eds) Mycorrhizal technology in agriculture: from genes to bioproducts. Birkhäuser Verlag, Basel, pp 187–197
Brundrett MC, Piché Y, Peterson RL (1984) A new method for observing the morphology of vesicular-arbuscular mycorrhizae. Can J Bot 62:2128–2134
Calvet C, Pera J, Barea JM (1993) Growth response of marigold (Tagetes erecta L.) to inoculation with Glomus mosseae, Trichoderma aureoviride and Pythium ultimum in a peat-perlite mixture. Plant Soil 148:1–6
Castro-Sowinski S, Herschkovitz Y, Okon Y, Jurkevitch E (2007) Effects of co-inoculation with plant growth-promoting rhizobacteria on resident rhizosphere microorganisms. FEMS Microbiol Lett 276:1–11
Chandanie WA, Kubota M, Hyakumachi M (2005) Interaction between arbuscular mycorrhizal fungus Glomus mosseae and plant growth promoting fungus Phoma sp. on their root colonization and growth promotion of cucumber. Mycoscience 46:201–204
Chandanie WA, Kubota M, Hyakumachi M (2006) Interactions between plant growth promoting fungi and arbuscular mycorrhizal fungus Glomus mosseae and induction of systemic resistance to anthracnose disease in cucumber. Plant Soil 286:209–217
Chandanie WA, Kubota M, Hyakumachi M (2009) Interactions between the arbuscular mycorrhizal fungus Glomus mosseae and plant growth promoting fungi and their significance for enhancing plant growth and suppressing damping-off of cucumber (Cucumis sativus L.). App Soil Ecol 41:336–341
Datnoff LE, Nemec S, Pernezny K (1995) Biological control of Fusarium crown and root rot of tomato in Florida using Trichoderma harzianum and Glomus intraradices. Biol Control 5:427–431
De Boer M, Bom P, Kindt F, Keurentjes JJB, van der Sluis I, van Loon LC, Bakker PAHM (2003) Control of Fusarium wilt of radish by combining Pseudomonas putida strains that have different disease-suppressive mechanisms. Phytopathology 93:626–632
Défago G, Keel C (1995) Pseudomonads as biocontrol agents of diseases caused by soil-borne pathogens. In: Hokkanen HMT, Lynch JM (eds) Biological control: benefits and risks. University Press, Cambridge, pp 230–257
Edwards SG, Young JPW, Fitter AH (1998) Interactions between Pseudomonas flourescens biocontrol agents and Glomus mosseae, an arbuscular mycorrhizal fungus, within the rhizosphere. FEMS Microbiol Lett 166:297–303
Fernández N, Patiño B, Vázquez C (1993) Pectin degrading enzymes secreted by six isolates of Fusarium oxysporum. Mycol Res 97:461–466
Filion M, St-Arnaud M, Jabaji-Hare SH (2003) Quantification of Fusarium solani f. sp. phaseoli in mycorrhizal bean plants and surrounding mycorrhizosphere soil using real-time polymerase chain reaction and direct isolations on selective media. Phytopathology 93:229–235
Fracchia S, García-Romera I, Godeas A, Ocampo JA (2000) Effect of the saprophytic fungus Fusarium oxysporum on arbuscular mycorrhizal colonization and growth of plants in greenhouse and field trials. Plant Soil 223:175–184
Garcia-Romera I, Garcia-Garrido JM, Martin J, Fracchia S, Mujica MT, Godeas A, Ocampo JA (1998) Interaction between saprophytic Fusarium strains and arbuscular mycorrhizas of soybean plants. Symbiosis 24:235–246
Green H, Larsen J, Olsson PA, Jensen DF, Jakobsen I (1999) Suppression of the biocontrol agent Trichoderma harzianum by mycelium of the arbuscular mycorrhizal fungus Glomus intraradices in root-free soil. App Environ Microbiol 65:1428–1434
Guetsky R, Shtienberg D, Elad Y, Dinoor A (2001) Combining biocontrol agents to reduce the variability of biological control. Phytopathology 91:621–627
Haggag WM, Abd-El latif FM (2001) Interaction between vesicular arbuscular mycorrhizae and antagonistic biocontrol microorganisms on controlling root rot disease incidence of geranium plants. Online J Biological Sci 1:1147–1153
Haggag WM, Nofal MA (2006) Improving the biological control of Botryodiplodia disease on some Annona cultivars using single or multi-biogents in Egypt. Biological Cont 38:341–349
Hale MG, Moore LD (1979) Factors affecting root exudation II: 1970–1978. In: Brady NC (ed) Advances in agronomy, vol 31, Academic Press. Inc, New York, pp 93–124
Hart MM, Reader RJ (2002) Does percent root len gth colonization and soil hyphal length reflect the extent of colonization for all AMF? Mycorrhiza 12:297–301
Horinouchi H, Muslim A, Suzuki T, Hyakumachi M (2007) Fusarium equiseti GF191 as an effective biocontrol agent against Fusarium crown and root rot of tomato in rock wool systems. Crop Prot 26:1514–1523
Hyakumachi M (1994) Plant growth promoting fungi from turfgrass rhizosphere with potential for disease suppression. Soil Microorg 44:53–68
Hyakumachi M, Kubota M (2004) Fungi as plant growth promoter and disease suppressor. In: Arora DK (ed) Fungal biotechnology in agricultural, food, and environmental applications, vol 21. Marcel Dekker, New York, pp 101–110
Jeffries P, Barea JM (2001) Arbuscular mycorrhiza: a key component of sustainable plant-soil ecocsystems. In: Hock B (ed) The Mycota: Vol IX: fungal associations. Springer, Berlin, pp 95–113
Jeffries P, Gianinazzi S, Perotto S, Turnau K, Barea JM (2003) The contribution of arbuscular mycorrhizal fungi in sustainable maintenance of plant health and soil fertility. Biol Fertil Soils 37:1–16
Khaosaad T, García-Garrido JM, Steinkellner S, Vierheilig H (2007) Take-all disease is systematically reduced in roots of mycorrhizal barley plants. Soil Biol Biochem 39:727–734
Koike N, Hyakumachi M, Kageyama K, Tsuyumu S, Doke N (2001) Induction of systemic resistance in cucumber against several diseases by plant growth promoting fungi: lignification and superoxide generation. Eur J Plant Pathol 107:523–533
Komada H (1975) Development of a selective medium for quantitative isolation of Fusarium oxysporum from natural soil. Rev Plant Prot Res 8:114–124
Linderman RG (1988) Mycorrhizal interactions with the rhizosphere microflora: the mycorrhizosphere effect. Phytopathology 78:366–371
Marschner P, Timonen S (2005) Interactions between plant species and mycorrhizal colonization on the bacterial community composition in the rhizosphere. Appl Soil Ecol 28:23–36
Marschner P, Crowley DE, Higashi RM (1997) Root exudation and physiological status of a root-colonizing fluorescent pseudomonad in mycorrhizal and non-mycorrhizal pepper (Capsicum annuum L.). Plant Soil 189:11–20
Martin JP (1950) Use of acid, rose bengal and streptomycin in the plate method for estimating soil fungi. Soil Sci 69:215–232
Martinez A, Obertello M, Pardo A, Ocampo JA, Godeas A (2004) Interactions between Trichoderma pseudokoningii strains and the arbuscular mycorrhizal fungi Glomus mosseae and Gigaspora rosea. Mycorrhiza 14:79–84
Martínez-Medina A, Pascual JA, Lloret E, Roldán A (2009) Interactions between arbuscular mycorrhizal fungi and Trichoderma harzianum and their effects on Fusarium wilt in melon plants grown in seedling nurseries. J Sci Food Agric 89:1843–1850
Martínez-Medina A, Roldán A, Pascual JA (2011) Interaction between arbuscular mycorrhizal fungi and Trichoderma harzianum under conventional and low input fertilization field condition in melon crops: growth response and Fusarium wilt biocontrol. App Soil Ecol 47:98–105
McAllister CB, Garcia-Garrido JM, García-Romera I, Godeas A, Ocampo JA (1997) Interaction between Alternaria alternata or Fusarium equiseti and Glomus mosseae and its effects on plant growth. Biol Fert Soil 24:301–305
McGonigle TP, Miller MH, Evans DG, Fairchild GL, Swan JA (1990) A new method which gives an objective measure of colonization of roots by vesicular-arbuscular mycorrhizal fungi. New Phytol 115:495–501
Meera MS, Shivanna MB, Kageyama K, Hyakumachi M (1994) Plant growth promoting fungi from zoysiagrass rhizosphere as potential inducers of systemic resistance in cucumbers. Phytopathology 84:1399–1406
Meera MS, Shivanna MB, Kageyama K, Hyakumachi M (1995a) Persistence of induced systemic resistance in cucumber in relation to root colonization by plant growth promoting fungal isolates. Crop Prot 14:123–130
Meera MS, Shivanna MB, Kageyama K, Hyakumachi M (1995b) Responses of cucumber cultivars to induction of systemic resistance against anthracnose by plant growth promoting fungi. Eur J Plant Pathol 101:421–430
Müller AK, Westergaard K, Christensen S, Sørensen SJ (2001) The effect of long-term mercury pollution on the soil microbial community. FEMS Microbiol Ecol 36:11–19
Muyzer G, De Waal EC, Uitterlinden AG (1993) Profiling of complex microbial populations by denaturing gradient gel electrophoresis analysis of polymerase chain reaction amplified genes coding for 16S ribosomal RNA. Appl Environ Microbiol 59:695–700
Norman JR, Hooker JE (2000) Sporulation of Phytophthora fragariae shows greater stimulation by exudates of non-mycorrhizal than by mycorrhizal strawberry roots. Mycol Res 104:1069–1073
Ozgonen H, Erkilic A (2007) Growth enhancement and Phytophthora blight (Phytophthora capsici Leonian) control by arbuscular mycorrhizal fungal inoculation in pepper. Crop Prot 26:1682–1688
Pozo MJ, Azcón-Aguilar C (2007) Unraveling mycorrhizal-induced resistance. Curr Opinion Plant Biol 10:393–398
Pozo MJ, Cordier C, Dumas-Gaudot E, Gianinazzi S, Barea JM, Azcón-Aguilar C (2002) Localized versus systemic effect of arbuscular mycorrhizal fungi on defence responses to Phytophthora infection in tomato plants. J Exp Bot 53:525–534
Pozo MJ, Jung SC, López-Ráez JA, Azcón-Aguilar C (2010) Impact of arbuscular mycorrhizal symbiosis on plant response to biotic stress: the role of plant defence mechanisms. In: Koltai H, Kapulnik J (eds) Arbuscular mycorrhizas: physiology and function, 2nd edn. Springer, Heidelberg, pp 193–208
Raupach GS, Kloepper JW (1998) Mixtures of plant growth-promoting rhizobacteria enhance biological control of multiple cucumber pathogens. Phytopathology 88:1158–1164
Ravnskov S, Jensen B, Knudsen IMB, Bødker L, Jensen DF, Karliński L, Larsen J (2006) Soil inoculation with the biocontrol agent Clonostachys rosea and the mycorrhizal fungus Glomus intraradices results in mutual inhibition, plant growth promotion and alteration of soil microbial communities. Soil Biol Biochem 38:3453–3462
Roberts DP, Lohrke SM, Meyer SLF, Buyer JS, Bowers JH, Baker CJ, Li W, de Souza JT, Lewis JA, Chung S (2005) Biocontrol agents applied individually and in combination for suppression of soilborne diseases of cucumber. Crop Prot 24:141–155
Rousseau A, Benhamou N, Chet I, Piché Y (1996) Mycoparasitism of the extramatrical phase of Glomus intraradices by Trichoderma harzianum. Phytopathology 86:434–443
Saldajeno MGB, Hyakumachi M (2011) The plant growth-promoting fungus Fusarium equiseti and the arbuscular mycorrhizal fungus Glomus mosseae stimulate plant growth and reduce severity of anthracnose and damping-off diseases in cucumber (Cucumis sativus L.) seedlings. Ann Appl Biol 159:28–40
Scheffknecht S, Mammerler R, Steinkellner S, Vierheilig H (2006) Root exudates of mycorrhizal tomato plants exhibit a different effect on microconidia germination of Fusarium oxysporum f. sp. lycopersici than root exudates from non-mycorrhizal tomato plants. Mycorrhiza 16:365–370
Shivanna MB, Meera MS, Kageyama K, Hyakumachi M (1996) Growth promotion ability of zoysiagrass rhizosphere fungi in consecutive plantings of wheat and soybean. Mycoscience 37:163–168
Shivanna MB, Meera MS, Kubota M, Hyakumachi M (2005) Promotion of growth and yield in cucumber by zoysiagrass rhizosphere fungi. Microbes Environ 20:34–40
Slezack S, Dumas-Gaudot E, Paynot M, Gianinazzi S (2000) Is a fully established mycorrhizal symbiosis required for bioprotection of Pisum sativum roots against Aphanomyces euteiches? Mol Plant-Microbe Interact 13:238–241
Smith SE, Read DJ (2008) Mycorrhizal symbiosis, 3rd edn. Elsevier, Great Britain
Toyota K, Kitamura M, Kimura M (1995) Suppression of Fusarium oxysporum f. sp. raphani PEG-4 in soil following colonization by other Fusarium spp. Soil Biol Biochem 27:41–46
Vainio EJ, Hantula J (2000) Direct analysis of wood-inhabiting fungi using denaturing gradient gel electrophoresis of amplified ribosomal DNA. Mycol Res 104:927–936
Vázquez MM, César S, Azcón R, Barea JM (2000) Interactions between arbuscular mycorrhizal fungi and other microbial inoculants (Azospirillum, Pseudomonas, Trichoderma) and their effects on microbial population and enzyme activities in the rhizosphere of maize plants. Appl Soil Ecol 15:261–272
Vierheilig H (2004) Regulatory mechanisms during the plant-arbuscular mycorrhizal fungus interaction. Can J Bot 82:1166–1176
Whipps JM (2004) Prospects and limitations for mycorrhizas in biocontrol of root pathogens. Can J Bot 82:1198–1227
Acknowledgment
We would like to thank F. Kawane (Idemitsu Kosan Co. Ltd., Tokyo, Japan) for the kind provision of G. mosseae inoculum and to the two anonymous reviewers for their suggestions in improving the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Saldajeno, M.G.B., Ito, M. & Hyakumachi, M. Interaction between the plant growth-promoting fungus Phoma sp. GS8-2 and the arbuscular mycorrhizal fungus Glomus mosseae: impact on biocontrol of soil-borne diseases, microbial population, and plant growth. Australasian Plant Pathol. 41, 271–281 (2012). https://doi.org/10.1007/s13313-011-0101-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13313-011-0101-7