Abstract
Glioblastoma has one of the highest mortality rates among cancers, and it is the most common and malignant form of brain cancer. Among the typical features of glioblastoma tumors, there is an aberrant vascularization: all gliomas are among the most vascularized/angiogenic tumors. In recent years, it has become clear that glioblastoma cells can secrete extracellular vesicles which are spherical and membrane-enclosed particles released, in vitro or in vivo, by both normal and tumor cells; they are involved in the regulation of both physiological and pathological processes; among the latter, cancer is the most widely studied. Extracellular vesicles from tumor cells convey messages to other tumor cells, but also to normal stromal cells in order to create a microenvironment that supports cancer growth and progression and are implicated in drug resistance, escape from immunosurveillance and from apoptosis, as well as in metastasis formation; they are also involved in angiogenesis stimulation, inducing endothelial cells proliferation, and other pro-angiogenic activities. To this aim, the present paper assesses in detail the extracellular vesicles phenomenon in the human glioblastoma cell line U251 and evaluates extracellular vesicles ability to promote the processes required to achieve the formation of new blood vessels in human brain microvascular endothelial cells, highlighting that they stimulate proliferation, motility, and tube formation in a dose-response manner. Moreover, a molecular characterization shows that extracellular vesicles are fully equipped for angiogenesis stimulation in terms of proteolytic enzymes (gelatinases and plasminogen activators), pro-angiogenic growth factors (VEGF and TGFβ), and the promoting-angiogenic CXCR4 chemokine receptor.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Glioblastoma, or glioblastoma multiforme (GBM), is the most malignant form of glioma and the most common brain cancer: it accounts for 12–15 % of all intracranial tumors and 50–60 % of astrocytic tumors [1]. GBM has one of the highest mortality rates among cancers: the prognosis remains unfavorable even if the tumor is surgically removed and subsequently treated with radiotherapy and chemotherapy [1]. Indeed, the highly infiltrative nature of GBM is associated to its innate ability to massively invade normal brain structures, hindering the complete removal of tumor cells through the surgical approach, as well as with radiation and chemotherapy [2].
Typical features of GBM tumors are the cellular heterogeneity (that includes stem cells), a high proliferative rate, and an extensive invasion [3, 4]; molecular features can be peculiar depending on whether the tumor arises as de novo or progresses from a lower grade astrocytoma (primary and secondary tumor, respectively) [2, 5].
Aberrant vascularization is another typical feature of GBM; all gliomas are among the most vascularized/angiogenic tumors, and it is known that the neurovascular unit (formed by endothelial cells, pericytes, and astrocytes) acts supporting the tumor progression [6]. Glioma vasculature, indeed, is characterized by microvascular proliferation and endothelial hyperplasia which are used as histological markers of high-grade gliomas and represents regions of angiogenesis [6], the process by which new blood vessels were formed from preexisting ones and that requires for endothelial cells to proliferate, migrate, and organize in new tubular structures [7]. Such regions, also called perivascular niches, seem to emerge as specific microenvironments in which brain tumor stem-like cells, a small population within the heterogeneous tumor cells with stem-like features, reside [8].
In recent years, it has become clear that GBM cells can secrete both exosomes (EXOs) and microvesicles (MVs) (collectively named extracellular vesicles, EVs) in their surrounding microenvironment [9–11] supporting tumor progression by several mechanisms, including angiogenesis stimulation [12]; EXOs contain a plethora of angiogenic molecules such as angiogenin, IL-6 and IL-8, and vascular endothelial growth factor (VEGF) [12], and EVs cargo contains some pro-angiogenic microRNAs (miRNAs), specifically miR-29a and miR-30e [13]. Moreover, once “programmed” by GBM EVs, endothelial cells are able, in turn, to stimulate pericytes proliferation in a paracrine fashion, as well as the migration and proliferation of GBM cells [14].
A deeper knowledge of the GBM tumor biology could help in the comprehension of biological mechanisms used in tumor progression. To this aim, the present paper assesses in detail the EVs phenomenon in the human GBM cell line U251 and evaluates the ability of both EXOs and MVs to promote in human brain microvascular endothelial cells the processes required to achieve the formation of new blood vessels.
Materials and methods
Cell culture
U251 cells, derived from a human malignant glioblastoma tumor, were grown as monolayer in DMEM containing 10 % (v/v) heat-inactivated fetal bovine serum, 1× Penicillin/Streptomycin, and 2 mM l-glutamine and maintained at 37 °C in a humidified atmosphere with 5 % CO2. All materials were purchased by Euroclone SpA, Milan, Italy.
Human brain microvascular endothelial cells (HBMEC) from ScienCell Research Laboratories (Carlsbad, CA, USA) were cultured in EGM-2MV (Lonza Bioresearch, Basel, Switzerland), a specified endothelial cell medium for microvascular endothelial cells. Cells were maintained at 37 °C in a humidified atmosphere with 5 % CO2.
All experiments were carried out when cells were sub-confluent and mycoplasma negative.
Isolation of extracellular vesicles from conditioned medium
U251 conditioned medium was obtained from cells stimulated with DMEM supplemented with 10 % of 40-nm-filtered heat-inactivated FBS Hyclone (Thermo Scientific, Rockford, IL, USA).
To isolate EVs, conditioned medium was centrifuged at 600 g for 15 min and then at 1500 g for 30 min to remove cells and large debris, respectively; resulting supernatants were centrifuged at 100,000 g for 2 h at 4 °C in a Beckmann ultracentrifuge. Pelleted EVs were resuspended in Dulbecco’s phosphate-buffered saline (Euroclone, Milan, Italy); double determination of EVs quantification was carried out by measuring their vesicle-associated protein levels using the method of Bradford (Bio-Rad, Milan, Italy), with bovine serum albumin (Sigma-Aldrich, St. Louis, MO, USA) as standard.
Electron microscopy
Scanning electron microscopy (SEM) was carried out on subconfluent cells grown on coverslips and fixed with 2 % glutaraldehyde (Electron Microscopy Sciences, Hatfield, PA, USA) in PBS for 30 min, then dehydrated through ethanol series beginning with 30 % and changing solutions of 50, 70, 90, and 100 %. Samples were critical point-dried, glued onto stubs, coated with gold in a SCD040 Balzer Sputterer, and observed using a Philips 505 SEM at 20 kV.
Transmission electron microscopy (TEM) was performed on isolated EVs, resuspended in PBS, to analyze their ultrastructural morphology. According to proper dilutions, the samples were adsorbed to 300 mesh carbon-coated copper grids (Electron Microscopy Sciences, Hatfield, PA, USA) for 5 min in a humidified chamber at room temperature. EVs on grids were then fixed in 2 % glutaraldehyde (Electron Microscopy Sciences, Hatfield, PA, USA) in PBS for 10 min and then briefly rinsed in Milli-Q water. Grids with adhered EVs were examined with a Philips CM 100 transmission electron microscope TEM at 80 kV, after negative staining with 2 % phosphotungstic acid, brought to pH 7.0 with NaOH. Images were captured by a Kodak digital camera [15, 16].
Zymography assays
Gelatin zymography was performed using sodium dodecyl sulfate-polyacrylamide gels (SDS-PAGE, 7.5 %) copolymerized with 1 mg/ml gelatin type B (Sigma-Aldrich, St. Louis, MO, USA). EVs (10 μg) were diluted in SDS-PAGE sample buffer under non-reducing conditions without heating; after electrophoresis, gels were washed three times, 15 min each ones, with 50 mM Tris HCl (pH 7.4) containing 2.5 % Triton X-100 (Sigma-Aldrich, St. Louis, MO, USA) under agitation at room temperature and then incubated overnight in gelatinases buffer (50 mM Tris-HCl pH 7.4 containing 120 mM NaCl, 5 mM CaCl2) at 37 °C. Gels were stained with 0.25 % Coomassie Blue R 250 (Bio-Rad, Milan, Italy) in a mixture of methanol–acetic acid–water (4:1:5) for 30 min and were destained in the same solution without dye. Gelatinases’ activities were visualized as distinct white bands on a blue background, indicating proteolysis of the substrate.
Casein-plasminogen zymography was performed on EVs (10 μg) diluted in SDS-PAGE sample buffer under non-reducing conditions without heating, using SDS-polyacrylamide (10 %) gels copolymerized with 0.2 % casein (Sigma-Aldrich, St. Louis, MO, USA) plus 10 μg/ml plasminogen (Sigma-Aldrich, St. Louis, MO, USA). After electrophoresis, gels were washed twice for 30 min in Tris 50 mM pH 7.4+ Triton-X 2.5 % at room temperature and incubated for 20 h in the same buffer without Triton-X at 37 °C. Gels staining and destaining were performed as for the gelatin zymography.
Western blot analysis
EVs (20 μg) were resolved by 10 % SDS-PAGE under non-reducing conditions without heating and transferred to nitrocellulose membrane (Whatman—GE Healthcare Life Sciences, UK). Nonspecific binding sites were blocked for 2 h in 10 % nonfat dry milk in TBS-T (TBS containing 0.5 % Tween-20) at room temperature. Blots were then incubated at 4° overnight with primary antibodies against CXCR4 (Rabbit polyclonal, 1:5000 dilution; NB100-74396 Novus Biologicals, Minneapolis, MN, USA) or TGFβ-1 (Mouse monoclonal, 1:2000 dilution, clone OTI3B6, TA506583 OriGene, Rockville, MD, USA). This step, after several washes in TBS-T, was followed by the membranes incubation in a peroxidase-conjugate secondary antibody (goat anti-rabbit IgG-HRP, 1:7500 dilution, sc-2004 or goat anti-mouse IgG-HRP, 1:10,000 dilution, sc-2005, both from Santa Cruz Biotechnology, Santa Cruz, CA, USA) for 1 h. All antibodies were diluted in blocking buffer (TBS-T containing 1 % nonfat dry milk).
After washing in TBS-T, the reactive bands were visualized with a chemiluminescence detection kit (SuperSignal West Pico Chemiluminescent Substrate, Thermo Scientific, Rockford, IL, USA) and images were recorded with the gel documentation system Alliance LD2 (Uvitec, Cambridge, UK).
The optical density of the target bands was determined using the Alliance LD2 gel documentation system or the ImageJ public domain software.
ELISA
VEGF concentrations in EVs from U251 conditioned medium were determined by ELISA. The amount of VEGF was assessed using sandwich RayBio® Human VEGF (RayBiotech, Inc.) according to the manufacturer’s instructions. All experimental points were assayed in triplicate. VEGF was within the dynamic range of the ELISA standard curve and calculated as picograms/ml/g of protein.
Proliferation
To quantify the effects of U251 EVs on endothelial cell proliferation, the colorimetric crystal violet technique was used. Briefly, HBMECs were detached from the culture flasks by a trypsin solution (0.05 % w/v). Upon cells centrifugation, the pellets were resuspended in specific medium but in the presence of 2 % FBS (starved media) and cells cultured for 12 h in order to allow synchronization before treatments. The media were then replaced by the appropriate complete medium as indicated in cell culture section and cells cultured for 24 or 72 h in the presence of EVs at increasing concentration (2, 4, 8 μg/ml). At the end of incubations, all plates were washed with PBS (three times), and the cells were fixed for 15 min with 100 μl/well of formaldehyde 3 % and sucrose 2 % solution. After drying, cells were stained with a solution of 5 % crystal violet in 20 % methanol for 30 min at room temperature as described. Stain was eluted with a solution of 0.1 M sodium citrate (pH 4.2) and ethanol, and the relative absorbance value was read at 540 nm. Growth of treated samples was determined as a percentage of growth with respect to controls (CTR).
In vitro scratch wound assay
The potential of U251 EVs to stimulate endothelial cell migration was assessed by in vitro scratch wound assay. Culture inserts from IBIDI (Munich, Germany) were placed on a plate surface and HBMECs at a concentration of 5 × 105 cells/ml (applied in 70 μl per well) were seeded in each reservoir. After cell attachment, culture silicone inserts were removed washing gently with medium. EVs at a concentration of 6 and 8 μg/ml were added to refresh medium. EGM-2MV served as control. The scratched area was photographed using light microscopy at start and after 24 h of incubation at 37 °C, 5 % CO2; to make the result more visible, cells photographed after 24 h were stained with Crystal Violet 1 % in methanol. Each sample was measured in two wells and two picture-fields per well were examined. Results were averaged for analysis.
Tube formation assay
The tube formation assay was carried out by using the in vitro Matrigel assay kit (Chemicon Millipore) following the manufacturer’s instructions to coat 15-wells microslides (10 μl/well) of IBIDI. Matrigel was allowed to solidify at 37 °C for 30 min and HBMECs at the concentration of 2 × 104/well were added to chilled pellets and incubated with EGM-2 medium for up to 12 h. The degree of the angiogenic response was assessed after 16 h from the beginning of the treatment using an inverted phase contrast microscope by evaluating the number of branching points. Each well was photographed and the relative acquired images quantified using an ImageJ analysis system. Several morphometric parameters are computed including total and average vessel length, branching point density, and vascular density and among those we chose the most representative ones: the branching index. Branching index was obtained by measuring the number of junctions formed per vessel area. Mean values and standard deviation (SD) were determined for each analysis.
Data analysis
Data shown are from at least three independent experiments and are presented as mean ± SD. Statistical significance was determined using the Student’s t test. Calculations were performed using GraphPad Prism 4 software (GraphPad, San Diego, CA, USA). Statistical significance was set at P < 0.05.
Results
U251 cells release both exosomes and microvesicles
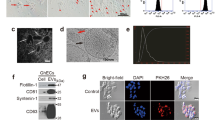
To verify which type of extracellular vesicles is released by U251 cell, TEM analyses of isolated EVs were performed: the assay shows the presence of intact vesicles enclosed by the plasma membrane (Fig. 1a–c) and characterized by heterogeneous size, ranging from 40 to 760 nm (Fig. 1d). Given for granted the limit of 100 nm conventionally used as discriminating between exosomes (size <100 nm) and microvesicles (size >100 nm) [17], the results show that the microvesicles population is slightly more represented than the exosomes (56 vs. 44 %).
Transmission electron microscopy (TEM) analysis of EVs isolated from U251 conditioned media. a–c Images of negatively stained EVs reveal a size heterogeneity that suggests the presence of both MVs and EXOs. Measured sizes are 70 nm in (a) (presumably an EXO), 115 nm and 315 nm (presumably MVs) in (b) and (c), respectively. Size bars are 500 nm for all the images. d Graph represents the size of EVs analyzed from TEM pictures: each EV observed on grids corresponds to one dot. The horizontal bar represents the size limit (100 nm) that usually distinguishes EXOs and MVs subpopulations
The release of microvesicles was also analyzed by the means of SEM analysis, which points out an abundant budding of MVs from cell surface (Fig. 2a, b). In some cells, even if a considerable dynamism of cell membrane is evident in the main body of the cell, the release of MVs involves only the cell periphery (Fig. 2a) while in other cells, the MVs budding interests the whole cell surface (Fig. 2b).
Molecular characterization
By means of zymography techniques, the presence of gelatinases and plasminogen activators was assessed; by Western blot, CXCR4 and TGF-β expression was also evaluated (Fig. 3).
U251 EVs molecular characterization. a Gelatinases in U251 EVs. Deduced molecular weights are 98 (pro-MMP-9) and 68 and 63 kDa (pro-MMP-2 and MMP-2, respectively). b Plasminogen activators in U251 EVs. Deduced molecular weights are 95 (PAI-PA complexes), 64 (tPA), and 49 kDa (uPA HMW). c CXCR4 in U251 EVs. Deduced molecular weights are 77 and 42,8 kDa for the upper and lower band, respectively. d TGF-β in U251 EVs. Deduced molecular weight is 51 kDa
Gelatin zymography allowed to identify in EVs samples the gelatinases MMP-2 and-9, the first one both in pro-enzyme and active form (calculated molecular weights: proMMP-9, 97 kDa; proMMP-2, 68 kDa; MMP-2, 63 kDa); MMPs complexes and/or dimers are also present at high molecular weights (Fig. 3a).
Casein-plasminogen zymography showed the presence of plasminogen activators in EVs. The assay underlines that PA-PAI complexes (95 kDa), tissue type-PA (67 kDa), and urokinase type-PA (49 kDa—HMW forms) are associated to EVs (Fig. 3b).
Anti-CXCR4 antibody revealed two bands with calculated size of 77 and 43 kDa, probably corresponding to monomer and dimer forms (Fig. 3c). Anti-TGF-β was also associated in EVs: the calculated size of band is about 51 kDa maybe corresponding to the glycosylated unprocessed form (Fig. 3d).
To verify whether the pro-angiogenic effect of U251 extracellular vesicles was dependent by VEGF presence, a known pro-angiogenic factor, we determined by ELISA the level of VEGF contained in vesicles. The value of VEGF with respect to the total amount of proteins was 0.26 ± 0.04 μg/μl.
Dose-dependent effect of U251 vesicles on endothelial growth
HBMEC exposed to U251 vesicles (2, 4, 8 μg/ml), with respect to CTR, demonstrated significant differences in growth rate after 24 and 72 h of treatment with respect to CTR (Fig. 4). In details, as shown in histogram after 24 h, this stimulation produced an increase of growth rate of HBMEC only by using 8 μg/ml EVs concentration. Differently, after 72 h of EVs stimulation, vesicles seemed able to stimulate HBMEC growth at all concentrations tested.
U251 extracellular vesicles effectively induce angiogenesis and wound closure
To further evaluate the effectiveness of U251 EVs in vitro, a wound closure assay and tube formation assay were performed.
EVs treatment showed increased wound closure in an in vitro scratch wound assay: EVs-treated HBMEC showed increased wound closure compared to CTR treatment (Fig. 5).
In the tube formation assay, the representative images show a dose-dependent effect of EVs on HBMEC tube formation (Fig. 6a–e). Histogram in Fig. 6f shows that U251 vesicles at higher concentration (6 and 8 μg/ml) were able to significantly induce tube formation compared to CTR.
U251 EVs stimulate angiogenesis. Representative images of tube formation showing a dose-dependent effect of EVs on HBMEC. The tube formation has been evaluated as branching index, measured after vesicles treatments (2, 4, 6, 8 μg/ml) (b–e) compared to CTR (a). f Depicts the graphical representation of tube formation assay; *P < 0.05 EVs vs. CTR
Discussion
It is known for several years that cells can take advantage of extracellular vesicles (EVs) as bioactive molecular shuttles to modulate their microenvironment and to interact with the neighboring cells, their cargo being composed by biologically active proteins (e.g., growth factors, cytokines, proteolytic enzymes), lipids, and nucleic acids (e.g., mRNA and miRNAs) [18–20].
The term “extracellular vesicles” refers specifically to spherical and membrane-enclosed particles released, in vitro or in vivo, by both normal and tumor cells [15, 19, 21, 22]; according to their biogenesis, EVs can be distinguished into exosomes (EXOs), microvesicles (MVs), and apoptotic bodies [21]. EXOs have a size ranging from 40 to 100 nm in diameter and originate from the fusion of multivesicular bodies with plasma membrane, whereas MVs are 100–1000 nm in size and are released through the outward budding of the plasma membrane [21]. Apoptotic bodies, the last class of EVs, vary in size between 1 and 5 μm in diameter and are released from the plasma membrane as blebs when cells undergo apoptosis [21]. Upon release, EVs can circulate in the local microenvironment or travel long distances diffusing through biological fluids (actually, EVs have been isolated from several biological fluids such as blood, urine, breast milk, semen, cerebrospinal fluid, saliva, synovial fluid) [17]; delivering of their messages to target cells happens through several mechanisms: bounding of ligands expressed on their surface to target cells receptors, membrane fusion with target cell membrane, and uptake by endocytosis or discharge of their contents directly into the extracellular space after the loss of their structural entirety [22].
EVs are involved in the regulation of both physiological (immune response, coagulation, and tissue repair) [23–25] and pathological processes (cardiovascular and inflammatory diseases or neurodegenerative disorders) [26–28]. To date, among all pathological conditions, the most studied one is represented by cancer, since EVs have been demonstrated to be involved in several processes leading to cancer progression.
Tumor cell EVs convey messages to other tumor cells, but also to normal stromal cells in order to create a microenvironment that supports cancer growth and progression, especially in virtue of their proteolytic cargo [29–33] and are involved in drug resistance and escape from immunosurveillance and from apoptosis too [34–37] as well as in metastasis formation [38–40]. They are also implicated in angiogenesis stimulation, inducing endothelial cells proliferation and other pro-angiogenic activities [40–43].
From several years, the role of EVs has been highlighted in GBM tumors too; the studies support the concept that GBM cells use EVs to modify the microenvironment in order to make it more advantageous to tumor progression [9–11, 13]. Among other processes, GBM EVs seem to be involved in angiogenesis; this process is a basic requirement for the sustainment of the rapid tumor growth and depends on the activation of endothelial cells through angiogenic factors (among the most important ones there are VEGF, HIF-1α, and TGFβ) [44]. Specifically, EXOs from GBM cells have been considered for their role in this process; such EXOs contain a lot of pro-angiogenic molecules (angiogenin, IL-6, IL-8, VEGF, TIMP-1/2) and are able to stimulate human brain microvascular endothelial cells tube formation in vitro [12]. EXOs are also able to promote proliferation and survival in endothelial cells activating cell surface receptor involved in angiogenic response, such as epidermal growth factor receptor (EGFR), vascular endothelial growth factor receptor 2 (VEGFR-2), and ephrin type A receptor 2 (EPHA2) [14]; moreover, once programmed by GBM EXOs, at least in hypoxic conditions, endothelial cells exhibit an enhanced ability to stimulate perycites survival and GBM cells proliferation and migration in a paracrine fashion [14].
In in vitro hypoxic conditions, GBM cells also release EXOs bearing the tissue factor (TF), which triggers the protease-activated receptor-2 (PAR-2) that in turn leads to an increased level of the pro-angiogenic molecules pERK1/2 and heparin-binding EGF-like growth factor (HB-EGF), resulting in tube formation stimulation [45].
Finally, it should be mentioned that some miRNAs carried in GBM EVs cargo, specifically miR-29a and miR-30e, also act as stimulators of angiogenesis [13].
So, although several studies on the pro-angiogenic properties of the whole GBM secretome (which include both soluble and vesicles-enclosed molecules) [46, 47] or on the EXOs only have been previously reported [12, 14, 45], our interest focus on the effect of the whole EVs population on angiogenesis.
Our studies by electron microscopy confirm the presence of both MVs and EXOs in human U251 GBM cells and highlight an abundant shed of heterogeneous in size MVs from the whole cell surface ranged between 150 and 800 nm.
These EVs convey several proteolytic enzymes, whose role in tumor growth and progression, both at primary and metastatic sites, has been widely highlighted: proteases secretion is positively correlated to tumor aggressiveness [48]. During cancer progression, indeed, tumor cells interact with surrounding microenvironment, i.e., extracellular matrix (to which growth factors and cytokines can be associated) and surrounding cells. All the typical hallmarks of cancer (migration, invasion, metastasis, and angiogenesis) depend on the ability of cancer cells to modify the surrounding microenvironment, and proteolytic enzymes, especially gelatinases, are critical molecules in cell-ECM interactions [49, 50]. Other studies have already shown that such enzymes can be carried by tumor extracellular vesicles, thus contributing to matrix remodeling and cancer progression [30–33, 51, 52]; the present paper confirms that U251 EVs carry both gelatinases and plasminogen activators and, specifically, gelatinases are contained in U251 EVs as pro-MMP-9, pro-MMP-2, and MMP-2, whereas plasminogen activators are represented by PAI-PA complexes, tPA, and high molecular weight uPA.
We also questioned whether the EVs could carry the CXCL12-chemokine-receptor CXCR4; it is expressed on a variety of normal cell types, such as hematopoietic cells, vascular endothelial cells, neurons, microglia, and astrocytes [53]; it is involved in several physiologic processes and migration response of CXCR4-expressing cells along CXCL12 gradients contributes to embryonic hematopoiesis, organogenesis, vascularization, and organ homeostasis [53]. CXCR4 is also expressed in more than 20 different types of human cancers (such as prostate, kidney, brain, breast, ovarian, small-cell lung, and pancreatic tumors) [54]; it promotes, directly and indirectly, the tumor progression [54] being fundamental, along with its ligand, for tumor cell proliferation, metastatic spread and therapeutic resistance, and for tumor angiogenesis too [54–57], and it is also expressed on cancer stem cells, contributing to cancer recurrence [53]. Its expression is a prognostic marker in various types of cancer, such as leukemia or breast carcinoma [54]; specifically, elevated levels have been correlated with high risk for recurrence and identified as a poor prognostic biomarker in many cancer patients (including lymphoma and leukemia, breast, lung, kidney, colon, ovarian, and brain cancers) [53, 58]. The CXCR4 presence has been already reported in EVs from bone marrow mesenchymal stem cells; they promote cardioprotection following myocardial infarction stimulating, among other things, angiogenesis [59].
Our assay shows that U251 EVs contain CXCR4 with molecular weight of approximately 43 and 77 kDa, suggesting the presence of both monomers and dimers; the dimers presence is intriguing since the CXCR4 homodimerization has been suggested to induce the JAK/STAT signaling pathway, which is known to promote changes in cell morphology that lead to chemotactic responses [53].
CXCR4, in fact, can be expressed in different isoforms in cells, sometimes in a predominant isoform and other times in multiple isoforms, glycosylated or non-glycosylated [60]. It exhibits, indeed, numerous post-translational modifications ranging from disulfide formation to tyrosine or serine chondroitin sulfation and including oligomerization and glycosylation, swinging the molecular weight between 40 and 110 kDa; 40 kDa molecules are believed to be monomers and 80 kDa as dimers [60].
To verify if EVs could affect angiogenic response, HBMEC cells were evaluated in term of proliferation, motility, and tube formation ability after EVs treatment. Data showed that U251 EVs promote cell proliferation in a dose-dependent manner; the more important effect is obtained after 72 h stimulating with 8 μg/ml EVs. Motility response, evaluated through a wound healing assay, had a similar trend; after 24 h, the wounds are more healed in the EVs treated points with respect to the untreated ones: specifically, cells treated with 8 μg/ml EVs almost completely closed the wound. Results obtained in the tube formation assay were also in line with the previous data confirming the ability of U251 EVs to stimulate endothelial cells activities in a dose-dependent manner. The proved pro-angiogenic abilities displayed by EVs could be explained, at least partially, in virtue of their VEGF content, as highlighted by ELISA, and TGFβ, analyzed by Western blot. VEGF, indeed, is well known for a long time as pro-angiogenic factors [44], and its release by tumor cells as a component of membrane vesicles has been already highlighted [61]; TGF-β also has potent effects on the function of endothelial cells affecting their proliferation, migration, and the extracellular matrix production [62].
In addition to angiogenesis stimulation, TGF-β, which is often abundant in tumor microenvironment, can affect tumor progression in several other ways since its signaling is involved in the regulation of proliferation, differentiation, and survival/apoptosis of all cells within the tumor microenvironment such as cancer cells, fibroblasts, endothelial cells, and immune cells [63, 64]. It can, also, affect the self-renewal of glioma-initiating stem cells and promote the immune suppression [63, 64]: TGF-β, in fact, enhances motility of monocytes, macrophages, NK cells, dendritic cells, and T cells increasing their recruitment in the tumor microenvironment but, in the meanwhile, directly inhibits their anti-tumor functions [64]. Since recruited cells, anyway, secrete growth factors, cytokines, chemokines, proteases, and extra-cellular matrix modifying enzymes, the result is an inflammation (which is a typical feature of GBMs) mediated by TGF-β that promotes tumorigenesis [64, 65].
So, altogether, our results indicate that human glioblastoma cells release, in vitro, extracellular vesicles fully equipped for angiogenesis stimulation in terms of proteolytic enzymes (gelatinases and plasminogen activators) and pro-angiogenic growth factors (VEGF and TGF-β); they also carry the CXCR4 chemokine receptor, which is already known for its promoting-angiogenic properties. To such molecules, at least partially, could be attributed the stimulating effects of U251 EVs on endothelial cells in term of proliferation, motility, and tube formation.
References
Veliz I, Loo Y, Castillo O, Karachaliou N, Nigro O, Rosell R. Advances and challenges in the molecular biology and treatment of glioblastoma-is there any hope for the future? Ann Transl Med. 2015;3(1):7.
Agnihotri S, Burrell KE, Wolf A, Jalali S, Hawkins C, Rutka JT, Zadeh G. Glioblastoma, a brief review of history, molecular genetics, animal models and novel therapeutic strategies. Arch Immunol Ther Exp. 2013;61(1):25–41.
Furnari FB, Fenton T, Bachoo RM, Mukasa A, Stommel JM, Stegh A, Hahn WC, Ligon KL, Louis DN, Brennan C, Chin L, DePinho RA, Cavenee WK. Malignant astrocytic glioma: genetics, biology, and paths to treatment. Genes Dev. 2007;21(21):2683–710.
Lathia JD, Heddleston JM, Venere M, Rich JN. Deadly teamwork: neural cancer stem cells and the tumor microenvironment. Cell Stem Cell. 2011;8(5):482–5.
Hochberg FH, Atai NA, Gonda D, Hughes MS, Mawejje B, Balaj L, Carter RS. Glioma diagnostics and biomarkers: an ongoing challenge in the field of medicine and science. Expert Rev Mol Diagn. 2014;14(4):439–52.
Charles NA, Holland EC, Gilbertson R, Glass R, Kettenmann H. The brain tumor microenvironment. Glia. 2011;59(8):1169–80.
Gatson NN, Chiocca EA, Kaur B. Anti-angiogenic gene therapy in the treatment of malignant gliomas. Neurosci Lett. 2012;527(2):62–70.
Charles N, Holland EC. The perivascular niche microenvironment in brain tumor progression. Cell Cycle. 2010;9(15):3012–21.
Godlewski J, Krichevsky AM, Johnson MD, Chiocca EA, Bronisz A. Belonging to a network-microRNAs, extracellular vesicles, and the glioblastoma microenvironment. Neuro-Oncology. 2015;17(5):652–62.
van der Vos KE, Balaj L, Skog J, Breakefield XO. Brain tumor microvesicles: insights into intercellular communication in the nervous system. Cell Mol Neurobiol. 2011;31(6):949–59.
Chistiakov DA, Chekhonin VP. Extracellular vesicles shed by glioma cells: pathogenic role and clinical value. Tumour Biol. 2014;35(9):8425–38.
Skog J, Würdinger T, van Rijn S, Meijer DH, Gainche L, Sena-Esteves M, Curry Jr WT, Carter BS, Krichevsky AM, Breakefield XO. Glioblastoma microvesicles transport RNA and proteins that promote tumour growth and provide diagnostic biomarkers. Nat Cell Biol. 2008;10(12):1470–6.
Li CC, Eaton SA, Young PE, Lee M, Shuttleworth R, Humphreys DT, Grau GE, Combes V, Bebawy M, Gong J, Brammah S, Buckland ME, Suter CM. Glioma microvesicles carry selectively packaged coding and non-coding RNAs which alter gene expression in recipient cells. RNA Biol. 2013;10(8):1333–44.
Kucharzewska P, Christianson HC, Welch JE, Svensson KJ, Fredlund E, Ringnér M, Mörgelin M, Bourseau-Guilmain E, Bengzon J, Belting M. Exosomes reflect the hypoxic status of glioma cells and mediate hypoxia-dependent activation of vascular cells during tumor development. Proc Natl Acad Sci U S A. 2013;110(18):7312–7.
Dolo V, D’Ascenzo S, Giusti I, Millimaggi D, Taraboletti G, Pavan A. Shedding of membrane vesicles by tumor and endothelial cells. Ital J Anat Embryol. 2005;110(2 Suppl 1):127–33.
Coticchio G, Borini A, Distratis V, Maione M, Scaravelli G, Bianchi V, Macchiarelli G, Nottola SA. Qualitative and morphometric analysis of the ultrastructure of human oocytes cryopreserved by two alternative slow cooling protocols. J Assist Reprod Genet. 2010;27(4):131–40.
Raposo G, Stoorvogel W. Extracellular vesicles: exosomes, microvesicles, and friends. J Cell Biol. 2013;200(4):373–83.
Shifrin Jr DA, Demory Beckler M, Coffey RJ, Tyska MJ. Extracellular vesicles: communication, coercion, and conditioning. Mol Biol Cell. 2013;24(9):1253–9.
Tetta C, Ghigo E, Silengo L, Deregibus MC, Camussi G. Extracellular vesicles as an emerging mechanism of cell-to-cell communication. Endocrine. 2013;44(1):11–9.
D’Souza-Schorey C, Clancy JW. Tumor-derived microvesicles: shedding light on novel microenvironment modulators and prospective cancer biomarkers. Genes Dev. 2012;26(12):1287–99.
György B, Szabó TG, Pásztói M, Pál Z, Misják P, Aradi B, László V, Pállinger E, Pap E, Kittel A, Nagy G, Falus A, Buzás EI. Membrane vesicles, current state-of-the-art: emerging role of extracellular vesicles. Cell Mol Life Sci. 2011;68(16):2667–88.
Turturici G, Tinnirello R, Sconzo G, Geraci F. Extracellular membrane vesicles as a mechanism of cell-to-cell communication: advantages and disadvantages. Am J Phys Cell Phys. 2014;306(7):C621–33.
Owens AP, Mackman N. Microparticles in hemostasis and thrombosis. Circ Res. 2011;108(10):1284–97.
Frühbeis C, Fröhlich D, Kuo WP, Krämer-Albers EM. Extracellular vesicles as mediators of neuron-glia communication. Front Cell Neurosci. 2013;7:182.
De Jong OG, Van Balkom BW, Schiffelers RM, Bouten CV, Verhaar MC. Extracellular vesicles: potential roles in regenerative medicine. Front Immunol. 2014;5:608.
Gaceb A, Martinez MC, Andriantsitohaina R. Extracellular vesicles: new players in cardiovascular diseases. Int J Biochem Cell Biol. 2014;50:24–8.
Buzas EI, György B, Nagy G, Falus A, Gay S. Emerging role of extracellular vesicles in inflammatory diseases. Nat Rev Rheumatol. 2014;10(6):356–64.
Candelario KM, Steindler DA. The role of extracellular vesicles in the progression of neurodegenerative disease and cancer. Trends Mol Med. 2014;20(7):368–74.
Webber J, Steadman R, Mason MD, Tabi Z, Clayton A. Cancer exosomes trigger fibroblast to myofibroblast differentiation. Cancer Res. 2010;70(23):9621–30.
Dolo V, D’Ascenzo S, Violini S, Pompucci L, Festuccia C, Ginestra A, Vittorelli ML, Canevari S, Pavan A. Matrix-degrading proteinases are shed in membrane vesicles by ovarian cancer cells in vivo and in vitro. Clin Exp Metastasis. 1999;17(2):131–40.
Dolo V, Ginestra A, Cassara D, Ghersi G, Nagase H, Vittorelli ML. Shed membrane vesicles and selective localization of gelatinases and MMP-9/TIMP-1 complexes. Ann N Y Acad Sci. 1999;878:497–9.
Angelucci A, D’Ascenzo S, Festuccia C, Gravina GL, Bologna M, Dolo V, Pavan A. Vesicle-associated urokinase plasminogen activator promotes invasion in prostate cancer cell lines. Clin Exp Metastasis. 2000;18(2):163–70.
Giusti I, D’Ascenzo S, Millimaggi D, Taraboletti G, Carta G, Franceschini N, Pavan A, Dolo V. Cathepsin B mediates the pH-dependent proinvasive activity of tumor-shed microvesicles. Neoplasia. 2008;10(5):481–8.
Abusamra AJ, Zhong Z, Zheng X, Li M, Ichim TE, Chin JL, Min WP. Tumor exosomes expressing Fas ligand mediate CD8+ T-cell apoptosis. Blood Cells Mol Dis. 2005;35(2):169–73.
Bebawy M, Combes V, Lee E, Jaiswal R, Gong J, Bonhoure A, Grau GE. Membrane microparticles mediate transfer of P-glycoprotein to drug sensitive cancer cells. Leukemia. 2009;23(9):1643–9.
Lv MM, Zhu XY, Chen WX, Zhong SL, Hu Q, Ma TF, Zhang J, Chen L, Tang JH, Zhao JH. Exosomes mediate drug resistance transfer in MCF-7 breast cancer cells and a probable mechanism is delivery of P-glycoprotein. Tumour Biol. 2014;35(11):10773–9.
Chen WX, Cai YQ, Lv MM, Chen L, Zhong SL, Ma TF, Zhao JH, Tang JH. Exosomes from docetaxel-resistant breast cancer cells alter chemosensitivity by delivering microRNAs. Tumour Biol. 2014;35(10):964959.
Kosaka N, Iguchi H, Hagiwara K, Yoshioka Y, Takeshita F, Ochiya T. Neutral sphingomyelinase 2 (nSMase2)-dependent exosomal transfer of angiogenic microRNAs regulate cancer cell metastasis. J Biol Chem. 2013;288(15):10849–59.
Hood JL, San RS, Wickline SA. Exosomes released by melanoma cells prepare sentinel lymph nodes for tumor metastasis. Cancer Res. 2011;71(11):3792–801.
Grange C, Tapparo M, Collino F, Vitillo L, Damasco C, Deregibus MC, Tetta C, Bussolati B, Camussi G. Microvesicles released from human renal cancer stem cells stimulate angiogenesis and formation of lung premetastatic niche. Cancer Res. 2011;71(15):5346–56.
Millimaggi D, Mari M, D’Ascenzo S, Carosa E, Jannini EA, Zucker S, Carta G, Pavan A, Dolo V. Tumor vesicle-associated CD147 modulates the angiogenic capability of endothelial cells. Neoplasia. 2007;9(4):34957.
Hong BS, Cho JH, Kim H, Choi EJ, Rho S, Kim J, Kim JH, Choi DS, Kim YK, Hwang D, Gho YS. Colorectal cancer cell-derived microvesicles are enriched in cell cycle-related mRNAs that promote proliferation of endothelial cells. BMC Genomics. 2009;10:556.
Zhuang G, Wu X, Jiang Z, Kasman I, Yao J, Guan Y, Oeh J, Modrusan Z, Bais C, Sampath D, Ferrara N. Tumour-secreted miR-9 promotes endothelial cell migration and angiogenesis by activating the JAK-STAT pathway. EMBO J. 2012;31(17):3513–23.
Sakurai T, Kudo M. Signaling pathways governing tumor angiogenesis. Oncology. 2011;81 Suppl 1:24–9.
Svensson KJ, Kucharzewska P, Christianson HC, Sköld S, Löfstedt T, Johansson MC, Mörgelin M, Bengzon J, Ruf W, Belting M. Hypoxia triggers a proangiogenic pathway involving cancer cell microvesicles and PAR-2-mediated heparin-binding EGF signaling in endothelial cells. Proc Natl Acad Sci U S A. 2011;108(32):1314752.
Venugopal C, Wang XS, Manoranjan B, McFarlane N, Nolte S, Li M, Murty N, Siu KW, Singh SK. GBM secretome induces transient transformation of human neural precursor cells. J Neuro-Oncol. 2012;109:457–66.
Tarassishin L, Lim J, Weatherly DB, Angeletti RH, Lee SC. Interleukin-1-induced changes in the glioblastoma secretome suggest its role in tumor progression. J Proteome. 2014;99:152–68.
Rakashanda S, Rana F, Rafiq S, Masood A, Amin S. Role of proteases in cancer: a review. Biotechnol Mol Biol Rev. 2012;7(4):90–101.
Gialeli C, Theocharis AD, Karamanos NK. Roles of matrix metalloproteinases in cancer progression and their pharmacological targeting. FEBS J. 2011;278(1):16–27.
Nakada M, Okada Y, Yamashita J. The role of matrix metalloproteinases in glioma invasion. Front Biosci. 2003;8:e261–9.
Ginestra A, La Placa MD, Saladino F, Cassarà D, Nagase H, Vittorelli ML. The amount and proteolytic content of vesicles shed by human cancer cell lines correlates with their in vitro invasiveness. Anticancer Res. 1998;18(5):3433–7.
Graves LE, Ariztia EV, Navari JR, Matzel HJ, Stack MS, Fishman DA. Proinvasive properties of ovarian cancer ascites-derived membrane vesicles. Cancer Res. 2004;64(19):7045–9.
Chatterjee S, Azad BB, Nimmagadda S. The intricate role of CXCR4 in cancer. Adv Cancer Res. 2014;124:31–82.
Burger JA, Kipps TJ. CXCR4: a key receptor in the crosstalk between tumor cells and their microenvironment. Blood. 2006;107(5):1761–7.
Wang J, He L, Combs CA, Roderiquez G, Norcross MA. Dimerization of CXCR4 in living malignant cells: control of cell migration by a synthetic peptide that reduces homologous CXCR4 interactions. Mol Cancer Ther. 2006;5(10):2474–83.
Teicher BA, Fricker SP. CXCL12 (SDF-1)/CXCR4 pathway in cancer. Clin Cancer Res. 2010;16(11):292731.
Gravina GL, Mancini A, Muzi P, Ventura L, Biordi L, Ricevuto E, Pompili S, Mattei C, Di Cesare E, Jannini EA, Festuccia C. CXCR4 pharmacogical inhibition reduces bone and soft tissue metastatic burden by affecting tumor growth and tumorigenic potential in prostate cancer preclinical models. Prostate. 2015;75(12):1227–46.
Rodríguez M, Silva J, Herrera A, Herrera M, Peña C, Martín P, Gil-Calderón B, Larriba MJ, Coronado MJ, Soldevilla B, Turrión VS, Provencio M, Sánchez A, Bonilla F, García-Barberán V. Exosomes enriched in stemness/metastatic-related mRNAS promote oncogenic potential in breast cancer. Oncotarget. 2015;6(38):40575–87.
Kang K, Ma R, Cai W, Huang W, Paul C, Liang J, Wang Y, Zhao T, Kim HW, Xu M, Millard RW, Wen Z, Wang Y. Exosomes secreted from CXCR4 overexpressing mesenchymal cells promote cardioprotection via Akt signaling pathway following myocardial infarction. Stem Cells Int. 2015;2015:659890.
Sloane AJ, Raso V, Dimitrov DS, Xiao X, Deo S, Muljadi N, Restuccia D, Turville S, Kearney C, Broder CC, Zoellner H, Cunningham AL, Bendall L, Lynch GW. Marked structural and functional heterogeneity in CXCR4: separation of HIV-1 and SDF-1alpha responses. Immunol Cell Biol. 2005;83(2):129–43.
Taraboletti G, D’Ascenzo S, Giusti I, Marchetti D, Borsotti P, Millimaggi D, Giavazzi R, Pavan A, Dolo V. Bioavailability of VEGF in tumor-shed vesicles depends on vesicle burst induced by acidic pH. Neoplasia. 2006;8(2):96–103.
Goumans MJ, Liu Z, ten Dijke P. TGF-beta signaling in vascular biology and dysfunction. Cell Res. 2009;19(1):116–27.
Kaminska B, Kocyk M, Kijewska M. TGF beta signaling and its role in glioma pathogenesis. Adv Exp Med Biol. 2013;986:171–87.
Bierie B, Moses HL. Transforming growth factor beta (TGF-beta) and inflammation in cancer. Cytokine Growth Factor Rev. 2010;21(1):49–59.
Kore RA, Abraham EC. Inflammatory cytokines, interleukin-1 beta and tumor necrosis factor-alpha, upregulated in glioblastomamultiforme, raise the levels of CRYAB in exosomes secreted by U373 glioma cell. Biochem Biophys Res Commun. 2014;453(3):326–31.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None
Rights and permissions
About this article
Cite this article
Giusti, I., Delle Monache, S., Di Francesco, M. et al. From glioblastoma to endothelial cells through extracellular vesicles: messages for angiogenesis. Tumor Biol. 37, 12743–12753 (2016). https://doi.org/10.1007/s13277-016-5165-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-016-5165-0