Abstract
Glioblastoma (GBM) is the most aggressive primary brain tumor in humans, with a uniformly poor prognosis. The tumor microenvironment is composed of both supportive cellular substrates and exogenous factors. We hypothesize that exogenous factors secreted by brain tumor initiating cells (BTICs) could predispose normal neural precursor cells (NPCs) to transformation. When NPCs are grown in GBM-conditioned media, and designated as “tumor-conditioned NPCs” (tcNPCs), they become highly proliferative and exhibit increased stem cell self-renewal, or the unique ability of stem cells to asymmetrically generate another stem cell and a daughter cell. tcNPCs also show an increased transcript level of stem cell markers such as CD133 and ALDH and growth factor receptors such as VEGFR1, VEGFR2, EGFR and PDGFRα. Media analysis by ELISA of GBM-conditioned media reveals an elevated secretion of growth factors such as EGF, VEGF and PDGF-AA when compared to normal neural stem cell-conditioned media. We also demonstrate that tcNPCs require prolonged or continuous exposure to the GBM secretome in vitro to retain GBM BTIC characteristics. Our in vivo studies reveal that tcNPCs are unable to form tumors, confirming that irreversible transformation events may require sustained or prolonged presence of the GBM secretome. Analysis of GBM-conditioned media by mass spectrometry reveals the presence of secreted proteins Chitinase-3-like 1 (CHI3L1) and H2A histone family member H2AX. Collectively, our data suggest that GBM-secreted factors are capable of transiently altering normal NPCs, although for retention of the transformed phenotype, sustained or prolonged secretome exposure or additional transformation events are likely necessary.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Studies of tumorigenesis suggest that neural stem cells (NSCs) may represent the most plausible targets for transformation [1]. Transformation is defined as the collective changes, such as uncontrolled cell proliferation, that convert a normal cell into a cancer cell [2].
A subpopulation of cells with stem cell properties termed brain tumor-initiating cells (BTICs) were isolated from glioblastoma (GBM) and were exclusively capable of driving brain tumor formation in a human–mouse xenograft model [3]. The invasive nature of this tumor is likely perpetuated by the GBM secretome [4], the cocktail of secreted factors released by GBM cells.
Given the invasiveness of GBM [5] and the long-standing presence of NSCs in the human brain [6], the occupancy of NSCs in the GBM tumor niche is likely. Moreover, neural progenitors have migratory capacities [7] and show tremendous tropism to regions of neural damage [8]. Not only have endogenous neural precursor cells (NPCs) been shown to migrate to the site of experimentally induced brain tumors [9], they can also be used to deliver therapeutic gene products to tumor cells [10]. NPC-based strategies may provide effective antitumor responses; however, their use in treatment would be compromised if they are vulnerable to transformation.
In this study, we have investigated the effects of the GBM secretome on NPCs by culturing them in BTIC-conditioned media. Our data suggest that factors secreted by GBM are capable of rendering normal NPCs vulnerable to transient transformation, although for retention of the transformed phenotype, sustained secretome exposure or additional transformation events are likely necessary.
Methods
Dissociation and culture of primary GBM tissue and human fetal brain
Human brain tumors (BT191, BT192, BT241) and fetal brain samples were obtained from consenting patients, as approved by the Hamilton Health Sciences/McMaster Health Sciences Research Ethics Board. Samples were dissociated in artificial cerebrospinal fluid containing 0.2 Wunisch U/mL Liberase Blendzyme 3 (Roche), and incubated at 37 °C in a shaker for 15 min. The dissociated tissue was then filtered through a 70 μm cell strainer and collected by centrifugation at 1,500 rpm for 3 min. Tumor cells were resuspended in tumor sphere medium (TSM), a chemically defined serum-free neural stem cell medium, and plated in ultra-low attachment plates (Corning). The components of our complete neural stem cell media per 500 mL include: Dulbecco’s modified Eagle’s medium/F12 (480 mL; Invitrogen), N2-supplement (5 mL; Invitrogen), 1 M HEPES (5 mL; Wisent), glucose (3 g; Invitrogen), N-acetylcysteine (1 mL of 60 mg/mL solution; Sigma) and neural survival factor-1 (10 mL; Lonza). Growth factors added to the media include human recombinant epidermal growth factor (20 ng/mL: Sigma), basic fibroblast growth factor (20 ng/ml; Invitrogen), leukemic inhibitory factor (10 ng/mL; Chemicon) and antibiotic–antimycotic (10 mg/mL; Wisent). Red blood cells were lysed using ammonium chloride solution (STEMCELL Technologies).
Acquisition of GBM conditioned media
GBM tumorspheres were grown in TSM and after 24–48 h of incubation, the tumorsphere containing media was centrifuged at 1,500 rpm for 3 min. The supernatant, which is the conditioned media, was collected and immediately filtered using a 0.2 μm filter, aliquoted and frozen at −80 °C.
Culture of NPCs in normal and GBM-conditioned media
NPC cultures were grown in TSM for 2 days prior to the treatment experiments. After two days, half of the neurospheres were grown in NPC-conditioned media (NPCs) which served as a control and the other half were grown in GBM-conditioned media (tcNPCs) for 7 days.
Sphere-formation assay
After primary sphere formation, spheres were dissociated, and single cells were plated at limiting dilution in 0.2 mL TSM. Total number of spheres per well were counted after seven days, and used to calculate the mean number of spheres formed per 2,000 cells, estimating stem cell frequency [11].
Cell proliferation assay
Cell proliferation assay was performed using Alamar Blue (Invitrogen), as per manufacturer’s instructions.
Differentiation assay and immunofluorescence (IF) staining
IF staining was performed as described previously [3, 12]. Briefly, the NPCs and tcNPCs were dissociated into a single cell suspension and cultured on coverslips in TSM without growth factors, supplemented with 20 % FBS. Media was replaced every other day. After 7 days, IF staining was performed. GFAP-Alexa 488 (1:100—Cell Signaling) and MAP2-Alexa 647 (1:5—BD Pharmingen) antibodies were incubated overnight at 4 °C. After staining with Hoechst 33342 (Molecular Probes), the slides were coverslipped with fluorescence mounting medium (DAKO). Images were taken using an Olympus spinning disk confocal microscope. Nuclei from ten high power fields were counted and positivity for lineage marker expression was quantified. Cells staining positive for both markers were not included in overall counts.
Flow cytometry analysis
Differentiated cells were stained for cell surface CD133-PE (Mitenyi Biotec; 1:20) followed by fixation and permeabilization using the BD Cytofix/Cytoperm™ Kit (BD Biosciences) and intracellular staining for MAP2-Alexa647 (BD Biosciences; 1:100) and GFAP-Alexa488 (Cell Signaling Technologies; 1:100). Appropriately matched isotype controls were used as negative controls. Cells were run on a Moflo XDP and analysed using Summit software (Beckman Coulter).
Real-time PCR
Total RNA was extracted using a RNeasy Micro kit (Qiagen) as per manufacturer’s instructions and quantified using the NanoDrop Spectrophotometer ND-1000. Complementary DNA was synthesized from 0.5 to 1 μg RNA and qScript cDNA Super Mix (Quanta) by a BIORAD C1000 Thermo Cycler (Bio-Rad) using the following protocol: 4 min at 25 °C, 30 min at 42 °C, 5 min at 85 °C, hold at 4 °C. qRT-PCR was performed using Perfecta Sybr Green (Quanta) on Opticon Chroma4 (Biorad). Gene expression analysis was quantified using Opticon software and expression levels were normalized to GAPDH expression. Primers are listed in Supplementary Table 1.
ELISA
Equal numbers of GBM cells and NPCs were grown in TSM media in triplicate for 2 days (3 biological samples). Following incubation, the filtered supernatant was tested for VEGF, EGF and PDGF-AA levels by Quantakine human ELISA kits according to manufacturer’s instructions (R & D Systems). Results are expressed as pg/mL of growth medium. The final values of growth factor concentration shown are subtracted values from control (i.e. TSM).
In vivo injections and immunostaining of xenografts
Xenografts from NPCs and tcNPCs were generated as previously described [3]. Briefly, the samples were injected into the right frontal lobe of NOD-CB17-SCID mice according to REB-approved protocols. Mice were injected with biological replicates of NPCs or tcNPCs (150,000–500,000 cells). The resulting human xenografts were fixed, embedded in paraffin, and stained with anti-human nestin (Millipore). Briefly, 4 μm formalin fixed, paraffin embedded tissue sections were dewaxed in xylene and rehydrated to distilled water. After antigen retrieval or unmasking procedures were applied, the slides were incubated with Anti-nestin (1:15,000) for 1 h. Endogenous peroxidase was blocked with 3 % hydrogen peroxide. The detection system used ImmPRESS polymer system (Vector Labs). After following kit instructions, color development was performed with freshly prepared DAB (DAKO). Finally, sections were counterstained lightly with Mayer’s Hematoxylin, dehydrated in alcohol, cleared in xylene and mounted with Permount mounting medium (Fisher).
Mass spectrometry analysis of GBM secretome
Acetone-precipitated GBM secretome proteins (50 μg) were digested with trypsin (Promega) using an established in-solution digestion protocol [13]. Peptides were separated by two-dimensional liquid chromatography with strong cation exchange being the first dimension, followed by reverse phase chromatography in the second dimension as previously published [14]. The MS/MS spectra were processed by Proteinpilot software version 2.0.1 (Paragon algorithm), against a concatenated Swissprot/Panther database of 66082 distinct human protein entries. Protein identification was performed using a confidence threshold of 95 % (Proteinpilot Unused score ≥1.31) with MMTS selected as cysteine modification, with search option emphasis on biological modifications.
Statistical analysis
Biological replicates from at least three patient samples were compiled for each experiment. Respective data represent mean ± SEM, n values are listed in figure legends. Student’s t test analyses were performed using GraphPad Prism 4.03. P < 0.05 was considered significant.
Results
tcNPCs show enhanced proliferation when compared to NPCs
Human fetal NPCs were grown in TSM and propagated as neurospheres in two different conditions: in normal NPC-conditioned media as a control to eliminate the ‘conditioned’ effects of normal cells, and in GBM-conditioned media. Cells grown in the latter conditions were designated as tcNPCs.
After 7 days in culture, tcNPCs formed significantly larger neurospheres (average = 212 μm) than NPCs (average = 110 μm) (Fig. 1a, b, P = 0.0001), indicating increased proliferation of tcNPCs compared to NPCs. This observation was further confirmed by the alamar blue cell proliferation assay (Fig. 1c, P = 0.010), suggesting that GBM-secreted factors increase the proliferation rate of NPCs. GBM tumorspheres (average = 289 μm) and proliferation of GBM are shown for comparison. Adhesive interactions are seen in a wide variety of biological processes including tumor invasion [15]. In the current study, we also observed that some tcNPC cells seem to acquire adhesive properties and grew adherently similar to primary GBM cells freshly isolated from a patient tumor (Supplementary Fig. 1).
The GBM secretome induces proliferation of tcNPCs. a Representative photomicrographs of NPCs, tcNPCs and GBM BTICs grown as spheres. b tcNPCs grown in GBM-conditioned media form bigger neurospheres than NPCs. (n = 3; P = 0.0001). GBM spheres are shown for comparison. c tcNPCs, like GBM show an increase in proliferation when compared to NPCs as depicted by alamar blue assay (n = 4; P = 0.010). RFU relative fluorescence units. *P < 0.05; ***P < 0.001
tcNPCs acquire BTIC properties
BTICs were originally identified as a subpopulation of CD133+ cells20 which exhibited stem cell properties both in vitro [12, 16] and in vivo [3, 17]. We assayed for CD133 transcript levels in both populations by RT-PCR and found that tcNPCs showed an increased expression of CD133 mRNA when compared to normal NPCs (Fig. 2a; P = 0.008). tcNPCs also showed elevated levels of aldehyde dehydrogenase (ALDH) mRNA (Fig 2a; P = 0.032), a marker shown to identify GBM BTIC populations [18] .
tcNPCs display enhanced neural stem cell properties. a tcNPCs grown in GBM-conditioned media show elevated mRNA expression of neural stem cell markers CD133 and ALDH as analyzed by RT-PCR (n = 4; P = 0.008 for CD133 and 0.032 for ALDH). b tcNPCs like GBM show a significant increase in secondary sphere formation when compared to NPCs by sphere formation assay (n = 6; P = 0.0067). #*P < 0.05; **P < 0.01
Limiting dilution analysis estimates the clonogenic stem cell frequency of a whole cell population by quantifying secondary sphere formation, which correlates directly with self-renewal capacity [3, 12]. We found that the self-renewal capacity of tcNPCs was significantly higher than NPCs, as measured by an increased number of secondary spheres (Fig. 2b; P = 0.0067).
tcNPCs show a decline in overall differentiation and a relative increase in neuronal marker expression
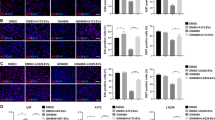
Differentiation is a key stem cell property, during which CD133+ stem and early progenitor cells lose their CD133 expression to give rise to late progenitors, and finally fully differentiated neural progeny [12]. We quantified CD133 protein expression of differentiated NPCs and tcNPCs by flow cytometry and found that CD133 is retained in a small percentage (7.1 ± 1.8) of tcNPCs when compared to NPCs (Fig. 3a; representative plot shown; P = 0.04). This indicates that a small percentage of tcNPC cells are refractory to differentiation. To further characterize the differentiated cells using IF, we stained both populations with antibodies against the mature neuronal marker MAP2 (microtubule-associated protein 2) and the astrocytic marker GFAP (glial fibrillary acidic protein). We found that tcNPCs exhibited an overall reduction in differentiation when compared to NPCs, and those tcNPCs that did differentiate showed increased MAP2 and relatively reduced GFAP expression (Fig. 3b, c, P = 0.0294 for MAP2 and P = 0.008 for GFAP). This qualitative observation was further confirmed by flow analysis of differentiated cells (Fig. 3d, P = 0.03 for MAP2 and P = 0.0248 for GFAP). Neuronal marker expression has been observed in GBM, and is associated with more aggressive GBM phenotypes and poor survival [19].
tcNPCs show reduced overall differentiation and an increase in neuronal marker expression a despite differentiation, tcNPCs were found to retain CD133 expression as analyzed by flow cytometry (n = 5; representative sample shown). b tcNPCs show reduced expression of astroglial marker GFAP (green) and increased expression of neuronal marker MAP2 (red) when compared to NPCs as shown by IF images (n = 3; representative images shown). c Quantification of immunofluorescence data shows reduced GFAP and increased MAP2 expression in tcNPCs compared to NPCs (n = 3; P = 0.008 for GFAP and P = 0.0294 for MAP2). d Flow analysis of differentiated tcNPCs show reduced expression of GFAP and increased expression of MAP2 (n = 5; P = 0.0248 for GFAP and P = 0.0314 for MAP2). #*P < 0.05; **P < 0.01
These findings show that the GBM secretome can promote acquisition of the BTIC marker CD133, which is accompanied by increased stemness, and can partially impede differentiation of NPCs.
GBM tumorspheres secrete higher levels of endothelial growth factor (EGF), vascular endothelial growth factor (VEGF) and Platelet-derived growth factor (PDGF) when compared to NPC neurospheres
The role of VEGF in glioma angiogenesis has been convincingly demonstrated [20] and VEGF is overexpressed in GBM. EGF and its receptor EGFR are thought to be important in mediating proliferative and transformation responses in numerous cancers [21, 22]. Elevated levels of PDGF-AA and its receptor expression are observed with a corresponding increase in glioma WHO grade [23].
In order to study the comparative levels of VEGF, EGF and PDGF-AA in the GBM and NPC secretomes, we quantified their levels by ELISA. We found that both VEGF (Fig. 4a; P = 0.0251), EGF (Fig. 4b; P = 0.006) and PGDF (Fig. 4c; P = 0.0001) levels were higher in GBM-conditioned media compared to NPC-conditioned media. Since growth factors modulate their activity through their cognate receptors, we next analyzed the transcript levels of receptors of VEGF, EGF and PDGF: VEGFR1 and VEGFR2, EGFR and PDGFRα respectively by RTPCR. We found that tcNPCs show an increased level of receptor expression when compared to NPCs (Fig. 4d; P = 0.009 for VEGFR1, 0.01 for VEGFR2 and Fig. 4e; P = 0.04 for EGFR). We also found an increase in the PDGFRα expression in tcNPCs (Fig. 4f; P = 0.04). This data confirms that the GBM secretome contains increased levels of growth factors such as VEGF, EGF and PGDF-AA and tcNPCs grown in GBM-conditioned media respond to secretome exposure by increasing cognate receptor expression.
GBM tumorspheres secrete elevated levels of growth factors VEGF and EGF when compared to NPCs. a VEGF, b EGF, c PDGF-AA levels were elevated in the GBM secretome media when compared to the normal NPC secretome as shown by ELISA (P = 0.0251 for VEGF, 0.006 for EGF and 0.0001 for PDGF-AA). RTPCR data reveals that tcNPCs show an elevated mRNA expression of d VEGF receptors VEGFR1 (n = 3, P = 0.009) and VEGFR2 (n = 3, 0.010). e EGF receptor EGFR levels (n = 3, P = 0.04) and f PDBF-AA receptor PDGFR alpha when compared to NPCs (n = 3, P = 0.04). #*P < 0.05, **P < 0.01, ***P < 0.001
tcNPCs require prolonged or continuous exposure to the GBM secretome to retain features of transformation
Results of our in vivo studies showed that although both NPCs and tcNPCs cells were observed in xenografts (Fig. 5a), tcNPCs did not form discrete tumors in our model system. As a positive control, we have shown xenografts generated by intracranial injections of 10,000 cells of primary GBM BT241 (Supplementary Fig. 2). However, we observed intracranially injected human tcNPCs integrating into host murine blood vessels (Fig. 5b), whereas normal NPCs did not. Insulin-like growth factor-binding protein 7 (IGFBP7), a selective biomarker of GBM vessels, was found to be upregulated in human brain endothelial cells exposed to GBM secretome [24]. In the current work, RT-PCR analysis revealed increased mRNA levels of IGFBP7 in tcNPCs (Supplementary Fig. 3, P = 0.03).
tcNPCs require prolonged or continuous exposure to the GBM secretome to retain features of transformation. a Immunohistochemistry of xenografts from mice injected tcNPCs (n = 7) stained for anti-human nestin and hematoxylin shows the presence of nestin-positive human cells (brown cells, red arrows) without formation of a discrete tumor mass at 6 months post-injection. Representative images of control mouse brain (left panel) and tcNPC xenograft (right panel) are shown (Scale bar 100 μm). Non-reactive nuclei stain blue. b Anti-human nestin staining of NPC and tcNPC xenografts show that tcNPCs integrate into host murine blood vessels (right panel). NPC xenografts are shown in left panel (Scale bar 100 μm). c tcNPCs growth in GBM-conditioned media yielded comparable viability as revealed by dye exclusion assay, even when grown for 2 weeks in GBM-conditioned media (n = 5). d tcrNPCs (tcNPCs reverted back to normal NPC-conditioned media) show decreased viability as measured by dye exclusion assay (n = 6, P = 0.0001) and e decreased proliferation as measured by alamar blue assay when they are reverted back to normal NPC-conditioned media (n = 3, P = 0.007). RFU relative fluorescence units. **P < 0.01, ***P < 0.001
Our in vivo studies showed that BTIC properties acquired by tcNPCs may be transient. In order to verify this effect, we performed in vitro studies on tcNPCs re-cultured in normal media. Sphere-formation and proliferation assays performed after 14 days of tcNPCs grown in GBM-conditioned media yielded similar results as observed earlier, with comparable viability (Fig. 5c). However, tcNPCs grown in GBM-conditioned media for a week and then reverted back to normal conditions for 7 days (designated as tcrNPCs) showed decreased viability (Fig. 5d, P = 0.0001) and a decrease in proliferation (Fig. 5e, P = 0.007). It is possible that tcNPCs may have acquired dependence on GBM secreted factors for survival and proliferation. This indicates that sustained exposure of GBM-conditioned media may be required for tcNPCs to show a sustained progression toward transformation.
GBM secretome analysis by mass spectrometry
We next wanted to identify key proteins present in GBM-conditioned media that may play a role in induction of transformation of NPCs. The high concentration of bovine serum albumin (BSA), which served as a stabilization factor for various supplemented neural growth proteins in media, posed a significant challenge for MS analysis and repeated efforts in immunoextraction of BSA prior to MS analyses were only partially successful. However, we were able to identify two interesting proteins, Chitinase-3-like protein 1 (CHI3L1; also known as YKL40) and Histone H2A-X, in GBM-conditioned media (Supplementary Fig. 4). CHI3L1 plays a known role in regulation of malignant transformation and local invasiveness of gliomas [25]. Histone H2A-X, one of four isoproteins of the core histone H2A [26, 27] is reported to be a secretory protein which not only organizes chromatin architecture, but also regulates growth through the cell cycle [28].
Discussion
Tumor cells take advantage of the neighboring stromal environment through recruitment of surrounding normal cells which provide physiological resources to aid tumor progression [29]. Although the tumor microenvironment is known to be regulated by both autocrine and paracrine factors secreted by tumor cells, the interaction between tumor cells and surrounding normal cells is otherwise poorly characterized. During malignant transformation, cancer cells are thought to gain mutations that supersede normal mechanisms controlling cellular proliferation [30]. Since normal somatic stem cells must self-renew and maintain a relative balance between self-renewal and differentiation, cancer can be conceptualized as a disease of unregulated self-renewal [31]. NSCs possess self-renewal machinery that is primed and could be harnessed to create a cancer cell [31, 32] and their longevity predisposes them to the accumulation of genetic mutations.
Various studies show that altered NSCs could initiate gliomagenesis. For example, Bachoo et al. [33] showed that transduction of NSC with EGFRvIII can induce glioma-like lesions and Holland et al. [34], have shown combined activation of Ras and Akt signaling pathways to induce GBM formation. Gene expression patterns of myoepithelial cells derived from a normal breast niche compared to those surrounding invasive breast carcinoma were significantly different, suggesting that the cancer cells can trigger changes in host tissues to promote invasion [35]. In the same way, it is likely that GBM cells can induce changes in neighboring NSCs to help in disease progression. For example, self-renewal, a complicated, tightly controlled phenomenon restricted to stem cells, was found to be enhanced in tcNPCs (Fig. 2b). Although the regulatory mechanisms of self-renewal in NSCs are not yet clearly delineated, it seems that secreted factors from GBMs may play a role in enhancing the NSC self-renewal machinery. tcNPCs exhibited an overall reduction in differentiation when compared to NPCs as measured by retention of primitive stem cell marker CD133 (Fig. 3a). Those tcNPCs that did differentiate showed increased neuronal and relatively reduced astrocytic differentiation compared to normal NPCs (Fig. 3b).
Angiogenesis is a characteristic feature of solid tumors and aids in providing oxygen and nutrition [36]. It has been shown that CD133 + BTICs facilitate angiogenesis by increasing the secretion of the prominent pro-angiogenic factor, VEGF [37]. We found that GBM grown in BTIC-enriching TSM release increased levels of VEGF when compared to normal NPCs (Fig. 3a). The EGF signaling pathway also plays an important role in gliomagenesis and NSC regulation [38], and we show that GBMs release enhanced levels of EGF while tcNPCs show an elevated expression of its cognate receptor EGFR (Fig. 3d). Amplification of the EGFR gene is observed in the classical subtype of GBM [39], and EGFR activation is thought to drive the transformation process in this GBM subtype [40, 41].
We observed both NPCs and tcNPCs cells to be present in xenografts (Fig. 5a), but tcNPCs did not form discrete tumors in our model system. This shows that the exposure of NPCs to GBM secretome exerts a transient effect on tcNPCs. NSCs are capable of differentiation toward the endothelial lineage [42] and CD133 + BTICs in GBM are capable of forming vessel-like structures positive for the endothelial marker CD31 [17, 43, 44]. We observed intracranially injected human tcNPCs integrating into host murine blood vessels (Fig. 5b), whereas normal NPCs did not. Our data suggests that through exposure to growth factors (such as VEGF) in GBM-conditioned media, the tcNPCs may have acquired an expanded differentiation capacity, and could contribute to the tumor vasculature.
Proteomics allows for the identification of extracellular proteins by means of LC–MS/MS [45]. Our mass spectrometric analysis of GBM-conditioned media revealed the presence of CHI3L1and H2AX (Supplementary Fig. 4). CH3L1 levels have been shown to increase with tumor grade in GBM [46, 47] and have been postulated as a marker for glioma subtypes [48, 49].
Although our data suggests that normal NPCs are vulnerable to initial transformation effects when exposed to GBM-conditioned media, tcNPCs do not appear to be fully transformed, or equivalent to BTICs. Indeed, transformation should be considered a spectrum of cellular changes, the endpoint of which is neoplasia. tcNPCS may be shifted further down this gradient towards transformation through exposure to the GBM secretome. Studies carried out with conditioned media can only address the paracrine effects of adjacent cells in a system. Limitations of the current study include lack of data examining cell–cell interactions, another major component of the tumor microenvironment. To comprehensively study the complex interactions of tumor cells with adjacent cells and recapitulate a natural tumor niche, future co-culture studies are required.
References
Koestner A, Swenberg JA, Wechsler W (1971) Transplacental production with ethylnitrosourea of neoplasms of the nervous system in Sprague–Dawley rats. Am J Pathol 63:37–56
Werbowetski-Ogilvie TE, Schnerch A, Rampalli S, Mills CE, Lee JB et al (2011) Evidence for the transmission of neoplastic properties from transformed to normal human stem cells. Oncogene 30:4632–4644
Singh SK, Hawkins C, Clarke ID, Squire JA, Bayani J et al (2004) Identification of human brain tumour initiating cells. Nature 432:396–401
Formolo CA, Williams R, Gordish-Dressman H, MacDonald TJ, Lee NH et al (2011) Secretome signature of invasive glioblastoma multiforme. J Proteome Res 10:3149–3159
Huse JT, Holland EC (2010) Targeting brain cancer: advances in the molecular pathology of malignant glioma and medulloblastoma. Nat Rev Cancer 10:319–331
Sutter R, Yadirgi G, Marino S (2007) Neural stem cells, tumour stem cells and brain tumours: dangerous relationships? Biochim Biophys Acta 1776:125–137
Fricker RA, Carpenter MK, Winkler C, Greco C, Gates MA et al (1999) Site-specific migration and neuronal differentiation of human neural progenitor cells after transplantation in the adult rat brain. J Neurosci 19:5990–6005
Aboody KS, Brown A, Rainov NG, Bower KA, Liu S et al (2000) Neural stem cells display extensive tropism for pathology in adult brain: evidence from intracranial gliomas. Proc Natl Acad Sci USA 97:12846–12851
Assanah MC, Bruce JN, Suzuki SO, Chen A, Goldman JE et al (2009) PDGF stimulates the massive expansion of glial progenitors in the neonatal forebrain. Glia 57:1835–1847
Aboody KS, Najbauer J, Schmidt NO, Yang W, Wu JK et al (2006) Targeting of melanoma brain metastases using engineered neural stem/progenitor cells. Neuro Oncol 8:119–126
Wang X, Venugopal C, Manoranjan B, McFarlane N, O’Farrell E et al (2012) Sonic hedgehog regulates Bmi1 in human medulloblastoma brain tumor-initiating cells. Oncogene 31:187–199
Singh SK, Clarke ID, Terasaki M, Bonn VE, Hawkins C et al (2003) Identification of a cancer stem cell in human brain tumors. Cancer Res 63:5821–5828
Li H, DeSouza LV, Ghanny S, Li W, Romaschin AD et al (2007) Identification of candidate biomarker proteins released by human endometrial and cervical cancer cells using two-dimensional liquid chromatography/tandem mass spectrometry. J Proteome Res 6:2615–2622
DeSouza L, Diehl G, Rodrigues MJ, Guo J, Romaschin AD et al (2005) Search for cancer markers from endometrial tissues using differentially labeled tags iTRAQ and cICAT with multidimensional liquid chromatography and tandem mass spectrometry. J Proteome Res 4:377–386
Hynes RO (1987) Integrins: a family of cell surface receptors. Cell 48:549–554
Reynolds BA, Weiss S (1992) Generation of neurons and astrocytes from isolated cells of the adult mammalian central nervous system. Science 255:1707–1710
Wang R, Chadalavada K, Wilshire J, Kowalik U, Hovinga KE et al (2010) Glioblastoma stem-like cells give rise to tumour endothelium. Nature 468:829–833
Rasper M, Schafer A, Piontek G, Teufel J, Brockhoff G et al (2010) Aldehyde dehydrogenase 1 positive glioblastoma cells show brain tumor stem cell capacity. Neuro Oncol 12:1024–1033
Donev K, Scheithauer BW, Rodriguez FJ, Jenkins S (2010) Expression of diagnostic neuronal markers and outcome in glioblastoma. Neuropathol Appl Neurobiol 36:411–421
Plate KH, Breier G, Weich HA, Risau W (1992) Vascular endothelial growth factor is a potential tumour angiogenesis factor in human gliomas in vivo. Nature 359:845–848
Alroy I, Yarden Y (1997) The ErbB signaling network in embryogenesis and oncogenesis: signal diversification through combinatorial ligand-receptor interactions. FEBS Lett 410:83–86
Carpenter G, Cohen S (1979) Epidermal growth factor. Annu Rev Biochem 48:193–216
Shih AH, Holland EC (2006) Platelet-derived growth factor (PDGF) and glial tumorigenesis. Cancer Lett 232:139–147
Pen A, Moreno MJ, Durocher Y, Deb-Rinker P, Stanimirovic DB (2008) Glioblastoma-secreted factors induce IGFBP7 and angiogenesis by modulating Smad-2-dependent TGF-beta signaling. Oncogene 27:6834–6844
Ku BM, Lee YK, Ryu J, Jeong JY, Choi J et al (2011) CHI3L1 (YKL-40) is expressed in human gliomas and regulates the invasion, growth and survival of glioma cells. Int J Cancer 128:1316–1326
West MH, Bonner WM (1980) Histone 2A, a heteromorphous family of eight protein species. Biochemistry 19:3238–3245
West MH, Bonner WM (1983) Structural comparisons of mouse histones 2A.X and 2A.Z with 2A.1 and 2A.2. Comp Biochem Physiol B 76:455–464
Watabe Y, Kuramochi H, Furuya Y, Inagaki N, Seino S et al (1996) Identification of histone H2A.X as a growth factor secreted by an androgen-independent subline of mouse mammary carcinoma cells. J Biol Chem 271:25126–25130
Charles NA, Holland EC, Gilbertson R, Glass R, Kettenmann H (2011) The brain tumor microenvironment. Glia 59:1169–1180
Hahn WC, Counter CM, Lundberg AS, Beijersbergen RL, Brooks MW et al (1999) Creation of human tumour cells with defined genetic elements. Nature 400:464–468
Reya T, Morrison SJ, Clarke MF, Weissman IL (2001) Stem cells, cancer, and cancer stem cells. Nature 414:105–111
Passegue E, Jamieson CH, Ailles LE, Weissman IL (2003) Normal and leukemic hematopoiesis: are leukemias a stem cell disorder or a reacquisition of stem cell characteristics? Proc Natl Acad Sci USA 100(Suppl 1):11842–11849
Bachoo RM, Maher EA, Ligon KL, Sharpless NE, Chan SS et al (2002) Epidermal growth factor receptor and Ink4a/Arf: convergent mechanisms governing terminal differentiation and transformation along the neural stem cell to astrocyte axis. Cancer Cell 1:269–277
Holland EC, Celestino J, Dai C, Schaefer L, Sawaya RE et al (2000) Combined activation of Ras and Akt in neural progenitors induces glioblastoma formation in mice. Nat Genet 25:55–57
Allinen M, Beroukhim R, Cai L, Brennan C, Lahti-Domenici J et al (2004) Molecular characterization of the tumor microenvironment in breast cancer. Cancer Cell 6:17–32
Li Z, Wang H, Eyler CE, Hjelmeland AB, Rich JN (2009) Turning cancer stem cells inside out: an exploration of glioma stem cell signaling pathways. J Biol Chem 284:16705–16709
Bao S, Wu Q, Sathornsumetee S, Hao Y, Li Z et al (2006) Stem cell-like glioma cells promote tumor angiogenesis through vascular endothelial growth factor. Cancer Res 66:7843–7848
Sanai N, Alvarez-Buylla A, Berger MS (2005) Neural stem cells and the origin of gliomas. N Engl J Med 353:811–822
Verhaak RG, Hoadley KA, Purdom E, Wang V, Qi Y et al (2010) Integrated genomic analysis identifies clinically relevant subtypes of glioblastoma characterized by abnormalities in PDGFRA, IDH1, EGFR, and NF1. Cancer Cell 17:98–110
Maher EA, Furnari FB, Bachoo RM, Rowitch DH, Louis DN et al (2001) Malignant glioma: genetics and biology of a grave matter. Genes Dev 15:1311–1333
Wechsler-Reya R, Scott MP (2001) The developmental biology of brain tumors. Annu Rev Neurosci 24:385–428
Wurmser AE, Nakashima K, Summers RG, Toni N, D’Amour KA et al (2004) Cell fusion-independent differentiation of neural stem cells to the endothelial lineage. Nature 430:350–356
Ricci-Vitiani L, Pallini R, Biffoni M, Todaro M, Invernici G et al (2010) Tumour vascularization via endothelial differentiation of glioblastoma stem-like cells. Nature 468:824–828
Zhao Y, Dong J, Huang Q, Lou M, Wang A et al (2010) Endothelial cell transdifferentiation of human glioma stem progenitor cells in vitro. Brain Res Bull 82: 308-312
Chan CY, Masui O, Krakovska O, Belozerov VE, Voisin S et al (2011) Identification of differentially regulated secretome components during skeletal myogenesis. Mol Cell Proteomics 10: M110 004804
Nigro JM, Misra A, Zhang L, Smirnov I, Colman H et al (2005) Integrated array-comparative genomic hybridization and expression array profiles identify clinically relevant molecular subtypes of glioblastoma. Cancer Res 65:1678–1686
Zhang W, Murao K, Zhang X, Matsumoto K, Diah S et al (2010) Resveratrol represses YKL-40 expression in human glioma U87 cells. BMC Cancer 10: 593
Hormigo A, Gu B, Karimi S, Riedel E, Panageas KS et al (2006) YKL-40 and matrix metalloproteinase-9 as potential serum biomarkers for patients with high-grade gliomas. Clin Cancer Res 12:5698–5704
Rousseau A, Nutt CL, Betensky RA, Iafrate AJ, Han M et al (2006) Expression of oligodendroglial and astrocytic lineage markers in diffuse gliomas: use of YKL-40, ApoE, ASCL1, and NKX2-2. J Neuropathol Exp Neurol 65:1149–1156
Acknowledgments
This work was supported by funds from the Department of Surgery at McMaster University, and the Ontario Institute for Cancer Research (OICR).
Conflict of interest
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Venugopal, C., Wang, X.S., Manoranjan, B. et al. GBM secretome induces transient transformation of human neural precursor cells. J Neurooncol 109, 457–466 (2012). https://doi.org/10.1007/s11060-012-0917-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-012-0917-1