Abstract
Purpose
In diabetics, elevated preoperative hemoglobin A1c (HbA1c) levels are associated with increased complication rates after cardiac surgery. While many non-diabetics also have elevated HbA1c, the relationship with outcome in these patients is not well understood. Therefore, in a cohort of non-diabetic patients, we tested the hypothesis that preoperative HbA1c is associated with early mortality risk after cardiac surgery.
Methods
In this retrospective observational study, we accessed data from a prospectively collected quality assurance database for a cohort of 1,474 non-diabetic elective cardiac surgery patients with documented preoperative HbA1c levels. The relationship of HbA1c with death within 30 days of surgery was examined using logistic regression modeling. Acute kidney injury and infection were similarly assessed using multivariable linear and logistic regression.
Results
Thirty-one percent of patients (n = 456) had elevated HbA1c values (>6.0%). Patients with elevated HbA1c had higher fasting and peak intraoperative blood glucose values. Also, an elevated HbA1c level was independently associated with increased 30-day mortality (odds ratio 1.53 per percent increase [1.24-1.91]; P = 0.0005). This relationship persisted even after “borderline” diabetics were excluded. Furthermore, acute kidney injury was associated with elevated baseline HbA1c (P = 0.01). No association was found between HbA1c and postoperative infection risk (P = 0.48).
Conclusion
In non-diabetics, an elevated preoperative HbA1c level (>6.0%) is independently associated with significantly greater early mortality risk after elective cardiac surgery. Our findings suggest that HbA1c may have value as a screening tool to identify high-risk non-diabetic cardiac surgery patients.
Rèsumè
Objectif
Chez les diabétiques, des niveaux élevés d’hémoglobine A1c (HbA1c) préopératoire sont associés à une incidence accrue de complications après une chirurgie cardiaque. Alors que nombre de personnes non diabétiques ont également des niveaux élevés de HbA1c, la relation entre ces niveaux et le devenir de ces patients demeure mal comprise. Pour cette raison, dans une cohorte de patients non diabétiques, nous avons testé l’hypothèse selon laquelle la HbA1c préopératoire est associée à un risque de mortalité précoce après une chirurgie cardiaque.
Méthode
Dans cette étude observationnelle rétrospective, nous avons utilisé des données tirées d’une base de données d’assurance de la qualité récoltées de façon prospective et portant sur une cohorte de 1474 patients non diabétiques devant subir une chirurgie cardiaque non urgente et dont les niveaux préopératoires de HbA1c étaient documentés. La relation entre les niveaux de HbA1c et le décès dans les 30 jours suivant la chirurgie a été examinée à l’aide d’un modèle de régression logistique. Les lésions rénales aiguës et les infections ont été évaluées de la même façon à l’aide de modèles de régression logistique et linéaire multivariée.
Résultats
Trente et un pour cent des patients (n = 456) présentaient des valeurs de HbA1c élevées (>6,0%). Chez les patients présentant des valeurs de HbA1c élevées, la glycémie était plus élevée à jeun et atteignait des valeurs maximales plus grandes pendant l’opération. De plus, un niveau élevé de HbA1c était associé de façon indépendante à une mortalité accrue à 30 jours (rapport de cotes 1,53 par augmentation de pour cent [1,24-1,91]; P = 0,0005). Cette relation est demeurée apparente même après l’exclusion des diabétiques «limite» du calcul. En outre, les lésions rénales aiguës étaient associées à des valeurs de HbA1c de base élevées (P = 0,01). Aucune association n’a été découverte entre la HbA1c et le risque d’infection postopératoire (P = 0,48).
Conclusion
Chez les personnes non diabétiques, un niveau préopératoire élevé de HbA1c (>6,0%) est associé de façon indépendante à un risque de mortalité précoce significativement plus élevé après une chirurgie cardiaque non urgente. Nos résultats suggèrent que le niveau de HbA1c pourrait être utile comme outil de dépistage afin d’identifier les patients de chirurgie cardiaque non diabétiques mais à risque élevé.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Coronary artery disease is the leading cause of death in the United States.1 Current cardiac surgery risk stratification models weigh heavily towards patient characteristics and chronic conditions that are not amenable to optimization.2 - 4 While diabetics are a high-risk group for early complication following cardiac surgery,5 - 7 some have suggested that tight postoperative glycemic control may improve outcome, though this is somewhat controversial.8 - 12
Monitoring glycosylated hemoglobin (hemoglobin A1c, HbA1c) is a recommended method for assessing the long-term effectiveness of diabetes therapies,13 and it has prognostic value as well;14 lower HbA1c levels are associated with better glycemic control, and they also predict reduced long-term complication rates, e.g., nephropathy and retinopathy, and forecast improved survival. In diabetics requiring cardiac surgery, lower HbA1c levels correlate with lower adverse event rates and 30-day mortality risk.15 , 16 Although the role of HbA1c as a marker of complication risk is established for diabetics, studies exploring its value in non-diabetics are limited.17 , 18
Thirty percent of non-diabetics undergoing percutaneous coronary interventions have elevated HbA1c levels, and these patients are at higher risk of cardiac mortality and adverse events.19 Complication rates are also higher in non-diabetic vascular surgery patients with elevated HbA1c.18 While elevated HbA1c levels are also common in non-diabetic cardiac surgery patients (57% in one study),20 the relationship between HbA1c and perioperative mortality risk is not well understood. Therefore, to assess HbA1c further as a preoperative screening tool, we tested the hypothesis that elevated HbA1c is associated with early mortality risk in non-diabetic patients following elective cardiac surgery.
Methods
Study population
Following institutional review board approval and in compliance with the Health Insurance Portability and Accountability Act, we reviewed the Duke Cardiovascular Database, automated anesthesia records, and an electronic laboratory results archive for all non-diabetic adults undergoing primary non-emergent cardiac surgery at Duke Heart Center from January 1, 1999 to December 31, 2005.
Demographics and clinical covariates
The following information was obtained from the prospectively gathered Duke Quality Measurement and Management Institute database and an intraoperative automated anesthesia recordkeeping system: patient and procedural characteristics, postoperative outcome data, and variables required for the Parsonnet score,3 including age, gender, body habitus, preoperative cardiac status (left ventricular ejection fraction/aneurysm, redo surgery, need for intra-aortic balloon counterpulsation), history of hypertension, diabetes, renal failure requiring dialysis and other major medical conditions.
Patients with diabetes were defined as those with a history of either type 1 or type 2 diabetes mellitus and/or those taking either insulin and/or oral hypoglycemic agents. The diagnosis of diabetes was determined from the patient’s admission history and medications and, therefore, did not involve index admission HbA1c or fasting blood glucose values. With the exception of morning medications taken with a small sip of water, all patients were fasted preoperatively for at least six hours for solids and four hours for clear fluids. Approximately 45% of cases involved a more extended fast due to an afternoon start time. To assure conformity, the baseline glucose value was the first blood glucose assessment recorded in the anesthesia record, as routinely assessed at the study institution after placement of the arterial catheter but prior to induction of general anesthesia. Arterial and intravenous line placement usually occurred concurrently in an induction room adjacent to the operating room. Then, according to routine, a certified anesthesia technologist performed point-of-care arterial blood gas determination, including serum glucose assessment, and hand entered the particulars into the automated anesthesia record. Any related fluid infusions were modest in volume, typically Ringer’s lactate or 0.9% sodium chloride, and would not be expected to affect the determination of serum glucose significantly.
As per institutional protocol, preoperative and daily postoperative serum creatinine values were measured until hospital discharge. Missing post-discharge serum creatinine values from postoperative days four to ten were imputed and were assumed to equal the final in-hospital serum creatinine values. Serum creatinine was measured using a dry-slide enzymatic reflectance technique (Vitros 950, Johnson and Johnson, New Brunswick, NJ, USA) with a normal range of 44-133 μmol·L−1 (0.5-1.5 mg·dL−1). Preoperative creatinine was obtained within one week prior to surgery and defined as the value recorded closest to but not on the day of surgery.
Intraoperative management
All patients received general anesthesia and conventional cardiac surgery through a midline sternotomy. The agents that were used in the induction and maintenance of anesthesia were at the discretion of the attending anesthesiologist and most commonly involved doses of intravenous midazolam, fentanyl, propofol, and pancuronium. Supplemental isoflurane (0.5-1.0%) was used as required to maintain heart rate and mean arterial pressure within 25% of pre-induction values, and propofol was infused at 20-30 ug·kg−1·min−1 at the start of rewarming during cardiopulmonary bypass. Extracorporeal perfusion was established using a non-pulsatile hypothermic (28-34°C) flow of 2.0-2.4 L·min−1·m−2, with mean arterial pressure maintained from 50-70 mmHg and α-stat blood gas management. A cold blood cardioplegia solution was utilized for myocardial protection and infused either antegrade, retrograde, or both, according to the clinical circumstance. High- and low-dose potassium blood cardioplegia was created by mixing a blood and cardioplegic solution 4:1. The cardioplegic solution contained 5% dextrose and additives.21 Standard cardioplegia strategy included an induction dose of 1,000 mL at the time of the aortic cross-clamp application (5% dextrose solution 200 mL), and 500 mL (5% dextrose solution 100 mL) every 20 min thereafter until aortic cross-clamp removal. During the time period of this study, our centre’s standard of care for antifibrinolytic therapy was aminocaproic acid, with aprotonin being used for high-risk (primarily redo) procedures.
Perioperative management of glucose and antibiotics
Intraoperative management included serial measurement of blood glucose every 30 min during extracorporeal perfusion and intravenous insulin by bolus and infusion to maintain a target serum glucose below 200 mg·dL−1 (11.1 mmol·L−1). In the postoperative intensive care unit, normoglycemia was achieved following the Duke cardiac surgery insulin protocol. The protocol targets a blood glucose level from 120-140 mg·dL−1 (6.7-7.8 mmol·L−1), and blood glucose levels are measured hourly. Transition away from the intravenous insulin protocol and subsequent glucose management is at the discretion of the cardiac surgical service.
Perioperative antibiotic prophylaxis for non-diabetic patients admitted from home on the day of surgery consisted of cefuroxime 1.5 g iv within one hour prior to skin incision, on separation from cardiopulmonary bypass, and every eight hours for a total duration of 48 hr. In addition, vancomycin 20 mg·kg−1 (up to a maximum 2 g) was administered to inpatients and those transferred hospital-to-hospital from another referral facility. Intravenous cefuroxime was diluted in normal saline, and vancomycin hydrochloride was infused in a 100 mL ready-to-use infusion bag that contained approximately 5 g of dextrose.
Endpoints
The primary clinical endpoint was death within 30 days of surgery. The two secondary clinical endpoints were acute kidney injury and infection. Renal function was analyzed as a binary variable using Acute Kidney Injury Network criteria defined as ≥50% postoperative increase from baseline creatinine to peak postoperative creatinine level in the first ten postoperative days. Infection data were derived from the prospectively collected Duke Cardiovascular Database; infectious complication definitions were as follows: positive blood culture (positive blood culture in the postoperative period), leg wound infection (incisional infection requiring dressings and intravenous antibiotics), sternal wound infection (incisional infection requiring dressings and intravenous antibiotics), mediastinitis (documented infection without return to operating room), pneumonia (radiographic infiltrate and growth of typical respiratory pathogen or heavy growth of atypical respiratory pathogen), and urinary tract infection (positive urine culture).
Statistical analysis
Continuous variables were described as means (±standard deviation) or medians (interquartile range) as appropriate; categorical variables were described as a percentage. For descriptive purposes, patients were grouped according to HbA1c status: ≤6.0% and >6.0% (the threshold limit for normal values at the study institution). For analytic purposes, HbA1c was considered as a continuous variable, using HbA1c level as a linear predictor. A logistic regression model for the outcome 30-day mortality was constructed with four predictors: HbA1c level, preoperative fasting blood glucose level, surgery date, and Parsonnet score. The Parsonnet score was included to reduce the statistical “cost” of adjusting for known mortality risk factors. The secondary outcomes of acute kidney injury and infection and their association to HbA1c were examined using multivariable linear and logistic regression, respectively. The multivariable analyses to assess the relationship of HbA1c with acute kidney injury (postoperative creatinine rise ≥ 50%)22 included known renal risk factors: age, gender, body mass index, hypertension, use of intra-aortic balloon pump, congestive heart failure, ethnicity, cardiopulmonary bypass duration, cross-clamp time, baseline blood pressure, preoperative creatinine, and fasting and peak intraoperative serum glucose.23 - 26
The multivariable analyses to assess the relationship of HbA1c with infection included mortality risk score, fasting and peak intraoperative serum glucose, body weight, and transfusion.
In order to assess for any role of undiagnosed diabetics within the sample, we performed a secondary analysis similar to the primary outcome analysis for 30-day mortality, excluding patients with fasting blood glucose levels of >126 mg·dL−1 (7.0 mmol·L−1). Due to the reduced number of deaths in this dataset compared with the primary analysis (26 vs 43, respectively), we limited the predictors in this logistic regression model to HbA1c and Parsonnet score.
Rationale for the abovementioned analysis strategy involves the view of our patient sample as a representative cohort of individuals presenting to preoperative assessment units, i.e., containing a subset of as yet undiagnosed and borderline diabetics. Many diabetics are undiagnosed or recognized but not yet receiving standard therapy.20 While pertinent to our analysis, determination of the true diabetic status is not possible on the basis of a single elevated blood glucose or HbA1c measurement. Since hyperglycemia may be stress-induced or related to pain, myocardial ischemia, or even emotional disturbance, particularly in preoperative cardiac surgery patients, the diagnosis of diabetes should not be made hastily.27 Interpretation of a single glucose test rather than two tests overestimates the prevalence of diabetes,28 and the American Diabetes Association recommends repeating abnormal fasting values.13 Current guidelines do not accept abnormally high HbA1c values as sufficient to warrant the diagnosis of diabetes.13
The data were managed and analyzed using SAS version 9.1.3 (SAS Inc, Cary, NC, USA). A P value of 0.05 was considered statistically significant.
Results
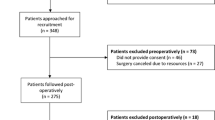
Twenty-nine percent of all non-diabetic patients undergoing cardiac surgery (1,615/5,559) at our institution from January 1, 1999 to December 31, 2005 were tested preoperatively for HbA1c levels based primarily on surgeon screening preference. Procedures included isolated aortocoronary bypass (71%), isolated valve (13%), and combined aortocoronary bypass/valve (16%) surgery. During the same period, 2,872 patients with a preoperative diagnosis of diabetes underwent surgery.
From the group of HbA1c-tested patients, a further 141 (13%) were excluded due to missing variables, primarily missing left ventricular ejection fraction data for calculation of the Parsonnet score. Mortality rates among patients with and without data to calculate a Parsonnet score were similar (P = 0.65). A further 87 (5%) patients were excluded due to incomplete 30-day follow-up data, resulting in a study sample of 1,474 patients. Survival and infection data were available for all patients. Serial serum creatinine values at baseline and daily to postoperative day ten were available for 1,230 (83%) patients.
Patient and procedural characteristics were similar to those observed in other cardiac surgery cohorts (Table 1).7 , 15 , 16 , 20 HbA1c levels exceeded 6.0% in 31% (n = 456) of the study cohort (Figure 1A). Compared with the normal group, patients with HbA1c > 6.0% were heavier (90 ± 22 vs 84 ± 21 kg; P < 0.0001), more likely to be obese (body mass index [BMI] 30.1 ± 7.7 vs 28.1 ± 6.4 kg·m−2; P < 0.0001), and more likely to be Caucasian than African American (88 vs 77%; P < 0.0001).
Distribution of preoperative hemoglobin A1c (HbA1c) levels (A), fasting blood glucose levels (B), and peak intraoperative blood glucose levels during cardiopulmonary bypass (CPB) (C) in 1,474 non-diabetic patients undergoing cardiac surgery. Conversion of HbA1c from 1993 Diabetes Control and Complications Trial (DCCT) (%) values to International Federation of Clinical Chemistry (IFCC) units can be achieved using the following equation: IFCC-HbA1c (mmol/mol) = [DCCT-HbA1c (%) − 2.15] × 10.929
Intraoperatively, mean blood glucose was 118 ± 36 mg·dL−1 (6.6 ± 2 mmol·L−1), and mean peak blood glucose was 212 ± 44 mg·dL−1 (11.8 ± 2.4 mmol·L−1) (Figure 1B, C). Blood glucose levels were significantly higher among patients with HbA1c > 6.0% compared with those with values ≤ 6.0%, including the baseline fasting (129 ± 40 mg·dL−1 [7.2 ± 2.2 mmol·L−1] vs 113 ± 34 mg·dL−1 [6.3 ± 1.9 mmol·L−1], respectively; P < 0.0001) and intraoperative peak (216 ± 57 mg·dL−1 [12.0 ± 3.2 mmol·L−1] vs 209 ± 55 mg·dL−1 [11.6 ± 3.1 mmol·dL−1], respectively; P = 0.03) blood glucose values (Figure 1B, C). Both baseline and peak blood glucose levels correlated with HbA1c (r = 0.30; P < 0.001 and r = 0.8; P = 0.004, respectively).
The overall 30-day mortality rate was 3.1% (n = 43). Higher HbA1c levels were associated with increased 30-day mortality (odds ratio 1.53 [1.24-1.91] per unit change of HbA1c; P = 0.0005) after adjusting for fasting blood glucose, surgery date, and Parsonnet score (Figure 2). The c-index for this multivariable model, including HbA1c, baseline fasting blood glucose, and Parsonnet score, was 0.76, which indicates that the model is a moderately good predictor of 30-day mortality risk. The relationship between HbA1c and 30-day mortality persisted in the secondary analysis (P = 0.05) after “borderline” diabetic patients with baseline fasting blood glucose values of > 126 mg·dL−1 (7.0 mmol·dL−1) were excluded. Aprotinin was used in 32.09% of the study patients. When aprotinin was included in the multivariable model, its use was associated with 30-day mortality (P = 0.02), but the association of HbA1c with mortality remained significant (P = 0.0002, odds ratio 1.52, 95% confidence interval [CI] 1.21-1.91).
Early postoperative mortality and preoperative hemoglobin A1c (HbA1c) levels in non-diabetic patients undergoing cardiac surgery. Conversion of HbA1c from 1993 Diabetes Control and Complications Trial (DCCT) (%) values to International Federation of Clinical Chemistry (IFCC) units can be achieved using the following equation: IFCC-HbA1c (mmol·mol−1) = [DCCT-HbA1c (%) − 2.15] × 10.929
Acute kidney injury was independently associated with increasing levels of HbA1c after adjusting for known renal risk factors, including age, gender, body weight, baseline serum creatinine, and peak intraoperative serum glucose (P = 0.04; odds ratio 1.148, 95% CI 1.003-1.313) (Figure 3). We found no association between HbA1c levels and postoperative infection (P = 0.48).
Graphic representation of the association between HbA1c and acute kidney injury (AKI), as reflected by a serum creatinine rise ≥ 50% above baseline,22 after adjustment for age, sex, preoperative creatinine, weight, and maximum glucose, with 95% confidence intervals (A). Conversion of HbA1c from 1993 Diabetes Control and Complications Trial (DCCT) (%) values to International Federation of Clinical Chemistry (IFCC) units can be achieved using the following equation: IFCC-HbA1c (mmol/mol) = [DCCT-HbA1c (%) − 2.15] × 10.929
Discussion
In our cohort of non-diabetic cardiac surgery patients, we found elevated preoperative HbA1c levels (>6%) to be common and independently associated with a 53% increase in risk of early postoperative mortality per percent increase in HbA1c. This effect persisted even when patients with evidence of “borderline” diabetes were excluded. Escalating acute kidney injury was also related to higher baseline HbA1c levels, even when HbA1c was within the normal range. Elevated HbA1c also predicted higher fasting and peak intraoperative glucose levels. However, infection risk was not associated with baseline HbA1c levels. Our findings suggest that preoperative serum HbA1c assessment may be useful as a screening tool for non-diabetic cardiac surgery patients to stratify for early postoperative mortality risk. In addition, baseline HbA1c may be informative to anticipate intraoperative elevations of blood glucose as part of glycemic control strategies and postoperative acute kidney injury.
HbA1c has gained acceptance in diabetics readily as an index of long-term blood glucose control and a predictor of complications.29 - 31 Also, two large studies in diabetics highlight the relationship of intensive therapy targeted to maintain normoglycemia with lower HbA1c levels and improved long-term outcome.29 , 31 Other long-term studies involving both diabetics and non-diabetics correlate elevated HbA1c with poorer survival both in the general population32 and specifically following cardiac surgery.15 In a cohort of over 3,000 diabetic and non-diabetic, on-pump and off-pump aortocoronary bypass surgery patients, Halkos et al. reported an association between elevated HbA1c and several short-term adverse outcomes, including postoperative myocardial infarction, deep sternal wound infection, renal failure, stroke, and short-term mortality.16 A similar but smaller study by Medhi et al. noted prolonged hospital stays after aortocoronary bypass surgery in patients with elevated preoperative HbA1c levels.33
Reports specific to HbA1c in non-diabetics are limited but include a study linking elevated HbA1c with early morbidity in vascular surgery patients.18 A second study by Anderson et al. found that non-diabetic aortocoronary bypass surgery patients with mildly elevated fasting glucose values were at increased risk for 30-day and one-year mortality.34 While these studies do not ask the same question addressed in our study, they are thematically consistent with our observation that evidence of insulin resistance is associated with worse perioperative outcomes. Although our study is restricted to non-diabetics, our findings are similar to the study of Halkos et al. that involves diabetics and non-diabetics. In addition to early mortality, both studies found an association between renal injury and elevated HbA1c levels but not between HbA1c and overall infection rates.16 In contrast to our study, the Halkos study found a relationship between deep sternal wound infection and HbA1c levels (odds ratio 1.38; P = 0.029). Notably, a strong association exists between diabetes and sternal wound infections.35 The difference between our findings and those of Halkos et al. with regard to sternal wound infection may involve the lack of diabetics in our study sample, but it may also be related to our smaller sample size and/or the low rate of sternal wound infection in our study. The rate of Hb1Ac elevation seen in our study (31%) and a cohort of non-diabetic patients undergoing percutaneous coronary interventions (30%)19 are lower than in two other studies of vascular (57%) and cardiac (58%) surgery patients.18 , 20 A possible explanation for this discrepancy may be the potential for elevated HbA1c to be associated with risk for emergent surgery, procedures which were not included in the first two studies. Our findings are consistent with a body of evidence associating elevated HbA1c and other biochemical evidence of glucose intolerance with adverse short- and long-term outcome, including the limited data involving non-diabetics. Our data also suggest that knowledge of baseline HbA1c values in non-diabetics may assist in anticipation of hyperglycemic responses to the stress of surgery and in implementation of perioperative glycemic control strategies.
Our study carries the limitations and biases inherent to any retrospective assessment of prospectively collected databases. The non-random selection of non-diabetic patients (by surgeon preference and other unknown factors) for preoperative HbA1c testing may introduce selection bias into the study. Despite controlling for multiple variables in our analyses, including the use of a validated mortality risk index, confounding factors may remain. In addition, our study is vulnerable to the accuracy of the preoperative diagnosis of diabetic status. Although the rate of “undiagnosed” diabetics may seem high, it is similar or lower than that seen in other reports and, thus, is likely an accurate representation of typical patient populations presenting for preoperative assessment prior to elective cardiac surgery.20 , 28 While the association we observed between HbA1c level and early postoperative mortality may include an effect related to a subset of undiagnosed diabetics, our findings are supported in a subgroup analysis excluding patients with baseline glucose values > 126 mg·dL−1 (7.0 mmol·dL−1).
Although not a primary focus of our study, it is tempting to speculate on the potential value of screening HbA1c to anticipate perioperative insulin dosing for glycemic control in non-diabetics. HbA1c values may not predict intraoperative hyperglycemia, particularly if dextrose-containing solutions have been used intraoperatively, but they may predict problems of intraoperative glycemic control above and beyond the hyperglycemia observed. While our study suggests that elevated baseline HbA1c predicts intraoperative hyperglycemia, it is unclear that aggressive perioperative insulin therapy in non-diabetics improves outcome. While the continuous infusion of insulin may reduce deep sternal wound infections in diabetics undergoing cardiac surgery,8 the broader role and merits of tight perioperative glucose control in all patients are still in question.10 - 12 van den Berghe et al.’s well-known intensive care study involving a large number of post-cardiac surgery patients showed a decrease in mortality from 8-4.6% with tight serum glucose targets and intensive insulin therapy.10 Unfortunately, subsequent studies have failed to confirm similar survival benefit but have served to highlight the increased risk of severe hypoglycemia in patients receiving aggressive insulin therapy.11 , 12
In summary, our findings suggest that HbA1c assessment may have value as a preoperative screening tool to identify the sizeable subset (approximately one-third) of non-diabetic cardiac surgery patients with elevated levels who are at high risk for both intraoperative hyperglycemia and adverse outcome, including a 53% greater risk of early postoperative mortality per percent increase in HbA1c and vulnerability to acute kidney injury. Further studies are required to confirm our observations and to explore and characterize the potential value of being able to anticipate perioperative hyperglycemia in non-diabetics. Predicting the glycemic response to cardiac surgery may help to guide earlier and possibly more aggressive insulin therapy, with the hope of improved safety and outcomes in non-diabetic patients with elevated HbA1c undergoing cardiac surgery.
References
Rosamond W, Flegal K, Furie K, et al. Heart disease and stroke statistics–2008 update: a report from the American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Circulation 2008; 117: e25-146.
Nashef SA, Roques F, Michel P, Gauducheau E, Lemeshow S, Salamon R. European system for cardiac operative risk evaluation (EuroSCORE). Eur J Cardiothorac Surg 1999; 16: 9-13.
Parsonnet V, Dean D, Bernstein AD. A method of uniform stratification of risk for evaluating the results of surgery in acquired adult heart disease. Circulation 1989; 79: I3-12.
Nilsson J, Algotsson L, Hoglund P, Luhrs C, Brandt J. Comparison of 19 pre-operative risk stratification models in open-heart surgery. Eur Heart J 2006; 27: 867-74.
Thourani VH, Weintraub WS, Stein B, et al. Influence of diabetes mellitus on early and late outcome after coronary artery bypass grafting. Ann Thorac Surg 1999; 67: 1045-52.
Carson JL, Scholz PM, Chen AY, Peterson ED, Gold J, Schneider SH. Diabetes mellitus increases short-term mortality and morbidity in patients undergoing coronary artery bypass graft surgery. J Am Coll Cardiol 2002; 40: 418-23.
Kubal C, Srinivasan AK, Grayson AD, Fabri BM, Chalmers JA. Effect of risk-adjusted diabetes on mortality and morbidity after coronary artery bypass surgery. Ann Thorac Surg 2005; 79: 1570-6.
Furnary AP, Zerr KJ, Grunkemeier GL, Starr A. Continuous intravenous insulin infusion reduces the incidence of deep sternal wound infection in diabetic patients after cardiac surgical procedures. Ann Thorac Surg 1999; 67: 352-60. discussion 60-2.
Lazar HL, Chipkin SR, Fitzgerald CA, Bao Y, Cabral H, Apstein CS. Tight glycemic control in diabetic coronary artery bypass graft patients improves perioperative outcomes and decreases recurrent ischemic events. Circulation 2004; 109: 1497-502.
van den Berghe G, Wouters P, Weekers F, et al. Intensive insulin therapy in the critically ill patients. N Engl J Med 2001; 345: 1359-67.
Griesdale DE, de Souza RJ, van Dam RM, et al. Intensive insulin therapy and mortality among critically ill patients: a meta-analysis including NICE-SUGAR study data. CMAJ 2009; 180: 821-7.
Reinhart K, Brunkhorst FM, Engel C, et al. Study protocol of the VISEP study. Response of the SepNet study group (German). Anaesthesist 2008; 57: 723-8.
American Diabetes Association. Standards of medical care in diabetes. Diabetes Care 2005; 28(Suppl 1): S4-36.
Saudek CD, Derr RL, Kalyani RR. Assessing glycemia in diabetes using self-monitoring blood glucose and hemoglobin A1c. JAMA 2006; 295: 1688-97.
Halkos ME, Lattouf OM, Puskas JD, et al. Elevated preoperative hemoglobin A1c level is associated with reduced long-term survival after coronary artery bypass surgery. Ann Thorac Surg 2008; 86: 1431-7.
Halkos ME, Puskas JD, Lattouf OM, et al. Elevated preoperative hemoglobin A1c level is predictive of adverse events after coronary artery bypass surgery. J Thorac Cardiovasc Surg 2008; 136: 631-40.
Khaw KT, Wareham N, Bingham S, Luben R, Welch A, Day N. Association of hemoglobin A1c with cardiovascular disease and mortality in adults: the European prospective investigation into cancer in Norfolk. Ann Intern Med 2004; 141: 413-20.
O’Sullivan CJ, Hynes N, Mahendran B, et al. Haemoglobin A1c (HbA1C) in non-diabetic and diabetic vascular patients. Is HbA1C an independent risk factor and predictor of adverse outcome? Eur J Vasc Endovasc Surg 2006; 32: 188-97.
Corpus RA, O’Neill WW, Dixon SR, Timmis GC, Devlin WH. Relation of hemoglobin A1c to rate of major adverse cardiac events in nondiabetic patients undergoing percutaneous coronary revascularization. Am J Cardiol 2003; 92: 1282-6.
Engoren M, Habib RH, Zacharias A, et al. The prevalence of elevated hemoglobin A1c in patients undergoing coronary artery bypass surgery. J Cardiothorac Surg 2008; 3: 63.
Buckberg GD, Beyersdorf F, Allen BS, Robertson JM. Integrated myocardial management: background and initial application. J Card Surg 1995; 10: 68-89.
Bellomo R, Ronco C, Kellum JA, Mehta RL, Palevsky P; Acute Dialysis Quality Initiative workgroup. Acute renal failure - definition, outcome measures, animal models, fluid therapy and information technology needs: the Second International Consensus Conference of the Acute Dialysis Quality Initiative (ADQI) Group. Crit Care 2004; 8: R204-12.
Thakar CV, Arrigain S, Worley S, Yared JP, Paganini EP. A clinical score to predict acute renal failure after cardiac surgery. J Am Soc Nephrol 2005; 16: 162-8.
Chertow GM, Lazarus JM, Christiansen CL, et al. Preoperative renal risk stratification. Circulation 1997; 95: 878-84.
Wijeysundera DN, Karkouti K, Dupuis JY, et al. Derivation and validation of a simplified predictive index for renal replacement therapy after cardiac surgery. JAMA 2007; 297: 1801-9.
Palomba H, de Castro I, Neto AL, Lage S, Yu L. Acute kidney injury prediction following elective cardiac surgery: AKICS Score. Kidney Int 2007; 72: 624-31.
Collier B, Dossett LA, May AK, Diaz JJ. Glucose control and the inflammatory response. Nutr Clin Pract 2008; 23: 3-15.
Selvin E, Zhu H, Brancati FL. Elevated A1C in adults without a history of diabetes in the U.S. Diabetes Care 2009; 32: 828-33.
Anonymous. The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. The Diabetes Control and Complications Trial Research Group. N Engl J Med 1993; 329: 977-86.
Anonymous. Effect of intensive blood-glucose control with metformin on complications in overweight patients with type 2 diabetes (UKPDS 34). UK Prospective Diabetes Study (UKPDS) Group. Lancet 1998; 352: 854-65.
Anonymous. Incidence and prevalence of ESRD United States Renal Data System. Am J Kidney Dis 1998; 32: S38-49.
Nakanishi S, Yamada M, Hattori N, Suzuki G. Relationship between HbA(1)c and mortality in a Japanese population. Diabetologia 2005; 48: 230-4.
Medhi M, Marshall MC Jr, Burke HB, et al. HbA1c predicts length of stay in patients admitted for coronary artery bypass surgery. Heart Dis 2001; 3: 77-9.
Anderson RE, Klerdal K, Ivert T, Hammar N, Barr G, Owall A. Are even impaired fasting blood glucose levels preoperatively associated with increased mortality after CABG surgery? Eur Heart J 2005; 26: 1513-8.
Paul M, Raz A, Leibovici L, Madar H, Holinger R, Rubinovitch B. Sternal wound infection after coronary artery bypass graft surgery: validation of existing risk scores. J Thorac Cardiovasc Surg 2007; 133: 397-403.
Financial support
Department of Anesthesiology, Duke University Medical Center.
Competing interests
None declared.
Author information
Authors and Affiliations
Consortia
Corresponding author
Appendix
Appendix
Members of the Cardiothoracic Anesthesiology Research Endeavors (C.A.R.E.), Department of Anesthesiology, Duke University Medical Center.
Director: Joseph P. Mathew MD
Anesthesiology: Solomon Aronson MD, Katherine P. Grichnik MD, Steven Hill MD, G. Burkhard Mackensen MD, PhD, Joseph P. Mathew MD, Mark F. Newman MD, Barbara Phillips-Bute PhD, Mihai V. Podgoreanu MD, Andrew D. Shaw MD, Mark Stafford-Smith MD, Madhav Swaminathan MD, Ian Welsby MD, William D. White MPH, Lisa Anderson, Lauren Baker BS, Jerry Dove RN, Bonita L. Funk RN, Roger L. Hall AAS, Gladwell Mbochi AAS, Tiffany Bisanar RN, Prometheus T. Solon MD, Peter Waweru.
Perfusion Services: Kevin Collins, BS, CCP, Greg Smigla, BS, CCP, Ian Shearer, BS, CCP.
Surgery: Thomas A. D’Amico MD, R. Duane Davis MD, Donald D. Glower MD, R. David Harpole MD, G. Chad Hughes MD, James Jaggers MD, Shu Lin MD, Andrew Lodge MD, James E. Lowe MD, Carmelo Milano MD, Peter K. Smith MD, Jeffrey Gaca MD, Mark Onatis MD.
Rights and permissions
About this article
Cite this article
Hudson, C.C.C., Welsby, I.J., Phillips-Bute, B. et al. Glycosylated hemoglobin levels and outcome in non-diabetic cardiac surgery patients. Can J Anesth/J Can Anesth 57, 565–572 (2010). https://doi.org/10.1007/s12630-010-9294-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12630-010-9294-4