Abstract
The exchange of cold polar waters from the north and warm North Atlantic waters from the south is limited by the Greenland-Iceland-Scotland Ridge (GSR) which then may act as a barrier for faunal exchange from the deep Artic and deep Atlantic basins. We investigated the meiofauna density and distribution patterns from different regions north and south of the GSR at water depths between 307 and 2749 m. A total of 84 multicore samples were examined collected during the IceAGE1 (Icelandic marine Animals: Genetics and Ecology) project in summer 2011. We used a gradientForest approach to assess the magnitude of compositional change and the thresholds of remarkable community changes along environmental variables. randomForest regression was applied to predict the meiofauna on a spatial contiguous scale with a set of 23 environmental variables. Meiofauna density ranged between 187 and 3185 individuals per 10 cm2 with the highest densities observed north of the GSR. Nematoda was the most abundant taxon among the meiofauna community, followed by Copepoda and Nauplii. In the Irminger Basin, Gastrotricha was the third most abundant taxon, while in all other regions, Ostracoda was the third most abundant. Food supply, water depth, bottom water oxygen, and hydrographic activity being the most important variables for community changes explain up to 86% of the variation observed in the meiofauna communities.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The sea around Iceland is a perfect natural laboratory for studying the effects of climatic changes to benthic marine communities. It is located at the transition of the Greenland, Iceland, and Norwegian Seas (GIN Seas) to the north (characterized by cold, highly oxygenated and less saline waters) and the northernmost North Atlantic waters to the south (characterized by comparatively warm, less oxygenated and more saline waters) (Hansen and Østerhus 2000). Although the GIN Seas and the northernmost North Atlantic Ocean are separated by the Greenland-Iceland-Scotland Ridge (GSR), extending from west to east, the exchange of waters is not fully blocked, but there exists an overflow water exchange (e.g., Beaird et al. 2013; Rudels et al. 2002).
Meiofauna organisms are among the most abundant group of the benthic communities (Rex and Etter 2010), even in the deep sea (Wei et al. 2010). Meiofauna organisms are influenced by various environmental variables such as food availability (Danovaro et al. 1996; Hoste et al. 2007; Pfannkuche 1985; Pfannkuche and Thiel 1987; Vanreusel et al. 1995b), sediment granulometry, temperature, and salinity (Kennedy and Jacoby 1999; Soltwedel 2000). Meiofauna densities follow bathymetrical gradients as reported for the Antarctic Ocean (Gutzmann et al. 2004; Herman and Dahms 1992; Vanhove et al. 1995), Pacific Ocean (Kitahashi et al. 2013), North Atlantic Ocean (Pfannkuche 1985; Soetaert et al. 2002; Vanaverbeke et al. 1997a), and Arctic Ocean (Pfannkuche and Thiel 1987; Schewe 2001; Schewe and Soltwedel 1999; Vanaverbeke et al. 1997b; Vanreusel et al. 2000).
Considering the Icelandic waters, however, research on meiofauna communities and their relationship to environmental variables are rather scarce. Despite some studies related to Harpacticoida Copepoda in an intertidal pool, freshwater Harpacticoida, and Icelandic Ostracoda (Apostolov 2007; Steinarsdóttir et al. 2003; Yasuhara et al. 2014), there are two main research expeditions to the Norwegian Sea (NORBI) and the Faroe Ridge (RV Anton Dohrn) (Dinet 1979; Thiel 1971, 1975), indicating an influence of hydrographic conditions concomitant with changes in food supply to the sea floor.
In the present study, we will investigate the Icelandic meiofauna community in different regions north and south of the GSR along bathymetrical gradients. Further, a gradientForest approach will be used to assess community changes along environmental variables, and finally, we will generate predictive models using randomForest regression (Breiman 2001; Liaw and Wiener 2002) to predict selected meiobenthic taxa on a contiguous geographic space at regional scale around Iceland.
The following research questions are addressed:
-
1.
Does the meiofauna community differ between the geographical regions?
-
2.
Which are the most important variables causing changes in the community structure and which taxa are most affected by these changes?
-
3.
Do the predictive models reflect observed differences in the study area?
Material and methods
Study site
The study area covers an area of about 750,000 km2, including the northernmost North Atlantic and the south-western part of the Greenland, Iceland, and Norwegian Seas (GIN Seas). The Greenland-Scotland Ridge (GSR) extends from west to east, featuring a mean depth of less than 500 m and three deep sills.
West of Iceland, cold and less saline water is transported across the Greenland-Iceland Ridge (GIR) into the Atlantic as Denmark Strait Overflow Water (DSOW), which flows along the Greenlandic coast as the Deep Water Boundary Current (DWBC). The surface East Greenland Current (EGC) transports PW across the ridge. East of Iceland cold water is transported across the Iceland-Faroe Ridge (IFR) as Iceland Scotland Overflow Water (ISOW), which flows along the Reykjanes Ridge into the Irminger Basin (Hansen and Østerhus 2000; Pickart et al. 2005; Logemann et al. 2013; Våge et al. 2011, 2013).
Due to geostrophic forces, there is a warm water inflow from the Atlantic into the Arctic Ocean. Two surface currents, the North Icelandic Irminger Current (NIIC) and the North Atlantic Current (NAC), transport Modified North Atlantic Water (MNAW) into the Arctic Ocean.
Processing of sediment samples
Sediment samples were collected with a multicore (company Oktopus GmbH, MUC, 12 cores, inner area 72.38 cm2 per core) during the IceAGE1 cruise (M85/3) from 27 August to 28 September 2011 on board the German RV Meteor at 24 stations. The study area was divided into four regions, Iceland Basin (IceBas, 7 stations), Irminger Basin (IrmBas, 4 stations), Denmark Strait (DenStr, 6 stations), and Norwegian Sea (NorwSea, 7 stations), located north and south of the GSR (Fig. 1, Table 1). Sediment samples were collected along bathymetrical gradients ranging from 307 to 2749 m. At each station, one deployment was conducted. Of each deployment, four cores were used to study density and distribution of metazoan meiofauna. The top 5 cm sediment of each core was fixed in 4% buffered formalin. Due to logistic difficulties with the deployment of the multicore in the Denmark Strait (broken cores and low number of samples), the multicore was deployed twice at each station there. From these deployments, two cores each were chosen for analysis.
Back in the laboratory, the fixed samples were washed through a 40-μm sieve and centrifuged 3 times at 4000 rpm for 6 min using a colloidal silica polymer (Levasil) as floating medium and kaolin to cover the heavier particles according to McIntyre and Warwick (1984). The supernatant, which contains organic material and organisms, was decanted with tap water after each step and stained with Rose Bengal. Meiofauna was sorted to higher taxon level and counted using a Leica MZ12.5/9 dissecting microscope.
Statistical analyses
Any procedure written in italics refers to analyses performed in R (R Core Team 2016). We tested for differences in meiofauna density between (a) northern and southern samples (of the GSR), (b) 4 regions, and (c) depths classes (upper slope [0–1000 m], lower slope [1000–2000 m], and abyssal [2000–3000 m]) using permutational multivariate analysis of variance (PERMANOVA, adonis) (Anderson 2001; Anderson and Walsh 2013; McArdle and Anderson 2001). Post hoc, pairwise comparisons were performed for water depths and regions, with p values Bonferroni corrected using the custom function pairwise.adonis (https://github.com/pmartinezarbizu/pairwiseAdonis). The analyses were performed on fourth root transformed data using a Bray-Curtis dissimilarity matrix (999 permutations).
Multivariate statistics were performed using the package vegan (Oksanen et al. 2016). We used non-metric Multidimensional Scaling (nMDS, function metaMDS) as an unconstrained ordination method (Minchin 1987). The nMDS was based on Bray-Curtis dissimilarity and fourth root transformed data. The function envfit was used to fit environmental variables as vectors onto the ordination of the nMDS (999 permutations). This method shows which environmental variable correlates most with the axis of the ordination. The relative length of the vector in comparison to the vector length of other variables is the strength of the variable. Therefore, the direction and length of the respective variable can be related to relative importance for structuring the communities. For image clarity, we display only the 11 most important variables, out of 23.
To explore the nonlinear multispecies response of meiofauna to environmental gradients, we applied the gradientForest method (Ellis et al. 2012) as a coherent extension to our randomForest regression approach, favoring this method over most popular linear alternatives like CANOCO (ter Braak 1986). gradientForest cannot only be used to explore the magnitude of community change along environmental gradients but also to discover threshold values, at which community (or single taxon) changes are most relevant (Pitcher et al. 2012). The R2 value provides the variance explained weighted over all species. gradientForest uses the conditional rather than the marginal importance (by performing conditional permutation of predictors over non-correlated groups of variables) to account for correlated predictors (Strobl et al. 2008; Ellis et al. 2012).
For each predictor, the split values of the regression trees are associated with the model accuracy improvement to produce response functions to community (and taxa) change along predictors. These curves are standardized and normalized to become comparable between species and predictors. For implementing gradientForest with our Icelandic dataset, we followed the vignette of the R package gradientForest (Pitcher et al. 2011) but growing 1000 rather than 500 trees.
Regression models
The spatial distribution of meiofauna (Nematoda, Copepoda, Nauplii, Ostracoda, Kinorhyncha, Tardigrada, Gastrotricha, Tantulocarida) was predicted using a set of 23 environmental predictor variables. Spatial layers of sediment grain sizes (sand, silt clay) and organic matter (total organic carbon [TOC], total nitrogen [N], C/N ratio, chlorophyll a, and pigment derivates) were taken from Ostmann et al. (2014), who worked on sediment samples from the exact locations and deployments. Hydrographic layers (bottom salinity and temperature including temperature maximum, temperature minimum, and inter-annual temperature range variation) were taken from NISE data (Norwegian Iceland Seas Experiment, 1998–2008, Nilse et al. 2008). Bottom oxygen was based on the dataset provided by Seiter et al. (2005). Further, particulate organic carbon (POC) flux to the bottom and the seasonal variation index (SVI) of POC flux (Lutz et al. 2007), depth (GEBCO_08 layer, 30 arc seconds’ resolution from www.gebco.net), sediment thickness (NOAA; Divins 2003), and bottom roughness (NOAA) were used as environmental layers.
The point sampling tool of QGIS (QGIS Development Team 2016, version 2.4.0, Chugiak) was used to extract the values of the 23 prediction layers at the exact position of IceAGE1 stations and for producing the predicting dataset.
randomForest regression is a nonparametric and nonlinear method to predict a quantitative variable using an ensemble of regression trees (Breiman 2001). At each split of the randomForest tree, the model is using a random subset of the predictors for the best split. The decision tree is built from 2/3 of the data (in-bag) and the other 1/3 of the dataset is used for prediction error and importance of predictors. The randomForest models were generated with the R package randomForest (Liaw and Wiener 2002) using 2000 random trees (ntree = 2000) and 8 random variables (mtry = 8). The performance of the model is given as the % of explained variance of the model (R2). The resulting models were used to predict meiofauna density on a grid of 22,139 points distributed around Iceland.
Maps were generated on a regional scale interpolating the values of this point-grid using the Generic Mapping Tools (Wessel et al. 2013, GMT 5.1.0, SOEST, Hawaii). Interpolation was done with the surface function (continuous curvature surface gridding algorithm) using a gridding space of 0.005 geographic degrees (WGS84) and a tension factor of 0.5.
Results
Meiofauna density (per 10 cm2)
In 84 samples, 680,701 meiofauna individuals out of 25 taxa were counted (total area of all corers covering 0.608 m2). The highest densities (per corer) were observed at stations in the Iceland Basin and Norwegian Sea (2554–3185 ind/10 cm2). Lowest densities (187–486 ind/10 cm2) were found in the Irminger Basin as well as the Iceland Basin (Table 2). In the other samples, density was intermediate varying between 500 and 2000 ind/10 cm2. Most taxa, such as Kinorhyncha (max. 19 ind/10 cm2), Ostracoda (max. 28 ind/10 cm2), and Copepoda (max. 109 ind/10 cm2) reached their maximum density in the Norwegian Sea. Tardigrada reached a maximum of 25 ind/10 cm2 in the Denmark Strait. The density of Gastrotricha was highest in the Irminger Basin. Significant differences were found between (a) northern and southern sites, (b) between the basins, and (c) different water depths (PERMANOVA, Table 3, Fig. 2). Sites located north of the GSR exhibit a higher meiofauna density compared to the southern sites (Fig. 2a). Meiofauna density was significantly higher in the Norwegian Sea compared to all other regions, while the lowest densities were observed in the Irminger Basin (Fig. 2b). The overall meiofauna density was significantly higher at the upper slope compared to the abyssal and lower slope (Fig. 2c). The highest taxa richness (18 taxa) was observed at stations in the Iceland Basin, Denmark Strait, and Norwegian Sea, while other stations in the Iceland Basin, Irminger Basin, and Norwegian Sea showed the lowest richness (9 taxa).
Non-metrical multidimensional scaling (nMDS, abundance data fourth root transformed) displaying the differences observed between factors (Bray-Curtis dissimilarity, fourth root transformed data. Stress: 0.142), a north-south direction; b regions; c depth classes (upper slope [0–1000 m], lower slope [1000–2000 m], abyssal [2000–3000 m]); d environmental variables plotted as vectors into the ordination. For significance levels, seeTable 4
Meiofauna community structure
Nematoda were most abundant among the meiofauna community. They dominated with values from 78.38% (Irminger Basin) to 95.71% in the Norwegian Sea with an overall median at 88.13%. Copepoda and their Nauplii followed in dominance (~ 1.42–5.96 and 1.6–8.82%, respectively), with an overall median of 3.58 and 5.11%, respectively.
Together with Annelida (excluded from further analyses, because fragments were count instead of specimens) and Ostracoda, the mentioned groups were observed in all samples. Kinorhyncha, Tardigrada, Loricifera and Bivalvia (both latter excluded from further analyses), and Gastrotricha were recorded in more than 60 out of 84 samples. Other taxa were found only sporadically (Table 2). In the Irminger Basin, Gastrotricha were the third most abundant taxa, while in the other regions, Ostracoda dominated following Nematoda and Copepoda.
Almost every environmental variable correlated with the ordination of the nMDS (envfit, Fig. 2d, Table 4). The vectors of oxygen, chlorophyll a, nitrogen, clay, and silt direct to sites in the Norwegian Sea, while POC, bottom roughness, temperature, and sand mostly reflect sites located in the Iceland Basin, Denmark Strait, and Irminger Basin. The C/N value (not shown in the graph) directed to the Irminger Basin, reflecting the second axis of the nMDS. Results indicate an influence of environmental variables on the meiofauna community in the Icelandic waters.
Meiofauna along environmental gradients
Meiofauna density follows bathymetrical gradients in the Norwegian Sea and Iceland Basin, mainly represented by a decline in densities of Nematoda, Copepoda, and their Nauplii. In the Irminger Basin and the Denmark Strait, overall meiofauna density did not decrease with depth (Table 2).
The eight most important variables shaping the overall meiofauna community are in decreasing order POC (particulate organic carbon flux), water depth, TOC (total organic carbon), bottom roughness, oxygen, nitrogen, sediment chlorophyll a content, and SVI (seasonal variation index of the POC flux) (Fig. 3). The shapes of the cumulative importance curves of the meiofauna to environmental change are nonlinear and some of them show thresholds that produce step changes in community composition (Figs. 4 and 5). The upper panels show the effect on the five most responsive taxa. The cumulative effect on all taxa in displayed in the lower panels (community level response). Meiofauna community changes occurred at around 17 and 27 mg C/m2 day (POC flux), with the greatest influence on Kinorhyncha and Copepoda with its Nauplii. Water depth, as second most important factor, influences mostly Copepoda, Kinorhyncha, and Ostracoda with thresholds around 1050 and 650 m which shall correspond with predicted abundance maxima for these taxa at the northern and southern slopes respectively (Fig. 5c, e, f). The largest changes in the meiofauna community occur at 1.7 and 2.1% TOC in the sediments, mainly influencing Nauplii, Copepoda, and Ostracoda. The most abundant taxon, the Nematoda is influenced mostly by bottom water oxygen with a big step at 289 μmol/kg, chlorophyll a in the sediment with a step at 0.08 μg/g, and seasonal variation of POC flux (SVI) with two steps around 1.1 and 1.42 g/m2. The amount of nitrogen influences the meiofauna at a threshold of 0.088%, which has effects on Ostracoda.
Cumulative plots. a–d Taxa cumulative plots showing the value of the environmental variable at which a split change of respective taxa occurs (in respective color). e–h Predictor cumulative plots (particulate organic carbon [POC], water depth, total organic carbon ([TOC], sediment), bottom roughness) showing the value of the environmental variable which is responsible for the split changes in the overall meiofauna abundance
Cumulative plots. a–d Taxa cumulative plots showing the value of the environmental variable at which a split change of respective taxa occurs (in respective color). (e–h) Predictor cumulative plots (oxygen, nitrogen, chlorophyll α, seasonal variation index of the POC flux) showing the value of the environmental variable which is responsible for the split changes in the overall meiofauna abundance
Regression models
The prediction models were generated with a set of 23 predictors and 84 samples. For each model, a random set of predictors was used. A more intuitive approach to assess model performance was to calculate the Pearson product moment correlation between observed values used for training and predicted values at these sites. The variation of the abundance of the meiofauna taxa could be explained with up to 86% (Table 5). Maps were generated on a regional spatial scale for the overall meiofauna density per 10 cm2, the taxa richness, and density of six selected meiofauna taxa (Figs. 6 and 7).
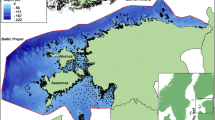
For the six taxa, the explained variation was higher than 65%. Gastrotricha were predicted with a variation of 35.11% and for the Tardigrada, predictor variables could explain 43.62% of the variation. A poor model was performed on Tantulocarida (− 19.24% of the variance). High densities of total meiofauna were predicted along the Greenland-Iceland-Scotland Ridge on the upper slope (1500–2900 ind/10 cm2) and north of the GSR in the Norwegian Sea (Fig. 6a). North-east of Iceland (6–12° W, 68–70° N), the model predicted one spot of high meiofauna densities. Generally, meiofauna density decreased from the upper slope to the abyssal. Nematoda, Copepoda and their nauplii, Ostracoda, and Kinorhyncha densities (Fig. 6b–f) were predicted to be highest at the Icelandic upper slope and along the Greenland-Iceland-Scotland Ridge.
Furthermore, the densities of these groups were predicted slightly higher north than south of the GSR. Tardigrada (Fig. 7a) were predicted to be highest in the Irminger Basin, southwest of Iceland, and south of the Faroe Islands (5–10 ind/10 cm2). High Gastrotricha density (Fig. 7b) was predicted north of the GSR (~ 2 ind/10 cm2) on the upper Icelandic slope, along the Iceland-Faroe Ridge (2 ind/10 cm2) and at the Reykjanes Ridge south of Iceland (2.4 ind/10 cm2). Lower densities were found in the deep sea north and south of the GSR (~ 0.3 ind/10 cm2). Tantulocarida showed high densities in the deep sea (Fig. 7c), while density was less along the GSR and on the upper slope (0.15 ind/10 cm2). Meiofaunal taxa richness (Fig. 7d) was highest west of Iceland at the Greenland-Iceland Ridge, on the upper slope north of Iceland, and north of the Iceland-Faroe Ridge (15.6–18 taxa).
Discussion
Meiofauna density and community structure
The present study aimed at describing the meiofauna abundance and distribution patterns in Icelandic waters. We identified several environmental variables explaining the regional geographic differences in meiofauna. Based on the knowledge on the complex hydrographic and topographic conditions, collinearity of these environmental variables has to be taken into account. Meiofauna densities in Icelandic waters varied between 187 and 3185 individuals per 10 cm2 and fall within the range of meiofauna densities reported from other Arctic and North Atlantic regions. The meiofauna community was dominated by Nematoda, followed by Copepoda, which is similar to other meiofauna studies from the Atlantic and Arctic Ocean (see Rex and Etter 2010; Soltwedel 2000; and Vincx et al. 1994 for reviews). Meiofauna density and community structure in Icelandic waters differed regionally. The highest densities were present north of the GSR. Thus, meiofauna density shows a latitudinal limit in the vicinity of the GSR. Studies on meiofauna abundance (Thiel 1971; Soltwedel 2000 for a summary) and Amphipoda abundance (Weisshappel and Svavarsson 1998) show similar results.
Meiofauna abundances, and furthermore abundances of Amphipoda, are described to differ north and south of the GSR (Thiel 1971; Weisshappel and Svavarsson 1998). Regional differences have been explained by the separation of the northern and southern deep-sea basins (topographic barrier), temperature, salinity, hydrographic activity, water depth, as well as food availability (Weisshappel and Svavarsson 1998; Thiel 1971).
Deep-sea ridges and their effect on the distribution and abundance of faunal organisms are to some extend contradictory. In the Atlantic Ocean, studies showed that deep-sea ridges or wide deep-sea areas do not prevent an exchange of meiofauna (Menzel and George 2012; Pointner et al. 2013), while other studies (here on macrofauna diversity) indicated that distribution patterns of macro- and meiofauna are to some extent limited at ridges (e.g., Brix and Svavarsson 2010; Svavarsson et al. 1993; Thiel 1971). Hence, depending on the ridge structure and the studied spatial scale, the answer whether or not ridges limit distributions patterns is varying. Even the faunal groups studied (taxa, genus, species) show different patterns (Brix and Svavarsson 2010; Menzel et al. 2011; Schnurr et al. 2014).
Northern and southern waters differ in temperature, salinity, and oxygen content (Ostmann et al. 2014). There is evidence, that temperature limits the faunal distribution to either warm or cold waters (Brix and Svavarsson 2010) and that the rapid changes in temperatures along the GSR (outflow of Arctic waters into the North Atlantic and inflow of Atlantic waters into the Arctic Ocean) affect the distribution capacities of Icelandic fauna (Svavarsson 1997). However, in the present study, neither temperature nor salinity could be identified as major factors in structuring meiofauna. Near bottom oxygen content, however, was observed to be correlated with the nMDS axis, particularly pointing towards the Norwegian Sea and furthermore, the gradientForest analysis identified two thresholds, at which the meiofauna community changes most. In both regions, Nematoda represent the main part of the meiofauna community. The observed changes and differences north and south are most probably related to changes within this group, although studies showed that Nematoda are less affected by varying oxygen content (e.g., Neira et al. 2001).
The results observed in the Norwegian Sea and the Iceland Basin are congruent with the general trend of decreasing faunal abundance with increasing depth (e.g., Rex et al. 2005, 2006; Smith et al. 2008). Meiofauna density follows similar patterns in different regions, including the Arctic Ocean (Netto et al. 2005), North Atlantic Ocean (e.g.; Pfannkuche 1985; Vanreusel et al. 2000), Gulf of Mexico (Baguley et al. 2006), and the Antarctic waters (Herman and Dahms 1992; Vanhove et al. 1995; Rose et al. 2015). Although faunal abundances decrease with increasing water depth, depth does not play the dominant role in this pattern (Rex et al. 2005, 2006; Smith et al. 2008). Various environmental variables change with increasing water depth, such as food supply. This was already observed by Thiel (1971) and Dinet (1979) along the Faroe Ridge and the Norwegian Sea, and the present study supports their results.
The proxies for food supply in the present study are POC, TOC, nitrogen, chlorophyll a, and SVI. The meiofauna abundance was positively correlated with increasing food supply (Figs. 2, 3, 4, and 5). The ordination of the nMDS was correlated with variables such as POC, TOC, nitrogen, and chlorophyll a. The seasonal variation index (SVI) showed a minor correlation to the ordination. Chlorophyll a and nitrogen were correlated to the nMDS, pointing to sites located in the Norwegian Sea, while POC was pointing to stations of the Iceland Basin, being indicative of a relationship between the meiofauna and POC in that region. Sites in the Irminger Basin showed an influence of TOC (Fig. 2d).
The nonlinear gradientForest analysis supported the observed nMDS results. Food supply was most presumably responsible for the observed meiofauna patterns in Icelandic waters. Greatest changes in the overall community occurred at values of 17 and 27 mg C/m2 day (POC), 0.08 μg/g (chlorophyll a), 2.1% (TOC), 0.088% (N), and 1.1 and 1.42 g/m2 (SVI). Meiofauna taxa were influenced differently by food supply. Chlorophyll a and SVI were highly responsible for changes in Nematoda, while Kinorhyncha, Copepoda, and their Nauplii as well as Ostracoda changed with other proxies for food supply (Figs. 4 and 5).
Food-driven changes in meiofauna community structure, standing stocks, and abundance are reported in several studies from Atlantic and Arctic regions (e.g., Pfannkuche 1985; Pfannkuche and Thiel 1987; Soltwedel 2000, Thiel 1983; Vanreusel et al. 1995a). The decrease in abundances and biomasses along depth gradients are often associated with changes in food availability. The deep-sea benthos depends on sea surface production, where no chemoautotrophic systems are present (Rex et al. 2005, 2006; Smith et al. 2008). With increasing depth and distance to the coast, food availability decreases. The uptake of particles in the water column, degradation, and current activity influence the amount of organic material reaching the sea floor. Hence, approximately 1% of the surface production serves benthic communities as food (Rex and Etter 2010 and references therein).
Along the GSR cold, nutrient-rich deep-sea water and warm nutrient-poor surface water mix and provide a perfect habitat for plankton. Regions with high planktonic activity show high detritus production. Thus, the higher production results in higher faunal densities along the GSR. On the upper slope, the organic material is still of more quality and quantity compared to the lower slope and abyssal. Therefore, food supply explains the densities observed along depth gradients in the Iceland Basin and the Norwegian Sea.
To explain the distribution pattern and abundance of meiofauna in the Irminger Basin and the Denmark Strait, another variable has to be taken into account. When Arctic water crosses the GSR into the Atlantic Ocean, the outflow current can reach velocities of about 1 m/s. We therefore assume a complex system of water depth, food supply, and hydrographic conditions to be the limiting factor for meiofauna densities. The correlation between hydrographic activity and food supply was already described at the Iceland-Faroe Ridge and the Norwegian Sea (Dinet 1979; Thiel 1971). The strong overflow currents west and east of Iceland erode and resuspend small grains and organic material.
West of Iceland, in the Denmark Strait, meiofauna abundance was highly variable, with the highest densities at the deepest station. In this region, we observe an outflow (East Greenland Current) of cold waters across the Greenland-Iceland Ridge (Ostmann et al. 2014), which erodes fine sediments and organic material at the shallower stations. As the deepest station is less affected by the current, food availability is higher, sediments are finer. Consequently, meiofauna abundances are higher.
In the Irminger Basin, meiofauna density was high at the shallow station and at the deepest station. In the Irminger Basin, we observe the outflow of the EGC, represented further as Denmark Strait Overflow Water (DSOW), which sinks down with high velocities into the Irminger Basin (Ostmann et al. 2014). Similar to the study by Thiel (1971), the strong hydrographic activity causes a lower organic matter supply to the Irminger Basin. As a result, meiofauna densities are lower.
Nevertheless, for some taxa the coarse, nutrient-poor sediments show advantages.
Gastrotricha, the third most abundant taxon in that region, was highly diverse in the Irminger Basin and comprises various genera (pers. observation, unpublished). This meiofauna taxon is known to be interstitial (Higgins and Thiel 1988; Leasi and Todaro 2010) and their abundance is lower in muddy sediments (Coull 1988). Gastrotricha seem to be adapted morphologically, through the presence of lateral and terminal adhesive tubes. Besides Gastrotricha, Tardigrada were present in high numbers in one of the most disturbed and sandy areas observed during this study, indicating that both depend on coarse sediments rather that water depth. Hansen et al. (2001) already hypothesized this for Tardigrada from the Faroe Bank.
Regression models
To predict selected meiobenthic taxa on a contiguous geographic space at regional scale around Iceland, we generated predictive models using randomForest regression (Breiman 2001; Liaw and Wiener 2002). The randomForest approach is widely used for classification and regression modeling (Breiman 2001; Cutler et al. 2007; Grömping 2009; Evans and Cushman 2009). In marine ecology, these methods begin to establish (Ostmann et al. 2014; Prasad et al. 2006; Reiss et al. 2011; Wei et al. 2010). Compared to other methods, random forests improve the model performance by using many trees and selection of a random number of predictors at each node (Elith et al. 2008).
Our model explained 65–86% of the variance observed in meiofauna densities. This performance is similar the study performed by Wei et al. (2010). Sampling density plays an important role for the prediction performance (Li and Heap 2008). This is congruent with our best performances for taxa observed in more than 75 samples.
Less predictive performance was found for Tardigrada (43%) and Gastrotricha (35%). Tantulocarida were predicted with a negative performance (− 19%). This happens when the mean error is larger than the explained variance of the predicted value (see Liaw and Wiener 2002 for the equation of %variance explained). The lack of samples and a patchy distribution in the study area may explain this result (Bučas et al. 2013 and references therein). However, assessing model performance by looking only at randomForest own index for explained variance may be misleading. A more intuitive approach to assess model performance was to calculate the Pearson product moment correlation between observed values used for training and predicted values at these sites (Table 5). The correlation values are higher than 0.8, indicating good prediction accuracy, with exception of Tantulocarida (0.6) and Gastrotricha (0.64) displaying moderate accuracy.
Although, the prediction performances were moderate for some taxa, the model could support the results observed in the study area. Moreover, when comparing total meiofauna abundances predicted in our model and with abundance data of Thiel (1971) for the Faroe Ridge or data from Dinet (1979) in the Norwegian Sea, the results are quite comparable. Tantulocarida (having the lowest prediction accuracy) is the only taxon for which the model predicts an increase of abundance with depth. This however corresponds to a natural pattern, as already pointed out by Gutzmann et al. (2004).
Conclusion
-
1.
Does the meiofauna community differ between the geographical regions?
Meiofauna communities differ in abundance between the geographical regions. Meiofauna abundances north of the Greenland-Iceland-Scotland Ridge were higher compared to the south.
-
2.
Which are the most important variables causing changes in the community structure and which taxa are most affected by these changes?
There are several environmental variables causing differences in the meiofauna community. We found that food supply, hydrographic conditions as well as the hydrographic activity influence the meiofauna community, and we assume that all these variables show a collinearity to some extent that, e.g., food availability is regulated by hydrographic activity and sediment grain sizes and that the GSR limits the exchange of northern and southern waters which has an effect on the different temperatures and salinities observed. As a result, primary production at the surface differs north and south. We further observed that some taxa adapted to harsh environmental conditions, e.g., in the Irminger Basin.
-
3.
Do the predictive models reflect observed differences in the study area?
The models used to predict different meiofauna taxa on a contiguous spatial scale are quite comparable to the observed data. The differences in abundance and also the number of taxa were quite well modeled with the randomForest approach.
References
Anderson MJ (2001) A new method for non-parametric multivariate analysis of variance. Austral Ecol 26:32–46
Anderson MJ, Walsh DCI (2013) PERMANOVA, ANOSIM, and the Mantel test in the face of heterogeneous dispersions: what null hypothesis are you testing? Ecol Monogr 83:557–574
Apostolov A (2007) Harpacticoïdes Marins (Copepoda, Harpacticoida) D'Islande, 1 Le Genre Halectinosoma Lang, 1944 Et Le Genre Leptocaris T Scott, 1899. Crustaceana 80(10):1153–1169
Baguley JG, Montagna PA, Lee W, Hyde LJ, Rowe GT (2006) Spatial and bathymetric trends in Harpacticoida (Copepoda) community structure in the Northern Gulf of Mexico deep-sea. J Exp Mar Biol Ecol 330(1):327–341
Beaird NL, Rhines PB, Eriksen CC (2013) Overflow waters at the Iceland–Faroe Ridge observed in multiyear seaglider surveys. J Phys Oceanogr 43:2334–2351
Breiman L (2001) Random Forests. Mach Learn 45:5–32
Brix S, Svavarsson J (2010) Distribution and diversity of desmosomatid and nannoniscid isopods (Crustacea) on the Greenland–Iceland–Faeroe Ridge. Pol Biol 33:515–530
Bučas M, Bergström U, Downie A-L, Sundblad G, Gullström M, von Numers M, Šiaulys A, Lindegarth M (2013) Empirical modelling of benthic species distribution, abundance, and diversity in the Baltic Sea: evaluating the scope for predictive mapping using different modelling approaches. ICES J Mar Sci. https://doi.org/10.1093/icesjms/fst036
Coull BC (1988) Ecology of the marine meiofauna. In: Higgins RP, Thiel H (eds) Introduction to the study of meiofauna. Smithsonian Institution Press, Washington DC, pp 18–38
Cutler DR, Edwards TC, Beard KH, Cutler A, Hess KT, Gibson JC, Lawler JJ (2007) Random forests for classification in ecology. Ecol 88(11):2783–2792
Danovaro R, Della Croce N, Fabiano M (1996) Microbial response to oil disturbance in the coastal sediments of the Ligurian Sea (NW Mediterranean). Chem Ecol 12(3):187–198
Dinet A (1979) A quantitative survey of meiobenthos in the deep norwegian sea, Ambio Special Report, No 6, The Deep Sea: Ecology and Exploitation, pp 75–77
Divins DL (2003) NGDC total sediment thickness of the world’s oceans & marginal seas. Retrieved https://www.ngdcnoaagov/mgg/sedthick/sedthickhtml
Elith J, Leathwick JR, Hastie T (2008) A working guide to boosted regression trees. J Anim Ecol 77:802–813
Ellis N, Smith SJ, Pitcher CR (2012) Gradient forests: calculating the importance gradients on physical predictors. Ecology 93:156–168
Evans JS, Cushman SA (2009) Gradient modeling of conifer species using random forests. Landsc Ecol 24:673–683
Grömping U (2009) Variable importance assessment in regression: linear regression versus random forest. Am Stat 63(4):308–319
Gutzmann E, Martínez Arbizu P, Rose A, Veit-Köhler G (2004) Meiofauna communities along an abyssal depth gradient in the Drake Passage. Deep-Sea Res 51:1617–1628
Hansen B, Østerhus S (2000) North Atlantic-Nordic Seas exchanges. Prog Ocean 45:109–208
Hansen JG, Jorgensen A, Kristensen RM (2001) Preliminary Studies of the Tardigrade Fauna of the Faroe Bank. Zool Anz 240:385–393
Herman RL, Dahms HU (1992) Meiofauna communities along a depth transect off Halley Bay (Weddell Sea-Antarctica). Pol Biol 12:313–320
Higgins RP, Thiel H (1988) Introduction to the study of meiofauna. Smithsonian Institution Press, Washington DC 488pp
Hoste E, Vanhove S, Schewe I, Soltwedel T, Vanreusel A (2007) Spatial and temporal variations in deep-sea meiofauna assemblages in the marginal ice zone of the Arctic Ocean. Deep-Sea Res I 54:109–129
Kennedy AD, Jacoby CA (1999) Biological indicators of marine environmental health: meiofauna—a neglected benthic component? Environ Monit Assess 54:47–68
Kitahashi T, Kawamura K, Kojima S, Shimanaga M (2013) Assemblages gradually change from bathyal to hadal depth: a case study on harpacticoid copepods around the Kuril Trench (north-west Pacific Ocean). Deep-Sea Res I 74:39–47
Leasi F, Todaro MA (2010) The gastrotrich community of a north Adriatic Sea site, with a redescription of Musellifer profundus (Chaetonotida: Muselliferidae). J Mar Biol Assoc UK 90(4):645–653
Li J, Heap AD (2008) A review of spatial interpolation methods for environmental statistics. Geosci Austr 23:137
Liaw A, Wiener M (2002) Classification and regression by randomForest. R News 2(3):18–22
Logemann K, Ólafsson J, Snorrason Á, Valdimarsson H, Marteinsdóttir G (2013) The circulation of Icelandic waters—a modelling study. Ocean Sci 9:931–955
Lutz MJ, Caldeira K, Dunbar RB, Behrenfeld MJ (2007) Seasonal rhythms of net primary production and particulate organic carbon flux to depth describe the efficiency of biological pump in the global ocean. J Geophys Res 112:1–26
McArdle BH, Anderson MJ (2001) Fitting multivariate models to community data: a comment on distance-based redundancy analysis. Ecol 82:290–297
McIntyre AD, Warwick RM (1984) Meiofauna techniques. In: Holme NA, McIntyre AD (eds) Methods for the study of marine benthos. Blackwell Scientific Publishers, Oxford, pp 217–244
Menzel L, George KH (2012) Copepodid and adult Argestidae Por, 1986 (Copepoda: Harpacticoida) in the southeastern Atlantic deep sea: diversity and community structure at the species level. Mar Biol 159:1223–1238
Menzel L, George KH, Martinez Arbizu P (2011) Submarine ridges do not prevent large-scale dispersal of abyssal fauna: a case study of Mesocletodes (Crustacea, Copepoda, Harpacticoida). Deep-Sea Res II 58:839–864
Minchin PR (1987) An evaluation of relative robustness of techniques for ecological ordinations. Vegetatio 69:89–107
Neira C, Sellanes J, Levin LA (2001) Meiofaunal distributions on the Peru margin: relationship to oxygen and organic matter availability. Deep-Sea Res I 48:2453–2472
Netto SA, Gallucci F, Fonseca GFC (2005) Meiofauna communities of continental slope and deep-sea sites off SE Brazil. Deep-Sea Res I 52:845–859
Nilse J, Hátún H, Mork K, Valdimarsson H (2008) The NISE Dataset Technical Report 08-07. Tórshavn, Faroe Islands, pp 1–17
Oksanen J, Blanchet FG, Kindt R, Legendre P, Minchin PR, O’Hara RB, Simpson GL, Solymos P, Stevens MHH, Wagner H (2016) vegan: Community Ecology Package R package version 23-3 https://cranr-project.org/package=vegan
Ostmann A, Schnurr S, Martínez Arbizu P (2014) The marine environment around Iceland: hydrography, sediments and first predictive models of Icelandic deep-sea sediment characteristics. Pol Polar Res 35(2):151–176
Pfannkuche O (1985) The deep-sea meiofauna of the Porcupine Seabight and abyssal plain (NE Atlantic): population structure, distribution and standing stocks. Oceanol Act 8:343–353
Pfannkuche O, Thiel H (1987) Meiobenthic stocks and benthic activity on the NE-Svalbard Shelf and in the Nansen Basin. Polar Biol 7:253–266
Pickart RS, Torres DJ, Fratantoni PS (2005) The East Greenland Spill Jet. J Phys Ocean 35:1037–1053
Pitcher CR, Ellis N, Smith SJ (2011) Example analysis of biodiversity survey data with R package gradientForest. https://gradientforestr-forger-project.org/biodiversity-surveypdf
Pitcher CR, Lawton P, Ellis N, Smith SJ, Incze LS, Wei C-L, Greenlaw ME, Wolff NH, Sameoto JA, Snelgrove PVR (2012) Exploring the role of environmental variables in shaping patterns of seabed biodiversity composition in regional-scale ecosystems. J Appl Ecol 49:670–679
Pointner K, Kihara TC, Glatzel T, Veit-Köhler G (2013) Two new closely related deep-sea species of Paramesochridae (Copepoda, Harpacticoida) with extremely differing geographical range sizes. Mar Biodivers 43:293–319
Prasad AM, Iverson LR, Liaw A (2006) Newer classification and regression tree techniques: bagging and random forests for ecological prediction. Ecosystems 9:181–199
QGIS Development Team (2016) QGIS Geographic Information System Open Source Geospatial Foundation Project https://qgisosgeo.org
R Core Team (2016) R: a language and environment for statistical computing R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Reiss H, Cunze S, König K, Neumann H, Kröncke I (2011) Species distribution modelling of marine benthos: a North Sea case study. Mar Ecol Prog Ser 442:71–86
Rex MA, Etter RJ (2010) Deep-sea biodiversity pattern and scale. Harvard University Press, Cambridge 354 pp
Rex MA, McClain CR, Johnson NA, Etter RJ, Allen JA, Bouchet P, Warén A (2005) A source-sink hypothesis for abyssal biodiversity. Am Nat 165:163–178
Rex MA, Etter RJ, Morris JS, Crouse J, McClain CR, Johnson NA, Stuart CT, Deming JW, Thies R, Avery R (2006) Global bathymetric patterns of standing stock and body size in the deep-sea benthos. Mar Ecol Prog Ser 317:1–8
Rose A, Ingels J, Raes M, Vanreusel A, Martínez Arbizu P (2015) Long-term iceshelf-covered meiobenthic communities of the Antarctic continental shelf resemble those of the deep sea. Mar Biodivers 45(4):743–762
Rudels B, Fahrbach E, Meincke J, Budeus G, Eriksson P (2002) The East Greenland Current and its contribution to the Denmark Strait overflow. ICES J Mar Sci 59:1133–1154
Schewe I (2001) Small-sized organisms of the Alpha Ridge, Central Arctic Ocean. Int Rev Hydrobiol 86(3):317–335
Schewe I, Soltwedel T (1999) Deep-sea meiobenthos of the central Arctic Ocean: distribution patterns and size-structure under extreme oligotrophic conditions. Vie et Milieu 49(2):79–92
Schnurr S, Brand A, Brix S, Fiorentino D, Malyutina M, Svavarsson J (2014) Composition and distribution of selected munnopsid genera (Crustacea, Isopoda, Asellota) in Icelandic waters. Deep-Sea Res I 84:142–155
Seiter K, Hensen C, Zabel M (2005) Benthic carbon mineralization on a global scale. Global Biogeochem Cycles 19:1–26
Smith CR, De Leo FC, Bernardino AF, Sweetman AK, Martínez Arbizu P (2008) Abyssal food limitation, ecosystem structure and climate change. Trends Ecol Evol 23:518–528
Soetaert K, Muthumbi A, Heip C (2002) Size and shape of ocean margin nematodes: morphological diversity and depth-related patterns. Mar Ecol Prog Ser 242:179–193
Soltwedel T (2000) Metazoan meiobenthos along continental margins: a review. Prog Oceanogr 46(1):59–84
Steinarsdóttir MB, Ingólfsson A, Ólafsson E (2003) Seasonality of harpacticoids (Crustacea, Copepoda) in a tidal pool in subarctic south-western Iceland. Hydrobiologia 503:211–221
Strobl C, Boulesteix AL, Kneib T, Augustin T, Zeileis A (2008) Conditional variable importance for random forests. BMC Bioinf 9(1):307
Svavarsson J (1997) Diversity of isopods (Crustacea): new data from the Arctic and Atlantic Oceans. Biodivers Conserv 6:1571–1579
Svavarsson J, Strömberg JO, Brattegard T (1993) The deep-sea asellote (Isopoda, Crustacea) fauna of the Northern Seas: species composition, distributional patterns and origin. J Biogeogr 20:537–555
ter Braak CJF (1986) Canonical correspondence analysis: a new eigenvector method for multivariate direct gradient analysis. Ecol 67:1167–1179
Thiel H (1971) Häufigkeit und Verteilung der Meiofauna im Bereich des Island-Färöer-Rückens. Ber D Wiss Komm Meeresforsch 22:99–128
Thiel H (1975) The size structure of the deep-sea benthos. Int Rev Ges Hydrobiol 60:576–606
Thiel H (1983) Meiobenthos and nanobenthos of the deep sea. In: Rowe GT (ed) The sea, vol 8. Wiley, New York, pp 167–230
Våge K, Pickart RS, Spall MA, Valdimarsson H, Jónsson S, Torres DJ, Østerhus S, Eldevik T (2011) Significant role of the North Icelandic Jet in the formation of Denmark Strait overflow water. Nat Geosci. https://doi.org/10.1038/ngeo1234
Våge K, Pickart RS, Spall MA, Moore GWK, Valdimarsson H, Torres DJ, Erofeeva SY, Nilsen JEØ (2013) Revised circulation scheme north of the Denmark Strait. Deep-Sea Res I 79:20–39
Vanaverbeke J, Soetaert K, Heip C, Vanreusel A (1997a) The metazoan meiobenthos along the continental slope of the Goban Spur (NE Atlantic). J Sea Res 38:93–107
Vanaverbeke J, Martínez Arbizu P, Dahms HU, Schminke HK (1997b) The metazoan meiobenthos along a depth gradient in the Arctic Laptev Sea with special attentions to nematode communities. Pol Biol 18:391–401
Vanhove S, Wittoeck J, Desmet G, Van den Berghe B, Herman RL, Bak RPM, Nieuwland G, Vosjan JH, Boldrin A, Rabitti S, Vincx M (1995) Deep-sea meiofauna communities in Antarctica: structural analysis and relation with the environment. Mar Ecol Prog Ser 127:65–76
Vanreusel A, Vincx M, Schram D, Gansbeke D (1995a) On the vertical distribution of the metazoan meiofauna in shelf break and upper slope habitats of the NE Atlantic. Int Rev Ges Hydrobiol 80:313–326
Vanreusel A, Vincx M, Bett BJ, Rice AL (1995b) Nematode biomass spectra at two abyssal sites in the NE Atlantic with a contrasting food supply. Int Rev Ges Hydrobiol 80:287–296
Vanreusel A, Clough L, Jacobsen K, Ambrose W, Jivaluk J, Ryheul V, Herman R, Vincx M (2000) Meiobenthos of the central Arctic Ocean with special emphasis on the nematode community structure. Deep-Sea Res 47:1855–1879
Vincx M, Bett BJ, Dinet A, Ferrero T, Gooday AJ, Lambshead PJD, Pfannkuche O, Soltwedel T, Vanreusel A (1994) Meiobenthos of the Northeast Atlantic. Adv Mar Biol 30:1–88
Wei C-L, Rowe GT, Escobar-Briones E, Boetius A, Soltwedel T (2010) Global patterns and predictions of seafloor biomass using random forests. PLoS ONE 5(12):e15323. https://doi.org/10.1371/journalpone0015323
Weisshappel JBF, Svavarsson J (1998) Benthic amphipods (Crustacea: Malacostraca) in Icelandic waters: diversity in relation to faunal patterns from shallow to intermediate deep Arctic and North Atlantic Oceans. Mar Biol 131:133–143
Wessel P, Smith WHF, Scharroo R, Luis JF, Wobbe F (2013) Generic Mapping Tools: improved version released. EOS, Trans Am Geophys Union 94:409–410
Yasuhara M, Grimm M, Brandão SN, Jöst A, Okahashi H, Iwatani H, Ostmann A, Martínez Arbizu P (2014) Deep-sea benthic ostracodes from multiple core and epibenthic sledge samples in Icelandic waters. Pol Polar Res 35(2):341–360
Acknowledgements
We would like to thank Captain Michael Schneider and his entire crew from the RV Meteor (leg M85/3), without whom the sample collection would not have been possible at all. Thanks to all those who helped to take the samples aboard. We are grateful to the technicians of Senckenberg am Meer for their help with meiofauna sorting. We also want to thank Dr. Saskia Brix for giving the possibility to work in the frame of the IceAGE project. We further thank two anonymous reviewers for their helpful comments and valuable suggestions, which improved this study.
Funding
The DFG funded the research expeditions on RV Meteor (BR3843/3-1) and RV Poseidon (BR3843/4-1). The first author received a research grant from the Senckenberg Gesellschaft für Naturforschung.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed by the authors.
Sampling and field studies
All necessary permits for sampling and observational field studies have been obtained by the authors from the competent authorities and are mentioned in the acknowledgements, if applicable.
Additional information
Communicated by S. Brix
This article is part of the Topical Collection on Biodiversity of Icelandic Waters by Karin Meißner, Saskia Brix, Ken M. Halanych and Anna Jazdzewska
Rights and permissions
About this article
Cite this article
Ostmann, A., Martínez Arbizu, P. Predictive models using randomForest regression for distribution patterns of meiofauna in Icelandic waters. Mar Biodiv 48, 719–735 (2018). https://doi.org/10.1007/s12526-018-0882-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12526-018-0882-9