Abstract
Background
Very long chain acyl-CoA dehydrogenase deficiency (VLCADD) is an inherited metabolic disease caused by deleterious mutations in the ACADVL gene that encodes very long chain acyl-CoA dehydrogenase (VLCAD), and which can present as cardiomyopathy in neonates, as hypoketotic hypoglycemia in infancy, and as myopathy in late-onset patients. Although many ACADVL mutations have been described, no prevalent mutations in the ACADVL gene have been associated with VLCADD. Herein, we report the clinical course of the disease and explore the genetic mutation spectrum in seven Chinese patients with VLCADD.
Methods
Seven Chinese patients, from newborn to 17 years old, were included in this study. Tandem mass spectrometry was performed to screen for VLCAD deficiency. All exons and flanking introns of the ACADVL gene were analyzed using polymerase chain reaction and direct sequencing. Online analysis tools were used to predict the impact of novel mutations.
Results
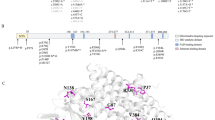
All cases had elevated serum levels of tetradecanoylcarnitine (C14:1) which is the characteristic biomarker for VLCADD. The phenotype of VLCADD is heterogeneous. Two patients were hospitalized for hypoactivity and hypoglycemia shortly after birth. Three patients showed hepatomegaly and hypoglycemia in infancy. The other two adolescent patients showed initial manifestations of exercise intolerance or rhabdomyolysis. Three of the patients died at the age of 6–8 months. Eleven different mutations in the ACADVL gene in the 7 patients were identified, including seven reported mutations (p.S22X, p.W427X, p.A213T, p.G222R, p.R450H, c.296-297delCA, c.1605+1G>T) and four novel mutations (p.S72F, p.Q100X, p.M437T, p.D466Y). The p.R450H and p.D466Y (14.28%, 2/14 alleles) mutations were identified in two alleles respectively.
Conclusions
The clinical manifestations were heterog-eneous and ACADVL gene mutations were heterozygous in the seven VLCADD Chinese patients. R450H may be a relatively common mutation in Asian populations. The genotype and phenotype had a certain correlation in our patients.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Lindner M, Hoffmann GF, Matern D. Newborn screening for disorders of fatty-acid oxidation: experience and recommendations from an expert meeting. J Inherit Metab Dis 2010;33:521–526.
Andresen BS, Bross P, Vianey-Saban C, Divry P, Zabot MT, Roe CR, et al. Cloning and characterization of human verylong-chain acyl-CoA dehydrogenase cDNA, chromosomal assignment of the gene and identification in four patients of nine different mutations within the VLCAD gene. Hum Mol Genet 1996;5:461–476.
Souri M, Aoyama T, Orii K, Yamaguchi S, Hashimoto T. Mutation analysis of very-long-chain acyl-coenzyme A dehydrogenase (VLCAD) deficiency: identification and characterization of mutant VLCAD cDNAs from four patients. Am J Hum Genet 1996;58:97–106.
Andresen BS, Olpin S, Poorthuis BJ, Scholte HR, Vianey-Saban C, Wanders R, et al. Clear correlation of genotype with disease phenotype in very-long-chain acyl-CoA dehydrogenase deficiency. Am J Hum Genet 1999;64:479–494.
Tong MK, Lam CS, Mak TW, Fu MY, Ng SH, Wanders RJ, et al. Very long-chain acyl-CoA dehydrogenase deficiency presenting as acute hypercapnic respiratory failure. Eur Respir J 2006;28:447–450.
Vianey-Saban C, Divry P, Brivet M, Nada M, Zabot MT, Mathieu M, et al. Mitochondrial very-long-chain acylcoenzyme A dehydrogenase deficiency: clinical characteristics and diagnostic considerations in 30 patients. Clin Chim Acta 1998;269:43–62.
Wood JC, Magera MJ, Rinaldo P, Seashore MR, Strauss AW, Friedman A. Diagnosis of very long chain acyl-dehydrogenase deficiency from an infant’s newborn screening card. Pediatrics 2001;108:E19.
Schiff M, Mohsen AW, Karunanidhi A, McCracken E, Yeasted R, Vockley J. Molecular and cellular pathology of very-longchain acyl-CoA dehydrogenase deficiency. Mol Genet Metab 2013;109:21–27
Wanders RJ, Vreken P, den Boer ME, Wijburg FA, van Gennip AH, IJlst L. Disorders of mitochondrial fatty acyl-CoA betaoxidation. J Inherit Metab Dis 1999;22:442–487.
Goetzman ES, Wang Y, He M, Mohsen AW, Ninness BK, Vockley J. Expression and characterization of mutations in human very long-chain acyl-CoA dehydrogenase using a prokaryotic system. Mol Genet Metab 2007;91:138–147.
Hoffmann L, Haussmann U, Mueller M, Spiekerkoetter U. VLCAD enzyme activity determinations in newborns identified by screening: a valuable tool for risk assessment. J Inherit Metab Dis 2012;35:269–277.
Zytkovicz TH, Fitzgerald EF, Marsden D, Larson CA, Shih VE, Johnson DM, et al. Tandem mass spectrometric analysis for amino, organic, and fatty acid disorders in newborn dried blood spots: a two-year summary from the New England Newborn Screening Program. Clin Chem 2001;47:1945–1955.
Han LS, Ye J, Qiu WJ, Gao XL, Wang Y, Gu XF. Selective screening for inborn errors of metabolism on clinical patients using tandem mass spectrometry in China: a four-year report. J Inherit Metab Dis 2007;30:507–514.
Tajima G, Sakura N, Shirao K, Okada S, Tsumura M, Nishimura Y, et al. Development of a new enzymatic diagnosis method for very-long-chain Acyl-CoA dehydrogenase deficiency by detecting 2-hexadecenoyl-CoA production and its application in tandem mass spectrometry-based selective screening and newborn screening in Japan. Pediatr Res 2008;64:667–672.
Law LK, Tang NL, Lam CW, Tong MK, Mak TW, Zhang WM, et al. Novel missense mutations in the first Chinese patient with very-long-chain acyl-CoA dehydrogenase deficiency. Clin Chim Acta 2007;375:173–174.
Ohashi Y, Hasegawa Y, Murayama K, Ogawa M, Hasegawa T, Kawai M, et al. A new diagnostic test for VLCAD deficiency using immunohistochemistry. Neurology 2004;62:2209–2213.
Mathur A, Sims HF, Gopalakrishnan D, Gibson B, Rinaldo P, Vockley J, et al. Molecular heterogeneity in very-long-chain acyl-CoA dehydrogenase deficiency causing pediatric cardiomyopathy and sudden death. Circulation 1999;99:1337–1343.
Gregersen N, Andresen BS, Corydon MJ, Corydon TJ, Olsen RK, Bolund L, et al. Mutation analysis in mitochondrial fatty acid oxidation defects: Exemplified by acyl-CoA dehydrogenase deficiencies, with special focus on genotype-phenotype relationship. Hum Mutat 2001;18:169–189.
Fukao T, Watanabe H, Orii K, Takahashi Y, Hirano A, Kondo T, et al. Myopathic form of very-long chain acyl-coa dehydrogenase deficiency: evidence for temperature-sensitive mild mutations in both mutant alleles in a Japanese girl. Pediatr Res 2001;49:227–231.
Smelt AH, Poorthuis BJ, Onkenhout W, Scholte HR, Andresen BS, van Duinen SG, et al. Very long chain acyl-coenzyme A dehydrogenase deficiency with adult onset. Ann Neurol 1998;43:540–544.
Gobin-Limballe S, McAndrew RP, Djouadi F, Kim JJ, Bastin J. Compared effects of missense mutations in Very-Long-Chain Acyl-CoA Dehydrogenase deficiency: combined analysis by structural, functional and pharmacological approaches. Biochim Biophys Acta 2010;1802:478–484.
Straussberg R, Strauss AW. A novel mutation of late-onset verylong-chain acyl-CoA dehydrogenase deficiency. Pediatr Neurol 2002;27:136–137.
Djouadi F, Aubey F, Schlemmer D, Ruiter JP, Wanders RJ, Strauss AW, et al. Bezafibrate increases very-long-chain acyl-CoA dehydrogenase protein and mRNA expression in deficient fibroblasts and is a potential therapy for fatty acid oxidation disorders. Hum Mol Genet 2005;14:2695–2703.
Kabuyama Y, Suzuki T, Nakazawa N, Yamaki J, Homma MK, Homma Y. Dysregulation of very long chain acyl-CoA dehydrogenase coupled with lipid peroxidation. Am J Physiol Cell Physiol 2010;298:C107–113.
Potter BK, Little J, Chakraborty P, Kronick JB, Evans J, Frei J, et al. Variability in the clinical management of fatty acid oxidation disorders: results of a survey of Canadian metabolic physicians. J Inherit Metab Dis 2012;35:115–123.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, RN., Li, YF., Qiu, WJ. et al. Clinical features and mutations in seven Chinese patients with very long chain acyl-CoA dehydrogenase deficiency. World J Pediatr 10, 119–125 (2014). https://doi.org/10.1007/s12519-014-0480-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12519-014-0480-2