Abstract
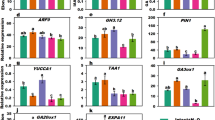
Plant height is determined by the processes of cell proliferation and elongation. Plant hormones play key roles in a species-dependent manner in these processes. We used paclobutrazol (PAC) at 400 mg·L-1 in this study to spray Agapanthus praecox ssp. orientalis plants in order to induce dwarf scape (inflorescence stem). Morphological examination showed that PAC reduced scape height by inhibiting the cell elongation by 54.56% and reducing cell proliferation by 10.45% compared to the control. Quantification and immunolocalization of endogenous gibberellins (GAs) and indole-3-acetic acid (IAA) showed that the GA1, GA3, and GA4 levels and the IAA gradient were reduced. Among these hormones, GA4 was the key component of GAs, which decreased 59.51-92.01% compared to the control in scape. The expression of cell wall synthesis related genes cellulose synthase (CESA) and UDP-glucuronic acid decarboxylase (UXS) were upregulated significantly, whereas cell wall loosening gene xyloglucan endotransglucosylase 2 (XET2) was downregulated by 99.99% surprisingly. Correlation analysis suggested GA regulated cell elongation and auxin modulated cell proliferation in Agapanthus scape. Additionally, the accumulation of sugars played roles in cell wall synthesis and cell expansion. These results indicated GA and IAA signals triggered a downstream signaling cascade, controlled cell expansion and proliferation during scape elongation.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Ayano M, Kani T, Kojima M, Sakakibara H, Kitaoka T, Kuroha T, Angeles-Shim RB, Kitano H, Nagai K, Ashikari M (2014) Gibberellin biosynthesis and signal transduction is essential for internode elongation in deepwater rice. Plant Cell Environ 37:2313–2324
Bulankova P, Akimcheva S, Fellner N, Riha K (2013) Identification of Arabidopsis meiotic cyclins reveals functional diversification among plant cyclin genes. PLoS Genet 9:e1003508
Carpita NC, Gibeaut DM (1993) Structural models of primary cell walls in flowering plants: consistency of molecular structure with the physical properties of the walls during growth. Plant J 3:1–30
Chen IJ, Lo W, Chuang J, Cheuh C, Fan Y, Lin L, Wu S, Wang L (2013) A chemical genetics approach reveals a role of brassinolide and cellulose synthase in hypocotyl elongation of etiolated Arabidopsis seedlings. Plant Sci 209:46–57
Chen S, Gao R, Wang H, Wen M, Xiao J, Bian N, Zhang R, Hu W, Cheng S, Bie T, Wang X (2015) Characterization of a novel reduced height gene (Rht23) regulating panicle morphology and plant architecture in bread wheat. Euphytica 203:583–594
Christiaens A, Pauwels E, Gobin B, Van Labeke MC (2015) Flower differentiation of azalea depends on genotype and not on the use of plant growth regulators. Plant Growth Regul 75:245–252
Claeys H, De Bodt S, Inzé D (2014) Gibberellins and DELLAs: central nodes in growth regulatory networks. Trends Plant Sci 19:231–239
Cosgrove DJ (2005) Growth of the plant cell wall. Nat Rev Mol Cell Biol 6:850–861
Dai Y, Wang H, Li B, Huang J, Liu X, Zhou Y, Mou Z, Li J (2006) Increased expression of MAP KINASE KINASE7 causes deficiency in polar auxin transport and leads to plant architectural abnormality in Arabidopsis. Plant Cell 18:308–320
Davière J, Achard P (2016) A pivotal role of DELLAs in regulating multiple hormone signals. Mol Plant 9:10–20
Dayan J, Voronin N, Gong F, Sun T, Hedden P, Fromm H, Aloni R (2012) Leaf-induced gibberellin signaling is essential for internode elongation, cambial activity, and fiber differentiation in tobacco stems. Plant Cell 24:66–79
Donnelly PM, Bonetta D, Tsukaya H, Dengler RE, Dengler NG (1999) Cell cycling and cell enlargement in developing leaves of Arabidopsis. Dev Biol 215:407–419
Feng S, Martinez C, Gusmaroli G, Wang Y, Zhou J, Wang F, Chen L, Yu L, Iglesias-Pedraz JM, Kircher S, Schäfer E, Fu X, Fan L-M, Deng XW (2008) Coordinated regulation of Arabidopsis thaliana development by light and gibberellins. Nature 451:475–479
Gao S, Xie X, Yang S, Chen Z, Wang X (2012) The changes of GA level and signaling are involved in the regulation of mesocotyl elongation during blue light mediated de-etiolation in Sorghum bicolor. Mol Biol Rep 39:4091–4100
Gou J, Strauss SH, Tsai CJ, Fang K, Chen Y, Jiang X, Busov VB (2010) Gibberellins regulate lateral root formation in Populus through interactions with auxin and other hormones. Plant Cell 22:623–639
Guanfang W, Hongzhi K, Yujin S, Zhang X, Zhang W, Altman N, de Pamphilis CW, Ma H (2004) Genome-wide analysis of the cyclin family in Arabidopsis and comparative phylogenetic analysis of plant cyclin-like proteins. Plant Physiol 135:1084–1099
Hedden P, Sponsel V (2015) A century of gibberellin research. J Plant Growth Regul 34:740–760
Hepworth J, Lenhard M (2014) Regulation of plant lateral-organ growth by modulating cell number and size. Curr Opin Plant Biol 17:36–42
Jan A, Yang G, Nakamura H, Ichikawa H, Kitano H, Matsuoka M, Matsumoto H, Komatsu S (2004) Characterization of a xyloglucan endotransglucosylase gene that is up-regulated by gibberellin in rice. Plant Physiol 136:3670–3681
Kevei Z, Baloban M, Da Ines O, Tiricz H, Kroll A, Regulski K, Mergaert P, Kondorosi E (2011) Conserved CDC20 cell cycle functions are carried out by two of the five isoforms in Arabidopsis thaliana. PLoS One 6:e20618
Kutschera U, Niklas KJ (2013) Cell division and turgor-driven stem elongation in juvenile plants: a synthesis. Plant Sci 207:45–56
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2-ΔΔCT method. Methods 25:402–408
Lv H, Zheng J, Wang T, Fu J, Huai J, Min H, Zhang X, Tian B, Shi Y, Wang G (2014) The maize d2003, a novel allele of VP8, is required for maize internode elongation. Plant Mol Biol 84:243–257
Mashiguchi K, Tanaka K, Sakai T, Sugawara S, Kawaide H, Natsume M, Hanada A, Yaeno T, Shirasu K, Yao H, McSteen P, Zhao Y, Hayashi K, Kamiya Y, Kasahara H (2011) The main auxin biosynthesis pathway in Arabidopsis. Proc Natl Acad Sci USA 108:18512–18517
Middleton AM, Úbeda-Tomás S, Griffiths J, Holman T, Hedden P, Thomas SG, Phillips AL, Holdsworth MJ, Bennett MJ, King JR (2012) Mathematical modeling elucidates the role of transcriptional feedback in gibberellin signaling. Proc Natl Acad Sci USA 109: 7571–7576
Nemhauser JL, Hong F, Chory J (2006) Different plant hormones regulate similar processes through largely nonoverlapping transcriptional responses. Cell 126:467–475
Peng L, Hocart CH, Redmond JW, Williamson RE (2000) Fractionation of carbohydrates in Arabidopsis root cell walls shows that three radial swelling loci are specifically involved in cellulose production. Planta 211:406–414
Serrani J, Fos M, Atarés A, García Martínez J (2007) Effect of gibberellin and auxin on parthenocarpic fruit growth induction in the cv Micro-Tom of tomato. J Plant Growth Regul 26:211–221
Stamm P, Kumar P (2013) Auxin and gibberellin responsive Arabidopsis SMALL AUXIN UP RNA36 regulates hypocotyl elongation in the light. Plant Cell Rep 32:759–769
Sun T (2011) The molecular mechanism and evolution of the GA–GID1–DELLA signaling module in plants. Curr Biol 21:338–345
Szymanski DB, Cosgrove DJ (2009) Dynamic coordination of cytoskeletal and cell wall systems during plant cell morphogenesis. Curr Biol 19:800–811
Takatsuka H, Ohno R, Umeda M (2009) The Arabidopsis cyclindependent kinase-activating kinase CDKF;1 is a major regulator of cell proliferation and cell expansion but is dispensable for CDKA activation. Plant J 59:475–487
Topp SH, Rasmussen SK (2012) Evaluating the potential of SHI expression as a compacting tool for ornamental plants. Plant Sci 187:19–30
Vanneste S, Friml J (2009) Auxin: a trigger for change in plant development. Cell 136:1005–1016
Vivian-Smith A, Koltunow AM (1999) Genetic analysis of growthregulator-induced parthenocarpy in Arabidopsis. Plant Physiol 121:437–452
Wang G, Que F, Xu Z, Wang F, Xiong A (2015) Exogenous gibberellin altered morphology, anatomic and transcriptional regulatory networks of hormones in carrot root and shoot. BMC Plant Biol 15:1–12
Wang L, Mu C, Du M, Chen Y, Tian X, Zhang M, Li Z (2014) The effect of mepiquat chloride on elongation of cotton (Gossypium hirsutum L.) internode is associated with low concentration of gibberellic acid. Plant Sci 225:15–23
Xie Q, Chen G, Chen X, Deng L, Liu Q, Zhang Y, Hu Z (2014) Jointly silencing BoDWARF, BoGA20ox and BoSP (SELFPRUNING) produces a novel miniature ornamental Brassica oleracea var. acephala f. tricolor variety. Mol Breeding 34:99–113
Yamaguchi S (2008) Gibberellin metabolism and its regulation. Annu Rev Plant Biol 59:225–251
Zhang D, Ren L, Yue J, Wang L, Zhuo L, Shen X (2013) A comprehensive analysis of flowering transition in Agapanthus praecox ssp. orientalis (Leighton) Leighton by using transcriptomic and proteomic techniques. J Proteomics 80:1–25
Zhang D, Ren L, Yue J, Wang L, Zhuo L, Shen X (2014) GA4 and IAA were involved in the morphogenesis and development of flowers in Agapanthus praecox ssp. orientalis. J Plant Physiol 171:966–976
Zheng R, Wu Y, Xia Y (2012) Chlorocholine chloride and paclobutrazol treatments promote carbohydrate accumulation in bulbs of Lilium Oriental hybrids ‘Sorbonne’. J Zhejiang Univ Sci B 13:136–144
Zhu X, Shi Y, Lei G, Fry SC, Zhang B, Zhou Y, Braam J, Jiang T, Xu X, Mao C, Pan Y, Yang J, Wu P, Zheng S (2012) XTH31, encoding an in vitro XEH/XET-active enzyme, regulates aluminum sensitivity by modulating in vivo XET action, cell wall xyloglucan content, and aluminum binding capacity in Arabidopsis. Plant Cell 24:4731–4747
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Yue, Jh., Zhang, D., Ren, L. et al. Gibberellin and auxin signals control scape cell elongation and proliferation in Agapanthus praecox ssp. orientalis . J. Plant Biol. 59, 358–368 (2016). https://doi.org/10.1007/s12374-016-0056-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12374-016-0056-x