Abstract
In 2015, many original articles pertaining to cardiovascular imaging with impressive quality were published in the Journal of Nuclear Cardiology. In a set of 2 articles, we provide an overview of these contributions to facilitate for the interested reader a quick review of the advancements that occurred in the field over this year. In this first article, we focus on arterial plaque imaging, cardiac positron emission tomography, computed tomography, and magnetic resonance imaging.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
One year ago, we reviewed the most interesting original articles in cardiovascular imaging that were published in the Journal of Nuclear Cardiology in 2014.1,2 Two of these articles were selected by the editors of the Journal for the best basic/technical and clinical research awards and were recognized at the 20th Annual Scientific Session of the American Society of Nuclear Cardiology.3,4 In 2015, many original contributions with impressive quality were published in the Journal on a variety of topics encompassing myocardial perfusion imaging (MPI) using single-photon emission computed tomography (SPECT) and positron emission tomography (PET), computed tomography (CT), cardiac magnetic resonance imaging (CMR), and multimodality imaging extending from basic science to translational and clinical investigations pertaining to the field. Just as last year, we will divide our review into two parts: Part 1 will focus on plaque imaging, PET, CT, and CMR while Part 2 will focus on SPECT.
Atherosclerotic Plaque Imaging
The visualization of atherosclerotic plaques, and more specifically vulnerable plaques, has received increasing attention with several breakthroughs over the last few years, and more recently this year. A particular group at high-risk are HIV-infected patients who often have increased arterial inflammation that leads to vasculopathy and premature cardiovascular events.5 In a recent paper, Knudsen et al used F18-fluorodeoxyglucose (FDG) to assess arterial inflammation of the carotid arteries and aorta in 26 HIV-infected patients without known cardiac disease, low Framingham score and fully viral-suppressed, and compared them to 25 healthy controls.6 Interestingly, there was no difference in standardized uptake values (SUV) in the arterial vasculatures, intimal media thickness, or soluble biomarkers of inflammation between the groups (Figure 1).6 Although prior studies showed conflicting results with increased arterial F18-FDG uptake in the arterial vasculature of HIV-infected patients,7 the study cohort by Krudsen et al consisted of fully viral-suppressed patients with no significant immune activation, and therefore minimal arterial inflammation.6 A potential clinical research and application of such findings is to assess whether treatment-related changes in F18-FDG uptake, which appears to be a surrogate of arterial inflammation and possibly immune activation, can be used as predictor of clinical response to HIV treatment, and therefore identify patients at lower risk of developing arterial vasculopathy.8
Fusion PET/CT axial images from the ascending (panel A) and abdominal aorta (panel B) of an HIV-infected patient and a healthy control showing modest FDG uptake in the arterial wall. AA, abdominal aorta; FDG, fluorodeoxyglucose; SVC, superior vena cava. Reproduced with permission from Ref. 6 (Figure 4, p 378)
While F18-FDG PET imaging for vascular inflammation has great potential, it remains hindered by several significant technical and biological difficulties leading still to unresolved issues.9 F18-Flotegatide, emerged as a novel αvβ3 integrin tracer that binds to aortic plaque and preferentially to areas of inflammation and angiogenesis, hence potentially distinguishing vulnerable from non-vulnerable plaques, and overcoming several of the limitations of F18-FDG.3 This paper by Su et al,3 which was published in 2014 in the Journal, was recently awarded the best basic science paper. As a continuation of this work and translation to clinical application, Golestani et al assessed the feasibility of using radiolabeled F18-arginine-glycine-aspartate for target imaging of the same αvβ3 integrin receptor in an ex-vivo human model.10 Indeed, using 19 carotid endarterectomy specimens, segmental-to-total ratio of radioactivity was calculated and was shown to modestly correlate with immunohistochemical staining score of the integrin receptor.10 The study further supports the use of non-invasive imaging for the identification and quantification of biological markers of atherosclerosis, and puts us one step closer to the clinical goal of assessment of vulnerable plaques, plaque progression, and regression in vivo.11
Cardiac Amyloidosis
Cardiac amyloidosis is an infiltrative disease associated with heart failure and restrictive cardiomyopathy. The two major types of cardiac amyloidosis are cardiac amyloid light chain and transthyretin-related cardiac amyloidosis. While early recognition of cardiac amyloidosis is of major clinical importance, so is the ability to differentiate between subtypes. Indeed, both prognosis and therapeutic options vary drastically depending on the subtype.12 Apical sparing pattern on echocardiography and diffused gadolinium enhancement on CMR have good sensitivity and specificity to diagnose cardiac amyloidosis; however, neither can distinguish the different subtypes.12,13 Recently, nuclear imaging using Tc-99m phosphate derivatives have shown promising results as they selectively bind transthyretin amyloid fibrils, hence distinguishing both subtypes and limiting the need for endomyocardial biopsy.12,13 Similarly, F18-sodium fluoride is another PET tracer that was recently described as a potential noninvasive imaging tool to distinguish transthyretin from light chain cardiac amyloidosis as illustrated in few case reports.14
The quantification of cardiac amyloidosis is an equally important breakthrough in the field of nuclear cardiology as it enables adequate and accurate monitoring of response to therapy. Indeed, Kero et al used C11-PIB (Pittburg compound B) PET imaging in 10 patients with cardiac amyloidosis and 5 healthy volunteers.15 Using fractional uptake rate model and integrating time-activity curve, the left ventricular retention index of C11-PIB was calculated using standard manual technique and compared to a semi-automated quantitative process (Figure 2). Not only did the results show excellent correlation (r = 0.98), the inter-reader reproducibility of the quantitative software was exceedingly high (interclass correlation coefficient ICC > 0.98).15 In an accompanied editorial, Dorbola et al discussed the potential application for this software for “precise imaging guided management of cardiac amyloidosis”.16 The role of serial scanning to evaluate the clinical response to therapy has been recently evaluated by Castano et al; despite clinical worsening of disease, there were no significant changes in quantitative scores with serial Tc99m-pyrophosphate scans over 1.5 years of follow-up imaging.17 Further advances in quantitative molecular imaging of cardiac amyloidosis are crucial as they may alter the course of the disease.
Analysis results of 11C-PIB with semiautomatic software at 10-20 min; images are modified from Carimas showing polar plots and histograms for five different subjects: A Healthy volunteer. B Transthyretin amyloidosis. C Amyloid light chain kappa amyloidosis. D Amyloid light chain lambda amyloidosis. E Amyloid light chain lambda amyloidosis. The histogram y-axis shows frequency (relative number of pixels) and the x-axis shows retention index value. Reproduced with permission from Ref. 15 (Figure 6)
Cardiac CT
Increasing efforts have been placed on performing stress only MPI to decrease radiation exposure and simplify the imaging protocol18; however, rest images are often required particularly when the stress study is not completely normal. Uretsky et al showed that combining a low-dose coronary artery calcium score by cardiac CT with stress images reduced the need for rest images in 72% of patients with a probably normal stress study, 47% with an equivocal, and 40% with probably abnormal study.19 Indeed, a normal coronary calcium scan increased the readers’ confidence concerning the interpretation of minimal abnormalities.
The routine use of CT is increasing in the field of nuclear cardiology, particularly with PET imaging where CT attenuation correction is routinely performed. The CT scan for attenuation correction allows the visualization of coronary calcification but not adequate quantification. In a recent paper, Kaster et al showed that a single low-dose rest coronary calcium score CT scan can be used for accurate attenuation correction for both rest and stress PET MPI, and allows quantification of calcium score without a significant increase in the radiation dose.20
Another advantage of performing cardiac CT is the assessment and quantification of epicardial fat volume. Indeed, epicardial adipose tissue has been linked to coronary artery disease through the release of pro-inflammatory cytokines. In a recent paper, Khawaja et al quantified epicardial fat volume of 396 patients undergoing SPECT MPI with CT attenuation correction; patients with reversible perfusion defect has significant increase in regional epicardial fat as compared to those with normal perfusion or scar.21 Hence, detection of increased epicardial adipose tissue adjacent to a coronary artery is a potential new risk factor for developing coronary artery disease.21 Future studies are warranted however to validate these findings, assess causality, and define a threshold for epicardial fat volume that could be used in clinical practice.
The classic teaching that diabetes is a coronary artery disease equivalent has been recently challenged. Van den Hoogen et al evaluated the prognostic value of coronary CT angiogram in 525 diabetic patients without chest pain syndrome; those with normal CT angiogram had very low event rates (3%), while those with obstructive disease had significant increase in events (hazard ratio 11.1, P = 0.001).22 This paper adds to the existing literature of screening asymptomatic diabetic patients showing that a substantial proportion of patients have zero calcium score, low event rates, and behave differently than patients with known coronary heart disease; the debate however is still on.23-26
An important issue that pertains to the use of cardiac CT, whether low-dose for attenuation correction or of diagnostic quality in symptomatic or asymptomatic individuals, is that it may reveal incidental findings. Qureshi et al showed that at least 1 in 10 patients undergoing CT attenuated MPI will have an incidental finding, lung nodules being the most common (almost two-third).27 Each incidental finding was associated with 14% incidental-related hospitalization, threefold increase in cancer-specific mortality after multivariate adjustment (Figure 3), and average of 6699$ in additional related cost.27 The challenge we face is balancing on one hand the fact that a majority of the incidental findings are benign with the “downstream interventions, related costs, patient anxiety, morbidity, potential diagnosis of malignancy, and the ugly possibility of poor patient outcomes when making recommendations for further testing or treatment.”28
Figure 2. Kaplan Meier survival curve for cancer-specific mortality of patients with incidental findings compared to patients without incidental findings. Reproduced with permission from Ref. 2 (Figure 2)
CMR
Stress CMR has been shown in several studies to have a high diagnostic accuracy for the diagnosis of coronary artery disease. The MR-IMPACT,29 MR-IMPACT II30 and CE-MARC31 trials showed higher sensitivity of CMR as compared to stress MPI SPECT; these studies greatly influenced the clinical practice in Europe in favor of shifting stress imaging toward CMR. However, these studies had many limitations including the lack of a core lab for MPI interpretation which likely resulted in significant bias favoring CMR. In a recent study, Ahmad et al compared the diagnostic accuracy of stress CMR using visual assessment (most commonly used in clinical practice) vs SPECT MPI among patients presenting to the emergency room, and showed similar diagnostic accuracy of 89% and 88%, respectively.32 The cohort of Ahmad et al, however, was different from other published studies as it is focused on the evaluation of chest pain in the emergency room. This is a very common clinical scenario and therefore represents an area of great interest and further research. The pretest probability of disease remains key in the clinical decision for noninvasive imaging and downstream further testing.33
PET MPI
MPI with PET is performed with pharmacological stress testing given the short half-life of most PET perfusion tracers such as rubidium. The ability to perform exercise stress MPI PET has been of interest for many years. Nitrogen-13 ammonia is a PET perfusion racer with longer half-life compared to rubidium. Aggarwal et al tested the feasibility and diagnostic accuracy of exercise stress PET with nitrogen-13 ammonia on 265 patients between 2009 and 2012.34 The test was feasible in all patients, yielded good image quality for more than 98% of patients, and had a sensitivity and specificity of 86 and 74%, respectively. Abnormal summed stress score was an independent predictor of coronary artery stenosis after multivariate adjustment.34 What is gained with exercise stress PET in terms of assessing functional capacity, metabolic equivalent test, heart rate recovery, and others, is lost with the inability to assess dynamic perfusion and coronary flow reserve.35 Also, the diagnostic accuracy of exercise stress PET seems lower than the published literature for the pharmacological counterpart, and needs further evaluation including head-to-head comparison.35
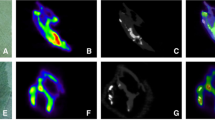
F18-FDG is another tracer used in PET cardiac imaging. Although a metabolic rather than a perfusion tracer, it has been recently used for direct ischemia imaging or “hot imaging” as opposed to “cold imaging” used with traditional perfusion tracers. Arun et al performed stress testing for 45 patients without known coronary artery disease that underwent exercise F18-FDG PET imaging and Tc-99m SPECT MPI.36 Coronary angiogram was subsequently performed to assess for coronary artery stenosis. Exercise F18-FDG exhibited better performance parameters than SPECT MPI (Figures 4 and 5).36 The use of F18-FDG for MPI is attractive since it does not require a cyclotron on site (as opposed to N13-ammonia) or generator (as opposed to rubidium) and is easily available. While the concept of hot imaging seems interesting, the preparation of patients for F18-FDG is complex and can be challenging particularly in patients with diabetes, as it involves reducing sugar intake and having high-fat diet prior to the stress test to ensure no FDG uptake by normal myocytes.36 Also, the issue of low specificity (44%) with a high false positive FDG uptake in the lateral wall is of particular concern.37 Thus, although the concept is interesting and of high clinical utility, further research and fine tuning of the technique is warranted before abandoning cold imaging.37
Coronary angiography-LAO cranial view showing the normal RCA (A), normal LAD (B), and RAO caudal view showing the normal LCX and OM major (C). SPECT-MPS attenuation corrected images showing no evidence of any stress-induced reversible perfusion defect in the LV myocardium (D). Exercise F18-FDG images showing no abnormal FDG uptake in the LV myocardium (E). Reproduced with permission from Ref. 36 (Figure 2, p 104)
Coronary angiography-LAO cranial view showing normal RCA (B), RAO caudal view showing LCX total cut off (A), and RAO cranial view showing normal LAD (C). SPECT-MPS images showing stress-induced reversible defect of mild severity in part of the inferolateral wall involving 10% to 12% of the LV myocardium (D). Exercise 18F-FDG images showing exercise induced FDG avid ischemia in mid and basal inferolateral segments involving 10% to 12% of the LV myocardium (E). Reproduced with permission from Ref. 36 (Figure 5, p 106)
Moreover, in patients with dilated cardiomyopathy, F18-FDG PET imaging is often performed to assess for viability. While the focus has always been on the left ventricular myocardium, Wang et al studied F18-FDG uptake in the right ventricle in 63 patients. Increased tracer uptake was associated with worse right ventricular ejection fraction (r = −0.571, P < 0.001).38 A relative right ventricular FDG uptake >0.795 (ratio of right to left ventricle corrected SUV) was an independent predictor of death or transplantation (Hazard ratio 5.0, P = 0.004).38 The ability to image both ventricles with FDG PET provides useful information about myocardial substrate and an additional tool for risk stratification, and perhaps early identification of patients who might benefit from resynchronization, metabolic modulators, or transplantation.39
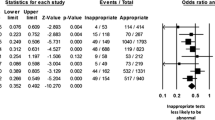
Although PET is a powerful non-invasive diagnostic and prognostic tool, there are concerns regarding the inappropriate use of this test. Winchester et al showed that among 521 patients undergoing cardiac PET, 10% of the tests were inappropriate; they were almost all normal, associated with low catheterization rate, failed to detect occult ischemia in any patient, and had no clinical utility.40 The study is one of the first to assess the use of appropriateness criteria for PET imaging which should be readily available in the clinical practice to optimize use while emphasizing clinical value in a cost-effective manner.41
Abbreviations
- CMR:
-
Cardiac magnetic resonance
- CT:
-
Computed tomography
- FDG:
-
Fluorodeoxyglucose
- MPI:
-
Myocardial perfusion imaging
- PET:
-
Positron emission tomography
- SPECT:
-
Single-photon emission computed tomography
- SUV:
-
Standardized uptake value
References
AlJaroudi WA, Hage FG. Review of cardiovascular imaging in the journal of nuclear cardiology in 2014: Part 1 of 2: Positron emission tomography, computed tomography, and neuronal imaging. J Nucl Cardiol 2015;22:507-12.
Hage FG, AlJaroudi WA. Review of cardiovascular imaging in the journal of nuclear cardiology in 2014: Part 2 of 2: Myocardial perfusion imaging. J Nucl Cardiol 2015;22:714-9.
Su H, Gorodny N, Gomez LF, Gangadharmath UB, Mu F, Chen G, et al. Atherosclerotic plaque uptake of a novel integrin tracer (1)(8)f-flotegatide in a mouse model of atherosclerosis. J Nucl Cardiol 2014;21:553-62.
Al Badarin FJ, Wimmer AP, Kennedy KF, Jacobson AF, Bateman TM. The utility of admire-hf risk score in predicting serious arrhythmic events in heart failure patients: Incremental prognostic benefit of cardiac 123i-mibg scintigraphy. J Nucl Cardiol 2014;21:756-62 quiz 753-755, 763-755.
Post WS, Budoff M, Kingsley L, Palella FJ Jr, Witt MD, Li X, et al. Associations between HIV infection and subclinical coronary atherosclerosis. Ann Intern Med 2014;160:458-67.
Knudsen A, Hag AM, Loft A, von Benzon E, Keller SH, Moller HJ, et al. HIV infection and arterial inflammation assessed by (18)f-fluorodeoxyglucose (fdg) positron emission tomography (pet): A prospective cross-sectional study. J Nucl Cardiol 2015;22:372-80.
Subramanian S, Tawakol A, Burdo TH, Abbara S, Wei J, Vijayakumar J, et al. Arterial inflammation in patients with hiv. JAMA 2012;308:379-86.
Tawakol A, Grinspoon SK. Imaging atherosclerotic burden and inflammation: Insights into the spectrum of atherosclerotic disease in HIV. J Nucl Cardiol 2015;22:381-4.
Sadeghi MM. (18)f-fdg pet and vascular inflammation: Time to refine the paradigm? J Nucl Cardiol 2015;22:319-24.
Golestani R, Mirfeizi L, Zeebregts CJ, Westra J, de Haas HJ, Glaudemans AW, et al. Feasibility of [18f]-rgd for ex vivo imaging of atherosclerosis in detection of alphavbeta3 integrin expression. J Nucl Cardiol 2015. doi:10.1007/s12350-014-0061-8.
Reyes E. A novel pet tracer for targeted imaging of atherosclerosis. J Nucl Cardiol 2015. doi:10.1007/s12350-015-0088-5.
Aljaroudi WA, Desai MY, Tang WH, Phelan D, Cerqueira MD, Jaber WA. Role of imaging in the diagnosis and management of patients with cardiac amyloidosis: State of the art review and focus on emerging nuclear techniques. J Nucl Cardiol 2014;21:271-83.
Bokhari S, Shahzad R, Castano A, Maurer MS. Nuclear imaging modalities for cardiac amyloidosis. J Nucl Cardiol 2014;21:175-84.
Van Der Gucht A, Galat A, Rosso J, Guellich A, Garot J, Bodez D, et al. [18f]-naf pet/ct imaging in cardiac amyloidosis. J Nucl Cardiol 2015. doi:10.1007/s12350-015-0287-0.
Kero T, Lindsjo L, Sorensen J, Lubberink M. Accurate analysis and visualization of cardiac c-pib uptake in amyloidosis with semiautomatic software. J Nucl Cardiol 2015. doi:10.1007/s12350-015-0149-9.
Dorbala S, Kijewski MF, Park MA. Quantitative molecular imaging of cardiac amyloidosis: The journey has begun. J Nucl Cardiol 2015
Castano A, DeLuca A, Weinberg R, Pozniakoff T, Blaner WS, Pirmohamed A, et al. Serial scanning with technetium pyrophosphate (Tc-PYP) in advanced ATTR cardiac amyloidosis. J Nucl Cardiol 2015. doi:10.1007/s12350-015-0261-x.
Gowd BM, Heller GV, Parker MW. Stress-only spect myocardial perfusion imaging: A review. J Nucl Cardiol 2014;21:1200-12.
Uretsky S, Cohen R, Argulian E, Balasundaram K, Supariwala A, Subero M, et al. Combining stress-only myocardial perfusion imaging with coronary calcium scanning as a new paradigm for initial patient work-up: An exploratory analysis. J Nucl Cardiol 2015;22:89-97.
Kaster TS, Dwivedi G, Susser L, Renaud JM, Beanlands RS, Chow BJ, et al. Single low-dose ct scan optimized for rest-stress pet attenuation correction and quantification of coronary artery calcium. J Nucl Cardiol 2015;22:419-28.
Khawaja T, Greer C, Thadani SR, Kato TS, Bhatia K, Shimbo D, et al. Increased regional epicardial fat volume associated with reversible myocardial ischemia in patients with suspected coronary artery disease. J Nucl Cardiol 2015;22:325-33.
van den Hoogen IJ, de Graaf MA, Roos CJ, Leen AC, Kharagjitsingh AV, Wolterbeek R, et al. Prognostic value of coronary computed tomography angiography in diabetic patients without chest pain syndrome. J Nucl Cardiol 2015. doi:10.1007/s12350-015-0213-5.
Beller GA, Bourque JM. Screening asymptomatic patients with type 2 diabetes: The debates persist. J Nucl Cardiol 2015. doi:10.1007/s12350-015-0283-4.
Berman DS, Budoff MJ, Min JK, Raggi P, Rozanski A, Shaw L, et al. Non-invasive imaging in assessment of the asymptomatic diabetic patient: Is it of value? J Nucl Cardiol 2015. doi:10.1007/s12350-015-0241-1.
Gibbons RJ. Screening asymptomatic patients with type 2 diabetes is recommended-con. J Nucl Cardiol 2015. doi:10.1007/s12350-015-0248-7.
Petretta M, Cuocolo A. Screening asymptomatic patients with type 2 diabetes is recommended: Pro. J Nucl Cardiol 2015. doi:10.1007/s12350-015-0250-0.
Qureshi WT, Alirhayim Z, Khalid F, Al-Mallah MH. Prognostic value of extracardiac incidental findings on attenuation correction cardiac computed tomography. J Nucl Cardiol 2015. doi:10.1007/s12350-015-0223-3.
Bhambhvani P. The good, bad, and ugly of incidental findings on cardiovascular-computed tomography. J Nucl Cardiol 2015. doi:10.1007/s12350-015-0244-y.
Schwitter J, Wacker CM, van Rossum AC, Lombardi M, Al-Saadi N, Ahlstrom H, et al. Mr-impact: Comparison of perfusion-cardiac magnetic resonance with single-photon emission computed tomography for the detection of coronary artery disease in a multicentre, multivendor, randomized trial. Eur Heart J 2008;29:480-9.
Schwitter J, Wacker CM, Wilke N, Al-Saadi N, Sauer E, Huettle K, et al. Mr-impact ii: Magnetic resonance imaging for myocardial perfusion assessment in coronary artery disease trial: Perfusion-cardiac magnetic resonance vs. Single-photon emission computed tomography for the detection of coronary artery disease: A comparative multicentre, multivendor trial. Eur Heart J 2013;34:775-81.
Greenwood JP, Maredia N, Younger JF, Brown JM, Nixon J, Everett CC, et al. Cardiovascular magnetic resonance and single-photon emission computed tomography for diagnosis of coronary heart disease (CE-MARC): A prospective trial Lancet. 2012;379:453-60.
Ahmad IG, Abdulla RK, Klem I, Margulis R, Ivanov A, Mohamed A, et al. Comparison of stress cardiovascular magnetic resonance imaging (CMR) with stress nuclear perfusion for the diagnosis of coronary artery disease. J Nucl Cardiol 2015. doi:10.1007/s12350-015-0242-0.
Gupta H. Functional imaging for chest pain in the emergency room: To do or not to do? J Nucl Cardiol 2015. doi:10.1007/s12350-015-0225-1.
Aggarwal NR, Drozdova A, Wells Askew J 3rd, Kemp BJ, Chareonthaitawee P. Feasibility and diagnostic accuracy of exercise treadmill nitrogen-13 ammonia pet myocardial perfusion imaging of obese patients. J Nucl Cardiol 2015. doi:10.1007/s12350-015-0073-z.
Gould KL, Johnson NP. Exercise pet: More insight or more complex? J Nucl Cardiol 2015. doi:10.1007/s12350-015-0099-2.
Arun S, Mittal BR, Bhattacharya A, Rohit MK. Comparison of tc-99m tetrofosmin myocardial perfusion scintigraphy and exercise f18-fdg imaging in detection of myocardial ischemia in patients with coronary artery disease. J Nucl Cardiol 2015;22:98-110.
Jain D, He ZX. Direct myocardial ischemia imaging with exercise (1)(8)fdg. J Nucl Cardiol 2015;22:111-4.
Wang L, Ma X, Xiang L, Lu M, Yan C, Zhao S, et al. The characterization and prognostic significance of right ventricular glucose metabolism in non-ischemic dilated cardiomyopathy. J Nucl Cardiol 2015. doi:10.1007/s12350-015-0165-9.
Neglia D. Positron emission tomography: An additional prognostic tool in dilated cardiomyopathy? J Nucl Cardiol 2015. doi:10.1007/s12350-015-0254-9.
Winchester DE, Chauffe RJ, Meral R, Nguyen D, Ryals S, Dusaj R, et al. Clinical utility of inappropriate positron emission tomography myocardial perfusion imaging: Test results and cardiovascular events. J Nucl Cardiol 2015;22:9-15.
Hendel RC. The value and appropriateness of positron emission tomography: An evolving tale. J Nucl Cardiol 2015;22:16-21.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
AlJaroudi, W.A., Hage, F.G. Review of cardiovascular imaging in the journal of nuclear cardiology in 2015. Part 1 of 2: Plaque imaging, positron emission tomography, computed tomography, and magnetic resonance. J. Nucl. Cardiol. 23, 122–130 (2016). https://doi.org/10.1007/s12350-015-0319-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12350-015-0319-9