Abstract
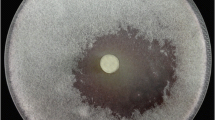
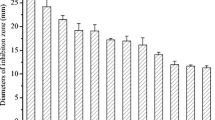
Control of plant pathogen Sclerotinia sclerotiorum is an ongoing challenge because of its wide host range and the persistence of its sclerotia in soil. Fungicides are the most commonly used method to control this fungus but these can have ecotoxicity impacts. Chitinolytic Streptomyces strains isolated from Brazilian tropical soils were capable of inhibiting S. sclerotiorum growth in vitro, offering new possibilities for integrated pest management and biocontrol, with a new approach to dealing with an old problem. Strain Streptomyces sp. 80 was capable of irreversibly inhibiting fungal growth. Compared to other strains, its crude enzymes had the highest chitinolytic levels when measured at 25°C and strongly inhibited sclerotia from S. sclerotiorum. It produced four hydrolytic enzymes involved in fungal cell wall degradation when cultured in presence of the fungal mycelium. The best production, obtained after three days, was 0.75 U/ml for exochitinase, 0.9 U/ml for endochitinase, 0.16 U/ml for glucanase, and 1.78 U/ml for peptidase. Zymogram analysis confirmed two hydrolytic bands of chitinolytic activity with apparent molecular masses of 45.8 and 206.8 kDa. One glucanase activity with an apparent molecular mass of 55 kDa was also recorded, as well as seven bands of peptidase activity with apparent molecular masses ranging from 15.5 to 108.4 kDa. Differential interference contrast microscopy also showed alterations of hyphal morphology after co-culture. Streptomyces sp. 80 seems to be promising as a biocontrol agent against S. sclerotiorum, contributing to the development of new methods for controlling plant diseases and reducing the negative impact of using fungicides.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Adams, P.B. and Ayers, W.A. 1979. Ecology of Sclerotinia species. Phytopathology 69, 896–899.
Bae, Y.S. and Knudsen, G.R. 2007. Effect of sclerotial distribution pattern of Sclerotinia sclerotiorum on biocontrol efficacy of Trichoderma harzianum. Appl. Soil Ecol. 35, 21–24.
Bara, M.T.F., Lima, A.L., and Ulhoa, C.J. 2003. Purification and characterization of an exo-beta-1,3-glucanase produced by Trichoderma asperellum. FEMS Microbiol. Lett. 219, 81–85.
Buroker-Kilgore, M. and Wang, K.K. 1993. A Coomassie brilliant blue G-250-based colorimetric assay for measuring activity of calpain and other proteases. Anal. Biochem. 208, 387–392.
Cole, J., Wang, Q., Cardenas, E., Fish, J., Chai, B., Farris, R., Kulam-Syed-Mohideen, A., McGarrell, D., Marsh, T., Garrity, G., and Tiedje, J. 2009. The Ribosomal Database Project: improved alignments and new tools for rRNA analysis. Nucleic Acids Res. 37, 141–145.
Dastager, S.G., Kim, C.J., Lee, J.C., Agasar, D., Park, D.J., and Li, W.J. 2008. Streptomyces deccanensis sp. nov., an alkaliphilic species isolated from soil. Int. J. Syst. Evol. Microbiol. 58, 1089–1093.
De Marco, J.L. and Felix, C.R. 2007. Purification and characterization of a β-glucanase produced by Trichoderma harzianum showing biocontrol potential. Braz. Arch. Biol. Technol. 50, 21–29.
De Marco, J.L., Valadares-Inglis, M.C., and Felix, C.R. 2003. Production of hydrolytic enzymes by Trichoderma isolates with antagonistic activity against Crinipellis perniciosa, the causal agent of witches’ broom of cocoa. Braz. J. Microbiol. 34, 33–38.
Dunne, C., Crowley, J.J., Moenne-Loccoz, Y., Dowling, D.N., De Bruijn, F.J., and O’Gara, F. 1997. Biological control of Pythium ultimum by Stenotrophomonas maltophilia W81 is mediated by an extracellular proteolytic activity. Microbiology 143, 3921–3931.
El-Tarabily, K.A. 2006. Rhizosphere-competent isolates of streptomycete and non-streptomycete actinomycetes capable of producing cell-wall-degrading enzymes to control Pythium aphanidermatum damping-off disease of cucumber. Can. J. Bot. 84, 211–222.
El-Tarabily, K.A., Nassar, A.H., Hardy, G.E.S.J., and Sivasithamparam, K. 2009. Plant growth promotion and biological control of Pythium aphanidermatum, a pathogen of cucumber, by endophytic actinomycetes. J. Appl. Microbiol. 106, 13–26.
El-Tarabily, K.A., Soliman, M.H., Nassar, A.H., Al-Hassani, H.A., Sivasithamparam, K., McKenna, F., and Hardy, G.E.S.J. 2000. Biological control of Sclerotinia minor using a chitinolytic bacterium and actinomycetes. Plant Pathol. 49, 573–583.
Ferraz, L.C.L. and Café Filho, A.C. 1998. Meios de cultura de fatores culturais para a produ..o de escleródios e apotécio de Sclerotinia sclerotiorum in vitro. Fitopatol. Bras. 23, 364–369.
Gomes, R.C., Semêdo, L.T.A.S., Linhares, A.A., Guimarães, A.C.C., Alviano, C.S., Linhares, L.F., and Coelho, R.R.R. 1999. Efficiency of the dispersion and differential centrifugation technique in the isolation of chitinolytic actinomycetes from soil. World J. Microbiol. Biotechnol. 15, 47–50.
Gomes, R.C., Semêdo, L.T., Soares, R.M.A., Alviano, C.S., Linhares, L.F., and Coelho, R.R.R. 2000. Chitinolytic activity of actinomycetes from a cerrado soil and their potential in biocontrol. Lett. Appl. Microbiol. 30, 146–150.
Gomes, R.C., Sêmedo, L.T.A.S., Soares, R.M.A., Linhares, L.F., Ulhoa, C.J., Alviano, C.S., and Coelho, R.R.R. 2001. Purification of a thermostable endochitinase from Streptomyces RC1071 isolated from a cerrado soil and its antagonism against phytopathogenic fungi. J. Appl. Microbiol. 90, 653–661.
Gopalakrishnan, S., Pande, S., Sharma, M., Humayun, P., Kiran, B.K., Sandeep, D., Vidya, M.S., Deepthi, K., and Rupela, O. 2011. Evaluation of actinomycete isolates obtained from herbal vermicompost for the biological control of Fusarium wilt of chickpea. Crop Protection 30, 1070–1078.
Gracia-Garza, J.A., Reeleder, R.D., and Paulitz, T.C. 1997. Degradation of sclerotia of Sclerotinia sclerotiorum by fungus gnats (Bradysia coprophila) and the biocontrol fungi Trichoderma spp. Soil Biol. Biochem. 29, 123–129.
Gupta, C., Kumar, B., Dubey, R., and Maheshwari, D. 2006. Chitinase-mediated destructive antagonistic potential of Pseudomonas aeruginosa against Sclerotinia sclerotiorum causing stem rot of peanut. Biocontrol 51, 821–835.
Han, Y., Yang, B., Zhang, F., Miao, X., and Li, Z. 2009. Characterization of antifungal chitinase from marine Streptomyces sp. DA11 associated with South China Sea sponge Craniella australiensis. Mar. Biotechnol. 11, 132–140.
Hayashi, H., Sakamoto, M., and Benno, Y. 2004. Evaluation of three different forward primers by terminal restriction fragment length polymorphism analysis for determination of fecal Bifidobacterium spp. in healthy subjects. Microbiol. Immunol. 48, 1–6.
Hegedus, D.D. and Rimmer, S.R. 2005. Sclerotinia sclerotiorum: When “to be or not to be” a pathogen? FEMS Microbiol. Lett. 251, 177–184.
Heussen, C. and Dowdle, E.B. 1980. Electrophoretic analysis of plasminogen activators in polyacrylamide gels containing sodium dodecyl sulfate and copolymerized substrates. Anal. Biochem. 102, 196–202.
Hou, X., Boyetchko, S.M., Brkic, M., Olson, D., Ross, A., and Hegedus, D. 2006. Characterization of the anti-fungal activity of a Bacillus spp. associated with sclerotia from Sclerotinia sclerotiorum. Appl. Microbiol. Biotechnol. 72, 644–653.
Inbar, J., Menendez, A.N.A., and Chet, I. 1996. Hyphal interaction between Trichoderma harzianum and Sclerotinia sclerotiorum and its role in biological control. Soil Biol. Biochem. 28, 757–763.
Jones, D. 1970. Ultrastructure and composition of the cell walls of Sclerotinia sclerotiorum. Transact. Brit. Mycol. Soc. 54, 351–360.
Jones, B.L., Fontanini, D., Jarvinen, M., and Pekkarinen, A. 1998. Simplified endoproteinase assays using gelatin or azogelatin. Anal. Biochem. 263, 214–220.
Junior, M.L. and Abreu, M.S.D. 1994. Inibição do crescimento micelial de Sclerotinia sclerotiorum por metabólitos voláteis produzidos por alguns antagonistas em diferentes temperaturas e pH. Cienc. Agrotecnol. 24, 3–8.
Kamensky, M., Ovadis, M., Chet, I., and Chernin, L. 2003. Soil-borne strain IC14 of Serratia plymuthica with multiple mechanisms of antifungal activity provides biocontrol of Botrytis cinerea and Sclerotinia sclerotiorum diseases. Soil Biol. Biochem. 35, 323–331.
Kurtzman, C.P. and Robnett, C.J. 1998. Identification and phylogeny of ascomycetous yeasts from analysis of nuclear large subunit (26S) ribosomal DNA partial sequences. Antonie van Leeuwenhoek 73, 331–371.
Leelasuphakul, W., Sivanunsakul, P., and Phongpaichit, S. 2006. Purification, characterization and synergistic activity of β-1,3-glucanase and antibiotic extract from an antagonistic Bacillus subtilis NSRS 89-24 against rice blast and sheath blight. Enzyme Microb. Technol. 38, 990–997.
Loffler, F.E., Sun, Q., Li, J., and Tiedje, J.M. 2000. 16S rRNA gene-based detection of tetrachloroethene-dechlorinating Desulfuromonas and Dehalococcoides species. Appl. Environ. Microbiol. 66, 1369–1374.
Monteiro, V.N. and Ulhoa, C.J. 2006. Biochemical characterization of a β-1,3-glucanase from Trichoderma koningii induced by cell wall of Rhizoctonia solani. Curr. Microbiol. 52, 92–96.
Napoleão, R., Nasser, L., Lopes, C., and Café Filho, A. 2006. Neon-S, novo meio para detecção de Sclerotinia sclerotiorum em sementes. Summ. Phytopathol. 32, 180–182.
Pan, S.Q., Ye, X.S., and Kuć, J. 1989. Direct detection of beta-1,3-glucanase isozymes on polyacrylamide electrophoresis and isoelectrofocusing gels. Anal. Biochem. 182, 136–140.
Prapagdee, B., Kuekulvong, C., and Mongkolsuk, S. 2008. Antifungal potential of extracellular metabolites produced by Streptomyces hygroscopicus against phytopathogenic fungi. Int. J. Biol. Sci. 4, 330–337.
Shirling, E.B. and Gottlieb, D. 1966. Methods for characterization of Streptomyces species. Int. J. Syst. Bacteriol. 16, 313–340.
Souza, R.F., Coelho, R.R.R., Macrae, A., Soares, R.M.A., Nery, D.C.M., Semêdo, L.T.A.S., Alviano, C.S., and Gomes, R.C. 2008. Streptomyces lunalinharesii sp. nov., a chitinolytic streptomycete isolated from cerrado soil in Brazil. Int. J. Syst. Evol. Microbiol. 58, 2774–2778.
Souza, R.F., Gomes, R.C., Coelho, R.R.R., Alviano, C.S., and Soares, R.M.A. 2003. Purification and characterization of an endochitinase produced by Colletotrichum gloeosporioides. FEMS Microbiol. Lett. 222, 45–50.
Spiro, R.G. 1966. The Nelson-Somogyi copper reduction method. Methods Enzymol. 8, 3–26.
Sunna, A. and Antranikian, G. 1997. Xylanolytic enzymes from fungi and bacteria. Crit. Rev. Biotechnol. 17, 39–67.
Sutherland, E.D. and Papavizas, G.C. 1991. Evaluation of oospore hyperparasites for the control of phytophthora crown rot of pepper. J. Phytopathol. 131, 33–39.
Tahtamouni, M.E.W., Hameed, K.M., and Saadoun, I.M. 2006. Biological control of Sclerotinia sclerotiorum using indigenous chitinolytic actinomycetes in Jordan. Plant Pathol. J. 22, 107–114.
Tamura, K., Peterson, D., Peterson, N., Stecher, G., Nei, M., and Kumar, S. 2011. MEGA5: Molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol. Biol. Evol. 28, 2731–2739.
Thompson, J.D., Higgins, D.G., and Gibson, T.J. 1994. CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res. 22, 4673–4680.
Trudel, J. and Asselin, A. 1989. Detection of chitinase activity after polyacrylamide gel electrophoresis. Anal. Biochem. 178, 362–366.
Valois, D., Fayad, K., Barasubiye, T., Garon, M., Dery, C., Brzezinski, R., and Beaulieu, C. 1996. Glucanolytic actinomycetes antagonistic to Phytophthora fragariae var. rubi, the causal agent of raspberry root rot. Appl. Environ. Microbiol. 62, 1630–1635.
Young, C.S., Clarkson, J.P., Smith, J.A., Watling, M., Phelps, K., and Whipps, J.M. 2004. Environmental conditions influencing Sclerotinia sclerotiorum infection and disease development in lettuce. Plant Pathol. 53, 387–397.
Yuan, W.M. and Crawford, D.L. 1995. Characterization of Streptomyces lydicus WYEC108 as a potential biocontrol agent against fungal root and seed rots. Appl. Environ. Microbiol. 61, 3119–3128.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Fróes, A., Macrae, A., Rosa, J. et al. Selection of a Streptomyces strain able to produce cell wall degrading enzymes and active against Sclerotinia sclerotiorum . J Microbiol. 50, 798–806 (2012). https://doi.org/10.1007/s12275-012-2060-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12275-012-2060-2